Mixed Enzyme Systems for Delignification of Lignocellulosic Biomass
Abstract
:1. Introduction
1.1. Precedent
1.2. Impact
1.3. Objective
| Lignocellulosic material | Cellulose (%) | Hemicellulose (%) | Lignin (%) |
|---|---|---|---|
| Hardwood Stems | 40–55 | 24–40 | 18–25 |
| Softwood Stems | 45–50 | 25–35 | 25–35 |
| Corn Cobs | 45 | 35 | 15 |
| Grasses | 25–40 | 35–50 | 10–30 |
| Paper | 85–99 | 0 | 0–15 |
| Wheat Straw | 30 | 50 | 15 |
| Leaves | 15–20 | 80–85 | 0 |
| Newspaper | 40–55 | 25–40 | 18–30 |
| Switchgrass | 45 | 31 | 12 |
2. Oxidative Enzymes for Lignin Removal
2.1. Laccase and the Mediator System
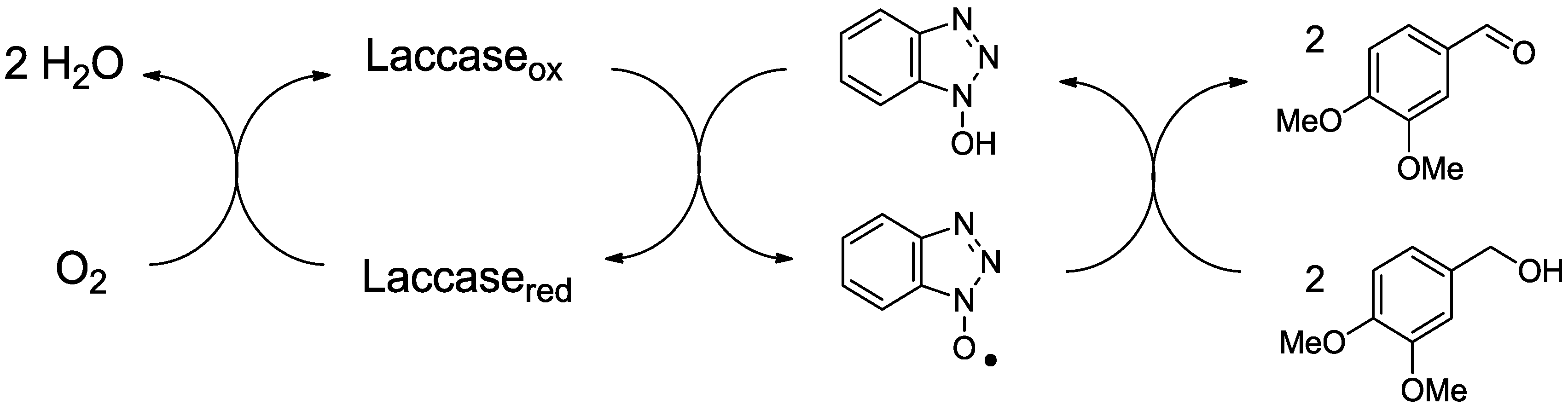
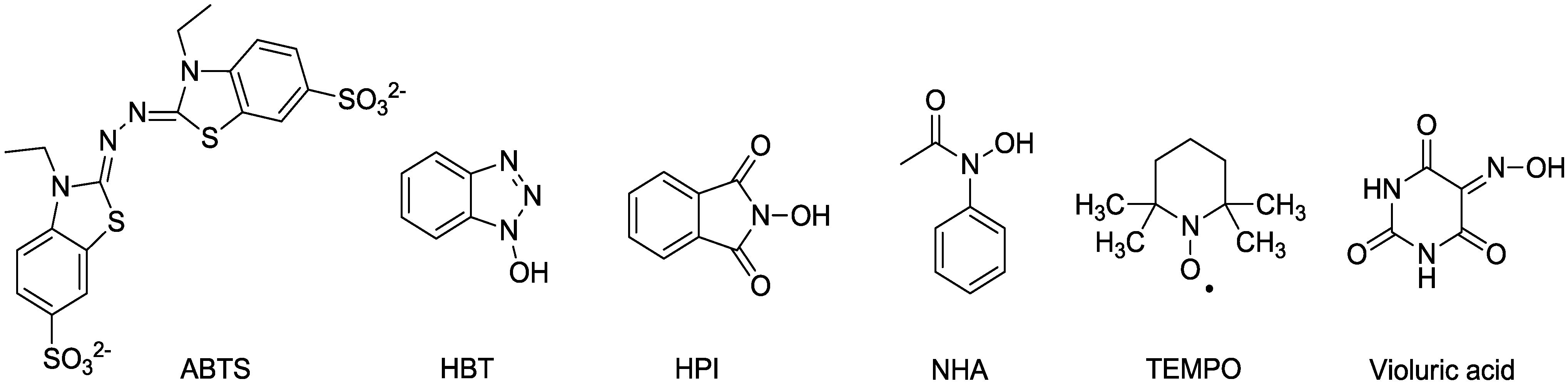
2.2. MnP
3. Hydrolytic Enzymes for Lignin Removal
3.1. Xylanase
3.2. Aspects of Xylanase Structure Relative to Catalysis and Substrate Binding
| Xylanase † | Family | Mass (Da) ‡ | pI ‡ | Entry | Reference |
|---|---|---|---|---|---|
| Aspergillus niger Xylanase C | 10 | 35,486 | 6.17 | XYNC_ASPNC | [96] |
| Cellulomonas fimi CEX/Xylanase B | 10 | 51,219 | 6.19 | GUX_CELFI | [79,90] |
| Clostridium thermocellum Xylanase Z | 10 | 92,263 | 5.56 | XYNZ_CLOTH | [79,90] |
| Pseudomonas fluorescens Xylanase A | 10 | 64,804 | 6.08 | XYNA_CELJU | [79,90] |
| Pseudomonas fluorescens Xylanase B | 10 | 64,363 | 6.37 | XYNB_CELJU | [79,90] |
| Streptomyces lividans Xylanase A | 10 | 51,163 | 6.18 | XYNA_STRLI | [79,91] |
| Thermoascus aurantiacus Xylanase | 10 | 35,686 | 5.82 | XYNA_THEAU | [90] |
| Aspergillus kawachii Xylanase 3 | 11 | 22,627 | 4.07 | XYN3_ASPKW | [96] |
| Bacillus agaradhaerens Xylanase | 11 | 23,152 | 8.58 | Q7SIE3_BACAG | [96,103] |
| Bacillus circulans Xylanase | 11 | 23,359 | 9.44 | XYNA_BACCI | [79,90,96] |
| Bacillus pumilus Xylanase A | 11 | 25,491 | 9.33 | XYNA_BACPU | [90,96] |
| Bacillus subtilis Xylanase A | 11 | 23,345 | 9.44 | XYNA_BACSU | [90] |
| Clostridium saccharobutylicum Xylanase | 11 | 29,033 | 9.24 | XYNA_CLOSA | [90] |
| Fibrobacter succinogenes Xylanase C | 11 | 66,403 | 7.96 | XYNC_FIBSS | [96] |
| Penicillium citrinium Xylanase | 11 | 23,480 | 4.69 | Q2PGY1 | [104] |
| Thermomyces lanuginosus Xylanase | 11 | 24,356 | 4.77 | XYNA_THELA | [79,96] |
| Trichoderma harzianum Xylanase | 11 | 20,703 | 8.71 | XYN_TRIHA | [79] |
| Trichoderma reesei Xylanase I | 11 | 24,583 | 5.00 | XYN1_HYPJE | [79,96] |
| Trichoderma reesei Xylanase II | 11 | 24,172 | 8.89 | XYN2_HYPJE | [79,96] |

3.3. Susceptibility of Xylanase to Oxidative Inactivation
3.4. Protection of Xylanase via Adsorption to Pulp
4. Mixed Enzyme Systems on Wood, Straw, and Grass (Biopulping)
5. Mixed Enzyme Systems on Pulp
5.1. Sequential Xylanase and Laccase Application
5.2. Simultaneous Xylanase and Laccase Application
5.3. Xylanase and MnP Application
6. Obstacles
| Year/Reference | Xylanase | Laccase | Mediator | Sequential or simultaneous? | Reaction conditions | Pulp type | Initial kappa | Bleach sequence following enzyme(s) | End bleach sequence | Delignification (%) | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Dose (per g od pulp) | Source organism or commercial product | Dose (per g od pulp) | Source organism or commercial product | Dose | ID | pH | Buffer | Temperature (°C) | Pulp consistency (%) | Time (h) | Brightness | Kappa | ||||||
| 1993/[160] | - | - | - | - | - | - | - | - | - | - | - | - | Pine Kraft | 33.7 | P | 35.9 | 21.8 | 35.3 |
| 2 units | Streptomyces olivochromogenes | - | - | - | - | - | 5.0 | citrate | 45 | 2.5 | 24 | Pine Kraft | 31.8 | P | 55.8 | 16.5 | 48.1 | |
| - | - | 6 units | Phlebia radita | none | none | - | 4.5 | dimethyl- succinate | room temperature | 2.5 | 24 | Pine Kraft | 33.7 | P | 38.8 | 22.1 | 34.4 | |
| 2 units | Streptomyces olivochromogenes | 6 units | Phlebia radita | none | none | Sequential XL | 5.0 then 4.5 | citrate then dimethyl- succinate | 45 then room temperature | 2.5 | 24 then 24 | Pine Kraft | 31.8 | P | 56.5 | 15.8 | 50.3 | |
| 1997/[7] | - | - | - | - | - | - | - | - | - | - | - | - | O2 Pine Kraft | 8.6 | EQP | 80.3 | 4.8 | 44.2 |
| 6 units | Ecopulp X-200 | - | - | - | - | - | 4.5 | none | 45 | 10 | 2 | O2 Pine Kraft | 8.6 | QP | 83.2 | 3.4 | 60.5 | |
| - | - | 40 units | Trametes hirsuta | 1% | HBT | - | 4.5 | none | 45 | 10 | 2 | O2 Pine Kraft | 8.6 | QP | 87.5 | 2.8 | 67.4 | |
| 6 units | Ecopulp X-200 | 40 units | Trametes hirsuta | 1% | HBT | Sequential XL | 4.5 | none | 45 | 10 | 2 | O2 Pine Kraft | 8.6 | QP | 88.5 | 2.3 | 73.3 | |
| 6 units | Ecopulp X-200 | 40 units | Trametes hirsuta | 1% | HBT | Sequential LX | 4.5 | none | 45 | 10 | 2 | O2 Pine Kraft | 8.6 | QP | 87.6 | 2.3 | 73.3 | |
| 6 units | Ecopulp X-200 | 40 units | Trametes hirsuta | 1% | HBT | Simultaneous | 4.5 | none | 45 | 10 | 2 | O2 Pine Kraft | 8.6 | QP | 88.1 | 2.5 | 70.9 | |
| 1997/[13] | - | - | - | - | - | - | - | - | - | - | - | - | Pine Kraft | 22.6 | EP | 40.1 | 19.3 | 14.6 |
| 12 units | Trichoderma reesei | - | - | - | - | - | 5.0 | none | 45 | 5 | 2 | Pine Kraft | 22.6 | EP | 43.9 | 15.7 | 30.5 | |
| - | - | 30 units | Trametes versicolor | 1% | HBT | - | 4.5 | none | 45 | 10 | 2 | Pine Kraft | 22.6 | EP | 46.4 | 12.8 | 43.4 | |
| 12 units | Trichoderma reesei | 30 units | Trametes versicolor | 1% | HBT | Sequential XL | 5.0 then 4.5 | none | 45 | 5 then 10 | 2 then 2 | Pine Kraft | 22.6 | EP | 50.8 | 10.4 | 54.0 | |
| 2002/[8] | - | - | - | - | - | - | - | 4.5 | none | 45 | 4 | 2 | Pine Kraft | 23.5 | EP | 52.1 | 13.5 | 42.5 |
| 6 units | Trichoderma reesei | - | - | - | - | - | 4.5 | none | 45 | 4 | 2 | Pine Kraft | 23.5 | EP | 55.4 | 11.4 | 51.5 | |
| - | - | 30 units | Trametes hirsuta | 1% | HBT | - | 4.5 | none | 45 | 4 | 2 | Pine Kraft | 23.5 | EP | 58.4 | 9.7 | 58.7 | |
| - | - | 30 units | Trametes hirsuta | 1% | NHA | - | 4.5 | none | 45 | 4 | 2 | Pine Kraft | 23.5 | EP | 59.5 | 8.5 | 63.8 | |
| 6 units | Trichoderma reesei | 30 units | Trametes hirsuta | 1% | HBT | Sequential XL | 4.5 | none | 45 | 4 | 2 | Pine Kraft | 23.5 | EP | 61.1 | 8.1 | 65.5 | |
| 6 units | Trichoderma reesei | 30 units | Trametes hirsuta | 1% | NHA | Sequential XL | 4.5 | none | 45 | 4 | 2 | Pine Kraft | 23.5 | EP | 62.2 | 6.9 | 70.6 | |
| 6 units | Trichoderma reesei | 30 units | Trametes hirsuta | 1% | HBT | Simultaneous | 4.5 | none | 45 | 4 | 2 | Pine Kraft | 23.5 | EP | 58.3 | 9.4 | 60.0 | |
| 6 units | Trichoderma reesei | 30 units | Trametes hirsuta | 1% | NHA | Simultaneous | 4.5 | none | 45 | 4 | 2 | Pine Kraft | 23.5 | EP | 61.3 | 7.7 | 67.2 | |
| 2002/[9] | - | - | - | - | - | - | - | - | - | - | - | - | Wheat Straw | 24.0 | E | nr | 17.3 | 27.9 |
| 1 unit | Pulpzyme HC | - | - | - | - | - | 7.0 | phosphate | 50 | 5 | 2 | Wheat Straw | 24.0 | E | nr | 15.5 | 35.4 | |
| - | - | 25 units | Pycnoporus cinnabarinus | 3% | HBT | - | 5.0 | acetate | 50 | 5 | 4 | Wheat Straw | 24.0 | E | nr | 12.7 | 47.0 | |
| 1 unit | Pulpzyme HC | 25 units | Pycnoporus cinnabarinus | 3% | HBT | Sequential XL | 7.0 then 5.0 | phosphate then acetate | 50 | 5 | 2 then 4 | Wheat Straw | 24.0 | E | nr | 8.5 | 64.6 | |
| 2006/[170] | - | - | - | Trametes versicolor | 3% | HBT | - | 4.0 | acetate | 45 | 15 | 5 | Eucalyptus Kraft | 18.2 | EpDoEpD1 (0.15 kappa factor) | 88.0 | nr | nr |
| 0.75 mg | Pulpzyme-HC | 60 units | Trametes versicolor | 3% | HBT | Sequential XL | 8.0 then 4.0 | none then acetate | 50 then 45 | 10 then 15 | 2 then 5 | Eucalyptus Kraft | 18.2 | EpDoEpD1 (0.12 kappa factor) | 88.0 | nr | nr | |
| 0.75 mg | Pulpzyme-HC | 60 units | Trametes versicolor | 3% | HBT | Simultaneous | 4.0 | acetate | 45 | 15 | 5 | Eucalyptus Kraft | 18.2 | EpDoEpD1 (0.12 kappa factor) | 88.3 | nr | nr | |
| 2009/[49] | 3 units | Bacillus sp. | - | - | - | - | - | 7.0 | Tris | 50 | 10 | 2 | O2 Eucalyptus Kraft | 8.4 | none | 53.7 | 8.4 | 0 |
| - | - | 10.5 units | NS-51002
(T. villosa) | 1.5% | HBT | - | 4.0 | tartrate | 30 | 5 | 4 | O2 Eucalyptus Kraft | 8.4 | E | 67.0 | 5.0 | 40.5 | |
| 3 units | Bacillus sp. | 10.5 units | NS-51002
(T. villosa) | 1.5% | HBT | Sequential XL | 7.0 then 4.0 | Tris then tartrate | 50 then 30 | 10 then 5 | 2 then 4 | O2 Eucalyptus Kraft | 8.4 | E | 70.2 | 4.0 | 52.4 | |
| 2010/[161] | 3 units | Bacillus sp. | - | - | - | - | - | 7.0 | Tris | 50 | 10 | 2 | O2 Eucalyptus Kraft | 8.4 | none | 53.7 | 8.4 | 0 |
| - | - | 10.5 units | NS-51002
(T. villosa) | 1.5% | HBT | - | 4.0 | tartrate | 30 | 5 | 4 | O2 Eucalyptus Kraft | 8.4 | none | 59.8 | 7.0 | 16.7 | |
| 3 units | Bacillus sp. | 10.5 units | NS-51002
(T. villosa) | 1.5% | HBT | Sequential XL | 7.0 then 4.0 | Tris then tartrate | 50 then 30 | 10 then 5 | 2 then 4 | O2 Eucalyptus Kraft | 8.4 | none | 58.8 | 5.2 | 38.1 | |
| 2010/[16] | 10 units | Penicillium oxalicum | - | - | - | - | - | 9.0 | none | 55 | 10 | 2 | 60:40 Eucalyptus: Poplar | 14.0 | none | 37.0 | 13.4 | 4.3 |
| 9.6 units | Penicillium oxalicum | 0.4 units | Pleurotus ostreatus | none | none | Simultaneous | 9.0 | none | 55 | 10 | 2 | 60:40 Eucalyptus: Poplar | 14.0 | none | 38.3 | 12.7 | 9.3 | |
| 7.7 units | Penicillium oxalicum | 0.3 units | Pleurotus ostreatus | none | none | Simultaneous | 9.0 | none | 55 | 10 | 3 | 60:40 Eucalyptus: Poplar | 14.0 | none | 38.3 | 11.1 | 20.7 | |
| Treatment † prior to Ep stage | Kappa number | Delignification (%) | ISO brightness (%) |
|---|---|---|---|
| None | 8.0 | - | 59.3 |
| Xylanase (no H2O2) | 6.2 | 23 | 65.6 |
| MnP (0.0612 mmole H2O2/g pulp) * | 6.6 ‡ | 18 | 62.5‡ |
| Xylanase + MnP (simultaneous) (0.0612 mmole H2O2/g pulp) * | 5.2 | 35 | 70.5 |
| Xylanase + MnP (simultaneous) ** | 4.3 | 46 | 78.1 |
| Xylanase→MnP (sequential) ** | 6.6 | 18 | 64.4 |
| MnP→Xylanase (sequential) ** | 6.4 | 20 | 65.9 |
7. Conclusions
Acknowledgements
Abbreviations
| ABTS | 2,2'-azinobis(3-ethylbenzothiazoline-6-sulfonic acid) |
| Ep | peroxide fortified extraction |
| EPR | electron paramagnetic resonance |
| ESEM | environmental scanning electron microscopy |
| HBT | 1‑hydroxybenzotriazole |
| HexA | hexenuronic acid |
| HPI | N-hydroxyphthalimide |
| kDa | kilodalton |
| HWKP | hardwood kraft pulp |
| L | laccase treatment stage |
| LCC | lignin carbohydrate complex |
| LiP | lignin peroxidase |
| LMS | laccase mediator system |
| MnP | manganese peroxidase |
| NHA | N-hydroxyacetanilide |
| NMR | nuclear magnetic resonance |
| od | oven dried |
| ONP | old newspaper pulp |
| P | peroxide stage |
| TEMPO | 2,2,6,6-tetramethyl-1-piperidinyloxy, free radical |
| TMB | 1,2,3,4-tetramethoxybenzene |
| X | xylanase treatment stage |
Conflicts of Interest
References
- Sigoillot, C.; Camarero, S.; Vidal, T.; Record, E.; Asther, M.; Pérez-Boada, M.; Martínez, M.J.; Sigoillot, J.-C.; Asther, M.; Colom, J.F.; et al. Comparison of different fungal enzymes for bleaching high-quality paper pulps. J. Biotechnol. 2005, 115, 333–343. [Google Scholar] [CrossRef]
- Eriksson, K.-E.L.; Blanchette, R.A.; Ander, P. Microbial and Enzymatic Degradation of Wood and Wood Components; Springer-Verlag: New York, NY, USA, 1990; pp. 1–87. [Google Scholar]
- Sweeney, M.D.; Xu, F. Biomass converting enzymes as industrial biocatalysts for fuels and chemicals: Recent developments. Catalysts 2012, 2, 244–263. [Google Scholar] [CrossRef]
- Kirk, T.K.; Farrell, R.L. Enzymatic “combustion”: The microbial degradation of lignin. Annu. Rev. Microbiol. 1987, 41, 465–505. [Google Scholar] [CrossRef]
- Tien, M.; Kirk, T.K. Lignin-degrading enzyme from the Hymenomycete Phanerochaete chrysosporium burds. Science 1983, 221, 661–663. [Google Scholar]
- Glenn, J.K.; Morgan, M.A.; Mayfield, M.B.; Kuwahara, M.; Gold, M.H. An extracellular H2O2-requiring enzyme preparation involved in lignin biodegradation by the white rot basidiomycete Phanerochaete chrysosporium. Biochem. Biophys. Res. Commun. 1983, 114, 1077–1083. [Google Scholar] [CrossRef]
- Poppius-Levlin, K.; Wang, W.; Ranua, M.; Niku-Paavola, M.L.; Viikari, L. Biobleaching of chemical pulps by laccase/mediator systems. In Proceedings of TAPPI Biological Science Symposium, San Francisco, CA, USA, 20–23 October 1997; TAPPI: San Francisco, CA, USA, 1997; pp. 329–333. [Google Scholar]
- Oksanen, T.; Buchert, J.; Amann, M.; Candussio, A.; Viikari, L. Boosting of LMS-bleaching with hemicellulases. In Progress in Biotechnology; Viikari, L., Lantto, R., Eds.; Elsevier: Amsterdam, The Netherlands, 2002; Volume 21, pp. 255–262. [Google Scholar]
- Herpoël, I.; Jeller, H.; Fang, G.; Petit-Conil, M.; Bourbonnais, R.; Robert, J.L.; Asther, M.; Sigoillot, J.C. Efficient enzymatic delignification of wheat straw pulp by a sequential xylanase-laccase mediator treatment. J. Pulp Pap. Sci. 2002, 28, 67–71. [Google Scholar]
- Surma-Ślusarska, B.; Leks-Stępień, J. TCF bleaching of kraft pulps with laccase and xylanase. J. Wood Chem. Technol. 2001, 21, 361–370. [Google Scholar] [CrossRef]
- Viikari, L.; Oksanen, T.; Buchert, J.; Amann, M.; Candussio, A. Combined action of hemicellulases and oxidases in bleaching. In Proceedings of 10th International Symposium on Wood and Pulping Chemistry–10th Biennial ISWPC, Yokohama, Japan, 7–10 June 1999; Japan TAPPI: Yokohama, Japan, 1999; pp. 504–508. [Google Scholar]
- Freudenreich, J.; Amann, M.; Fritz-Langhals, E.; Stohrer, J. Understanding the Lignozym-process. In Proceedings of International Pulp Bleaching Conference, Helsinki, Finland, 1998; IBBC: Helsinki, Finland, 1998; pp. 71–76. [Google Scholar]
- Oksanen, T.; Amann, M.; Candussio, A.; Buchert, J.; Viikari, L. Role of xylanase in laccase-mediator delignification of kraft pulps. In Proceedings of 9th International Symposium on Wood and Pulping Chemistry, Montreal, Canada, 9–12 June 1997; CPPA: Montreal, Canada, 1997; pp. 83-81–83-84. [Google Scholar]
- Nelson, P.J.; Chin, C.W.J.; Viikari, L.; Tenkanen, M. The use of a laccase-mediator stage in bleaching of eucalypt kraft pulps. Appita J. 1998, 51, 451–455. [Google Scholar]
- Richard, T.L. Challenges in Scaling Up Biofuels Infrastructure. Science 2010, 329, 793–796. [Google Scholar] [CrossRef]
- Dwivedi, P.; Vivekanand, V.; Pareek, N.; Sharma, A.; Singh, R.P. Bleach enhancement of mixed wood pulp by xylanase-laccase concoction derived through co-culture strategy. Appl. Biochem. Biotechnol. 2010, 160, 255–268. [Google Scholar] [CrossRef]
- Feijoo, G.; Moreira, M.T.; Álvarez, P.; Lú-Chau, T.A.; Lema, J.M. Evaluation of the enzyme manganese peroxidase in an industrial sequence for the lignin oxidation and bleaching of eucalyptus kraft pulp. J. Appl. Polym. Sci. 2008, 109, 1319–1327. [Google Scholar] [CrossRef]
- Jørgensen, H.; Kristensen, J.B.; Felby, C. Enzymatic conversion of lignocellulose into fermentable sugars: Challenges and opportunities. Biofuel Bioprod. Bior. 2007, 1, 119–134. [Google Scholar] [CrossRef]
- Kumar, P.; Barrett, D.M.; Delwiche, M.J.; Stroeve, P. Methods for pretreatment of lignocellulosic biomass for efficient hydrolysis and biofuel production. Ind. Eng. Chem. Res. 2009, 48, 3713–3729. [Google Scholar] [CrossRef]
- Bermek, H.; Li, K.; Eriksson, K.-E.L. Pulp bleaching with manganese peroxidase and xylanase: A synergistic effect. Tappi J. 2000, 83, 69. [Google Scholar]
- Olsen, W.L.; Gallagher, H.P.; Burris, K.A.; Bhattacharjee, S.S.; Slocomb, J.P.; DeWitt, D.M. Enzymatic delignification of lignocellulosic material. EP0406617, 9 January 1991. [Google Scholar]
- Niku-Paavola, M.L.; Ranua, M.; Suurnäkki, A.; Kantelinen, A. Effects of lignin-modifying enzymes on pine kraft pulp. Bioresour. Technol. 1994, 50, 73–77. [Google Scholar] [CrossRef]
- Thurston, C.F. The structure and function of fungal laccases. Microbiology 1994, 140, 19–26. [Google Scholar] [CrossRef]
- Rochefort, D.; Leech, D.; Bourbonnais, R. Electron transfer mediator systems for bleaching of paper pulp. Green Chem. 2004, 6, 14–24. [Google Scholar] [CrossRef]
- Cañas, A.I.; Camarero, S. Laccases and their natural mediators: Biotechnological tools for sustainable eco-friendly processes. Biotechnol. Adv. 2010, 28, 694–705. [Google Scholar] [CrossRef]
- Solomon, E.I.; Sundaram, U.M.; Machonkin, T.E. Multicopper oxidases and oxygenases. Chem. Rev. 1996, 96, 2563–2606. [Google Scholar] [CrossRef]
- Morozova, O.V.; Shumakovich, G.P.; Gorbacheva, M.A.; Shleev, S.V.; Yaropolov, A.I. “Blue” laccases. Biochemistry 2007, 72, 1136–1150. [Google Scholar]
- Yaropolov, A.I.; Skorobogat’ko, O.V.; Vartanov, S.S.; Varfolomeyev, S.D. Laccase. Appl. Biochem. Biotechnol. 1994, 49, 257–280. [Google Scholar]
- Higuchi, T. Lignin biochemistry: Biosynthesis and biodegradation. Wood Sci. Technol. 1990, 24, 23–63. [Google Scholar] [CrossRef]
- Kawai, S.; Umezawa, T.; Shimada, M.; Higuchi, T.; Koide, K.; Nishida, T.; Morohoshi, N.; Haraguchi, T. Cα-Cβ cleavage of phenolic β-1 lignin substructure model compound by laccase of Coriolus versicolor. Mokuzai Gakkaishi 1987, 33, 792–797. [Google Scholar]
- Sigoillot, C.; Lomascolo, A.; Record, E.; Robert, J.L.; Asther, M.; Sigoillot, J.-C. Lignocellulolytic and hemicellulolytic system of Pycnoporus cinnabarinus: Isolation and characterization of a cellobiose dehydrogenase and a new xylanase. Enzyme Microb. Technol. 2002, 31, 876–883. [Google Scholar] [CrossRef]
- Kawai, S.; Umezawa, T.; Higuchi, T. Oxidation of methoxylated benzyl alcohols by laccase of Coriolus versicolor in the presence of syringaldehyde. Wood Res. 1989, 76, 10–16. [Google Scholar]
- Bourbonnais, R.; Paice, M.G. Oxidation of non-phenolic substrates: An expanded role for laccase in lignin biodegradation. FEBS Lett. 1990, 267, 99–102. [Google Scholar] [CrossRef]
- Paice, M.G.; Bourbonnais, R.; Reid, I.; Archibald, F.S. Kraft pulp bleaching by redox enzymes. In Proceedings of International Symposium on Wood and Pulping Chemistry, Montreal, Canada, 9–12 June 1997; CPPA: Montreal, Canada, 1997; pp. PL1–1–PL1–4. [Google Scholar]
- Bourbonnais, R.; Paice, M.G. Demethylation and delignification of kraft pulp by Trametes versicolor laccase in the presence of 2,2′-azinobis-(3-ethylbenzthiazoline-6-sulphonate). Appl. Microbiol. Biotechnol. 1992, 36, 823–827. [Google Scholar]
- Bajpai, P.; Anand, A.; Bajpai, P.K. Bleaching with lignin-oxidizing enzymes. Biotechnol. Annu. Rev. 2006, 12, 349–378. [Google Scholar] [CrossRef]
- Call, H.P. Process for modifying, breaking down or bleaching lignin, materials containing lignin or like substances. WO/1994/029510, 22 December 1994. [Google Scholar]
- Call, H.P.; Mücke, I. History, overview and applications of mediated lignolytic systems, especially laccase-mediator-systems (Lignozym®-process). J. Biotechnol. 1997, 53, 163–202. [Google Scholar] [CrossRef]
- Bourbonnais, R.; Paice, M.; Freiermuth, B.; Bodie, E.; Borneman, S. Reactivities of various mediators and laccases with kraft pulp and lignin model compounds. Appl. Environ. Microbiol. 1997, 63, 4627–4632. [Google Scholar]
- Bendl, R.F.; Kandel, J.M.; Amodeo, K.D.; Ryder, A.M.; Woolridge, E.M. Characterization of the oxidative inactivation of xylanase by laccase and a redox mediator. Enzyme Microb. Technol. 2008, 43, 149–156. [Google Scholar] [CrossRef]
- Fabbrini, M.; Galli, C.; Gentili, P. Comparing the catalytic efficiency of some mediators of laccase. J. Mol. Catal. 2002, 16, 231–240. [Google Scholar]
- Barreca, A.M.; Fabbrini, M.; Galli, C.; Gentili, P.; Ljunggren, S. Laccase/mediated oxidation of a lignin model for improved delignification procedures. J. Mol. Catal. 2003, 26, 105–110. [Google Scholar]
- Cantarella, G.; Galli, C.; Gentili, P. Free radical versus electron-transfer routes of oxidation of hydrocarbons by laccase/mediator systems: Catalytic or stoichiometric procedures. J. Mol. Catal. 2003, 22, 135–144. [Google Scholar]
- Chakar, F.S.; Ragauskas, A.J. The effects of oxidative alkaline extraction stages after laccase HBT and laccase NHAA treatments—An NMR study of residual lignins. J. Wood Chem. Technol. 2000, 20, 169–184. [Google Scholar] [CrossRef]
- Camarero, S.; García, O.; Vidal, T.; Colom, J.; del Río, J.C.; Gutiérrez, A.; Gras, J.M.; Monje, R.; Martínez, M.J.; Martínez, Á.T. Efficient bleaching of non-wood high-quality paper pulp using laccase-mediator system. Enzyme Microb. Technol. 2004, 35, 113–120. [Google Scholar] [CrossRef] [Green Version]
- Garcia, O.; Camarero, S.; Colom, J.F.; Martínez, A.T.; Martínez, M.J.; Monje, R.; Vidal, T. Optimization of a laccase-mediator stage for TCF bleaching of flax pulp. Holzforschung 2003, 57, 513–519. [Google Scholar]
- Ibarra, D.; Camarero, S.; Romero, J.; Martínez, M.J.; Martínez, A.T. Integrating laccase–mediator treatment into an industrial-type sequence for totally chlorine-free bleaching of eucalypt kraft pulp. J. Chem. Technol. Biotechnol. 2006, 81, 1159–1165. [Google Scholar] [CrossRef]
- Poppius-Levlin, K.; Wang, W.; Tamminen, T.; Hortling, B.; Viikari, L.; Niku-Paavola, M.L. Effects of laccase/HBT treatment on pulp and lignin structures. J. Pulp Pap. Sci. 1999, 25, 90–94. [Google Scholar]
- Valls, C.; Roncero, M.B. Using both xylanase and laccase enzymes for pulp bleaching. Bioresour. Technol. 2009, 100, 2032–2039. [Google Scholar] [CrossRef]
- Moldes, D.; Vidal, T. Laccase-HBT bleaching of eucalyptus kraft pulp: Influence of the operating conditions. Bioresour. Technol. 2008, 99, 8565–8570. [Google Scholar] [CrossRef]
- Fillat, U.; Roncero, M.B. Effect of process parameters in laccase mediator system delignification of flax pulp. Part II. Impact on effluents properties. Chem. Eng. J. 2009, 152, 330–338. [Google Scholar] [CrossRef] [Green Version]
- Camarero, S.; Ibarra, D.; Martínez, Á.T.; Romero, J.; Gutiérrez, A.; del Río, J.C. Paper pulp delignification using laccase and natural mediators. Enzyme Microb. Technol. 2007, 40, 1264–1271. [Google Scholar] [CrossRef] [Green Version]
- Johannes, C.; Majcherczyk, A. Natural mediators in the oxidation of polycyclic aromatic hydrocarbons by laccase mediator systems. Appl. Environ. Microbiol. 2000, 66, 524–528. [Google Scholar] [CrossRef]
- Roncero, M.B.; Colom, J.F.; Vidal, T. Cellulose protection during ozone treatments of oxygen delignified Eucalyptus kraft pulp. Carbohydr. Polym. 2003, 51, 243–254. [Google Scholar] [CrossRef]
- Fillat, U.; Blanca Roncero, M. Effect of process parameters in laccase-mediator system delignification of flax pulp. Part I. Pulp properties. Chem. Eng. J. 2009, 152, 322–329. [Google Scholar] [CrossRef]
- Moldes, D.; Díaz, M.; Tzanov, T.; Vidal, T. Comparative study of the efficiency of synthetic and natural mediators in laccase-assisted bleaching of eucalyptus kraft pulp. Bioresour. Technol. 2008, 99, 7959–7965. [Google Scholar] [CrossRef]
- Shleev, S.; Persson, P.; Shumakovich, G.; Mazhugo, Y.; Yaropolov, A.; Ruzgas, T.; Gorton, L. Interaction of fungal laccases and laccase-mediator systems with lignin. Enzyme Microb. Technol. 2006, 39, 841–847. [Google Scholar] [CrossRef]
- Valls, C.; Colom, J.F.; Baffert, C.; Gimbert, I.; Roncero, M.B.; Sigoillot, J.-C. Comparing the efficiency of the laccase-NHA and laccase-HBT systems in eucalyptus pulp bleaching. Biochem. Eng. J. 2010, 49, 401–407. [Google Scholar] [CrossRef]
- Aracri, E.; Colom, J.F.; Vidal, T. Application of laccase-natural mediator systems to sisal pulp: An effective approach to biobleaching or functionalizing pulp fibres? Bioresour. Technol. 2009, 100, 5911–5916. [Google Scholar] [CrossRef]
- Camarero, S.; Ibarra, D.; Martinez, M.J.; Martinez, A.T. Lignin-derived compounds as efficient laccase mediators for decolorization of different types of recalcitrant dyes. Appl. Environ. Microbiol. 2005, 71, 1775–1784. [Google Scholar] [CrossRef]
- Eggert, C.; Temp, U.; Dean, J.F.D.; Eriksson, K.-E.L. A fungal metabolite mediates degradation of non-phenolic lignin structures and synthetic lignin by laccase. FEBS Lett. 1996, 391, 144–148. [Google Scholar] [CrossRef]
- Fillat, A.; Colom, J.F.; Vidal, T. A new approach to the biobleaching of flax pulp with laccase using natural mediators. Bioresour. Technol. 2010, 101, 4104–4110. [Google Scholar] [CrossRef]
- Amann, M. The Lignozym process coming closer to the mill. In Proceedings of International Symposium of Wood and Pulping Chemistry, Montreal, Canada, 9–12 June 1997; CPPA: Montreal, Canada, 1997; pp. F4–1–F4–5. [Google Scholar]
- Geng, X.; Li, K.; Xu, F. Investigation of hydroxamic acids as laccase-mediators for pulp bleaching. Appl. Microbiol. Biotechnol. 2004, 64, 493–496. [Google Scholar] [CrossRef]
- Xu, F.; Kulys, J.J.; Duke, K.; Li, K.; Krikstopaitis, K.; Deussen, H.-J.W.; Abbate, E.; Galinyte, V.; Schneider, P. Redox chemistry in laccase-catalyzed oxidation of N-hydroxy compounds. Appl. Environ. Microbiol. 2000, 66, 2052–2056. [Google Scholar] [CrossRef]
- Xu, F.; Shin, W.; Brown, S.H.; Wahleithner, J.A.; Sundaram, U.M.; Solomon, E.I. A study of a series of recombinant fungal laccases and bilirubin oxidase that exhibit significant differences in redox potential, substrate specificity, and stability. Biochim. Biophys. Acta 1996, 1292, 303–311. [Google Scholar] [CrossRef]
- Bourbonnais, R.; Paice, M.G. Enzymatic delignification of kraft pulp using laccase and a mediator. Tappi J. 1996, 79, 199–204. [Google Scholar]
- Baldrian, P. Fungal laccases—Occurrence and properties. FEMS Microbiol. Rev. 2006, 30, 215–242. [Google Scholar] [CrossRef]
- Fukushima, Y.; Kirk, T. Laccase component of the Ceriporiopsis subvermispora lignin-degrading system. Appl. Environ. Microbiol. 1995, 61, 872–876. [Google Scholar]
- Gold, M.H.; Glenn, J.K. Manganese peroxidase from Phanerochaete chrysosporium. Methods Enzymol. 1988, 161, 258–264. [Google Scholar]
- Cui, F.; Dolphin, D. The role of manganese in model systems related to lignin biodegradation. Holzforschung 1990, 44, 279–283. [Google Scholar] [CrossRef]
- Paice, M.G.; Bourbonnais, R.; Reid, I.D. Bleaching kraft pulps with oxidative enzymes and alkaline hydrogen peroxide. Tappi J. 1995, 78, 161–169. [Google Scholar]
- Katagiri, N.; Tsutsumi, Y.; Nishida, T. Correlation of brightening with cumulative enzyme activity related to lignin biodegradation during biobleaching of kraft pulp by white rot fungi in the solid-state fermentation system. Appl. Environ. Microbiol. 1995, 61, 617–622. [Google Scholar]
- Ni, Y.; Ju, Y.; Ohi, H. Further understanding of the manganese-induced decomposition of hydrogen peroxide. J. Pulp Pap. Sci. 2000, 26, 90–94. [Google Scholar]
- Leary, G.; Giampaolo, D. The darkening reactions of TMP and BTMP during alkaline peroxide bleaching. J. Pulp Pap. Sci. 1999, 25, 141–147. [Google Scholar]
- Glenn, J.K.; Gold, M.H. Purification and characterization of an extracellular Mn(II)-dependent peroxidase from the lignin-degrading basidiomycete, Phanerochaete chrysosporium. Arch. Biochem. Biophys. 1985, 242, 329–341. [Google Scholar] [CrossRef]
- Moreira, M.T.; Sierra-Alvarez, R.; Lema, J.M.; Feijoo, G.; Field, J.A. Oxidation of lignin in eucalyptus kraft pulp by manganese peroxidase from Bjerkandera sp. strain BOS55. Bioresour. Technol. 2001, 78, 71–79. [Google Scholar] [CrossRef]
- Bermek, H.; Li, K.; Eriksson, K.-E.L. Studies on mediators of manganese peroxidase for bleaching of wood pulps. Bioresour. Technol. 2002, 85, 249–252. [Google Scholar] [CrossRef]
- Gruber, K.; Klintschar, G.; Hayn, M.; Schlacher, A.; Steiner, W.; Kratky, C. Thermophilic xylanase from Thermomyces lanuginosus: High-resolution X-ray structure and modeling studies. Biochemistry 1998, 37, 13475–13485. [Google Scholar] [CrossRef]
- Bandivadekar, K.R.; Deshpande, V.V. Structure-function relationship of xylanase: Fluorimetric analysis of the tryptophan environment. Biochem. J. 1996, 315, 583–587. [Google Scholar]
- Jeffries, T.W. Conversion of xylose to ethanol under aerobic conditions by Candida tropicalis. Biotechnol. Lett. 1981, 3, 213–218. [Google Scholar] [CrossRef]
- Balakrishnan, H.; Satyanarayana, L.; Gaikwad, S.M.; Suresh, C.G. Structural and active site modification studies implicate Glu, Trp and Arg in the activity of xylanase from alkalophilic Bacillus sp. (NCL 87–6-10). Enzyme Microb. Technol. 2006, 39, 67–73. [Google Scholar] [CrossRef]
- Heidorne, F.O.; Magalhães, P.O.; Ferraz, A.L.; Milagres, A.M.F. Characterization of hemicellulases and cellulases produced by Ceriporiopsis subvermispora grown on wood under biopulping conditions. Enzyme Microb. Technol. 2006, 38, 436–442. [Google Scholar] [CrossRef]
- Vieira, D.S.; Degrève, L.; Ward, R.J. Characterization of temperature dependent and substrate-binding cleft movements in Bacillus circulans family 11 xylanase: A molecular dynamics investigation. Biochim. Biophys. Acta 2009, 1790, 1301–1306. [Google Scholar] [CrossRef]
- Viikari, L.; Ranua, M.; Kantelinen, A.; Sundquist, J.; Linko, M. Bleaching with enzymes. In Proceedings of the Third International Conference on Biotechnology in the Pulp and Paper Industry, Stockholm, 16–19 June 1986; STFI: Stockholm, Sweden, 1986; p. 67. [Google Scholar]
- Scott, B.P.; Young, F.; Paice, M.G. Mill-scale enzyme treatment of a softwood kraft pulp prior to bleaching: Brightness was maintained while reducing the active chlorine multiple. Pulp Pap. Can. 1993, 94, 57–61. [Google Scholar]
- Kantelinen, A.; Hortling, B.; Sundquist, J.; Linko, M.; Viikari, L. Proposed mechanism of the enzymatic bleaching of kraft pulp with xylanases. Holzforschung 1993, 47, 318–324. [Google Scholar] [CrossRef]
- Viikari, L.; Suurnäkki, A.; Grönqvist, S.; Raaska, L.; Ragauskas, A. Forest Products: Biotechnology in Pulp and Paper Processing. In Encyclopedia of Microbiology; Moselio, S., Ed.; Academic Press: Oxford, UK, 2009; pp. 80–94. [Google Scholar]
- Viikari, L.; Kantelinen, A.; Sundquist, J.; Linko, M. Xylanases in bleaching: From an idea to the industry. FEMS Microbiol. Rev. 1994, 13, 335–350. [Google Scholar] [CrossRef]
- Henrissat, B. A classification of glycosyl hydrolases based on amino acid sequence similarities. Biochem. J. 1991, 280, 309–316. [Google Scholar]
- Davies, G.; Henrissat, B. Structures and mechanisms of glycosyl hydrolases. Structure 1995, 3, 853–859. [Google Scholar] [CrossRef]
- Wong, K.K.; Tan, L.U.; Saddler, J.N. Multiplicity of beta-1,4-xylanase in microorganisms: Functions and applications. Microbiol. Rev. 1988, 52, 305–317. [Google Scholar]
- Georis, J.; Esteves, F.D.L.; Lamotte-Brasseur, J.; Bougnet, V.; Giannotta, F.; Frère, J.-M.; Devreese, B.; Granier, B. An additional aromatic interaction improves the thermostability and thermophilicity of a mesophilic family 11 xylanase: Structural basis and molecular study. Protein Sci. 2000, 9, 466–475. [Google Scholar]
- Collins, T.; Meuwis, M.-A.; Stals, I.; Claeyssens, M.; Feller, G.; Gerday, C. A novel family 8 xylanase, functional and physicochemical characterization. J. Biol. Chem. 2002, 277, 35133–35139. [Google Scholar] [CrossRef]
- Collins, T.; Gerday, C.; Feller, G. Xylanases, xylanase families and extremophilic xylanases. FEMS Microbiol. Rev. 2005, 29, 3–23. [Google Scholar] [CrossRef]
- McAllister, K.A.; Marrone, L.; Clarke, A.J. The role of tryptophan residues in substrate binding to catalytic domains A and B of xylanase C from Fibrobacter succinogenes S85. Biochim. Biophys. Acta 2000, 1480, 342–352. [Google Scholar] [CrossRef]
- Maio, S.; Ziser, L.; Aebersold, R.; Withers, S.G. Identification of glutamic acid 78 as the active site nucleophile in Bacillus subtilis xylanase using electrospray tandem mass spectrometry. Biochemistry 1994, 33, 7027–7032. [Google Scholar] [CrossRef]
- Wakarchuk, W.W.; Sung, W.L.; Campbell, R.L.; Cunningham, A.; Watson, D.C.; Yaguchi, M. Thermostabilization of the Bacillus circulans xylanase by the introduction of disulfide bonds. Protein Eng. 1994, 7, 1379–1386. [Google Scholar] [CrossRef]
- Wakarchuk, W.W.; Campbell, R.L.; Sung, W.L.; Davoodi, J.; Yaguchi, M. Mutational and crystallographic analysis of the active site residues of the Bacillus circulans xylanase. Protein Sci. 1994, 3, 467–475. [Google Scholar]
- Sabini, E.; Wilson, K.S.; Danielson, S.; Schülein, M.; Davies, G.J. Oligosaccharide binding to family 11 xylanases: Both covalent intermediate and mutant product complexes display 2,5B conformations at the active centre. Acta Crystallogr. D 2001, 57, 1344–1347. [Google Scholar] [CrossRef]
- Törrönen, A.; Harkki, A.; Rouvinen, J. Three-dimensional structure of endo-1,4-β-xylanase II from Trichoderma reesei: Two conformational states in the active site. EMBO J. 1994, 13, 2493–2501. [Google Scholar]
- Havukainen, R.; Torronen, A.; Laitinen, T.; Rouvinen, J. Covalent binding of three epoxyalkyl xylosides to the active site of endo-1,4-xylanase II from Trichoderma reesei. Biochemistry 1996, 35, 9617–9624. [Google Scholar] [CrossRef]
- Sabini, E.; Sulzenbacher, G.; Dauter, M.; Dauter, Z.; Jørgensen, P.L.; Schülein, M.; Dupont, C.; Davies, G.J.; Wilson, K.S. Catalysis and specificity in enzymatic glycoside hydrolysis: A 2,5B conformation for the glycosyl-enzyme intermediate revealed by the structure of the Bacillus agaradhaerens family 11 xylanase. Chem. Biol. 1999, 6, 483–492. [Google Scholar] [CrossRef]
- Dutta, T.; Sahoo, R.; Sinha Ray, S.; Bhattacharjee, A.; Sengupta, R.; Ghosh, S. Probing the active site environment of alkaliphilic family 11 xylanase from Penicillium citrinum: Evidence of essential histidine residue at the active site. Enzyme Microb. Technol. 2007, 41, 440–446. [Google Scholar] [CrossRef]
- Wouters, J.; Gerois, J.; Engher, D.; Vandenhaute, J.; Dusart, J.; Frere, J.M.; Depiereux, E.; Charlier, P. Crystallographic analysis of family 11 endo-β-1,4-xylanase Xyl1 from Streptomyces sp. S38. Acta Crystallogr. D 2001, 57, 1813–1819. [Google Scholar] [CrossRef]
- Krengel, U.B.; Dijkstra, W. Three dimensional analysis of endo-1,4-β-xylanase I from Aspergillus niger: Molecular basis for its low pH optimum. J. Mol. Biol. 1996, 263, 70–78. [Google Scholar] [CrossRef]
- Törrönen, A.; Rovinen, J. Structural comparison of two major endo-1,4-β-xylanases from Trichoderma reesei. Biochemistry 1995, 34, 847–856. [Google Scholar] [CrossRef]
- Withers, S.G.; Aebersold, R.R. Approaches to labeling and identification of active site residues in glycosidases. Protein Sci. 1995, 4, 361–372. [Google Scholar] [CrossRef]
- Juy, M.; Amrt, A.G.; Alzari, P.M.; Poljak, R.J.; Claeyssens, M.; Béguin, P.; Aubert, J.-P. Three-dimensional structure of a thermostable bacterial cellulase. Nature 1992, 357, 89–91. [Google Scholar] [CrossRef]
- Vyas, N.K. Atomic features of protein-carbohydrate interactions. Curr. Opin. Struct. Biol. 1991, 1, 732–740. [Google Scholar] [CrossRef]
- Sierks, M.R.; Ford, C.; Reilly, P.J.; Sevensson, B. Catalytic mechanism of fungal glucoamylase as defined by mutagenesis of Asp176, Glu179 and Glu180 in the enzyme from Aspergillus awamori. Protein Eng. 1990, 3, 193–198. [Google Scholar] [CrossRef]
- Clarke, A.J. Essential tryptophan residues in the function of cellulase from Schizophyllum commune. Biochim. Biophys. Acta 1987, 912, 424–431. [Google Scholar] [CrossRef]
- Polizeli, M.L.; Rizzatti, A.C.; Monti, R.; Terenzi, H.F.; Jorge, J.A.; Amorim, D.S. Xylanases from fungi: Properties and industrial applications. Appl. Microbiol. Biotechnol. 2005, 67, 577–591. [Google Scholar] [CrossRef]
- Larkin, M.A.; Blackshields, G.; Brown, N.P.; Chenna, R.; McGettigan, P.A.; McWilliam, H.; Valentin, F.; Wallace, I.M.; Wilm, A.; Lopez, R.; et al. Clustal W and Clustal X version 2.0. Bioinformatics 2007, 23, 2947–2948. [Google Scholar] [CrossRef]
- Vandermarliere, E.; Bourgois, T.M.; Rombouts, S.; van Campenhout, S.; Volckaert, G.; Strelkov, S.V.; Delcour, J.A.; Rabijns, A.; Courtin, C.M. Crystallographic analysis shows substrate binding at the −3 to +1 active-site subsites and at the surface of glycoside hydrolase family 11 endo-1,4-β-xylanases. Biochem. J. 2008, 410, 71–79. [Google Scholar] [CrossRef]
- Marrone, L.; McAllister, K.A.; Clarke, A.J. Characterization of function and activity of domains A, B and C of xylanase C from Fibrobacter succinogenes S85. Protein Eng. 2000, 13, 593–601. [Google Scholar] [CrossRef]
- Paradis, F.W.; Zhu, H.; Krell, P.J.; Phillips, J.P.; Forsberg, C.W. The xynC gene from Fibrobacter succinogenes S85 codes for a xylanase with two similar catalytic domains. J. Bacteriol. 1993, 175, 7666–7672. [Google Scholar]
- Tanaka, H.; Nakamura, T.; Hayashi, S.; Ohta, K. Purification and properties of an extracellular endo-1,4-beta-xylanase from Penicillium citrinum and characterization of the encoding gene. J. Biosci. Bioeng. 2005, 100, 623–630. [Google Scholar] [CrossRef]
- Schlacher, A.; Holzmann, K.; Hayn, M.; Steiner, W.; Schwab, H. Cloning and characterization of the gene for the thermostable xylanase XynA from Thermomyces lanuginosus. J. Biotechnol. 1996, 49, 211–218. [Google Scholar] [CrossRef]
- He, J.; Yu, B.; Zhang, K.; Ding, X.; Chen, D. Expression of a Trichoderma reesei β-xylanase gene in Escherichia coli and activity of the enzyme on fiber-bound substrates. Protein Expression Purif. 2009, 67, 1–6. [Google Scholar] [CrossRef]
- Deshpande, V.; Hinge, J.; Rao, M. Chemical modification of xylanases: Evidence for essential tryptophan and cysteine residues at the active site. Biochim. Biophys. Acta 1990, 1041, 172–177. [Google Scholar] [CrossRef]
- Zhu, H.; Paradis, F.W.; Krell, P.J.; Phillips, J.P.; Forsberg, C.W. Enzymatic specificities and modes of action of the two catalytic domains of the XynC xylanase from Fibrobacter succinogenes S85. J. Bacteriol. 1994, 176, 3885–3894. [Google Scholar]
- Bray, M.R.; Clarke, A.J. Identification of an essential tyrosyl residue in the binding site of Schizophyllum commune xylanase A. Biochemistry 1995, 34, 2006–2014. [Google Scholar] [CrossRef]
- Keskar, S.S.; Srinivasan, M.C.; Deshpande, V.V. Chemical modification of a xylanase from thermotolerant Streptomyces. Biochem. J. 1989, 261, 49–55. [Google Scholar]
- Blake, C.C.F.; Cassels, R.; Dobson, C.M.; Poulsen, F.M.; Williams, R.J.P.; Wilson, K.S. Structure and binding properties of hen lysozyme modified at tryptophan 62. J. Mol. Biol. 1981, 147, 73–95. [Google Scholar] [CrossRef]
- Spande, T.F.; Witkop, B. Determination of the tryptophan content of proteins with N-bromosuccinimide. Methods Enzymol. 1967, 11, 498–506. [Google Scholar]
- Witkop, B. Nonenzymatic methods for the preferential and selective cleavage and modification of proteins. Adv. Protein Chem. 1962, 16, 221–321. [Google Scholar] [CrossRef]
- Bray, M.R.; Carriere, A.D.; Clarke, A.J. Quantitation of tryptophan and tyrosine residues in proteins by fourth-derivative spectroscopy. Anal. Biochem. 1994, 221, 278–284. [Google Scholar] [CrossRef]
- Poon, D.K.Y.; Webster, P.; Withers, S.G.; McIntosh, L.P. Characterizing the pH-dependent stability and catalytic mechanism of the family 11 xylanase from the alkalophilic Bacillus agaradhaerens. Carbohydr. Res. 2003, 338, 415–421. [Google Scholar] [CrossRef]
- Biswas, S.R.; Jana, S.C.; Mishra, A.K.; Nanda, G. Production, purification and characterization of xylanase from hyperxylanolytic mutant of Aspergillus ochraceus. Biotechnol. Bioeng. 1990, 35, 244–251. [Google Scholar] [CrossRef]
- Kubacková, M.; Karácsonyi, S.; Bilisics, L.; Toman, R. Some properties of an endo-1,4-beta-d-xylanase from the ligniperdous fungus Trametes hirsuta. Folia Microbiol. 1978, 23, 202–209. [Google Scholar] [CrossRef]
- Nath, D.; Rao, M. Structural and functional role of tryptophan in xylanase from an extremophilic Bacillus: Assessment of the active site. Biochem. Biophys Res. Commun. 1998, 249, 207–212. [Google Scholar] [CrossRef]
- Hakulinen, N.; Turunen, O.; Jänis, J.; Leisola, M.; Rouvinen, J. Three-dimensional structures of thermophilic β-1,4-xylanases from Chaetomium thermophilum and Nonomuraea flexuosa. Eur. J. Biochem. 2003, 270, 1399–1412. [Google Scholar] [CrossRef]
- Senior, D.J.; Mayers, P.R.; Saddler, J.N. The interaction of xylanases with commercial pulps. Biotechnol. Bioeng. 1991, 37, 274–279. [Google Scholar] [CrossRef]
- Tatsumoto, K.; Baker, J.O.; Tucker, M.P.; Oh, K.K.; Mohagheghi, A.; Grohmann, K.; Himmel, M.E. Digestion of pretreated aspen substrates: Hydrolysis rates and adsorptive loss of cellulase enzymes. Appl. Biochem. Biotechnol. 1988, 18, 159–174. [Google Scholar] [CrossRef]
- Montgomery, L.; Flesher, B.; Stahl, D. Transfer of Bacteroides succinogenes (Hungate) to Fibrobacter gen. nov. as Fibrobacter succinogenes comb. nov. and description of Fibrobacter intestinalis sp. nov. Int. J. Syst Bacteriol 1988, 38, 430–435. [Google Scholar] [CrossRef]
- Várnai, A.; Viikari, L.; Marjamaa, K.; Siika-aho, M. Adsorption of monocomponent enzymes in enzyme mixture analyzed quantitatively during hydrolysis of lignocellulose substrates. Bioresour. Technol. 2011, 102, 1220–1227. [Google Scholar] [CrossRef]
- Kyriacou, A.; Neufeld, R.J.; MacKenzie, C.R. Effect of physical parameters on the adsorption characteristics of fractionated Trichoderma reesei cellulase components. Enzyme Microb. Technol. 1988, 10, 675–681. [Google Scholar] [CrossRef]
- Bernier, R.L., Jr.; Gray, A.L.; Moser, G.P.; Hamilton, J.; Roberge, M.; Senior, D.J. Fate of residual xylanase after treatment and bleaching of softwood kraft pulp. Bioresour. Technol. 1994, 50, 79–83. [Google Scholar] [CrossRef]
- Ferraz, A.; Guerra, A.; Mendonça, R.; Masarin, F.; Vicentim, M.P.; Aguiar, A.; Pavan, P.C. Technological advances and mechanistic basis for fungal biopulping. Enzyme Microb. Technol. 2008, 43, 178–185. [Google Scholar] [CrossRef]
- de Souza-Cruz, P.B.; Freer, J.; Siika-Aho, M.; Ferraz, A. Extraction and determination of enzymes produced by Ceriporiopsis subvermispora during biopulping of Pinus taeda wood chips. Enzyme Microb. Technol. 2004, 34, 228–234. [Google Scholar] [CrossRef]
- Machuca, A.; Durán, N. Phenol oxidases production and wood degradation by a thermophilic fungus Thermoascus aurantiacus. Appl. Biochem. Biotechnol. 1993, 43, 37–44. [Google Scholar] [CrossRef]
- Guerra, A.; Mendonça, R.; Ferraz, A. Molecular weight distribution of wood components extracted from Pinus taeda biotreated by Ceriporiopsis subvermispora. Enzyme Microb. Technol 2003, 33, 12–18. [Google Scholar] [CrossRef]
- Aguiar, A.; Souza-Cruz, P.B.D.; Ferraz, A. Oxalic acid, Fe3+-reduction activity and oxidative enzymes detected in culture extracts recovered from Pinus taeda wood chips biotreated by Ceriporiopsis subvermispora. Enzyme Microb. Technol. 2006, 38, 873–878. [Google Scholar] [CrossRef]
- Blanchette, R.A.; Krueger, E.W.; Haight, J.E.; Masood, A.; Akin, D.E. Cell wall alterations in loblolly pine wood decayed by the white-rot fungus, Ceriporiopsis subvermispora. J. Biotechnol. 1997, 53, 203–213. [Google Scholar] [CrossRef]
- Ferraz, A.; Córdova, A.M.; Machuca, A. Wood biodegradation and enzyme production by Ceriporiopsis subvermispora during solid-state fermentation of Eucalyptus grandis. Enzyme Microb. Technol. 2003, 32, 59–65. [Google Scholar] [CrossRef]
- Watanabe, T.; Koller, K.; Messner, K. Copper-dependent depolymerization of lignin in the presence of fungal metabolite, pyridine. J. Biotechnol. 1998, 62, 221–230. [Google Scholar] [CrossRef]
- Enoki, M.; Watanabe, T.; Nakagame, S.; Koller, K.; Messner, K.; Honda, Y.; Kuwahara, M. Extracellular lipid peroxidation of selective white-rot fungus, Ceriporiopsis subvermispora. FEMS Microbiol. Lett. 1999, 180, 205–211. [Google Scholar] [CrossRef]
- Sethuraman, A.; Akin, D.E.; Eriksson, K.-E.L. Plant-cell-wall-degrading enzymes produced by the white-rot fungus Ceriporiopsis subvermispora. Biotechnol. Appl. Biochem. 1998, 27, 37–47. [Google Scholar]
- Srebotnik, E.; Jensen, K.A.; Kawai, S.; Hammel, K. Evidence that Ceriporiopsis subvermispora degrades nonphenolic lignin structures by a one-electron-oxidation mechanism. Appl. Environ. Microbiol. 1997, 63, 4435–4440. [Google Scholar]
- Bao, W.; Fukushima, Y.; Jensen, K.A., Jr.; Moen, M.A.; Hammel, K.E. Oxidative degradation of non-phenolic lignin during lipid peroxidation by fungal manganese peroxidase. FEBS Lett. 1994, 354, 297–300. [Google Scholar] [CrossRef]
- Kapich, A.N.; Jensen, K.A.; Hammel, K.E. Peroxyl radicals are potential agents of lignin biodegradation. FEBS Lett. 1999, 461, 115–119. [Google Scholar] [CrossRef]
- Jensen, K.A.; Bao, W.; Kawai, S.; Srebotnik, E.; Hammel, K. Manganese-dependent cleavage of nonphenolic lignin structures by Ceriporiopsis subvermispora in the absence of lignin peroxidase. Appl. Environ. Microbiol. 1996, 62, 3679–3686. [Google Scholar]
- Vicentim, M.P.; Ferraz, A. Enzyme production and chemical alterations of Eucalyptus grandis wood during biodegradation by Ceriporiopsis subvermispora in cultures supplemented with Mn2+, corn steep liquor and glucose. Enzyme Microb. Technol. 2007, 40, 645–652. [Google Scholar] [CrossRef]
- Machuca, A.; Ferraz, A. Hydrolytic and oxidative enzymes produced by white- and brown-rot fungi during Eucalyptus grandis decay in solid medium. Enzyme Microb. Technol. 2001, 29, 386–391. [Google Scholar] [CrossRef]
- Addleman, K.; Dumonceaux, T.; Paice, M.G.; Bourbonnais, R.; Archibald, F.S. Production and characterization of Trametes versicolor mutants unable to bleach hardwood kraft pulp. Appl. Environ. Microbiol. 1995, 61, 3687–3694. [Google Scholar]
- Torres, A.L.; Roncero, M.B.; Colom, J.F.; Pastor, F.I.J.; Blanco, A.; Vidal, T. Effect of a novel enzyme on fibre morphology during ECF bleaching of oxygen delignified Eucalyptus kraft pulps. Bioresour. Technol. 2000, 74, 135–140. [Google Scholar] [CrossRef]
- Roncero, M.B.; Torres, A.L.; Colom, J.F.; Vidal, T. The effect of xylanase on lignocellulosic components during the bleaching of wood pulps. Bioresour. Technol. 2005, 96, 21–30. [Google Scholar]
- Roncero, M.B.; Torres, A.L.; Colom, J.F.; Vidal, T. Effect of xylanase on ozone bleaching kinetics and properties of Eucalyptus kraft pulp. J. Chem. Technol. Biotechnol. 2003, 78, 1023–1031. [Google Scholar] [CrossRef]
- Kantelinen, A.; Hortling, B.; Ranua, M.; Viikari, L. Effects of fungal and enzymatic treatments on isolated lignins and on pulp bleachability. Holzforschung 1993, 47, 29–35. [Google Scholar] [CrossRef]
- Valls, C.; Vidal, T.; Roncero, M.B. Boosting the effect of a laccase-mediator system by using a xylanase stage in pulp bleaching. J. Hazard. Mater. 2010, 177, 586–592. [Google Scholar] [CrossRef]
- Chakar, F.S.; Ragauskas, A.J. Formation of quinonoid structures in laccase-mediator reactions. In Oxidative Delignification Chemistry; American Chemical Society: Washington, DC, USA, 2001; Volume 785, pp. 444–455. [Google Scholar]
- Ibarra, D.; Romero, J.; Martínez, M.J.; Martínez, A.T.; Camarero, S. Exploring the enzymatic parameters for optimal delignification of eucalypt pulp by laccase-mediator. Enzyme Microb. Technol. 2006, 39, 1319–1327. [Google Scholar]
- Aracri, E.; Vidal, T. Xylanase- and laccase-aided hexenuronic acids and lignin removal from specialty sisal fibres. Carbohydr. Polym. 2011, 83, 1355–1362. [Google Scholar] [CrossRef]
- Valls, C.; Vidal, T.; Roncero, M.B. The role of xylanases and laccases on hexenuronic acid and lignin removal. Process. Biochem. 2010, 45, 425–430. [Google Scholar] [CrossRef]
- Cadena, E.M.; Vidal, T.; Torres, A.L. Can the laccase mediator system affect the chemical and refining properties of the eucalyptus pulp? Bioresour. Technol. 2010, 101, 8199–8204. [Google Scholar] [CrossRef]
- Xu, Q.; Fu, Y.; Gao, Y.; Qin, M. Performance and efficiency of old newspaper deinking by combining cellulase/hemicellulase with laccase-violuric acid system. Waste Manag. 2009, 29, 1486–1490. [Google Scholar] [CrossRef]
- Chandra, R.P.; Ragauskas, A.J. Modification of high-lignin kraft pulps with laccase. Part 2. Xylanase-enhanced strength benefits. Biotechnol. Prog. 2005, 21, 1302–1306. [Google Scholar] [CrossRef]
- Pfaller, R.; Amann, M.; Freudenreich, J. Analysis of laccase and mediator interactions in the laccase mediator system. In Proceedings of 7th International Conference on Biotechnology in the Pulp and Paper Industry, Vancouver, Canada, 16–19 June 1998; CPPA: Vancouver, Canada, 1998; pp. A99–A102. [Google Scholar]
- Bajpai, P.; Anand, A.; Sharma, N.; Mishra, S.P.; Bajpai, P.K.; Lachenal, D. Enzymes improve ECF bleaching of pulp. BioResources 2006, 1, 34–44. [Google Scholar]
- Freixo, M.D.R.; Karmali, A.; Frazão, C.; Arteiro, J.M. Production of laccase and xylanase from Coriolus versicolor grown on tomato pomace and their chromatographic behaviour on immobilized metal chelates. Process. Biochem. 2008, 43, 1265–1274. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Woolridge, E.M. Mixed Enzyme Systems for Delignification of Lignocellulosic Biomass. Catalysts 2014, 4, 1-35. https://doi.org/10.3390/catal4010001
Woolridge EM. Mixed Enzyme Systems for Delignification of Lignocellulosic Biomass. Catalysts. 2014; 4(1):1-35. https://doi.org/10.3390/catal4010001
Chicago/Turabian StyleWoolridge, Elisa M. 2014. "Mixed Enzyme Systems for Delignification of Lignocellulosic Biomass" Catalysts 4, no. 1: 1-35. https://doi.org/10.3390/catal4010001
APA StyleWoolridge, E. M. (2014). Mixed Enzyme Systems for Delignification of Lignocellulosic Biomass. Catalysts, 4(1), 1-35. https://doi.org/10.3390/catal4010001




