Chalcogenides and Chalcogenide-Based Heterostructures as Photocatalysts for Water Splitting
Abstract
1. Introduction
2. General Synthesis Approaches of Chalcogenides
| Synthesized Materials | Synthesis Method | Metal Precursor Used | Source of Sulfur | Solvent Used | Ref. |
|---|---|---|---|---|---|
| AgInS2 | Low-temperature liquid method | AgNO3 In(NO3)3·xH2O | Thioglycollic acid Thioacetamide | Water | [26] |
| AgInS2 | Microwave | AgNO3 In(NO3)3∙ 4.5H2O | Sulfur powder | Glycerol, Oleic acid, Oleylamine, 1-dodecanethiol and 1-octadecene | [28] |
| Cd0.5Zn0.5S | Hydrothermal | Cd(CH3COO)2·2H2O Zn(CH3COO)2·2H2O | Na2S·9H2O | Water | [29] |
| CdS | Solvothermal | Cd(CH3COO)2·2H2O | Sulfur powder | Dodecylamine | [27] |
| Cu2WS4 | Hydrothermal | Cu(NO3)2· 3H2O Na2WO4 2H2O | L-Cysteine | Water | [22] |
| Cu2WSe4 | Hot injection | CuCl2·2H2O WCl4 | Se powder | Oleylamine | [23] |
| Cu2ZnSnS4 | Hot injection | Cu(acac)2 Zn(OAc)2 ·2H2O Sn(OAc)4 | 1-dodecylthiol and tert-dodecylthiol | 1-octadecene | [30] |
| CuCdS2 | Solvothermal | Copper nitrate Cadmium acetate | Sodium thiosulphate pentahydrate | Ethylene glycol | [24] |
| CuS | Hydrothermal | Copper acetate dihydrate | Thiourea | Water | [31] |
| MoS2 | One-pot liquid-phase reaction | (NH4)6Mo7O24 | Na2S | Water | [32] |
| NiS2 | Hydrothermal | Ni(NO3)3·6H2O | Thioacetamide | Water | [33] |
| VS2 | Single-step chemical vapor deposition | VCl3 | Sulphur powder | - | [34] |
| Zn0.5Cd0.5S | Combustion method | Zn(NO3)2⋅4H2O, Cd(NO3)2⋅6H2O | Thioacetamide | Water | [35] |
| ZnS | Co- precipitation | Zn(NO3)2 | Na2S | Water | [36] |
3. Binary Chalcogenides and Their Photocatalytic Water-Splitting Activities
3.1. Cd-Based Chalcogenides
3.2. Cu-Based Chalcogenides
3.3. Ga-Based Chalcogenides
3.4. Mo-Based Chalcogenides
3.5. Sn-Based Chalcogenides
3.6. Ti-Based Chalcogenides
3.7. V-Based Chalcogenides
3.8. Zn-Based Chalcogenides
4. Ternary Chalcogenide Heterostructures for Water Splitting
5. Prospects of Chalcogenides and Chalcogenide-Based Heterostructures
- In order to further boost the activity and stability of chalcogenides for water splitting, it is important to create precise methods for obtaining pure phase, active interface, exposed active surface, optimized electronic structure, and enhanced electronic conductivity.
- Recent research has shown that chalcogenides are very promising materials for H2 evolution by photocatalytic water splitting. It is expected that, with further knowledge, controlled doping, surface engineering, and development of their performance can be further improved [101].
- Binary metal chalcogenides such as CdS and CdSe are unstable in acidic media and are also susceptible to photocorrosion. As such, potential replacements that are more stable in acidic media and that do not exhibit photocorrosion should be explored.
- Despite many recent studies on the use of ternary and quaternary chalcogenides as photocatalysts for H2 production, the exact cause of photocorrosion in these materials is yet to be explored in detail and should be researched thoroughly in order to synthesize highly stable, multifunctional chalcogenides.
- Preparation of chalcogenide using low-cost methods while can produce large quantities of products also require more attention. Optimization of different parameters in the synthetic reactions such as precursors, temperature, pH, and reaction time should be studied for optimal yield to facilitate the production of these materials at a commercial scale.
- Detailed studies on extending the lifetime of photo-generated carriers and suppressing recombination are required to improve the photocatalytic activity of these materials for broader applications. Several approaches that could be employed include coupling with other semiconductors, loading of noble metals, and doping with metal or non-metal ions.
6. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Khan, M.M.; Ansari, S.A.; Pradhan, D.; Ansari, M.O.; Han, D.H.; Lee, J.; Cho, M.H. Band gap engineered TiO2 nanoparticles for visible light induced photoelectrochemical and photocatalytic studies. J. Mater. Chem. A 2013, 2, 637–644. [Google Scholar] [CrossRef]
- Rahman, A.; Harunsani, M.H.; Tan, A.L.; Khan, M.M. Zinc oxide and zinc oxide-based nanostructures: Biogenic and phytogenic synthesis, properties and applications. Bioprocess Biosyst. Eng. 2021, 44, 1333–1372. [Google Scholar] [CrossRef]
- Matussin, S.; Harunsani, M.H.; Tan, A.L.; Khan, M.M. Plant-Extract-Mediated SnO2 Nanoparticles: Synthesis and Applications. ACS Sustain. Chem. Eng. 2020, 8, 3040–3054. [Google Scholar] [CrossRef]
- Khan, M.M.; Adil, S.F.; Al-Mayouf, A. Metal oxides as photocatalysts. J. Saudi Chem. Soc. 2015, 19, 462–464. [Google Scholar] [CrossRef]
- Khan, M.M.; Rahman, A.; Matussin, S.N. Recent Progress of Metal-Organic Frameworks and Metal-Organic Frameworks-Based Heterostructures as Photocatalysts. Nanomaterials 2022, 12, 2820. [Google Scholar] [CrossRef]
- Khan, M.M.; Pradhan, D.; Sohn, Y. Nanocomposites for Visible Light-Induced Photocatalysis; Springer: Berlin/Heidelberg, Germany, 2017. [Google Scholar] [CrossRef]
- Hojamberdiev, M.; Cai, Y.; Vequizo, J.J.M.; Khan, M.M.; Vargas, R.; Yubuta, K.; Yamakata, A.; Teshima, K.; Hasegawa, M. Binary flux-promoted formation of trigonal ZnIn2S4 layered crystals using ZnS-containing industrial waste and their photocatalytic performance for H2 production. Green Chem. 2018, 20, 3845–3856. [Google Scholar] [CrossRef]
- Matussin, S.N.; Rahman, A.; Khan, M.M. Role of Anions in the Synthesis and Crystal Growth of Selected Semiconductors. Front. Chem. 2022, 10, 881518. [Google Scholar] [CrossRef]
- Bouroushian, M. Chalcogens and Metal Chalcogenides. In Electrochemistry of Metal Chalcogenides; Monographs in Electrochemistry; Springer: Berlin/Heidelberg, Germany, 2010; pp. 1–56. [Google Scholar] [CrossRef]
- Rahman, A.; Khan, M.M. Chalcogenides as photocatalysts. New J. Chem. 2021, 45, 19622–19635. [Google Scholar] [CrossRef]
- Choi, Y.I.; Lee, S.; Kim, S.K.; Kim, Y.-I.; Cho, D.W.; Khan, M.M.; Sohn, Y. Fabrication of ZnO, ZnS, Ag-ZnS, and Au-ZnS microspheres for photocatalytic activities, CO oxidation and 2-hydroxyterephthalic acid synthesis. J. Alloys Compd. 2016, 675, 46–56. [Google Scholar] [CrossRef]
- Khan, M.E.; Cho, M.H. CdS-graphene Nanocomposite for Efficient Visible-light-driven Photocatalytic and Photoelectrochemical Applications. J. Colloid Interface Sci. 2016, 482, 221–232. [Google Scholar] [CrossRef]
- Khan, M.M. Chalcogenide-Based Nanomaterials as Photocatalysts; Elsevier: Amsterdam, The Netherlands, 2021. [Google Scholar] [CrossRef]
- Tian, M.-W.; Yuen, H.-C.; Yan, S.-R.; Huang, W.-L. The multiple selections of fostering applications of hydrogen energy by integrating economic and industrial evaluation of different regions. Int. J. Hydrogen Energy 2019, 44, 29390–29398. [Google Scholar] [CrossRef]
- Chen, Y.; Feng, X.; Liu, M.; Su, J.; Shen, S. Towards efficient solar-to-hydrogen conversion: Fundamentals and recent progress in copper-based chalcogenide photocathodes. Nanophotonics 2016, 5, 524–547. [Google Scholar] [CrossRef]
- Revankar, S.T. Nuclear hydrogen production. In Storage and Hybridization of Nuclear Energy: Techno-Economic Integration of Renewable and Nuclear Energy; Elsevier: Amsterdam, The Netherlands, 2018; pp. 49–117. [Google Scholar] [CrossRef]
- Yerga, R.M.N.; Alvarez-Galvan, M.C.; Del Valle, F.; De La Mano, J.A.V.; Fierro, J.L.G. Water Splitting on Semiconductor Catalysts under Visible-Light Irradiation. ChemSusChem 2009, 2, 471–485. [Google Scholar] [CrossRef] [PubMed]
- Maeda, K.; Domen, K. New Non-Oxide Photocatalysts Designed for Overall Water Splitting under Visible Light. J. Phys. Chem. C 2007, 111, 7851–7861. [Google Scholar] [CrossRef]
- Maeda, K.; Lu, D.; Domen, K. Direct Water Splitting into Hydrogen and Oxygen under Visible Light by using Modified TaON Photocatalysts with d0Electronic Configuration. Chem. A Eur. J. 2013, 19, 4986–4991. [Google Scholar] [CrossRef] [PubMed]
- Walter, M.G.; Warren, E.L.; McKone, J.R.; Boettcher, S.W.; Mi, Q.; Santori, E.A.; Lewis, N.S. Solar Water Splitting Cells. Chem. Rev. 2010, 110, 6446–6473. [Google Scholar] [CrossRef]
- Li, X.; Zuo, X.; Jiang, X.; Li, D.; Cui, B.; Liu, D. Enhanced photocatalysis for water splitting in layered tin chalcogenides with high carrier mobility. Phys. Chem. Chem. Phys. 2019, 21, 7559–7566. [Google Scholar] [CrossRef]
- Kannan, S.; Vinitha, P.; Mohanraj, K.; Sivakumar, G. Antibacterial studies of novel Cu2WS4 ternary chalcogenide synthesized by hydrothermal process. J. Solid State Chem. 2018, 258, 376–382. [Google Scholar] [CrossRef]
- Sarilmaz, A.; Can, M.; Ozel, F. Ternary copper tungsten selenide nanosheets synthesized by a facile hot-injection method. J. Alloys Compd. 2017, 699, 479–483. [Google Scholar] [CrossRef]
- Saravanan, K.; Selladurai, S.; Ananthakumar, S.; Suriakarthick, R. Solvothermal synthesis of copper cadmium sulphide (CuCdS2) nanoparticles and its structural, optical and morphological properties. Mater. Sci. Semicond. Process. 2019, 93, 345–356. [Google Scholar] [CrossRef]
- Khalil, A.A.I.; El-Gawad, A.-S.H.A.; Gadallah, A.-S. Impact of silver dopants on structural, morphological, optical, and electrical properties of copper-zinc sulfide thin films prepared via sol-gel spin coating method. Opt. Mater. 2020, 109, 110250. [Google Scholar] [CrossRef]
- Gu, X.; Tan, C.; He, L.; Guo, J.; Zhao, X.; Qi, K.; Yan, Y. Mn2+ doped AgInS2 photocatalyst for formaldehyde degradation and hydrogen production from water splitting by carbon tube enhancement. Chemosphere 2022, 304, 135292. [Google Scholar] [CrossRef]
- Wang, X.; Feng, Z.; Fan, D.; Fan, F.; Li, C. Shape-Controlled Synthesis of CdS Nanostructures via a Solvothermal Method. Cryst. Growth Des. 2010, 10, 5312–5318. [Google Scholar] [CrossRef]
- Hu, Z.; Chen, T.; Xie, Z.; Guo, C.; Jiang, W.; Chen, Y.; Xu, Y. Emission tunable AgInS2 quantum dots synthesized via microwave method for white light-emitting diodes application. Opt. Mater. 2022, 124, 111975. [Google Scholar] [CrossRef]
- Li, S.; Cai, M.; Liu, Y.; Wang, C.; Yan, R.; Chen, X. Constructing Cd0.5Zn0.5S/Bi2WO6 S-scheme heterojunction for boosted photocatalytic antibiotic oxidation and Cr(VI) reduction. Adv. Powder Mater. 2023, 2, 100073. [Google Scholar] [CrossRef]
- Thompson, M.J.; Ruberu, T.P.A.; Blakeney, K.J.; Torres, K.V.; Dilsaver, P.S.; Vela, J. Axial Composition Gradients and Phase Segregation Regulate the Aspect Ratio of Cu2ZnSnS4 Nanorods. J. Phys. Chem. Lett. 2013, 4, 3918–3923. [Google Scholar] [CrossRef][Green Version]
- Mandari, K.K.; Son, N.; Kang, M. CuS/Ag2O nanoparticles on ultrathin g-C3N4 nanosheets to achieve high performance solar hydrogen evolution. J. Colloid Interface Sci. 2022, 615, 740–751. [Google Scholar] [CrossRef]
- Yin, M.; Sun, J.; Li, Y.; Ye, Y.; Liang, K.; Fan, Y.; Li, Z. Efficient photocatalytic hydrogen evolution over MoS2/activated carbon composite sensitized by Erythrosin B under LED light irradiation. Catal. Commun. 2020, 142, 106029. [Google Scholar] [CrossRef]
- Yang, Z.; Xie, X.; Zhang, Z.; Yang, J.; Yu, C.; Dong, S.; Xiang, M.; Qin, H. NiS2@V2O5/VS2 ternary heterojunction for a high-performance electrocatalyst in overall water splitting. Int. J. Hydrogen Energy 2022, 47, 27338–27346. [Google Scholar] [CrossRef]
- Gopalakrishnan, S.; Paulraj, G.; Eswaran, M.K.; Ray, A.; Singh, N.; Jeganathan, K. VS2 wrapped Si nanowires as core-shell heterostructure photocathode for highly efficient photoelectrochemical water reduction performance. Chemosphere 2022, 302, 134708. [Google Scholar] [CrossRef]
- Tang, Y.; Li, X.; Zhang, D.; Pu, X.; Ge, B.; Huang, Y. Noble metal-free ternary MoS2/Zn0.5Cd0.5S/g-C3N4 heterojunction composite for highly efficient photocatalytic H2 production. Mater. Res. Bull. 2018, 110, 214–222. [Google Scholar] [CrossRef]
- Arai, T.; Senda, S.-I.; Sato, Y.; Takahashi, H.; Shinoda, K.; Jeyadevan, B.; Tohji, K. Cu-Doped ZnS Hollow Particle with High Activity for Hydrogen Generation from Alkaline Sulfide Solution under Visible Light. Chem. Mater. 2008, 20, 1997–2000. [Google Scholar] [CrossRef]
- Oliva, A. Formation of the band gap energy on CdS thin films growth by two different techniques. Thin Solid Films 2001, 391, 28–35. [Google Scholar] [CrossRef]
- Cheng, L.; Xiang, Q.; Liao, Y.; Zhang, H. CdS-Based photocatalysts. Energy Environ. Sci. 2018, 11, 1362–1391. [Google Scholar] [CrossRef]
- Li, C.; Yuan, J.; Han, B.; Shangguan, W. Synthesis and photochemical performance of morphology-controlled CdS photocatalysts for hydrogen evolution under visible light. Int. J. Hydrogen Energy 2011, 36, 4271–4279. [Google Scholar] [CrossRef]
- Garg, P.; Bhauriyal, P.; Mahata, A.; Rawat, K.S.; Pathak, B. Role of Dimensionality for Photocatalytic Water Splitting: CdS Nanotube versus Bulk Structure. ChemPhysChem 2018, 20, 383–391. [Google Scholar] [CrossRef]
- Luo, M.; Liu, Y.; Hu, J.; Liu, H.; Li, J. One-Pot Synthesis of CdS and Ni-Doped CdS Hollow Spheres with Enhanced Photocatalytic Activity and Durability. ACS Appl. Mater. Interfaces 2012, 4, 1813–1821. [Google Scholar] [CrossRef]
- Zhen, W.; Ning, X.; Yang, B.; Wu, Y.; Li, Z.; Lu, G. The enhancement of CdS photocatalytic activity for water splitting via anti-photocorrosion by coating Ni2P shell and removing nascent formed oxygen with artificial gill. Appl. Catal. B Environ. 2018, 221, 243–257. [Google Scholar] [CrossRef]
- Li, Y.-H.; Qi, M.-Y.; Li, J.-Y.; Tang, Z.-R.; Xu, Y.-J. Noble metal free CdS@CuS-NixP hybrid with modulated charge transfer for enhanced photocatalytic performance. Appl. Catal. B Environ. 2019, 257, 117934. [Google Scholar] [CrossRef]
- Yang, J.; Yan, H.; Wang, X.; Wen, F.; Wang, Z.; Fan, D.; Shi, J.; Li, C. Roles of cocatalysts in Pt–PdS/CdS with exceptionally high quantum efficiency for photocatalytic hydrogen production. J. Catal. 2012, 290, 151–157. [Google Scholar] [CrossRef]
- Yang, T.-T.; Chen, W.-T.; Hsu, Y.-J.; Wei, K.-H.; Lin, T.-Y. Interfacial Charge Carrier Dynamics in Core−Shell Au-CdS Nanocrystals. J. Phys. Chem. C 2010, 114, 11414–11420. [Google Scholar] [CrossRef]
- Tso, S.; Li, W.-S.; Wu, B.-H.; Chen, L.-J. Enhanced H2 production in water splitting with CdS-ZnO core-shell nanowires. Nano Energy 2017, 43, 270–277. [Google Scholar] [CrossRef]
- Zhang, F.; Zhuang, H.-Q.; Zhang, W.; Yin, J.; Cao, F.-H.; Pan, Y.-X. Noble-metal-free CuS/CdS photocatalyst for efficient visible-light-driven photocatalytic H2 production from water. Catal. Today 2018, 330, 203–208. [Google Scholar] [CrossRef]
- Cheng, F.; Xiang, Q. A solid-state approach to fabricate a CdS/CuS nano-heterojunction with promoted visible-light photocatalytic H2-evolution activity. RSC Adv. 2016, 6, 76269–76272. [Google Scholar] [CrossRef]
- Wu, Z.; Zhao, G.; Zhang, Y.-N.; Tian, H.; Li, D. Enhanced Photocurrent Responses and Antiphotocorrosion Performance of CdS Hybrid Derived from Triple Heterojunction. J. Phys. Chem. C 2012, 116, 12829–12835. [Google Scholar] [CrossRef]
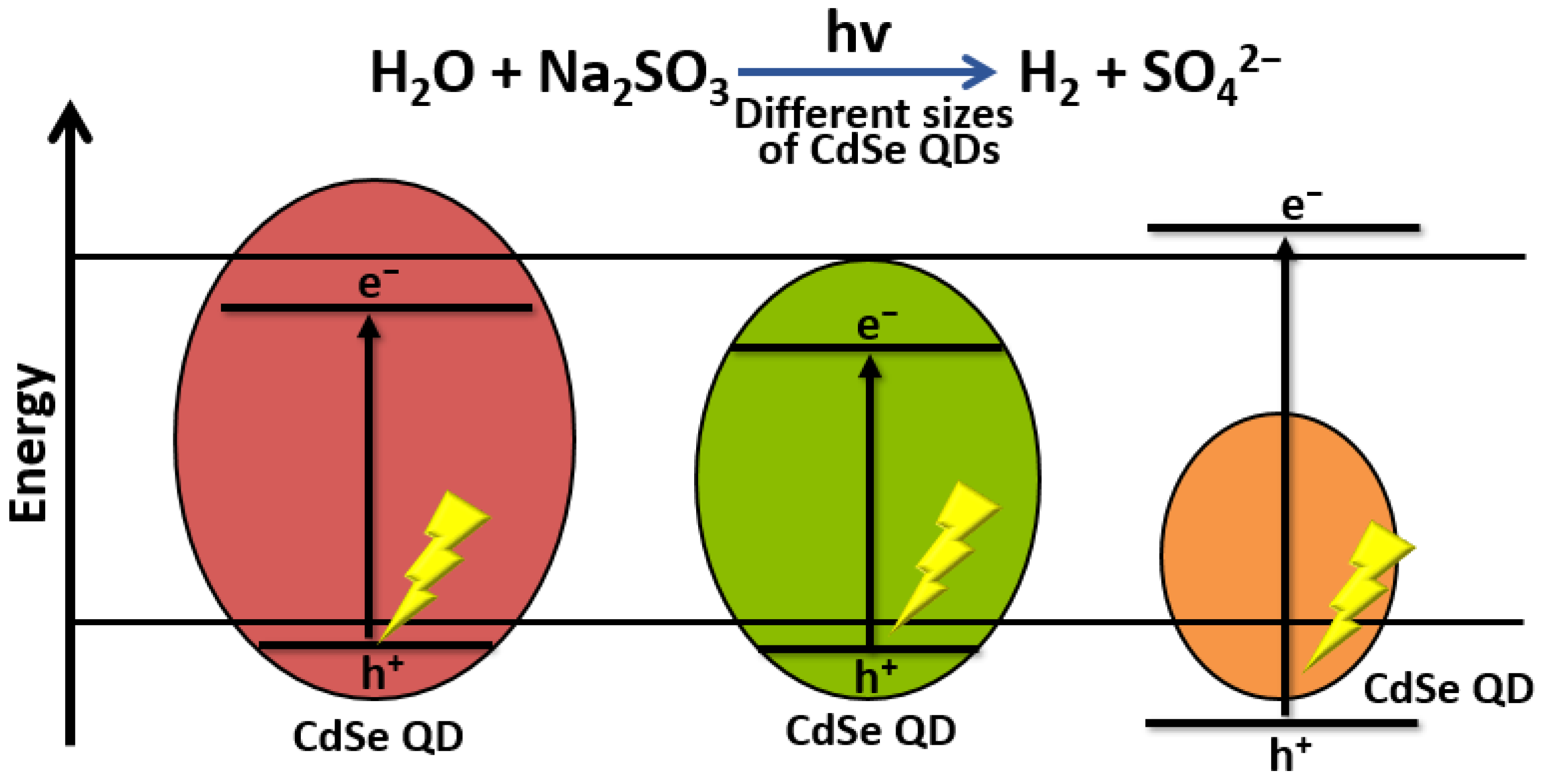
- Holmes, M.A.; Townsend, T.K.; Osterloh, F.E. Quantum confinement controlled photocatalytic water splitting by suspended CdSe nanocrystals. Chem. Commun. 2011, 48, 371–373. [Google Scholar] [CrossRef]
- Yang, Q.; Luo, M.; Liu, K.; Cao, H.; Yan, H. Covalent organic frameworks for photocatalytic applications. Appl. Catal. B Environ. 2020, 276, 119174. [Google Scholar] [CrossRef]
- You, J.; Zhao, Y.; Wang, L.; Bao, W. Recent developments in the photocatalytic applications of covalent organic frameworks: A review. J. Clean. Prod. 2021, 291, 125822. [Google Scholar] [CrossRef]
- You, D.; Pan, Z.; Cheng, Q. COFs-Ph@CdS S-scheme heterojunctions with photocatalytic hydrogen evolution and efficient degradation properties. J. Alloys Compd. 2023, 930, 167069. [Google Scholar] [CrossRef]
- Das, A.; Han, Z.; Haghighi, M.G.; Eisenberg, R. Photogeneration of hydrogen from water using CdSe nanocrystals demonstrating the importance of surface exchange. Proc. Natl. Acad. Sci. USA 2013, 110, 16716–16723. [Google Scholar] [CrossRef]
- Park, Y.; Park, B. Effect of ligand exchange on photocurrent enhancement in cadmium selenide (CdSe) quantum dot water splitting cells. Results Phys. 2018, 11, 162–165. [Google Scholar] [CrossRef]
- Zhou, W.; Li, F.; Yang, X.; Yang, W.; Wang, C.; Cao, R.; Zhou, C.; Tian, M. Peanut-chocolate-ball-inspired construction of the interface engineering between CdS and intergrown Cd: Boosting both the photocatalytic activity and photocorrosion resistance. J. Energy Chem. 2023, 76, 75–89. [Google Scholar] [CrossRef]
- Li, K.; Pan, H.; Wang, F.; Zhang, Z.; Min, S. In-situ exsolved NiS nanoparticle-socketed CdS with strongly coupled interfaces as a superior visible-light-driven photocatalyst for hydrogen evolution. Appl. Catal. B Environ. 2023, 321, 122028. [Google Scholar] [CrossRef]
- Basu, M.; Nazir, R.; Fageria, P.; Pande, S. Construction of CuS/Au Heterostructure through a Simple Photoreduction Route for Enhanced Electrochemical Hydrogen Evolution and Photocatalysis. Sci. Rep. 2016, 6, 34738. [Google Scholar] [CrossRef]
- Chandra, M.; Bhunia, K.; Pradhan, D. Controlled Synthesis of CuS/TiO2 Heterostructured Nanocomposites for Enhanced Photocatalytic Hydrogen Generation through Water Splitting. Inorg. Chem. 2018, 57, 4524–4533. [Google Scholar] [CrossRef]
- Dubale, A.A.; Tamirat, A.G.; Chen, H.-M.; Berhe, T.A.; Pan, C.-J.; Su, W.-N.; Hwang, B.-J. A highly stable CuS and CuS–Pt modified Cu2O/CuO heterostructure as an efficient photocathode for the hydrogen evolution reaction. J. Mater. Chem. A 2015, 4, 2205–2216. [Google Scholar] [CrossRef]
- Ma, L.; Yang, D.-J.; Song, X.-P.; Li, H.-X.; Ding, S.-J.; Xiong, L.; Qin, P.-L.; Chen, X.-B. Pt Decorated (Au Nanosphere)/(CuSe Ultrathin Nanoplate) Tangential Hybrids for Efficient Photocatalytic Hydrogen Generation via Dual-Plasmon-Induced Strong VIS–NIR Light Absorption and Interfacial Electric Field Coupling. Sol. RRL 2019, 4, 1900376. [Google Scholar] [CrossRef]
- Liang, H.; Mei, J.; Sun, H.; Cao, L. Enhanced photocatalytic hydrogen evolution of CdS@CuS core-shell nanorods under visible light. Mater. Sci. Semicond. Process. 2023, 153, 107105. [Google Scholar] [CrossRef]
- Hou, J.; Huang, B.; Kong, L.; Xie, Y.; Liu, Y.; Chen, M.; Wang, Q. One-pot hydrothermal synthesis of CdS–CuS decorated TiO2 NTs for improved photocatalytic dye degradation and hydrogen production. Ceram. Int. 2021, 47, 30860–30868. [Google Scholar] [CrossRef]
- Zhuang, H.L.; Hennig, R.G. Single-Layer Group-III Monochalcogenide Photocatalysts for Water Splitting. Chem. Mater. 2013, 25, 3232–3238. [Google Scholar] [CrossRef]
- Peng, Q.; Xiong, R.; Sa, B.; Zhou, J.; Wen, C.; Wu, B.; Anpo, M.; Sun, Z. Computational mining of photocatalysts for water splitting hydrogen production: Two-dimensional InSe-family monolayers. Catal. Sci. Technol. 2017, 7, 2744–2752. [Google Scholar] [CrossRef]
- Peng, Q.; Guo, Z.; Sa, B.; Zhou, J.; Sun, Z. New gallium chalcogenides/arsenene van der Waals heterostructures promising for photocatalytic water splitting. Int. J. Hydrogen Energy 2018, 43, 15995–16004. [Google Scholar] [CrossRef]
- Gupta, U.; Rao, C. Hydrogen generation by water splitting using MoS2 and other transition metal dichalcogenides. Nano Energy 2017, 41, 49–65. [Google Scholar] [CrossRef]
- Rahman, A.; Jennings, J.R.; Tan, A.L.; Khan, M.M. Molybdenum Disulfide-Based Nanomaterials for Visible-Light-Induced Photocatalysis. ACS Omega 2022, 7, 22089–22110. [Google Scholar] [CrossRef]
- Li, M.; Cui, Z.; Li, E. Silver-modified MoS2 nanosheets as a high-efficiency visible-light photocatalyst for water splitting. Ceram. Int. 2019, 45, 14449–14456. [Google Scholar] [CrossRef]
- Yuan, Y.-J.; Chen, D.; Zhong, J.; Yang, L.-X.; Wang, J.; Liu, M.-J.; Tu, W.-G.; Yu, Z.-T.; Zou, Z.-G. Interface engineering of a noble-metal-free 2D–2D MoS2/Cu-ZnIn2S4 photocatalyst for enhanced photocatalytic H2 production. J. Mater. Chem. A 2017, 5, 15771–15779. [Google Scholar] [CrossRef]
- Hassan, M.A.; Kim, M.-W.; Johar, M.A.; Waseem, A.; Kwon, M.-K.; Ryu, S.-W. Transferred monolayer MoS2 onto GaN for heterostructure photoanode: Toward stable and efficient photoelectrochemical water splitting. Sci. Rep. 2019, 9, 20141. [Google Scholar] [CrossRef]
- Zhao, G.; Wang, X.; Wang, S.; Rui, K.; Chen, Y.; Yu, H.; Ma, J.; Dou, S.X.; Sun, W. Heteroatom-doped MoSe 2 Nanosheets with Enhanced Hydrogen Evolution Kinetics for Alkaline Water Splitting. Chem. Asian J. 2018, 14, 301–306. [Google Scholar] [CrossRef]
- Lei, W.; Wang, F.; Pan, X.; Ye, Z.; Lu, B. Z-scheme MoO3-2D SnS nanosheets heterojunction assisted g-C3N4 composite for enhanced photocatalytic hydrogen evolutions. Int. J. Hydrogen Energy 2022, 47, 10877–10890. [Google Scholar] [CrossRef]
- Liu, Y.; Zhou, Y.; Zhou, X.; Jin, X.; Li, B.; Liu, J.; Chen, G. Cu doped SnS2 nanostructure induced sulfur vacancy towards boosted photocatalytic hydrogen evolution. Chem. Eng. J. 2020, 407, 127180. [Google Scholar] [CrossRef]
- Geng, Y.; Zou, X.; Lu, Y.; Wang, L. Fabrication of the SnS2/ZnIn2S4 heterojunction for highly efficient visible light photocatalytic H2 evolution. Int. J. Hydrogen Energy 2022, 47, 11520–11527. [Google Scholar] [CrossRef]
- Li, Y.-Y.; Wang, J.-G.; Sun, H.-H.; Hua, W.; Liu, X.-R. Heterostructured SnS2/SnO2 nanotubes with enhanced charge separation and excellent photocatalytic hydrogen production. Int. J. Hydrogen Energy 2018, 43, 14121–14129. [Google Scholar] [CrossRef]
- Mangiri, R.; Kumar, K.S.; Subramanyam, K.; Ratnakaram, Y.; Sudharani, A.; Reddy, D.A.; Vijayalakshmi, R. Boosting solar driven hydrogen evolution rate of CdS nanorods adorned with MoS2 and SnS2 nanostructures. Colloids Interface Sci. Commun. 2021, 43, 100437. [Google Scholar] [CrossRef]
- Barawi, M.; Flores, E.; Ferrer, I.J.; Ares, J.R.; Sánchez, C. Titanium trisulphide (TiS3) nanoribbons for easy hydrogen photogeneration under visible light. J. Mater. Chem. A 2015, 3, 7959–7965. [Google Scholar] [CrossRef]
- Flores, E.; Ares, J.; Sánchez, C.; Ferrer, I. Ternary transition titanium-niobium trisulfide as photoanode for assisted water splitting. Catal. Today 2018, 321–322, 107–112. [Google Scholar] [CrossRef]
- Guo, W.; Wu, D. Facile synthesis of VS4/graphene nanocomposites and their visible-light-driven photocatalytic water splitting activities. Int. J. Hydrogen Energy 2014, 39, 16832–16840. [Google Scholar] [CrossRef]
- Ikeda, S.; Aono, N.; Iwase, A.; Kobayashi, H.; Kudo, A. Cu3MS4 (M=V, Nb, Ta) and its Solid Solutions with Sulvanite Structure for Photocatalytic and Photoelectrochemical H2 Evolution under Visible-Light Irradiation. ChemSusChem 2018, 12, 1977–1983. [Google Scholar] [CrossRef]
- Li, G.; Deng, X.; Chen, P.; Wang, X.; Ma, J.; Liu, F.; Yin, S.-F. Sulphur vacancies-VS2@C3N4 drived by in situ supramolecular self-assembly for synergistic photocatalytic degradation of real wastewater and H2 production: Vacancies taming interfacial compact heterojunction and carriers transfer. Chem. Eng. J. 2022, 433, 134505. [Google Scholar] [CrossRef]
- Zhong, X.; Tang, J.; Wang, J.; Shao, M.; Chai, J.; Wang, S.; Yang, M.; Yang, Y.; Wang, N.; Wang, S.; et al. 3D heterostructured pure and N-Doped Ni3S2/VS2 nanosheets for high efficient overall water splitting. Electrochim. Acta 2018, 269, 55–61. [Google Scholar] [CrossRef]
- Kurnia, F.; Ng, Y.H.; Amal, R.; Valanoor, N.; Hart, J.N. Defect engineering of ZnS thin films for photoelectrochemical water-splitting under visible light. Sol. Energy Mater. Sol. Cells 2016, 153, 179–185. [Google Scholar] [CrossRef]
- Sánchez-Tovar, R.; Fernández-Domene, R.M.; Montañés, M.T.; Sanz-Marco, A.; Garcia-Antón, J. ZnO/ZnS heterostructures for hydrogen production by photoelectrochemical water splitting. RSC Adv. 2016, 6, 30425–30435. [Google Scholar] [CrossRef]
- Zhang, X.; Zhou, Y.-Z.; Wu, D.-Y.; Liu, X.-H.; Zhang, R.; Liu, H.; Dong, C.-K.; Yang, J.; Kulinich, S.A.; Du, X.-W. ZnO nanosheets with atomically thin ZnS overlayers for photocatalytic water splitting. J. Mater. Chem. A 2018, 6, 9057–9063. [Google Scholar] [CrossRef]
- Lee, G.-J.; Anandan, S.; Masten, S.J.; Wu, J.J. Photocatalytic hydrogen evolution from water splitting using Cu doped ZnS microspheres under visible light irradiation. Renew. Energy 2016, 89, 18–26. [Google Scholar] [CrossRef]
- Wu, Y.; Wang, H.; Tu, W.; Wu, S.; Chew, J.W. Effects of composition faults in ternary metal chalcogenides (Zn In2S3+, x = 1 − 5) layered crystals for visible-light-driven catalytic hydrogen generation and carbon dioxide reduction. Appl. Catal. B Environ. 2019, 256, 117810. [Google Scholar] [CrossRef]
- Fan, H.-T.; Wu, Z.; Liu, K.-C.; Liu, W.-S. Fabrication of 3D CuS@ZnIn2S4 hierarchical nanocages with 2D/2D nanosheet subunits p-n heterojunctions for improved photocatalytic hydrogen evolution. Chem. Eng. J. 2022, 433, 134474. [Google Scholar] [CrossRef]
- Ni, T.; Yang, Z.; Cao, Y.; Lv, H.; Liu, Y. Rational design of MoS2/g-C3N4/ZnIn2S4 hierarchical heterostructures with efficient charge transfer for significantly enhanced photocatalytic H2 production. Ceram. Int. 2021, 47, 22985–22993. [Google Scholar] [CrossRef]
- Guo, X.; Peng, Y.; Liu, G.; Xie, G.; Guo, Y.; Zhang, Y.; Yu, J. An Efficient ZnIn2S4@CuInS2 Core–Shell p–n Heterojunction to Boost Visible-Light Photocatalytic Hydrogen Evolution. J. Phys. Chem. C 2020, 124, 5934–5943. [Google Scholar] [CrossRef]
- Yang, W.; Ma, G.; Fu, Y.; Peng, K.; Yang, H.; Zhan, X.; Yang, W.; Wang, L.; Hou, H. Rationally designed Ti3C2 MXene@TiO2/CuInS2 Schottky/S-scheme integrated heterojunction for enhanced photocatalytic hydrogen evolution. Chem. Eng. J. 2021, 429, 132381. [Google Scholar] [CrossRef]
- Raja, A.; Son, N.; Swaminathan, M.; Kang, M. Facile synthesis of sphere-like structured ZnIn2S4-rGO-CuInS2 ternary heterojunction catalyst for efficient visible-active photocatalytic hydrogen evolution. J. Colloid Interface Sci. 2021, 602, 669–679. [Google Scholar] [CrossRef]
- Li, Q.; Qiao, X.-Q.; Jia, Y.; Hou, D.; Li, D.-S. Amorphous CoMoS4 Nanostructure for Photocatalytic H2 Generation, Nitrophenol Reduction, and Methylene Blue Adsorption. ACS Appl. Nano Mater. 2019, 3, 68–76. [Google Scholar] [CrossRef]
- Yao, Z.; Wang, L.; Zhang, Y.; Yu, Z.; Jiang, Z. Carbon nanotube modified Zn0.83Cd0.17S nanocomposite photocatalyst and its hydrogen production under visible-light. Int. J. Hydrogen Energy 2014, 39, 15380–15386. [Google Scholar] [CrossRef]
- Lv, H.; Kong, Y.; Gong, Z.; Zheng, J.; Liu, Y.; Wang, G. Engineering multifunctional carbon black interface over Mn0.5Cd0.5S nanoparticles/CuS nanotubes heterojunction for boosting photocatalytic hydrogen generation activity. Appl. Surf. Sci. 2022, 604, 154513. [Google Scholar] [CrossRef]
- Bai, Y.; Zhang, Q.; Xu, N.; Deng, K.; Kan, E. The Janus structures of group-III chalcogenide monolayers as promising photocatalysts for water splitting. Appl. Surf. Sci. 2019, 478, 522–531. [Google Scholar] [CrossRef]
- Hu, L.; Wei, D. Janus Group-III Chalcogenide Monolayers and Derivative Type-II Heterojunctions as Water-Splitting Photocatalysts with Strong Visible-Light Absorbance. J. Phys. Chem. C 2018, 122, 27795–27802. [Google Scholar] [CrossRef]
- Wang, P.; Zong, Y.; Liu, H.; Wen, H.; Wu, H.-B.; Xia, J.-B. Highly efficient photocatalytic water splitting and enhanced piezoelectric properties of 2D Janus group-III chalcogenides. J. Mater. Chem. C 2021, 9, 4989–4999. [Google Scholar] [CrossRef]
- Ahmad, I.; Shahid, I.; Ali, A.; Gao, L.; Cai, J. Electronic, mechanical, optical and photocatalytic properties of two-dimensional Janus XGaInY (X, Y;= S, Se and Te) monolayers. RSC Adv. 2021, 11, 17230–17239. [Google Scholar] [CrossRef]
- Chu, S.; Li, W.; Yan, Y.; Hamann, T.; Shih, I.; Wang, D.; Mi, Z. Roadmap on solar water splitting: Current status and future prospects. Nano Futur. 2017, 1, 022001. [Google Scholar] [CrossRef]









| Photocatalyst Used | Amount of Photocatalyst Used | Electrolyte/ Solvent Used | Morphology | H2 Produced | Ref. |
|---|---|---|---|---|---|
| MoS2/g-C3N4/ZnIn2S4 | 50 mg | 90 mL water and 10 mL triethanolamine | 3D flower-like nanospheres | ZnIn2S4: 904 μmol g−1h−1 MoS2/g-C3N4/ZnIn2S4 loaded with with 3 wt% g-C3N4 and 1.5 wt% MoS2: 6291 μmol g−1h−1 | [90] |
| ZnIn2S4@ CuInS2 | 50 mg | 0.25 mol/L Na2S and 0.35 mol/L Na2SO3 as a sacrificial agent in 100 mL of aqueous solution | Marigold-like spherical structure comprising numerous thin nanosheets | 1168 μmol g−1 | [91] |
| Ti3C2 MXene@ TiO2/CuInS2 | 10 mg | 100 mL water solution containing 20% methanol as sacrificial agent | Layered structure decorated with small CuInS2 nanoparticles | 356.27 μmol g−1h−1 | [92] |
| ZnIn2S4-rGO-CuInS2 | 15 mg | 50 mL of aqueous Na2S/Na2SO3 (10 vol%) solution | Dispersed marigold-like structured ZnIn2S4 and layer-structured CuInS2 on reduced graphene oxide sheets | 2531 μmol/g after 5 h | [93] |
| CdS/CoMoS4 | 80 mg | 8 mL of lactic acid was added into 80 mL of water | Nanorod structured | 7.5 at.% CdS/ CoMoS4: 208 μmol h−1 | [94] |
| Carbon nanotube modified Zn0.83Cd0.17S | 1 mg | 40 mL of an aqueous solution containing 0.1 M Na2S/0.02 M Na2SO3 | Irregular | 0.25 wt% carbon nanotube modified Zn0.83Cd0.17S: 5.41 mmol h−1g−1 | [95] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Khan, M.M.; Rahman, A. Chalcogenides and Chalcogenide-Based Heterostructures as Photocatalysts for Water Splitting. Catalysts 2022, 12, 1338. https://doi.org/10.3390/catal12111338
Khan MM, Rahman A. Chalcogenides and Chalcogenide-Based Heterostructures as Photocatalysts for Water Splitting. Catalysts. 2022; 12(11):1338. https://doi.org/10.3390/catal12111338
Chicago/Turabian StyleKhan, Mohammad Mansoob, and Ashmalina Rahman. 2022. "Chalcogenides and Chalcogenide-Based Heterostructures as Photocatalysts for Water Splitting" Catalysts 12, no. 11: 1338. https://doi.org/10.3390/catal12111338
APA StyleKhan, M. M., & Rahman, A. (2022). Chalcogenides and Chalcogenide-Based Heterostructures as Photocatalysts for Water Splitting. Catalysts, 12(11), 1338. https://doi.org/10.3390/catal12111338









