Valorisation of Vietnamese Rice Straw Waste: Catalytic Aqueous Phase Reforming of Hydrolysate from Steam Explosion to Platform Chemicals
Abstract
:1. Introduction
2. Results and Discussion
2.1. Catalyst Characterization
| Catalyst | Calcination Temperature/°C | pH a | Surface W Loading b/wt% | Surface Area c/m2.g−1 | Mesopore Diameter d/Å | Total Pore Volume d/cm3.g−1 | Surface W Density e/atoms.nm−2 |
|---|---|---|---|---|---|---|---|
| ZrO2 | 500 | 5.0 | - | 78 | 25 | 0.19 | - |
| 5 wt% WZ | 5.6 | 7.7 | 122 | 17 | 0.18 | 2.1 | |
| 10 wt% WZ | 5.3 | 12.6 | 184 | 17 | 0.12 | 2.7 | |
| ZrO2 | 600 | - | - | 44 | 50 | 0.15 | - |
| 5 wt% WZ | 5.7 | 9.3 | 81 | 22 | 0.15 | 3.7 | |
| 10 wt% WZ | 4.6 | 8.4 | 116 | 17 | 0.16 | 3.0 | |
| ZrO2 | 700 | - | - | 86 | 17 | 0.19 | - |
| 5 wt% WZ | 5.5 | 11.7 | 64 | 25 | 0.14 | 6.0 | |
| 10 wt% WZ | 3.3 | 17.7 | 98 | 20 | 0.18 | 7.1 |
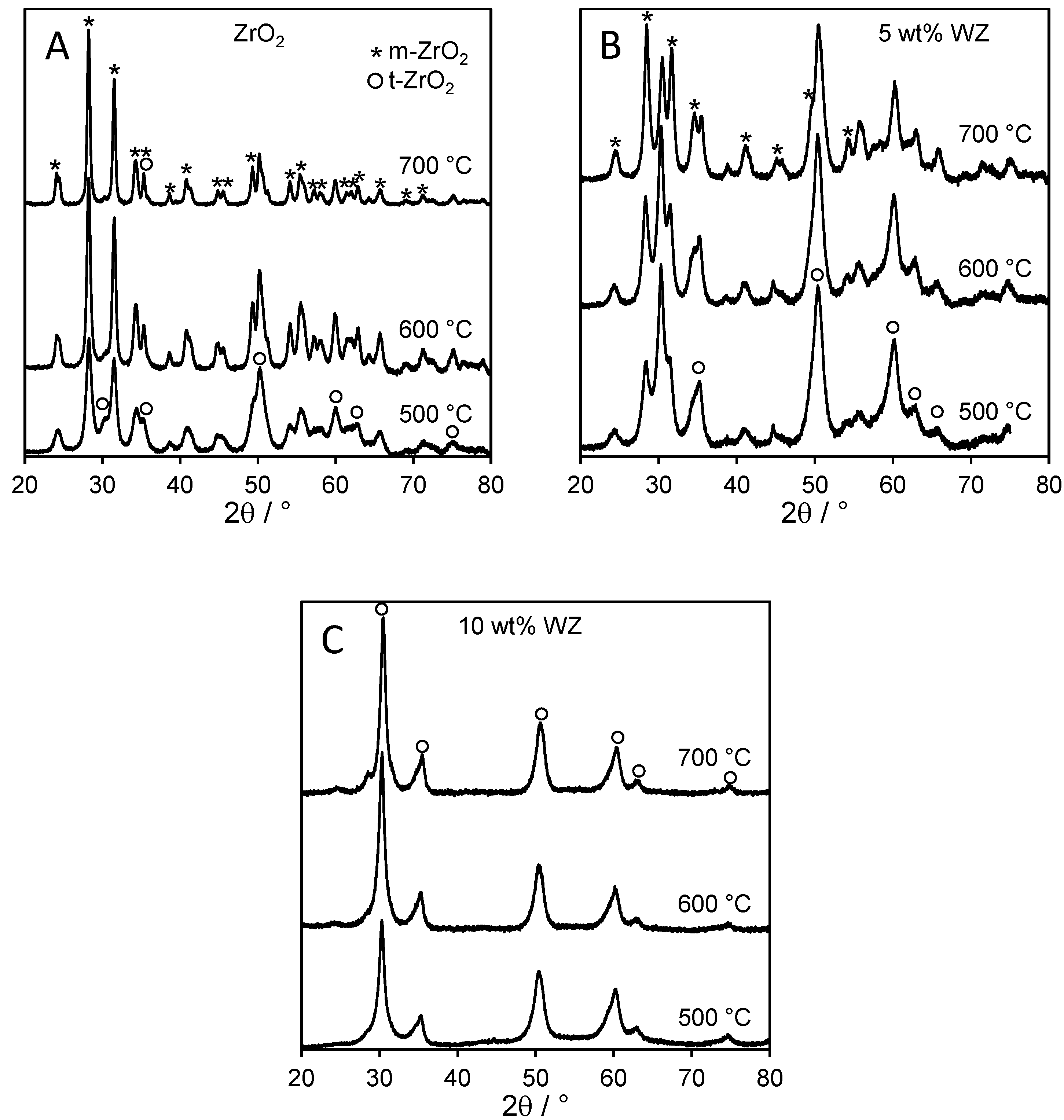
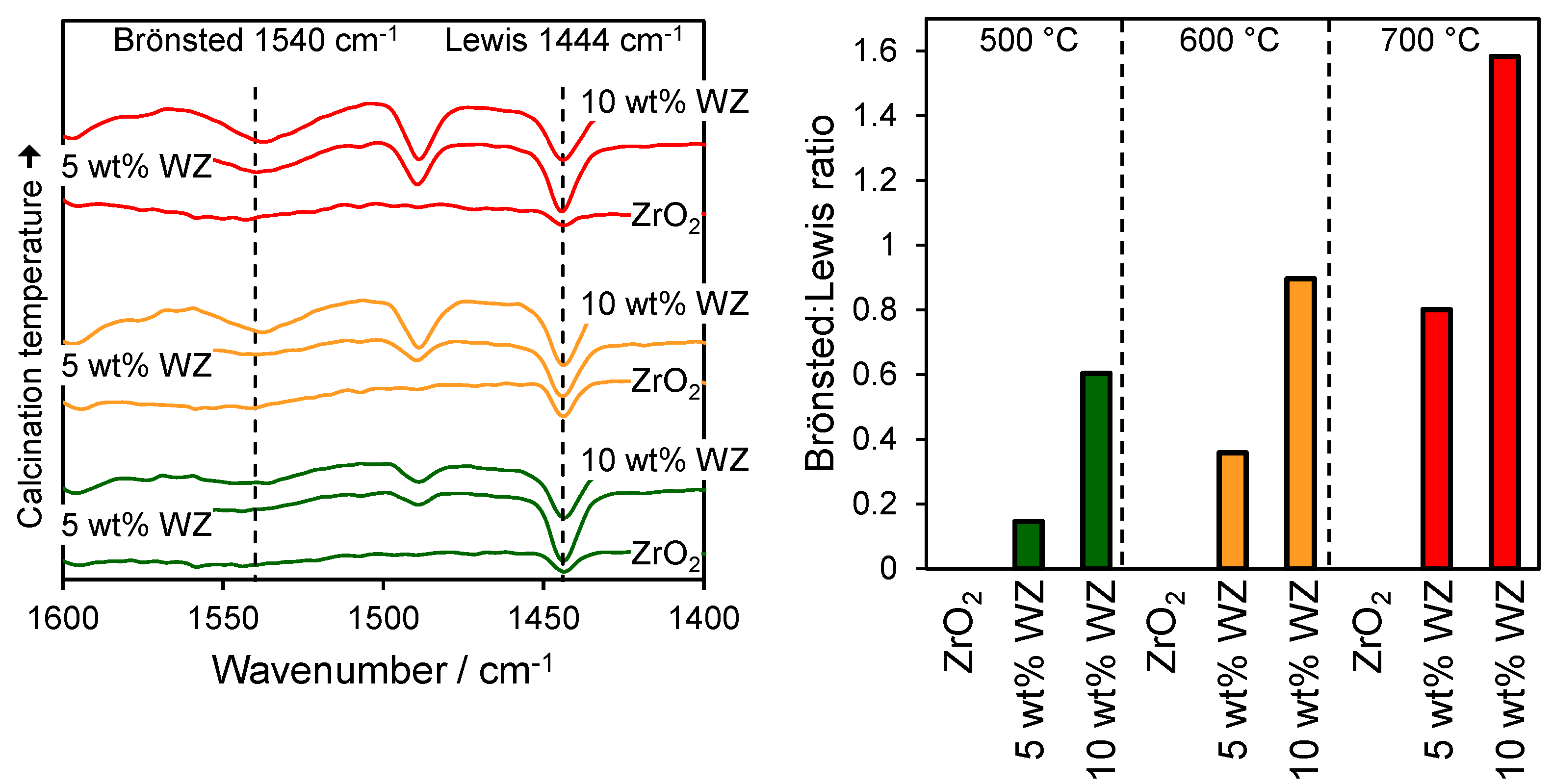
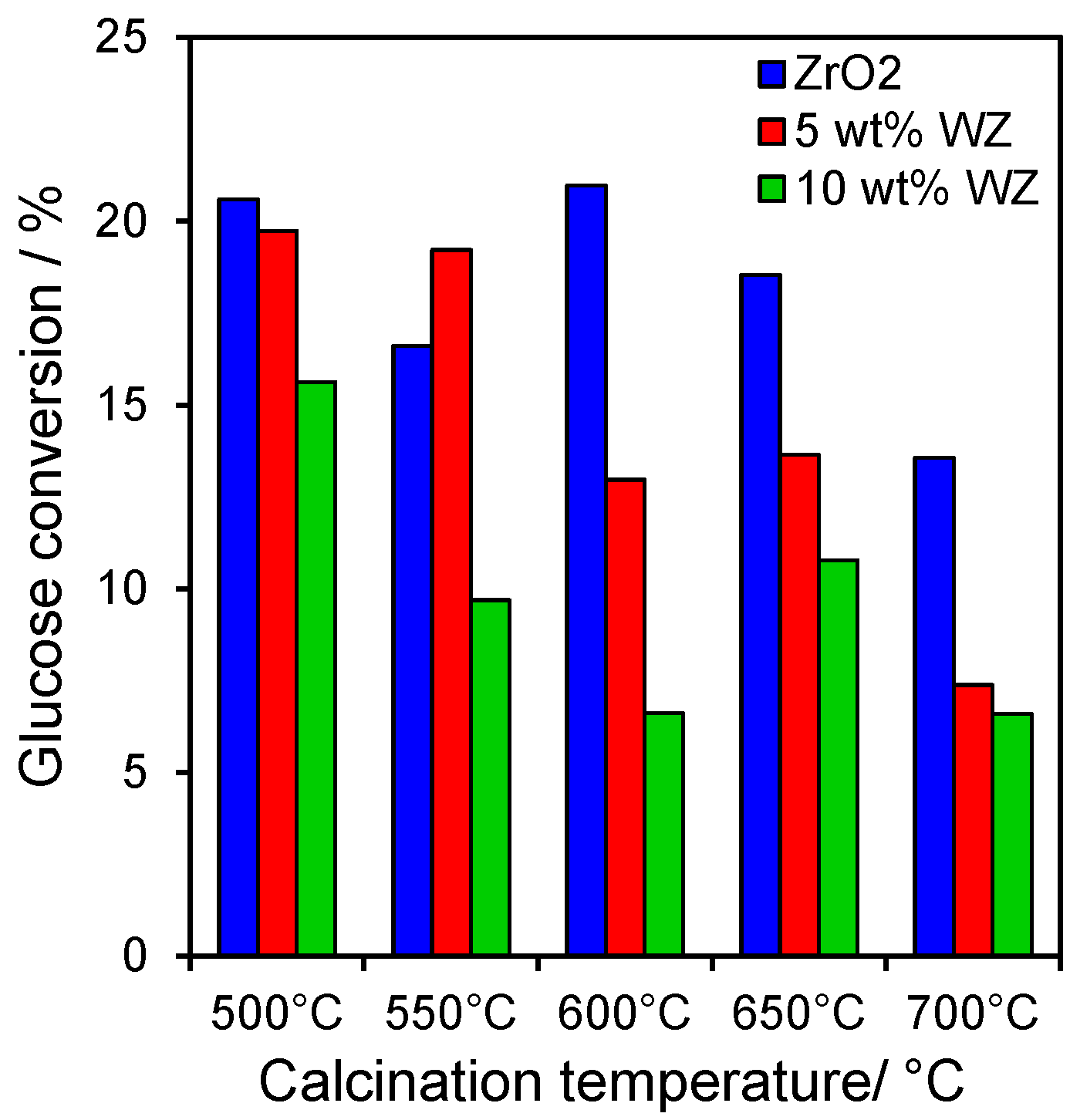
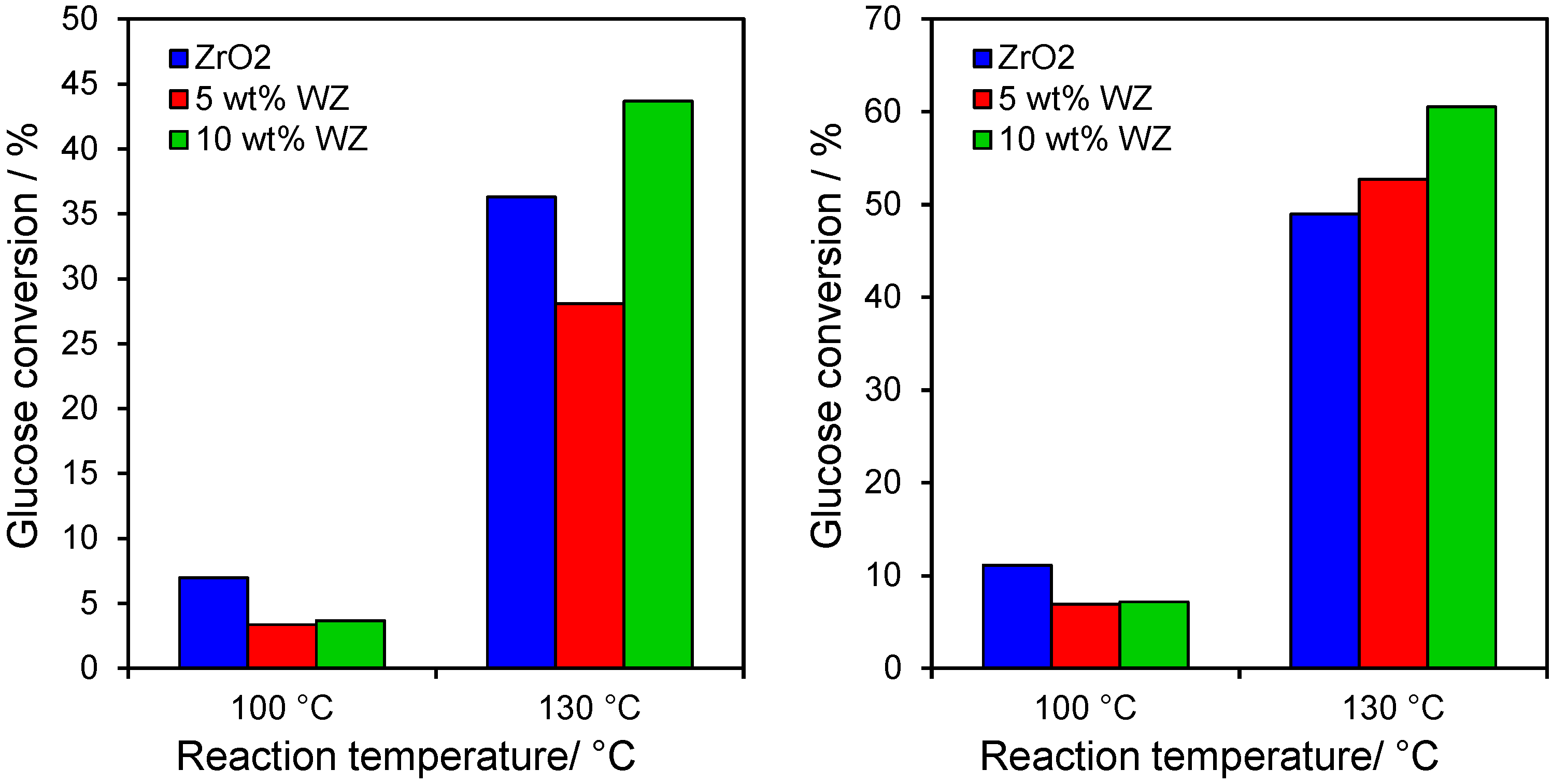
2.2. Catalytic Conversion of Glucose
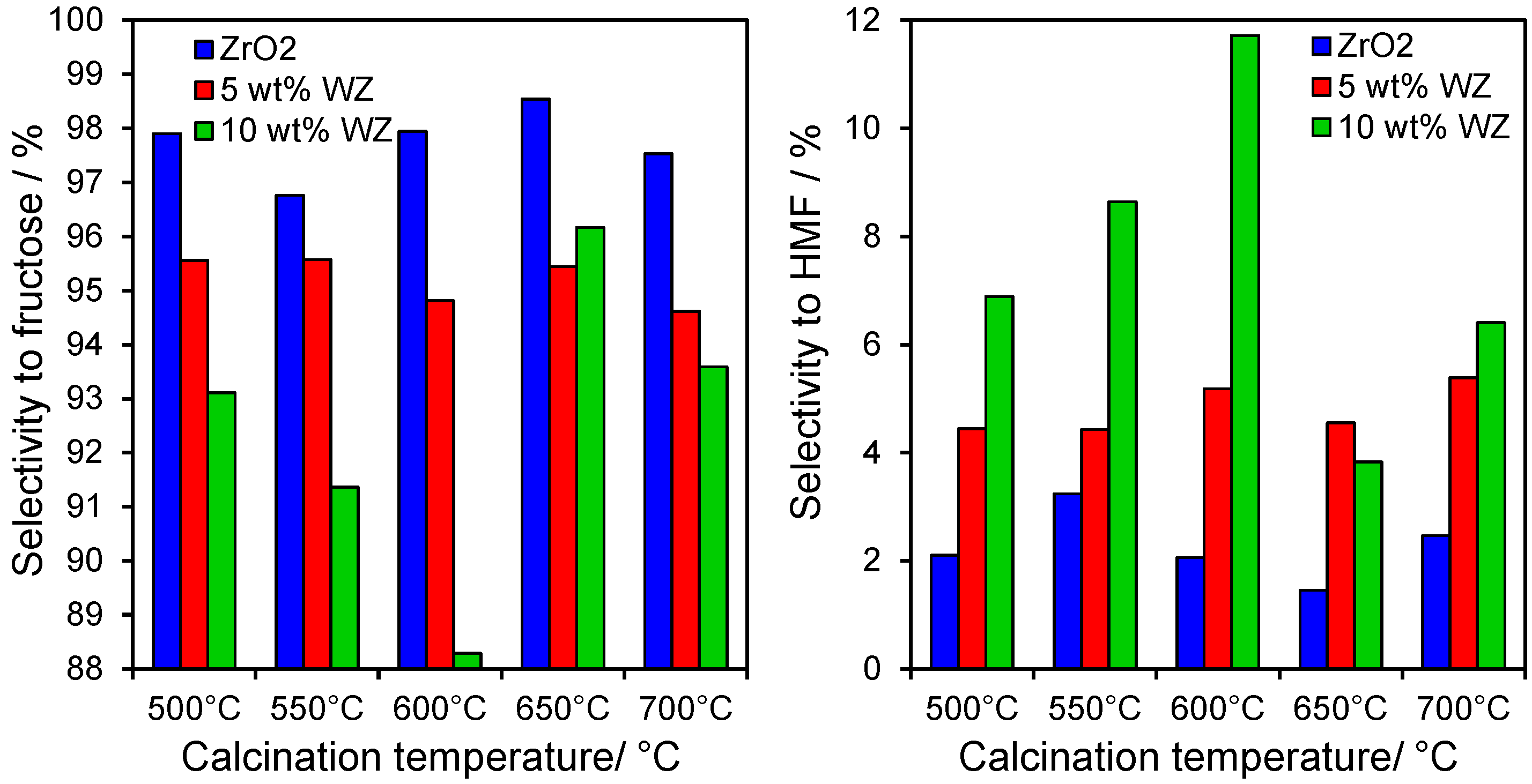
2.3. Catalytic Conversion of Fructose
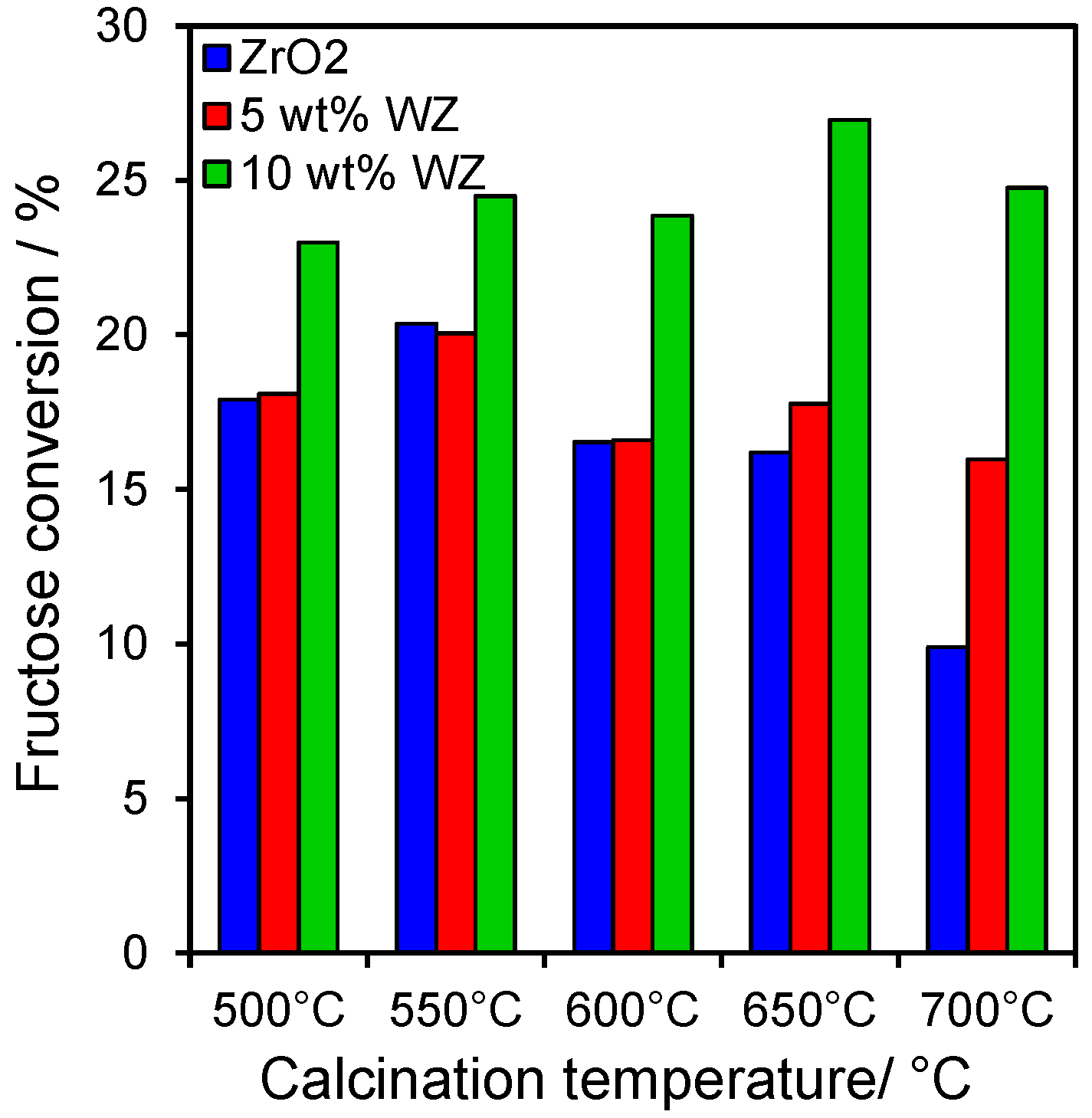
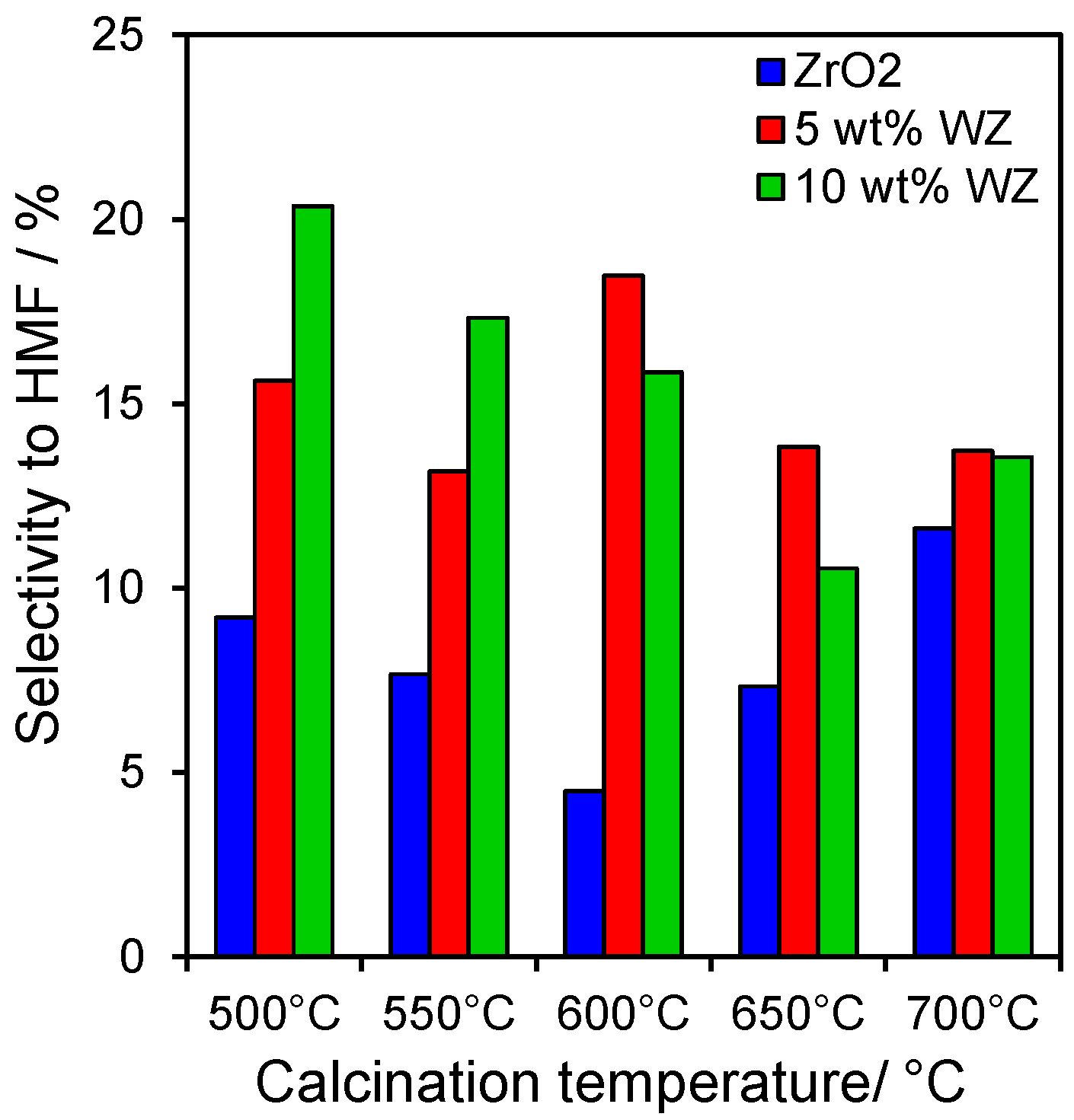
2.4. Catalytic Dehydration of Hydrolysate Sugar and Condensate
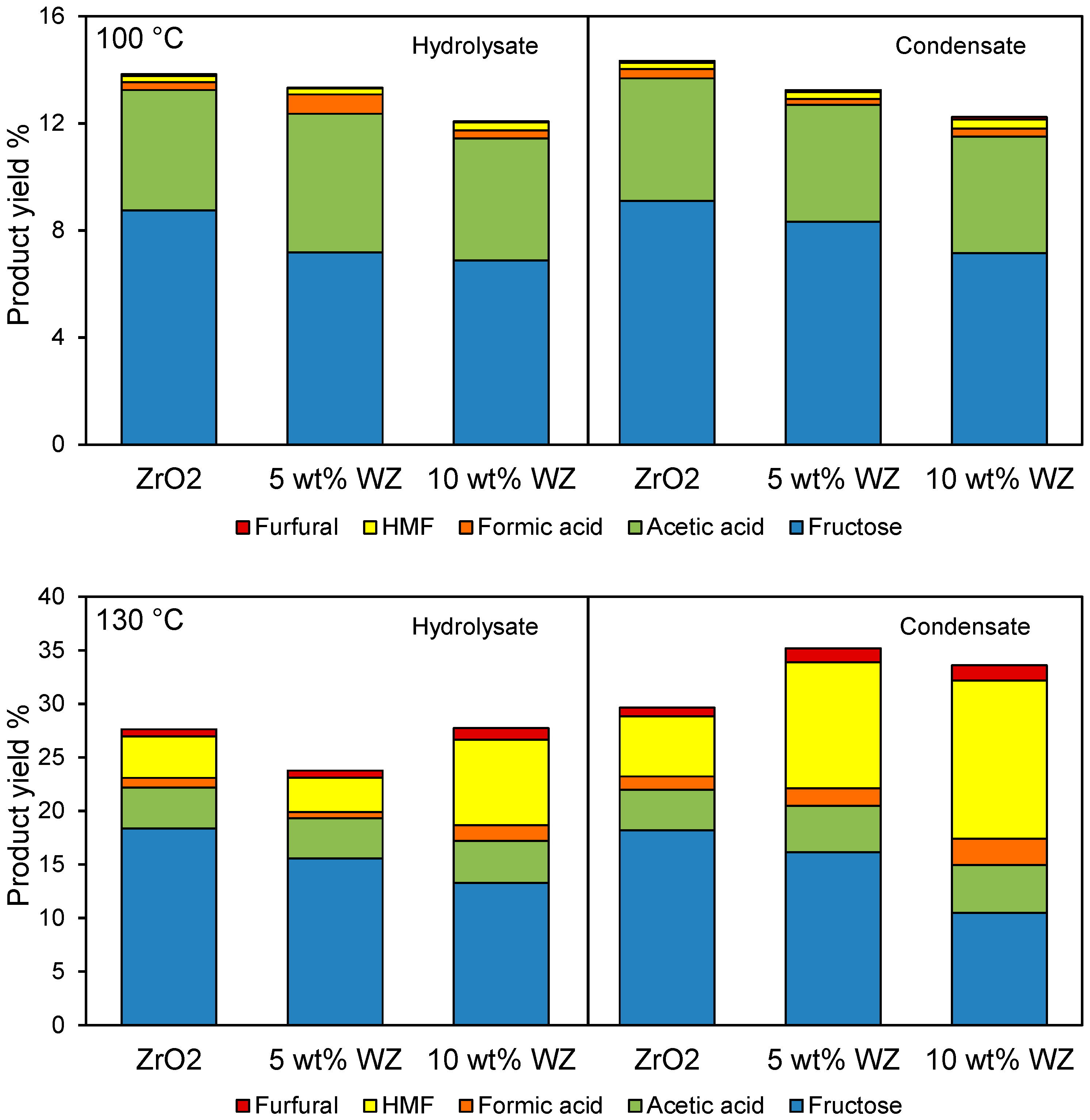
3. Experimental Section
3.1. Catalyst Preparation
3.2. Catalyst Characterization
3.3. Catalytic Reaction
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Lee, A.F.; Bennett, J.A.; Manayil, J.C.; Wilson, K. Heterogeneous catalysis for sustainable biodiesel production via esterification and transesterification. Chem. Soc. Rev. 2014, 43, 7887–7916. [Google Scholar] [CrossRef] [PubMed]
- Wilson, K.; Lee, A.F. Rational design of heterogeneous catalysts for biodiesel synthesis. Catal. Tech. Sci. 2012, 2, 884–897. [Google Scholar] [CrossRef]
- Lee, A. Catalysing sustainable fuel and chemical synthesis. Appl. Petrochem. Res. 2014, 4, 11–31. [Google Scholar] [CrossRef]
- Pilipski, M. Saccharification of cellulose. U.S. Patent 4,235,968, 25 November 1980. [Google Scholar]
- Onda, A.; Ochi, T.; Yanagisawa, K. Selective hydrolysis of cellulose into glucose over solid acid catalysts. Green Chem. 2008, 10, 1033–1037. [Google Scholar] [CrossRef]
- Pang, J.; Wang, A.; Zheng, M.; Zhang, T. Hydrolysis of cellulose into glucose over carbons sulfonated at elevated temperatures. Chem. Commun. 2010, 46, 6935–6937. [Google Scholar] [CrossRef]
- Zhou, C.H.; Xia, X.; Lin, C.X.; Tong, D.S.; Beltramini, J. Catalytic conversion of lignocellulosic biomass to fine chemicals and fuels. Chem. Soc. Rev. 2011, 40, 5588–5617. [Google Scholar] [CrossRef] [PubMed]
- Climent, M.J.; Corma, A.; Iborra, S. Converting carbohydrates to bulk chemicals and fine chemicals over heterogeneous catalysts. Green Chem. 2011, 13, 520–540. [Google Scholar] [CrossRef]
- Kobayashi, H.; Fukuoka, A. Synthesis and utilisation of sugar compounds derived from lignocellulosic biomass. Green Chem. 2013, 15, 1740–1763. [Google Scholar] [CrossRef]
- Moliner, M.; Román-Leshkov, Y.; Davis, M.E. Tin-containing zeolites are highly active catalysts for the isomerization of glucose in water. Proc. Natl. Acad. Sci. USA 2010, 107, 6164–6168. [Google Scholar] [CrossRef] [PubMed]
- Chareonlimkun, A.; Champreda, V.; Shotipruk, A.; Laosiripojana, N. Reactions of C5 and C6-sugars, cellulose, and lignocellulose under hot compressed water (HCW) in the presence of heterogeneous acid catalysts. Fuel 2010, 89, 2873–2880. [Google Scholar] [CrossRef]
- Nakajima, K.; Baba, Y.; Noma, R.; Kitano, M.; N. Kondo, J.; Hayashi, S.; Hara, M. Nb2O5·nH2O as a heterogeneous catalyst with water-tolerant lewis acid sites. J. Am. Chem. Soc. 2011, 133, 4224–4227. [Google Scholar] [CrossRef] [PubMed]
- Zeng, W.; Cheng, D.G.; Chen, F.; Zhan, X. Catalytic conversion of glucose on Al–Zr mixed oxides in hot compressed water. Catal. Lett. 2009, 133, 221–226. [Google Scholar] [CrossRef]
- Osatiashtiani, A.; Lee, A.F.; Brown, D.R.; Melero, J.A.; Morales, G.; Wilson, K. Bifunctional SO4/ZrO2 catalysts for 5-hydroxymethylfufural (5-HMF) production from glucose. Catal. Tech. Sci. 2014, 4, 333–342. [Google Scholar] [CrossRef]
- Qi, X.; Watanabe, M.; Aida, T.M.; Smith, R.L., Jr. Sulfated zirconia as a solid acid catalyst for the dehydration of fructose to 5-hydroxymethylfurfural. Catal. Comm. 2009, 10, 1771–1775. [Google Scholar] [CrossRef]
- Climent, M.J.; Corma, A.; Iborra, S. Conversion of biomass platform molecules into fuel additives and liquid hydrocarbon fuels. Green Chem. 2014, 16, 516–547. [Google Scholar] [CrossRef]
- Chen, F.R.; Coudurier, G.; Joly, J.F.; Vedrine, J.C. Superacid and catalytic properties of sulfated zirconia. J. Catal. 1993, 143, 616–626. [Google Scholar] [CrossRef]
- López, D.E.; Suwannakarn, K.; Bruce, D.A.; Goodwin, J.G., Jr. Esterification and transesterification on tungstated zirconia: Effect of calcination temperature. J. Catal. 2007, 247, 43–50. [Google Scholar] [CrossRef]
- Dos Santos, V.C.; Wilson, K.; Lee, A.F.; Nakagaki, S. Physicochemical properties of WOx/ZrO2 catalysts for palmitic acid esterification. Appl. Catal. B 2015, 162, 75–84. [Google Scholar]
- Karim, A.H.; Triwahyono, S.; Jalil, A.A.; Hattori, H. WO3 monolayer loaded on ZrO2: Property-activity relationship in n-butane isomerization evidenced by hydrogen adsorption and IR studies. Appl. Catal. A 2012, 433–434, 49–57. [Google Scholar] [CrossRef] [Green Version]
- Barton, D.G.; Shtein, M.; Wilson, R.D.; Soled, S.L.; Iglesia, E. Structure and electronic properties of solid acids based on tungsten oxide nanostructures. J. Phys. Chem. B 1999, 103, 630–640. [Google Scholar] [CrossRef]
- Hu, X.; Lievens, C.; Larcher, A.; Li, C.Z. Reaction pathways of glucose during esterification: Effects of reaction parameters on the formation of humin type polymers. Bioresour. Technol. 2011, 102, 10104–10113. [Google Scholar] [CrossRef] [PubMed]
- Elliston, A.; Collins, S.R.A.; Wilson, D.R.; Roberts, I.N.; Waldron, K.W. High concentrations of cellulosic ethanol achieved by fed batch semi simultaneous saccharification and fermentation of waste-paper. Bioresour. Technol. 2013, 134, 117–126. [Google Scholar] [CrossRef] [PubMed]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Giang, C.H.; Osatiashtiani, A.; Dos Santos, V.C.; Lee, A.F.; Wilson, D.R.; Waldron, K.W.; Wilson, K. Valorisation of Vietnamese Rice Straw Waste: Catalytic Aqueous Phase Reforming of Hydrolysate from Steam Explosion to Platform Chemicals. Catalysts 2014, 4, 414-426. https://doi.org/10.3390/catal4040414
Giang CH, Osatiashtiani A, Dos Santos VC, Lee AF, Wilson DR, Waldron KW, Wilson K. Valorisation of Vietnamese Rice Straw Waste: Catalytic Aqueous Phase Reforming of Hydrolysate from Steam Explosion to Platform Chemicals. Catalysts. 2014; 4(4):414-426. https://doi.org/10.3390/catal4040414
Chicago/Turabian StyleGiang, Cao Huong, Amin Osatiashtiani, Vannia Cristina Dos Santos, Adam F. Lee, David R. Wilson, Keith W. Waldron, and Karen Wilson. 2014. "Valorisation of Vietnamese Rice Straw Waste: Catalytic Aqueous Phase Reforming of Hydrolysate from Steam Explosion to Platform Chemicals" Catalysts 4, no. 4: 414-426. https://doi.org/10.3390/catal4040414
APA StyleGiang, C. H., Osatiashtiani, A., Dos Santos, V. C., Lee, A. F., Wilson, D. R., Waldron, K. W., & Wilson, K. (2014). Valorisation of Vietnamese Rice Straw Waste: Catalytic Aqueous Phase Reforming of Hydrolysate from Steam Explosion to Platform Chemicals. Catalysts, 4(4), 414-426. https://doi.org/10.3390/catal4040414






