Hydrogen Evolution from Napiergrass by the Combination of Biological Treatment and a Pt-Loaded TiO2-Photocatalytic Reaction
Abstract
:1. Introduction
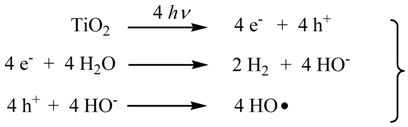
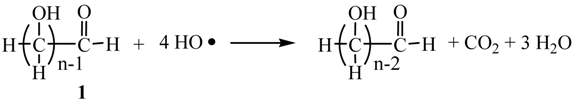
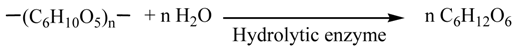
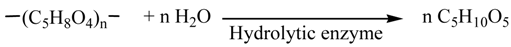
2. Results and Discussion
2.1. Alkali-Treatment of Napiergrass
2.2. Biological Treatment of Holocellulose
| Biological treatment | PC-treatment d | ∆H/kJ g | ||
|---|---|---|---|---|
| Method b | Product (yields/g) c | [N] e (yield/g) f | ||
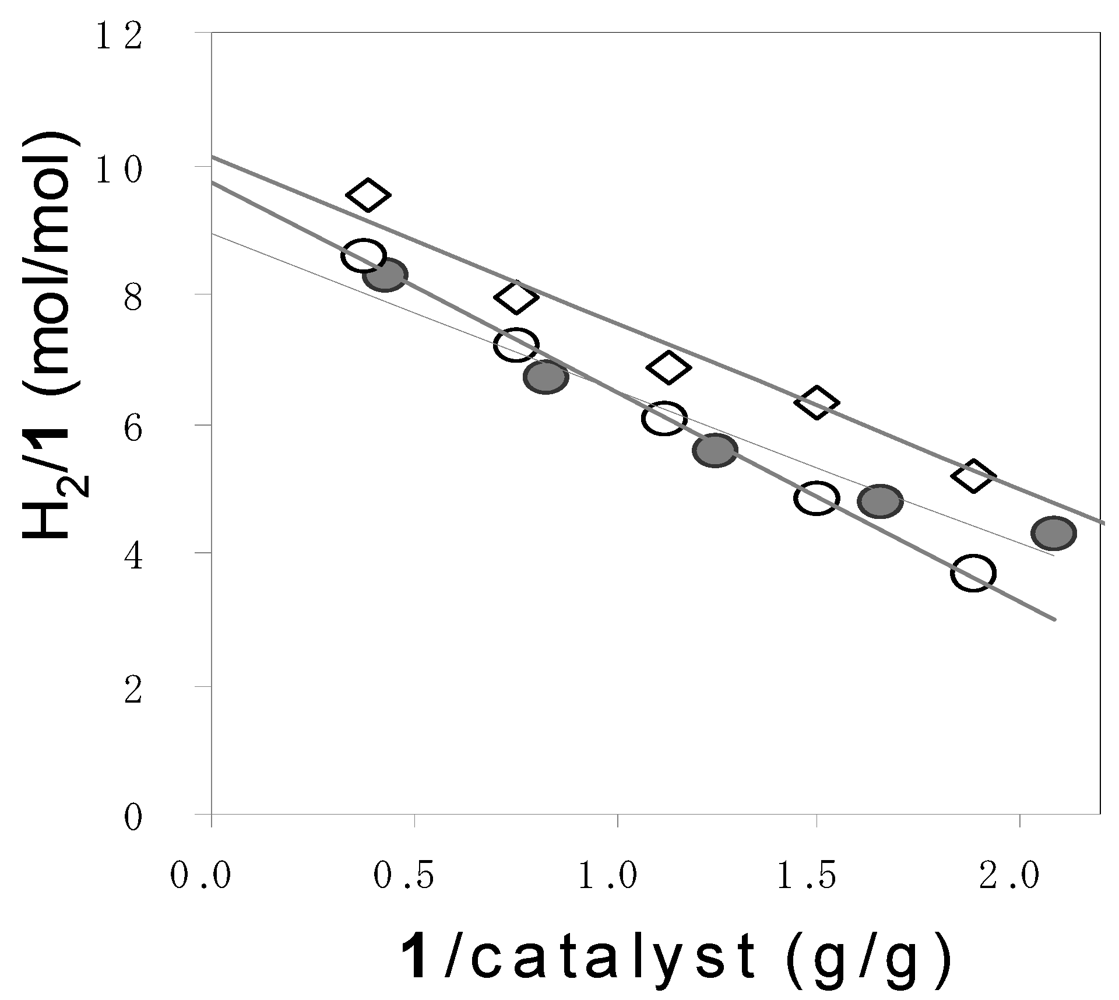
| A (SA) | Pentose (13.6) → | H2 [8.7] (4.23) | 603 | |
| Hexose (27.4) → | ||||
| B (SA/FE) | Pentose (12.0) | Pentose (12.1) → | H2 [9.7] (1.66) | 555 |
| Hexose (27.2) → | Hexose (0.9) → | |||
| EtOH (10.7) | ||||
| C (SSF) | Pentose (12.8) → | H2 [10.2] (1.74) | 615 | |
| Ethanol (12.3) | ||||
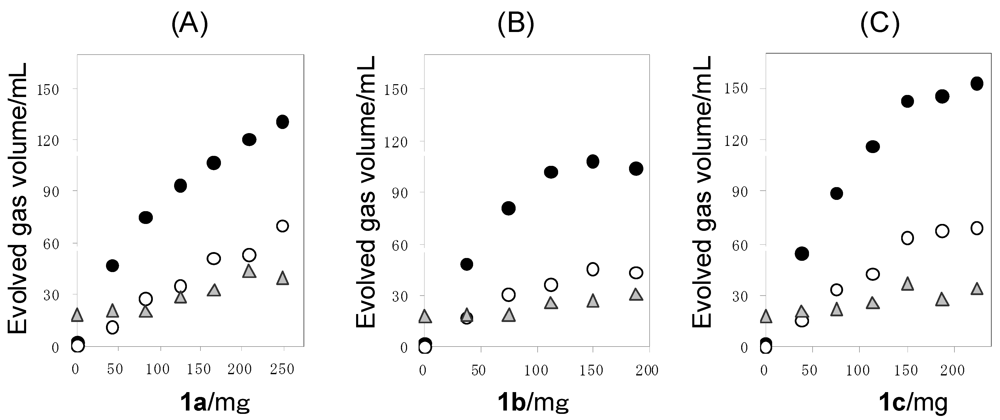
2.3. Photocatalytic Hydrogen Evolution (PC) Using the Saccharide (1) as Sacrificial Agent
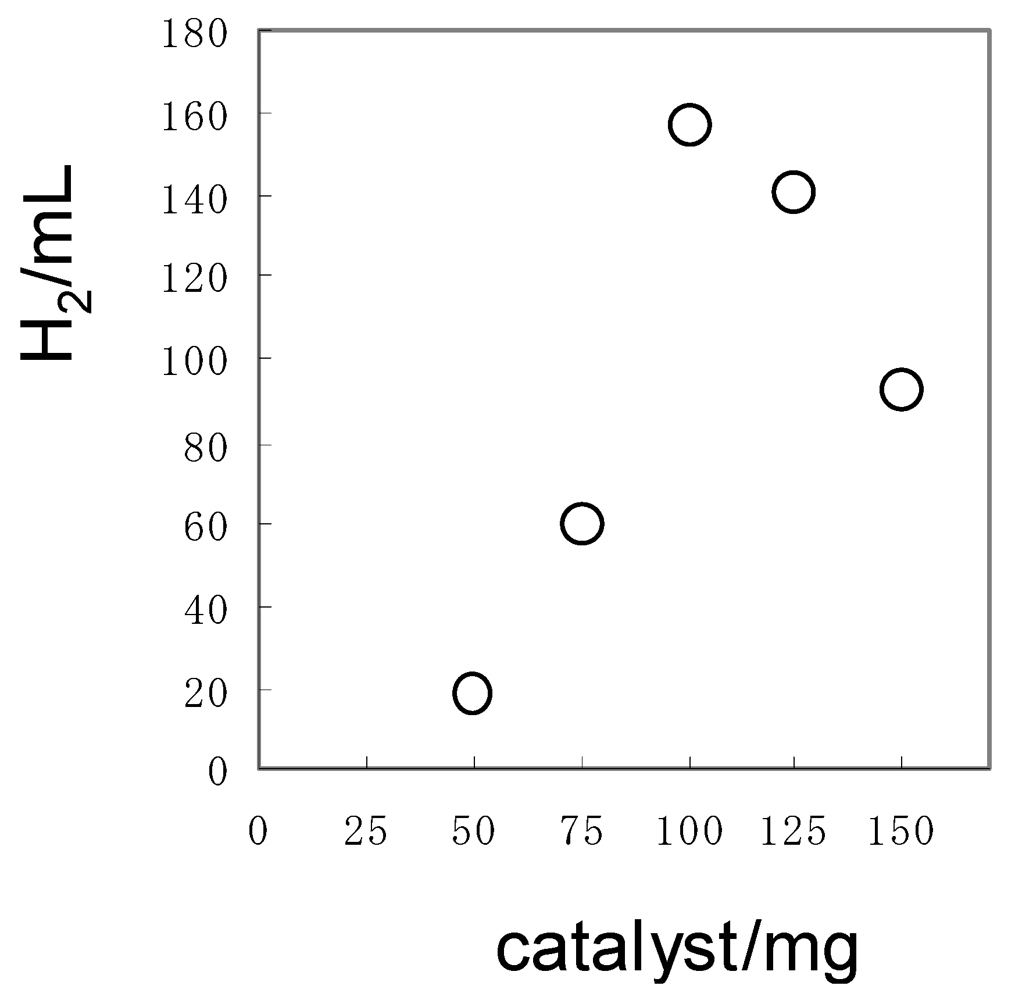
| 1/mg b | T/h c | Gas volume/mL | Molar ratio | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Total d | H2 | CO2 | O2 | N2e | H2/1 | H2/CO2 | |||
| 0 | 5 | 13 | 2 | 0 | 18 | −7 | - | - | |
| 1a | 42 | 10 | 72 | 47 | 11 | 21 | −7 | 8.4 | 4.3 |
| 1a | 83 | 21 | 110 | 75 | 27 | 20 | −12 | 6.7 | 2.8 |
| 1a | 125 | 30 | 158 | 94 | 35 | 29 | 0 | 5.6 | 2.7 |
| 1a | 166 | 49 | 184 | 107 | 51 | 33 | −7 | 4.8 | 2.1 |
| 1a | 208 | 53 | 215 | 120 | 53 | 44 | −2 | 4.3 | 2.3 |
| 1a | 249 | 74 | 240 | 131 | 70 | 40 | −1 | 3.9 | 1.9 |
| 1b | 38 | 17 | 80 | 48 | 17 | 19 | −23 | 8.6 | 2.9 |
| 1b | 75 | 25 | 117 | 81 | 31 | 19 | −13 | 7.2 | 2.6 |
| 1b | 113 | 31 | 157 | 102 | 36 | 26 | −7 | 6.1 | 2.8 |
| 1b | 150 | 45 | 160 | 108 | 45 | 27 | −20 | 4.8 | 2.4 |
| 1b | 188 | 53 | 161 | 104 | 43 | 31 | −17 | 3.7 | 2.4 |
| 1c | 38 | 14 | 78 | 54 | 15 | 21 | −12 | 9.6 | 3.6 |
| 1c | 75 | 26 | 134 | 89 | 33 | 22 | −10 | 8.0 | 2.7 |
| 1c | 113 | 29 | 174 | 116 | 42 | 26 | −10 | 6.9 | 2.8 |
| 1c | 150 | 40 | 225 | 142 | 63 | 20 | 0 | 6.3 | 2.3 |
| 1c | 188 | 53 | 225 | 145 | 67 | 28 | −15 | 5.2 | 2.2 |
| 1c | 225 | 43 | 230 | 153 | 69 | 34 | −26 | 4.6 | 2.2 |
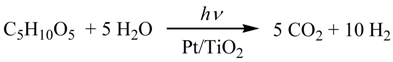
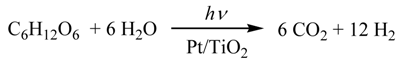
2.4. The Combustion Energy of the Products
3. Experimental Section
3.1. Preparation of the Photocatalyst
3.2. Analysis
3.3. Alkali-Treatment of Napiergrass
3.4. Saccharification of Hollocellulose with Enzyme (SA)
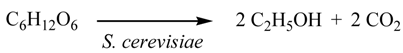
3.5. Fermentation of the Saccharide (1) with S. cerevisiae
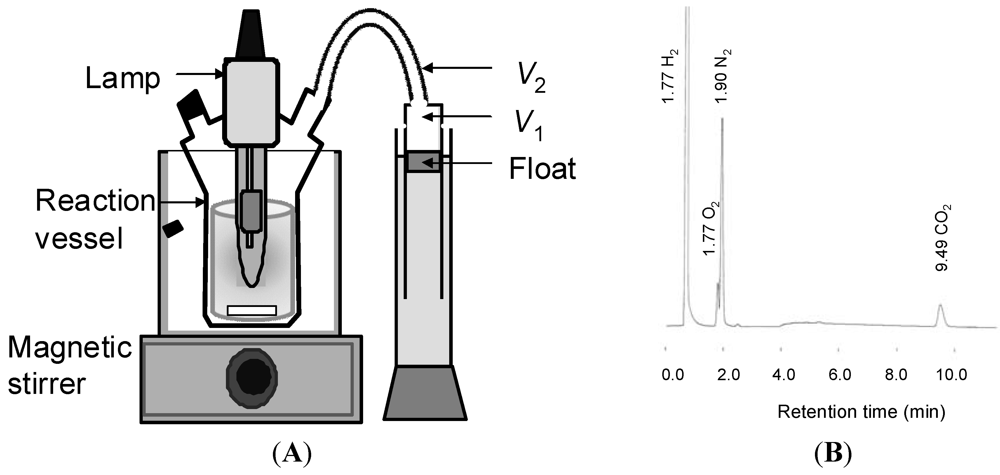
3.6. Photocatalytic Reaction
4. Conclusions

References
- Ward, O.P.; Singh, A. Bioethanol technology: Development and perspectives. Adv. Appl. Microbiol. 2002, 51, 53–80. [Google Scholar] [CrossRef]
- Balat, M.; Balat, H. Recent trends in global production and utilization of bio-ethanol fuel. Appl. Energy 2009, 86, 2273–2282. [Google Scholar] [CrossRef]
- Ishii, Y.; Mukhtar, M.; Idota, S.; Fukuyama, K. Rotational grazing system for beef cows on dwarf napiergrass pasture oversown with Italian ryegrass for 2 years after establishment. Grassl. Sci. 2005, 51, 209–220. [Google Scholar]
- Yasuda, M.; Miura, A.; Yuki, R.; Nakamura, Y.; Shiragami, T.; Ishii, Y.; Yokoi, H. The effect of TiO2-photocatalytic pretreatment on the biological production of ethanol from lignocelluloses. J. Photochem. Photobiol. A 2011, 220, 195–199. [Google Scholar] [CrossRef]
- Shiragami, T.; Tomo, T.; Tsumagari, H.; Yuki, R.; Yamashita, T.; Yasuda, M. Pentose acting as a sacrificial multi-electron source in photocatalytic hydrogen evolution from water by Pt-doped TiO2. Chem. Lett. 2012, in press.. [Google Scholar]
- Kawai, T.; Sakata, T. Conversion of carbohydrate into hydrogen fuel by a photocatalytic process. Nature 1980, 286, 474–476. [Google Scholar] [CrossRef]
- Fu, X.; Long, J.; Wang, X.; Leung, Y.; Ding, Z.; Wu, L.; Zhang, Z.; Li, Z.; Fu, X. Photocatalytic reforming of biomass: A systematic study of hydrogen evolution from glucose solution. Int. J. Hydrog. Energy 2008, 33, 6484–6491. [Google Scholar]
- Fujishima, A.; Rao, T.N.; Tryk, D.A. Titanium dioxide photocatalysis. J. Photochem. Photobiol. C 2000, 1, 1–21. [Google Scholar] [CrossRef]
- Gslinska, A.; Walendziewski, J. Photocatalytic water-splitting over Pt-TiO2 in the presence of sacrificial Reagents. J. Energy Fuels 2005, 19, 1143–1147. [Google Scholar] [CrossRef]
- Kitano, M.; Tsujimaru, K.; Anpo, M. Development of water in the separate evolution of hydrogen and oxygen using visible-responsive TiO2 thin film photocatalysts: Effect of the work function of the substrates on the yield of the reaction. Appl. Catal. A 2006, 314, 179–183. [Google Scholar] [CrossRef]
- Bahruji, H.; Bowker, M.; Davies, P.R.; Pedrono, F. New insights into the mechanism of photocatalytic reforming on Pd/TiO2. Appl. Catal. B 2011, 107, 205–209. [Google Scholar] [CrossRef]
- Atkins, P.W. Physical Chemistry, 5th ed; Oxford University Press: Oxford, UK, 1994; pp. 922–926. [Google Scholar]
- Kennedy, J.C., III; Datye, A.K. Photochemical heterogeneous oxidation of ethanol over Pt/TiO2. J. Catal. 1998, 179, 375–389. [Google Scholar]
- Kim, Y.-K.; Sakano, Y. Analyses of reducing sugars on a thin-layer chromatographic plate with modified Somogyi and Nelson reagents, and with copper bicinchoninate. Biosci. Biotechnol. Biochem. 1996, 60, 594–597. [Google Scholar] [CrossRef]
- Chiarello, G.L.; Aguirre, M.H.; Selli, E. Hydrogen production by photocatalytic stream reforming of methanol on noble metal-modified TiO2. J. Catal. 2010, 273, 182–190. [Google Scholar]
- Yang, Y.Z.; Chang, C.-H.; Idriss, H. Photo-catalytic production of hydrogen from ethanol over M/TiO2 catalysts (M = Pd, Pt, or Rh). Appl. Catal. 2006, 67, 217–222. [Google Scholar] [CrossRef]
- Daskalaki, V.D.; Kondarides, D.I. Efficient production of hydrogen by photo-induced reforming of glycerol at ambient conditions. Catal. Today 2009, 144, 75–80. [Google Scholar]
- Ho, C.-H.; Shieh, C.-Y.; Tseng, C.-L.; Chen, Y.-K.; Lin, J.-L. Decomposition pathways of glycolic acid on titanium dioxide. J. Catal. 2009, 261, 150–157. [Google Scholar]
- Zheng, X.-J.; Wei, L.-F.; Zhang, Z.-H.; Jiang, Q.-J.; Wei, Y.-J.; Xie, B.; Wei, M.-B. Research on photocatalytic H2 production from acetic acid solution by Pt/TiO2 nanoparticles under UV irradiation. Int. J. Hydrog. Energy 2009, 24, 9033–9041. [Google Scholar]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Shiragami, T.; Tomo, T.; Tsumagari, H.; Ishii, Y.; Yasuda, M. Hydrogen Evolution from Napiergrass by the Combination of Biological Treatment and a Pt-Loaded TiO2-Photocatalytic Reaction. Catalysts 2012, 2, 56-67. https://doi.org/10.3390/catal2010056
Shiragami T, Tomo T, Tsumagari H, Ishii Y, Yasuda M. Hydrogen Evolution from Napiergrass by the Combination of Biological Treatment and a Pt-Loaded TiO2-Photocatalytic Reaction. Catalysts. 2012; 2(1):56-67. https://doi.org/10.3390/catal2010056
Chicago/Turabian StyleShiragami, Tsutomu, Takayuki Tomo, Hikaru Tsumagari, Yasuyuki Ishii, and Masahide Yasuda. 2012. "Hydrogen Evolution from Napiergrass by the Combination of Biological Treatment and a Pt-Loaded TiO2-Photocatalytic Reaction" Catalysts 2, no. 1: 56-67. https://doi.org/10.3390/catal2010056
APA StyleShiragami, T., Tomo, T., Tsumagari, H., Ishii, Y., & Yasuda, M. (2012). Hydrogen Evolution from Napiergrass by the Combination of Biological Treatment and a Pt-Loaded TiO2-Photocatalytic Reaction. Catalysts, 2(1), 56-67. https://doi.org/10.3390/catal2010056




