Selectivity Regulation of Au/Titanate by Biochar Modification for Selective Oxidation of Benzyl Alcohol
Abstract
1. Introduction
2. Results and Discussions
2.1. Characterizations of Catalysts
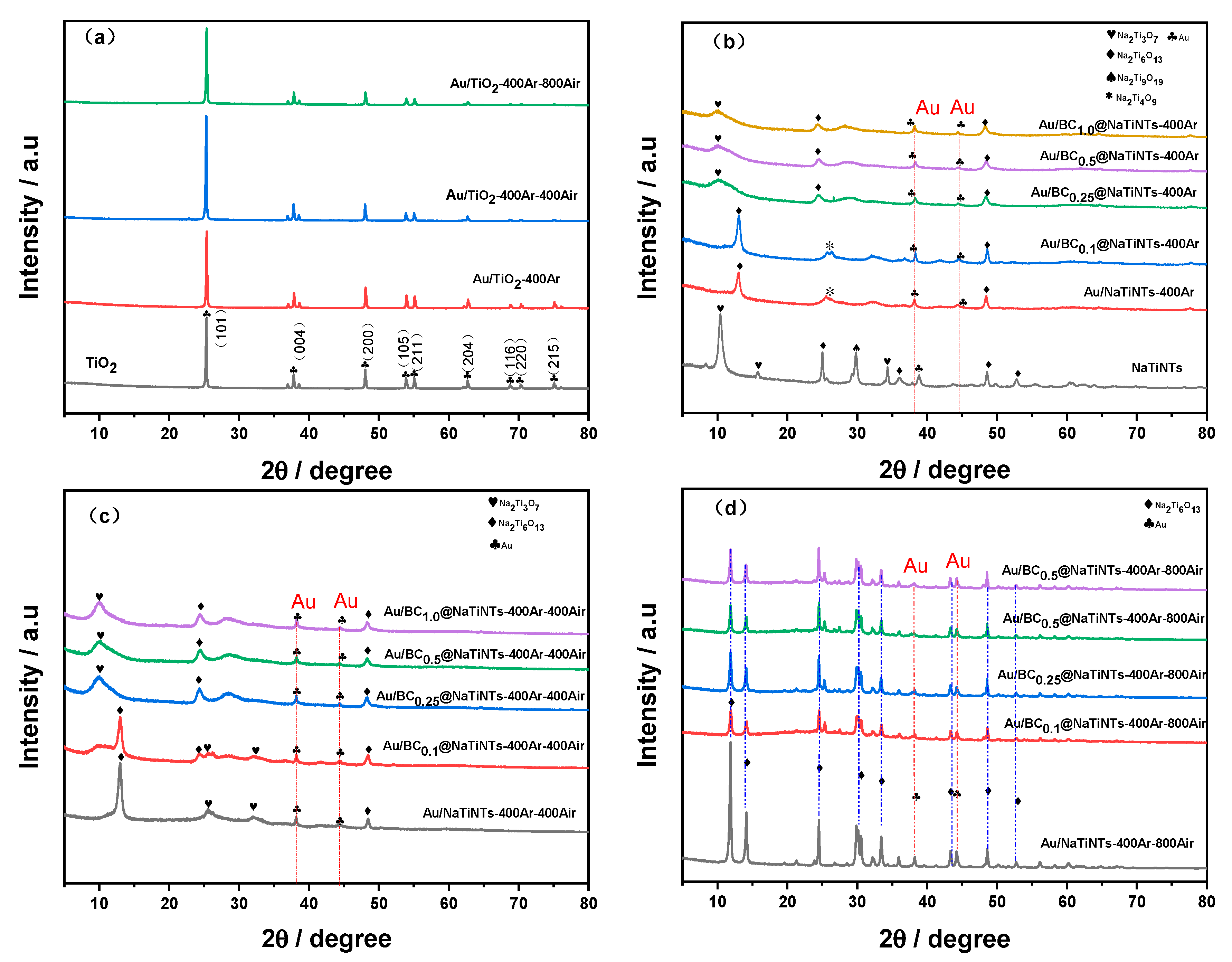
2.1.1. Crystal Structures Analysis by XRD
2.1.2. Morphology Analysis by SEM and TEM
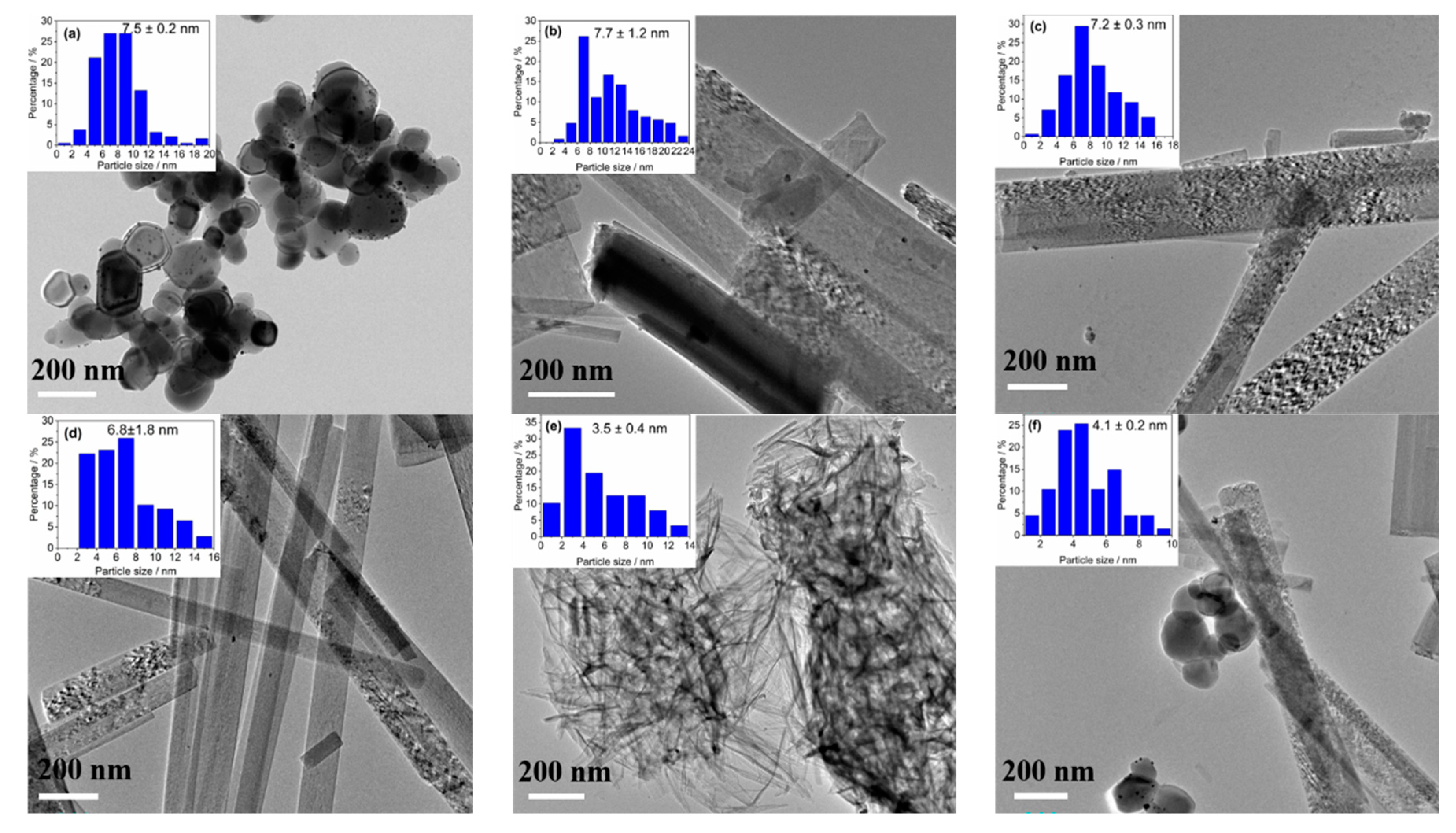
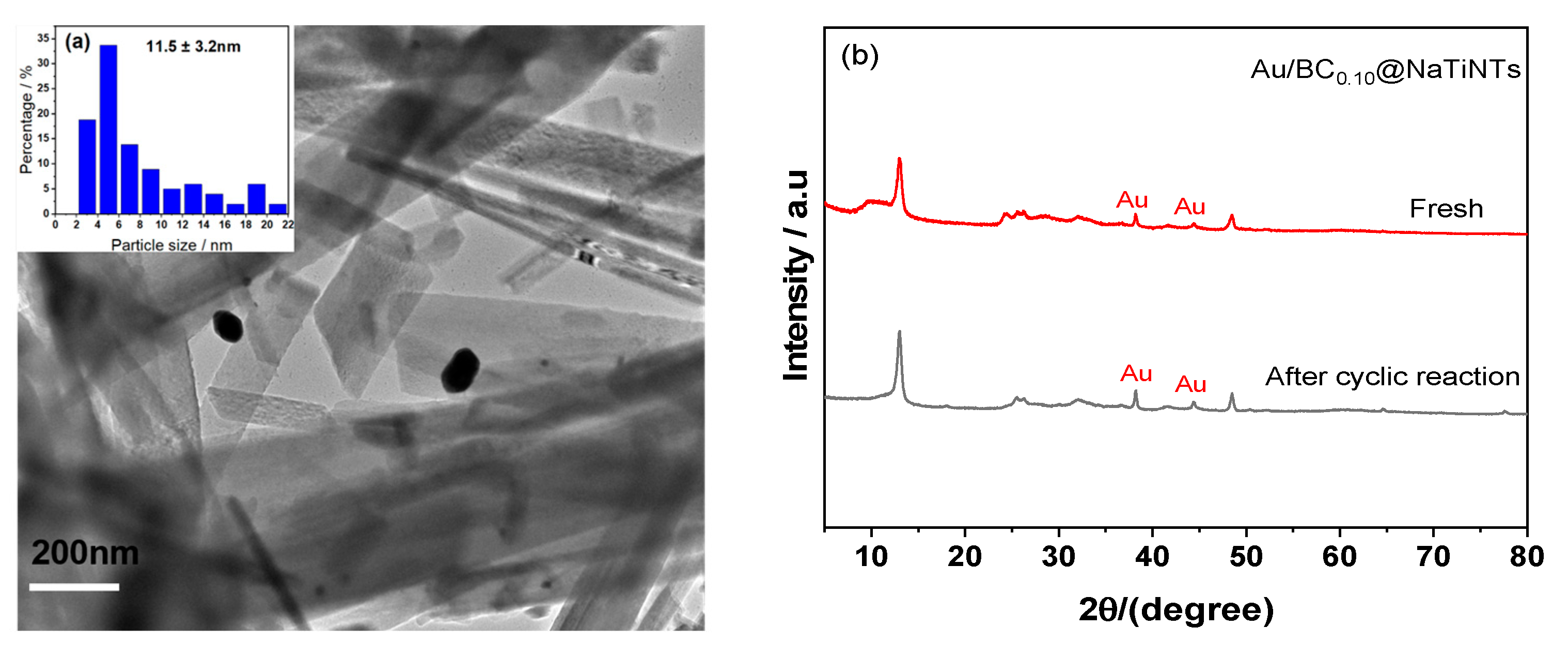
2.1.3. TEM Analyses and Size Distribution of Au NPs
2.1.4. Physicochemical Properties of Catalysts
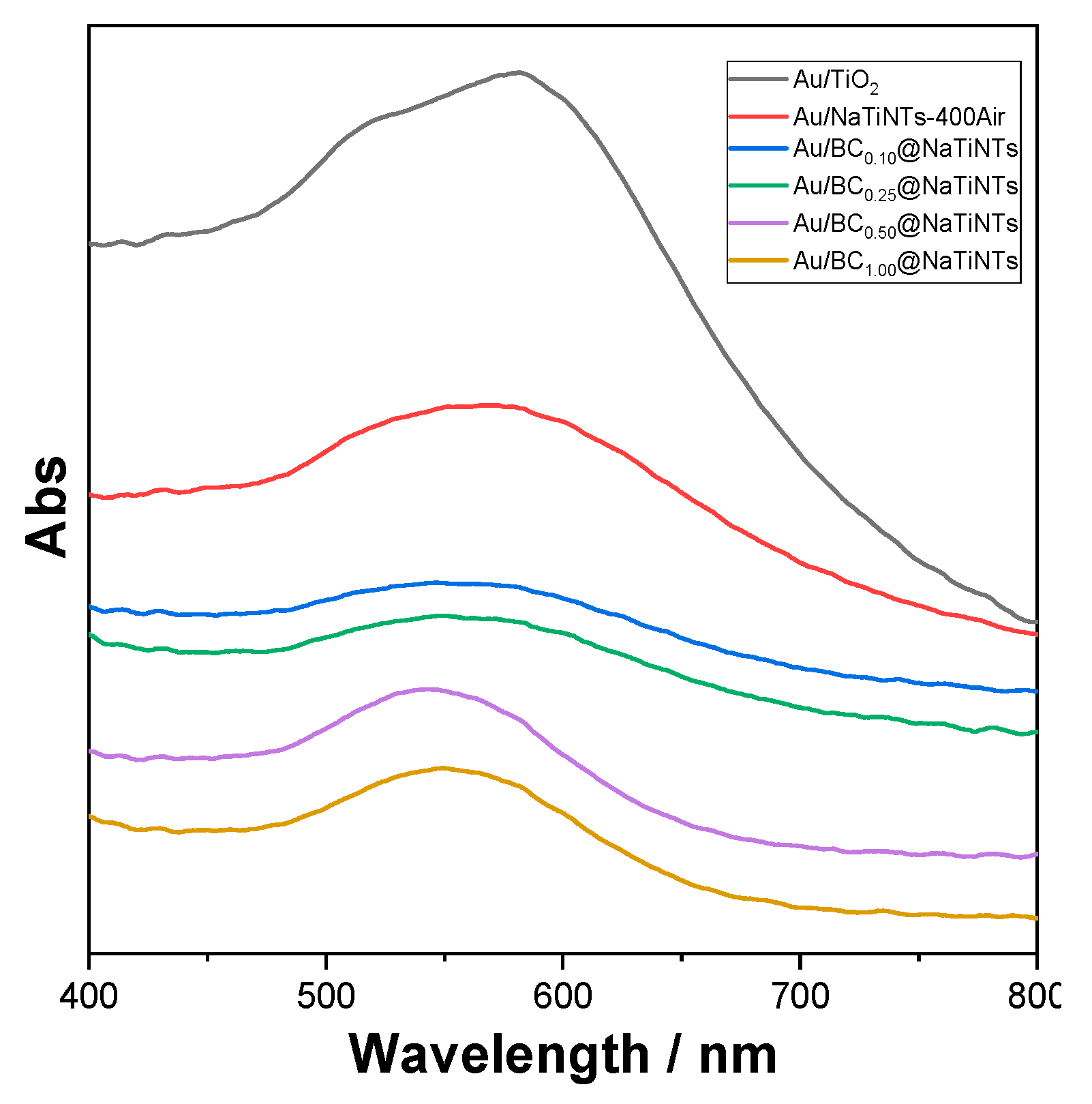
2.1.5. UV-Vis Characterization of the Catalysts
2.1.6. XPS Analyses of Key Elements
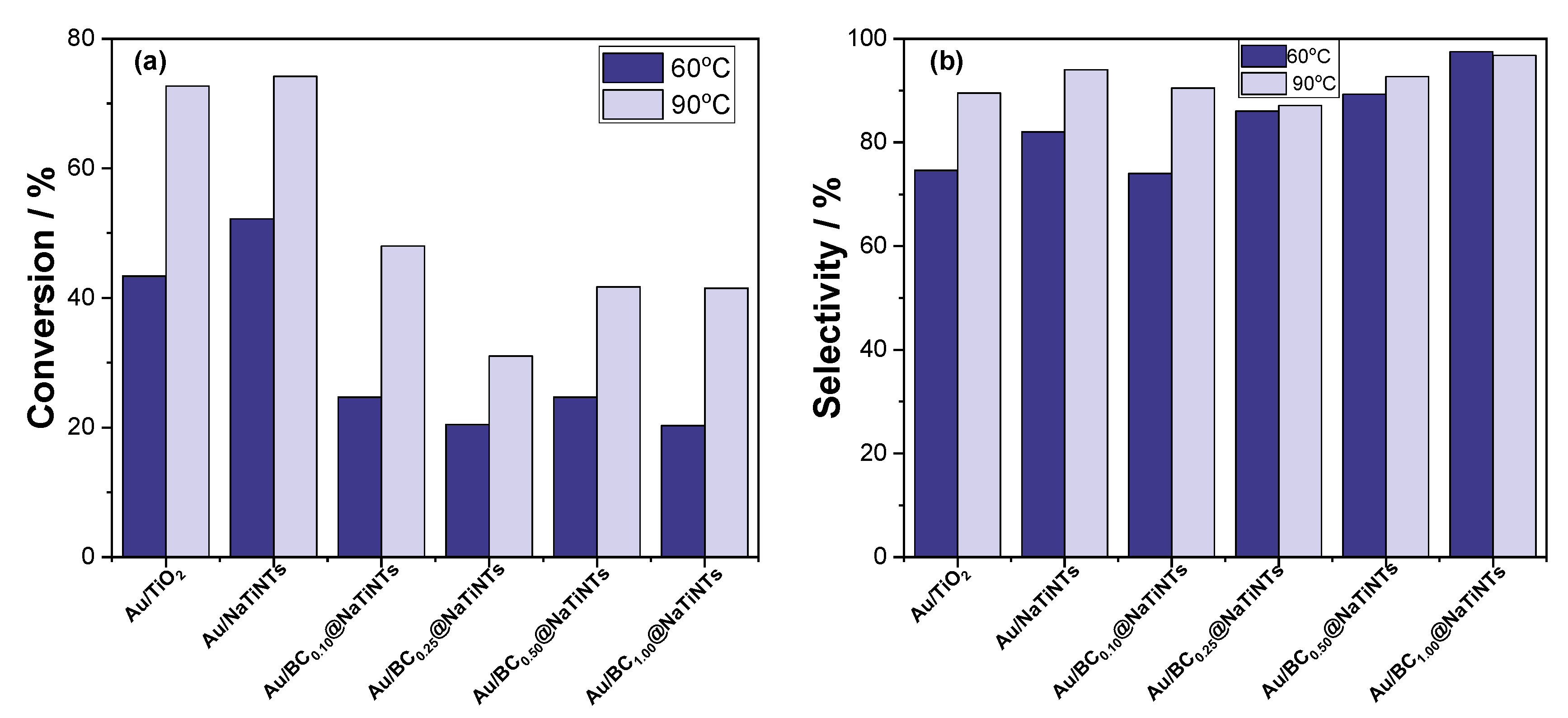
2.2. Catalytic Performances
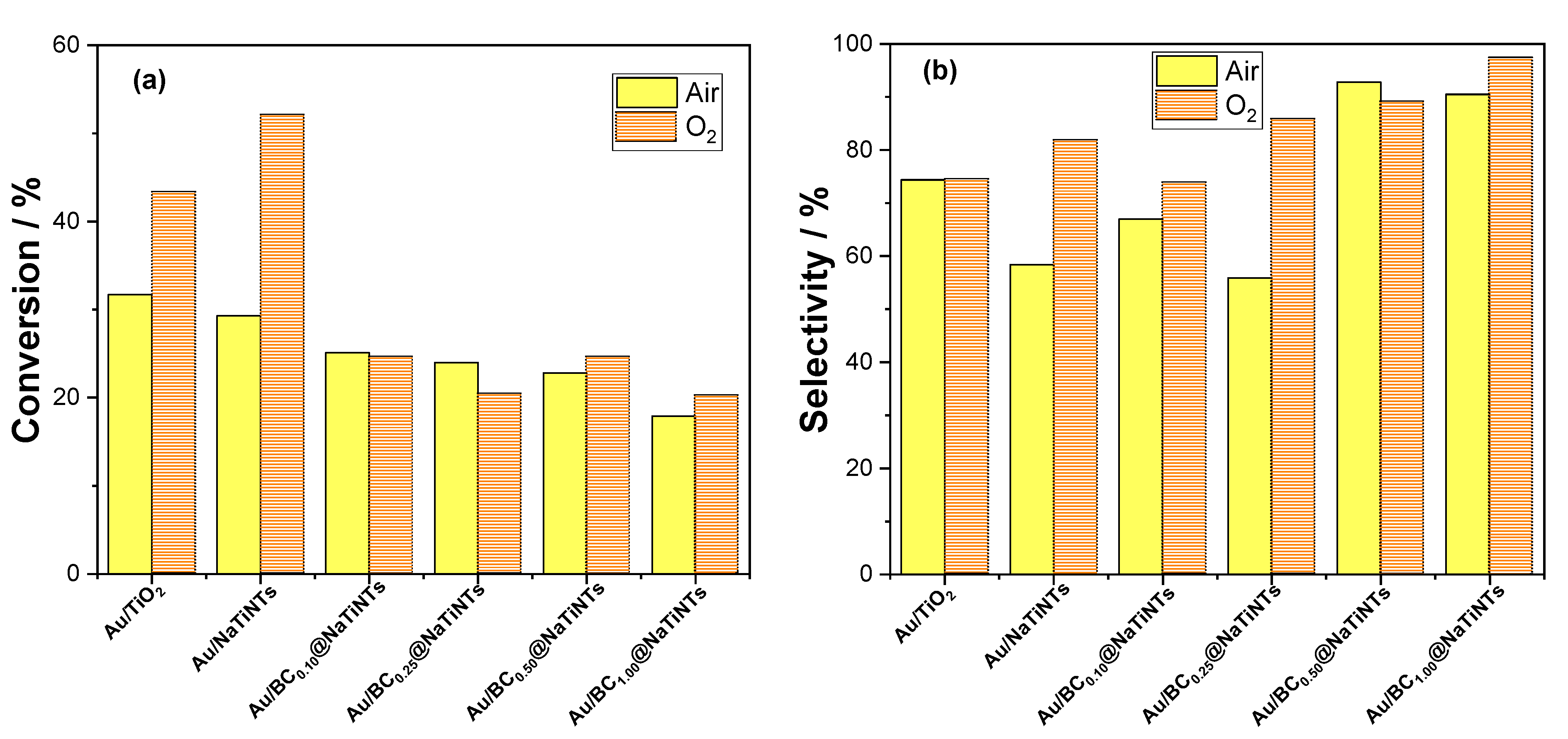
2.2.1. The Influence of Calcination Temperatures and Biochar Content
2.2.2. The Influence of Reaction Time, Atmospheres, and Temperatures
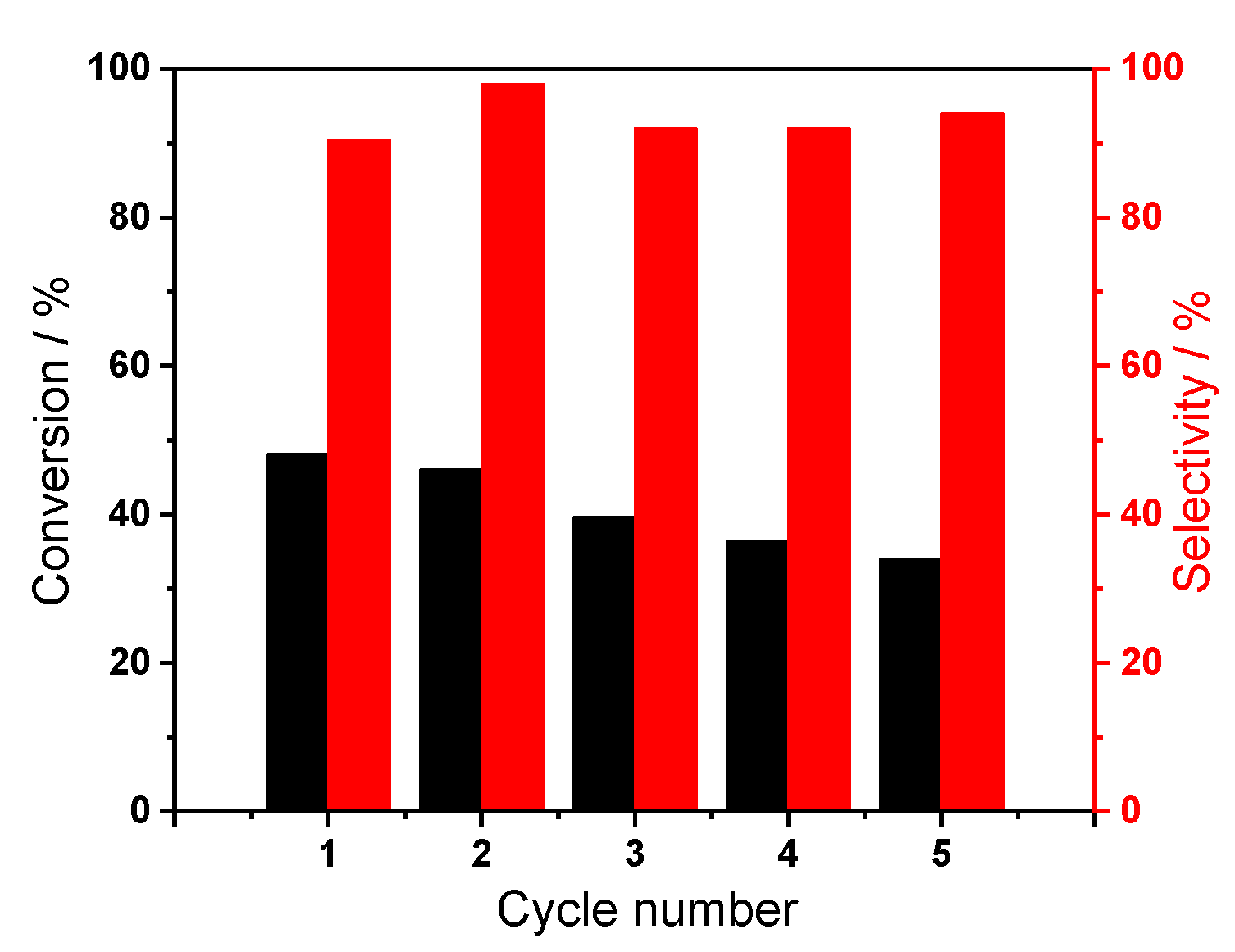
2.2.3. Reusability Test
2.2.4. The Influence of Solvent
3. Experimental Parts
3.1. Raw Materials
3.2. Catalyst Preparation
3.3. Catalyst Characterization
3.4. Catalytic Tests
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Yadav, G.D.; Mewada, R.K.; Manyar, H.; Wagh, D.P. Advances and future trends in selective oxidation catalysis: A critical review. Catal. Sci. Technol. 2022, 12, 7245–7269. [Google Scholar] [CrossRef]
- Xiao, Y.H.; Liang, L.; Liu, Z.Z.; Yin, X.; Yang, X.J.; Ding, Y.G.; Du, Z.P. Facile fabrication of size-controlled Pd nanoclusters supported on Al2O3 as excellent catalyst for solvent-free aerobic oxidation of benzyl alcohol. Appl. Surf. Sci. 2022, 585, 152668. [Google Scholar] [CrossRef]
- Li, Y.Y.; Sabbaghi, A.; Huang, J.; Li, K.C.; Tsui, L.S.; Lam, F.L.Y.; Hu, X.J. Aerobic oxidation of benzyl alcohol: Influence from catalysts basicity, acidity, and preparation methods. Mol. Catal. 2020, 485, 110789. [Google Scholar] [CrossRef]
- Lv, X.S.; Yuan, S.H.; Zhang, Y.W.; Fu, Y.F.; Wu, Y.J.; Zhou, Y.M.; Huang, R.; Huang, Z.W. Preparation of cyclonic Co3O4/Au/mesoporous SiO2 catalysts with core–shell structure for solvent-free oxidation of benzyl alcohol. J. Taiwain. Inst. Chem. Eng. 2019, 102, 448–455. [Google Scholar] [CrossRef]
- Villa, A.; Dimitratos, N.; Chan, T.C.E.; Hammond, C.; Veith, G.M.; Wang, D.; Manzoli, M.; Prati, L.; Hutchings, G.J. Characterisation of gold catalysts. Chem. Soc. Rev. 2016, 45, 4953–4994. [Google Scholar] [CrossRef]
- Xu, Y.; Li, J.; Zhou, J.; Liu, Y.F.; Wei, Z.J.; Zhang, H. Layered double hydroxides supported atomically precise Aun nanoclusters for air oxidation of benzyl alcohol: Effects of size and active site structure. J. Catal. 2020, 389, 409–420. [Google Scholar] [CrossRef]
- Sankar, M.; He, Q.; Engel, R.V.; Sainna, M.A.; Logsdail, A.J.; Roldan, A.; Willock, D.J.; Agarwal, N.; Kiely, C.J.; Hutchings, G.J. Role of the support in gold-containing nanoparticles as heterogeneous catalysts. Chem. Rev. 2020, 120, 3890–3938. [Google Scholar] [CrossRef]
- Brindle, J.; Sufyan, S.A.; Nigra, M.M. Support, composition, and ligand effects in partial oxidation of benzyl alcohol using gold–copper clusters. Catal. Sci. Technol. 2022, 12, 3846–3855. [Google Scholar] [CrossRef]
- Hao, Y.; Wang, S.; Sun, Q.; Shi, L.; Lu, A.H. Uniformly dispersed Pd nanoparticles on nitrogen-doped carbon nanospheres for aerobic benzyl alcohol oxidation. Chin. J. Catal. 2015, 36, 612–619. [Google Scholar] [CrossRef]
- Li, X.L.; Feng, J.J.; Sun, J.; Wang, Z.; Zhao, W. Solvent-free catalytic oxidation of benzyl alcohol over Au-Pd bimetal deposited on TiO2: Comparison of rutile, brookite, and anatase. Nanoscale Res. Lett. 2019, 14, 394. [Google Scholar] [CrossRef]
- Ndolomingo, M.J.; Meijboom, R. Selective liquid phase oxidation of benzyl alcohol to benzaldehyde by tert-butyl hydroperoxide over γ-Al2O3 supported copper and gold nanoparticles. Appl. Surf. Sci. 2017, 398, 19–32. [Google Scholar] [CrossRef]
- Zhou, Q.Y.; Zhou, C.Y.; Zhou, Y.H.; Hong, W.; Zou, S.H.; Gong, X.Q.; Liu, J.J.; Xiao, L.p.; Fan, J. More than oxygen vacancies: A collective crystal-plane effect of CeO2 in gas-phase selective oxidation of benzyl alcohol. Catal. Sci. Technol. 2019, 9, 2960–2967. [Google Scholar] [CrossRef]
- Ishida, T.; Murayama, T.; Taketoshi, A.; Haruta, M. Importance of size and contact structure of gold nanoparticles for the genesis of unique catalytic processes. Chem. Rev. 2019, 120, 464–525. [Google Scholar] [CrossRef] [PubMed]
- Sudheeshkumar, V.; Shivhare, A.; Scott, R.W.J. Synthesis of sinter-resistant Au@silica catalysts derived from Au25 clusters. Catal. Sci. Technol. 2016, 7, 272–280. [Google Scholar] [CrossRef]
- Khawaji, M.; Chadwick, D. Au–Pd NPs immobilised on nanostructured ceria and titania: Impact of support morphology on the catalytic activity for selective oxidation. Catal. Sci. Technol. 2018, 8, 2529–2539. [Google Scholar] [CrossRef]
- Zaarour, M.; Cazemier, J.; Ruiz-Martínez, J. Recent developments in the control of selectivity in hydrogenation reactions by confined metal functionalities. Catal. Sci. Technol. 2020, 10, 8140–8172. [Google Scholar] [CrossRef]
- Otor, H.O.; Steiner, J.B.; García-Sancho, C.; Alba-Rubio, A.C. Encapsulation methods for control of catalyst deactivation: A Review. ACS Catal. 2020, 10, 7630–7656. [Google Scholar] [CrossRef]
- Zhan, W.; He, Q.; Liu, X.; Guo, Y.; Wang, Y.; Wang, L.; Guo, Y.; Borisevich, A.Y.; Zhang, J.; Lu, G.; et al. A sacrificial coating strategy toward enhancement of metal–support interaction for ultrastable Au nanocatalysts. J. Am. Chem. Soc. 2016, 138, 16130–16139. [Google Scholar] [CrossRef]
- Liu, B.; Wang, P.; Lopes, A.; Jin, L.; Zhong, W.; Pei, Y.; Suib, S.L.; He, J. Au–carbon electronic interaction mediated selective oxidation of styrene. ACS Catal. 2017, 7, 3483–3488. [Google Scholar] [CrossRef]
- Do Minh, T.; Song, J.; Deb, A.; Cha, L.; Srivastava, V.; Sillanpää, M. Biochar based catalysts for the abatement of emerging pollutants: A review. Chem. Eng. J. 2020, 394, 124856. [Google Scholar] [CrossRef]
- Szczęśniak, B.; Phuriragpitikhon, J.; Choma, J.; Jaroniec, M. Recent advances in the development and applications of biomass-derived carbons with uniform porosity. J. Mater. Chem. A 2020, 8, 18464–18491. [Google Scholar] [CrossRef]
- Dong, X.S.; Wang, Y.Q.; Jia, M.M.; Niu, Z.Y.; Cai, J.M.; Yu, X.; Ke, X.B.; Yao, J.F.; Zhang, X.G. Sustainable and scalable in-situ synthesis of hydrochar-wrapped Ti3AlC2-derived nanofibers as adsorbents to remove heavy metals. Bioresource Technol. 2019, 282, 222–227. [Google Scholar] [CrossRef] [PubMed]
- Cojocaru, B.; Andrei, V.; Tudorache, M.; Lin, F.; Cadigan, C.; Richards, R.; Parvulescu, V.I. Enhanced photo-degradation of bisphenol pollutants onto gold-modified photocatalysts. Catal. Today 2016, 284, 153–159. [Google Scholar] [CrossRef]
- Gregor, Ž.; Matevž, R.; Janez, Z.; Janez, K.; Albin, P. Effect of Au loading on Schottky barrier height in TiO2 + Au plasmonic photocatalysts. Appl. Surf. Sci. 2021, 579, 152196. [Google Scholar]
- Lu, Y.M.; Zhu, H.Z.; Liu, J.W.; Yu, S.H. Palladium nanoparticles supported on titanate nanobelts for solvent-free aerobic oxidation of alcohols. ChemCatChem 2015, 7, 4131–4136. [Google Scholar] [CrossRef]
- Jamal, H.; Kang, B.S.; Lee, H.; Yu, J.S.; Lee, C.S. Comparative Studies of Electrochemical Performance and Characterization of TiO2/Graphene Nanocomposites as Anode Materials for Li-Secondary Batteries. J. Ind. Eng. Chem. 2018, 64, 151–166. [Google Scholar] [CrossRef]
- Song, K.; Wang, X.H.; Xiang, Q.; Xu, J.Q. Weakened negative effect of Au/TiO2 photocatalytic activity by CdS quantum dots deposited under UV-vis light illumination at different intensity ratios. Phys. Chem. Chem. Phys. 2016, 18, 29131–29138. [Google Scholar] [CrossRef]
- Corro, G.; Cebada, S.; Pal, U.; Fierro, J.L.G. Au0–Au3+ bifunctional site mediated enhanced catalytic activity of Au/ZnO composite in diesel particulate matter oxidation. J. Catal. 2017, 347, 148–156. [Google Scholar] [CrossRef]
- Hernández Ramírez, E.; Wang, J.A.; Chen, L.F.; Valenzuela, M.A.; Dalai, A.K. Partial oxidation of methanol catalyzed with Au/TiO2, Au/ZrO2 and Au/ZrO2-TiO2 catalysts. Appl. Surf. Sci. 2017, 399, 77–85. [Google Scholar] [CrossRef]
- Pan, Y.N.; Wu, G.D.; He, Y.F.; Feng, J.T.; Li, D.Q. Identification of the Au/ZnO interface as the specific active site for the selective oxidation of the secondary alcohol group in glycerol. J. Catal. 2018, 369, 222–232. [Google Scholar] [CrossRef]
- Wong, R.J.; Scott, J.; Kappen, P.; Low, G.K.C.; Hart, J.N.; Amal, R. Enhancing bimetallic synergy with light: The effect of UV light pre-treatment on catalytic oxygen activation by bimetallic Au–Pt nanoparticles on a TiO2 support. Catal. Sci. Technol. 2017, 7, 4792–4805. [Google Scholar] [CrossRef]
- Yu, Y.; Wen, W.; Qian, X.Y.; Liu, J.B.; Wu, J.M. UV and visible light photocatalytic activity of Au/TiO2 nanoforests with anatase/rutile phase junctions and controlled Au locations. Sci. Rep. 2017, 7, 41253. [Google Scholar] [CrossRef] [PubMed]
- Deas, R.; Pearce, S.; Goss, K.; Wang, Q.; Chen, W.T.; Waterhouse, G.I.N. Hierarchical Au/TiO2 nanoflower photocatalysts with outstanding performance for alcohol photoreforming under UV irradiation. Appl. Catal. A Gen. 2020, 602, 117706. [Google Scholar] [CrossRef]
- Khawaji, M.; Chadwick, D. Au-Pd bimetallic nanoparticles immobilised on titanate nanotubes: A highly active catalyst for selective oxidation. ChemCatChem 2017, 9, 4353–4363. [Google Scholar] [CrossRef]
- Khawaji, M.; Chadwick, D. Selective oxidation using Au-Pd catalysts: Role of the support in the stabilization of colloidal Au-Pd NPs. Catal. Today 2019, 348, 203–211. [Google Scholar] [CrossRef]
- Chen, D.; Shi, J.; Shen, H.Y. High-dispersed catalysts of core-shell structured Au@SiO2 for formaldehyde catalytic oxidation. Chem. Eng. J. 2019, 385, 123887. [Google Scholar] [CrossRef]
- Aramendía, M.A.; Borau, V.; Colmenares, J.C.; Marinas, A.; Marinas, J.M.; Navío, J.A.; Urbano, F.J. Modification of the photocatalytic activity of Pd/TiO2 and Zn/TiO2 systems through different oxidative and reductive calcination treatments. Appl. Catal. B Environ. 2008, 80, 88–97. [Google Scholar] [CrossRef]
- Soundiraraju, B.; George, B.K. Two-dimensional titanium nitride (Ti2N) MXene: Synthesis, characterization, and potential application as surface-enhanced raman scattering substrate. ACS Nano 2017, 11, 8892–8900. [Google Scholar] [CrossRef]
- Wu, Y.M.; Zhang, J.L.; Xiao, L.; Chen, F. Preparation and characterization of TiO2 photocatalysts by Fe3+ doping together with Au deposition for the degradation of organic pollutants. Appl. Catal. B Environ. 2008, 88, 525–532. [Google Scholar] [CrossRef]
- Peng, C.; Yang, X.F.; Li, Y.H.; Yu, H.; Wang, H.J.; Peng, F. Hybrids of two-dimensional Ti3C2 and TiO2 exposing {001} facets toward enhanced photocatalytic activity. ACS Appl. Mater. Interfaces 2016, 8, 6051–6060. [Google Scholar] [CrossRef]
- Li, D.F.; Wang, J.G.; Xu, F.X.; Zhang, N.C.; Men, Y. Mesoporous (001)-TiO2 nanocrystals with tailored Ti3+ and surface oxygen vacancies for boosting photocatalytic selective conversion of aromatic alcohols. Catal. Sci. Technol. 2021, 11, 2939–2947. [Google Scholar] [CrossRef]
- Wang, K.; Lu, J.B.; Lu, Y.; Lau, C.H.; Zheng, Y.; Fan, X.F. Unravelling the CC coupling in CO2 photocatalytic reduction with H2O on Au/TiO2-x: Combination of plasmonic excitation and oxygen vacancy. Appl. Catal. B Environ. 2021, 292, 120147. [Google Scholar] [CrossRef]
- Wang, Y.X.; Zhang, Y.Y.; Zhu, X.J.; Liu, Y.; Wu, Z.B. Fluorine-induced oxygen vacancies on TiO2 nanosheets for photocatalytic indoor VOCs degradation. Appl. Catal. B Environ. 2022, 316, 121610. [Google Scholar] [CrossRef]
- Santara, B.; Giri, P.K.; Imakita, K.; Fujii, M. Evidence of oxygen vacancy induced room temperature ferromagnetism in solvothermally synthesized undoped TiO2nanoribbons. Nanoscale 2013, 5, 5476–5488. [Google Scholar] [CrossRef] [PubMed]
- Xiao, Z.; Ji, S.; Li, Y.; Hou, F.; Zhang, H.; Zhang, X.; Wang, L.; Li, G. Tuning oxygen vacancies on mesoporous ceria nanorods by metal doping: Controllable magnetic property. Appl. Surf. Sci. 2018, 455, 1037–1104. [Google Scholar] [CrossRef]
- Chi, M.; Sun, X.; Sujan, A.; Davis, Z.; Tatarchuk, B.J. A quantitative XPS examination of UV induced surface modification of TiO2 sorbents for the increased saturation capacity of sulfur heterocycles. Fuel 2019, 238, 446–454. [Google Scholar] [CrossRef]
- Mao, D.L.; Jia, M.M.; Qiu, J.H.; Zhang, X.F.; Yao, J.F. N-Doped porous carbon supported Au nanoparticles for benzyl alcohol oxidation. Catal. Lett. 2019, 150, 74–81. [Google Scholar] [CrossRef]
- Zhang, X.G.; Ke, X.B.; Zhu, H.Y. Zeolite-supported gold nanoparticles for selective photooxidation of aromatic alcohols under visible-light irradiation. Chem. Eur. J. 2012, 18, 8048–8056. [Google Scholar] [CrossRef]
- Savara, A.; Rossetti, I.; Chan, C.E.T.; Prati, L.; Villa, A. Microkinetic modeling of benzyl alcohol oxidation on carbon-supported Palladium nanoparticles. ChemCatChem 2016, 8, 2482–2491. [Google Scholar] [CrossRef]
- Zhu, Y.F.; Wei, Y.F. Solvent-controlled copper-catalyzed oxidation of benzylic alcohols to aldehydes and esters. Eur. J. Org. Chem. 2013, 2013, 4503–4508. [Google Scholar] [CrossRef]
- Mao, D.L.; Qiu, J.H.; Jia, M.M.; Zhang, X.F.; Yao, J.F. Platinum supported cellulose-based carbon with oxygen-containing functional groups for benzyl alcohol oxidation. J. Phys. Chem. Solids. 2019, 135, 109095. [Google Scholar] [CrossRef]
- Duarte, H.A.; Luggren, P.J.; Zelin, J.; Sad, M.E.; Díez, V.K.; Cosimo, J.I.D. Selective aerobic oxidation of benzyl alcohol on inexpensive and reusable ZnO/MnCO3 catalysts. Catal. Today 2021, 394–396, 178–189. [Google Scholar] [CrossRef]
- Manash, J.B.; Tonmoy, J.B.; Rupjyoti, D.; Subhasish, R.; Ankur Kanti, G.; Kusum, K.B. Fe(III) superoxide radicals in halloysite nanotubes for visible-light-assisted benzyl alcohol oxidation and oxidative CC coupling of 2-naphthol. Mol. Catal. 2021, 515, 111858. [Google Scholar]
- Gogoi, G.; Nath, J.K.; Hoque, N.; Biswas, S.; Gour, N.K.; Kalita, D.J.; Bora, S.R.; Bania, K.K. Single and multiple site Cu(II) catalysts for benzyl alcohol and catechol oxidation reactions. Appl. Catal. A Gen. 2022, 644, 118816. [Google Scholar] [CrossRef]
- Yang, D.J.; Liu, H.W.; Zheng, Z.F.; Yuan, Y.; Zhao, J.C.; Waclawik, E.R.; Ke, X.B.; Zhu, H.Y. An efficient photocatalyst structure: TiO2(B) nanofibers with a shell of anatase nanocrystals. J. Am. Chem. Soc. 2009, 131, 17885–17893. [Google Scholar] [CrossRef]
- Aguilar Tapia, A.; Delannoy, L.; Louis, C.; Han, C.W.; Ortalan, V.; Zanella, R. Selective hydrogenation of 1,3-butadiene over bimetallic Au-Ni/TiO2 catalysts prepared by deposition-precipitation with urea. J. Catal. 2016, 344, 515–523. [Google Scholar] [CrossRef]
- Marques, J.; Gomes, T.D.; Forte, M.A.; Silva, R.F.; Tavares, C.J. A new route for the synthesis of highly-active N-doped TiO2 nanoparticles for visible light photocatalysis using urea as nitrogen precursor. Catal. Today 2018, 326, 36–45. [Google Scholar] [CrossRef]
- Zope, B.N.; Hibbitts, D.D.; Neurock, M.; Davis, R.J. Reactivity of the Gold/Water Interface During Selective Oxidation Catalysis. Science 2010, 330, 74–78. [Google Scholar] [CrossRef]
- Skupien, E.; Berger, R.; Santos, V.; Gascon, J.; Makkee, M.; Kreutzer, M.; Kooyman, P.; Moulijn, J.; Kapteijn, F. Inhibition of a Gold-Based Catalyst in Benzyl Alcohol Oxidation: Understanding and Remediation. Catalysts 2014, 4, 89–115. [Google Scholar] [CrossRef]
- Marianna, B.; Guillermo Escolano, C.; Leonardo, P.; Lorenzo, M. Surface processes in selective photocatalytic oxidation of hydroxybenzyl alcohols by TiO2 P25. Catal. Today 2022, 413–415, 113983. [Google Scholar]
- Zhang, X.G.; Durndell, L.J.; Isaacs, M.A.; Parlett, C.M.A.; Lee, A.F.; Wilson, K. Platinum-catalyzed aqueous-phase hydrogenation of d-glucose to d-sorbitol. ACS Catal. 2016, 6, 7409–7417. [Google Scholar] [CrossRef]



| Catalyst | Au Loading/%wt | SBET/m2g−1 | Vpore/cm3·g−1 |
|---|---|---|---|
| Au/TiO2 | 0.64 | 8.8 | 0.03 |
| Au/NaTiNTs | 1.00 | 21.3 | 0.07 |
| Au/BC0.10@NaTiNTs | 0.90 | 20.2 | 0.07 |
| Au/BC0.25@NaTiNTs | 0.96 | 167.5 | 0.65 |
| Au/BC0.50@NaTiNTs | 0.86 | 9.2 | 0.03 |
| Au/BC1.00@NaTiNTs | 0.56 | 142.9 | 0.56 |
| Calcined in Air | Au/TiO2 | Au/NaTiNTs | Au/BC0.10 @NaTiNTs | Au/BC0.25 @NaTiNTs | Au/BC0.50 @NaTiNTs | Au/BC1.00 @NaTiNTs | |
|---|---|---|---|---|---|---|---|
| Con. a % | 200 °C | 30.5 | 29.9 | 27.5 | 11.4 | 10.0 | 6.0 |
| 400 °C | 31.7 | 29.3 | 25.1 | 24.0 | 22.8 | 17.9 | |
| 600 °C | 27.4 | 25.9 | 21.9 | 23.5 | 1.9 | 0.9 | |
| 800 °C | 34.3 | 10.6 | 17.8 | 24.2 | 4.3 | 7.5 | |
| Sel. a % | 200 °C | 78.7 | 51.9 | 72.5 | 93.0 | 99.0 | 99.0 |
| 400 °C | 74.4 | 58.4 | 67.0 | 55.9 | 92.8 | 90.5 | |
| 600 °C | 76.7 | 66.1 | 73.1 | 56.1 | 99.0 | 99.0 | |
| 800 °C | 73.0 | 69.5 | 54.3 | 79.5 | 99.0 | 99.0 | |
| Yield% | 200 °C | 24.0 | 15.5 | 19.9 | 10.6 | 9.9 | 6.0 |
| 400 °C | 23.6 | 17.1 | 16.8 | 13.4 | 21.2 | 16.2 | |
| 600 °C | 21.0 | 17.1 | 16.0 | 13.2 | 1.8 | 0.8 | |
| 800 °C | 25.0 | 7.4 | 9.6 | 19.2 | 4.3 | 7.4 |
| Solvent | Au/TiO2-400Ar-400Air | Au/BC0.25@NaTiNTs-400Ar-400Air | ||||
|---|---|---|---|---|---|---|
| Con. a/% | Sel. a/% | Con. a/% | Sel. a/% | |||
| Benzaldehyde | Others | Benzaldehyde | Others | |||
| Toluene | 43.4 | 74.6 | --b | 20.5 | 86.0 | -- b |
| DMF | 67.9 | 84.0 | Benzene: 16 | 4.1 | 99.0 | -- c |
| Hexane | 22.8 | 57.6 | Benzene: 37.9 | 10.7 | 99 | -- c |
| THF | 1.9 | 99 | --c | 2.2 | 99 | -- c |
| Ethanol | 32.7 | 5.2 | Benzoic acid: 88.8 | <1 | --b | -- b |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, X.; Jiang, H.; Cui, D.; Lu, K.; Kong, X.; Cai, J.; Yu, S.; Zhang, X. Selectivity Regulation of Au/Titanate by Biochar Modification for Selective Oxidation of Benzyl Alcohol. Catalysts 2023, 13, 864. https://doi.org/10.3390/catal13050864
Chen X, Jiang H, Cui D, Lu K, Kong X, Cai J, Yu S, Zhang X. Selectivity Regulation of Au/Titanate by Biochar Modification for Selective Oxidation of Benzyl Alcohol. Catalysts. 2023; 13(5):864. https://doi.org/10.3390/catal13050864
Chicago/Turabian StyleChen, Xiya, Hangwei Jiang, Danlan Cui, Kun Lu, Xiao Kong, Junmeng Cai, Shirui Yu, and Xingguang Zhang. 2023. "Selectivity Regulation of Au/Titanate by Biochar Modification for Selective Oxidation of Benzyl Alcohol" Catalysts 13, no. 5: 864. https://doi.org/10.3390/catal13050864
APA StyleChen, X., Jiang, H., Cui, D., Lu, K., Kong, X., Cai, J., Yu, S., & Zhang, X. (2023). Selectivity Regulation of Au/Titanate by Biochar Modification for Selective Oxidation of Benzyl Alcohol. Catalysts, 13(5), 864. https://doi.org/10.3390/catal13050864






