Modifications in the Composition of CuO/ZnO/Al2O3 Catalyst for the Synthesis of Methanol by CO2 Hydrogenation
Abstract
:1. Introduction
2. Results and Discussion
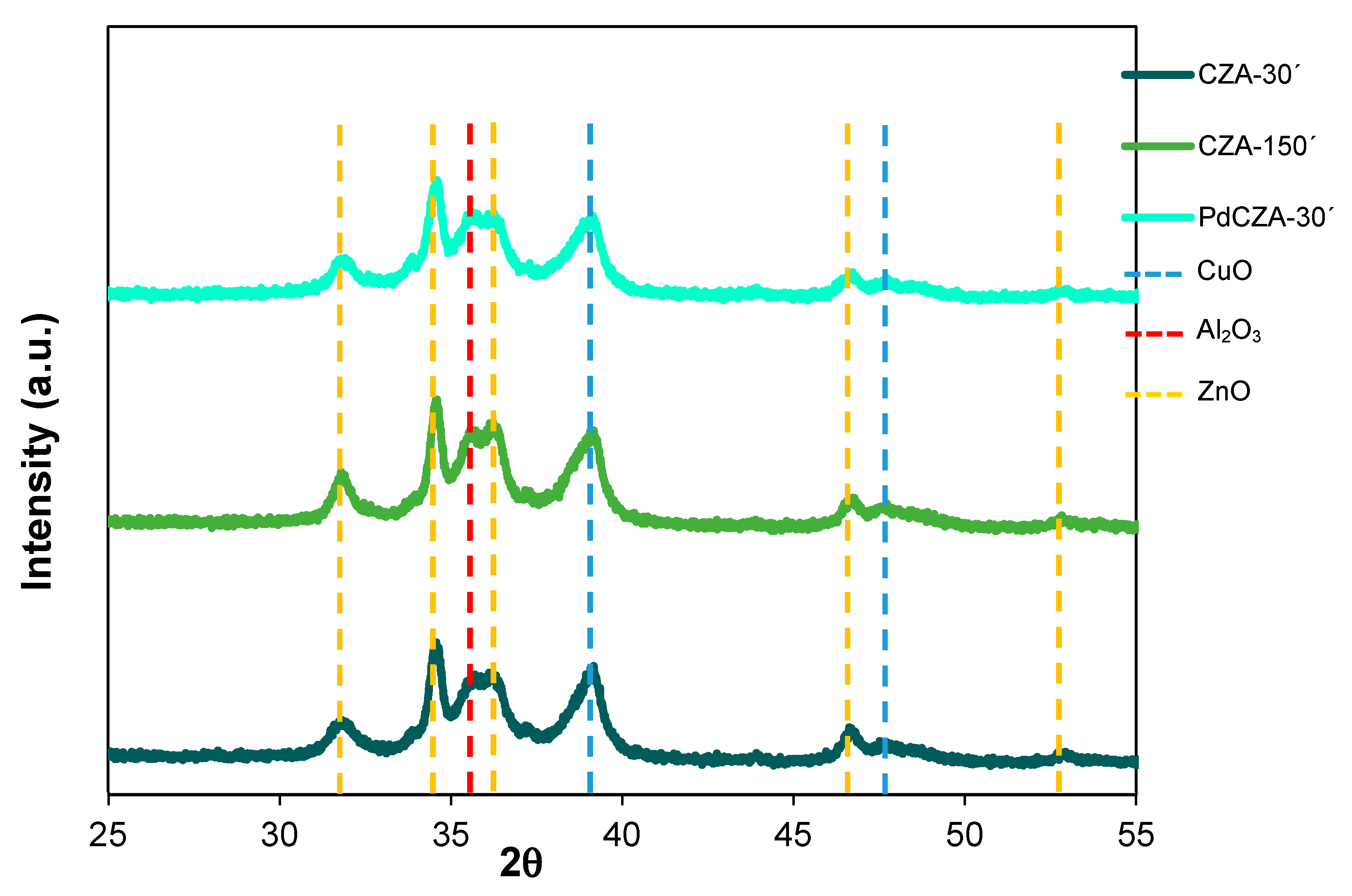
2.1. Catalyst Characterization
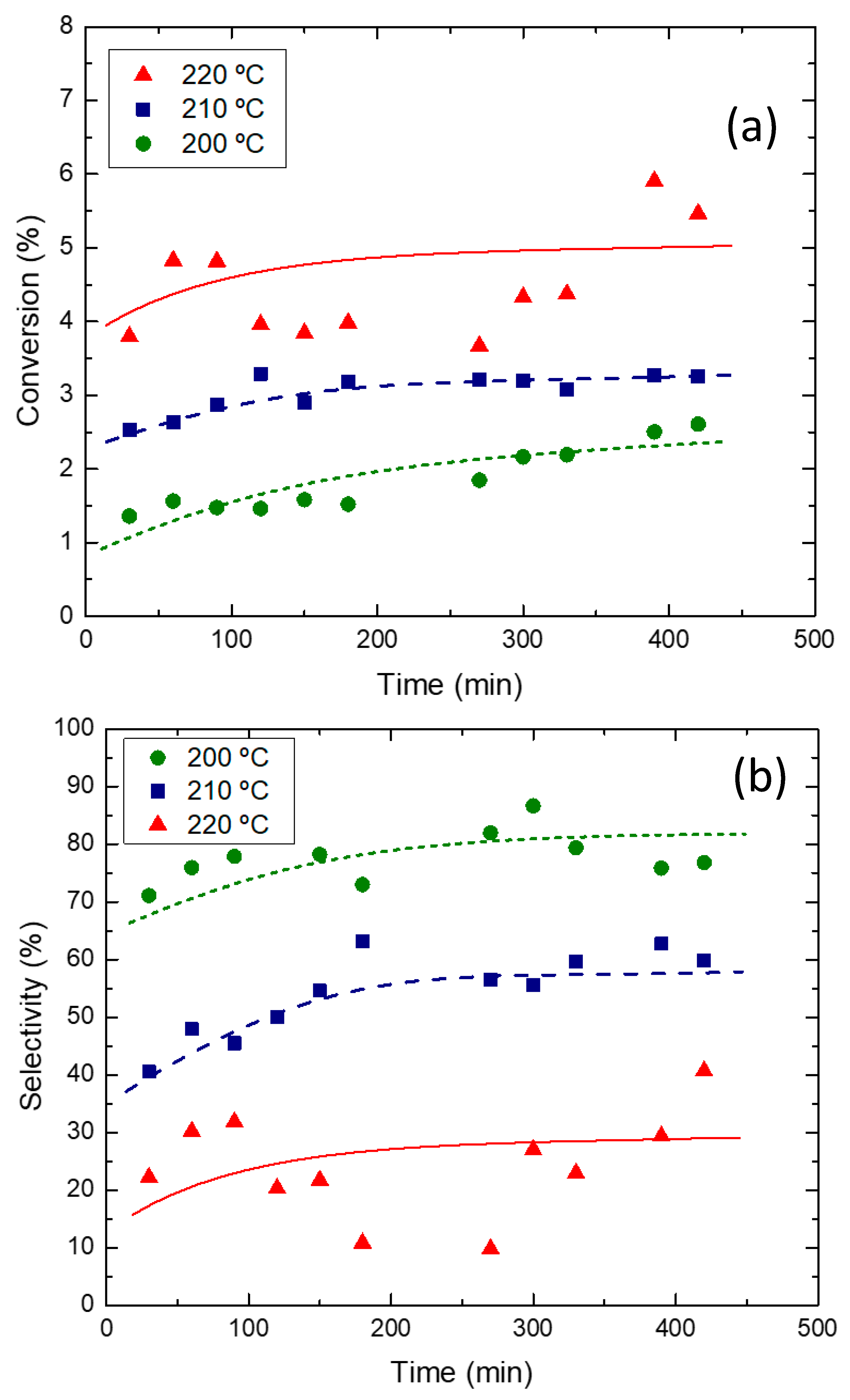
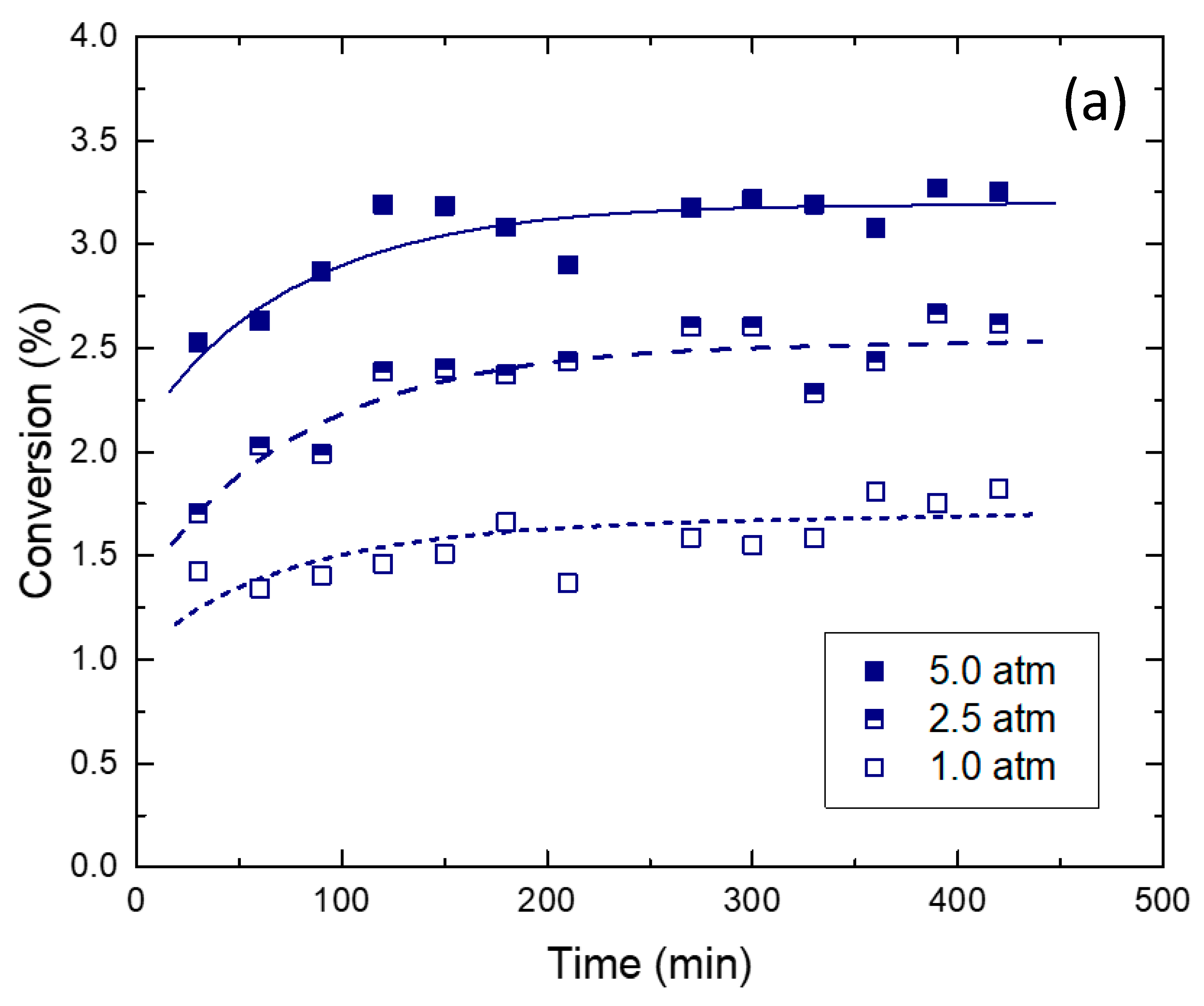
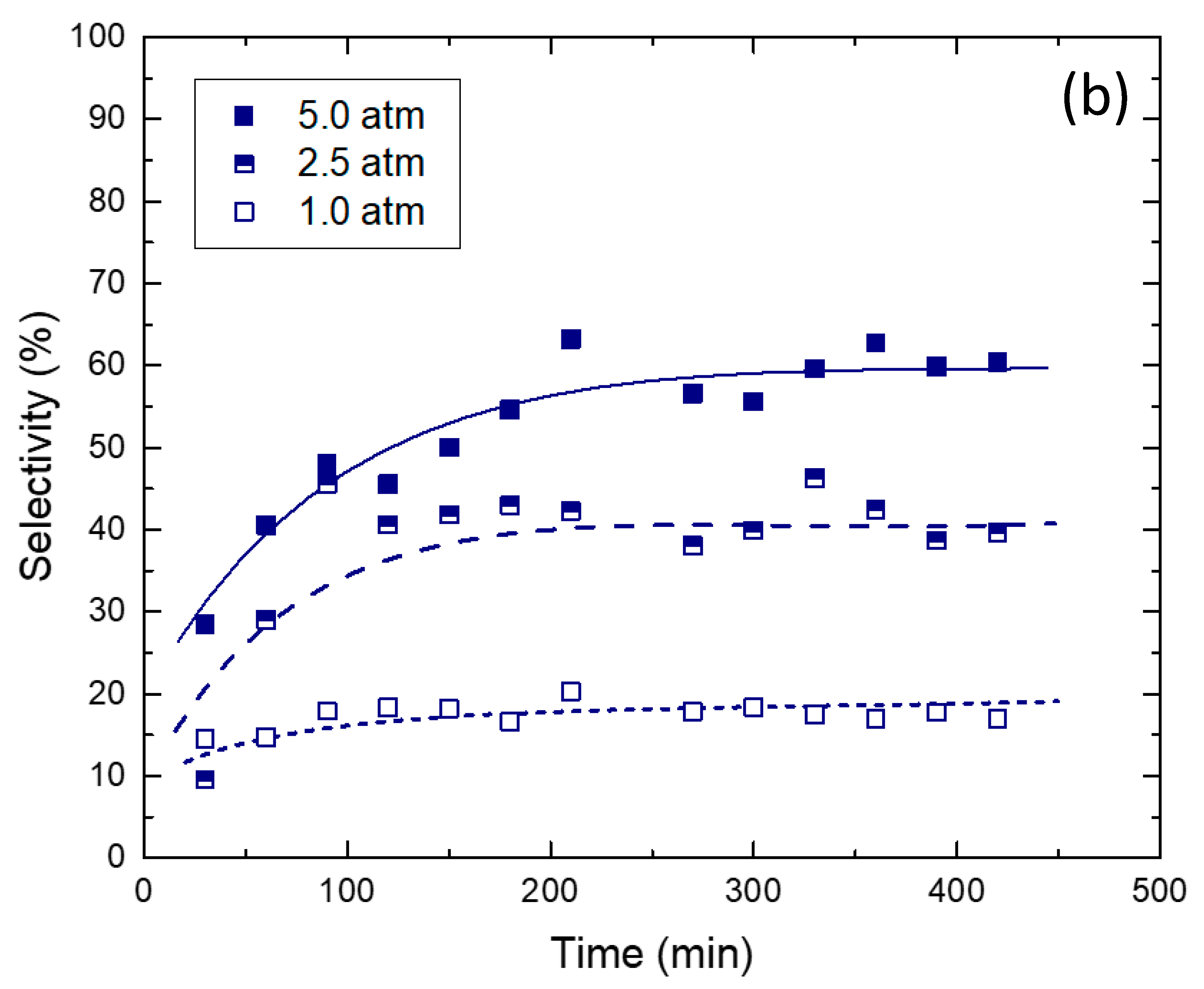
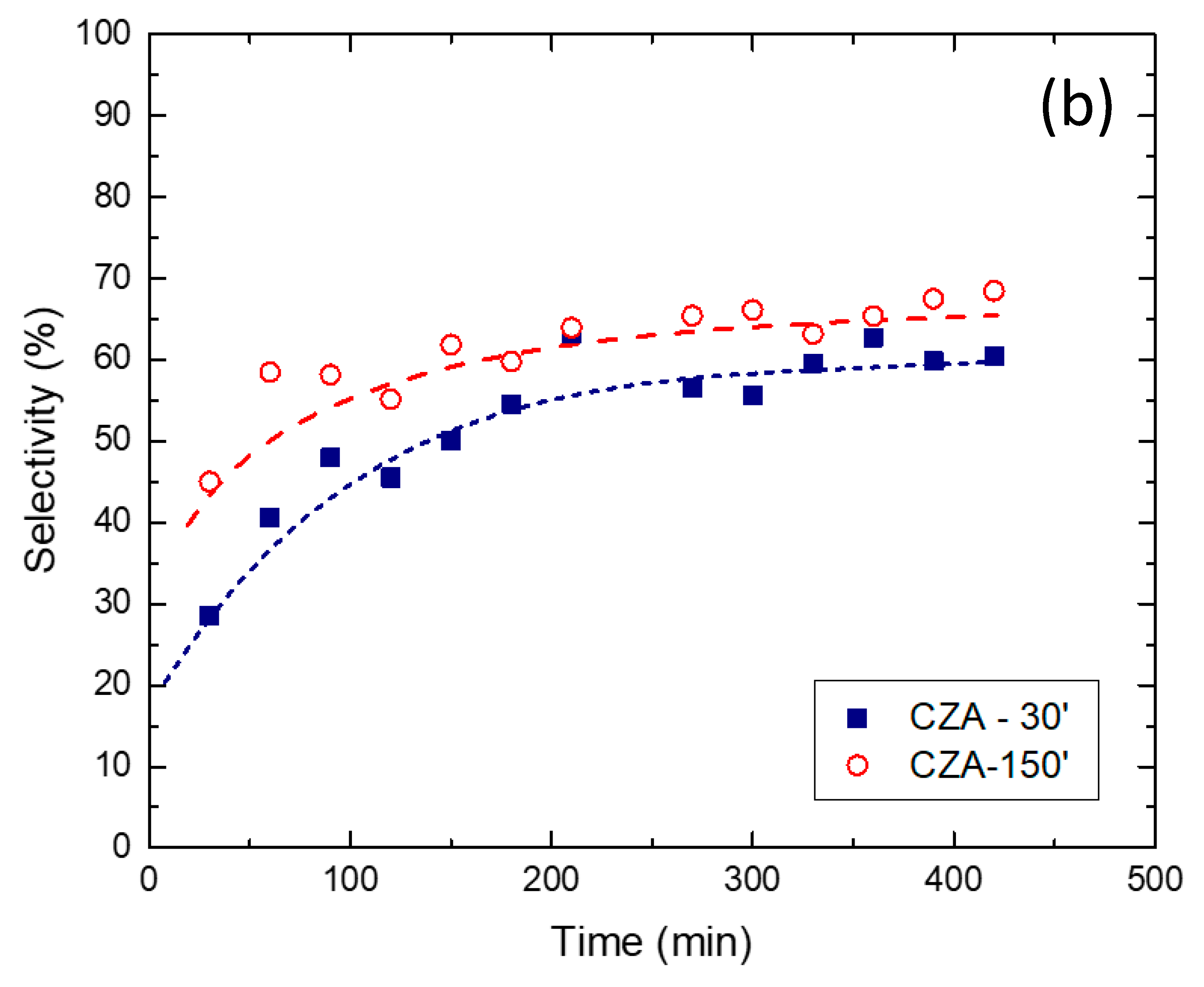
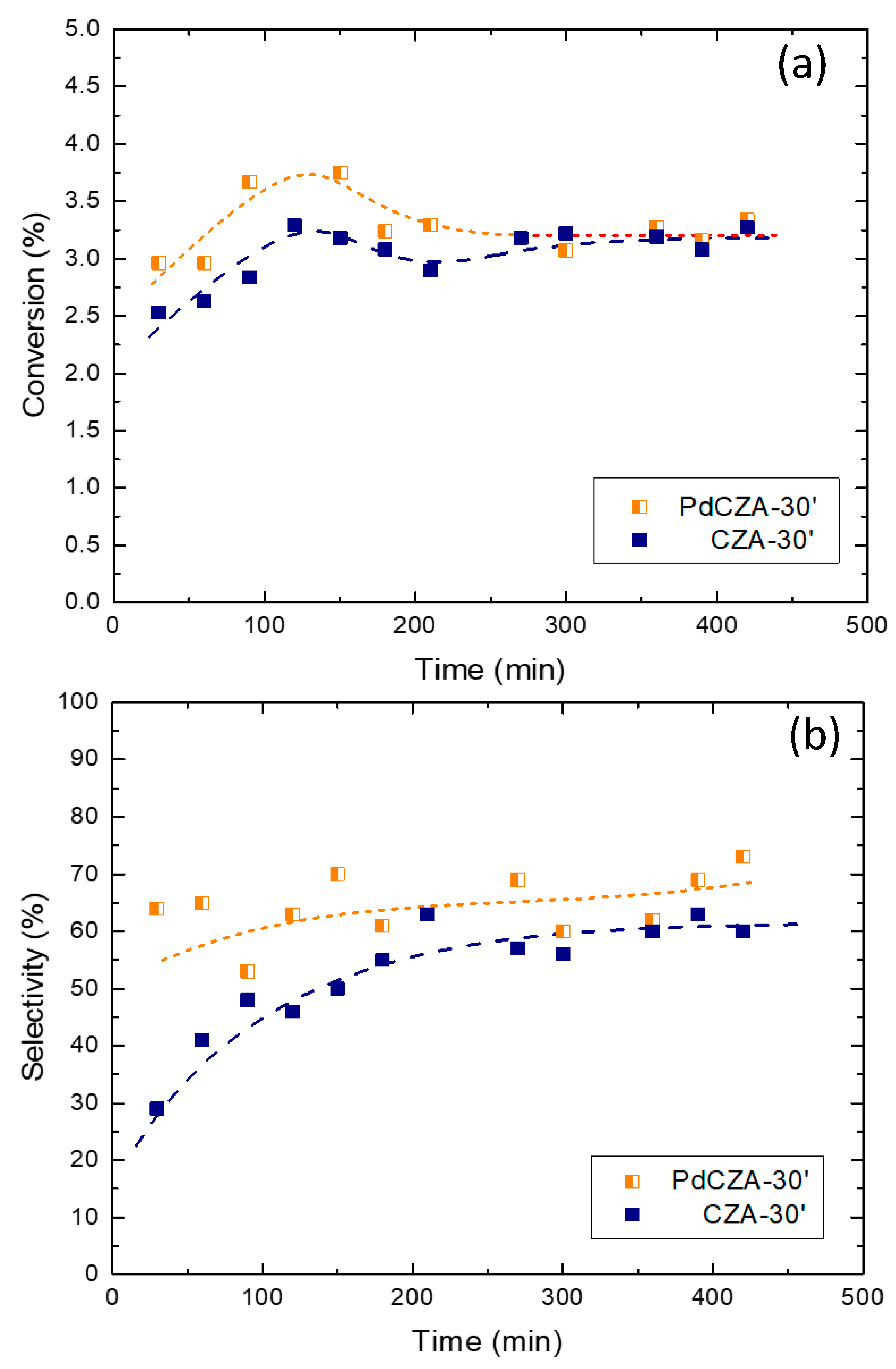
2.2. Reaction Tests
3. Experimental
3.1. Reaction System
3.2. Catalyst Synthesis
3.3. Catalyst Characterisation
3.4. Reaction Tests
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Atsonios, E.K.K.; Panopolous, K.D. Investigation of technical and economic aspects for methanol production through CO2 hydrogenation. Int. J. Hydrogen Energy 2016, 41, 220–232. [Google Scholar] [CrossRef]
- Saedi, S.; Amin, N.S.A.; Rahimpour, M.R. Hydrogenation of CO2 to value-added products—A review and potential future developments. J. CO2 Util. 2014, 5, 66–81. [Google Scholar] [CrossRef]
- Aresta, M.; Dibenedetto, A.; Angelini, A. The changing paradigm in CO2 utilization. J. CO2 Util 2013, 3, 65–73. [Google Scholar] [CrossRef]
- Olah, G.A. Beyond Oil and Gas: The Methanol Economy. Angew. Chem. Int. Ed. 2005, 44, 2636–2639. [Google Scholar] [CrossRef] [PubMed]
- Olah, G.A. Towards oil independence through renewable methanol chemistry. Angew. Chem. Int. Ed. 2013, 52, 104–107. [Google Scholar] [CrossRef] [PubMed]
- Centi, G.; Perathoner, S. Opportunities and prospects in the chemical recycling of carbon dioxide to fuels. Catal. Today 2009, 148, 191–205. [Google Scholar] [CrossRef]
- Andersson, J.; Grönkvist, S. Large-scale storage of hydrogen. Int. J. Hydrogen Energy 2019, 44, 11901–11919. [Google Scholar] [CrossRef]
- Jadhav, S.G.; Vaidya, P.D.; Bhanage, B.M.; Joshi, J.B. Catalytic carbon dioxide hydrogenation to methanol: A review of recent studies. Chem. Eng. Res. Dev. 2014, 92, 2557–2567. [Google Scholar] [CrossRef]
- Tursunov, O.; Kutstov, L.; Tilybaev, Z. Methanol synthesis from the catalytic hydrogenation of CO2 over CuO–ZnO supported on aluminum and silicon oxides. J. Taiwan Inst. Chem. 2017, 78, 416–422. [Google Scholar] [CrossRef]
- Baltes, C.; Vukojevic, S.; Schuth, F. Correlations between synthesis, precursor, and catalyst structure and activity of a large set of CuO/ZnO/Al2O3 catalysts for methanol synthesis. J. Catal. 2008, 258, 334–344. [Google Scholar] [CrossRef]
- Ahoba-Sam, C.; Olsbye, U.; Jens, K.J. Low temperature methanol synthesis catalyzed by copper nanoparticles. Catal. Today 2018, 299, 112–119. [Google Scholar] [CrossRef] [Green Version]
- Riaz, A.; Zahedi, G.; Klemeš, J.J. A review of cleaner production methods for the manufacture of methanol. J. Clean. Prod. 2013, 57, 19–37. [Google Scholar] [CrossRef]
- Chinchen, G.C.; Spencer, M.S.; Waugh, K.C.; Whan, D.A. Promotion of methanol synthesis and the water-gas shift reactions by adsorbed oxygen on supported copper catalyst. J. Chem. Soc. Faraday Trans. 1 Phys. Chem. Condens. Phases 1987, 83, 2193–2212. [Google Scholar] [CrossRef]
- Ayodele, O.B. Eliminating reverse water gas shift reaction in CO2 hydrogenation to primary oxygenates over MFI-type zeolite supported Cu/ZnO nanocatalysts. J. CO2 Util. 2017, 20, 368–377. [Google Scholar] [CrossRef]
- Struis, R.; Stucki, S.; Wiedorn, M.A. membrane reactor for methanol synthesis. J. Membr. Sci. 1996, 113, 93–100. [Google Scholar] [CrossRef]
- Menéndez, M.; Piera, E.; Coronas, J.; Santamaría, J. Reactor de Membrana Zeolítica Para la Obtención de Metanol y Otros Alcoholes a Partir de Gas de Síntesis. Spanish Patent ES2164544, July 1999. [Google Scholar]
- Gallucci, F.; Paturzo, L.; Basile, A. An experimental study of CO2 hydrogenation into methanol involving a zeolite membrane reactor. Chem. Eng. Proc. Process Intens. 2004, 43, 1029–1036. [Google Scholar] [CrossRef]
- Raso, R.; Tovar, M.; Lasobras, J.; Herguido, J.; Kumakiri, I.; Araki, S. Comparison of zeolite membranes towards their use in membrane reactors for CO2 hydrogenation to methanol. Catal. Today 2020, 364, 270–275. [Google Scholar] [CrossRef]
- Hong, Z.S.; Cao, Y.; Deng, J.F.; Fan, K.N. CO2 hydrogenation to methanol over Cu/ZnO/Al2O3 catalysts prepared by a novel gel-network-coprecipitation method. Catal. Lett. 2002, 82, 37–44. [Google Scholar] [CrossRef]
- Melián-Cabrera, I.; López Granados, M.; Fierro, J.L.G. Reverse Topotactic Transformation of a Cu–Zn–Al Catalyst during Wet Pd Impregnation: Relevance for the Performance in Methanol Synthesis from CO2/H2 Mixtures. J. Catal. 2002, 210, 273–284. [Google Scholar] [CrossRef]
- Regabulto, J. Catalyst Preparation: Science and Engineering; CRC Press: Boca Raton, FL, USA, 2007. [Google Scholar]

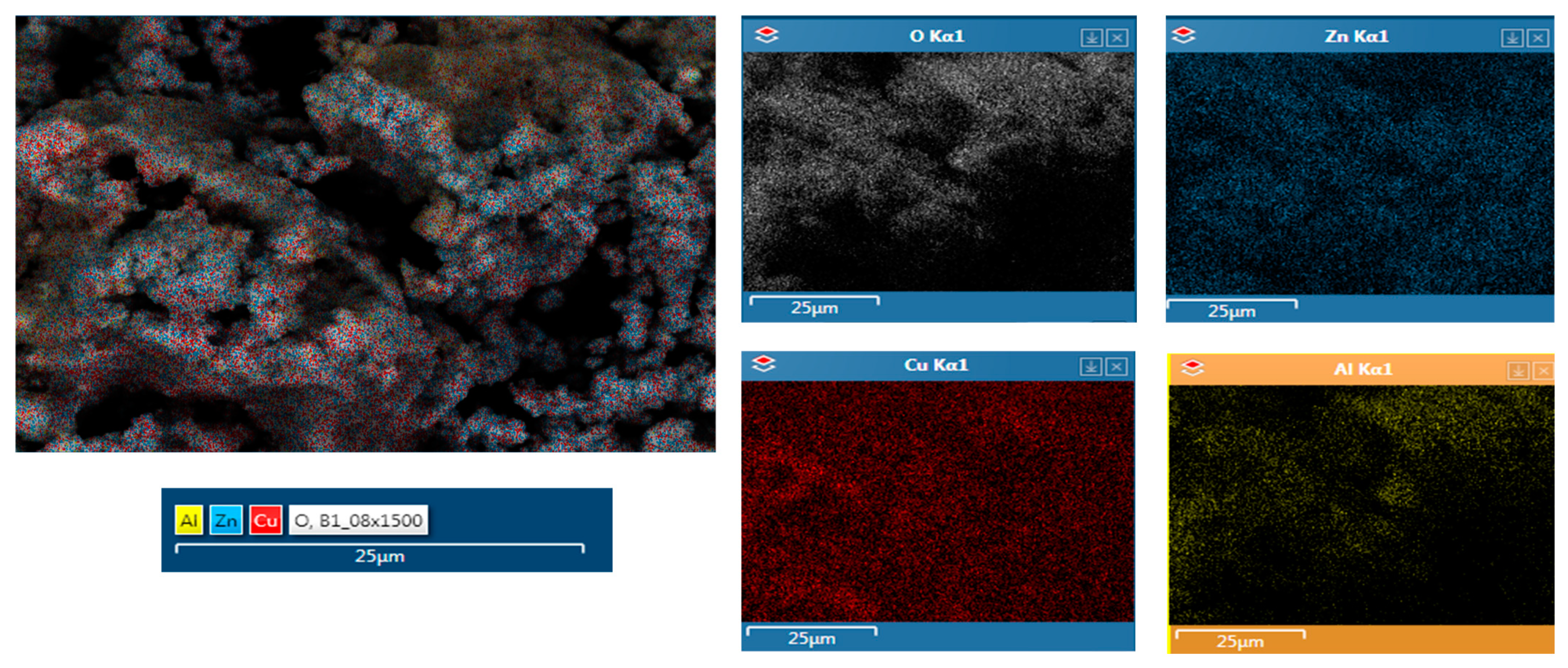



| Sample | O | Pd | Al | Cu | Zn |
|---|---|---|---|---|---|
| CZA-30′ | 34.4 | - | 6.3 | 34.7 | 24.6 |
| PdCZA-30′ | 21.3 | 3.9 | 2.9 | 30.8 | 37.4 |
| CZA-150′ | 21.9 | - | 4.2 | 30.5 | 38.7 |
| Sample | Specific Surface Area (m2/g) | Pore Volume (cm3/g) | Pore Diameter (nm) | |||
|---|---|---|---|---|---|---|
| SBET | Smicropore | Vpore | Vmicropore | dpore | dmicropore | |
| CZA-30′ | 111.9 | 12.3 | 0.5 | 0.005 | 18.2 | 1.6 |
| PdCZA-30′ | 123.7 | - | 0.3 | - | 10.7 | - |
| CZA-150′ | 65.9 | 4.7 | 0.2 | 0.002 | 14.6 | 1.7 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Trifan, B.; Lasobras, J.; Soler, J.; Herguido, J.; Menéndez, M. Modifications in the Composition of CuO/ZnO/Al2O3 Catalyst for the Synthesis of Methanol by CO2 Hydrogenation. Catalysts 2021, 11, 774. https://doi.org/10.3390/catal11070774
Trifan B, Lasobras J, Soler J, Herguido J, Menéndez M. Modifications in the Composition of CuO/ZnO/Al2O3 Catalyst for the Synthesis of Methanol by CO2 Hydrogenation. Catalysts. 2021; 11(7):774. https://doi.org/10.3390/catal11070774
Chicago/Turabian StyleTrifan, Bianca, Javier Lasobras, Jaime Soler, Javier Herguido, and Miguel Menéndez. 2021. "Modifications in the Composition of CuO/ZnO/Al2O3 Catalyst for the Synthesis of Methanol by CO2 Hydrogenation" Catalysts 11, no. 7: 774. https://doi.org/10.3390/catal11070774
APA StyleTrifan, B., Lasobras, J., Soler, J., Herguido, J., & Menéndez, M. (2021). Modifications in the Composition of CuO/ZnO/Al2O3 Catalyst for the Synthesis of Methanol by CO2 Hydrogenation. Catalysts, 11(7), 774. https://doi.org/10.3390/catal11070774