Development of a Graphene Oxide-Supported N-Heterocyclic Carbene Copper(I) Complex as a Heterogeneous Catalyst for the Selective N-Monoalkylation of Amines
Abstract
1. Introduction
2. Results and Discussion
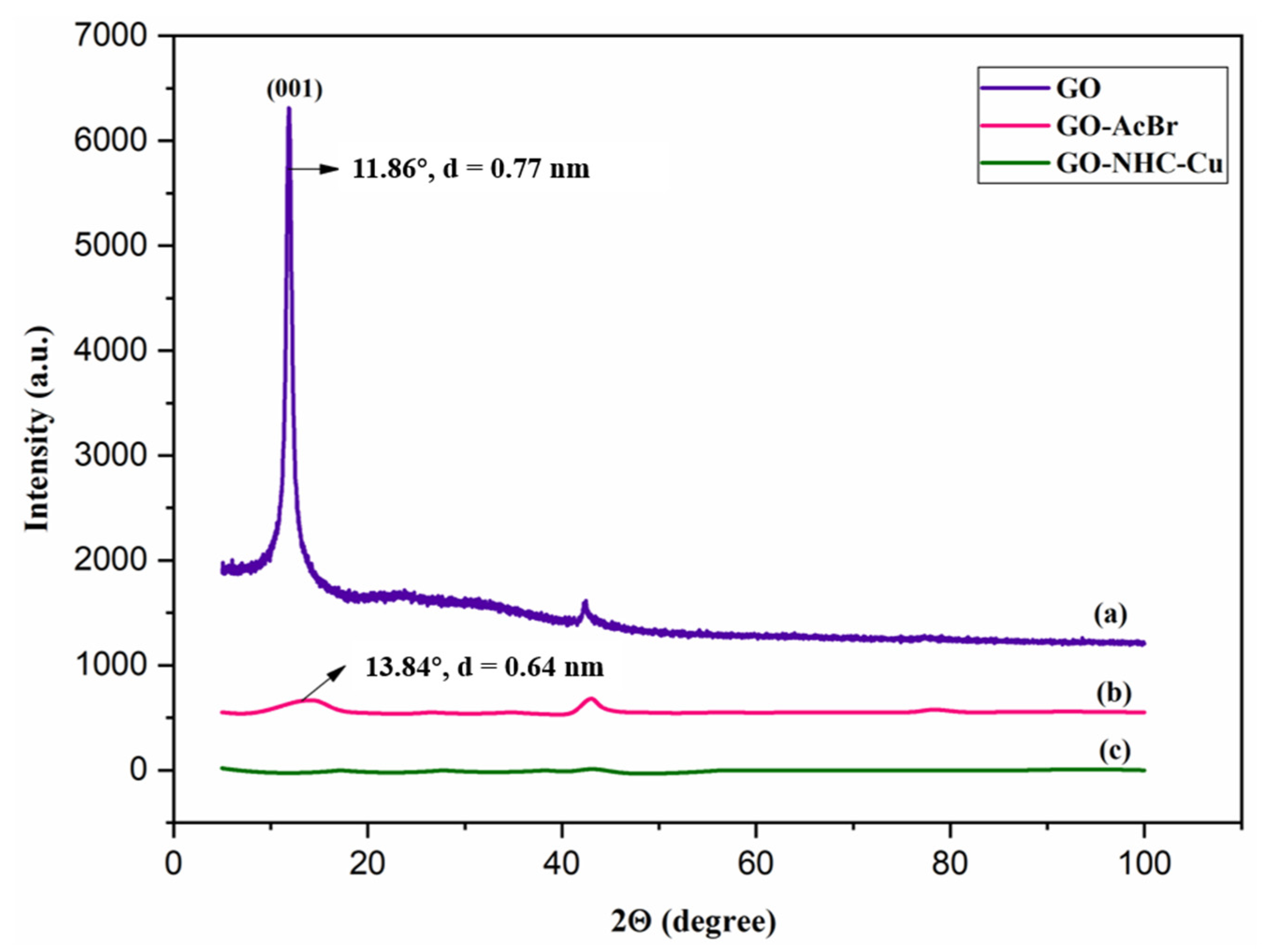
2.1. Characterisation of GO-NH--Cu
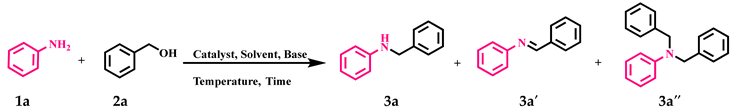
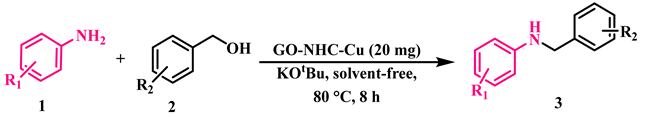
2.2. Catalytic Application of GO-NHC-Cu in the N-Alkylation of Amines with Alcohols
2.3. Discussion on the Reaction Mechanism
2.4. Plausible Mechanism
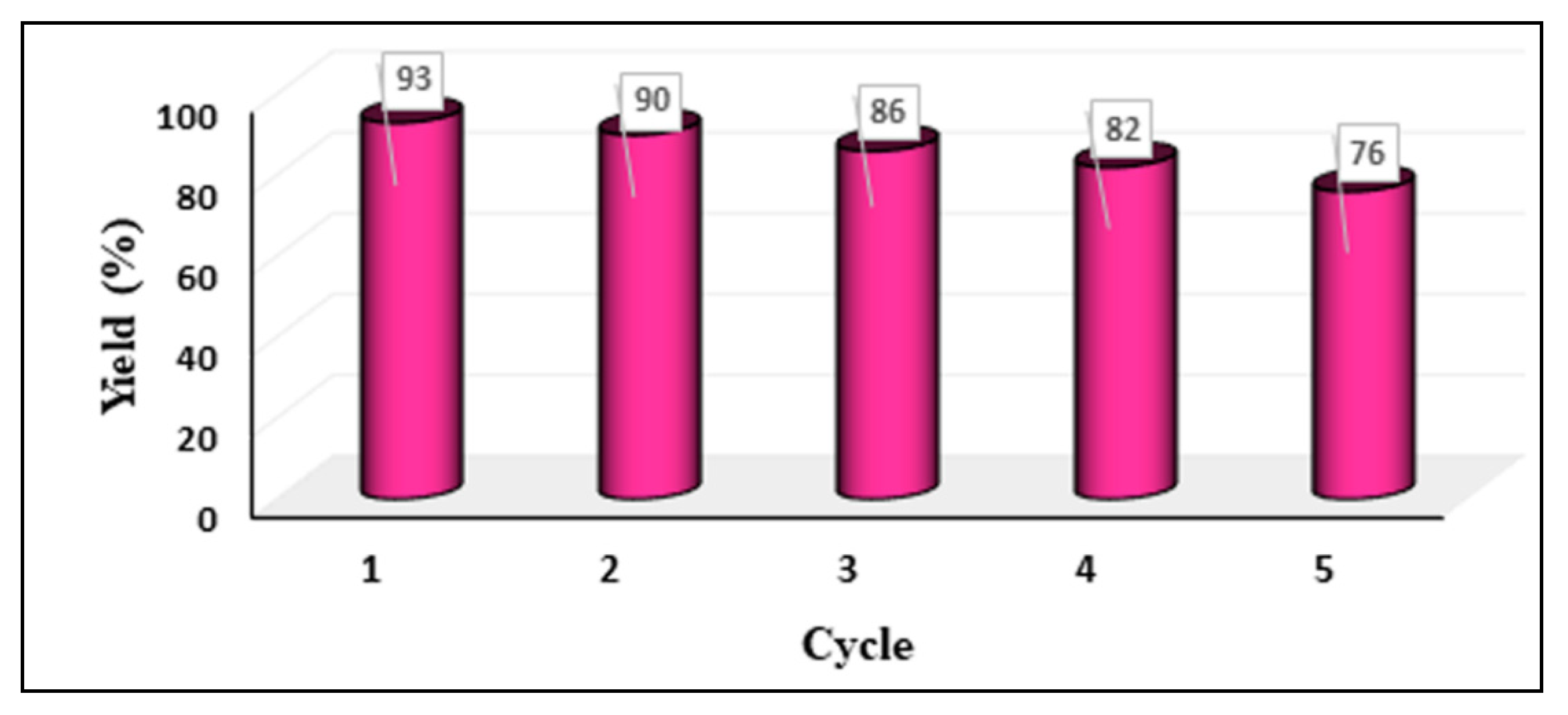
2.5. Recyclability and Hot Filtration Test
2.6. Comparison of GO-NHC-Cu with Reported Heterogeneous Catalysts
2.7. Calculation of Green Chemistry Metrics
3. Experimental Section
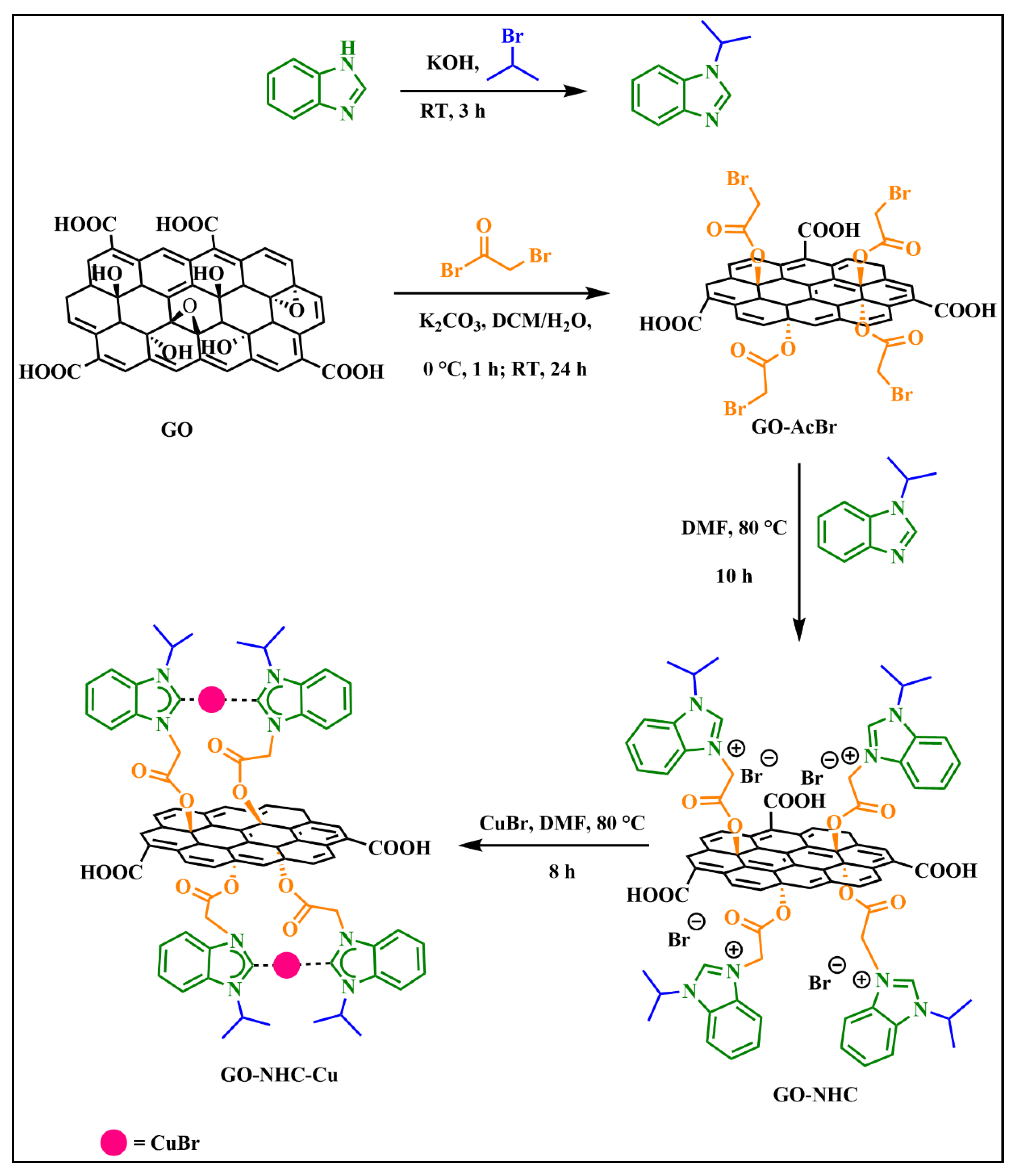
3.1. Synthesis of GO and GO-AcBr
3.2. Synthesis of GO-NHC and GO-NHC-Cu
3.3. N-Monoalkylation of Amines with Alcohol Using GO-NHC-Cu
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Vitaku, E.; Smith, D.T.; Njardarson, J.T. Analysis of the Structural Diversity, Substitution Patterns, and Frequency of Nitrogen Heterocycles among U.S. FDA Approved Pharmaceuticals. J. Med. Chem. 2014, 57, 10257–10274. [Google Scholar] [CrossRef] [PubMed]
- Kaloğlu, M.; Gürbüz, N.; Sémeril, D.; Özdemir, İ. Ruthenium(II)-(p-Cymene)-N-Heterocyclic Carbene Complexes for the N-Alkylation of Amine Using the Green Hydrogen Borrowing Methodology. Eur. J. Inorg. Chem. 2018, 2018, 1236–1243. [Google Scholar] [CrossRef]
- Guillena, G.; Ramón, D.J.; Yus, M. Hydrogen Autotransfer in the N-Alkylation of Amines and Related Compounds Using Alcohols and Amines as Electrophiles. Chem. Rev. 2010, 110, 1611–1641. [Google Scholar] [CrossRef]
- Salvatore, R.N.; Yoon, C.H.; Jung, K.W. Synthesis of Secondary Amines. Tetrahedron 2001, 57, 7785–7811. [Google Scholar] [CrossRef]
- Yan, L.; Liu, X.X.; Fu, Y. N-Alkylation of Amines with Phenols over Highly Active Heterogeneous Palladium Hydride Catalysts. RSC Adv. 2016, 6, 109702–109705. [Google Scholar] [CrossRef]
- Ju, Y.; Varma, R.S. Aqueous N-Alkylation of Amines Using Alkyl Halides: Direct Generation of Tertiary Amines under Microwave Irradiation. Green Chem. 2004, 6, 219–221. [Google Scholar] [CrossRef]
- Bissember, A.C.; Lundgren, R.J.; Creutz, S.E.; Peters, J.C.; Fu, G.C. Transition-Metal-Catalyzed Alkylations of Amines with Alkyl Halides: Photoinduced, Copper-Catalyzed Couplings of Carbazoles. Angew. Chem. Int. Ed. 2013, 52, 5129–5133. [Google Scholar] [CrossRef]
- Tararov, V.I.; Börner, A. Approaching Highly Enantioselective Reductive Animation. Synlett 2005, 2005, 203–211. [Google Scholar] [CrossRef]
- Reed-Berendt, B.G.; Latham, D.E.; Dambatta, M.B.; Morrill, L.C. Borrowing Hydrogen for Organic Synthesis. ACS Cent. Sci. 2021, 7, 570–585. [Google Scholar] [CrossRef]
- Irrgang, T.; Kempe, R. Transition-Metal-Catalyzed Reductive Amination Employing Hydrogen. Chem. Rev. 2020, 120, 9583–9674. [Google Scholar] [CrossRef]
- Sankar, V.; Kathiresan, M.; Sivakumar, B.; Mannathan, S. Zinc-Catalyzed N-Alkylation of Aromatic Amines with Alcohols: A Ligand-Free Approach. Adv. Synth. Catal. 2020, 362, 4409–4414. [Google Scholar] [CrossRef]
- Bains, A.K.; Kundu, A.; Yadav, S.; Adhikari, D. Borrowing Hydrogen-Mediated N-Alkylation Reactions by a Well-Defined Homogeneous Nickel Catalyst. ACS Catal. 2019, 9, 9051–9059. [Google Scholar] [CrossRef]
- Irrgang, T.; Kempe, R. 3d-Metal Catalyzed N- and C-Alkylation Reactions via Borrowing Hydrogen or Hydrogen Autotransfer. Chem. Rev. 2019, 119, 2524–2549. [Google Scholar] [CrossRef] [PubMed]
- Dang, T.T.; Shan, S.P.; Ramalingam, B.; Seayad, A.M. An Efficient Heterogenized Palladium Catalyst for N-Alkylation of Amines and α-Alkylation of Ketones Using Alcohols. RSC Adv. 2015, 5, 42399–42406. [Google Scholar] [CrossRef]
- Prades, A.; Corberán, R.; Poyatos, M.; Peris, E. [IrCl2Cp*(NHC)] Complexes as Highly Versatile Efficient Catalysts for the Cross-Coupling of Alcohols and Amines. Chem. A Eur. J. 2008, 14, 11474–11479. [Google Scholar] [CrossRef] [PubMed]
- Agrawal, S.; Lenormand, M.; Martín-Matute, B. Selective Alkylation of (Hetero)Aromatic Amines with Alcohols Catalyzed by a Ruthenium Pincer Complex. Org. Lett. 2012, 14, 1456–1459. [Google Scholar] [CrossRef] [PubMed]
- Putro, W.S.; Hara, T.; Ichikuni, N.; Shimazu, S. One-Pot Synthesis of Aniline N-Alkylation from Benzyl Alcohol over Cu-Fe Catalyst. Appl. Catal. A Gen. 2020, 602, 117519. [Google Scholar] [CrossRef]
- Lu, G.P.; Shan, H.; Lin, Y.; Zhang, K.; Zhou, B.; Zhong, Q.; Wang, P. A Fe Single Atom on N,S-Doped Carbon Catalyst for Performing N-Alkylation of Aromatic Amines under Solvent-Free Conditions. J. Mater. Chem. A Mater. 2021, 9, 25128–25135. [Google Scholar] [CrossRef]
- Li, Q.Q.; Xiao, Z.F.; Yao, C.Z.; Zheng, H.X.; Kang, Y.B. Direct Alkylation of Amines with Alcohols Catalyzed by Base. Org. Lett. 2015, 17, 5328–5331. [Google Scholar] [CrossRef]
- Allen, S.E.; Walvoord, R.R.; Padilla-Salinas, R.; Kozlowski, M.C. Aerobic Copper-Catalyzed Organic Reactions. Chem. Rev. 2013, 113, 6234–6458. [Google Scholar] [CrossRef]
- Pawar, R.C.; Choi, D.H.; Lee, J.S.; Lee, C.S. Formation of Polar Surfaces in Microstructured ZnO by Doping with Cu and Applications in Photocatalysis Using Visible Light. Mater. Chem. Phys. 2015, 151, 167–180. [Google Scholar] [CrossRef]
- Shih, Z.Y.; Periasamy, A.P.; Hsu, P.C.; Chang, H.T. Synthesis and Catalysis of Copper Sulfide/Carbon Nanodots for Oxygen Reduction in Direct Methanol Fuel Cells. Appl. Catal. B 2013, 132, 363–369. [Google Scholar] [CrossRef]
- Aneeja, T.; Neetha, M.; Afsina, C.M.A.; Anilkumar, G. Progress and Prospects in Copper-Catalyzed C–H Functionalization. RSC Adv. 2020, 10, 34429–34458. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.; Wu, X.; Sun, L. Copper-Based Homogeneous and Heterogeneous Catalysts for Electrochemical Water Oxidation. Nanoscale 2020, 12, 4187–4218. [Google Scholar] [CrossRef]
- Koy, M.; Bellotti, P.; Das, M.; Glorius, F. N-Heterocyclic Carbenes as Tunable Ligands for Catalytic Metal Surfaces. Nat. Catal. 2021, 4, 352–363. [Google Scholar] [CrossRef]
- Ranganath, K.V.S.; Onitsuka, S.; Kumar, A.K.; Inanaga, J. Recent Progress of N-Heterocyclic Carbenes in Heterogeneous Catalysis. Catal. Sci. Technol. 2013, 3, 2161–2181. [Google Scholar] [CrossRef]
- Jalal, M.; Hammouti, B.; Touzani, R.; Aouniti, A.; Ozdemir, I. Metal-NHC Heterocycle Complexes in Catalysis and Biological Applications: Systematic Review. Mater. Today Proc. 2020, 31, S122–S129. [Google Scholar] [CrossRef]
- Lazreg, F.; Nahra, F.; Cazin, C.S.J. Copper–NHC Complexes in Catalysis. Coord. Chem. Rev. 2015, 293–294, 48–79. [Google Scholar] [CrossRef]
- Herrmann, W.A.; Köcher, C. N-Heterocyclic Carbenes. Angew. Chem. Int. Ed. Engl. 1997, 36, 2162–2187. [Google Scholar] [CrossRef]
- Peris, E.; Crabtree, R.H. Recent Homogeneous Catalytic Applications of Chelate and Pincer N-Heterocyclic Carbenes. Coord. Chem. Rev. 2004, 248, 2239–2246. [Google Scholar] [CrossRef]
- Lee, Y.; Hoveyda, A.H. Efficient Boron-Copper Additions to Aryl-Substituted Alkenes Promoted by NHC-Based Catalysts. Enantioselective Cu-Catalyzed Hydroboration Reactions. J. Am. Chem. Soc. 2009, 131, 3160–3161. [Google Scholar] [CrossRef] [PubMed]
- Liu, P.; Yang, J.; Li, P.; Wang, L. An Efficient and Recyclable Silica-Supported Carbene–Cu(II) Catalyst for the Oxidative Coupling Reaction of Terminal Alkynes with H-Phosphonates under Base-Free Reaction Conditions. Appl. Organomet. Chem. 2011, 25, 830–835. [Google Scholar] [CrossRef]
- Kaur, H.; Zinn, F.K.; Stevens, E.D.; Nolan, S.P. (NHC) CuI (NHC = N-Heterocyclic Carbene) Complexes as Efficient Catalysts for the Reduction of Carbonyl Compounds. Organometallics 2004, 23, 1157–1160. [Google Scholar] [CrossRef]
- Ohishi, T.; Zhang, L.; Nishiura, M.; Hou, Z. Carboxylation of Alkylboranes by N-Heterocyclic Carbene Copper Catalysts: Synthesis of Carboxylic Acids from Terminal Alkenes and Carbon Dioxide. Angew. Chem. Int. Ed. 2011, 50, 8114–8117. [Google Scholar] [CrossRef] [PubMed]
- Neshat, A.; Mastrorilli, P.; Mobarakeh, A.M. Recent Advances in Catalysis Involving Bidentate N-Heterocyclic Carbene Ligands. Molecules 2021, 27, 95. [Google Scholar] [CrossRef]
- Fadhel, A.Z.; Pollet, P.; Liotta, C.L.; Eckert, C.A. Combining the Benefits of Homogeneous and Heterogeneous Catalysis with Tunable Solvents and Nearcritical Water. Molecules 2010, 15, 8400–8424. [Google Scholar] [CrossRef]
- Brisebois, P.P.; Siaj, M. Harvesting Graphene Oxide—Years 1859 to 2019: A Review of Its Structure, Synthesis, Properties and Exfoliation. J. Mater. Chem. C 2020, 8, 1517–1547. [Google Scholar] [CrossRef]
- Cheng, Z.; Liao, J.; He, B.; Zhang, F.; Zhang, F.; Huang, X.; Zhou, L. One-Step Fabrication of Graphene Oxide Enhanced Magnetic Composite Gel for Highly Efficient Dye Adsorption and Catalysis. ACS Sustain. Chem. Eng. 2015, 3, 1677–1685. [Google Scholar] [CrossRef]
- Shangguan, Q.; Chen, Z.; Yang, H.; Cheng, S.; Yang, W.; Yi, Z.; Wu, X.; Wang, S.; Yi, Y.; Wu, P. Design of Ultra-Narrow Band Graphene Refractive Index Sensor. Sensors 2022, 22, 6483. [Google Scholar] [CrossRef]
- Chen, H.; Chen, Z.; Yang, H.; Wen, L.; Yi, Z.; Zhou, Z.; Dai, B.; Zhang, J.; Wu, X.; Wu, P. Multi-Mode Surface Plasmon Resonance Absorber Based on Dart-Type Single-Layer Graphene. RSC Adv. 2022, 12, 7821–7829. [Google Scholar] [CrossRef]
- Zhang, Z.; Cai, R.; Long, F.; Wang, J. Development and Application of Tetrabromobisphenol A Imprinted Electrochemical Sensor Based on Graphene/Carbon Nanotubes Three-Dimensional Nanocomposites Modified Carbon Electrode. Talanta 2015, 134, 435–442. [Google Scholar] [CrossRef] [PubMed]
- Biswas, A.; Banerjee, A. Tailored Synthesis of Various Nanomaterials by Using a Graphene-Oxide-Based Gel as a Nanoreactor and Nanohybrid-Catalyzed C-C Bond Formation. Chem. Asian J. 2014, 9, 3451–3456. [Google Scholar] [CrossRef] [PubMed]
- Navalón, S.; Herance, J.R.; Álvaro, M.; García, H. Covalently Modified Graphenes in Catalysis, Electrocatalysis and Photoresponsive Materials. Chem. A Eur. J. 2017, 23, 15244–15275. [Google Scholar] [CrossRef] [PubMed]
- Kandathil, V.; Siddiqa, A.; Patra, A.; Kulkarni, B.; Kempasiddaiah, M.; Sasidhar, B.S.; Patil, S.A.; Rout, C.S.; Patil, S.A. NHC-Pd Complex Heterogenized on Graphene Oxide for Cross-Coupling Reactions and Supercapacitor Applications. Appl. Organomet. Chem. 2020, 34, e5924. [Google Scholar] [CrossRef]
- Park, J.H.; Raza, F.; Jeon, S.J.; Kim, H.I.; Kang, T.W.; Yim, D.; Kim, J.H. Recyclable N-Heterocyclic Carbene/Palladium Catalyst on Graphene Oxide for the Aqueous-Phase Suzuki Reaction. Tetrahedron Lett. 2014, 55, 3426–3430. [Google Scholar] [CrossRef]
- Qian, Y.; So, J.; Jung, S.Y.; Hwang, S.; Jin, M.J.; Shim, S.E. A Graphene Oxide Nanosheet Supported NHC–Palladium Complex as a Highly Efficient and Recyclable Suzuki Coupling Catalyst. Synthesis 2019, 51, 2287–2292. [Google Scholar] [CrossRef]
- Coman, S.M.; Podolean, I.; Tudorache, M.; Cojocaru, B.; Parvulescu, V.I.; Puche, M.; Garcia, H. Graphene Oxide as a Catalyst for the Diastereoselective Transfer Hydrogenation in the Synthesis of Prostaglandin Derivatives. Chem. Commun. 2017, 53, 10271–10274. [Google Scholar] [CrossRef]
- Ding, J.; Tang, S.; Chen, X.; Ding, M.; Kang, J.; Wu, R.; Fu, Z.; Jin, Y.; Li, L.; Feng, X.; et al. Introduction of Benzotriazole into Graphene Oxide for Highly Selective Coadsorption of An and Ln: Facile Synthesis and Theoretical Study. Chem. Eng. J. 2018, 344, 594–603. [Google Scholar] [CrossRef]
- Pawar, A.; Gajare, S.; Jagdale, A.; Patil, S.; Chandane, W.; Rashinkar, G.; Patil, S. Supported NHC-Benzimi@Cu Complex as a Magnetically Separable and Reusable Catalyst for the Multicomponent and Click Synthesis of 1,4-Disubstituted 1,2,3-Triazoles via Huisgen 1,3-Dipolar Cycloaddition. Catal. Lett. 2022, 152, 1854–1868. [Google Scholar] [CrossRef]
- Vinoth, R.; Babu, S.G.; Bharti, V.; Gupta, V.; Navaneethan, M.; Bhat, S.V.; Muthamizhchelvan, C.; Ramamurthy, P.C.; Sharma, C.; Aswal, D.K.; et al. Ruthenium Based Metallopolymer Grafted Reduced Graphene Oxide as a New Hybrid Solar Light Harvester in Polymer Solar Cells. Sci. Rep. 2017, 7, 43133. [Google Scholar] [CrossRef]
- Gou, F.; Liu, J.; Ye, N.; Jiang, X.; Qi, C. Cobalt-Porphyrin Modified Graphene Oxide as a Heterogeneous Catalyst for Solvent-Free CO2 Fixation to Cyclic Carbonates. J. CO2 Util. 2021, 48, 101534. [Google Scholar] [CrossRef]
- Zhao, Q.; Bai, C.; Zhang, W.; Li, Y.; Zhang, G.; Zhang, F.; Fan, X. Catalytic Epoxidation of Olefins with Graphene Oxide Supported Copper (Salen) Complex. Ind. Eng. Chem. Res. 2014, 53, 4232–4238. [Google Scholar] [CrossRef]
- Sharma, R.K.; Sharma, A.; Sharma, S.; Dutta, S.; Yadav, S.; Arora, B. Design and Exploration of Catalytic Activity of Two-Dimensional Surface-Engineered Graphene Oxide Nanosheets in the Transannulation of N-Heterocyclic Aldehydes or Ketones with Alkylamines. ACS Omega 2019, 4, 3146–3158. [Google Scholar] [CrossRef]
- Prabha, D.; Pachisia, S.; Gupta, R. Cobalt Mediated N-Alkylation of Amines by Alcohols: Role of Hydrogen Bonding Pocket. Inorg. Chem. Front. 2021, 8, 1599–1609. [Google Scholar] [CrossRef]
- Wang, L.; Jv, X.; Wang, R.; Ma, L.; Liu, J.; Sun, J.; Shi, T.; Zhao, L.; Zhang, X.; Wang, B. Highly Selective Synergistic N-Alkylation of Amines with ROH Catalyzed by Nickel–Ruthenium. ACS Sustain. Chem. Eng. 2022, 10, 8342–8349. [Google Scholar] [CrossRef]
- Verma, S.; Kujur, S.; Sharma, R.; Pathak, D.D. Cucurbit[6]Uril-Supported Fe3O4 Magnetic Nanoparticles Catalyzed Green and Sustainable Synthesis of 2-Substituted Benzimidazoles via Acceptorless Dehydrogenative Coupling. ACS Omega 2022, 7, 9754–9764. [Google Scholar] [CrossRef]
- Nallagangula, M.; Sujatha, C.; Bhat, V.T.; Namitharan, K. A Nanoscale Iron Catalyst for Heterogeneous Direct N- and C-Alkylations of Anilines and Ketones Using Alcohols under Hydrogen Autotransfer Conditions. Chem. Commun. 2019, 55, 8490–8493. [Google Scholar] [CrossRef]
- Goyal, V.; Sarki, N.; Poddar, M.K.; Narani, A.; Tripathi, D.; Ray, A.; Natte, K. Biorenewable Carbon-Supported Ru Catalyst for N -Alkylation of Amines with Alcohols and Selective Hydrogenation of Nitroarenes. New J. Chem. 2021, 45, 14687–14694. [Google Scholar] [CrossRef]
- Takahashi, Y.; Kondo, R.; Utsunomiya, M.; Suzuki, T.; Takeshita, H.T.; Obora, Y. Ti−Pd Alloys as Heterogeneous Catalysts for the Hydrogen Autotransfer Reaction and Catalytic Improvement by Hydrogenation Effects. ChemCatChem 2019, 11, 2432–2437. [Google Scholar] [CrossRef]
- Rasero-Almansa, A.M.; Corma, A.; Iglesias, M.; Sánchez, F. Design of a Bifunctional Ir–Zr Based Metal–Organic Framework Heterogeneous Catalyst for the N-Alkylation of Amines with Alcohols. ChemCatChem 2014, 6, 1794–1800. [Google Scholar] [CrossRef]
- Kumar, G.; Mogha, N.K.; Kumar, M.; Masram, D.T. NiO Nanocomposites/RGO as a Heterogeneous Catalyst for Imidazole Scaffolds with Applications in Inhibiting the DNA Binding Activity. Dalton Trans. 2020, 49, 1963–1974. [Google Scholar] [CrossRef] [PubMed]
- Krishnamoorthy, K.; Veerapandian, M.; Yun, K.; Kim, S.-J. The Chemical and Structural Analysis of Graphene Oxide with Different Degrees of Oxidation. Carbon 2013, 53, 38–49. [Google Scholar] [CrossRef]
- Lin, Y.R.; Chiu, C.C.; Chiu, H.T.; Lee, D.S.; Lu, T.J. Bis-Benzimidazolium-Palladium System Catalyzed Suzuki-Miyaura Coupling Reaction of Aryl Bromides under Mild Conditions. Appl. Organomet. Chem. 2018, 32, e3896. [Google Scholar] [CrossRef]
- Li, Q.; Fan, S.J.; Sun, Q.; Tian, H.W.; Yu, X.C.; Xu, Q. Copper-Catalyzed N-alkylation of Amides and Amines with Alcohols Employing the Aerobic Relay Race Methodology. Org. Biomol. Chem. 2012, 10, 2966–2972. [Google Scholar] [CrossRef] [PubMed]
- Yu, X.; Jiang, L.; Li, Q.; Xie, Y.; Xu, Q. Palladium-Catalyzed N-Alkylation of Amides and Amines with Alcohols Employing the Aerobic Relay Race Methodology. Chin. J. Chem. 2012, 30, 2322–2332. [Google Scholar] [CrossRef]
- Liu, C.; Liao, S.; Li, Q.; Feng, S.; Sun, Q.; Yu, X.; Xu, Q. Discovery and Mechanistic Studies of a General Air-Promoted Metal-Catalyzed Aerobic N-Alkylation Reaction of Amides and Amines with Alcohols. J. Org. Chem 2011, 76, 5759–5773. [Google Scholar] [CrossRef] [PubMed]
- Lan, X.B.; Ye, Z.; Yang, C.; Li, W.; Liu, J.; Huang, M.; Liu, Y.; Ke, Z. Tungsten-Catalyzed Direct N-Alkylation of Anilines with Alcohols. ChemSusChem 2021, 14, 860–865. [Google Scholar] [CrossRef]
- Langde, V.G.; Mondal, A.; Kumar, V.; Nandakumar, A.; Balaram, E. Manganese Catalyzed N-Alkylation of Anilines with Alcohols: Ligand Enabled Selectivity. Org. Biomol. Chem. 2018, 16, 8175–8180. [Google Scholar] [CrossRef]
- Ramachandran, R.; Prakash, G.; Selvamurugan, S.; Viswanathamurthi, P.; Malecki, J.G.; Ramkumar, V. Efficient and Versatile Catalysis of N-Alkylation of Heterocyclic Amines with Alcohols and One-Pot Synthesis of 2-Aryl Substituted Benzazoles with Newly Designed Ruthenium (II) Complexes of PNS Thiosemicarbazones. Dalton Trans. 2014, 43, 7889–7902. [Google Scholar] [CrossRef]





| Sample | D-Band (cm−1) | G-Band (cm−1) | ID/IG |
|---|---|---|---|
| GO | 1353 | 1605 | 0.77 |
| GO-AcBr | 1348 | 1597 | 0.92 |
| GO-NHC | 1345 | 1595 | 1.05 |
| GO-NHC-Cu | 1340 | 1589 | 1.13 |
 | ||||||
| S.No. | Catalyst (mg) | Solvent | Base | Temperature (°C) | Time (h) | Yield b (%) |
|---|---|---|---|---|---|---|
| 1 | - | Toluene | KOtBu | 110 | 24 | - |
| 2 | GO (30 mg) | Toluene | KOtBu | 110 | 24 | - |
| 3 | GO-NHC-Cu (30 mg) | Toluene | KOtBu | 110 | 24 | 88/0/0 |
| 4 | GO-NHC-Cu (30 mg) | 1,4-dioxane | KOtBu | 101 | 24 | 62/0/0 |
| 5 | GO-NHC-Cu (30 mg) | THF | KOtBu | 66 | 24 | 40/0/0 |
| 6 | GO-NHC-Cu (30 mg) | DMSO | KOtBu | 120 | 24 | 29/0/0 |
| 7 | GO-NHC-Cu (30 mg) | - | KOtBu | 110 | 24 | 93/0/0 |
| 8 | GO-NHC-Cu (25 mg) | - | KOtBu | 110 | 24 | 93/0/0 |
| 9 | GO-NHC-Cu (20 mg) | - | KOtBu | 110 | 24 | 93/0/0 |
| 10 | GO-NHC-Cu (15 mg) | - | KOtBu | 110 | 24 | 85/0/0 |
| 11 | GO-NHC-Cu (20 mg) | - | KOH | 110 | 24 | 82/0/0 |
| 12 | GO-NHC-Cu (20 mg) | - | Cs2CO3 | 110 | 24 | 66/0/0 |
| 13 | GO-NHC-Cu (20 mg) | - | K2CO3 | 110 | 24 | 48/0/0 |
| 14 | GO-NHC-Cu (20 mg) | - | - | 110 | 24 | 41/0/0 |
| 15 | GO-NHC-Cu (20 mg) | - | KOtBu | 100 | 24 | 93/0/0 |
| 16 | GO-NHC-Cu (20 mg) | - | KOtBu | 90 | 24 | 93/0/0 |
| 17 | GO-NHC-Cu (20 mg) | - | KOtBu | 80 | 24 | 93/0/0 |
| 18 | GO-NHC-Cu (20 mg) | - | KOtBu | 70 | 24 | 75/0/0 |
| 19 | GO-NHC-Cu (20 mg) | - | KOtBu | 80 | 12 | 93/0/0 |
| 20 | GO-NHC-Cu (20 mg) | - | KOtBu | 80 | 10 | 93/0/0 |
| 21 | GO–NHC–Cu (20 mg) | - | KOtBu | 80 | 8 | 93/0/0 |
| 22 | GO-NHC-Cu (20 mg) | - | KOtBu | 80 | 7 | 89/0/0 |
| 23 | GO-NHC-Cu (20 mg) | - | KOtBu | 80 | 8 | trace c |
| 24 | CuBr (5 mg) | - | KOtBu | 80 | 24 | 10/30/0 |
 | ||
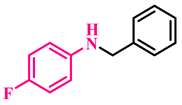
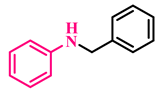 3a, 93% | 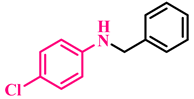 3b, 91% |  3c, 92% |
 3d, 88% | 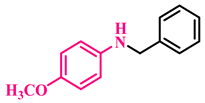 3e, 96% | 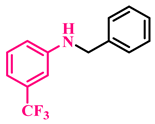 3f, 91% |
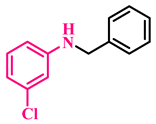 3g, 92% |  3h, 94% | 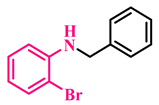 3i, 90% |
 3j, 87% |  3k, 91% | 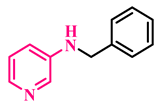 3l, 89% |
 3m, 90% | 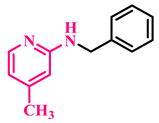 3n, 93% |  3o, 90% |
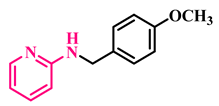 3p, 96% | 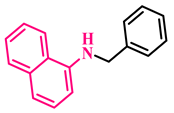 3q, 86% | 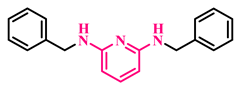 3r, 83% |
 3s, 95% | 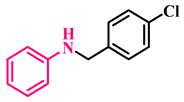 3t, 92% | |
| Entry | Catalyst | Conditions | Yield (%) | Reference |
|---|---|---|---|---|
| 1. | Nano Fe2O3 | KOH, toluene, 135 °C, 24 h | 83 | [57] |
| 2. | Ru/PNC-700 | KOtBu, THF, 150 °C, 48 h | 94 | [58] |
| 3. | Ti–Pd (Hy) | KOH, toluene, 135 °C, 48 h | 85 | [59] |
| 4. | Ir–Zr–MOF | 1–6 bar H2, 80 °C, microwave reactor, 2.5 h | 90 | [60] |
| 5. | GO-NHC-Cu | KOtBu, solvent-free, 80 °C, sealed tube, 8 h | 93 | This work |
| Entry | Metric | Calculated Value | Ideal Value |
|---|---|---|---|
| 1 | E-factor | 0.15 | 0 |
| 2 | Atom economy | 92.14% | 100% |
| 3 | Carbon efficiency | 95.9% | 100% |
| 4 | Process Mass intensity | 1.15 | 1 |
| 5 | Reaction mass efficiency | 86.80% | 100% |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kujur, S.; Verma, S.; Pathak, D.D. Development of a Graphene Oxide-Supported N-Heterocyclic Carbene Copper(I) Complex as a Heterogeneous Catalyst for the Selective N-Monoalkylation of Amines. Catalysts 2022, 12, 1458. https://doi.org/10.3390/catal12111458
Kujur S, Verma S, Pathak DD. Development of a Graphene Oxide-Supported N-Heterocyclic Carbene Copper(I) Complex as a Heterogeneous Catalyst for the Selective N-Monoalkylation of Amines. Catalysts. 2022; 12(11):1458. https://doi.org/10.3390/catal12111458
Chicago/Turabian StyleKujur, Shelly, Shruti Verma, and Devendra Deo Pathak. 2022. "Development of a Graphene Oxide-Supported N-Heterocyclic Carbene Copper(I) Complex as a Heterogeneous Catalyst for the Selective N-Monoalkylation of Amines" Catalysts 12, no. 11: 1458. https://doi.org/10.3390/catal12111458
APA StyleKujur, S., Verma, S., & Pathak, D. D. (2022). Development of a Graphene Oxide-Supported N-Heterocyclic Carbene Copper(I) Complex as a Heterogeneous Catalyst for the Selective N-Monoalkylation of Amines. Catalysts, 12(11), 1458. https://doi.org/10.3390/catal12111458







