2.1. Supported Catalysts versus Doped Catalysts
The employment of advanced carbon materials extends the ways to develop combustion catalysts by using different approaches and formats, such as powders, grains, pellets, films, coatings on different monolith or foam structures and so on (
Figure 1). Supported catalysts are traditionally prepared by a deposition method (e.g., impregnation, chemical vapor deposition–CVD) on the corresponding carbon support, previously prepared. Some years ago, our research group has proposed the synthesis of metal-doped carbon aerogels and their direct application as catalysts. These metal-doped materials are prepared by solving the precursor metal-salt into a starting solution of organic precursors, which is decomposed during a thermal treatment performed over the organic polymeric aerogel or xerogel [
57]. Thus, the metal is reduced during carbonization by the organic matrix and an additional pretreatment is not needed, in contrast of supported catalysts. The metals used for doping are also active along all synthesis steps of the material,
i.e., polymerization, carbonization and/or activation. Interactions between organic–inorganic phases determine the distribution and dispersion of metals, sintering resistance, porosity, graphitization, formation of different active phases,
etc. Therefore, new challenges are possible in catalysis because resulting metal particles are active in many catalyzed processes.
Figure 1.
Carbon materials obtained in different formats: grains, powders, pellets, films and coatings on monolith and foam structures.
Figure 1.
Carbon materials obtained in different formats: grains, powders, pellets, films and coatings on monolith and foam structures.
The issue of supported catalysts
versus doped catalysts was studied with two Pt-catalyst series in the gas-phase combustion reaction of toluene [
56]. The materials were prepared according the recipes regarded in
Table 7. The supported Pt catalysts (1 wt.% Pt loading) were prepared by impregnation of an organic aerogel carbonized at 500 or 1000 °C with an aqueous solution of [Pt(NH
3)
4]Cl
2 (
i.e., A500-Pt and A1000-Pt, respectively). The same precursor salt was used in the starting solution of Pt-doped catalysts (
i.e., B500-Pt and B1000-Pt), a similar Pt content (around 1.1 wt.%) than the impregnated catalysts being determined by thermogravimetric (TG) analysis after carbonization.
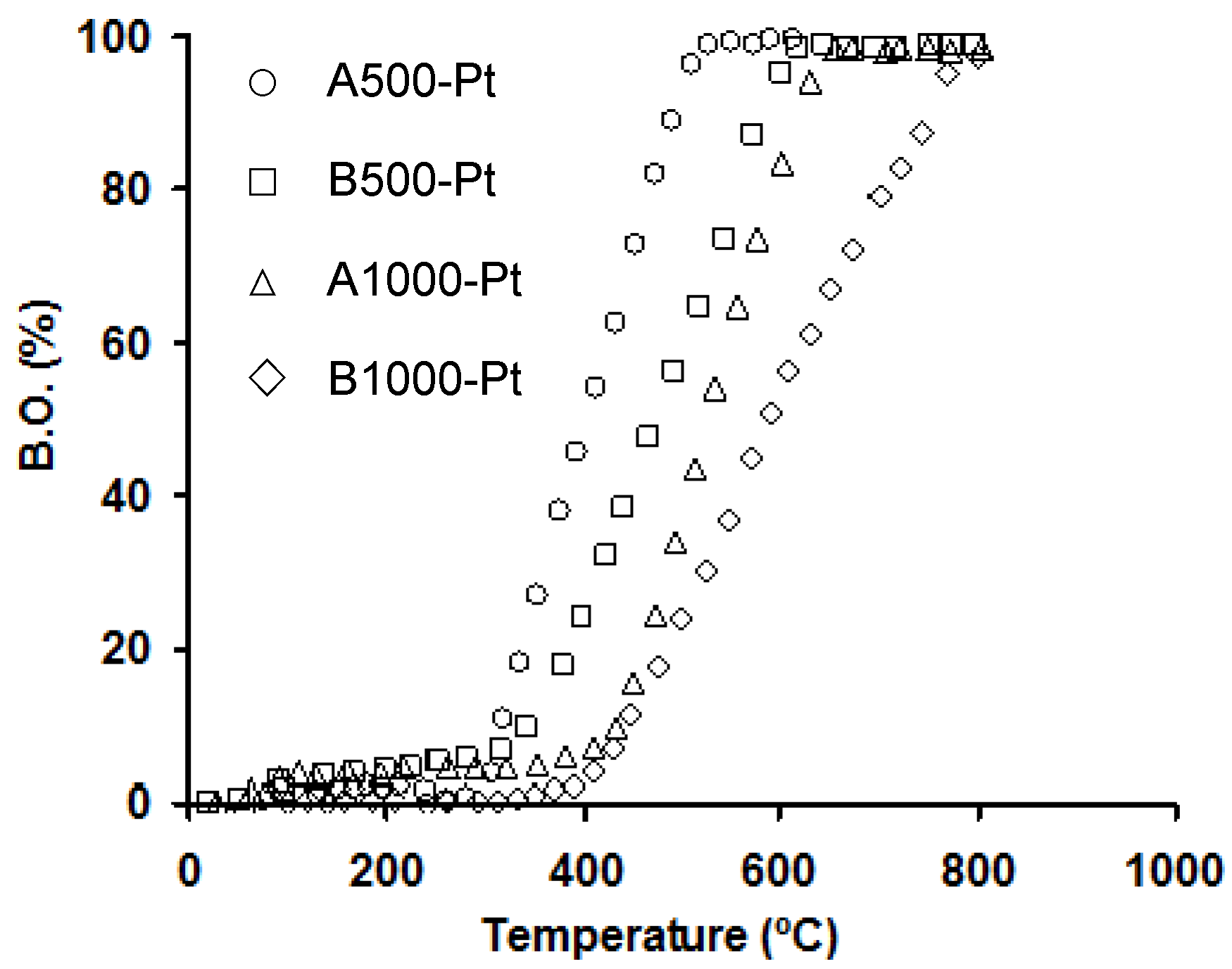
The thermal stability in air of carbon-based catalysts is an important parameter to study in reactions or processes requiring high and moderate temperatures. It depends not only on the previous thermal treatments of supports but also on the porous texture, active phase and metal dispersion. In general, the carbon supports are more stable when they are carbonized at higher temperature, while many metals may catalyze the carbon combustion reaction. For the Pt-catalysts series, the burn-off (B.O.) of the support was practically negligible below 300–400 °C (
Figure 2), which was much higher than the temperature used to perform the catalytic oxidation of VOCs and thereby, the support gasification did not occur.
Figure 2.
Percentage of burn-off (B.O.) in air against temperature for supported catalysts (A-samples) or doped (B-samples) carbonized at 500 and 1000 °C. Figure reprinted with permission from [
56]. Copyright 2004, Elsevier.
Figure 2.
Percentage of burn-off (B.O.) in air against temperature for supported catalysts (A-samples) or doped (B-samples) carbonized at 500 and 1000 °C. Figure reprinted with permission from [
56]. Copyright 2004, Elsevier.
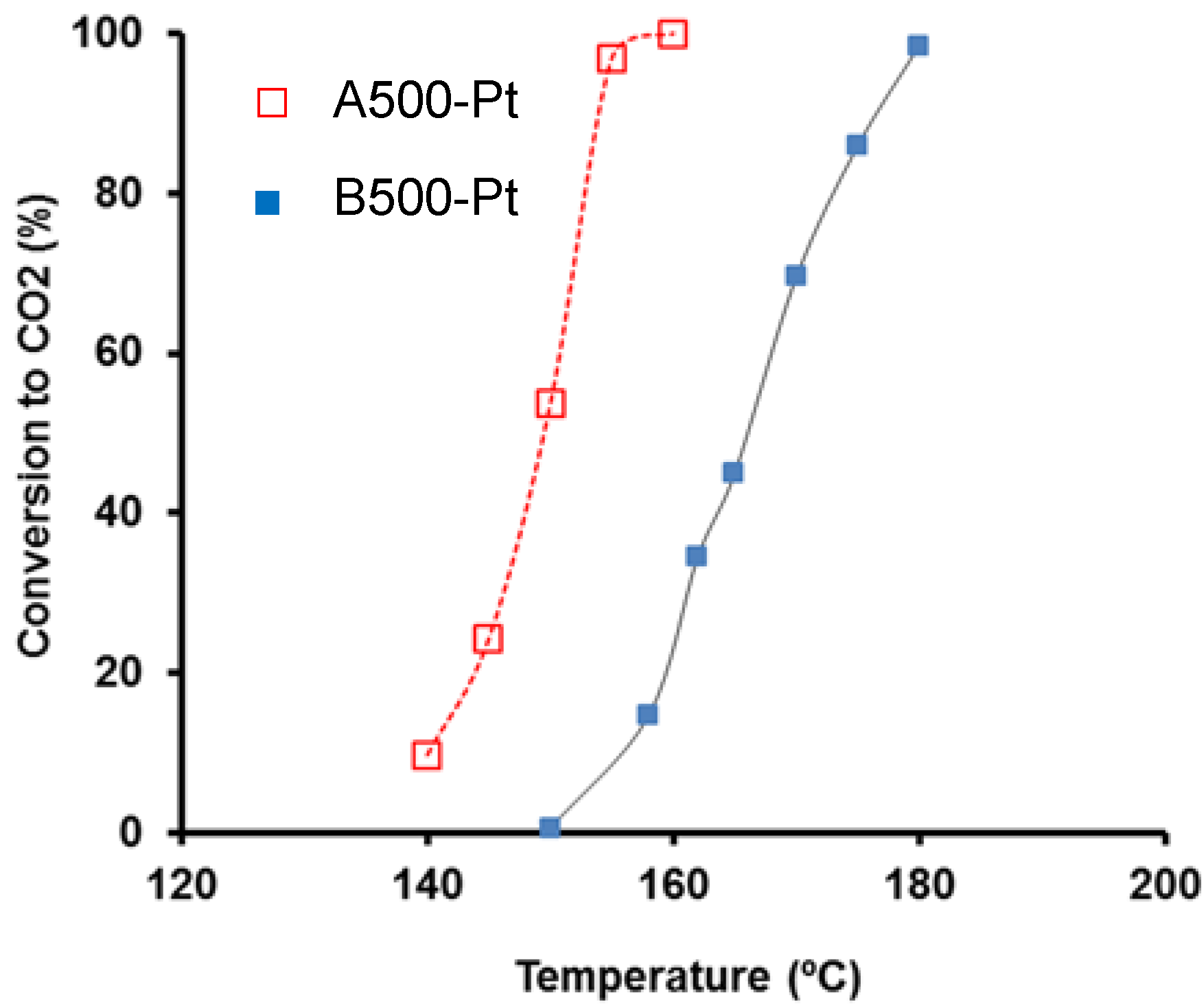
The toluene conversion to CO
2 was obtained by using supported Pt-catalysts (A500-Pt) and Pt-doped catalysts (B500-Pt) on carbon aerogels (
Figure 3). Only CO
2 and water were found as reaction products, while partial oxidation products including CO and oxygenated organic compounds, were not detected by both gas chromatography (GC) or mass spectrometry (MS) techniques. The total toluene conversion was achieved below 200 °C in both cases, although differences in the activity were observed to depend on several parameters, e.g., Pt-particle size. For catalysts prepared with supports at 500 °C, Pt-particle size was 3.9 nm for A500-Pt after H
2-pretreatment, while particles of 12 nm were obtained for B500-Pt. The higher activity of supported catalysts (A500-Pt) was related with a better Pt-dispersion and an easier accessibility of pollutants to Pt-particle sites. Moreover, in spite of the previously described advantages of metal-doped carbon gels, some metal particles were encapsulated by the organic matrix and thereby inactive in catalysis. In this context, both preparation methods present clear advantages and drawbacks, and the catalyst preparation (supported
vs. doped) should be determined by the experimental conditions and the final application.
Figure 3.
Catalytic performance obtained for supported Pt and Pt-doped catalysts on carbon aerogels in the toluene combustion ([toluene] = 0.45 vol.%,
Q = 3.6 L h
−1, 0.10 g
catalyst). Figure adapted with permission from [
56]. Copyright 2004, Elsevier.
Figure 3.
Catalytic performance obtained for supported Pt and Pt-doped catalysts on carbon aerogels in the toluene combustion ([toluene] = 0.45 vol.%,
Q = 3.6 L h
−1, 0.10 g
catalyst). Figure adapted with permission from [
56]. Copyright 2004, Elsevier.
2.2. Influence of the Support Porosity
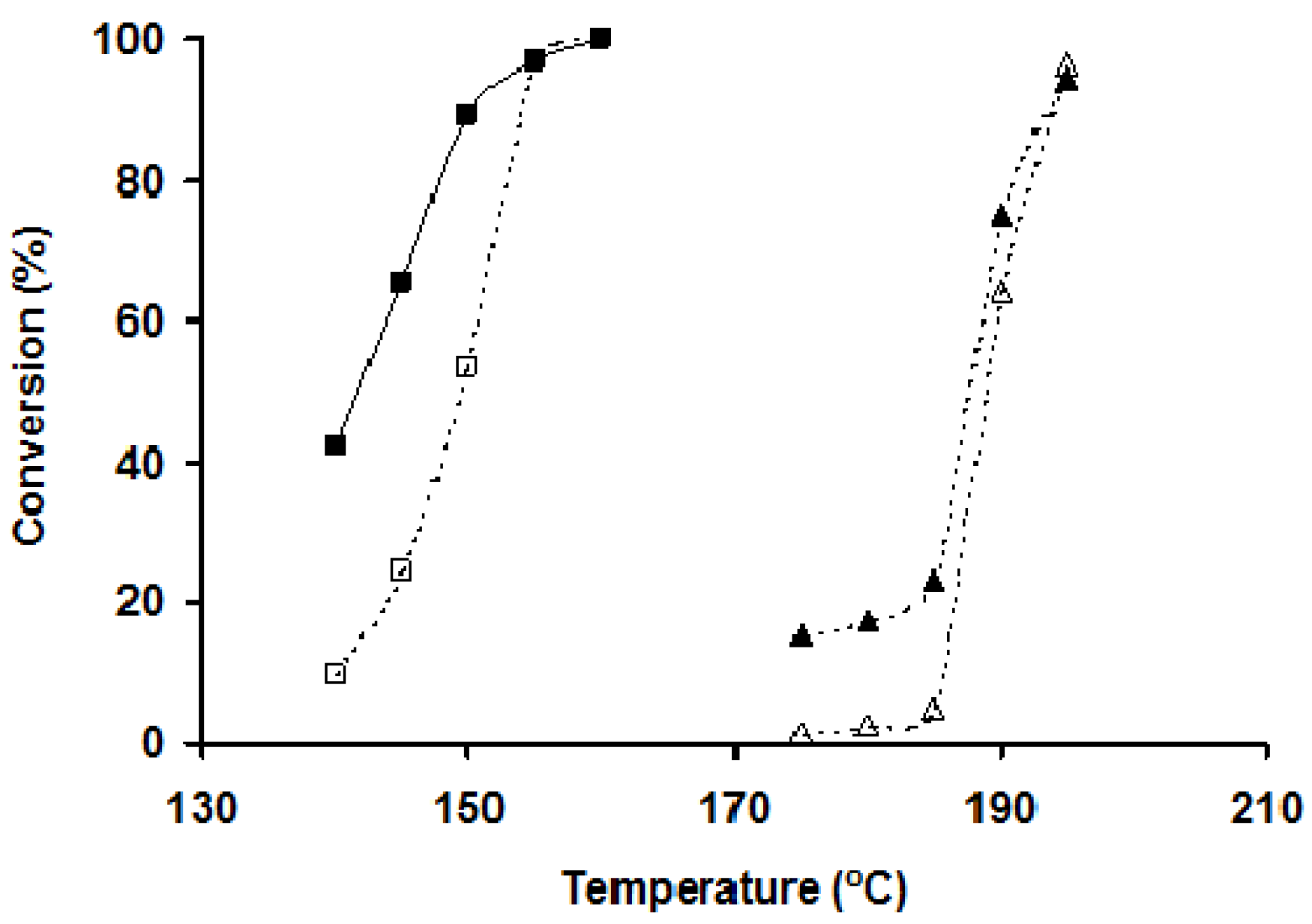
Light-off curves for the A1000-Pt catalyst pre-treated in H
2 were obtained under two different toluene concentrations in the feed stream (
Figure 4). These results showed that the light-off curve was shifted to a lower temperature when the toluene concentration decreased from 0.45 to 0.10 vol.%, in agreement with VOCs oxidation obtained by other Pt catalysts [
48,
58]. In addition, the conversion to CO
2 was lower than the total conversion of toluene when the catalyst was tested at low reaction temperatures, showing the participation of VOCs adsorption in the removal of pollutants under these experimental conditions, since partial oxidation products were not detected. Nevertheless, both conversions were matched when the reaction temperature increased due to a less favored adsorption of toluene and the enhancement of the combustion process.
Figure 4.
Light-off curves for A1000-Pt at two different toluene concentrations in the feeding gas: 0.10 vol.% (squares) and 0.45 vol.% (triangles). Open symbols: conversion to CO
2. Closed symbols: conversion of toluene (
Q = 3.6 L h
−1, 0.10 g
catalyst). Figure reprinted with permission from [
56]. Copyright 2004, Elsevier.
Figure 4.
Light-off curves for A1000-Pt at two different toluene concentrations in the feeding gas: 0.10 vol.% (squares) and 0.45 vol.% (triangles). Open symbols: conversion to CO
2. Closed symbols: conversion of toluene (
Q = 3.6 L h
−1, 0.10 g
catalyst). Figure reprinted with permission from [
56]. Copyright 2004, Elsevier.
VOCs’ adsorption on the carbon support is an important key for the respective oxidation because it increases the surface density of adsorbed molecules in the metal-carbon interphase region and enhances the catalytic oxidation at low temperatures [
17,
48]. VOCs’ adsorption is obviously controlled by the physical (porosity) and chemical (interactions) properties of carbon supports.
The macro-mesoporosity of carbon aerogels is commonly associated with the inter-particle voids, and thereby, can be controlled by modifying the synthesis conditions during the polymerization. On the other hand, the microporosity is generated by the release of gases during carbonization [
57,
59]. In this context, the pore size distribution (PSD) of the supports (carbon aerogels) was modulated by changing the polymerization catalyst (
Table 7). Thus, when Li
2CO
3 was used as polymerization catalyst, the resulting carbon aerogel was mesoporous, whereas a macroporous support was produced with Cs
2CO
3. The analysis by mercury porosimetry indicated that both carbon aerogels had a homogenous and unimodal PSD with pore sizes of 25 and 96 nm, respectively. The external and BET surface areas, as well as the meso- and macropore volumes are regarded in
Table 2.
Table 2.
Textural characterization of the carbon aerogels.
Table 2.
Textural characterization of the carbon aerogels.
| Support | *SBET (m2 g−1) | ρ (g cm−3) | Vmeso (cm3 g−1) | Vmacro (cm3 g−1) | Sext (m2 g−1) |
|---|
| ALi900 | 902 | 0.99 | 1.06 | 0.00 | 191 |
| ACs900 | 758 | 0.80 | 0.07 | 1.20 | 59 |
The mesoporous and macroporous aerogels (ALi900 and ACs900) were used to prepare supported Pt-catalysts, which after H
2-pretreatment at 300 °C showed a Pt-particle size (d
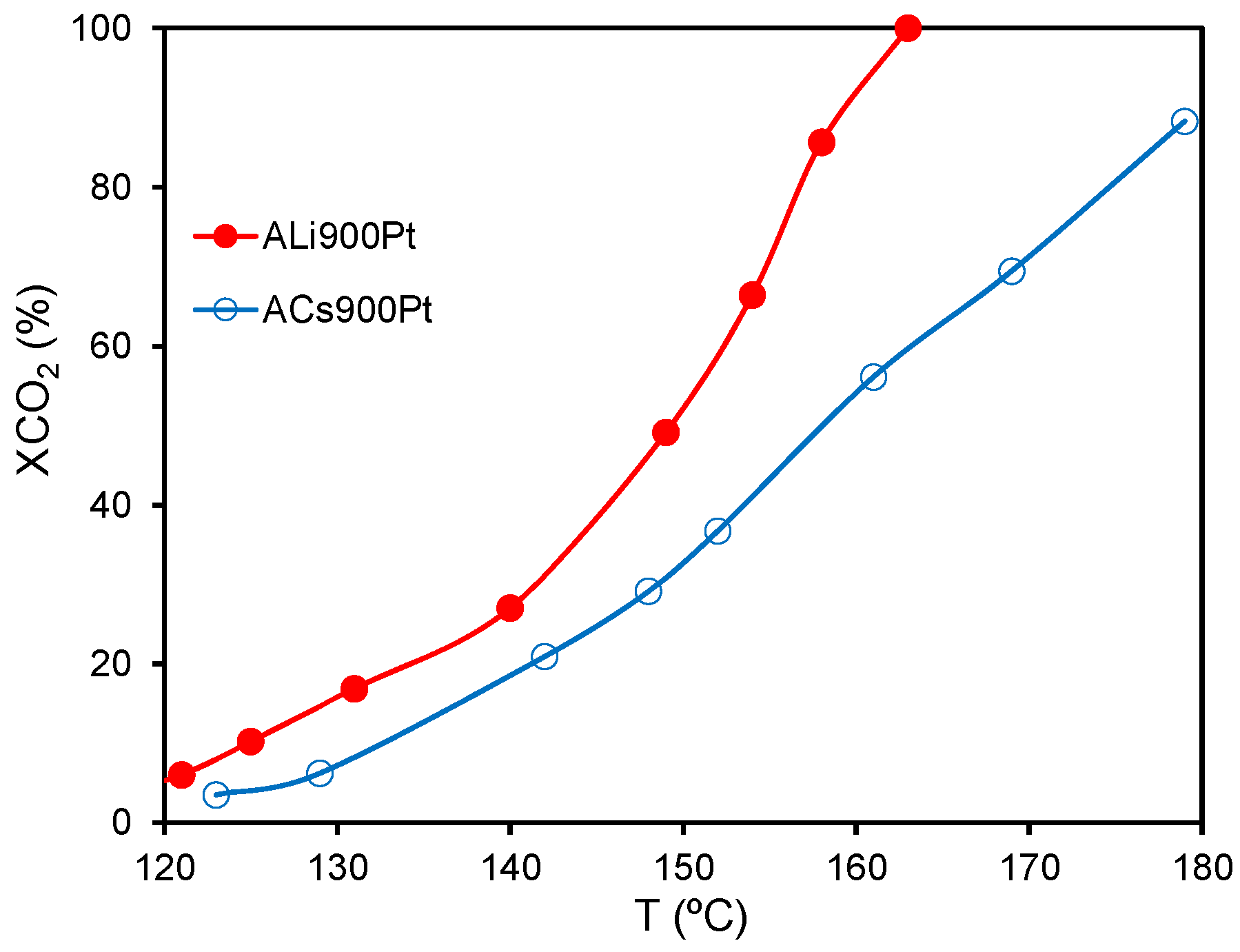
Pt) around 1.5 nm regardless the support used. When the catalytic activity of both catalysts pretreated at 300 °C was analyzed in the combustion of benzene (
Figure 5), the only parameter affecting on their activity was the porous texture, since the surface oxygen content, pH
PZC (point of zero charge) and Pt-dispersion were similar. ALi900Pt was more active than ACs900Pt for the temperatures range studied, benzene being totally and selectively converted to CO
2 at only 160 °C. This larger catalytic activity of ALi900Pt is related with the role of mesoporosity and S
ext of the support, as previously indicated. In the mesoporous support, benzene molecules easily access to the porosity and are adsorbed throughout the large S
ext containing a high concentration of Pt-particles. Thus, the high benzene concentration near to reactive Pt-Particles enhanced the benzene oxidation at low temperatures.
The role of the support porosity was also assessed by Pd-catalysts supported on honeycomb ceramic monoliths [
60], in particular three different carbon-based monoliths and their catalytic performance was studied in the total gas-phase combustion of
m-xylene. The first monolith (HPM) was a square channel cordierite modified with α-Al
2O
3, on which a carbon layer was deposited (
Figure 6c). The other two monolithic supports (WA and WB) were carbon/ceramic composites with different porosity (
Figure 6b). A cross-section detailed of the channels of a monolith coated with a γ-Al
2O
3 layer is also shown in
Figure 6a for comparison.
Figure 5.
Influence of the macro-mesoporosity of supports on the benzene catalytic combustion ([benzene] = 1000 ppm,
Q = 3.6 L h
−1, 0.10 g
catalyst). Figure adapted with permission from [
61]. Copyright 2010, Elsevier.
Figure 5.
Influence of the macro-mesoporosity of supports on the benzene catalytic combustion ([benzene] = 1000 ppm,
Q = 3.6 L h
−1, 0.10 g
catalyst). Figure adapted with permission from [
61]. Copyright 2010, Elsevier.
Figure 6.
SEM micrographs of the monolith channels: details of (
a) γ-Al
2O
3 layer; (
b) carbon/ceramic composite and (
c) carbon coating. (
d) Model of the porosity developed for carbon-based monoliths. Figures reprinted with permission from [
60,
62,
63]. Copyright 2008, 2009, Elsevier.
Figure 6.
SEM micrographs of the monolith channels: details of (
a) γ-Al
2O
3 layer; (
b) carbon/ceramic composite and (
c) carbon coating. (
d) Model of the porosity developed for carbon-based monoliths. Figures reprinted with permission from [
60,
62,
63]. Copyright 2008, 2009, Elsevier.
The textural properties of the carbon-based monoliths were analyzed (
Table 3), different porosities being determined depending on the type of support used. In this context, the carbon layer in HPM presented only small micropores, since
SBET was much lower than
SCO2. Both WA and WB monoliths had a heterogeneous micropore size distribution (
i.e.,
SBET was higher than
SCO2), while WA presented only macropores and WB had meso- and macropores (
Figure 6d) and consequently, a larger
Sext.
Table 3.
Surface characteristics of the monolithic supports. Data in parenthesis are given per gram of carbon.
Table 3.
Surface characteristics of the monolithic supports. Data in parenthesis are given per gram of carbon.
| Monolith | *SCO2 (m2 g−1) | SBET (m2 g−1) | Sext (m2 g−1) | Vmacro (cm3 g−1) | Vmeso (cm3 g−1) |
|---|
| WA | 329 (947) | 474 (1366) | 4 (12) | 0.325 (0.937) | 0 |
| WB | 242 (782) | 460 (1489) | 62 (199) | 0.233 (0.754) | 0.138 (0.447) |
| HPM | 17 (269) | (2) | (<1) | (n.d.) | (n.d.) |
Pd-catalysts supported on the previous monoliths presented a comparable Pd loading with very similar Pd particle sizes (around 5–6 nm), although differences were found in their location. Thus, Pd was situated only in the macropores of the Pd/WA catalyst, while for Pd/WB the Pd was distributed throughout the mesoporous texture. In the case of Pd/HPM, Pd-particles were deposited on the external surface of the carbon layer, since the size of the metal particles avoided their deposition in the respective micropores.
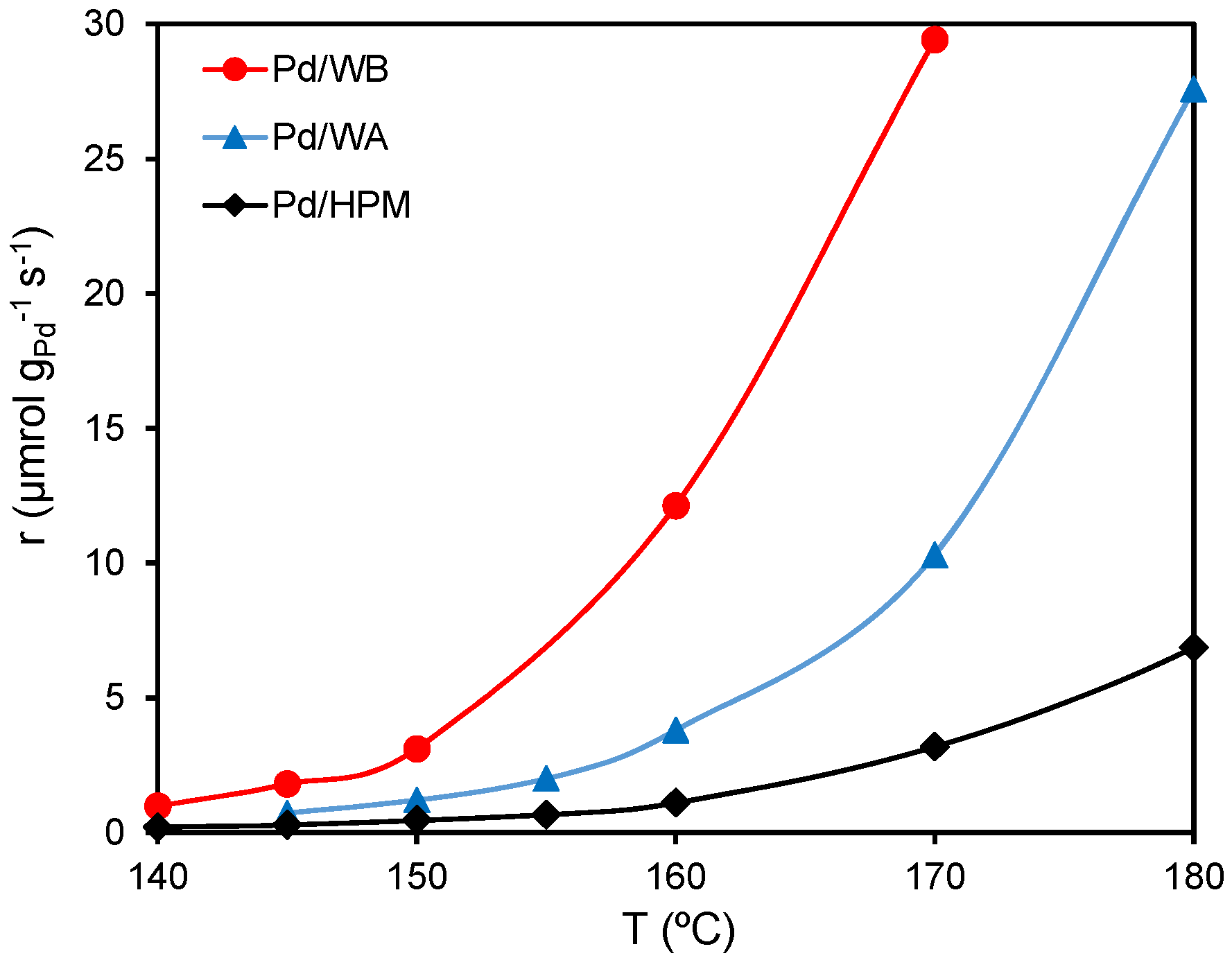
The reaction rates of the monolithic catalysts were obtained for the gas-phase catalytic combustion of
m-xylene (
Figure 7). The total combustion of
m-xylene was reached at 170 °C for Pd/WB, which was the most active catalyst, with a total selectivity to CO
2 and H
2O. In addition, the catalytic performance was very different, the rate of the catalysts being varied as follows: Pd/HPM < Pd/WA < Pd/WB,
i.e., in according to the external surface area of the corresponding support (
Table 3). Therefore, the macropores and mainly mesopores, play an important role in this type of gas-phase reactions, improving the contact between Pd-particles and
m-xylene molecules, in agreement with previous results for carbon aerogels supported catalysts.
Figure 7.
Catalytic performance of the monolithic catalysts normalized per gram of Pd in the
m-xylene combustion ([
m-xylene] = 0.10 vol.%,
Q = 2000 m
3gas h
−1 m
−3monolith). Figure adapted with permission from [
60]. Copyright 2008, Elsevier.
Figure 7.
Catalytic performance of the monolithic catalysts normalized per gram of Pd in the
m-xylene combustion ([
m-xylene] = 0.10 vol.%,
Q = 2000 m
3gas h
−1 m
−3monolith). Figure adapted with permission from [
60]. Copyright 2008, Elsevier.
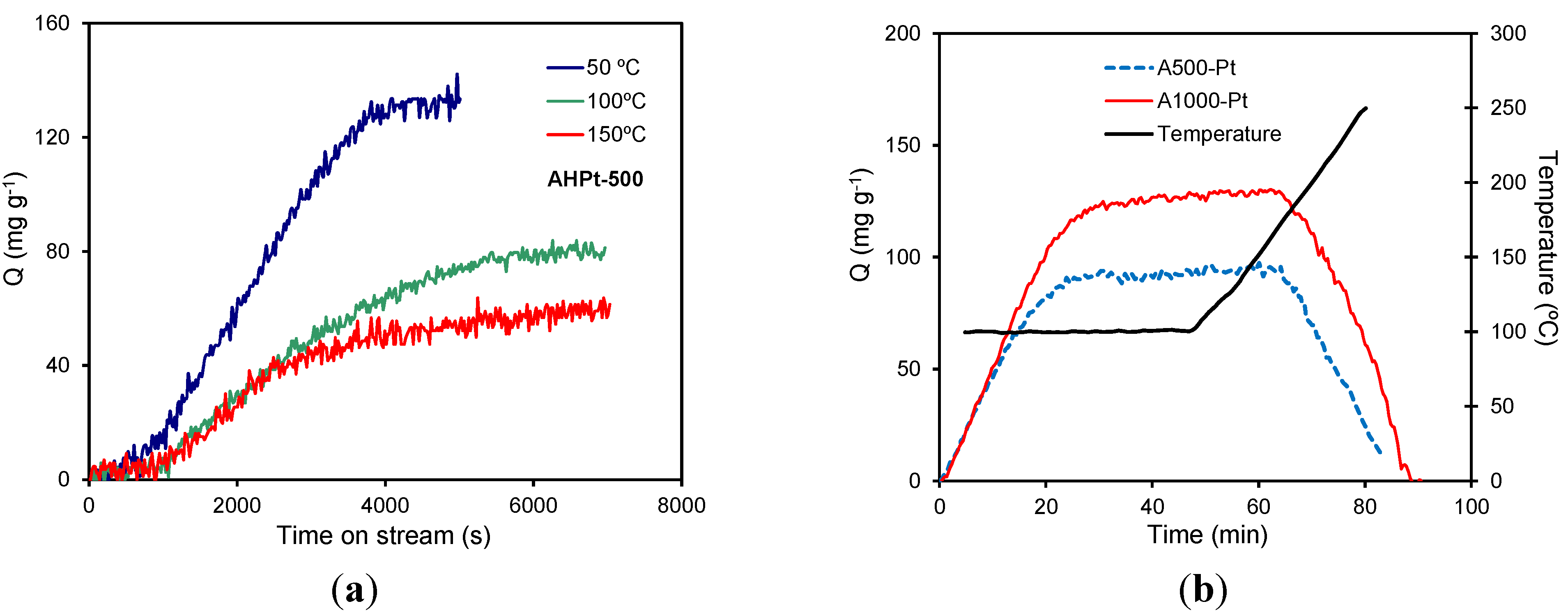
Since the carbon materials are highly porous solids, the preconcentration of VOCs in their surface by adsorption facilitates their combustion (as indicated), but also the synergy adsorbent/catalyst will have an important role. Therefore, the adsorptive behavior of this type of materials in the removal of VOCs will depend markedly on the operational conditions. In this context, the adsorption of different VOCs over carbon aerogel-based catalysts was studied by TG in dynamic mode (
Figure 8) [
59,
64]. As expected, the adsorption capacity and the adsorption rate for A500-Pt decrease with higher temperatures (
Figure 8a), since adsorption only takes place in progressively narrower pores and consequently, the adsorption capacity decreases. On the other hand, adsorption/desorption cycles for A500-Pt and A1000-Pt at constant temperature (
Figure 8b) showed a comparable toluene adsorption rate for both aerogels regardless the catalyst and the carbonization temperature used, because the mean micropore sizes (L
0) were always around 0.57 ± 0.03 nm. However, the adsorption capacity for both samples was favored for the sample carbonized at higher temperature (A1000-Pt) due to a larger development of its microporosity, compared to A500-Pt. These results also pointed out that the toluene adsorption is reversible by thermal treatment of the corresponding support (
Figure 8b).
Figure 8.
Dynamic toluene adsorption: (
a) effect of the adsorption temperature and (
b) effect of the carbonization temperature (
Q = 3.6 L h
−1, 0.10 g
catalyst). Figures adapted with permission from [
64]. Copyright 2011, Elsevier.
Figure 8.
Dynamic toluene adsorption: (
a) effect of the adsorption temperature and (
b) effect of the carbonization temperature (
Q = 3.6 L h
−1, 0.10 g
catalyst). Figures adapted with permission from [
64]. Copyright 2011, Elsevier.
Figure 9.
Catalytic stability of A500-Pt over the combustion of (
a)
p-xylene at 170 °C and (
b) acetone at 210 °C ([VOC] = 1500 ppm,
Q = 3.6 L h
−1, 0.10 g
catalyst). Figures adapted with permission from [
64]. Copyright 2011, Elsevier.
Figure 9.
Catalytic stability of A500-Pt over the combustion of (
a)
p-xylene at 170 °C and (
b) acetone at 210 °C ([VOC] = 1500 ppm,
Q = 3.6 L h
−1, 0.10 g
catalyst). Figures adapted with permission from [
64]. Copyright 2011, Elsevier.
The catalytic performance of A500-Pt was assessed in the combustion of
p-xylene at 170 °C and acetone at 210 °C (
Figure 9a,b, respectively). The total conversion (X
T) of
p-xylene was reached and maintained at 170 °C over time, however the corresponding conversion to CO
2 (X
CO2) showed small positive and negative oscillations around 100% (
Figure 9a). For the acetone combustion (
Figure 9b), the total conversion was lower than that obtained for
p-xylene,
i.e., acetone is less reactive than
p-xylene. In addition, both X
T and X
CO2 curves showed large oscillations, in particular X
CO2 under these experimental conditions. This oscillatory behavior was reported in the literature as a consequence of adsorbed VOCs or coke-precursor burning with Pt-catalysts supported on inorganic supports such as, zeolites or alumina [
65]. However, the toluene adsorption was completely reversible for carbon aerogels with different porous textures as previously showed [
59] and thereby, the VOCs interaction with the basic carbon surface should be weaker than that obtained with acidic inorganic supports. Thus, VOCs are only adsorbed and oxidized over carbon-based catalysts without any transformation and deposition of stable coke or coke precursor.
In summary, carbon-based catalysts present a versatile behavior, since they can behave as good adsorbents at low temperatures and as excellent combustion catalysts at high reaction temperatures. The temperature range depends obviously on material properties (e.g., porosity, surface chemistry, active phase and metal dispersion) and on VOCs nature. In this context, O’Malley and Hodnett [
66] defined a reactivity pattern for different Pt-inorganic supports as follows: alcohols > aromatics > ketones > carboxylic acids > alkanes, according to the weakness of C-H bonds.
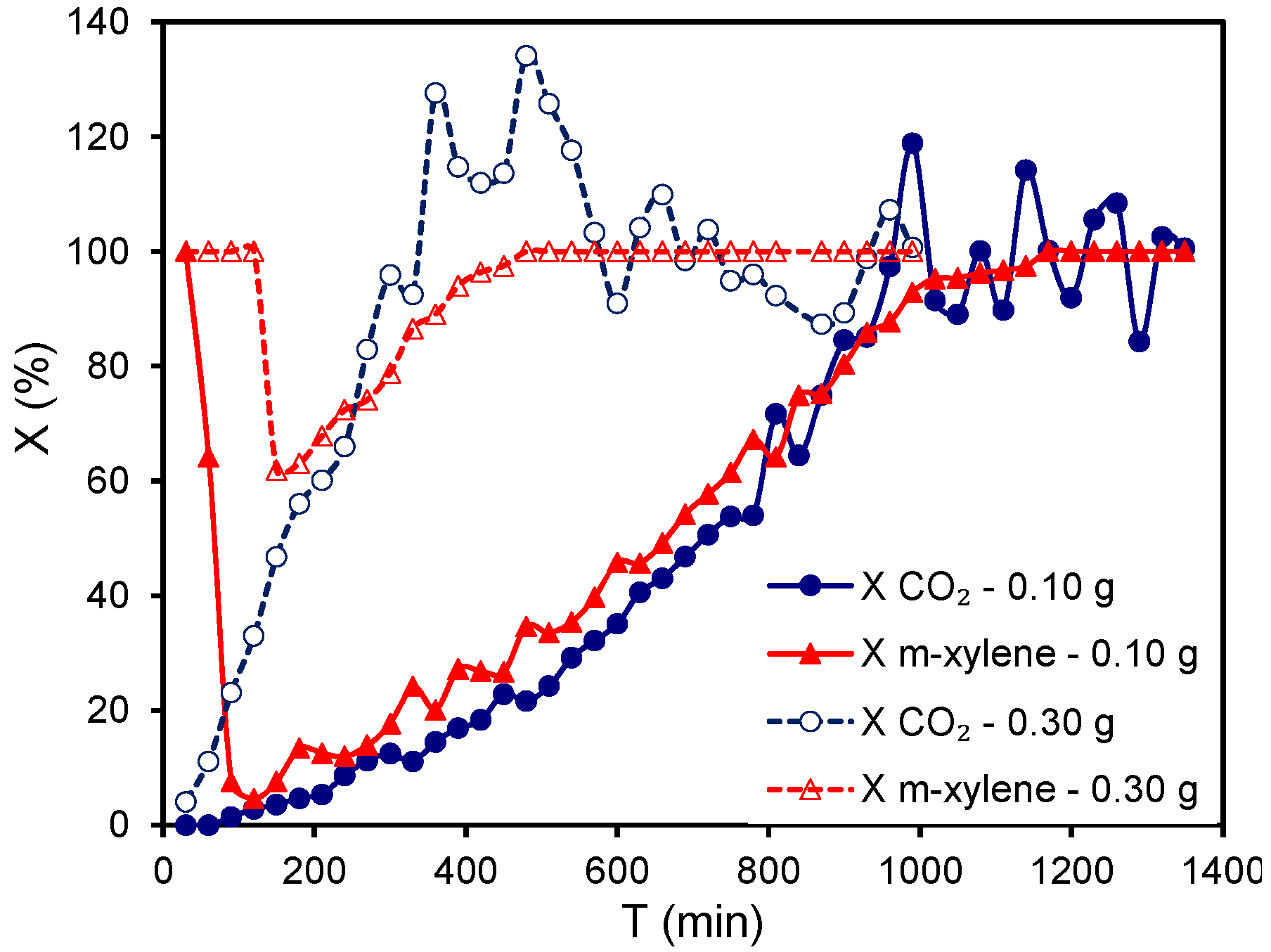
The equilibrium between both adsorption and catalytic processes was studied by changing the weight of catalyst used (0.10 or 0.30 g of A1000-Pt),
i.e., by varying the contact time, over the
m-xylene combustion at 190 °C (
Figure 10). When 0.10 g of catalyst was used, the
m-xylene removal reached at the beginning of the reaction was only due to adsorption, since X
CO2 was null. After saturation (at around 2 h on stream) X
T becomes also very low, but both X
T and X
CO2 simultaneously increased over reaction time because the
m-xylene removal by catalytic combustion increased for 17 h until total conversion. The enhanced catalytic activity can be also explained by the sintering of Pt particles, as previously reported [
56]. By increasing the contact time (using 0.30 g of A1000-Pt), the removal of
m-xylene was also total during the first 2 h but after saturation, the X
T decay was smaller than that observed when 0.10 g of catalyst was used, because the X
CO2 evolution was faster due to the combustion process improved by the presence of a larger amount of active sites. In this case, total removal of
m-xylene to CO
2 was reached after 7 h, compared to 17 h obtained when 0.10 g of catalyst was used. Therefore, at this temperature the adsorption process occurs initially faster than the combustion reaction, although the catalytic removal of VOCs is progressively favored by the preconcentration on the catalyst surface and by the sintering of Pt-particles. The influence of this parameter will be deeply studied in the subsequent Section. Oscillations in both X
T and X
CO2 indicate the establishment of a continuous equilibrium between different adsorption/reaction cycles, while X
CO2 values higher than 100% demonstrated the combustion of compounds previously adsorbed.
Figure 10.
Influence of the contact time on the
m-xylene combustion obtained for A1000-Pt at 190 °C ([
m-xylene] = 1500 ppm,
Q = 3.6 L h
−1, 0.10 or 0.30 g
catalyst). Figure adapted with permission from [
64]. Copyright 2011, Elsevier.
Figure 10.
Influence of the contact time on the
m-xylene combustion obtained for A1000-Pt at 190 °C ([
m-xylene] = 1500 ppm,
Q = 3.6 L h
−1, 0.10 or 0.30 g
catalyst). Figure adapted with permission from [
64]. Copyright 2011, Elsevier.
2.3. Influence of the Metal Particle Size
The influence of this parameter on the catalytic performance in gas-phase combustion reactions has generated certain controversy. Some authors classify the combustion as a structure–sensitive reaction with the Pt particle size, having a detrimental effect on the conversion [
56,
67]. However, Tsou
et al. [
65] did not find any dependency on Pt-dispersion for the
o-xylene combustion with Pt/HBEA zeolite. Therefore, these discrepancies suggest that the correlation between the Pt particle size and catalytic activity is influenced by the catalyst support selected.
For the sake of elucidating this issue, a Pt catalyst supported on a mesoporous carbon aerogel was pre-treated at different temperatures (400, 600 and 950 °C) in He flow (
i.e., APt400He, APt600He and APt950He, respectively) or at 400 °C in H
2 flow (
i.e., APt400H
2) [
67]. The Pt particle size for the different catalysts was determined by H
2 chemisorption (d
Pt) and high resolution transmission electron microscopy (d
Pt-HRTEM), obtaining a well agreement between both techniques (
Table 4). The He-pretreatments performed at 400 and 600 °C gave rise to small Pt particle sizes (between 2.3–2.8 nm). When the pre-treatment temperature increased up to 950 °C, the catalyst with the largest Pt-particle size was obtained (9.5 nm). The H
2-pretreatment at 400 °C led to a d
Pt-HRTEM value (2.9 nm) even slightly larger than that obtained after He-pre-treatment at 600 °C (2.4 nm). Pt catalysts prepared with identical metal precursor and supported on other carbon materials [
56,
68] and even zeolites [
69] also presented a loss of dispersion after H
2-pretreatments. This behavior is due to the formation of an intermediate hydride, [Pt(NH
3)
2H
2], which is unstable and mobile, leading to an agglomeration of Pt-particles. He-pretreatments give rise to a gradual decomposition of the amino complex along two intermediate reactions: (1) reaction takes place between 275 and 315 °C and (2) reaction between 315 and 370 °C [
70].
Table 4.
Hydrogen uptake (Qt), dispersion (D) and Pt particle size of supported catalysts.
Table 4.
Hydrogen uptake (Qt), dispersion (D) and Pt particle size of supported catalysts.
| Catalyst | *Qt (µmol H2 g−1) | *D (%) | *dPt (nm) | dPt-HRTEM (nm) |
|---|
| APt400He | 12.0 | 46.8 | 2.3 | - |
| APt600He | 10.0 | 39.2 | 2.8 | 2.4 |
| APt950He | 2.9 | 11.3 | 9.5 | 6.6 |
| APt400H2 | - | - | - | 2.9 |
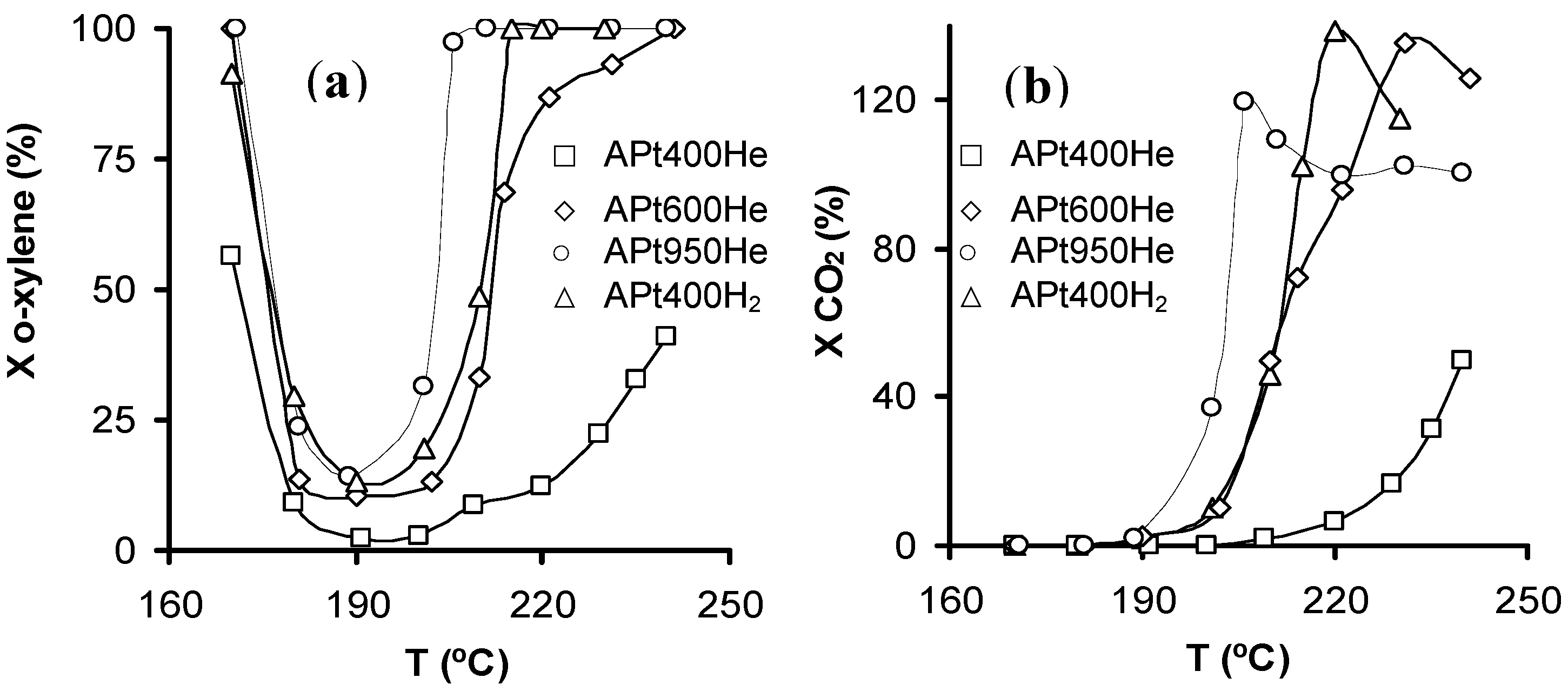
The pre-treated catalysts with different Pt particle sizes were tested in the gas-phase
o-xylene combustion (
Figure 11). The absence of CO
2 converted (X
CO2,
Figure 11b) and the total
o-xylene conversion (X
o-xylene,
Figure 11a) at low reaction temperatures, indicated an
o-xylene removal mainly by adsorption. The light-off curves shift to lower temperatures with increasing Pt particle size (
Figure 11), APt950He and APt400He being the most and least active catalysts, respectively. Therefore, these results suggest that gas-phase
o-xylene combustion over these Pt-catalysts is a structure-sensitive reaction.
Figure 11.
Light-off curves for the supported Pt catalysts: (
a) conversion of
o-xylene and (
b) conversion to CO
2 ([
o-xylene] = 0.10 vol.%,
Q = 3.6 L h
−1, 0.10 g
catalyst). Figures reprinted with permission from [
67]. Copyright 2005, Elsevier.
Figure 11.
Light-off curves for the supported Pt catalysts: (
a) conversion of
o-xylene and (
b) conversion to CO
2 ([
o-xylene] = 0.10 vol.%,
Q = 3.6 L h
−1, 0.10 g
catalyst). Figures reprinted with permission from [
67]. Copyright 2005, Elsevier.
Furthermore, the catalyst activation during the reaction by sintering (
Figure 10) was also reported by other authors [
71,
72]. This behavior seems to be markedly affected by the initial metal particle size of the catalyst. In this context, the CO
2 formation as a function of reaction time was monitored at 190 °C (
Figure 12), in which no
o-xylene combustion to CO
2 was detected (
Figure 11b). As previously mentioned, Pt catalysts are activated after a certain period of time under these experimental conditions and the conversion to CO
2 increases (
Figure 12). However, this behavior was not observed with APt400He, which is the catalyst with the highest Pt-dispersion.
Figure 12.
Conversion of
o-xylene to CO
2 obtained at 190 °C in function of time on stream (TOS) ([
o-xylene] = 0.10 vol.%,
Q = 3.6 L h
−1, 0.10 g
catalyst). Figures reprinted with permission from [
67]. Copyright 2005, Elsevier.
Figure 12.
Conversion of
o-xylene to CO
2 obtained at 190 °C in function of time on stream (TOS) ([
o-xylene] = 0.10 vol.%,
Q = 3.6 L h
−1, 0.10 g
catalyst). Figures reprinted with permission from [
67]. Copyright 2005, Elsevier.
The catalyst activation during reaction time was also pointed out with two consecutive light-off curves [
67]. Light-off curves in the second run were shifted to a lower temperature compared to those obtained with the first run. The difference between both curves, ΔT, is a measurement of the activation degree of the catalyst after the first combustion run. These results showed that the most activated catalyst was APt950He, which was also the most active catalyst during the
o-xylene combustion.
2.4. Influence of the Support Surface Chemistry
If there is a highlighted property of the carbon materials, it is their easily tuneable surface chemistry. Its influence on the catalytic performance in gas-phase VOCs combustion was pointed out by using supported Pt-catalysts on oxidized carbon aerogels. Thus, the mesoporous and macroporous carbon aerogel (ALi900 and ACs900, respectively) were oxidized with different agents: hydrogen peroxide (9.8 M H
2O
2 – AX900H samples) and ammonium peroxydisulfate ((NH
4)
2S
2O
8–AX900S samples) [
73], before being used as Pt catalyst supports.
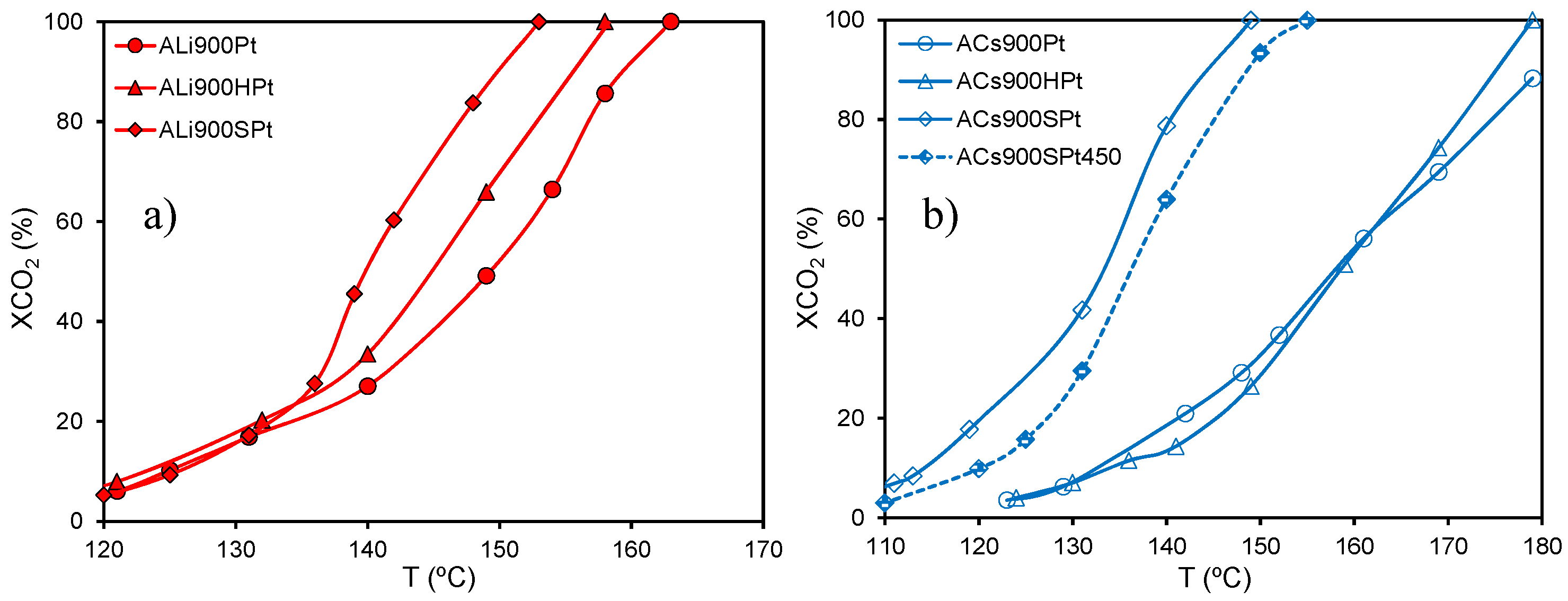
The oxidation treatments performed on the carbon aerogels did not modify significantly their porosity, but their surface chemistry changed from basic to acid as oxygen content increased (
Table 5). The porosity preservation allowed us to study accurately the influence of the surface chemistry on the Pt particle size of the corresponding catalysts, which increased for those prepared on oxidized supports (e.g., 1.6 and 2.4 nm for ALi900 and ALi900S, respectively). This loss of Pt-dispersion was more notorious when the amount of oxygen-containing surface groups increased and, in particular, for macroporous oxidized supports. In this context, the Pt particle size increased from 2.4 to 3.9 nm for ALi900SPt pre-treated at 300 and 450 °C, respectively, while a more evident increase was observed for ACs900SPt pre-treated under the same conditions,
i.e., d
Pt increased from 3.4 to 6.0 nm, respectively. Therefore, the increase of oxygen content in the supports favors the Pt sintering, while large external surface areas (mesoporous supports) prevent in certain extent the sintering, which is more marked with high pre-treatment temperatures.
Table 5.
Amounts of CO and CO2 evolved up to 1000 °C and oxygen content (OTPD) from TPD experiments, measurements of pHPZC and mean Pt particle size for the derivatives Pt/C.
Table 5.
Amounts of CO and CO2 evolved up to 1000 °C and oxygen content (OTPD) from TPD experiments, measurements of pHPZC and mean Pt particle size for the derivatives Pt/C.
| Support/catalyst | CO (µmol g−1) | CO2 (µmol g−1) | OTPD (%) | pHPZC | *dPt(nm) |
|---|
| ALi900 | 420 | 61 | 0.9 | 10.22 | 1.6 |
| ALi900H | 1110 | 335 | 2.9 | 4.94 | 2.0 |
| ALi900S | 2846 | 1621 | 9.7 | 3.16 | 2.4 (3.9) |
| ACs900 | 534 | 59 | 1.0 | 10.28 | 1.5 |
| ACs900H | 832 | 364 | 2.5 | 6.89 | 1.8 |
| ACs900S | 2651 | 1522 | 9.1 | 3.85 | 3.4 (6.0) |
The catalytic activity of the supported Pt catalysts was determined in the gas-phase combustion of benzene, studying the correlation with the surface chemistry once the porosity was comparable after oxidation treatments. In general, the employment of oxidized supports have two consequences: (i) a decrease of the hydrophobic character with the oxygen content of the support [
74], since the oxygen-containing surface groups (called as primary centers) may interact with water molecules by hydrogen bonds; and (ii) a decrease of Pt-dispersion (as indicated). The loss of support hydrophobicity has a detrimental effect on the performance of the catalyst, because the re-adsorption of water generated during the reaction can block the active sites, namely at low reaction temperatures. Nevertheless, when the catalytic behavior for each series is analyzed, the conversion of benzene to CO
2 increased with the order: un-oxidized < H-treated < S-treated supports (
Figure 13).
Figure 13.
Light-off curves obtained in the benzene combustion for Pt catalysts supported on (
a) mesoporous and (
b) macroporous carbon aerogels ([benzene] = 1000 ppm,
Q = 3.6 L h
−1, 0.10 g
catalyst). Figures adapted with permission from [
61]. Copyright 2010, Elsevier.
Figure 13.
Light-off curves obtained in the benzene combustion for Pt catalysts supported on (
a) mesoporous and (
b) macroporous carbon aerogels ([benzene] = 1000 ppm,
Q = 3.6 L h
−1, 0.10 g
catalyst). Figures adapted with permission from [
61]. Copyright 2010, Elsevier.
As previously demonstrated, VOCs combustion with Pt/C catalysts is a sensitive–structure reaction, in which Pt-dispersion would be the main parameter controlling the catalytic activity [
67]. The hydrophilic character increases therefore after oxidation, producing a weak effect on the catalytic performance of oxidation catalysts supported on carbon materials. Therefore, the possible water re-adsorption favored by the increased hydrophilicity of the oxidized support is clearly compensated by the increase of activity induced by large Pt particles formed.
2.5. Carbon versus Inorganic Supports: The Influence of the Hydrophobicity
In order to clarify the influence of hydrophobicity of supports, this study was carried out by analyzing the catalytic activity of Pd and Pt catalysts supported on both carbon and inorganic supports, looking for a deeper hydrophobicity difference taking into account that changes in the hydrophobicity of oxidized carbon aerogels did not seem to influence significantly on the catalytic activity. In this context, two types of coatings were developed on a cordierite monolithic substrate. The first one was formed by a γ-Al
2O
3 layer (A monolith) and the another contained 3.3 wt.% of carbon nanofibers (ACNF monolith) grown
in situ on the A monolith [
63].
The corresponding γ-Al
2O
3 and CNF layers were thin, homogeneous and well-adhered on the cordierite substrate (
Figure 14a). The CNFs layer was developed avoiding the formation of cracks and other defects on the cordierite monolith.
Figure 6a and
Figure 14b show views of A and ACNF monolith channels, respectively, with different topologies being observed for each monolith. Some individual CNFs can be clearly observed through the monolith surface (
Figure 14c). The different morphology obviously influenced on the textural properties of the monolithic supports. Thus,
SBET significantly increased with the CNF coating compared to A monolith (
Table 6). The analysis of the macro- and mesoporosity indicated that both samples are mainly macroporoses, the macropore volume in ACNF being larger than in A (
i.e., 0.157 and 0.134 cm
3 g
−1, respectively) and the macropores being narrower after the CNF coating.
Figure 14.
(a) Photograph of both γ-Al2O3 and CNF coatings on a cordierite monolith. SEM micrographs of the ACNF monolith: (b) cross-section of a monolith channel and (c) detail of CNF grown in situ.
Figure 14.
(a) Photograph of both γ-Al2O3 and CNF coatings on a cordierite monolith. SEM micrographs of the ACNF monolith: (b) cross-section of a monolith channel and (c) detail of CNF grown in situ.
Table 6.
Textural characteristics of the monolithic supports.
Table 6.
Textural characteristics of the monolithic supports.
| Monolith | *SBET (m2 g−1) | Vmacro (cm3 g−1) | Vmeso (cm3 g−1) | *,†Enthalpy (J g −1) |
|---|
| A | 164 | 0.134 | 0.027 | −138.2 |
| ACNF | 371 | 0.157 | 0.028 | −103.8 |
Figure 15.
Catalytic activity of the catalysts (normalized per gram of Pt or Pd) as a function of the reaction temperature: (
a) combustion of benzene, (
b) combustion of toluene and (
c) combustion of
m-xylene. (
d) Water desorption profiles obtained by TPD. Figures adapted with permission from [
63]. Copyright 2006, Elsevier.
Figure 15.
Catalytic activity of the catalysts (normalized per gram of Pt or Pd) as a function of the reaction temperature: (
a) combustion of benzene, (
b) combustion of toluene and (
c) combustion of
m-xylene. (
d) Water desorption profiles obtained by TPD. Figures adapted with permission from [
63]. Copyright 2006, Elsevier.
Pt and Pd catalysts were prepared on both A and ACNF monoliths by the impregnation method and their catalytic activity was studied in the gas-phase combustion of benzene, toluene and m-xylene (
Figure 15a–c, respectively). The conversion of the different VOCs tested, followed the sequence: X
benzene > X
toluene > X
m-xylene regardless the active phase and type of support used. In addition, Pt catalysts were more active than corresponding Pd-catalysts regardless the type of aromatic compound and support employed, the most active catalysts being always those supported on CNF coated monoliths. Furthermore, catalysts supported on A monoliths seem to burn equally any BTX, while those supported on ACNF showed preference to burn benzene better than toluene or
m-xylene (
Figure 15a–c), namely the Pt/ACNF catalyst.
A possible explanation of these results was deduced by determining the adsorptive capacity of water for the A support and the Pt/A catalyst. The water desorption profiles obtained by TPD (
Figure 15d) indicated that the A support (after calcined at 400 °C and exposed to ambient air at room temperature for a month), is able to reincorporate important amounts of water (like water itself or like hydroxyl groups by reaction with the Al atoms). Since the catalysts were pre-treated in H
2 flow at 300 °C before being used in BTX combustions, water should not appear below 300 °C during the TPD experiment. However, a large amount of water was evolved, which should have been produced during the combustion. Therefore, the water adsorption of the alumina phase could produce a decrease on the activity of the corresponding supported catalysts. These results are in agreement with the lower water immersion enthalpy (
Table 6) obtained for ACNF, which corroborates its large hydrophobic character. As a summary, the support hydrophobicity allowed enhancing the activity of supported Pt or Pd catalysts in the gas-phase combustion of BTX.