Abstract
Biocatalysis holds immense potential for pharmaceutical development as it enables synthetic routes to various chiral building blocks with unparalleled selectivity. Therein, solvent and water use account for a large contribution to the environmental impact of the reactions. In the spirit of Green Chemistry, a transition from traditional highly diluted aqueous systems to intensified non-aqueous media to overcome limitations (e.g., water shortages, recalcitrant wastewater treatments, and low substrate loadings) has been observed. Benefiting from the spectacular advances in various enzyme stabilization techniques, a plethora of biotransformations in non-conventional media have been established. Deep eutectic solvents (DESs) emerge as a sort of (potentially) greener non-aqueous medium with increasing use in biocatalysis. This review discusses the state-of-the-art of biotransformations in DESs with a focus on biocatalytic pathways for the synthesis of active pharmaceutical ingredients (APIs). Representative examples of different enzyme classes are discussed, together with a critical vision of the limitations and discussing prospects of using DESs for biocatalysis.
1. Introduction
Biocatalysis has become a powerful toolbox for the pharmaceutical industry, enabling elegant synthetic routes toward a broad range of active pharmaceutical ingredients (APIs) [1,2,3,4,5]. Around 57% of known APIs are chiral compounds and commercialized in an enantiomerically pure form [6]. Enzymes are nature-privileged chiral catalysts with unparalleled selectivity (regio-, enantio-, and chemo-selectivity), which makes them especially appealing for the synthesis of chiral complex motifs. Notably, enzymes have been used in the commercial synthesis of more than two-thirds of chiral compounds on an industrial scale [7]. Additionally, biocatalysts are produced from renewable resources through fermentation, being inherently benign and biodegradable. On that basis, the last two decades have witnessed a compounded growth of biocatalysis in the synthesis of building blocks in the pharmaceutical, flavor, and fragrance sectors. More enzyme classes have been involved including hydrolases, oxidoreductase, transaminases, and lyases [8]. The incorporation of biocatalysis into industrial processes is continuously expanding and their use may potentially become one of the major pillars for Green Chemistry. Herein, a holistic consideration is required on the path to sustainable chemistry [9], and several interrelated aspects should be taken into account for efficient process intensification including reactor design [10,11], catalyst design and selection [12,13], medium engineering [14], process analytical technologies, downstream process, and ultimate product and enzyme disposal [9,15]. In particular, an adequate trade-off solvent selection with an emphasis on its efficiency, economy, enzyme-compatibility, and greenness is fundamental and currently a main focus in both chemo- and bio-catalysis [16,17].
Solvents contribute up to 70–80% of the total process solution and up to 60% of the mass used in pharmaceutical syntheses [17,18]. Importantly, instead of just being a reaction space, solvents act as solubilizers for reaction components while providing a suitable reaction environment and compatible media for (bio)catalysts [19]. Moreover, many solvents act also as reactive media and display synergistic effects (e.g., activating/deactivating a catalyst) [14], significantly affecting the biocatalytic performance, substrate specificity, and even stereoselectivity. Customizing solvents for certain circumstances can thus be a game-changer for (bio)catalytic processes, providing more efficient and cleaner synthetic strategies.
Biocatalysts have naturally evolved to function optimally in aqueous media, and thus conducting biotransformations in various aqueous conditions is the traditional approach to maximize enzyme performance (activity and stability). Water is often claimed as a non-toxic and readily available green solvent, although its high polarity becomes a key obstacle when hydrophobic non-natural substrates are applied [20]. As a result, the concentrations of poorly water-soluble chemicals are downgraded to the millimolar range. These diluted biocatalyzed processes in aqueous media typically produce large volumes of wastewater, compromising the pursuit of economy and sustainability [21]. The wastewater treatment of such large effluents, an easily overlooked aspect, makes an additional negative contribution to the environmental impact of the entire process. To overcome this, and to improve water-based industrial (bio)catalytic processes, the use of surfactants to enhance substrate solubility has been proposed [22].
To fairly evaluate and compare the greenness of bioprocesses, several quantitative environmental metrics have been introduced, such as the E-factor (environmental factor, kg waste kg product−1) [23,24], the E+-factor (classical E-factor plus CO2 emissions caused by electricity generation) [25], and the total CO2 production (kg CO2·kg product−1) [26,27]. Other aspects, such as the inherent hazardousness of wastes, or energy consumption, should be addressed too. A recent study identified the CO2 production of generic bioconversions in water and non-conventional media by evaluating the upstream and the downstream (with a focus on the extraction of the aqueous media with an organic solvent). It revealed that, in general, the lower the substrate loadings, the higher the CO2 production (regardless of whether the biotransformation is conducted in water or in non-conventional media) [26]. Thus, the key to decreasing the environmental footprint is to maximize production titers, leading to an optimized use of solvents and water and improving process economics at the same time [28]. Industrial processes are, most of the time, performed at substrate loadings of >50 g L−1, and preferably at >100 g L−1 for fine chemicals [29]. Comparatively, pharmaceutical segments are confronting the challenge with 10-fold less production and >5-fold higher E-factor compared to other chemical industries [3,28,29]. The E-factor is, in fact, even higher in the current ‘chiral era’ than in the previous ‘racemic era’ [30]. Hence, economic and environmental improvements in the API manufacturing processes are continuously in demand.
In this context, the introduction of non-conventional media to enhance substrate loadings may become a straightforward solution for process intensification [31]. The use of non-aqueous solvents not only enables high substrate loadings to adapt to industrial scale-up and commercialization [29,32] but also allows for seamless integration of chemo-enzyme cascades in industry, where, typically, other chemical steps are conducted in non-aqueous media [33]. The systematic development of biotransformations in non-aqueous media dates back to the 1980s, starting with a focus on the workhorse of lipases in organic solvents [34,35], followed by new generations of master solvents including ionic liquids (ILs) and supercritical liquids in the subsequent decades. These solvents have historically seen ups and downs due to their strengths and weaknesses. In addition to the respective drawbacks of each type of solvent, the greatest concern of applying these conventional non-aqueous solvents is their inherent toxicity. This is driving the transaction from traditional fossil-based solvents to alternative biomass-based solvents or even solvent-free systems [36]. In particular, biomass-derived solvents from renewable natural resources (e.g., plants and animals) have become attractive for the large-scale production of a wide range of green solvents at acceptable prices. Compared to their counterparts, biomass-based solvents are safer and more effective from manufacture to application, and thus more sustainable [37].
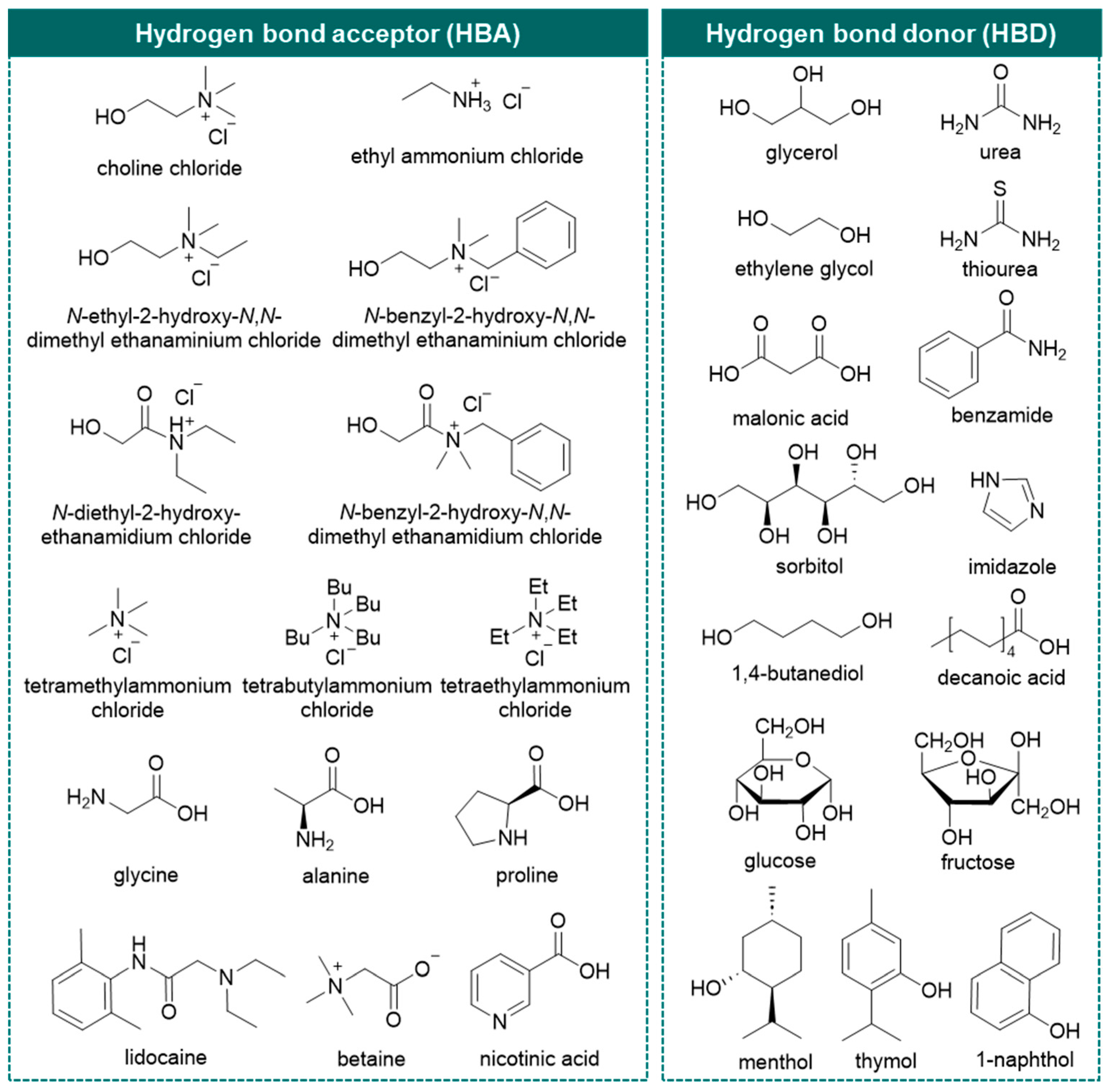
In this area, deep eutectic solvents (DESs) emerge as a neoteric class of (potentially) greener solvents and have received tremendous attention since their debut in Abbott’s seminal study [38]. The definition of DESs has gradually been completed through their in-depth understanding. They are eutectic mixtures formed from Lewis/Brønsted acids and bases as hydrogen bond donors (HBD) and hydrogen bond acceptors (HBA) forming hydrogen bond networks [39,40]. Nowadays, five types of DESs are defined based on the nature of compositions: Type I—quaternary ammonium salt and metal chloride; Type II—quaternary ammonium salt and metal chloride hydrate; Type III—quaternary ammonium salt and HBD; Type IV—metal chloride hydrate and HBD [39,41]; and a new Type V, comprising only nonionic molecular HBA and HBD [42]. Type III DESs, comprising of quaternary ammonium salts, typically choline chloride (ChCl), and organic molecular HBDs (amide, carboxylic acid, or polyol) are widely used (Figure 1) [40]. The usual expression of a DES as HBA: HBD (n:n, mol/mol) will be used in this review.
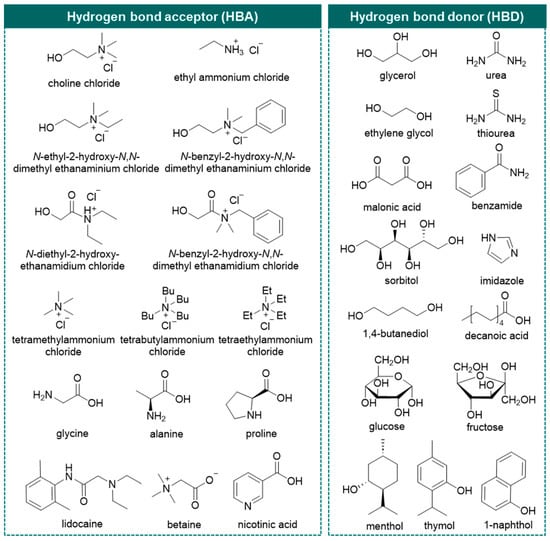
Figure 1.
Chemical structures of various representative hydrogen bond acceptors (HBA) and hydrogen bond donors (HBD) for type III deep eutectic solvents used in previous studies.
DESs may hold remarkable advantages in ecology, economy, and practicability, and have been hailed as ‘the solvents of the 21st century’ [43]. On the one hand, DESs share similar physicochemical properties with ILs (high thermostability, low volatility, non-flammability, and tunable polarity). On the other hand, DESs are ‘designer’ solvents that are a function of their compositions, which lends DESs enormous versatility and tunability to adapt for desired purposes. Via a defined combination of constituents, DESs with tailored hydrophilicity/hydrophobicity can be accessed to (de)solubilize various chemicals [44,45,46,47,48]. Besides this, the strong mutual attractive interaction between DES components decreases their reactivity, minimizing side reactions in many cases [49]. Thanks to that, DESs have found prospective applications in many fields, including metallurgy and electrodeposition, separation and gas capture, battery technology, nanotechnology, separations and gas capture, biomolecule stabilization, and organic syntheses [39,40,43,50,51]. Specifically, chemical catalysis involving DESs has been developed primarily for organometallic chemistry and metal-catalyzed reactions [52,53,54,55,56,57]. In the case of using DESs for biocatalytic purposes, DESs can offer special benefits. For example, the molecular interactions can be established between DESs and biocatalysts/cofactors, providing stabilization effects on them [58]. Using DESs can sometimes boost reaction efficiency, while the selectivity of biocatalysts can also be enhanced/modified due to a well-organized 3D-network [59]. Despite these advantages, however, when it comes to sustainability, making unsubstantiated claims about DESs greenness should be avoided. DESs can only be made affordable, non-toxic, and biodegradable if the composition is properly chosen. Natural DESs (NADESs) derived from biomass may be good examples to fulfill these criteria [43,60]. The high degree of freedom in designing DESs using a variety of (biogenic) substances and explorable molar ratios (HBA: HBD) allows for the generation of a versatile toolbox containing more sustainable customized solvents [61]. In general terms, the proposition of DESs’ greenness needs an in-depth and holistic analysis from a life-cycle perspective [62]. DESs can be prepared by mixing HBA (typically a quaternary ammonium salt) and HBD (e.g., glycerol, urea), enabling a large-scale supply of DESs at low cost. A major challenge for using DESs is their high viscosity (∼7−86,800 mPa·s at room temperature) compared to water (0.89 mPa·s) and most organic solvents [46,63]. Nevertheless, this has not hindered its rapid advancement in many fields, e.g., extractive agents, biorefineries, organic synthesis [64], and others [65,66].
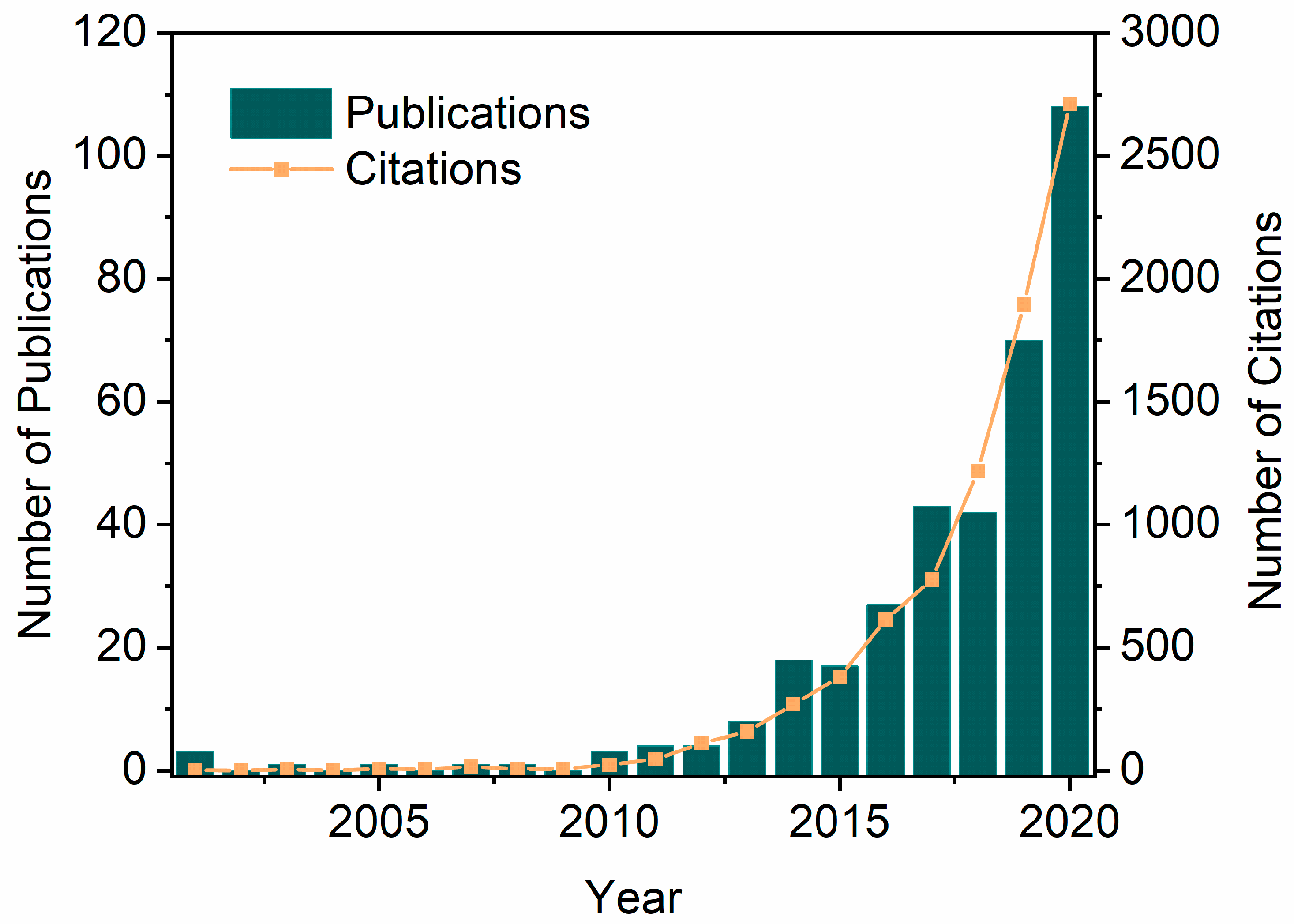
Biocatalysis in DESs combines strengths such as enzyme selectivity and DES tunability to maximize the range of their applications, thus seeing a spiral uptick in the last two decades. The number of publications and citations on the topic ‘enzymatic reactions or enzymes and eutectic solvents’ has been increasing considerably from 2001 to 2020, with the number of publications going from 2 up to 108 and citations going from 0 to 2713 (Figure 2). These values increase more drastically when the topic is expanded to chemo catalysis in DESs, which is beyond the scope of this review and therefore not shown. A variety of biotransformations have been established in DESs to synthesize high-value chemicals using hydrolases, oxidoreductases, lyases, and amine transaminases, wherein DESs may function as solvents, cosolvents, additives, or even as substrates [67,68,69,70]. Rational design of customized DES for biocatalytic applications requires a holistic consideration of several aspects, including water content to retain enzyme activity, polarity to solubilize reactants, and viscosity to enable practical operation. Correspondently, these considerations can be addressed by introducing water, varying DES components and their molar ratios, and increasing temperatures [71]. The relative low viscosity of most DESs can be achieved by elevating temperatures (200–500 mPa·s at 40 °C), which is, however, incompatible with many enzymes. Alternatively, water is often added as a cosolvent to DESs, effectively reducing viscosity while providing a sufficient hydration environment for enzymes [72]. The water capacity and its role in DESs have been explored to broaden the tunable window of DES–water mixtures while keeping their hydrogen-bonding network maintained. For instance, a computational study on choline chloride-glycerol (ChCl-Gly) has shown that water plays a Janus-faced de/structuring role in the interactive hydrogen-bonding network between ChCl and Gly [73]. The increase in hydration via the initial addition of water (10.1 wt.%) indeed reduced the hydrogen-bonding interaction between ChCl-Gly but, at the same time, water molecules function similarly to chloride to bridge the DES components of Ch and Gly, reaching the maximum structuring effect at 35.8 wt.%. Likewise, the nanostructure of widely-used choline chloride-urea (ChCl-U) was shown to remain even in the presence of a remarkably high level of water content (ca. 42 wt.%) [74]. In addition, when water and DES components are part of the hydrogen-bonding network, their reactivity is significantly reduced [49]. This avoids possible side reactions and unfavorable thermodynamic equilibria. As a result, DES–water mixtures become suitable and practical, and thus are being used in a wide range of catalysis scenarios for different purposes [71,72,75], e.g., solubility improvement, biocatalyst stabilization, selectivity modification, and ease of reusability.
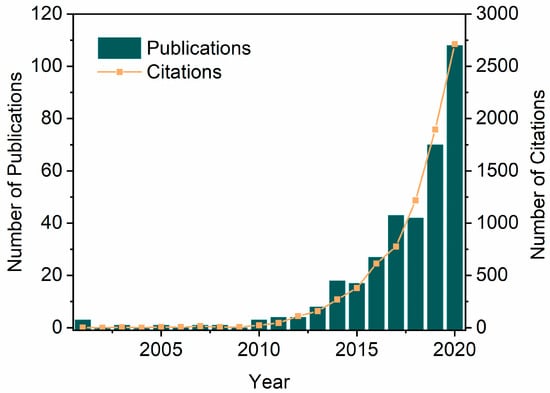
Figure 2.
Analysis of publications and citations on ‘enzymatic reactions or enzymes and eutectic solvents’ year on year from 2001 to 2020 (Web of Science, last update 28 November 2023).
Despite all the above-mentioned advantages of combining DESs and biocatalysis, there are still challenges prior to their prevalent practical applications. In particular, enzyme deactivation occurs quite often in the presence of DESs. On the one hand, this promotes the initiation of whole-cell catalysis in DESs, which naturally provides a protective environment to enzymes. On the other hand, this has led to numerous studies on the relation between enzyme-related features and DES-related properties. The impact of DESs has been explored, e.g., earlier, mainly on hydrolases [49,76], and recently on other enzymes such as lyases [77], peroxidases [78,79,80], alcohol dehydrogenases [75,81,82,83], and photosynthetic reaction centers [84]. To adapt enzymes to harsh DES conditions, a variety of immobilization techniques (e.g., physical adsorption/deposition, entrapment, covalent attachment, and cross-linked enzyme aggregates) can be employed to generate robust immobilized biocatalysts that can be easily processed and reused in DES systems. On this basis, a number of biotranformations have been developed using immobilized whole cells or enzymes, which are outlined in detail in the following sections. Moreover, it was revealed that some DESs even displayed stabilizing effects on nicotinamide cofactors compared to aqueous media, which lays another solid basis for applying cofactor-dependent oxidoreductases in DESs [58]. In addition, to optimize the biocatalytic processes in DESs, the DES-related physicochemical parameters such as dynamic viscosity [80], compound solubility [85,86,87], dynamic water activity [39], and oxygen transfer rates (OTRs) [71] were investigated comprehensively via experiments and computational analysis. The gained knowledge has been used to drive the rational design of tailored enzyme-compatible DESs for specific circumstances. Some comprehensive review articles on the topic of ‘combining enzymes and DESs for organic synthesis’ were published recently [88]. These organic syntheses target miscellaneous valuable chemicals including pharmaceutical-relevant compounds [89,90], cosmetics [81,91,92,93], food industrial chemicals [94,95,96], surfactants [97,98,99,100], polymers [101,102,103], biodiesel [104,105,106,107,108], and so on. To complement these works, this review is dedicated to biocatalysis in DESs with a specific focus on syntheses of various building blocks for APIs, potential pro-drugs, and pharmaceutical-relevant compounds.
2. Oxidoreductase-Meditated API Syntheses in DESs
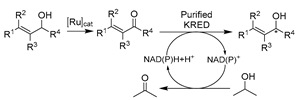
Oxidoreductases (E.C. 1)-catalyzed reactions account for around one-third of enzymatic transformations in the BRaunschweig ENzyme DAtabase (BRENDA) [109]. In particular, selective redox biocatalysis plays a pivotal role in the synthesis of complex APIs [110]. Some well-known oxidoreductases have been widely used for organic syntheses, including alcohol dehydrogenase (ADH, KRED, E.C. 1.1.), laccases (E.C. 1.10), peroxidases (E.C. 1.11), monooxygenases (E.C. 1.13), NAD(P)H oxidoreductases (E.C. 1.6), and sulfur oxidoreductases (E.C. 1.8). Amongst them, ADHs are mostly used in the asymmetric reduction of ketones for the synthesis of various value-added chiral alcohols [111]. ADHs are cofactor-dependent and require a stoichiometric amount of costly NAD(P)H. The in situ cofactor-regeneration is proposed to enable a cost-effective application [112,113,114,115]. That necessity can be fulfilled by either in vivo designer cell systems or in vitro artificial regeneration systems, using an ancillary substrate [116].
2.1. Whole-Cell Catalyzed Redox Catalysis in DESs
Redox catalysis in DESs was initiated with whole cells due to the provision of a natural environment, avoidance of enzyme purification, and in situ cofactor-regeneration. ChCl-based classic DESs were first adopted due to the wealth of relevant knowledge about them. Domínguez de María et al. seminally reported the use of wild-type whole-cell baker’s yeast in DES–water mixtures for the selective reduction of ethyl acetoacetate to afford (R/S)-ethyl 3-hydroxybutanoate (Table 1, entry 1) [117]. The (R)-enantiomer is of high interest as a building block for the preparation of (+)-decarestrictine L or ß-lactamase inhibitors, while the S-enantiomer is used in the synthesis of pheromones and carbapenem antibiotics [118]. A complete stereoinversion (ee 95% (S)- to 95% (R)-) was observed, presumably as a result of the deactivation of (S)-enantioselective enzymes within whole cells when pure ChCl-Gly was applied [117]. The same reaction was studied in more ChCl-based DESs with various hydrogen bond donors (sugars, alcohols, acids, or amides) [119]. This work demonstrated the significant effects of varying DES components and water contents on the activity and enantioselectivity for yeast-mediated reduction, leading to a promising bioprocess for (R/S)-enantiomer production. The gained selectivity reversal is presumably attributed to the interplay between the “solvation” of whole cells and the “selective inhibition” of specific enzymes, which was caused by the fine-tuning of DES constituents and water proportion. This finding was further observed in a baker’s-yeast-catalyzed bioreduction of various enantioenriched arylpropanones in water and 90 wt.% ChCl-Gly (1:2), giving rise to (S)- and (R)-1-phenoxy-2-propanol, respectively, as key synthons for the preparation of neuroprotective drugs (Table 1, entry 2) [120].
Additionally, other types of wild-type whole-cell biocatalysts were applied in DESs for the synthesis of chiral intermediates of APIs. Lou’s research group has focused on the free or immobilized cell-facilitated asymmetric oxidations or reductions in DES-enriched media to produce various chiral alcohols, including the oxidative kinetic resolution of racemic 1-(4-methoxyphenyl)ethanol (MOPE) [121,122] and the reductions of 3-chloropropiophenone [123], 2-octanone [124], and 2-hydroxyacetophenone [125] (Table 1, entries 3–6). The enantiopure (S)-MOPE is an important chiral building block for the synthesis of cycloalkyl[b]indoles as a treatment for general allergic responses [126]. The oxidation of MOPE catalyzed by immobilized Acetobacter sp. CCTCC M209061 cells was conducted in various ChCl-based DESs, revealing an optimal option of ChCl-Gly (1:2) with 10 wt.% water regarding reaction efficiency (Table 1, entry 3) [121]. A follow-up study found that the introduction of 10 vol% ChCl-Gly to an ionic liquid/buffer biphasic system drastically increased the reaction rate and enantioselectivity of the same catalysis [122]. Moreover, not only oxidations were assessed, but the research for the same immobilized cells was expanded to the asymmetric reduction of 3-chloropropiophenone in various ChCl-based DESs systems (Table 1, entry 4) [123]. The obtained (S)-3-chloro-1-phenylpropanol ((S)-CPL) is an essential building block for the synthesis of antidepressants (S)-fluoxetine, nisoxetine, and (R)-tomoxetine [127,128]. The overall productivity and enantioselectivity were improved in the optimized DES-IL-buffer system. In one of their contributions, an efficient biocatalytic route for (R)-1-phenyl-1,2-ethanediol (PED) production, a crucial part for the preparation of β-adrenergic antagonists for treating systemic hypertension [129], was established with Kurthia gibsonii SC0312 in ChCl-1,4-butanediol (ChCl-1,4-Bd)-containing buffer system (Table 1, entry 5) [125]. The presence of 2 vol.% ChCl-Bd enhanced the reaction rate of cells by 22%. In these accounts, various ChCl-based DESs were examined for specific reactions catalyzed by whole cells, revealing very different optimal DES-enriched systems. These reaction efficiencies were drastically boosted due to DES-stimulated enhancement of substrate loadings and improved cell membrane permeability. Similar findings were documented in other contributions about wild-type whole-cell-mediated biotransformations in the presence of various DESs. The stereoselective reduction of α-acetylbutyrolactone was developed with Candida viswanathi AM120 in 10 vol.% ChCl-Gly, leading to the production of α’-1′-hydroxyethyl-γ-butyrolactone, a chiral chemical similar to γ-butyrolactone (GBL) with potential psychoactivity (Table 1, entry 6) [130]. Likewise, another baker’s yeast Saccharomyces cerevisiae cell-catalyzed reduction of acetophenone derivatives was explored in three ChCl-based DESs, producing three chiral alcohols as building blocks for drug synthesis (Table 1, entry 7) [131]. The highest enantioselectivity was achieved in ChCl-Gly with 30 vol.% water, and the reduction was performed on a preparative scale with reuse of DESs. Last but not least, the reduction of 3,5-bistrifluoromethylphenone catalyzed by various whole cells to produce (R)- or (S)-3,5-bistrifluoromethylphenyl ethanol ((R)-/(S)-BTPE) was evaluated in several ChCl-based DESs (Table 1, entry 8) [132,133,134]. Both (R)-/(S)-BTPE are key respective intermediates for the synthesis of antiemetic drugs aprepitant (Emend®) and fosaprepitant (Ivemend®) and the preparation of NK-1 receptor antagonist. The introduction of 1 vol.% ChCl-U (1:1) or ChCl-glutathione (ChCl-GSH, 1:1) significantly increases cell membrane permeability and substrate solubility, finally leading to high enantioselectivity and yields. These studies further impart the knowledge that the effects of DESs can be quite diverse toward various reaction systems. In general, some of the studied DESs showed a beneficial impact on wild-type whole-cell biocatalysts (baker’s yeast, Candida viswanathi AM120, Acetobacter sp., and R. erythropolis), which expressed several analogous oxidoreductases at low levels. Therefore, the diverse effects of DESs are presumably attributed to the restraint of some enzymes by specific DESs, manifesting the great potential of DESs for manipulating various selective whole-cell bioprocesses.

Table 1.
Representative redox catalysis catalyzed by whole cells and isolated enzymes in DESs.
Table 1.
Representative redox catalysis catalyzed by whole cells and isolated enzymes in DESs.
| Entry | Reaction Scheme | Reaction Medium | Concluding Remark | Ref. |
|---|---|---|---|---|
| 1 | 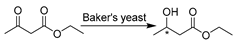 | ChCl-Gly (1:2) with 0–100 vol.% water, and other ChCl-based DESs | Stereoinversion, (S)- in water and (R)- in DES | [117,119] |
| 2 | 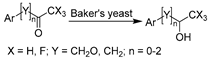 | ChCl-based DESs with 0–50 wt.% water | High selectivity and stereoinversion, (S)- in water and (R)- in DES | [120] |
| 3 | 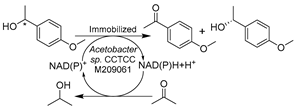 | ChCl-based DESs with 10 vol.% water, and ChCl-Gly/buffer/[C4MIM][PF6], 10/40/50 (vol.%) | Enhanced substrate loadings, membrane permeability, and reaction rate | [121,122] |
| 4 | 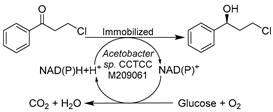 | Buffer with 5 vol.% ChCl-based DESs, and ChCl-U containing buffer/[C4MIM][PF6], 80/20 (vol.%) | Improved yield and productivity at 0.5 L scale | [123] |
| 5 |  | ChCl-based DESs, buffer with 2 vol.% ChCl-Bd (1:4) | Improved membrane permeability and reaction rate | [125] |
| 6 |  | Buffer with 10–25 vol.% ChCl-Gly (1:2) | Reduced reaction course and increased stereoselectivity, (85% dr., 76% ee) | [130] |
| 7 | 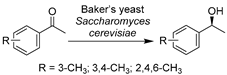 | ChCl-based DESs with 30/50/80 vol.% water, ChCl-Gly (1:2) with 30 vol.% water | Improved conversion at preparative scale with reuse of DESs | [131] |
| 8 |  | ChCl-based DESs, optimal ones of buffer with 1 w/v% ChCl-U (1:1) or ChCl-GSH (1:1) | Improved substrate solubility and yield, shortened reaction time | [132,133,134] |
| 9 | 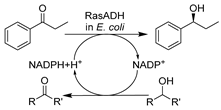 | ChCl-Gly (1:1.5, 1:2)/ChCl-U (1:2) /ChCl-EG (1:2) with 0–90 vol.% buffer | Improved enantioselectivity | [75,135] |
| 10 | 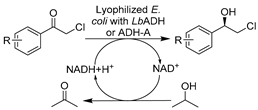 | ChCl-Gly (1:2) with 50–80 vol.% buffer | >90% conv. and >99% ee with 0.3–0.4 M substrate | [136] |
| 11 | 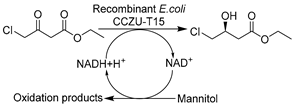 | ChCl-Gly (1:2) with 80–100 vol.% buffer | 93–100% conv. and >99% ee with 1–3 M substrate | [137] |
| 12 | 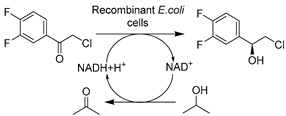 | ChAc-Lys (1:1) (1 w/v%) in buffer | 87% conv. and >99% ee with 1 M substrate; improved cofactor regeneration | [138] |
| 13 | 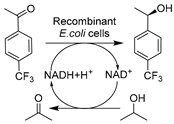 | Various betaine-based NADES, optimal one Bet-Lys (1:2) (3 w/v%) in buffer | 92.4% conv. with 400 mM substrate at 0.1 L scale; improved cell membrane integrity | [139] |
| 14 | 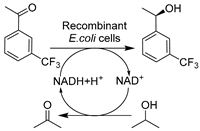 | 4 w/v% ChCl-Lys (1:1) with 0.6 w/v% Tween-20 in buffer | 4.0-fold enhanced substrate conc., >99% ee, and 91.5% conv. with 0.2 M substrate | [140] |
| 15 | 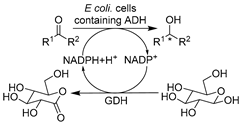 | 10–50 vol.% ChCl-Glu (1.5:1) in buffer | HBD glucose in ‘designer’ NADESs served as co-substrate and solubilizer | [141] |
| 16 | 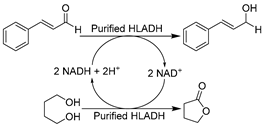 | 20 vol.% ChCl-Gly (1:9) in buffer; 80 vol.% ChCl-1,4-BD (1:4) in buffer | Improved STY of 15.3 g L−1 d−1; HBD 1,4-BD as cosubstrate with STY of 75 g L−1 d−1 | [81,93] |
| 17 | 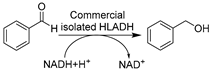 | 50 wt.% LID-OA (1:1) with lower critical solution temperature (LCST) in buffer | Enzyme reused 3 times | [142] |
| 18 | 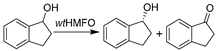 | Various ChCl-/sugar-based DESs, and 60 vol.% Glu-Fru-water (1:1:6) | Improved yield over pure buffer system (26% vs. 7%) | [143,144] |
| 19 | 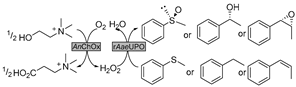 | 0–100 vol.% various ChCl-based DESs | HBA ChCl as co-solvent and stoichiometric reductant | [145,146] |
| 20 | 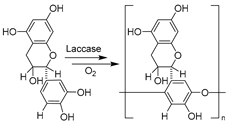 | ChCl-/Bet-based NADESs, 5 vol.% Bet-Man (5:2) in buffer | Highest molecular weight polycatechin | [147] |
To further boost the efficiency toward target reactions and broaden the biocatalytic toolbox, creating recombinant whole cells overexpressing specific oxidoreductases or employing precursors for biosynthesis in DES-enriched media can be advantageous. The initial studies proceeded with predominant ADHs with the goal of producing diverse chiral alcohols as precursors for API synthesis. It was disclosed that E. coli cells overexpressing ADHs remained active even at high DES concentration (80 vol.%) and could catalyze the bioreduction of various aromatic substrates with highly improved enantioselectivity over buffer systems (Table 1, entry 9) [135]. This finding is in line with that for wild-type whole cells in DESs and was further cemented by confirming the stabilizing effects of DES components [75]. Likewise, E. coli cells harboring overexpressed two stereo-complementary enzymes (Lactobacillus brevis ADH and Rhodococcus ruber ADH (so-called ADH-A)) were used in the synthesis of enantiopure halohydrins, key intermediates for many commercialized medicaments (e.g., mirabegron, sertraline, luliconazole, ezetimibe, and empagliflozin) [136]. The reduction of α-halogenated ketone precursors was performed in a eutectic mixture composed of ChCl-Gly and Tris-SO4 buffer, enabling high substrate loadings (0.3–0.4 mM) as well as excellent conversions and enantiomeric excesses (Table 1, entry 10). These studies maximize the potential of DESs to increase substrate loading, thereby improving productivity. In fact, an increase in substrate capacity can mitigate substrate inhibition, especially that caused by carbonyl compounds [137]. This finding was disclosed in an E. coli whole-cell mediated biosynthesis of ethyl (S)-4-chloro-3-hydroxybutanoate [(S)-CHBE], which is known as a key precursor for the synthesis of cholesterol-lowering HMG-CoA reductase inhibitors such as atorvastatin (Table 1, entry 11) [148]. In this regard, the use of 12.5 vol.% ChCl-Gly together with a surfactant tween-80 boosted the loading of water-insoluble substrate to 1–3 M while greatly improving cell permeability [137]. These discoveries broaden new research lines for managing intensified selective biocatalysis while tackling the economics and the environmental aspects of the process simultaneously.
Apart from conventional ChCl-based DESs, the development of novel biocompatible and bio-based natural DESs (NADESs) has become relevant to target process sustainability [134,138,139,141]. Recently, Wang et al. made substantial contributions in applying novel task-specific DESs in selective whole-cell catalysis for the synthesis of valuable pharmaceutical building blocks [134,138,139,140]. Various bio-based DESs were applied in the E. coli-catalyzed bioreduction of 2-chloro-1-(3,4-difluorophenyl)ethanone (CFPO) to produce (S)-2-chloro-1-(3,4-difluorophenyl)ethanol ((S)-CFPL) (Table 1, entry 12), which is a crucial synthon for synthesizing P2Y12 receptor antagonist Ticagrelor (Brilinta®) [138]. Introducing 1 w/v% choline acetate/lysine (ChAc-Lys) to aqueous systems enabled improved cofactor regeneration and a 3.3-fold increase in substrate loading, finally leading to productivity of 87% conversion and >99% ee at 1 M CFPO content [138]. The spectrum of biocompatible DES was broadened in a follow-up study on the E. coli-mediated bioproduction of (R)-1-[4-(trifluoromethyl)phenyl]ethanol (Table 1, entry 13), an important intermediate of many drugs like chemokine CCR5 antagonist [139]. Similarly, applying the least amount of 3 w/v% Betaine/lysine DESs (B-Lys, 1:2) in aqueous media enabled high substrate loadings. Furthermore, these lysine-based NADES exhibited better biocompatibility over traditional ChCl-based DESs by maintaining cell membrane integrity. The synergistic effect contributes to the enhanced catalytic efficiency, resulting in a yield of 92.4% and >99.9% ee at a substrate conc. of 0.4 M [139]. These studies firmly demonstrated the applicability and vast available choices of customized NADES. Some NADESs are even integrated with surfactants to further boost process intensification of whole-cell mediated reductive biotransformations. For example, in the case of yeast-catalyzed reduction of BTAP to produce (S)-BTPE, the addition of 1 w/v% L-carnitine/lysine (C-Lys, 1:2) and 5 g/L Tween-80 further boosted substrate solubility to 7.6-fold over a pure aqueous system (Table 1, entry 8) [134]. A similar finding was unraveled in an E. coli-catalyzed synthesis of (R)-1-[3-(trifluoromethyl)phenyl]ethanol as a useful building block for a neuroprotective compound, (R)-3-(1-(3-(trifluoromethyl)phenyl)ethoxy)azetidine-1-carboxamide (Table 1, entry 14) [140]. This combinatorial strategy presents more perspectives for designing tailored DESs for specific biocatalytic applications through delicate medium engineering, especially in the cases of using non-natural hydrophobic substrates. The prospective designability of DESs is further manifested in cofactor-dependent reductions. A NADES ChCl-Glu was designed for the bioreductions catalyzed by five (R)- and (S)-selective ADHs as lyophilized E. coli preparations (Table 1, entry 15), producing a variety of valuable chiral alcohols [141]. Glucose in DES plays bifunctional roles as the co-substrate for GDH-mediated nicotinamide cofactor regeneration and as a co-solvent for the enhancement of substrate loadings. The synergistic effects highly contributed to transformations and even enabled semi-preparative biocatalytic syntheses [141]. Overall, these studies demonstrate that using DESs can increase substrate loadings, reduce substrate negative effects, enhance cell membrane permeability, and exert a synergistic effect on cofactor recycling.
2.2. Isolated Enzyme-Catalyzed Redox Catalysis in DESs
The extensive achievements of whole-cell catalysis in DESs (vide supra) inspired the application of isolated enzymes in DESs, including dehydrogenases [81,82,83,93,142], peroxygenases [145], and oxidases [61,89,143,144]. Again, the most attractive feature of using DESs here is to reach higher substrate loading on the premise of maintaining enzyme activity and selectivity. However, without the protection of cell walls, the catalytic performance of most isolated enzymes is still compromised in the presence of DESs. It is necessary to understand the influence of DESs on enzymes for the knowledge-oriented design of biocatalysis in DESs. Therefore, most studies have focused more on investigating and elucidating the effects of DESs on enzymes via studying the structure–function relation of purified enzymes. Considering the significance of ketoreductases, the holistic investigation of DES impact on enzymes as well as trial catalysis in DESs was first launched with representative ADHs. A combination of experimental and computational analyses revealed and interpreted the diverse impacts of various DESs and DES individual components, e.g., the positive effect of Gly vs. the negative effect of ChCl [81,83]. The gained knowledge was used to generate a novel eutectic mixture with higher Gly loading, ChCl-Gly (1:9), which was used in the reduction of cinnamaldehyde catalyzed by purified horse liver ADH (HLADH) (Table 1, entry 16). This led to a highly improved specific activity and promising productivity of 15.3 g L−1 d−1 (space-time yield, STY) for cinnamyl alcohol, as an important synthon for flavor and fragrance industries while also being a starting material for the synthesis of drugs dapoxetine and taxol [149]. It is noticeable that a smart co-substrate 1,4-butanediol (1,4-BD) is coupled for cofactor regenerating, shifting the equilibrium toward product γ-butyrolactone (GBL) [150]. This promoted the combination of smart co-substrate with DES to establish an efficient and environmentally friendly synthetic alternative. A new DES ChCl-1,4-BD (1:4) was designed and used in combination with a 20 vol.% buffer. The purified HLADH turned out to be still active, resulting in high STY (75 g L−1 d−1) for the reduction of cinnamaldehyde (Table 1, entry 16) [93]. These contributions firmly demonstrate that DESs can be rationally designed to be both substrate-solubilizers and enzyme-compatible, opening new research lines for sustainable chemistry and biocatalysis. Not only ChCl-based classic DESs but also novel DESs owning specific physicochemical properties have been investigated for enzyme catalysis. For example, a designer novel DES with hydrophobicity and thermo-morphicity, lidocaine/oleic acid (LID-OA, 1:1), has been employed for HLADH-catalyzed reduction of ethyl benzene to produce valuable chiral alcohols (Table 1, entry 17) [142]. In particular, the temperature-controlled multicomponent solvent system allows for the easy reusability of biocatalysts and enhanced substrate loadings. The new designer DESs hold great potential for ADH-promoted energy-efficient asymmetric reduction of prochiral ketones to produce a variety of value-added enantiopure alcohols.
Research on other oxidoreductases is also gradually emerging. In this context, 5-hydroxymethylfurfural oxidases (HMFO, E.C. 1.1.3.47) are appreciated for their versatility in catalyzing various selective oxidations of alcohols, aldehydes, and even thiols, with clear links to biorefineries as well. De Gonzalo et al. examined the catalytic performance of HMFO in various ChCl- and sugar-based DESs [143,144]. In DES-enriched buffer mixture comprising 60 vol.% Glu-Fru-water (1:1:6), the first HMFO-catalyzed enantioselective oxidation of benzofused racemic secondary alcohol (1-indanol) was performed for kinetic resolution (Table 1, entry 18) [143], resulting in a 4-fold improvement of conversion over the pure aqueous system, leading to 1-indanone with 26% conversion despite a relatively low 17% ee for (R)-1-indanol. The derivatives of produced 1-indanone have been widely used in medicine and natural product synthesis [151]. Subsequent protein engineering in combination with designed carbohydrate-based NADES was proven to be a promising toolbox for further improving the selectivity of HMFO and solubility toward various substrates [144]. Intriguingly, the other underlying factor, solubility of oxygen, was first studied to gain a full understanding of DES’s impact on oxygen-dependent biocatalysis. A mild decrease in oxygen solubility in the studied DES systems was observed. Additionally, the depletion of oxygen in reaction systems could be compensated via sparling in the liquid phase or above gases phase. This consequently raises the demand to acquire knowledge on the oxygen transfer rates (OTRs) to DES–water mixtures. Later on, the OTRs were systematically investigated by quantifying the volumetric mass transfer coefficient (kLa) for various DES–water systems at different temperatures [71]. Therein, from 4.4-fold to 14-fold lower kLa values were revealed for DES–water mixtures compared to pure water, which was effectively enhanced by adding water or increasing temperatures to abate viscosity. The knowledge can be used to establish and optimize oxygen-dependent biocatalysis in DESs. In this context, a compelling study is the development of cascades in DESs which were catalyzed by choline oxidase (AnChOx) (E.C. 1.1.3.17) and a recombinant peroxygenase from Agrocybe aegerita (rAaeUPO) (E.C. 1.11.2.1) (Table 1, entry 19) [145,146]. The produced high-value-added enantiopure alcohols and sulfoxides, as well as epoxides, have seen applications in many natural products and pharmaceutical agents [152,153]. Therein, various ChCl-based NADESs were employed, serving as co-solvents and stoichiometric reductants for H2O2 generation at the same time. The studied DESs showed general stabilizing effects on rAaeUPO, whereas AnChOx showed reduced robustness in the presence of DESs [145]. In fact, discrepant findings were discovered for the cascades applying different substrates. Considering two studies together, ChCl-U-Gly was favorable for the hydroxylation of ethyl benzene and ChCl-Pro-H2O for the epoxidation of cis-ß-methylstyrene [145], while no specific benefit of the studied DESs was found for the sulfoxidation of thioanisole [146]. Some of these preliminary results lack plausible interpretation and propel further investigation for further practical applications.
In addition, laccases (oxygen oxidoreductase, E.C. 1.10.3.2) are multicopper-containing oxidases that use oxygen and substrate to initiate catalysis and can catalyze single-electron oxidization of a wide range of organic and inorganic compounds [61,89,154]. The research on laccase catalysis in DESs has mainly focused on the DES impact on enzymes’ activity and stability but less on process applications [61,89,147,154,155,156]. It has been proven that the activity and stability of laccases could be maintained in choline-based DESs and even enhanced in betaine-based NADESs [155,156], which can be attributed to the established hydrogen bonds between DES components and enzyme amino acids [61]. Based on these studies, the use of DESs was attempted for laccase-catalyzed polymerization of valuable compounds. For example, replacing the organic solvent with 5 vol.% betaine/mannose (Bet-Man, 5:2) in buffer enabled the production of the highest molecular weight polycatechin that holds potential use as an antioxidant molecule (Table 1, entry 20) [147]. Similar findings were discovered in the laccase-catalyzed oligomerization of rutin, which exhibits enhanced biological, pharmacological, antioxidant, and solubility properties over its monomer [89]. Bet-Man showed significant positive effects on laccase activity and caused spontaneous derivatization of rutin as well as oligomerization of rutin and these derivatives. The eco-friendly synthetic routes of other oligomeric or polymeric products owning biological and pharmacological properties were also realized with laccase in DESs, Bet-Gly (1:2) for flavonoid dihydroquercetin (DHQ, taxifolin) oligomers [154], and D,L-menthol/decanoic acid (Ment-DA, 1:1) polyaniline [157]. Overall, these studies provide insights into the development of green polymerization processes by using NADES–water mixtures. However, the recovery of target products (the downstream) remains a challenge. In the meantime, the other powerful catalyst for the oxidation of various compounds, horseradish peroxidase (HRP, E.C. 1.11.1.7), was subjected to basic studies on its activity and stability in various DESs [78,80]. A number of ChCl-based DESs were found to promote HPR activity and provide stabilization. Very different effects were observed when the DES composition and the molar ratio of HBD/HBA were varied, which was attributed to the changes in protein structure induced by the DESs [78]. Recently, the impact of a variety of betaine-based NADESs on HPR was comprehensively exploited by combing experiments and simulations, finally identifying an optimal beneficial system, Bet-Sor-Water (1:1:3) [80]. Both works studied conformational changes and proposed that an increment in α-helix secondary structure content contributes to enhanced stability and activity. The improvement mirrors the applicability of DESs as co-solvents along with enzyme preservatives against denaturation.
3. Hydrolase-Meditated API Syntheses in DESs
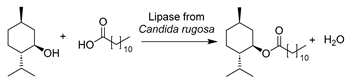
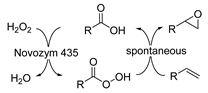
The pioneering research on biocatalysis in DESs commenced with the largely used cofactor-independent hydrolases (E.C. 3) [76]. The pioneering report was from the Kazlauskas group about transesterification catalyzed by various lipases in DESs [49]. They discovered the selected enzymes still remained active in DESs and resulted in enhanced reaction rates in DES–water mixtures. The components within DESs were found to be 20- to >600-fold less active toward enzyme denaturation and reaction when compared to individual forms due to the formed hydrogen network. This makes them suitable as solvents and promising alternatives for volatile organic solvents. Prior to the widespread use of DESs in biocatalysis, more efforts have been devoted to understanding the effects of diverse DESs on a variety of hydrolases including lipases (E.C. 3.1) [92,158,159,160,161], ß-glucosidases (E.C. 3.2) [161,162], epoxide hydrolases (E.C. 3.3) [163], proteases (E.C. 3.4) [164], cutinases (E.C. 3.1) [165], dehalogenases (E.C. 3.8) [166], and so on. As expected, DESs exerted very different effects on enzymes and biocatalytic processes as a function of their constituents, molar ratios, and water contents in reaction systems. Not only the activity but also the selectivity of enzymes can be drastically altered in the presence of specific DESs. Subsequently, the gained knowledge has been utilized to develop a variety of hydrolase-mediated biotransformations in DESs involving esterifications, transesterifications, hydrolysis, and even promiscuous aldol reactions. Also, due to the robustness and cofactor-independency, most hydrolases are used either in purified form or in crude isolated form. Pleasantly, large quantities of industrially marketable compounds have been produced [76], such as esters as flavor additives [167,168], glycolipids as surfactants [90,97,98,99,100,169], biodiesel [104,106,107,170,171], bio-based polyesters and polymers [99,101,172], and pharmaceutical and cosmeceutical intermediates [91,96,173,174,175,176,177,178].
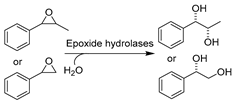
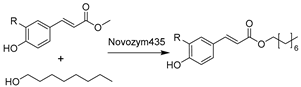
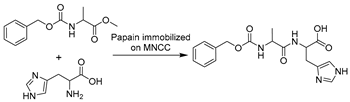
With respect to the synthesis of APIs catalyzed by hydrolase API in DESs the synthesis of chiral compounds remains of high interest due to their unlimited transformability. For example, epoxide hydrolases (EHs) are widely used for epoxide hydrolysis for the synthesis of enantiopure epoxides and vicinal diols, which are vital multipurpose building blocks for diverse pharmaceuticals [179]. In several studies, the application of various DESs was exploited to overcome the poor solubility of epoxides and non-enzymatic hydrolysis (Table 2, entries 1 and 2) [163,180,181]. Intriguingly, the regioselectivity of potato EHs toward the hydrolysis of a chiral (1,2)-trans-2-methylstyrene oxide was altered in the presence of 40 vol.% ChCl-Gly (1:2), while the KM was elevated 20-fold [163]. Likewise, in the hydrolysis of styrene oxide by mung bean EHs, an improvement of enantiopurity of (R)-1-phenyl-1,2-ethanediol was observed with the addition of 10–30 vol.% choline chloride/triethylene glycol (ChCl-TEG, 1:4), despite a general negative effect of studied DESs on enzymes (Table 2, entry 1) [180]. These findings substantiate that a delicate design of DESs or DES–water mixtures can provide a synergetic effect on enzymatic selectivity. However, the deleterious impact of DESs on enzymes is evident, propelling the generation and application of immobilized enzymes in DES-enriched systems. Exemplarily, the immobilized soybean epoxide hydrolase (SEH) on a new metal-organic framework (MOF) was applied in the asymmetric hydrolysis of 1,2-epoxyoctane to produce (R)-1,2-octanediol in the presence of 15 vol.% ChCl-U (1:2), leading to an improved yield of 41.4% and 81.2% ee (Table 2, entry 2) [181]. This work broadens the perspective to combine the use of DESs and enzyme immobilization to maximize biocatalytic efficiency. This claim was further demonstrated in the case study using Novozym435 (commercial immobilized lipase B from Candida antarctica, now renamed as Pseudozyma aphidis but remaining CalB here) for the lipophilization of phenolic acids in DES–water mixtures (Table 2, entry 3) [182]. As potential natural antioxidants, the lipophilic formulation shows improved antioxidant activity. Intriguingly, this contribution found that the introduction of a large amount of water is essential for quantitative conversion of phenolic compounds when compared to <2% conversion in pure DESs. This further explains why more and more DESs are often used as a co-solvent in buffer systems instead of in pure form to sustain enzyme hydration. This finding was also reflected in the biosynthesis of alanyl-histidine dipeptide catalyzed by papain immobilized on magnetic nanocrystalline cellulose in various ChCl-based DESs (Table 2, entry 4) [183]. The biological active dipeptide N-(benzyloxycarbonyl)-alanyl-histidine (Z-Ala-His), serving as a potential preventer of type II diabetes, was efficiently produced in ChCl-U (1:2) with a specific water content of 16.7 vol.%. Also, immobilization eliminates enzyme deactivation by DESs and leads to over 80% relative activity after seven batches [183]. In a nutshell, sustainable biosynthesis can be established by combining biocatalyst engineering and medium engineering.

Table 2.
Representative hydrolase-catalyzed syntheses of APIs in DESs.
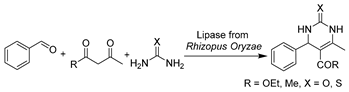
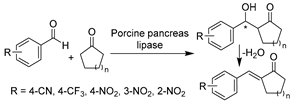
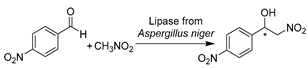
In addition to traditional hydrolysis and alcoholysis, lipase-catalyzed promiscuous reactions like Henry and aldol reactions were demonstrated using DESs as reaction media, leading to diverse valuable APIs [174,184,185,186]. A budding work was reported with lipase from Rhizopus oryzae for the synthesis of dihydropyrimidin-2(1H)-ones (DHPMs) moieties and derivatives (Table 2, entry 5) [174], which are commonly found in natural products and potent drugs such as anti-hypertensive agents, anti-carcinogenic agents, and anti-inflammatory agents. The use of ChCl-U (1:2) significantly increased yields and enabled the recyclability of DES and enzyme four times [174]. The continuation of lipase promiscuity extended to carbon–carbon bond-formation reactions. The aldol reactions were first set up with commercial crude porcine pancreas lipase (PPL) in ChCl-Gly (1:2) containing 5 vol.% water to produce a variety of chiral aldols as synthons for drugs (Table 2, entry 6) [184]. Similarly, the use of this neoteric solvent led to excellent conversion and allowed for the reuse of DES and enzyme, while the side products of unsaturated carbonyl compounds were significantly abated. Meanwhile, the DES–water mixture was first applied to lipase-catalyzed Henry and aza-Henry reactions for the synthesis of β-nitro alcohols, a starting material for the synthesis of various key API intermediates [185]. Within ChCl-Gly (1:2) containing 30 vol.% water, the enzyme activity was enhanced three-fold compared to pure water but without enantioselectivity (Table 2, entry 7) [185]. Nevertheless, the applicability of DESs for lipase-catalyzed syntheses was tentatively demonstrated, and further studies from the point of view of reaction media and catalyst type are indeed needed in the future. Later, it was found that tetraoctylammonium bromide-based hydrophobic DESs can be used to yield desired aldol products rather than unwanted olefin formation [186]. Furthermore, other types of enzymes like pepsin [187] and bovine pancreatic lipase [188] were explored in classic asymmetric aldol reactions in water-enriched DESs. These works display improved selectivity, showcasing the potential of using DESs as recoverable solvents with concentrated substrates for aldol catalysis.
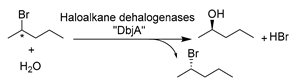
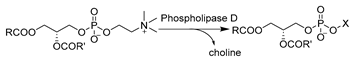
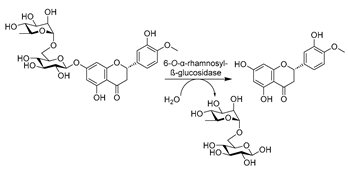
Other subclasses of hydrolases were systemically studied and used in DES–water mixtures to produce valuable compounds in a greener manner. For example, a budding work of haloalkane dehalogenases (HLDs) (E.C. 3.8) in DESs was reported, converting hazardous halogenated substrates to useful building blocks of chiral alcohols. Introducing 50 vol.% ChCl-EG (1:2) enabled improved enantioselectivity and compatibility, enhanced substrate loadings, and suppressed non-stereoselective hydrolysis for 2-bromopentane, which was attributed to the positive impact of EG (Table 2, entry 8) [166]. These observations demonstrated the potential of DES to be designed for specific biocatalytic applications. Driven by these encouraging results, attempts were also made to use another enzyme subclass in DESs, e.g., phospholipase D (PLD, E.C. 3.1.4.4), for the modification of naturally abundant phospholipids (PLs) such as phosphatidylcholine (PC). PLs present potential effective therapeutic agents for neurological pathologies such as Alzheimer’s dementia, Parkinson’s disease, and stress-related disorders [173,176]. In the transphosphatidylation of PC with L-serine to produce phosphatidylserine, alcohol-based DESs were proved to be superior to amine-based and acid-based DESs, and ChCl-EG in particular stands out as an optimal option for obtaining high yield and remarkable enzyme operation stability [173]. This was attributed to the fact that the introduction of DESs favors thermodynamic equilibrium. In addition, DESs were tailored to play multiple purposes as solvents and nucleophiles for choline substitution. This can be notably seen in the cases of using DES–buffer mixtures containing ChCl-Gly and ChCl-EG for the synthesis of phosphatidylglycerol (PG) and phosphatidylethyleneglycol (PEG) (Table 2, entry 9) [176]. This design led to high yield, simple product recovery, and higher product purity due to the avoidance of unwanted PC hydrolysis. The practicability of DESs was further exploited with glucosidases (E.C. 3.2.1) for the deglycosylation of flavonoids to produce diverse de-glycosylated flavanones. A representative compound, hesperetin, exhibits analgesic, anti-inflammatory, and antioxidant properties and is a synthon for dyes and sweeteners [189]. Various ChCl-based DESs with a wide range of water contents were applied as an alternative to DMSO to boost the solubility of flavonoids (Table 2, entry 10) [96]. Gratifyingly, the enzyme remained active in DES–aqueous mixtures with <40 vol.% DESs. DES individual components were proved to exert contrary effects on the enzyme, e.g., the beneficial effects of Gly and ethylene glycol (EG) as well as the deleterious effect of ChCl. On this basis, a plethora of opportunities for customizing DESs as enzyme-compatible solvents and substrate concentration enhancers can be envisaged. This was also demonstrated in the efficient enzymatic synthesis of daidzein by PLD in an optimized system of 30 vol.% ChCl-EG (1:2) [177]. Overall, these studies pave more research lines to establish efficient, mild, and environmentally friendly approaches for the production of various valuable APIs.
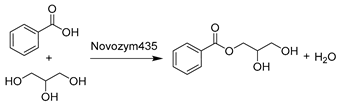
To further intensify biocatalytic processes, the first case study of using ChCl-Gly (1:2) in fed-batch and continuous packed-bed reactors as co-solvent and substrate for lipase-catalyzed esterification of benzoic acid and Gly was reported (Table 2, entry 11) [175]. The achieved α-MBG can be utilized as an emulsifier agent, plasticizer, and a building block in pharmaceuticals. In this case, the use of DES maximized the solubility of two substrates with different polarities, and this led to highly improved conversion in both modes. A higher enzyme stability and the reuse of reaction media were achieved [175]. Finally, it must be noted that the downstream processes in DESs are still not sufficiently addressed in the literature and clearly deserve more attention. The situation promoted the work to develop a product purification strategy for a DES-based reaction mixture in the lipase-catalyzed synthesis of (−)-menthol ester (Table 2, entry 12) [94]. The esterification with short-chain fatty acids is important for the extenuation of strong flavor for (−)-menthol and slows down its release in the application of ointments. The purification of a product and the re-use of an unconverted DES component [(−)-menthol] was accomplished via a thermal separation process due to the different boiling points of all reactants [94]. This success provides a new perspective on how to handle downstream processes when applying DESs for biocatalytic reactions.
4. Other Enzyme-Catalyzed API Syntheses in DESs
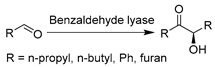
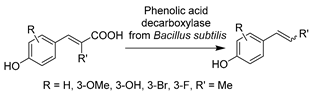
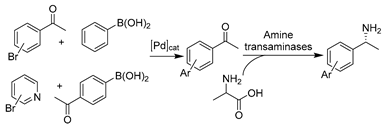
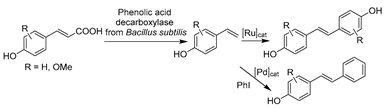
The advances in oxidoreductases (E.C. 1) and hydrolases (E.C. 3) have fueled the continuous development of biotransformations in DESs with other enzyme classes as well. In particular, the gained wealth of knowledge with prototypical ChCl-based DESs propelled their applications with amine transaminases (ATAs) (E.C. 2) and lyases (E.C. 4) to overcome the solubility dilemma of substrates. For example, benzaldehyde lyase (BAL) was initially explored in DESs for the enantioselective C-C bond carboligation (Table 3, entry 1) [190]. The afforded chiral α-hydroxy ketones are important building blocks for various pharmaceuticals (anti-depressants, fungicides, and anti-cancer drugs). Isolated BAL can effectively complete the reaction in 60 vol.% ChCl-Gly (1:2)-water mixture with high enantioselectivity and conversion, although it was markedly deactivated at a higher level of DESs (e.g., 70 vol.%) [190]. Surprisingly, the notorious DES, ChCl-U, is favorable for BAL, which calls for more research to fully understand the observed discrepancies before taking full advantage of DESs’ tunability in biocatalysis. Likewise, in the case of phenolic acid decarboxylase (PAD)-catalyzed decarboxylation, various classic ChCl-based DESs were employed to enhance the solubility of phenolic acids for the synthesis of diverse alkenes as starting materials for organic synthesis [33,85]. The exceptional solubilizing properties of ChCl-Gly (1:2) with 50 wt.% water content enabled conversion to the corresponding p-hydroxystyrene derivatives (>99%) at substrate concentrations of up to 300 mM (Table 3, entry 2) [85]. The observation of different substrate preferences dependent on the used solvents suggests that DESs have a specific effect on this isolated enzyme. Indeed, kinetic studies are necessary to gain a deeper insight into this remarkable variation in the selectivity of used DESs. The use of other enzyme class amine transaminases (E.C. 2) was also explored in ChCl-based DESs as an alternative to volatile and toxic organic solvents (Table 3, entry 3) [191]. Pleasantly, the ATAs lysate exhibited favorable stability in the presence of up to 75 wt.% ChCl-Gly (1:2). This contribution also disclosed a foreseeable possible combination of medium engineering and protein engineering to form a circular toolkit for efficient biotransformations. In the following studies, both PADs and ATAs have been successfully integrated with chemo-catalyzed steps to accomplish elegant tandem catalysis in the presence of DESs, which will be discussed in Section 5.

Table 3.
Representative syntheses of APIs catalyzed by the enzymes from other classes in DESs.
5. Chemo-Enzymatic Cascade and Miscellaneous Synthetic Routes in DESs
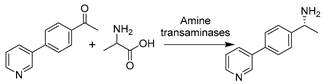
DESs may not only be used for one-step enzymatic biotransformations but also find widespread applications in chemo-enzymatic multi-step reactions. The combination of chemo catalysis and biocatalysis is a challenging but forward-looking topic in synthetic chemistry. The combination of the unique selectivity of enzymes and the wide range of applications of chemical catalysts allows for many novel processes, with reduced waste formation due to the set-up of fewer downstream units. As a neoteric and greener class of non-conventional media, the use of DESs could address the conflicts between chemo-enzymatic cascades regarding different optimal reaction conditions for chemical and biological catalysts while enhancing substrate loadings [192,193]. Gradually, more and more DESs have been applied as sustainable alternatives to toxic ILs and organic solvents. A number of chemo-enzymatic cascades have been established in DESs with various enzymes such as lipases [102,194,195], KREDs [196,197], ATAs [191,198], and PADs [33,199,200] and for the synthesis of valuable compounds, as well as intermediates, as potential APIs.
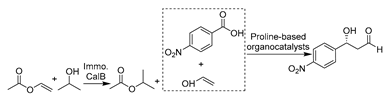
The first chemo-enzymatic cascade in DESs was established with immobilized lipase (CalB) for in situ acetaldehyde formation and proline-based organocatalysts for aldehyde-acetaldehyde aldol reactions (Table 4, entry 1) [194]. The presence of water was detrimental to aldol catalysis regarding selectivity, and the use of pure ChCl-Gly (1:2) led to efficient reaction rates. The aldol addition products were extracted to the organic phase before being subjected to sodium borohydride-catalyzed reduction to the corresponding 1,3-diols. The remaining DES and enzymes were reused for up to six cycles without any activity loss. A variety of aromatic 1,3-diols were synthesized with high selectivity, which can act as precursors for bioactive agents like anti-HIV agents [201]. This work developed a new, greener approach to synthesizing highly valuable optically building blocks and demonstrated the integrative concept with straightforward product recovery and catalyst reuse. Inspired by this promising report, the chemo-enzymatic epoxidation of alkenes with CalB was systematically investigated in various ChCl-based DESs. The cascade starts with lipase-catalyzed perhydrolysis of a carboxylic acid, with H2O2 forming the corresponding peracid, followed by a non-enzymatic Prileshajev reaction, producing epoxides as important building blocks (Table 4, entry 2) [195]. Notably, excellent stabilization of enzymes was observed in polyol-based DESs like ChCl-sorbitol (Sorb, 1:2) due to the preservation of structural integrity. This contribution further illustrates the potential of using DESs as a replacement for toxic non-aqueous media.

Table 4.
Representative chemo-enzymatic cascade for the synthesis of APIs in DESs.
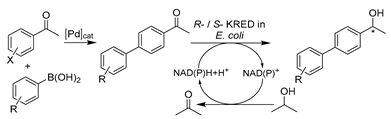
Recently, oxidoreductase-involved chemo-enzymatic tandem catalysis in DESs has been on the rise. Unlike lipases, enzymes are mostly combined with metal-catalyzed steps to synthesize substituted-chiral alcohols for pharmaceutical applications. The first example was reported by Cicco et al., involving a ruthenium (Ru)-catalyzed isomerization of racemic allylic alcohols combined with a KRED-catalzyed enantioselective reduction (Table 4, entry 3) [196]. Therein, KRED showed good catalytic performance (>99% conversion and up to >99% ee) in ad hoc DES–water mixtures including 20–50 wt.% ChCl-Gly (1:2) and ChCl-Sorb (1:1). Despite the slightly decreased enzyme stability compared to a pure aqueous system, a higher percentage of DESs favors enantioselectivity improvement. Benefiting from the use of DESs and the exceptional catalytic performance of enzymes, the one-pot cascade in both sequential and concurrent modes was successfully performed in 50 wt.% ChCl-Gly, achieving moderate conversion and remarkable enantiomeric excess (>99%) for all applied substrates [196]. This report opens the floodgate for the design of enzyme/reaction-compatible DESs to fulfill various biocatalytic demands for greener sustainable chemistry. Motivated by this pioneering example, another cascade was established in DES–water mixtures with palladium (Pd)-catalyzed Suzuki-cross coupling followed by an ADH-mediated reduction (Table 4, entry 4) [197]. After extensive optimization of the two catalytic steps, the cascade was efficiently performed in a two-step one-pot manner with improved substrate loadings. The connection with two ADHs overexpressed in E. coli with opposite enantioselectivity allows for the synthesis of various enantiomerically pure (R)- and (S)- biaryl alcohols [197]. This work further underlines the benefits of applying DESs in both chemocatalysis and biocatalysis. Subsequently, the stability of amine transaminases (ATAs) (E.C. 2) in DES-enriched aqueous media allows for the development of a chemo-enzymatic one-pot cascade together with Pd-catalyzed Suzuki-cross coupling (Table 4, entry 5) [191]. The produced biaryl-substituted amines are indispensable building blocks for pharmaceuticals (e.g., valsartane and odanacatib) [202]. Similarly, a higher DES content is favorable for the chemo-step while harmful for the enzymatic-step. Therefore, the cascade is conducted in a sequential mode. The Suzuki cross-coupling was completed with high substrate concentrations in 75 wt.% ChCl-Gly and diluted to 10 wt.% ChCl-Gly to be compatible with enzymatic transamination, leading to the obtainment of various enantiopure biaryl amines. Despite this progress, it remains clear that enzyme stability is one of the main limitations for bridging chemo- and bio- catalysis in DESs. Therefore, most established tandem catalysis was in one-pot two-step sequential mode, even when applying whole cells. For example, E. coli cells harboring ω-transaminase and L-alanine dehydrogenase were combined with sulfonated tin-based solid acid (Sn-SS) in 20 vol.% ChCl-EG for the efficient production of furfurylamine from sugarcane bagasse [198]. This strategy exhibited potential application for converting renewable biomass into valuable chemicals in the benign DES–water systems. In this sense, the unique extractive properties of NADESs are fully utilized in the valorization of agricultural wastes, where DESs serve multiple functions as a solvent, extractant, and substrate [102]. It is foreseeable that more and more eutectic solvents will be broadly applied in catalysis applied to multi-step integrated reactions.
Recently, the combination of DESs and phenolic acid decarboxylases (PADs) became attractive in tandem-catalytic processes, mainly to tackle the incompatibility of catalysts and solubility obstacles [33,199,200]. In particular, researchers attempted to combine PAD-catalyzed decarboxylation with Pd-catalyzed Heck coupling or Ru-catalyzed metathesis using DESs as solvents for the synthesis of stilbenes (Table 4, entry 6). Many stilbenes have biological properties (anti-oxidant, anti-inflammatory, anti-diabetic, and anti-aging) and are currently being investigated for use as APIs in cancer-preventive drugs [203]. In an integrated two-step continuous flow process, PAD encapsulated in alginate beads and heterogeneous Pd-catalyst were used in two individual steps in DES–water mixtures containing 50 vol.% and 20 vol.% ChCl-Gly (1:2), respectively [200]. This approach avoided the reciprocal poisoning of catalysts and finally led to the efficient synthesis of (E)-4-hydroxy-stilbene with enhanced substrate concentrations. However, the employment of DESs does not always work perfectly. For example, in the one-pot sequential synthetic route in the presence of DESs, PAD showed a strong deactivation impact on transition-metal complexes, resulting in very low overall conversions for the synthesis of aromatic olefins [199]. Immobilized enzymes were employed to avoid contact between catalysts but confronted the strong elution power of choline, resulting in no product for the first step. In the end, the use of aqueous micellar solutions was proved better than DES–water systems. Likewise, pure DESs showed outstanding solubilizing capacity, but they were not applicable for the cascade in the synthesis of bio-based styrene alternatives [33]. Nevertheless, using DESs as a replacement for hazardous organic solvents to deal with the challenges of substrate insolubility and solvent incompatibility is still a major step toward a broad application of chemo-enzymatic reactions.
6. Summary and Future Perspectives
Since the characterization of the first DESs decades ago, the field has flourished and many applications have been reported for a broad number of DESs, ranging from direct solvent use to other “finer” applications like performance additives, (co)catalysts, and therapeutic or reactive agents. The large tunability of DESs gives options to design the most proper solvent for each application. Among them, their use in biocatalysis has been broadly studied, with applications in most of the enzyme classes. In general terms, works dealing with DESs and enzymes are conceived when higher substrate loadings are needed (e.g., intensified processes), while keeping enzyme compatibility, higher (enantio)selectivity, and sustainability at the same time. Likewise, works focusing on the understanding of the behavior of enzymes in DES–water mixtures have started to appear as well, enabling the design of more compatible solvent systems for combined enzyme catalytic processes.
Other aspects, however, have not attracted sufficient interest from the community, and it is important that they are addressed in the coming future. One point herein is the set-up of efficient downstream processing alternatives that can enable DES reuse while leading to purified products at the same time. That aspect is important in terms of economics and the sustainability of the process. Likewise, considerations about the biodegradability of the used DESs need to be further assessed. Currently, little is known about the possible fate of DESs once used in several reaction cycles. The results on the way solvents can be ultimately treated are mandatory to the establishment of a sustainable strategy for their use. As an example, integrating used DESs into wastewater treatment plants (e.g., through further dilution) would be a plausible option, provided that the wastewater microorganisms could cope with it and that large effluents would not compromise the process economics. Given the tunability of DESs, it may be foreseeable that customized DESs covering all aspects (sustainability, biodegradability, enzyme compatibility, and substrate solubilizing) may be found in future research plans. Based on progress in all of these considerations, the upscale of biocatalytic processes in DESs could be envisaged in the future.
Author Contributions
Conceptualization, N.Z. and S.K.; writing—original draft preparation, N.Z.; writing—review and editing, P.D.d.M. and S.K.; project administration, S.K.; funding acquisition, S.K. All authors have read and agreed to the published version of the manuscript.
Funding
This project has received funding from the EU’s Horizon Europe Doctoral Network Program under the Marie Skłodowska-Curie grant agreement no. 101072731. The authors acknowledge the financial support from Deutsche Forschungsgemeinschaft (DFG, German Research Foundation) under grant no. 391127961.
Conflicts of Interest
Author Pablo Domínguez de María is the CEO & Founder of the company Sustainable Momentum SL. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
References
- Straathof, A.J. Transformation of biomass into commodity chemicals using enzymes or cells. Chem. Rev. 2014, 114, 1871–1908. [Google Scholar] [CrossRef] [PubMed]
- Truppo, M.D. Biocatalysis in the Pharmaceutical Industry: The Need for Speed. ACS Med. Chem. Lett. 2017, 8, 476–480. [Google Scholar] [CrossRef] [PubMed]
- Sun, H.; Zhang, H.; Ang, E.L.; Zhao, H. Biocatalysis for the synthesis of pharmaceuticals and pharmaceutical intermediates. Bioorg. Med. Chem. 2018, 26, 1275–1284. [Google Scholar] [CrossRef] [PubMed]
- de Gonzalo, G.; Alcantara, A.R.; Domínguez de María, P.; Sanchez-Montero, J.M. Biocatalysis for the asymmetric synthesis of Active Pharmaceutical Ingredients (APIs): This time is for real. Expert Opin. Drug Discov. 2022, 17, 1159–1171. [Google Scholar] [CrossRef]
- Buller, R.; Lutz, S.; Kazlauskas, R.J.; Snajdrova, R.; Moore, J.C.; Bornscheuer, U.T. From nature to industry: Harnessing enzymes for biocatalysis. Science 2023, 382, eadh8615. [Google Scholar] [CrossRef]
- Rossino, G.; Robescu, M.S.; Licastro, E.; Tedesco, C.; Martello, I.; Maffei, L.; Vincenti, G.; Bavaro, T.; Collina, S. Biocatalysis: A smart and green tool for the preparation of chiral drugs. Chirality 2022, 34, 1403–1418. [Google Scholar] [CrossRef]
- Wang, X.D.; Saba, T.; Yiu, H.H.P.; Howe, R.F.; Anderson, J.A.; Shi, J.F. Cofactor NAD(P)H Regeneration Inspired by Heterogeneous Pathways. Chem 2017, 2, 621–654. [Google Scholar] [CrossRef]
- Lewis, R.D.; France, S.P.; Martinez, C.A. Emerging Technologies for Biocatalysis in the Pharmaceutical Industry. ACS Catal. 2023, 13, 5571–5577. [Google Scholar] [CrossRef]
- Swati; Pathania, S.; Rawal, R.K. Current prospective of green chemistry in the pharmaceutical industry. In Green Sustainable Process for Chemical and Environmental Engineering and Science; Elsevier: Amsterdam, The Netherlands, 2023; pp. 419–450. [Google Scholar]
- De Santis, P.; Meyer, L.-E.; Kara, S. The rise of continuous flow biocatalysis–fundamentals, very recent developments and future perspectives. React. Chem. Eng. 2020, 5, 2155–2184. [Google Scholar] [CrossRef]
- Tamborini, L.; Fernandes, P.; Paradisi, F.; Molinari, F. Flow Bioreactors as Complementary Tools for Biocatalytic Process Intensification. Trends Biotechnol. 2018, 36, 73–88. [Google Scholar] [CrossRef]
- Žnidaršič-Plazl, P. Biocatalytic process intensification via efficient biocatalyst immobilization, miniaturization, and process integration. Curr. Opin. Green Sustain. Chem. 2021, 32, 100546. [Google Scholar] [CrossRef]
- Rocha, R.A.; Speight, R.E.; Scott, C. Engineering Enzyme Properties for Improved Biocatalytic Processes in Batch and Continuous Flow. Org. Process Res. Dev. 2022, 26, 1914–1924. [Google Scholar] [CrossRef]
- Carrea, G.; Riva, S. Medium Engineering. In Asymmetric Organic Synthesis with Enzymes; John Wiley & Sons: Hoboken, NJ, USA, 2008; pp. 1–20. [Google Scholar]
- Sheldon, R.A.; Pereira, P.C. Biocatalysis engineering: The big picture. Chem. Soc. Rev. 2017, 46, 2678–2691. [Google Scholar] [CrossRef]
- Prat, D.; Hayler, J.; Wells, A. A survey of solvent selection guides. Green Chem. 2014, 16, 4546–4551. [Google Scholar] [CrossRef]
- Henderson, R.K.; Jiménez-González, C.; Constable, D.J.C.; Alston, S.R.; Inglis, G.G.A.; Fisher, G.; Sherwood, J.; Binks, S.P.; Curzons, A.D. Expanding GSK’s solvent selection guide—Embedding sustainability into solvent selection starting at medicinal chemistry. Green Chem. 2011, 13, 854–862. [Google Scholar] [CrossRef]
- France, S.P.; Lewis, R.D.; Martinez, C.A. The Evolving Nature of Biocatalysis in Pharmaceutical Research and Development. JACS Au 2023, 3, 715–735. [Google Scholar] [CrossRef]
- Hessel, V.; Tran, N.N.; Asrami, M.R.; Tran, Q.D.; Van Duc Long, N.; Escribà-Gelonch, M.; Tejada, J.O.; Linke, S.; Sundmacher, K. Sustainability of green solvents—Review and perspective. Green Chem. 2022, 24, 410–437. [Google Scholar] [CrossRef]
- Holtmann, D.; Hollmann, F. Is water the best solvent for biocatalysis? Mol. Catal. 2022, 517, 112035. [Google Scholar] [CrossRef]
- Hollmann, F.; Opperman, D.J.; Paul, C.E. Biocatalytic Reduction Reactions from a Chemist’s Perspective. Angew. Chem. Int. Ed. 2021, 60, 5644–5665. [Google Scholar] [CrossRef]
- Krell, C.; Schreiber, R.; Hueber, L.; Sciascera, L.; Zheng, X.M.; Clarke, A.; Haenggi, R.; Parmentier, M.; Baguia, H.; Rodde, S.; et al. Strategies to Tackle the Waste Water from α-Tocopherol-Derived Surfactant Chemistry. Org. Process Res. Dev. 2021, 25, 900–915. [Google Scholar] [CrossRef]
- Ni, Y.; Holtmann, D.; Hollmann, F. How Green is Biocatalysis? To Calculate is To Know. ChemCatChem 2014, 6, 930–943. [Google Scholar] [CrossRef]
- Sheldon, R.A. Biocatalysis, solvents, and green metrics in sustainable chemistry. In Biocatalysis in Green Solvents; Lozano, P., Ed.; Elsevier Inc.: Amsterdam, The Netherlands, 2022; pp. 1–22. [Google Scholar]
- Tieves, F.; Tonin, F.; Fernández-Fueyo, E.; Robbins, J.M.; Bommarius, B.; Bommarius, A.S.; Alcalde, M.; Hollmann, F. Energising the E-factor: The E+-factor. Tetrahedron 2019, 75, 1311–1314. [Google Scholar] [CrossRef]
- Domínguez de María, P.; Kara, S.; Gallou, F. Biocatalysis in Water or in Non-Conventional Media? Adding the CO2 Production for the Debate. Molecules 2023, 28, 6452. [Google Scholar] [CrossRef]
- Onken, U.; Koettgen, A.; Scheidat, H.; Schueepp, P.; Gallou, F. Environmental Metrics to Drive a Cultural Change: Our Green Eco-Label. Chimia 2019, 73, 730–736. [Google Scholar] [CrossRef]
- Sheldon, R.A.; Bode, M.L.; Akakios, S.G. Metrics of green chemistry: Waste minimization. Curr. Opin. Green Sustain. Chem. 2022, 33, 100569. [Google Scholar] [CrossRef]
- Domínguez de María, P.; Hollmann, F. On the (Un)greenness of Biocatalysis: Some Challenging Figures and Some Promising Options. Front. Microbiol. 2015, 6, 1257. [Google Scholar] [CrossRef]
- Ciriminna, R.; Pagliaro, M. Green Chemistry in the Fine Chemicals and Pharmaceutical Industries. Org. Process Res. Dev. 2013, 17, 1479–1484. [Google Scholar] [CrossRef]
- Nieto, S.; Villa, R.; Donaire, A.; Lozano, P. Nonconventional biocatalysis: From organic solvents to green solvents. In Biocatalysis in Green Solvents; Lozano, P., Ed.; Academic Press: Cambridge, MA, USA, 2022; pp. 23–55. [Google Scholar]
- Illanes, A. CHAPTER 3: Biocatalysis in Organic Media. In White Biotechnology for Sustainable Chemistry; Coelho, M.A., Ribeiro, B.D., Eds.; Green Chemistry Series; The Royal Society of Chemistry: London, UK, 2015; pp. 36–51. [Google Scholar]
- Petermeier, P.; Bittner, J.P.; Müller, S.; Byström, E.; Kara, S. Design of a green chemoenzymatic cascade for scalable synthesis of bio-based styrene alternatives. Green Chem. 2022, 24, 6889–6899. [Google Scholar] [CrossRef]
- Klibanov, A.M. Enzymatic catalysis in anhydrous organic solvents. Trends Biochem. Sci. 1989, 14, 141–144. [Google Scholar] [CrossRef]
- Zaks, A.; Klibanov, A.M. Enzymatic Catalysis in Nonaqueous Solvents. J. Biol. Chem. 1988, 263, 3194–3201. [Google Scholar] [CrossRef]
- Domínguez de María, P. Biocatalysis, sustainability, and industrial applications: Show me the metrics. Curr. Opin. Green Sustain. Chem. 2021, 31, 100514. [Google Scholar] [CrossRef]
- Calvo-Flores, F.G.; Monteagudo-Arrebola, M.J.; Dobado, J.A.; Isac-Garcia, J. Green and Bio-Based Solvents. Top. Curr. Chem. 2018, 376, 18. [Google Scholar] [CrossRef]
- Abbott, A.P.; Capper, G.; Davies, D.L.; Rasheed, R.K.; Tambyrajah, V. Novel solvent properties of choline chloride/urea mixtures. Chem. Commun. 2003, 39, 70–71. [Google Scholar] [CrossRef]
- Smith, E.L.; Abbott, A.P.; Ryder, K.S. Deep eutectic solvents (DESs) and their applications. Chem. Rev. 2014, 114, 11060–11082. [Google Scholar] [CrossRef]
- Hansen, B.B.; Spittle, S.; Chen, B.; Poe, D.; Zhang, Y.; Klein, J.M.; Horton, A.; Adhikari, L.; Zelovich, T.; Doherty, B.W.; et al. Deep Eutectic Solvents: A Review of Fundamentals and Applications. Chem. Rev. 2021, 121, 1232–1285. [Google Scholar] [CrossRef]
- Abbott, A.P.; Barron, J.C.; Ryder, K.S.; Wilson, D. Eutectic-based ionic liquids with metal-containing anions and cations. Chem. Eur. J. 2007, 13, 6495–6501. [Google Scholar] [CrossRef]
- Abranches, D.O.; Martins, M.A.R.; Silva, L.P.; Schaeffer, N.; Pinho, S.P.; Coutinho, J.A.P. Phenolic hydrogen bond donors in the formation of non-ionic deep eutectic solvents: The quest for type V DES. Chem. Commun. 2019, 55, 10253–10256. [Google Scholar] [CrossRef]
- Paiva, A.; Craveiro, R.; Aroso, I.; Martins, M.; Reis, R.L.; Duarte, A.R.C. Natural Deep Eutectic Solvents—Solvents for the 21st Century. ACS Sustain. Chem. Eng. 2014, 2, 1063–1071. [Google Scholar] [CrossRef]
- van Osch, D.J.G.P.; Dietz, C.H.J.T.; van Spronsen, J.; Kroon, M.C.; Gallucci, F.; van Sint Annaland, M.; Tuinier, R. A Search for Natural Hydrophobic Deep Eutectic Solvents Based on Natural Components. ACS Sustain. Chem. Eng. 2019, 7, 2933–2942. [Google Scholar] [CrossRef]
- Longeras, O.; Gautier, A.; Ballerat-Busserolles, K.; Andanson, J.-M. Deep Eutectic Solvent with Thermo-Switchable Hydrophobicity. ACS Sustain. Chem. Eng. 2020, 8, 12516–12520. [Google Scholar] [CrossRef]
- van Osch, D.J.G.P.; Dietz, C.H.J.T.; Warrag, S.E.E.; Kroon, M.C. The Curious Case of Hydrophobic Deep Eutectic Solvents: A Story on the Discovery, Design, and Applications. ACS Sustain. Chem. Eng. 2020, 8, 10591–10612. [Google Scholar] [CrossRef]
- Cao, J.; Su, E. Hydrophobic deep eutectic solvents: The new generation of green solvents for diversified and colorful applications in green chemistry. J. Clean. Prod. 2021, 314, 127965. [Google Scholar] [CrossRef]
- Li, D. Natural deep eutectic solvents in phytonutrient extraction and other applications. Front. Plant Sci. 2022, 13, 1004332. [Google Scholar] [CrossRef] [PubMed]
- Gorke, J.T.; Srienc, F.; Kazlauskas, R.J. Hydrolase-catalyzed biotransformations in deep eutectic solvents. Chem. Commun. 2008, 10, 1235–1237. [Google Scholar] [CrossRef]
- Singh, M.B.; Kumar, V.S.; Chaudhary, M.; Singh, P. A mini review on synthesis, properties and applications of deep eutectic solvents. J. Indian Chem. Soc. 2021, 98, 100210. [Google Scholar] [CrossRef]
- Abo-Hamad, A.; Hayyan, M.; AlSaadi, M.A.; Hashim, M.A. Potential applications of deep eutectic solvents in nanotechnology. Chem. Eng. J. 2015, 273, 551–567. [Google Scholar] [CrossRef]
- Cheng, F.-F.; Zhu, J.-N.; Zhao, M.-Y.; Ma, Z.-J.; Xiong, W.-W. Preparing transition metal-organic frameworks based on oxalate-sulfate anions in deep eutectic solvents. J. Solid State Chem. 2019, 278, 120904. [Google Scholar] [CrossRef]
- Majid, M.F.; Mohd Zaid, H.F.; Kait, C.F.; Jumbri, K.; Yuan, L.C.; Rajasuriyan, S. Futuristic advance and perspective of deep eutectic solvent for extractive desulfurization of fuel oil: A review. J. Mol. Liq. 2020, 306, 112870. [Google Scholar] [CrossRef]
- Hong, S.; Lian, H.; Sun, X.; Pan, D.; Carranza, A.; Pojman, J.A.; Mota-Morales, J.D. Zinc-based deep eutectic solvent-mediated hydroxylation and demethoxylation of lignin for the production of wood adhesive. RSC Adv. 2016, 6, 89599–89608. [Google Scholar] [CrossRef]
- Hong, S.; Yuan, Y.; Yang, Q.; Chen, L.; Deng, J.; Chen, W.; Lian, H.; Mota-Morales, J.D.; Liimatainen, H. Choline chloride-zinc chloride deep eutectic solvent mediated preparation of partial O-acetylation of chitin nanocrystal in one step reaction. Carbohydr. Polym. 2019, 220, 211–218. [Google Scholar] [CrossRef]
- Di Carmine, G.; Abbott, A.P.; D’Agostino, C. Deep eutectic solvents: Alternative reaction media for organic oxidation reactions. React. Chem. Eng. 2021, 6, 582–598. [Google Scholar] [CrossRef]
- Perna, F.M.; Vitale, P.; Capriati, V. Deep eutectic solvents and their applications as green solvents. Curr. Opin. Green Sustain. Chem. 2020, 21, 27–33. [Google Scholar] [CrossRef]
- Radović, M.; Hok, L.; Panić, M.; Cvjetko Bubalo, M.; Vianello, R.; Vinković, M.; Radojčić Redovniković, I. Deep eutectic solvents as a stabilising medium for NAD coenzyme: Unravelling the mechanism behind coenzyme stabilisation effect. Green Chem. 2022, 24, 7661–7674. [Google Scholar] [CrossRef]
- Panić, M.; Bubalo, M.C.; Redovniković, I.R. Designing a biocatalytic process involving deep eutectic solvents. J. Chem. Technol. Biotechnol. 2021, 96, 14–30. [Google Scholar] [CrossRef]
- Liu, Y.; Friesen, J.B.; McAlpine, J.B.; Lankin, D.C.; Chen, S.N.; Pauli, G.F. Natural Deep Eutectic Solvents: Properties, Applications, and Perspectives. J. Nat. Prod. 2018, 81, 679–690. [Google Scholar] [CrossRef] [PubMed]
- Toledo, M.L.; Pereira, M.M.; Freire, M.G.; Silva, J.P.A.; Coutinho, J.A.P.; Tavares, A.P.M. Laccase Activation in Deep Eutectic Solvents. ACS Sustain. Chem. Eng. 2019, 7, 11806–11814. [Google Scholar] [CrossRef]
- Zaib, Q.; Eckelman, M.J.; Yang, Y.; Kyung, D. Are deep eutectic solvents really green? A life-cycle perspective. Green Chem. 2022, 24, 7924–7930. [Google Scholar] [CrossRef]
- Zhang, Q.; De Oliveira Vigier, K.; Royer, S.; Jerome, F. Deep eutectic solvents: Syntheses, properties and applications. Chem. Soc. Rev. 2012, 41, 7108–7146. [Google Scholar] [CrossRef]
- Liu, P.; Hao, J.W.; Mo, L.P.; Zhang, Z.H. Recent advances in the application of deep eutectic solvents as sustainable media as well as catalysts in organic reactions. RSC Adv. 2015, 5, 48675–48704. [Google Scholar] [CrossRef]
- Domínguez de María, P.; Guajardo, N.; González-Sabín, J. Recent granted patents related to Deep Eutectic Solvents. Curr. Opin. Green Sustain. Chem. 2022, 38, 100712. [Google Scholar] [CrossRef]
- Kalhor, P.; Ghandi, K. Deep Eutectic Solvents for Pretreatment, Extraction, and Catalysis of Biomass and Food Waste. Molecules 2019, 24, 4012. [Google Scholar] [CrossRef] [PubMed]
- Xu, P.; Zheng, G.W.; Zong, M.H.; Li, N.; Lou, W.Y. Recent progress on deep eutectic solvents in biocatalysis. Bioresour. Bioprocess. 2017, 4, 34. [Google Scholar] [CrossRef]
- Pätzold, M.; Siebenhaller, S.; Kara, S.; Liese, A.; Syldatk, C.; Holtmann, D. Deep Eutectic Solvents as Efficient Solvents in Biocatalysis. Trends Biotechnol. 2019, 37, 943–959. [Google Scholar] [CrossRef]
- Paul, C.E.; Gotor-Fernández, V. Applied biocatalysis in deep eutectic solvents. In Biocatalysis in Green Solvents; Lozano, P., Ed.; Elsevier Inc.: Amsterdam, The Netherlands, 2022; pp. 467–510. [Google Scholar]
- Domínguez de María, P.; Guajardo, N.; Kara, S. Enzyme Catalysis: In DES, with DES, and in the Presence of DES. In Deep Eutectic Solvents: Synthesis, Properties, and Applications; Ramón, D.J., Guillena, G., Eds.; Wiley-VCH Verlag GmbH: Weinheim, Germany, 2019; pp. 257–271. [Google Scholar]
- Zhang, N.; Steininger, F.; Meyer, L.-E.; Koren, K.; Kara, S. Can Deep Eutectic Solvents Sustain Oxygen-Dependent Bioprocesses?—Measurements of Oxygen Transfer Rates. ACS Sustain. Chem. Eng. 2021, 9, 8347–8353. [Google Scholar] [CrossRef]
- Guajardo, N.; Domínguez de María, P.; Ahumada, K.; Schrebler, R.A.; Ramírez-Tagle, R.; Crespo, F.A.; Carlesi, C. Water as Cosolvent: Nonviscous Deep Eutectic Solvents for Efficient Lipase-Catalyzed Esterifications. ChemCatChem 2017, 9, 1393–1396. [Google Scholar] [CrossRef]
- Weng, L.; Toner, M. Janus-faced role of water in defining nanostructure of choline chloride/glycerol deep eutectic solvent. Phys. Chem. Chem. Phys. 2018, 20, 22455–22462. [Google Scholar] [CrossRef] [PubMed]
- Hammond, O.S.; Bowron, D.T.; Edler, K.J. The Effect of Water upon Deep Eutectic Solvent Nanostructure: An Unusual Transition from Ionic Mixture to Aqueous Solution. Angew. Chem. Int. Ed. 2017, 56, 9782–9785. [Google Scholar] [CrossRef] [PubMed]
- Hassani, F.Z.I.M.; Lavandera, I.; Kreit, J. Stabilizing Effects of Deep Eutectic Solvents on Alcohol Dehydrogenase Mediated Systems. Int. J. Biol. Biomed. Eng. 2016, 10, 829. [Google Scholar]
- Durand, E.; Lecomte, J.; Villeneuve, P. Deep eutectic solvents: Synthesis, application, and focus on lipase-catalyzed reactions. Eur. J. Lipid Sci. Technol. 2013, 115, 379–385. [Google Scholar] [CrossRef]
- Daneshjou, S.; Khodaverdian, S.; Dabirmanesh, B.; Rahimi, F.; Daneshjoo, S.; Ghazi, F.; Khajeh, K. Improvement of chondroitinases ABCI stability in natural deep eutectic solvents. J. Mol. Liq. 2017, 227, 21–25. [Google Scholar] [CrossRef]
- Wu, B.P.; Wen, Q.; Xu, H.; Yang, Z. Insights into the impact of deep eutectic solvents on horseradish peroxidase: Activity, stability and structure. J. Mol. Catal. B Enzym. 2014, 101, 101–107. [Google Scholar] [CrossRef]
- Papadopoulou, A.A.; Efstathiadou, E.; Patila, M.; Polydera, A.C.; Stamatis, H. Deep Eutectic Solvents as Media for Peroxidation Reactions Catalyzed by Heme-Dependent Biocatalysts. Ind. Eng. Chem. Res. 2016, 55, 5145–5151. [Google Scholar] [CrossRef]
- Gajardo-Parra, N.F.; Meneses, L.; Duarte, A.R.C.; Paiva, A.; Held, C. Assessing the Influence of Betaine-Based Natural Deep Eutectic Systems on Horseradish Peroxidase. ACS Sustain. Chem. Eng. 2022, 10, 12873–12881. [Google Scholar] [CrossRef]
- Bittner, J.P.; Zhang, N.; Huang, L.; Domínguez de María, P.; Jakobtorweihen, S.; Kara, S. Impact of deep eutectic solvents (DESs) and individual DES components on alcohol dehydrogenase catalysis: Connecting experimental data and molecular dynamics simulations. Green Chem. 2022, 24, 1120–1131. [Google Scholar] [CrossRef]
- Gajardo-Parra, N.F.; Rodríguez, G.; Arroyo-Avirama, A.F.; Veliju, A.; Happe, T.; Canales, R.I.; Sadowski, G.; Held, C. Impact of Deep Eutectic Solvents on Kinetics and Folding Stability of Formate Dehydrogenase. Processes 2023, 11, 2815. [Google Scholar] [CrossRef]
- Huang, L.; Bittner, J.P.; Domínguez de María, P.; Jakobtorweihen, S.; Kara, S. Modeling Alcohol Dehydrogenase Catalysis in Deep Eutectic Solvent/Water Mixtures. ChemBioChem 2020, 21, 811–817. [Google Scholar] [CrossRef] [PubMed]
- Milano, F.; Giotta, L.; Guascito, M.R.; Agostiano, A.; Sblendorio, S.; Valli, L.; Perna, F.M.; Cicco, L.; Trotta, M.; Capriati, V. Functional Enzymes in Nonaqueous Environment: The Case of Photosynthetic Reaction Centers in Deep Eutectic Solvents. ACS Sustain. Chem. Eng. 2017, 5, 7768–7776. [Google Scholar] [CrossRef]
- Schweiger, A.K.; Ríos-Lombardía, N.; Winkler, C.K.; Schmidt, S.; Morís, F.; Kroutil, W.; González-Sabín, J.; Kourist, R. Using Deep Eutectic Solvents to Overcome Limited Substrate Solubility in the Enzymatic Decarboxylation of Bio-Based Phenolic Acids. ACS Sustain. Chem. Eng. 2019, 7, 16364–16370. [Google Scholar] [CrossRef]
- Palmelund, H.; Andersson, M.P.; Asgreen, C.J.; Boyd, B.J.; Rantanen, J.; Lobmann, K. Tailor-made solvents for pharmaceutical use? Experimental and computational approach for determining solubility in deep eutectic solvents (DES). Int. J. Pharm. X 2019, 1, 100034. [Google Scholar] [CrossRef]
- Chen, Y.F.; Ai, N.; Li, G.H.; Shan, H.F.; Cui, Y.H.; Deng, D.S. Solubilities of Carbon Dioxide in Eutectic Mixtures of Choline Chloride and Dihydric Alcohols. J. Chem. Eng. Data 2014, 59, 1247–1253. [Google Scholar] [CrossRef]
- Arnodo, D.; Maffeis, E.; Marra, F.; Nejrotti, S.; Prandi, C. Combination of Enzymes and Deep Eutectic Solvents as Powerful Toolbox for Organic Synthesis. Molecules 2023, 28, 516. [Google Scholar] [CrossRef]
- Ünlü, A.E.; Prasad, B.; Anavekar, K.; Bubenheim, P.; Liese, A. The effect of natural deep eutectic solvents on laccase activity and oligomerization of rutin. Biocatal. Biotransform. 2022, 41, 353–366. [Google Scholar] [CrossRef]
- Pohnlein, M.; Ulrich, J.; Kirschhofer, F.; Nusser, M.; Muhle-Goll, C.; Kannengiesser, B.; Brenner-Weiss, G.; Luy, B.; Liese, A.; Syldatk, C.; et al. Lipase-catalyzed synthesis of glucose-6-O-hexanoate in deep eutectic solvents. Eur. J. Lipid Sci. Technol. 2015, 117, 161–166. [Google Scholar] [CrossRef]
- Nieto, S.; Bernal, J.M.; Villa, R.; García-Verdugo, E.; Donaire, A.; Lozano, P. Sustainable Setups for the Biocatalytic Production and Scale-Up of Panthenyl Monoacyl Esters under Solvent-Free Conditions. ACS Sustain. Chem. Eng. 2023, 11, 5737–5747. [Google Scholar] [CrossRef]
- Cao, J.; Wu, R.; Zhu, F.; Dong, Q.; Su, E. How to improve the efficiency of biocatalysis in non-aqueous pure deep eutectic solvents: A case study on the lipase-catalyzed transesterification reaction. Biochem. Eng. J. 2022, 179, 108336. [Google Scholar] [CrossRef]
- Chanquía, S.N.; Huang, L.; Liñares, G.G.; Domínguez de María, P.; Kara, S. Deep Eutectic Solvents as Smart Cosubstrate in Alcohol Dehydrogenase-Catalyzed Reductions. Catalysts 2020, 10, 1013. [Google Scholar] [CrossRef]
- Patzold, M.; Burek, B.O.; Liese, A.; Bloh, J.Z.; Holtmann, D. Product recovery of an enzymatically synthesized (−)-menthol ester in a deep eutectic solvent. Bioprocess Biosyst. Eng. 2019, 42, 1385–1389. [Google Scholar] [CrossRef] [PubMed]
- Hümmer, M.; Kara, S.; Liese, A.; Huth, I.; Schrader, J.; Holtmann, D. Synthesis of (−)-menthol fatty acid esters in and from (−)-menthol and fatty acids—Novel concept for lipase catalyzed esterification based on eutectic solvents. Mol. Catal. 2018, 458, 67–72. [Google Scholar] [CrossRef]
- Weiz, G.; Braun, L.; Lopez, R.; Domínguez de María, P.; Breccia, J.D. Enzymatic deglycosylation of flavonoids in deep eutectic solvents-aqueous mixtures: Paving the way for sustainable flavonoid chemistry. J. Mol. Catal. B Enzym. 2016, 130, 70–73. [Google Scholar] [CrossRef]
- Buzatu, A.R.; Soler, M.A.; Fortuna, S.; Ozkilinc, O.; Dreavă, D.M.; Bîtcan, I.; Badea, V.; Giannozzi, P.; Fogolari, F.; Gardossi, L.; et al. Reactive natural deep eutectic solvents increase selectivity and efficiency of lipase catalyzed esterification of carbohydrate polyols. Catal. Today 2024, 426, 114373. [Google Scholar] [CrossRef]
- Hollenbach, R.; Delavault, A.; Gebhardt, L.; Soergel, H.; Muhle-Goll, C.; Ochsenreither, K.; Syldatk, C. Lipase-Mediated Mechanoenzymatic Synthesis of Sugar Esters in Dissolved Unconventional and Neat Reaction Systems. ACS Sustain. Chem. Eng. 2022, 10, 10192–10202. [Google Scholar] [CrossRef]
- Krystof, M.; Perez-Sanchez, M.; Domínguez de María, P. Lipase-catalyzed (trans)esterification of 5-hydroxy- methylfurfural and separation from HMF esters using deep-eutectic solvents. ChemSusChem 2013, 6, 630–634. [Google Scholar] [CrossRef] [PubMed]
- Semproli, R.; Chanquia, S.N.; Bittner, J.P.; Müller, S.; Domínguez de María, P.; Kara, S.; Ubiali, D. Deep Eutectic Solvents for the Enzymatic Synthesis of Sugar Esters: A Generalizable Strategy? ACS Sustain. Chem. Eng. 2023, 11, 5926–5936. [Google Scholar] [CrossRef]
- Silvianti, F.; Maniar, D.; Boetje, L.; Woortman, A.J.J.; van Dijken, J.; Loos, K. Greener Synthesis Route for Furanic-Aliphatic Polyester: Enzymatic Polymerization in Ionic Liquids and Deep Eutectic Solvents. ACS Polym. Au 2023, 3, 82–95. [Google Scholar] [CrossRef]
- Ma, Y.; Li, P.; Li, Y.; Willot, S.J.; Zhang, W.; Ribitsch, D.; Choi, Y.H.; Verpoorte, R.; Zhang, T.; Hollmann, F.; et al. Natural Deep Eutectic Solvents as Multifunctional Media for the Valorization of Agricultural Wastes. ChemSusChem 2019, 12, 1310–1315. [Google Scholar] [CrossRef]
- Sanchez-Leija, R.J.; Torres-Lubian, J.R.; Resendiz-Rubio, A.; Luna-Barcenas, G.; Mota-Morales, J.D. Enzyme-mediated free radical polymerization of acrylamide in deep eutectic solvents. RSC Adv. 2016, 6, 13072–13079. [Google Scholar] [CrossRef]
- He, Y.; Li, K.; Wang, J.; Xu, L.; Yan, J.; Yang, M.; Yan, Y. A novel strategy for biodiesel production by combination of liquid lipase, deep eutectic solvent and ultrasonic-assistance in scaled-up reactor: Optimization and kinetics. J. Clean. Prod. 2022, 372, 133740. [Google Scholar] [CrossRef]
- Kleiner, B.; Fleischer, P.; Schörken, U. Biocatalytic synthesis of biodiesel utilizing deep eutectic solvents: A two-step-one-pot approach with free lipases suitable for acidic and used oil processing. Process Biochem. 2016, 51, 1808–1816. [Google Scholar] [CrossRef]
- Zhao, H.; Zhang, C.; Crittle, T.D. Choline-based deep eutectic solvents for enzymatic preparation of biodiesel from soybean oil. J. Mol. Catal. B Enzym. 2013, 85–86, 243–247. [Google Scholar] [CrossRef]
- Zhao, H.; Baker, G.A.; Holmes, S. New eutectic ionic liquids for lipase activation and enzymatic preparation of biodiesel. Org. Biomol. Chem. 2011, 9, 1908–1916. [Google Scholar] [CrossRef]
- Chanquia, S.N.; Benfeldt, F.V.; Petrovai, N.; Santner, P.; Hollmann, F.; Eser, B.E.; Kara, S. Immobilization and Application of Fatty Acid Photodecarboxylase in Deep Eutectic Solvents. ChemBioChem 2022, 23, e202200482. [Google Scholar] [CrossRef] [PubMed]
- Vidal, L.S.; Kelly, C.L.; Mordaka, P.M.; Heap, J.T. Review of NAD(P)H-dependent oxidoreductases: Properties, engineering and application. Biochim. Biophys. Acta Proteins Proteom. 2018, 1866, 327–347. [Google Scholar] [CrossRef] [PubMed]
- Martinez, A.T.; Ruiz-Duenas, F.J.; Camarero, S.; Serrano, A.; Linde, D.; Lund, H.; Vind, J.; Tovborg, M.; Herold-Majumdar, O.M.; Hofrichter, M.; et al. Oxidoreductases on their way to industrial biotransformations. Biotechnol. Adv. 2017, 35, 815–831. [Google Scholar] [CrossRef]
- de Gonzalo, G.; Paul, C.E. Recent trends in synthetic enzymatic cascades promoted by alcohol dehydrogenases. Curr. Opin. Green Sustain. Chem. 2021, 32, 100548. [Google Scholar] [CrossRef]
- Weckbecker, A.; Gröger, H.; Hummel, W. Regeneration of nicotinamide coenzymes: Principles and applications for the synthesis of chiral compounds. In Biosystems Engineering I: Creating Superior Biocatalysts; Wittmann, C., Krull, R., Eds.; Springer Berlin Heidelberg: Berlin/Heidelberg, Germany, 2010; pp. 195–242. [Google Scholar]
- Kara, S.; Schrittwieser, J.H.; Hollmann, F.; Ansorge-Schumacher, M.B. Recent trends and novel concepts in cofactor-dependent biotransformations. Appl. Microbiol. Biotechnol. 2014, 98, 1517–1529. [Google Scholar] [CrossRef] [PubMed]
- Hollmann, F.; Arends, I.W.C.E.; Buehler, K. Biocatalytic Redox Reactions for Organic Synthesis: Nonconventional Regeneration Methods. ChemCatChem 2010, 2, 762–782. [Google Scholar] [CrossRef]
- Roth, S.; Niese, R.; Muller, M.; Hall, M. Redox Out of the Box: Catalytic Versatility Across NAD(P)H-Dependent Oxidoreductases. Angew. Chem. Int. Ed. 2024, e202314740. [Google Scholar] [CrossRef] [PubMed]
- Zhang, N.; Müller, B.; Kirkeby, T.Ø.; Kara, S.; Loderer, C. Development of a Thioredoxin-Based Cofactor Regeneration System for NADPH-Dependent Oxidoreductases. ChemCatChem 2022, 14, e202101625. [Google Scholar] [CrossRef]
- Maugeri, Z.; Domínguez de María, P. Whole-Cell Biocatalysis in Deep-Eutectic-Solvents/Aqueous Mixtures. ChemCatChem 2014, 6, 1535–1537. [Google Scholar] [CrossRef]
- Gopal, M.R.; Dickey, R.M.; Butler, N.D.; Talley, M.R.; Nakamura, D.T.; Mohapatra, A.; Watson, M.P.; Chen, W.; Kunjapur, A.M. Reductive Enzyme Cascades for Valorization of Polyethylene Terephthalate Deconstruction Products. ACS Catal. 2023, 13, 4778–4789. [Google Scholar] [CrossRef]
- Cvjetko Bubalo, M.; Mazur, M.; Radošević, K.; Radojčić Redovniković, I. Baker’s yeast-mediated asymmetric reduction of ethyl 3-oxobutanoate in deep eutectic solvents. Process Biochem. 2015, 50, 1788–1792. [Google Scholar] [CrossRef]
- Vitale, P.; Abbinante, V.M.; Perna, F.M.; Salomone, A.; Cardellicchio, C.; Capriati, V. Unveiling the Hidden Performance of Whole Cells in the Asymmetric Bioreduction of Aryl-containing Ketones in Aqueous Deep Eutectic Solvents. Adv. Synth. Catal. 2017, 359, 1049–1057. [Google Scholar] [CrossRef]
- Xu, P.; Cheng, J.; Lou, W.Y.; Zong, M.H. Using deep eutectic solvents to improve the resolution of racemic 1-(4-methoxyphenyl)ethanol through Acetobacter sp. CCTCC M209061 cell-mediated asymmetric oxidation. RSC Adv. 2015, 5, 6357–6364. [Google Scholar] [CrossRef]
- Wei, P.; Liang, J.; Cheng, J.; Zong, M.H.; Lou, W.Y. Markedly improving asymmetric oxidation of 1-(4-methoxyphenyl) ethanol with Acetobacter sp. CCTCC M209061 cells by adding deep eutectic solvent in a two-phase system. Microb. Cell Fact. 2016, 15, 5. [Google Scholar] [CrossRef]
- Xu, P.; Xu, Y.; Li, X.F.; Zhao, B.Y.; Zong, M.H.; Lou, W.Y. Enhancing Asymmetric Reduction of 3-Chloropropiophenone with Immobilized Acetobacter sp. CCTCC M209061 Cells by Using Deep Eutectic Solvents as Cosolvents. ACS Sustain. Chem. Eng. 2015, 3, 718–724. [Google Scholar] [CrossRef]
- Xu, P.; Du, P.X.; Zong, M.H.; Li, N.; Lou, W.Y. Combination of deep eutectic solvent and ionic liquid to improve biocatalytic reduction of 2-octanone with Acetobacter pasteurianus GIM1.158 cell. Sci. Rep. 2016, 6, 26158. [Google Scholar] [CrossRef]
- Peng, F.; Chen, Q.-S.; Li, F.-Z.; Ou, X.-Y.; Zong, M.-H.; Lou, W.-Y. Using deep eutectic solvents to improve the biocatalytic reduction of 2-hydroxyacetophenone to (R)-1-phenyl-1,2-ethanediol by Kurthia gibsonii SC0312. Mol. Catal. 2020, 484, 110773. [Google Scholar] [CrossRef]
- Hillier, M.C.; Marcoux, J.F.; Zhao, D.; Grabowski, E.J.; McKeown, A.E.; Tillyer, R.D. Stereoselective formation of carbon-carbon bonds via SN2-displacement: Synthesis of substituted cycloalkyl[b]indoles. J. Org. Chem. 2005, 70, 8385–8394. [Google Scholar] [CrossRef]
- Hilborn, J.W.; Lu, Z.-H.; Jurgens, A.R.; Fang, Q.K.; Byers, P.; Wald, S.A.; Senanayake, C.H. A practical asymmetric synthesis of (R)-fluoxetine and its major metabolite (R)-norfluoxetine. Tetrahedron Lett. 2001, 42, 8919–8921. [Google Scholar] [CrossRef]
- Hillhouse, T.M.; Porter, J.H. A brief history of the development of antidepressant drugs: From monoamines to glutamate. Exp. Clin. Psychopharmacol. 2015, 23, 1–21. [Google Scholar] [CrossRef]
- Kamal, A.; Khanna, G.B.R.; Krishnaji, T.; Tekumalla, V.; Ramu, R. New chemoenzymatic pathway for β-adrenergic blocking agents. Tetrahedron Asymmetry 2005, 16, 1485–1494. [Google Scholar] [CrossRef]
- Maczka, W.; Winska, K.; Grabarczyk, M.; Zarowska, B. Yeast-Mediated Stereoselective Reduction of α-Acetylbutyrolactone. Appl. Sci. 2018, 8, 1334. [Google Scholar] [CrossRef]
- Panic, M.; Delac, D.; Roje, M.; Radojcic Redovnikovic, I.; Cvjetko Bubalo, M. Green asymmetric reduction of acetophenone derivatives: Saccharomyces cerevisiae and aqueous natural deep eutectic solvent. Biotechnol. Lett. 2019, 41, 253–262. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.J.; Qian, F.; Lin, H.X.; Chen, W.Q.; Wang, P. Using Choline Chloride-Based DESs as Co-Solvent for 3,5-Bis(trifluoromethyl) Acetophenone Bioreduction with XS1012. Catalysts 2020, 10, 30. [Google Scholar] [CrossRef]
- Li, J.; Wang, P.; He, Y.S.; Zhu, Z.R.; Huang, J. Toward Designing a Novel Oligopeptide-Based Deep Eutectic Solvent: Applied in Biocatalytic Reduction. ACS Sustain. Chem. Eng. 2019, 7, 1318–1326. [Google Scholar] [CrossRef]
- Bi, S.D.; Liu, H.Y.; Lin, H.X.; Wang, P. Integration of natural deep-eutectic solvent and surfactant for efficient synthesis of chiral aromatic alcohol mediated by whole cells. Biochem. Eng. J. 2021, 172, 108053. [Google Scholar] [CrossRef]
- Müller, C.R.; Lavandera, I.; Gotor-Fernández, V.; Domínguez de María, P. Performance of Recombinant-Whole-Cell-Catalyzed Reductions in Deep-Eutectic-Solvent–Aqueous-Media Mixtures. ChemCatChem 2015, 7, 2654–2659. [Google Scholar] [CrossRef]
- Hassani, F.Z.I.; Amzazi, S.; Kreit, J.; Lavandera, I. Deep Eutectic Solvents as Media in Alcohol Dehydrogenase-Catalyzed Reductions of Halogenated Ketones. ChemCatChem 2020, 12, 832–836. [Google Scholar] [CrossRef]
- Dai, Y.; Huan, B.; Zhang, H.S.; He, Y.C. Effective Biotransformation of Ethyl 4-Chloro-3-Oxobutanoate into Ethyl (S)-4-Chloro-3-Hydroxybutanoate by Recombinant E. coli CCZU-T15 Whole Cells in [ChCl][Gly]-Water Media. Appl. Biochem. Biotechnol. 2017, 181, 1347–1359. [Google Scholar] [CrossRef]
- He, Y.S.; Huang, Q.Y.; Wang, P. Design and evaluation of novel bio-based deep eutectic solvents for highly efficient bioproduction of chiral aryl alcohol. J. Chem. Technol. Biotechnol. 2020, 95, 1980–1988. [Google Scholar] [CrossRef]
- Xia, N.; Xiong, L.; Bi, S.; Qian, F.; Wang, P. Development of biocompatible DES/NADES as co-solvents for efficient biosynthesis of chiral alcohols. Bioprocess Biosyst. Eng. 2020, 43, 1987–1997. [Google Scholar] [CrossRef] [PubMed]
- Zhuang, W.; Liu, H.; Zhang, Y.; He, J.; Wang, P. Effective asymmetric preparation of (R)-1-[3-(trifluoromethyl)phenyl]ethanol with recombinant E. coli whole cells in an aqueous Tween-20/natural deep eutectic solvent solution. AMB Express 2021, 11, 118. [Google Scholar] [CrossRef] [PubMed]
- Mourelle-Insua, A.; Lavandera, I.; Gotor-Fernández, V. A designer natural deep eutectic solvent to recycle the cofactor in alcohol dehydrogenase-catalysed processes. Green Chem. 2019, 21, 2946–2951. [Google Scholar] [CrossRef]
- Meyer, L.E.; Andersen, M.B.; Kara, S. A Deep Eutectic Solvent Thermomorphic Multiphasic System for Biocatalytic Applications. Angew. Chem. Int. Ed. 2022, 61, e202203823. [Google Scholar] [CrossRef]
- de Gonzalo, G.; Martin, C.; Fraaije, M.W. Positive Impact of Natural Deep Eutectic Solvents on the Biocatalytic Performance of 5-Hydroxymethyl-Furfural Oxidase. Catalysts 2020, 10, 447. [Google Scholar] [CrossRef]
- de Gonzalo, G.; Lončar, N.; Fraaije, M. Kinetic resolution of racemic benzofused alcohols catalysed by HMFO variants in presence of natural deep eutectic solvents. Biocatal. Biotransform. 2022, 41, 145–152. [Google Scholar] [CrossRef]
- Ma, Y.J.; Li, Y.R.; Ali, S.; Li, P.L.; Zhang, W.Y.; Rauch, M.C.R.; Willot, S.J.P.; Ribitsch, D.; Choi, Y.H.; Alcalde, M.; et al. Natural Deep Eutectic Solvents as Performance Additives for Peroxygenase Catalysis. ChemCatChem 2020, 12, 989–994. [Google Scholar] [CrossRef]
- Li, Y.; Ma, Y.; Li, P.; Zhang, X.; Ribitsch, D.; Alcalde, M.; Hollmann, F.; Wang, Y. Enantioselective Sulfoxidation of Thioanisole by Cascading a Choline Oxidase and a Peroxygenase in the Presence of Natural Deep Eutectic Solvents. ChemPlusChem 2020, 85, 254–257. [Google Scholar] [CrossRef]
- Ezgi Ünlü, A.; Prasad, B.; Anavekar, K.; Bubenheim, P.; Liese, A. Investigation of a green process for the polymerization of catechin. Prep. Biochem. Biotechnol. 2017, 47, 918–924. [Google Scholar] [CrossRef]
- Yang, Z.; Ye, W.; Xie, Y.; Liu, Q.; Chen, R.; Wang, H.; Wei, D. Efficient Asymmetric Synthesis of Ethyl (S)-4-Chloro-3-hydroxybutyrate Using Alcohol Dehydrogenase SmADH31 with High Tolerance of Substrate and Product in a Monophasic Aqueous System. Org. Process Res. Dev. 2020, 24, 1068–1076. [Google Scholar] [CrossRef]
- Klumbys, E.; Zebec, Z.; Weise, N.J.; Turner, N.J.; Scrutton, N.S. Bio-derived Production of Cinnamyl Alcohol via a Three Step Biocatalytic Cascade and Metabolic Engineering. Green Chem. 2019, 20, 658–663. [Google Scholar] [CrossRef] [PubMed]
- Kara, S.; Spickermann, D.; Schrittwieser, J.H.; Leggewie, C.; van Berkel, W.J.H.; Arends, I.W.C.E.; Hollmann, F. More efficient redox biocatalysis by utilising 1,4-butanediol as a ‘smart cosubstrate’. Green Chem. 2013, 15, 330–335. [Google Scholar] [CrossRef]
- Turek, M.; Szczesna, D.; Koprowski, M.; Balczewski, P. Synthesis of 1-indanones with a broad range of biological activity. Beilstein J. Org. Chem. 2017, 13, 451–494. [Google Scholar] [CrossRef]
- Anselmi, S.; Aggarwal, N.; Moody, T.S.; Castagnolo, D. Unconventional Biocatalytic Approaches to the Synthesis of Chiral Sulfoxides. ChemBioChem 2021, 22, 298–307. [Google Scholar] [CrossRef] [PubMed]
- Gomes, A.R.; Varela, C.L.; Tavares-da-Silva, E.J.; Roleira, F.M.F. Epoxide containing molecules: A good or a bad drug design approach. Eur. J. Med. Chem. 2020, 201, 112327. [Google Scholar] [CrossRef] [PubMed]
- Khlupova, M.; Vasil’eva, I.; Shumakovich, G.; Zaitseva, E.; Chertkov, V.; Shestakova, A.; Morozova, O.; Yaropolov, A. Enzymatic Polymerization of Dihydroquercetin (Taxifolin) in Betaine-Based Deep Eutectic Solvent and Product Characterization. Catalysts 2021, 11, 639. [Google Scholar] [CrossRef]
- Delorme, A.E.; Andanson, J.M.; Verney, V. Improving laccase thermostability with aqueous natural deep eutectic solvents. Int. J. Biol. Macromol. 2020, 163, 919–926. [Google Scholar] [CrossRef]
- Khodaverdian, S.; Dabirmanesh, B.; Heydari, A.; Dashtban-Moghadam, E.; Khajeh, K.; Ghazi, F. Activity, stability and structure of laccase in betaine based natural deep eutectic solvents. Int. J. Biol. Macromol. 2018, 107, 2574–2579. [Google Scholar] [CrossRef]
- Altundağ, A.; Ünlü, A.E.; Takaç, S. Deep eutectic solvent-assisted synthesis of polyaniline by laccase enzyme. J. Chem. Technol. Biotechnol. 2020, 96, 1107–1115. [Google Scholar] [CrossRef]
- Durand, E.; Lecomte, J.; Barea, B.; Piombo, G.; Dubreucq, E.; Villeneuve, P. Evaluation of deep eutectic solvents as new media for Candida antarctica B lipase catalyzed reactions. Process Biochem. 2012, 47, 2081–2089. [Google Scholar] [CrossRef]
- Kim, S.H.; Park, S.; Yu, H.; Kim, J.H.; Kim, H.J.; Yang, Y.H.; Kim, Y.H.; Kim, K.J.; Kan, E.; Lee, S.H. Effect of deep eutectic solvent mixtures on lipase activity and stability. J. Mol. Catal. B Enzym. 2016, 128, 65–72. [Google Scholar] [CrossRef]
- Juneidi, I.; Hayyan, M.; Hashim, M.A.; Hayyan, A. Pure and aqueous deep eutectic solvents for a lipase-catalysed hydrolysis reaction. Biochem. Eng. J. 2017, 117, 129–138. [Google Scholar] [CrossRef]
- Cao, J.; Wu, R.; Zhu, F.; Dong, Q.; Su, E. Enzymes in nearly anhydrous deep eutectic solvents: Insight into the biocompatibility and thermal stability. Enzyme Microb. Technol. 2022, 157, 110022. [Google Scholar] [CrossRef]
- Xu, W.-J.; Huang, Y.-K.; Li, F.; Wang, D.-D.; Yin, M.-N.; Wang, M.; Xia, Z.-N. Improving β-glucosidase biocatalysis with deep eutectic solvents based on choline chloride. Biochem. Eng. J. 2018, 138, 37–46. [Google Scholar] [CrossRef]
- Lindberg, D.; de la Fuente Revenga, M.; Widersten, M. Deep eutectic solvents (DESs) are viable cosolvents for enzyme-catalyzed epoxide hydrolysis. J. Biotechnol. 2010, 147, 169–171. [Google Scholar] [CrossRef] [PubMed]
- Zhao, H.; Baker, G.A.; Holmes, S. Protease activation in glycerol-based deep eutectic solvents. J. Mol. Catal. B Enzym. 2011, 72, 163–167. [Google Scholar] [CrossRef]
- Romano, A.; Varriale, S.; Pezzella, C.; Totaro, G.; Andanson, J.M.; Verney, V.; Sisti, L. Natural deep eutectic solvents as thermostabilizer for Humicola insolens cutinase. New Biotechnol. 2023, 76, 118–126. [Google Scholar] [CrossRef] [PubMed]
- Stepankova, V.; Vanacek, P.; Damborsky, J.; Chaloupkova, R. Comparison of catalysis by haloalkane dehalogenases in aqueous solutions of deep eutectic and organic solvents. Green Chem. 2014, 16, 2754–2761. [Google Scholar] [CrossRef]
- Cvjetko Bubalo, M.; Jurinjak Tušek, A.; VinkoviĿ, M.; RadoševiĿ, K.; Gaurina SrĿek, V.; RadojĿiĿ RedovnikoviĿ, I. Cholinium-based deep eutectic solvents and ionic liquids for lipase-catalyzed synthesis of butyl acetate. J. Mol. Catal. B Enzym. 2015, 122, 188–198. [Google Scholar] [CrossRef]
- Jawale, P.V.; Bhanage, B.M. Synthesis of decanoate compounds in deep eutectic solvent using lipase: Optimization using response surface methodology, kinetic and docking study. J. Indian Chem. Soc. 2023, 100, 100950. [Google Scholar] [CrossRef]
- Noro, J.; Cabo, J.; Freitas, D.S.; Roque, C.S.; de Castro, M.; Cavaco-Paulo, A.; Silva, C. Deep Eutectic Solvents as Suitable Solvents for Lipase-Catalyzed Transesterification Reactions. ChemSusChem 2023, 16, e202300615. [Google Scholar] [CrossRef]
- Huang, Z.L.; Wu, B.P.; Wen, Q.; Yang, T.X.; Yang, Z. Deep eutectic solvents can be viable enzyme activators and stabilizers. J. Chem. Technol. Biotechnol. 2014, 89, 1975–1981. [Google Scholar] [CrossRef]
- Šalić, A.; Ljubić, A.; Marčinko, T.; Jurinjak Tušek, A.; Cvjetko Bubalo, M.; Tišma, M.; Zelić, B. Shifting the natural deep eutectic solvent based liquid lipase extraction from batch to continuous for more efficient process performance. J. Clean. Prod. 2023, 405, 136899. [Google Scholar] [CrossRef]
- Freitas, D.S.; Quesado, V.; Rocha, D.; Noro, J.; Martins, M.; Cavaco-Paulo, A.; Silva, C. Lipase-Catalysed Polymerization of Eutectic Mixtures. ChemSusChem 2023, 16, e202202374. [Google Scholar] [CrossRef]
- Yang, S.L.; Duan, Z.Q. Insight into enzymatic synthesis of phosphatidylserine in deep eutectic solvents. Catal. Commun. 2016, 82, 16–19. [Google Scholar] [CrossRef]
- Borse, B.N.; Borude, V.S.; Shukla, S.R. Synthesis of novel dihydropyrimidin-2(1h)-ones derivatives using lipase and their antimicrobial activity. Curr. Chem. Lett. 2012, 1, 59–68. [Google Scholar] [CrossRef]
- Guajardo, N.; Schrebler, R.A.; Domínguez de María, P. From batch to fed-batch and to continuous packed-bed reactors: Lipase-catalyzed esterifications in low viscous deep-eutectic-solvents with buffer as cosolvent. Bioresour. Technol. 2019, 273, 320–325. [Google Scholar] [CrossRef]
- Allegretti, C.; Gatti, F.G.; Marzorati, S.; Rossato, L.A.M.; Serra, S.; Strini, A.; D’Arrigo, P. Reactive Deep Eutectic Solvents (RDESs): A New Tool for Phospholipase D-Catalyzed Preparation of Phospholipids. Catalysts 2021, 11, 655. [Google Scholar] [CrossRef]
- Cheng, Q.B.; Zhang, L.W. Highly Efficient Enzymatic Preparation of Daidzein in Deep Eutectic Solvents. Molecules 2017, 22, 186. [Google Scholar] [CrossRef]
- Jin, W.; Wang, K.; Xu, X.; Xu, Y.; Li, F.; Ji, Q.; Chen, X.; Yu, X. High efficiency production of ferulic acid using feruloyl esterase and xylanase in deep eutectic solvents. New J. Chem. 2023, 47, 17599–17606. [Google Scholar] [CrossRef]
- Arndt, S.; Kohlpaintner, P.J.; Donsbach, K.; Waldvogel, S.R. Synthesis and Applications of Periodate for Fine Chemicals and Important Pharmaceuticals. Org. Process Res. Dev. 2022, 26, 2564–2613. [Google Scholar] [CrossRef]
- Peng, F.; Zhao, Y.; Li, F.Z.; Zong, M.H.; Lou, W.Y. The effect of deep eutectic solvents on the asymmetric hydrolysis of styrene oxide by mung bean epoxide hydrolases. Bioresour. Bioprocess. 2018, 5, 5. [Google Scholar] [CrossRef]
- Cao, S.-L.; Yue, D.-M.; Li, X.-H.; Smith, T.J.; Li, N.; Zong, M.-H.; Wu, H.; Ma, Y.-Z.; Lou, W.-Y. Novel Nano-/Micro-Biocatalyst: Soybean Epoxide Hydrolase Immobilized on UiO-66-NH2 MOF for Efficient Biosynthesis of Enantiopure (R)-1, 2-Octanediol in Deep Eutectic Solvents. ACS Sustain. Chem. Eng. 2016, 4, 3586–3595. [Google Scholar] [CrossRef]
- Durand, E.; Lecomte, J.; Baréa, B.; Dubreucq, E.; Lortie, R.; Villeneuve, P. Evaluation of deep eutectic solvent-water binary mixtures for lipase-catalyzed lipophilization of phenolic acids. Green Chem. 2013, 15, 2275–2282. [Google Scholar] [CrossRef]
- Xiong, J.; Cao, S.L.; Zong, M.H.; Lou, W.Y.; Wu, X.L. Biosynthesis of Alanyl-Histidine Dipeptide Catalyzed by Papain Immobilized on Magnetic Nanocrystalline Cellulose in Deep Eutectic Solvents. Appl. Biochem. Biotechnol. 2020, 192, 573–584. [Google Scholar] [CrossRef]
- González-Martínez, D.; Gotor, V.; Gotor-Fernández, V. Application of Deep Eutectic Solvents in Promiscuous Lipase-Catalysed Aldol Reactions. Eur. J. Org. Chem. 2016, 2016, 1513–1519. [Google Scholar] [CrossRef]
- Tian, X.; Zhang, S.; Zheng, L. Enzyme-Catalyzed Henry Reaction in Choline Chloride-Based Deep Eutectic Solvents. J. Microbiol. Biotechnol. 2016, 26, 80–88. [Google Scholar] [CrossRef]
- Milker, S.; Pätzold, M.; Bloh, J.Z.; Holtmann, D. Comparison of deep eutectic solvents and solvent-free reaction conditions for aldol production. Mol. Catal. 2019, 466, 70–74. [Google Scholar] [CrossRef]
- Wang, Y.; Chen, X.Y.; Liang, X.Y.; Liang, Z.H.; Cheng, H.; Li, X.; Li, L.L. Pepsin-Catalyzed Asymmetric Cross Aldol Reaction Promoted by Ionic Liquids and Deep Eutectic Solvents. Catal. Lett. 2020, 150, 2549–2557. [Google Scholar] [CrossRef]
- Wang, Y.; Liang, X.Y.; Chen, X.Y.; Liang, Z.H.; Cheng, H.; Li, X.; Li, L.L. Promotion of water in BPL-catalyzed asymmetric cross aldol reaction. Biomass Convers. Biorefin. 2022, 12, 2017–2025. [Google Scholar] [CrossRef]
- Majo, D.D.; Giammanco, M.; Guardia, M.L.; Tripoli, E.; Giammanco, S.; Finotti, E. Flavanones in Citrus fruit: Structure–antioxidant activity relationships. Food Res. Int. 2005, 38, 1161–1166. [Google Scholar] [CrossRef]
- Maugeri, Z.; Domínguez de María, P. Benzaldehyde lyase (BAL)-catalyzed enantioselective CC bond formation in deep-eutectic-solvents–buffer mixtures. J. Mol. Catal. B Enzym. 2014, 107, 120–123. [Google Scholar] [CrossRef]
- Paris, J.; Telzerow, A.; Ríos-Lombardíia, N.; Steiner, K.; Schwab, H.; Morís, F.; Gröger, H.; González-Sabín, J. Enantioselective One-Pot Synthesis of Biaryl-Substituted Amines by Combining Palladium and Enzyme Catalysis in Deep Eutectic Solvents. ACS Sustain. Chem. Eng. 2019, 7, 5486–5493. [Google Scholar] [CrossRef]
- Kourist, R.; González-Sabín, J. Non-Conventional Media as Strategy to Overcome the Solvent Dilemma in Chemoenzymatic Tandem Catalysis. ChemCatChem 2020, 12, 1903–1912. [Google Scholar] [CrossRef]
- Cicco, L.; Dilauro, G.; Perna, F.M.; Vitale, P.; Capriati, V. Advances in deep eutectic solvents and water: Applications in metal- and biocatalyzed processes, in the synthesis of APIs, and other biologically active compounds. Org. Biomol. Chem. 2021, 19, 2558–2577. [Google Scholar] [CrossRef]
- Müller, C.R.; Meiners, I.; Domínguez de María, P. Highly enantioselective tandem enzyme–organocatalyst crossed aldol reactions with acetaldehyde in deep-eutectic-solvents. RSC Adv. 2014, 4, 46097–46101. [Google Scholar] [CrossRef]
- Zhou, P.F.; Wang, X.P.; Yang, B.; Hollmann, F.; Wang, Y.H. Chemoenzymatic epoxidation of alkenes with Candida antarctica lipase B and hydrogen peroxide in deep eutectic solvents. RSC Adv. 2017, 7, 12518–12523. [Google Scholar] [CrossRef]
- Cicco, L.; Rios-Lombardía, N.; Rodríguez-Alvarez, M.J.; Morís, F.; Perna, F.M.; Capriati, V.; García-Alvarez, J.; González-Sabín, J. Programming cascade reactions interfacing biocatalysis with transition-metal catalysis in Deep Eutectic Solvents as biorenewable reaction media. Green Chem. 2018, 20, 3468–3475. [Google Scholar] [CrossRef]
- Paris, J.; Ríos-Lombardía, N.; Morís, F.; Gröger, H.; González-Sabín, J. Novel Insights into the Combination of Metal- and Biocatalysis: Cascade One-Pot Synthesis of Enantiomerically Pure Biaryl Alcohols in Deep Eutectic Solvents. ChemCatChem 2018, 10, 4417–4423. [Google Scholar] [CrossRef]
- Li, Q.; Di, J.H.; Liao, X.L.; Ni, J.C.; Li, Q.; He, Y.C.; Ma, C.L. Exploration of benign deep eutectic solvent-water systems for the highly efficient production of furfurylamine from sugarcane bagasse chemoenzymatic cascade catalysis. Green Chem. 2021, 23, 8154–8168. [Google Scholar] [CrossRef]
- Rios-Lombardia, N.; Rodríguez-Álvarez, M.J.; Moris, F.; Kourist, R.; Comino, N.; López-Gallego, F.; González-Sabín, J.; García-Álvarez, J. DESign of Sustainable One-Pot Chemoenzymatic Organic Transformations in Deep Eutectic Solvents for the Synthesis of 1,2-Disubstituted Aromatic Olefins. Front. Chem. 2020, 8, 139. [Google Scholar] [CrossRef]
- Grabner, B.; Schweiger, A.K.; Gavric, K.; Kourist, R.; Gruber-Woelfler, H. A chemo-enzymatic tandem reaction in a mixture of deep eutectic solvent and water in continuous flow. React. Chem. Eng. 2020, 5, 263–269. [Google Scholar] [CrossRef]
- Liang, Y.; Narayanasamy, J.; Schinazi, R.F.; Chu, C.K. Phosphoramidate and phosphate prodrugs of (−)-beta-D-(2R,4R)-dioxolane-thymine: Synthesis, anti-HIV activity and stability studies. Bioorg. Med. Chem. 2006, 14, 2178–2189. [Google Scholar] [CrossRef]
- Bühlmayer, P.; Furet, P.; Criscione, L.; de Gasparo, M.; Whitebread, S.; Schmidlin, T.; Lattmann, R.; Wood, J. Valsartan, a potent, orally active angiotensin II antagonist developed from the structurally new amino acid series. Bioorg. Med. Chem. Lett. 1994, 4, 29–34. [Google Scholar] [CrossRef]
- Savio, M.; Ferraro, D.; Maccario, C.; Vaccarone, R.; Jensen, L.D.; Corana, F.; Mannucci, B.; Bianchi, L.; Cao, Y.; Stivala, L.A. Resveratrol analogue 4,4′-dihydroxy-trans-stilbene potently inhibits cancer invasion and metastasis. Sci. Rep. 2016, 6, 19973. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).