Pore Blocking by Phenolates as Deactivation Path during the Cracking of 4-Propylphenol over ZSM-5
Abstract
1. Introduction
2. Results
2.1. Catalyst Characterization
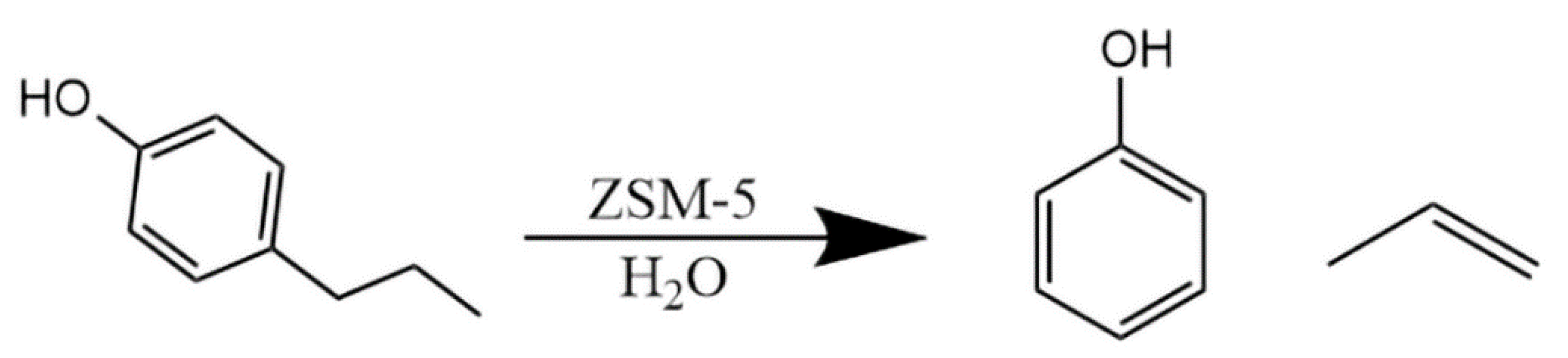
2.2. Catalytic Cracking of 4-Propylphenol
2.3. Characterization of the Deactivated ZSM-5
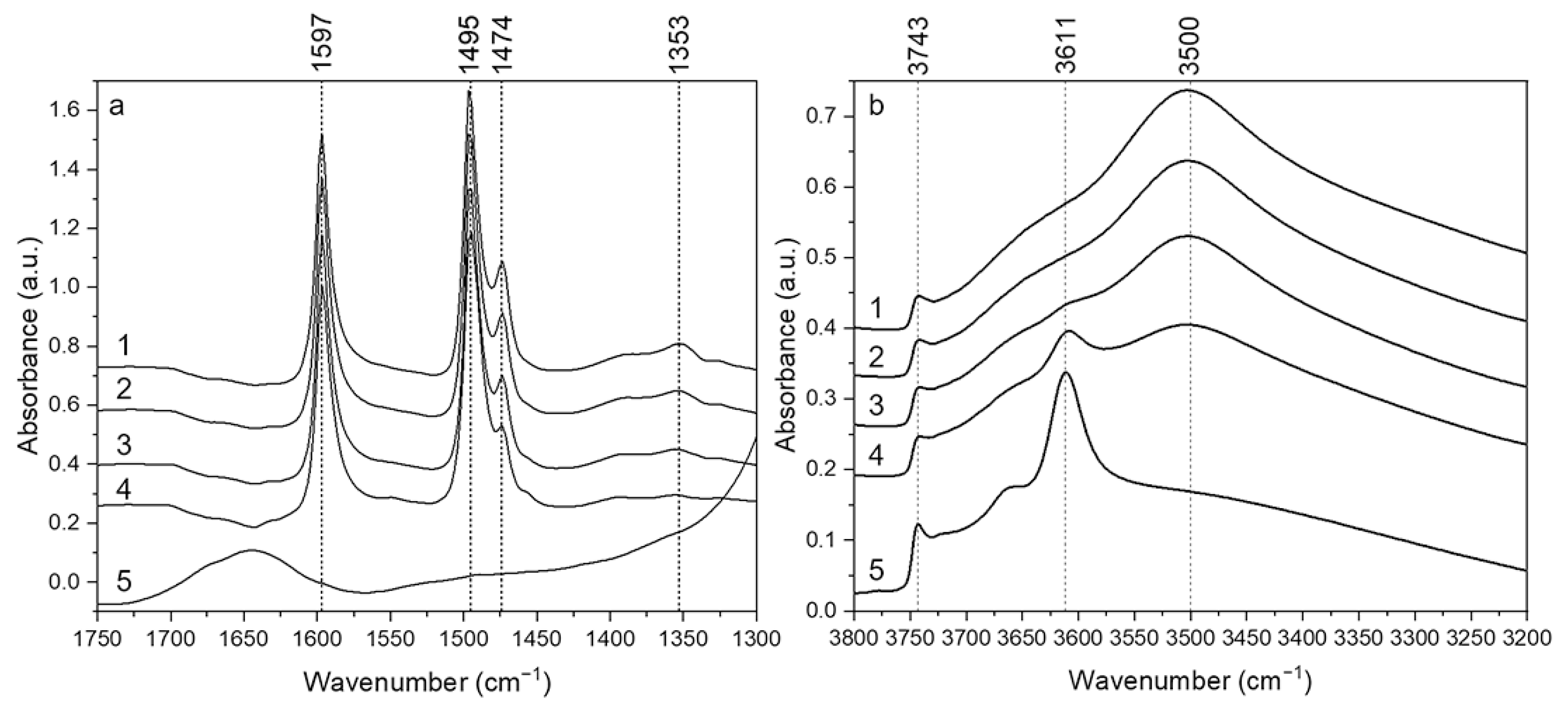
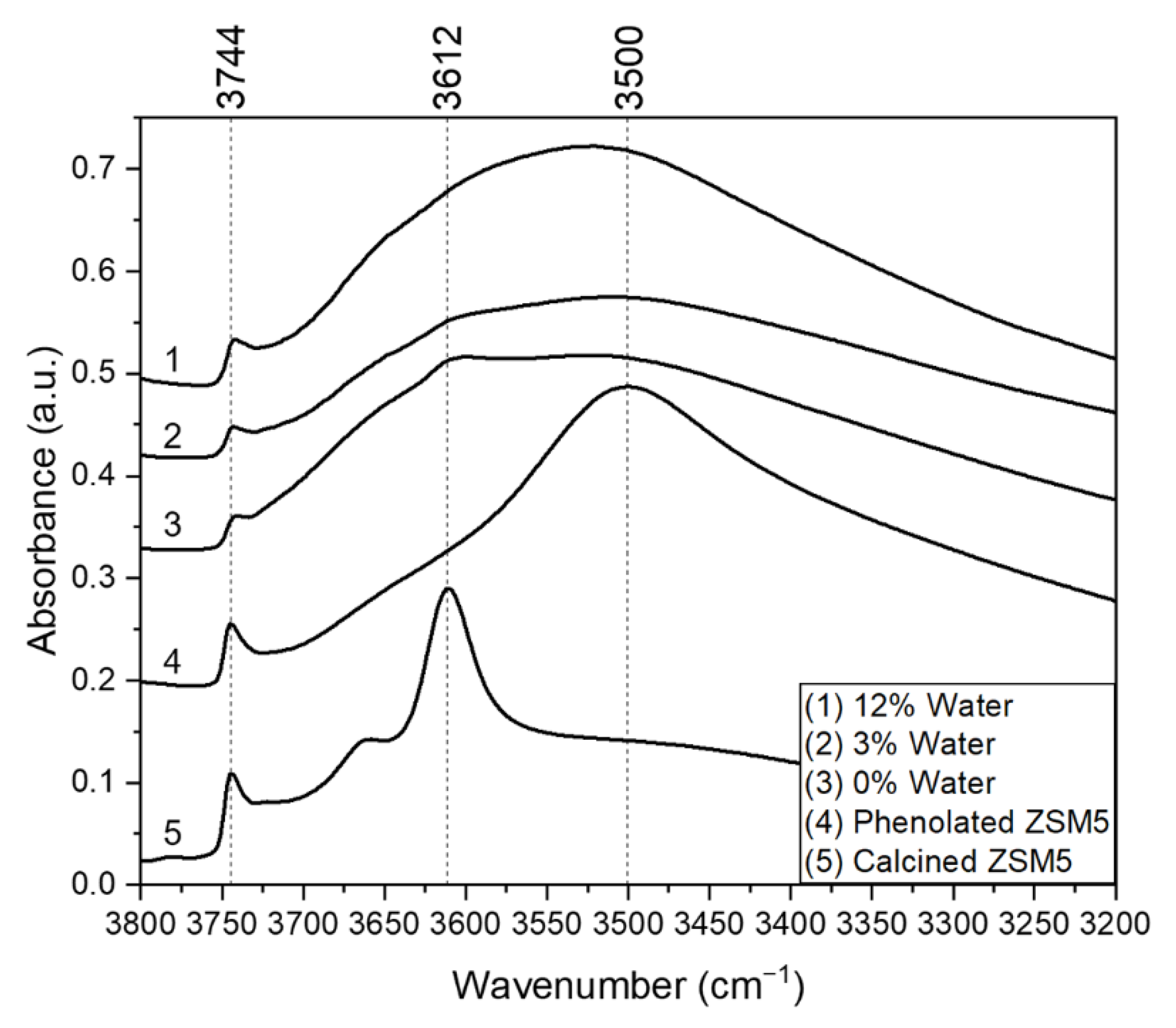
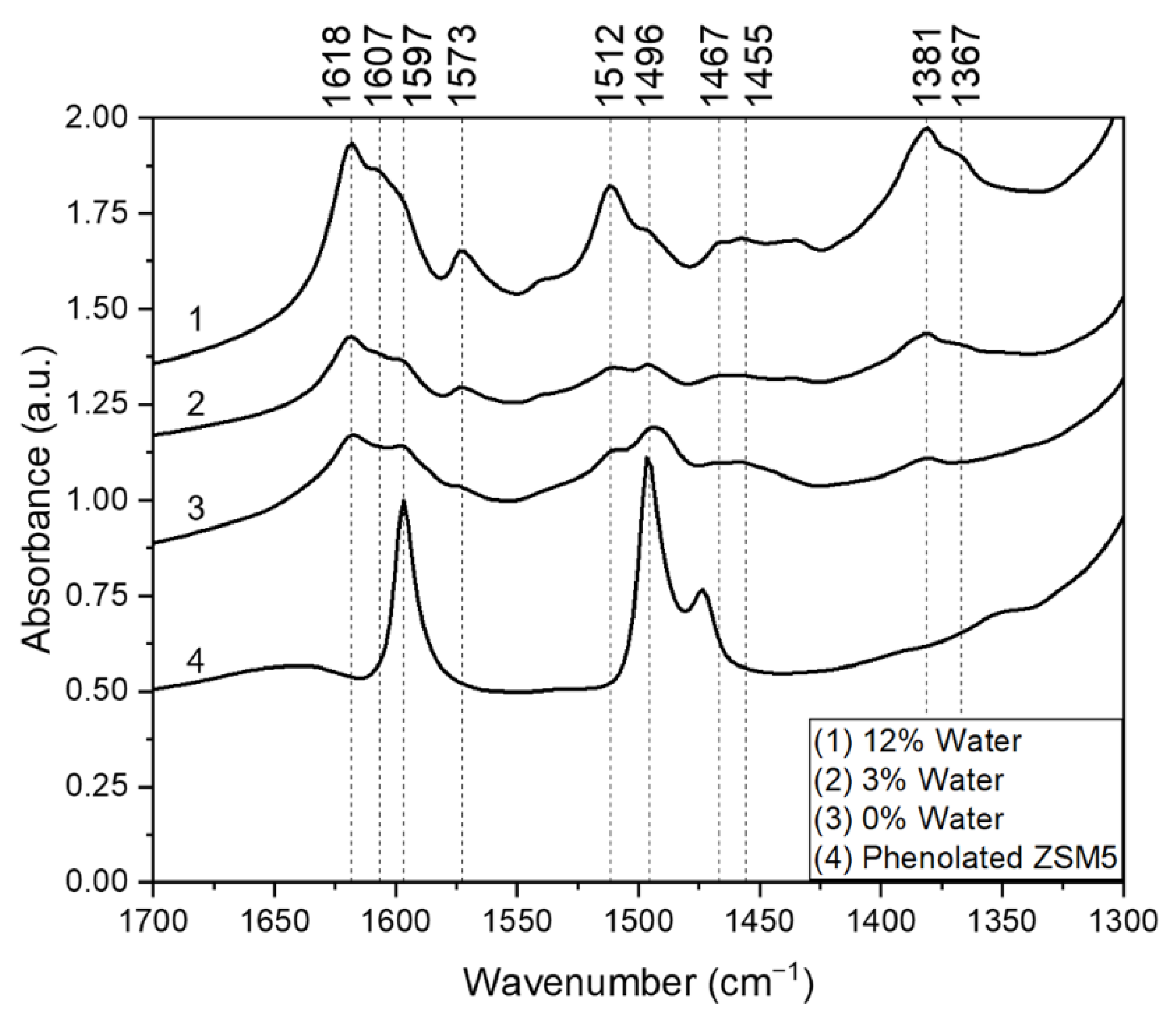
2.4. FTIR Spectroscopic Analysis of Adsorbed Phenol and Spent Catalysts
3. Discussion
3.1. Routes to Deactivation
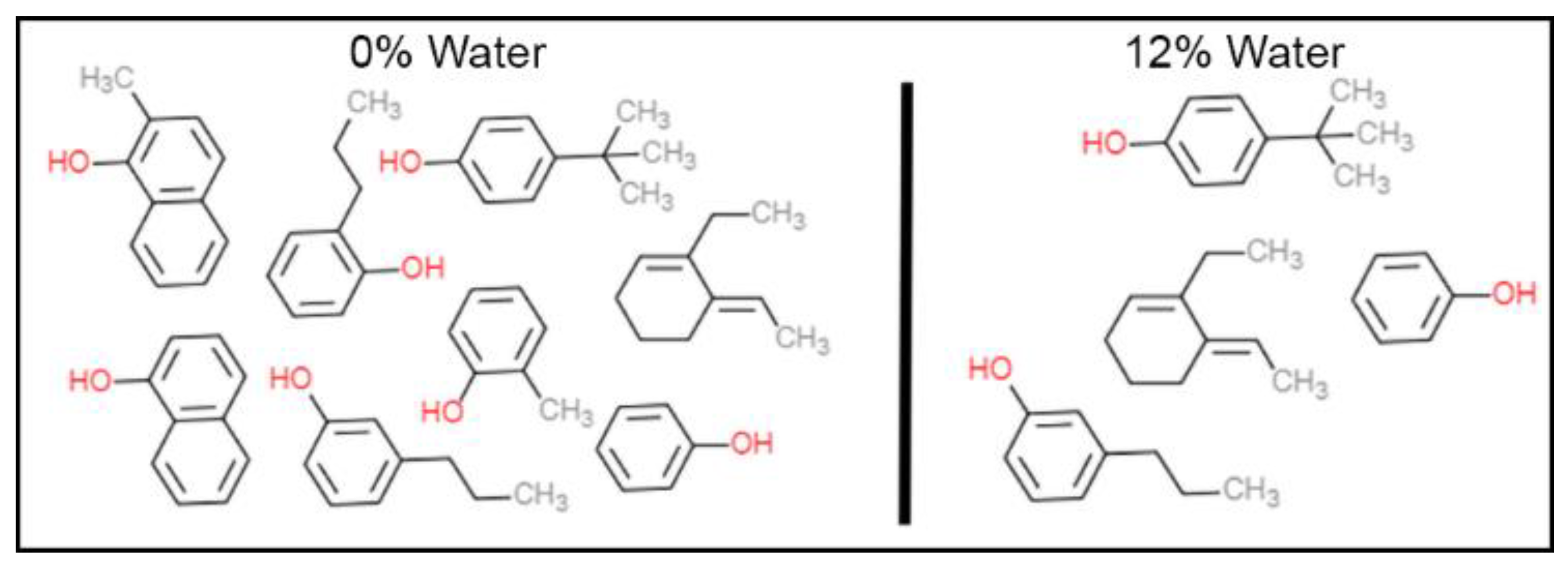
3.2. Nature of Species Causing Pore Blockage
3.3. Effect of Propylene Polymerization
3.4. Implications for Catalyst Design of Biorefining Processes
4. Materials and Methods
4.1. Materials
4.2. Catalyst Characterization
4.3. Catalytic Cracking Reactions
4.4. FTIR Spectroscopy of Adsorbed Phenol and Carbonaceous Deposits
4.5. Thermogravimetric Analysis
4.6. Raman Spectroscopy
4.7. Hydrofluoric Acid Digestion
4.8. Solid State 27Al NMR Spectroscopy
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Katelhon, A.; Meys, R.; Deutz, S.; Suh, S.; Bardow, A. Climate change mitigation potential of carbon capture and utilization in the chemical industry. Proc. Natl. Acad. Sci. USA 2019, 116, 11187–11194. [Google Scholar] [CrossRef] [PubMed]
- Alonso, D.M.; Hakim, S.H.; Zhou, S.; Won, W.; Hosseinaei, O.; Tao, J.; Garcia-Negron, V.; Motagamwala, A.H.; Mellmer, M.A.; Huang, K.; et al. Increasing the revenue from lignocellulosic biomass: Maximizing feedstock utilization. Sci. Adv. 2017, 3, e1603301. [Google Scholar] [CrossRef]
- Brodin, M.; Vallejos, M.; Opedal, M.T.; Area, M.C.; Chinga-Carrasco, G. Lignocellulosics as sustainable resources for production of bioplastics—A review. J. Clean. Prod. 2017, 162, 646–664. [Google Scholar] [CrossRef]
- Zakzeski, J.; Bruijnincx, P.C.; Jongerius, A.L.; Weckhuysen, B.M. The catalytic valorization of lignin for the production of renewable chemicals. Chem. Rev. 2010, 110, 3552–3599. [Google Scholar] [CrossRef]
- Ragauskas, A.J.; Beckham, G.T.; Biddy, M.J.; Chandra, R.; Chen, F.; Davis, M.F.; Davison, B.H.; Dixon, R.A.; Gilna, P.; Keller, M.; et al. Lignin valorization: Improving lignin processing in the biorefinery. Science 2014, 344, 1246843. [Google Scholar] [CrossRef]
- Renders, T.; Van den Bosch, S.; Koelewijn, S.F.; Schutyser, W.; Sels, B.F. Lignin-first biomass fractionation: The advent of active stabilisation strategies. Energy Environ. Sci. 2017, 10, 1551–1557. [Google Scholar] [CrossRef]
- Arato, C.; Pye, E.K.; Gjennestad, G. The lignol approach to biorefining of woody biomass to produce ethanol and chemicals. Appl. Biochem. Biotechnol. 2005, 123, 871–882. [Google Scholar] [CrossRef]
- Gillet, S.; Aguedo, M.; Petitjean, L.; Morais, A.R.C.; Lopes, A.M.D.; Lukasik, R.M.; Anastas, P.T. Lignin transformations for high value applications: Towards targeted modifications using green chemistry. Green Chem. 2017, 19, 4200–4233. [Google Scholar] [CrossRef]
- Scown, C.D.; Gokhale, A.A.; Willems, P.A.; Horvath, A.; McKone, T.E. Role of lignin in reducing life-cycle carbon emissions, water use, and cost for United States cellulosic biofuels. Environ. Sci. Technol. 2014, 48, 8446–8455. [Google Scholar] [CrossRef]
- Tricker, A.W.; Stellato, M.J.; Kwok, T.T.; Kruyer, N.S.; Wang, Z.; Nair, S.; Thomas, V.M.; Realff, M.J.; Bommarius, A.S.; Sievers, C. Similarities in recalcitrant structures of industrial non-kraft and kraft lignin. ChemSusChem 2020, 13, 4624–4632. [Google Scholar] [CrossRef]
- Kwok, T.T.; Luettgen, C.O.; Realff, M.J.; Bommarius, A.S. Lignin value prior to pulping (LVPP): An advanced pulping concept. Tappi J. 2017, 16, 553–555. [Google Scholar]
- Anderson, E.M.; Stone, M.L.; Katahira, R.; Reed, M.; Beckham, G.T.; Roman-Leshkov, Y. Flowthrough reductive catalytic fractionation of biomass. Joule 2017, 1, 613–622. [Google Scholar] [CrossRef]
- Van den Bosch, S.; Schutyser, W.; Vanholme, R.; Driessen, T.; Koelewijn, S.F.; Renders, T.; De Meester, B.; Huijgen, W.J.J.; Dehaen, W.; Courtin, C.M.; et al. Reductive lignocellulose fractionation into soluble lignin-derived phenolic monomers and dimers and processable carbohydrate pulps. Energy Environ. Sci. 2015, 8, 1748–1763. [Google Scholar] [CrossRef]
- Foo, G.S.; Rogers, A.K.; Yung, M.M.; Sievers, C. Steric effect and evolution of surface species in the hydrodeoxygenation of bio-oil model compounds over Pt/HBEA. ACS Catal. 2016, 6, 1292–1307. [Google Scholar] [CrossRef]
- Luo, Z.; Zheng, Z.; Li, L.; Cui, Y.-T.; Zhao, C. Bimetallic Ru–Ni catalyzed aqueous-phase guaiacol hydrogenolysis at low H2 pressures. ACS Catal. 2017, 7, 8304–8313. [Google Scholar] [CrossRef]
- Rezaei, P.S.; Shafaghat, H.; Daud, W.M.A.W. Production of green aromatics and olefins by catalytic cracking of oxygenate compounds derived from biomass pyrolysis: A review. Appl. Catal. A Gen. 2014, 469, 490–511. [Google Scholar] [CrossRef]
- Verboekend, D.; Liao, Y.H.; Schutyser, W.; Sels, B.F. Alkylphenols to phenol and olefins by zeolite catalysis: A pathway to valorize raw and fossilized lignocellulose. Green Chem. 2016, 18, 297–306. [Google Scholar] [CrossRef]
- Liao, Y.H.; d’Halluin, M.; Makshina, E.; Verboekend, D.; Sels, B.F. Shape selectivity vapor-phase conversion of lignin-derived 4-ethylphenol to phenol and ethylene over acidic aluminosilicates: Impact of acid properties and pore constraint. Appl. Catal. B Environ. 2018, 234, 117–129. [Google Scholar] [CrossRef]
- Liao, Y.H.; Zhong, R.Y.; d’Halluin, M.; Verboekend, D.; Sels, B.F. Aromatics production from lignocellulosic biomass: Shape selective dealkylation of lignin-derived phenolics over hierarchical ZSM-5. ACS Sustain. Chem. Eng. 2020, 8, 8713–8722. [Google Scholar] [CrossRef]
- De Lucas, A.; Canizares, P.; Durán, A.; Carrero, A. Dealumination of HZSM-5 zeolites: Effect of steaming on acidity and aromatization activity. Appl. Catal. A Gen. 1997, 154, 221–240. [Google Scholar] [CrossRef]
- Triantafillidis, C.S.; Vlessidis, A.G.; Nalbandian, L.; Evmiridis, N.P. Effect of the degree and type of the dealumination method on the structural, compositional and acidic characteristics of H-ZSM-5 zeolites. Microporous Mesoporous Mater. 2001, 47, 369–388. [Google Scholar] [CrossRef]
- Anderson, E.; Crisci, A.; Murugappan, K.; Roman-Leshkov, Y. Bifunctional Molybdenum polyoxometalates for the combined hydrodeoxygenation and alkylation of lignin-derived model phenolics. ChemSusChem 2017, 10, 2226–2234. [Google Scholar] [CrossRef]
- Ardizzi, M.; Ballarini, N.; Cavani, F.; Chiappini, E.; Dal Pozzo, L.; Maselli, L.; Monti, T. Environmentally friendly, heterogeneous acid and base catalysis for the methylation of catechol: Chances for the control of chemo-selectivity. Appl. Catal. B Environ. 2007, 70, 597–605. [Google Scholar] [CrossRef]
- Yung, M.M.; Foo, G.S.; Sievers, C. Role of Pt during hydrodeoxygenation of biomass pyrolysis vapors over Pt/HBEA. Catal. Today 2018, 302, 151–160. [Google Scholar] [CrossRef]
- Chen, T.-S.; Yang, W.-Y.; Du, Z.-Y.; Feng, J.; Li, W.-Y. Effects of mesopore introduction on the stability of zeolites for 4-iso-Propylphenol dealkylation. Catal. Today 2021, 371, 40–49. [Google Scholar] [CrossRef]
- Einaga, H.; Futamura, S.; Ibusuki, T. Heterogeneous photocatalytic oxidation of benzene, toluene, cyclohexene and cyclohexane in humidified air: Comparison of decomposition behavior on photoirradiated TiO2 catalyst. Appl. Catal. B Environ. 2002, 38, 215–225. [Google Scholar] [CrossRef]
- Pujari, S.P.; Scheres, L.; Marcelis, A.T.; Zuilhof, H. Covalent surface modification of oxide surfaces. Angew. Chem. Int. Ed. Engl. 2014, 53, 6322–6356. [Google Scholar] [CrossRef] [PubMed]
- Sedo, J.; Saiz-Poseu, J.; Busque, F.; Ruiz-Molina, D. Catechol-based biomimetic functional materials. Adv. Mater. 2013, 25, 653–701. [Google Scholar] [CrossRef]
- Ye, Q.; Zhou, F.; Liu, W. Bioinspired catecholic chemistry for surface modification. Chem. Soc. Rev. 2011, 40, 4244–4258. [Google Scholar] [CrossRef]
- Yuen, A.K.; Hutton, G.A.; Masters, A.F.; Maschmeyer, T. The interplay of catechol ligands with nanoparticulate iron oxides. Dalton Trans. 2012, 41, 2545–2559. [Google Scholar] [CrossRef]
- Yang, Y.; Yan, W.; Jing, C. Dynamic adsorption of catechol at the goethite/aqueous solution interface: A molecular-scale study. Langmuir 2012, 28, 14588–14597. [Google Scholar] [CrossRef]
- Popov, A.; Kondratieva, E.; Goupil, J.M.; Mariey, L.; Bazin, P.; Gilson, J.P.; Travert, A.; Mauge, F. Bio-oils hydrodeoxygenation: Adsorption of phenolic molecules on oxidic catalyst supports. J. Phys. Chem. C 2010, 114, 15661–15670. [Google Scholar] [CrossRef]
- Jiang, F.; Zeng, L.; Li, S.; Liu, G.; Wang, S.; Gong, J. Propane dehydrogenation over Pt/TiO2–Al2O3 catalysts. ACS Catal. 2014, 5, 438–447. [Google Scholar] [CrossRef]
- Tamura, M.; Shimizu, K.; Satsuma, A. Comprehensive IR study on acid/base properties of metal oxides. Appl. Catal. A Gen. 2012, 433, 135–145. [Google Scholar] [CrossRef]
- Liao, Y.H.; Zhong, R.Y.; Makshina, E.; d’Halluin, M.; van Limbergen, Y.; Verboekend, D.; Sels, B.F. Propylphenol to phenol and propylene over acidic zeolites: Role of shape selectivity and presence of steam. ACS Catal. 2018, 8, 7861–7878. [Google Scholar] [CrossRef]
- Cheng, Y.T.; Huber, G.W. Chemistry of furan conversion into aromatics and olefins over HZSM-5: A Model biomass conversion reaction. ACS Catal. 2011, 1, 611–628. [Google Scholar] [CrossRef]
- Zhang, H.; Shao, S.; Xiao, R.; Shen, D.; Zeng, J. Characterization of coke deposition in the catalytic fast pyrolysis of biomass derivates. Energy Fuels 2013, 28, 52–57. [Google Scholar] [CrossRef]
- Almas, Q.; Naeem, M.A.; Baldanza, M.A.S.; Solomon, J.; Kenvin, J.C.; Muller, C.R.; da Silva, V.T.; Jones, C.W.; Sievers, C. Transformations of FCC catalysts and carbonaceous deposits during repeated reaction-regeneration cycles. Catal. Sci. Technol. 2019, 9, 6977–6992. [Google Scholar] [CrossRef]
- Jiao, J.; Kanellopoulos, J.; Behera, B.; Jiang, Y.; Huang, J.; Reddy Marthala, V.R.; Ray, S.S.; Wang, W.; Hunger, M. Effects of adsorbate molecules on the quadrupolar interaction of framework aluminum atoms in dehydrated zeolite H,Na-Y. J. Phys. Chem. B 2006, 110, 13812–13818. [Google Scholar] [CrossRef]
- Wang, W.; Buchholz, A.; Arnold, A.; Xu, M.; Hunger, M. Effect of surface methoxy groups on the 27Al quadrupole parameters of framework aluminum atoms in calcined zeolite H–Y. Chem. Phys. Lett. 2003, 370, 88–93. [Google Scholar] [CrossRef]
- Chen, T.-H.; Houthoofd, K.; Grobet, P.J. Toward the aluminum coordination in dealuminated mordenite and amorphous silica–alumina: A high resolution 27Al MAS and MQ MAS NMR study. Microporous Mesoporous Mater. 2005, 86, 31–37. [Google Scholar] [CrossRef]
- Isobe, T.; Watanabe, T.; d’Espinose de la Caillerie, J.B.; Legrand, A.P.; Massiot, D. Solid-state 1H and 27Al NMR studies of amorphous aluminum hydroxides. J. Colloid Interface Sci. 2003, 261, 320–324. [Google Scholar] [CrossRef]
- Silaghi, M.C.; Chizallet, C.; Raybaud, P. Challenges on molecular aspects of dealumination and desilication of zeolites. Microporous Mesoporous Mater. 2014, 191, 82–96. [Google Scholar] [CrossRef]
- Colthup, N.B.; Daly, L.H.; Wiberley, S.E. Aromatic and Heteroaromatic Rings. In Introduction to Infrared and Raman Spectroscopy; Colthup, N.B., Daly, L.H., Wiberley, S.E., Eds.; Academic Press: San Diego, CA, USA, 1990; pp. 261–288. [Google Scholar]
- Popov, A.; Kondratieva, E.; Gilson, J.P.; Mariey, L.; Travert, A.; Mauge, F. IR study of the interaction of phenol with oxides and sulfided CoMo catalysts for bio-fuel hydrodeoxygenation. Catal. Today 2011, 172, 132–135. [Google Scholar] [CrossRef]
- Roth, W.; Imhof, P.; Gerhards, M.; Schumm, S.; Kleinermanns, K. Reassignment of ground and first excited state vibrations in phenol. Chem. Phys. 2000, 252, 247–256. [Google Scholar] [CrossRef]
- Karge, H.G. Characterization by IR spectroscopy. In Verified Syntheses of Zeolitic Materials; Robson, H., Lillerud, K.P., Eds.; Elsevier Science: Amsterdam, The Netherlands, 2001; pp. 69–71. [Google Scholar]
- Colthup, N.B.; Daly, L.H.; Wiberley, S.E. Methyl and Methylene Groups. In Introduction to Infrared and Raman Spectroscopy; Colthup, N.B., Daly, L.H., Wiberley, S.E., Eds.; Academic Press: San Diego, CA, USA, 1990; pp. 215–233. [Google Scholar]
- Jo, C.; Ryoo, R.; Zilkova, N.; Vitvarova, D.; Cejka, J. The effect of MFI zeolite lamellar and related mesostructures on toluene disproportionation and alkylation. Catal. Sci. Technol. 2013, 3, 2119–2129. [Google Scholar] [CrossRef]
- Bhatia, S.; Beltramini, J.; Do, D.D. Deactivation of zeolite catalysts. Catal. Rev. Sci. Eng. 1989, 31, 431–480. [Google Scholar] [CrossRef]
- Guisnet, M.; Costa, L.; Ribeiro, F.R. Prevention of zeolite deactivation by coking. J. Mol. Catal. A Chem. 2009, 305, 69–83. [Google Scholar] [CrossRef]
- Argyle, M.D.; Bartholomew, C.H. Heterogeneous catalyst deactivation and regeneration: A Review. Catalysts 2015, 5, 145–269. [Google Scholar] [CrossRef]
- Cerqueira, H.S.; Caeiro, G.; Costa, L.; Ramôa Ribeiro, F. Deactivation of FCC catalysts. J. Mol. Catal. A Chem. 2008, 292, 1–13. [Google Scholar] [CrossRef]
- Vogt, E.T.; Weckhuysen, B.M. Fluid catalytic cracking: Recent developments on the grand old lady of zeolite catalysis. Chem. Soc. Rev. 2015, 44, 7342–7370. [Google Scholar] [CrossRef]
- Luo, W.H.; Cao, W.X.; Bruijnincx, P.C.A.; Lin, L.; Wang, A.Q.; Zhang, T. Zeolite-supported metal catalysts for selective hydrodeoxygenation of biomass-derived platform molecules. Green Chem. 2019, 21, 3744–3768. [Google Scholar] [CrossRef]
- Shi, Y.C.; Xing, E.H.; Wu, K.J.; Wang, J.L.; Yang, M.D.; Wu, Y.L. Recent progress on upgrading of bio-oil to hydrocarbons over metal/zeolite bifunctional catalysts. Catal. Sci. Technol. 2017, 7, 2385–2415. [Google Scholar] [CrossRef]
- Maier, S.M.; Jentys, A.; Lercher, J.A. Steaming of zeolite BEA and its effect on acidity: A comparative NMR and IR spectroscopic study. J. Phys. Chem. C 2011, 115, 8005–8013. [Google Scholar] [CrossRef]
- Baerlocher, C.; McCusker, L.B.; Treacy, M.M.J. Database of Zeolite Structures. Available online: http://www.iza-structure.org/databases/ (accessed on 19 December 2020).
- Baertsch, C.D.; Funke, H.H.; Falconer, J.L.; Noble, R.D. Permeation of aromatic hydrocarbon vapors through silicalite-zeolite membranes. J. Phys. Chem. 1996, 100, 7676–7679. [Google Scholar] [CrossRef]
- Xu, W.; Miller, S.J.; Agrawal, P.K.; Jones, C.W. Positive effect of water on zeolite BEA Catalyzed alkylation of phenol with propylene. Catal. Lett. 2013, 144, 434–438. [Google Scholar] [CrossRef]
- NIST Mass Spectrometry Data Center; Wallace, W.E. (Director). Infrared Spectra. In NIST Chemistry WebBook, NIST Standard Reference Database Number 69; Linstrom, P.J., Mallard, W.G., Eds.; National Institute of Standards and Technology: Gaithersburg, MD, USA, 2018. [Google Scholar] [CrossRef]
- Rozwadowski, M.; Lezanska, M.; Wloch, J.; Erdmann, K.; Golembiewski, R.; Kornatowski, J. Investigation of coke deposits on Al-MCM-41. Chem. Mater. 2001, 13, 1609–1616. [Google Scholar] [CrossRef]
- Palumbo, L.; Bonino, F.; Beato, P.; Bjorgen, M.; Zecchina, A.; Bordiga, S. Conversion of methanol to hydrocarbons: Spectroscopic characterization of Carbonaceous species formed over H-ZSM-5. J. Phys. Chem. C 2008, 112, 9710–9716. [Google Scholar] [CrossRef]
- Haider, M.H.; D’Agostino, C.; Dummer, N.F.; Mantle, M.D.; Gladden, L.F.; Knight, D.W.; Willock, D.J.; Morgan, D.J.; Taylor, S.H.; Hutchings, G.J. The effect of grafting zirconia and ceria onto alumina as a support for silicotungstic acid for the catalytic dehydration of glycerol to acrolein. Chem. Eur. J. 2014, 20, 1743–1752. [Google Scholar] [CrossRef]
- Kortunov, P.; Vasenkov, S.; Kärger, J.; Fé Elía, M.; Perez, M.; Stöcker, M.; Papadopoulos, G.; Theodorou, D.; Drescher, B.; McElhiney, G. Diffusion in fluid catalytic cracking catalysts on various displacement scales and its role in catalytic performance. Chem. Mater. 2005, 17, 2466–2474. [Google Scholar] [CrossRef]
- D’Agostino, C.; Ryabenkova, Y.; Miedziak, P.J.; Taylor, S.H.; Hutchings, G.J.; Gladden, L.F.; Mantle, M.D. Deactivation studies of a carbon supported AuPt nanoparticulate catalyst in the liquid-phase aerobic oxidation of 1,2-propanediol. Catal. Sci. Technol. 2014, 4, 1313–1322. [Google Scholar] [CrossRef]
- Possato, L.G.; Diniz, R.N.; Garetto, T.; Pulcinelli, S.H.; Santilli, C.V.; Martins, L. A comparative study of glycerol dehydration catalyzed by micro/mesoporous MFI zeolites. J. Catal. 2013, 300, 102–112. [Google Scholar] [CrossRef]
- Rodrigues, M.V.; Vignatti, C.; Garetto, T.; Pulcinelli, S.H.; Santilli, C.V.; Martins, L. Glycerol dehydration catalyzed by MWW zeolites and the changes in the catalyst deactivation caused by porosity modification. Appl. Catal. A Gen. 2015, 495, 84–91. [Google Scholar] [CrossRef]
- de Miguel Mercader, F.; Groeneveld, M.J.; Kersten, S.R.A.; Way, N.W.J.; Schaverien, C.J.; Hogendoorn, J.A. Production of advanced biofuels: Co-processing of upgraded pyrolysis oil in standard refinery units. Appl. Catal. B Environ. 2010, 96, 57–66. [Google Scholar] [CrossRef]
- Demirbas, A. Competitive liquid biofuels from biomass. Appl. Energy 2011, 88, 17–28. [Google Scholar] [CrossRef]
- Barrett, E.P.; Joyner, L.G.; Halenda, P.P. The determination of pore volume and area distributions in porous substances. I. Computations from nitrogen isotherms. J. Am. Chem. Soc. 2002, 73, 373–380. [Google Scholar] [CrossRef]
- Emeis, C.A. Determination of integrated molar extinction coefficients for infrared-absorption bands of pyridine adsorbed on solid acid catalysts. J. Catal. 1993, 141, 347–354. [Google Scholar] [CrossRef]
- Sadezky, A.; Muckenhuber, H.; Grothe, H.; Niessner, R.; Pöschl, U. Raman microspectroscopy of soot and related carbonaceous materials: Spectral analysis and structural information. Carbon 2005, 43, 1731–1742. [Google Scholar] [CrossRef]
- Magnoux, P.; Roger, P.; Canaff, C.; Fouche, V.; Gnep, N.S.; Guisnet, M. New Technique for the Characterization of Carbonaceous Compounds Responsible for Zeolite Deactivation. In Studies in Surface Science and Catalysis; Delmon, B., Froment, G.F., Eds.; Elsevier: Amsterdam, The Netherlands, 1987; Volume 34, pp. 317–330. [Google Scholar] [CrossRef]
- Momma, K.; Izumi, F. VESTA 3for three-dimensional visualization of crystal, volumetric and morphology data. J. Appl. Crystallogr. 2011, 44, 1272–1276. [Google Scholar] [CrossRef]



| Water Content | 0% | 3% | 6% | 9% | 12% |
| Deactivation Constant kd/h−1 | 1.269 | 0.143 | 0.114 | 0.116 | 0.100 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Stellato, M.J.; Innocenti, G.; Bommarius, A.S.; Sievers, C. Pore Blocking by Phenolates as Deactivation Path during the Cracking of 4-Propylphenol over ZSM-5. Catalysts 2021, 11, 721. https://doi.org/10.3390/catal11060721
Stellato MJ, Innocenti G, Bommarius AS, Sievers C. Pore Blocking by Phenolates as Deactivation Path during the Cracking of 4-Propylphenol over ZSM-5. Catalysts. 2021; 11(6):721. https://doi.org/10.3390/catal11060721
Chicago/Turabian StyleStellato, Michael J., Giada Innocenti, Andreas S. Bommarius, and Carsten Sievers. 2021. "Pore Blocking by Phenolates as Deactivation Path during the Cracking of 4-Propylphenol over ZSM-5" Catalysts 11, no. 6: 721. https://doi.org/10.3390/catal11060721
APA StyleStellato, M. J., Innocenti, G., Bommarius, A. S., & Sievers, C. (2021). Pore Blocking by Phenolates as Deactivation Path during the Cracking of 4-Propylphenol over ZSM-5. Catalysts, 11(6), 721. https://doi.org/10.3390/catal11060721







