A Brief Review of Catalytic Cathode Materials for Na-CO2 Batteries
Abstract
1. Introduction
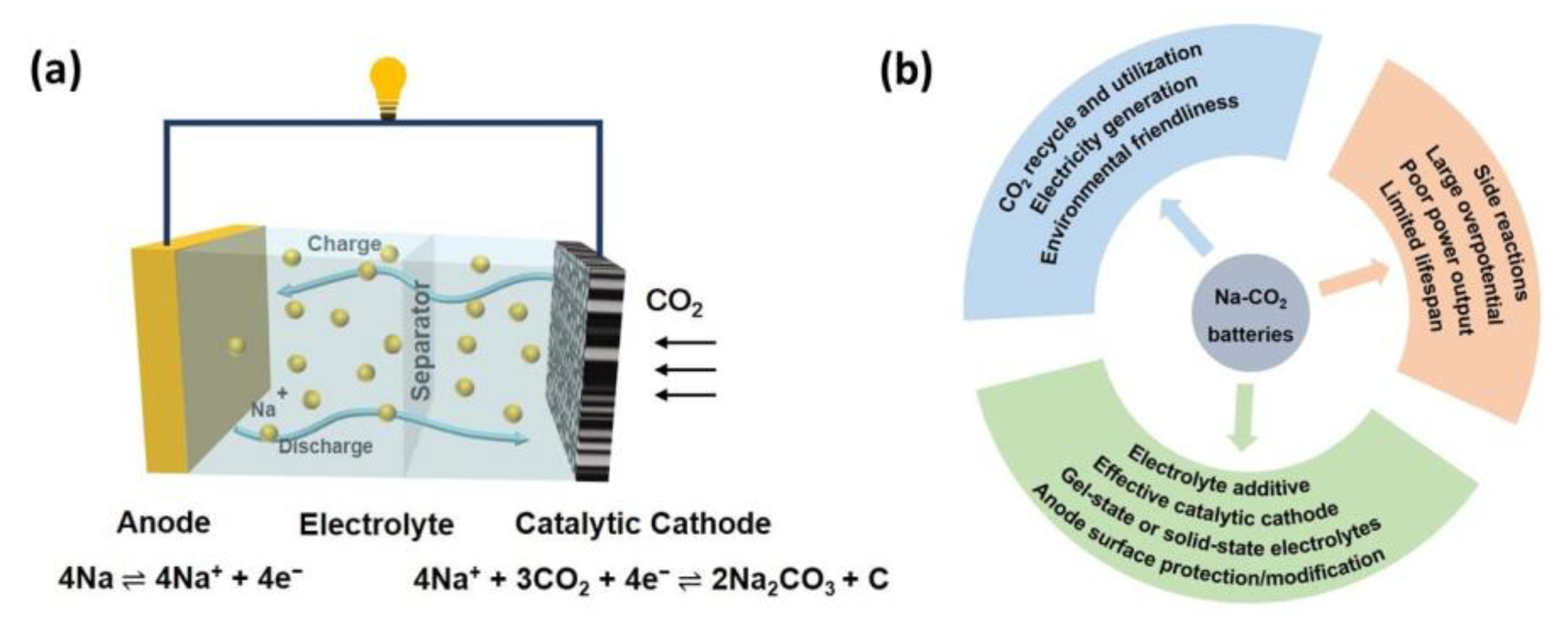
2. Na-CO2 Batteries
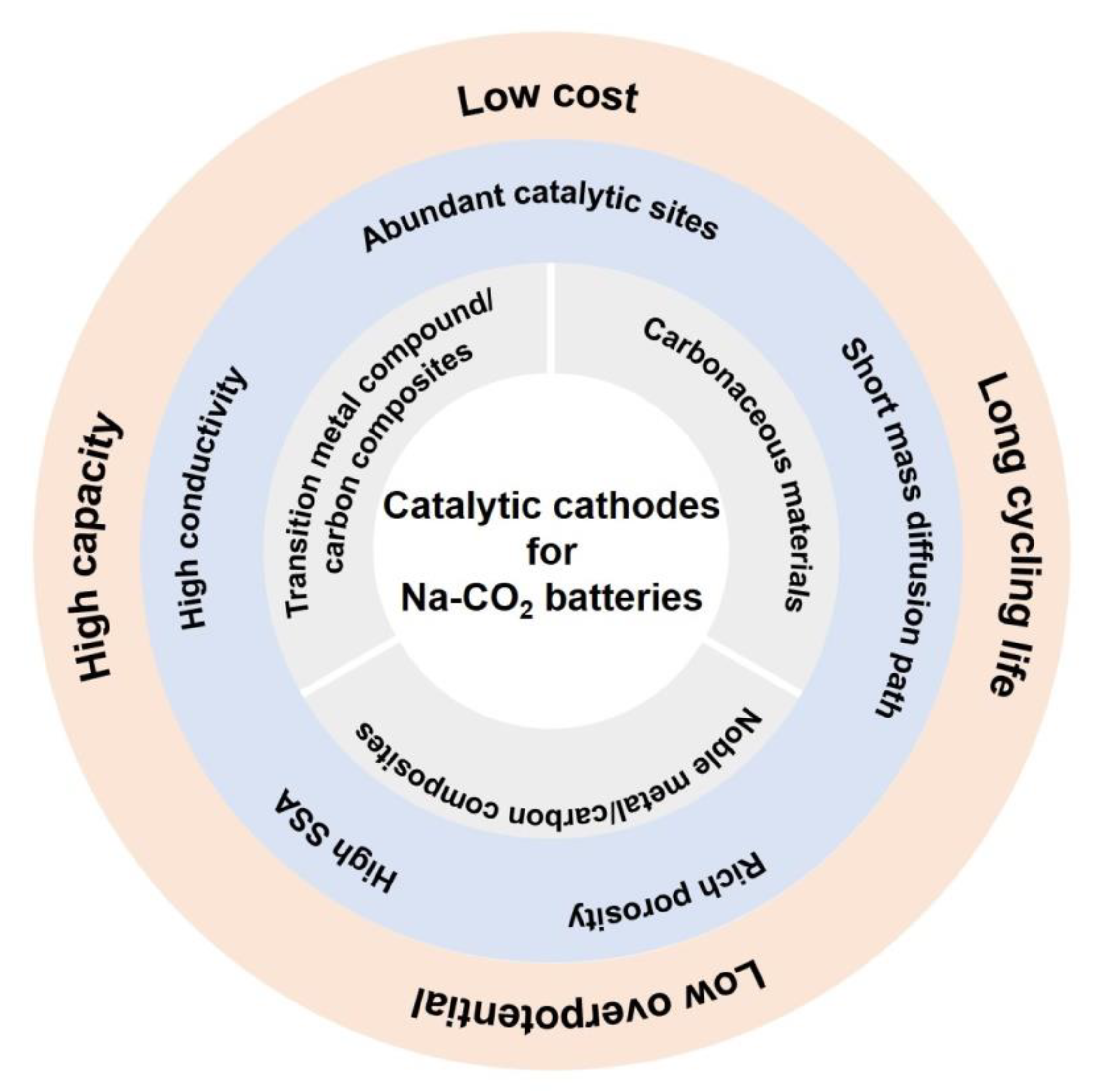
3. Catalytic Cathode Materials for Na-CO2 Batteries
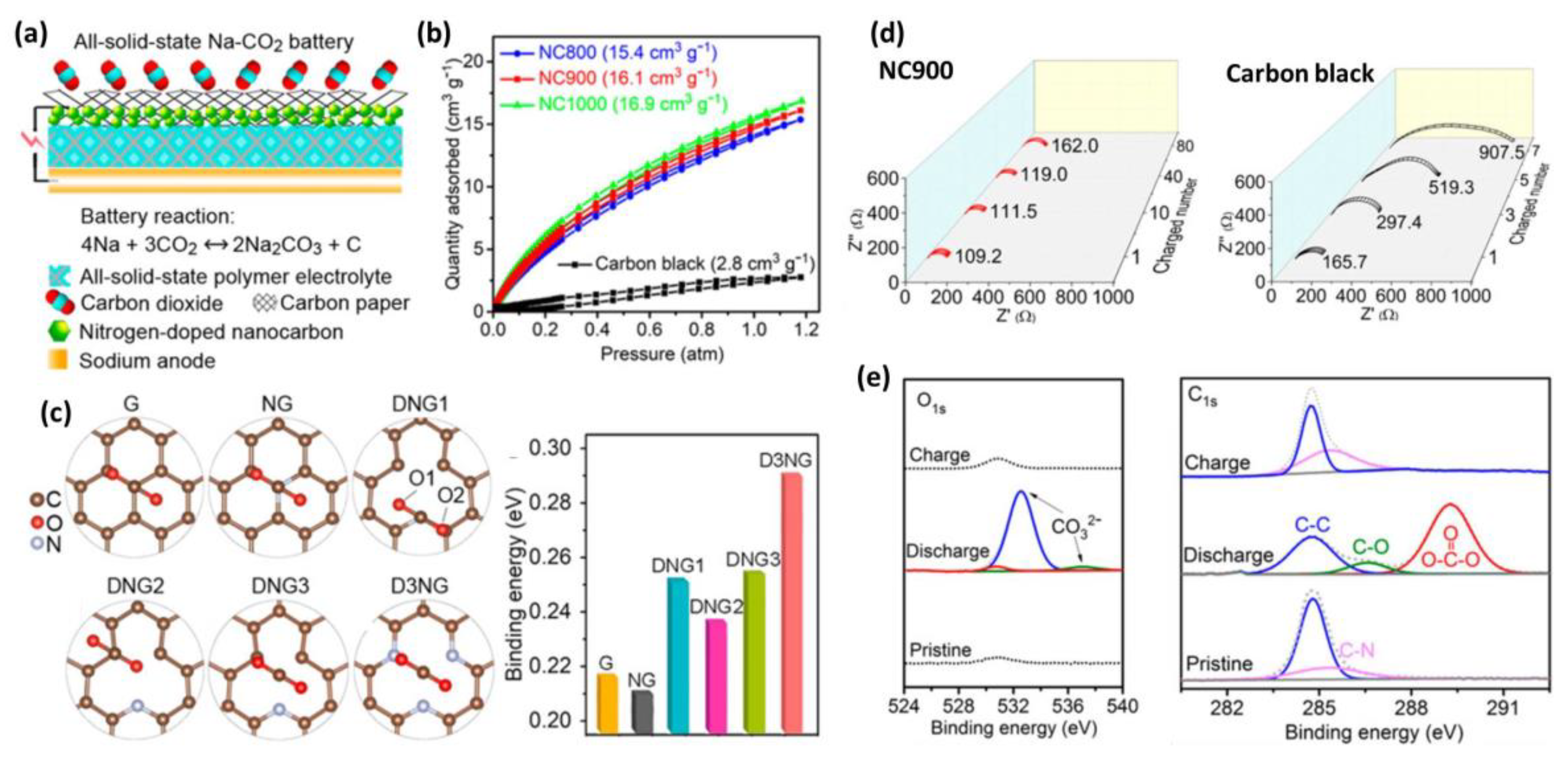
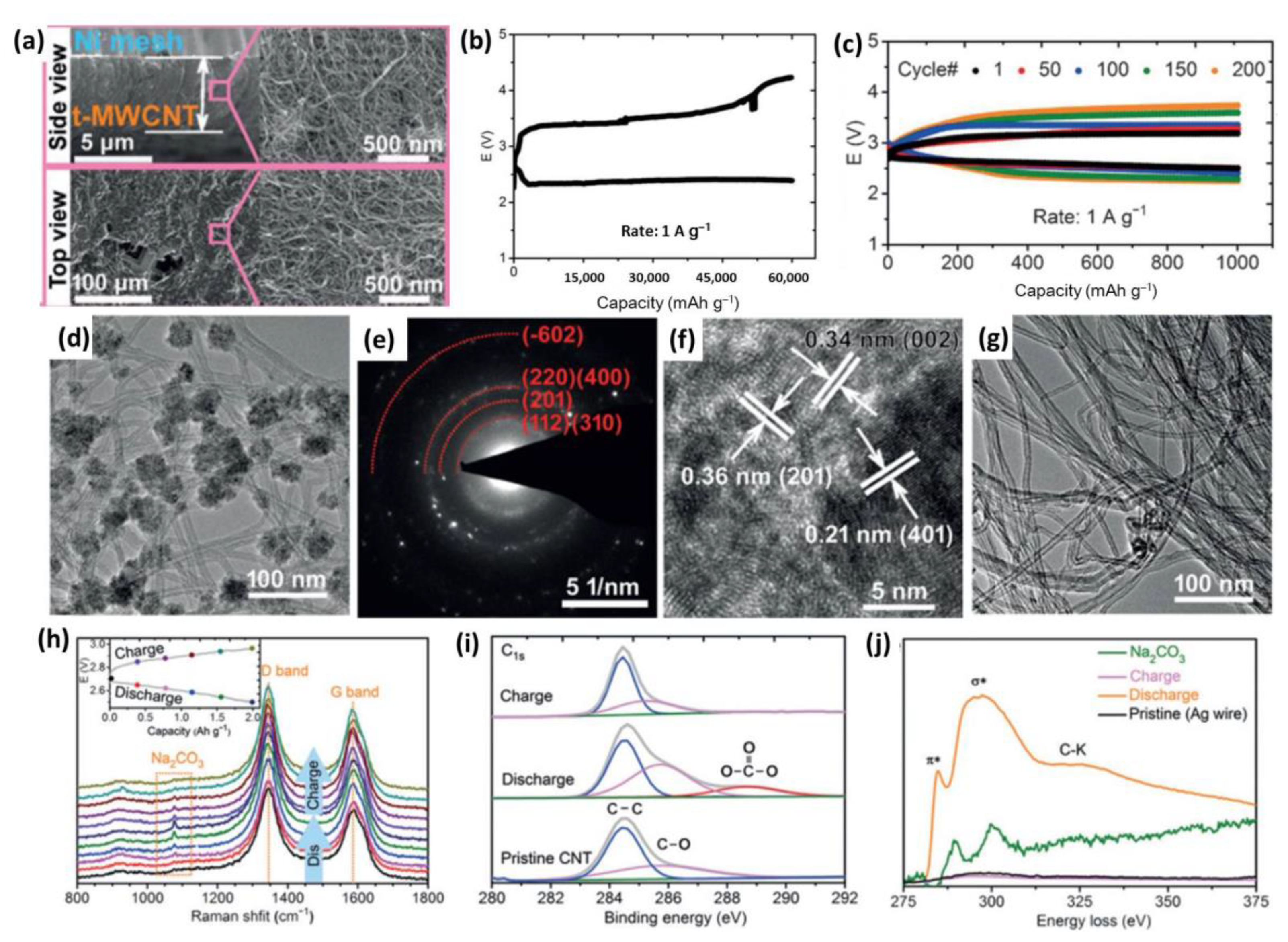
3.1. Carbonaceous Materials
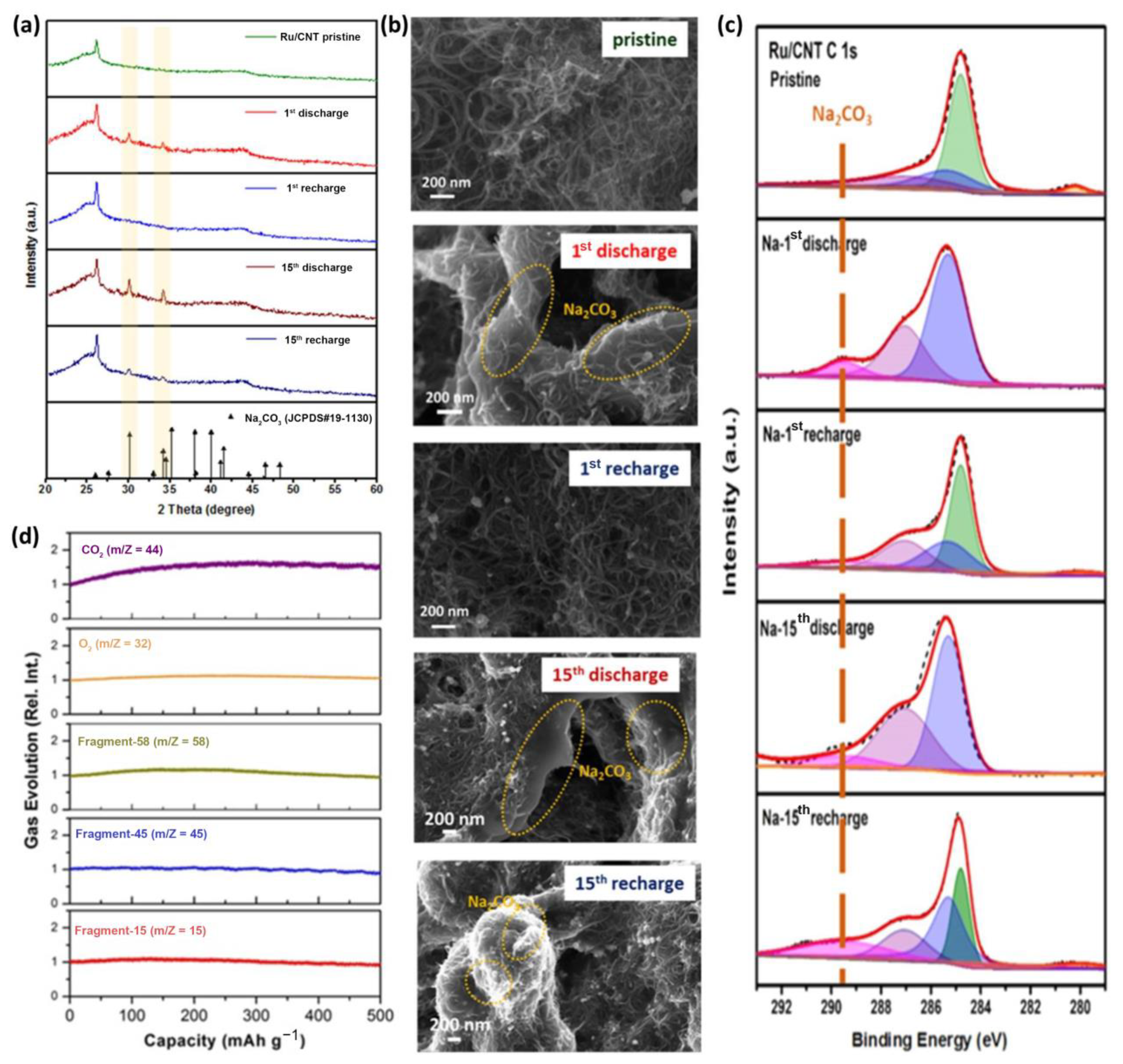
3.2. Noble Metal/Carbon Composites
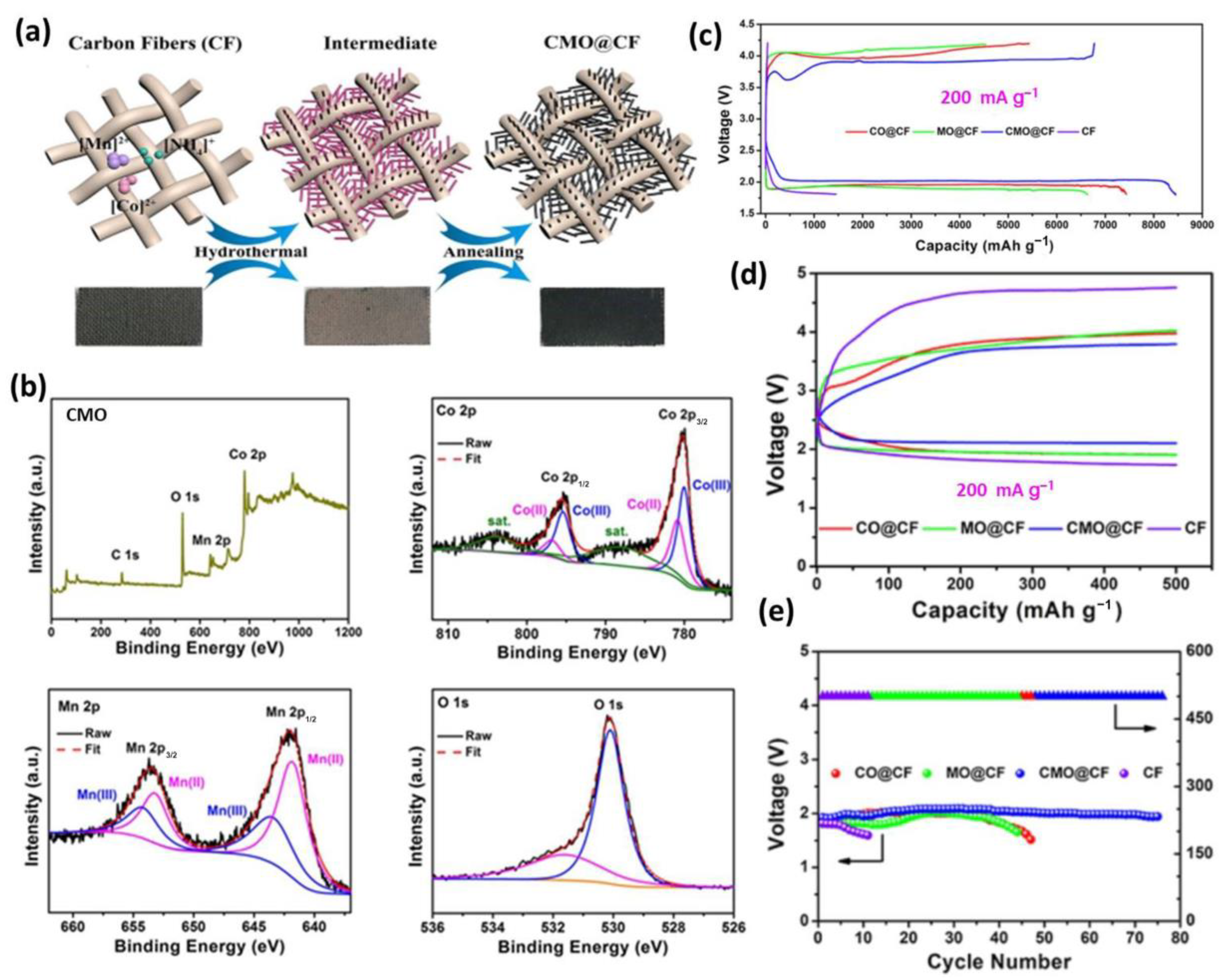
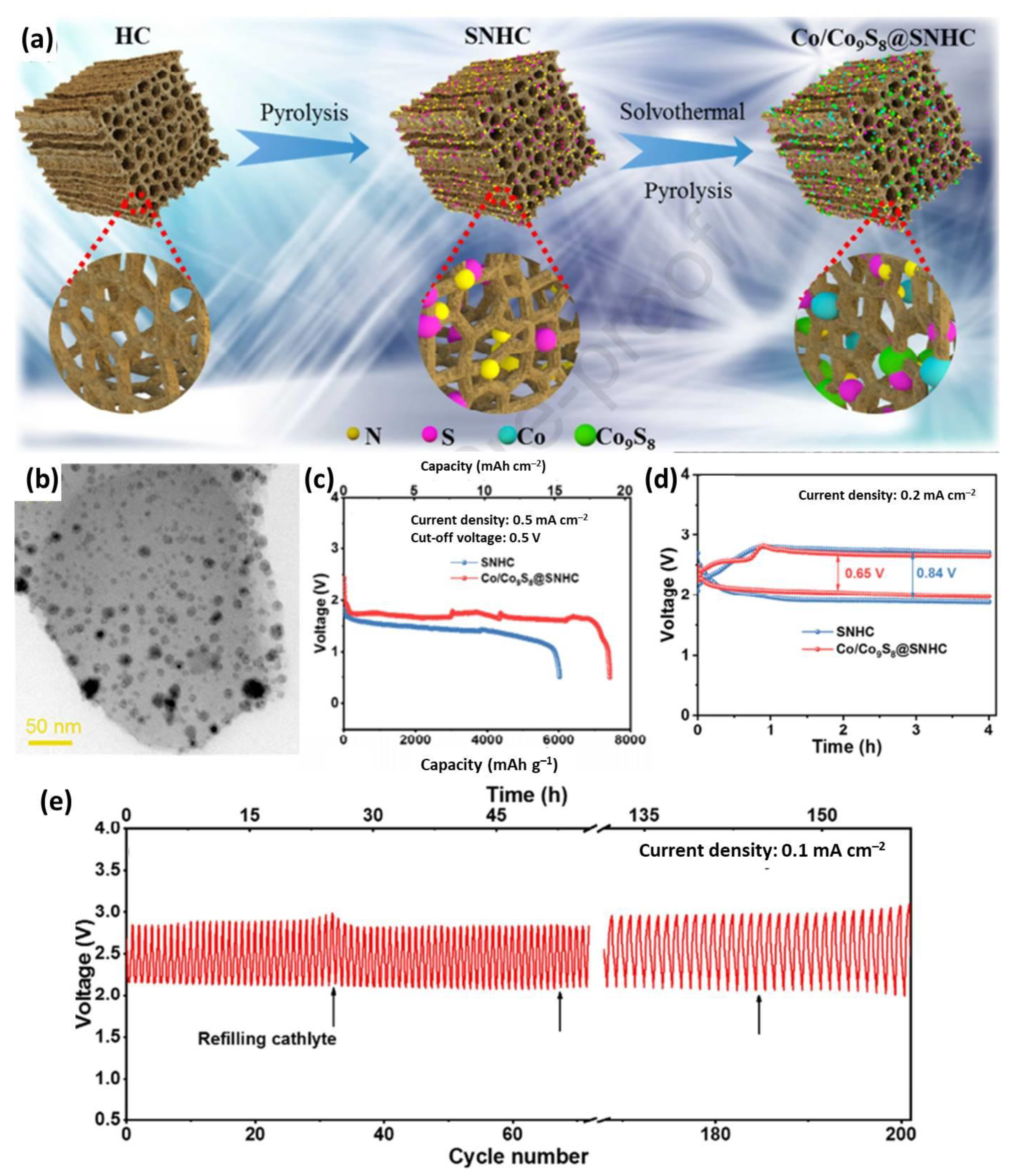
3.3. Transition Metal Compound/Carbon Composites
4. Conclusions and Outlook
Author Contributions
Funding
Conflicts of Interest
References
- Liu, Y.; Zhu, Y.; Cui, Y. Challenges and opportunities towards fast-charging battery materials. Nat. Energy 2019, 4, 540–550. [Google Scholar] [CrossRef]
- Wu, N.; Qiao, X.; Shen, J.; Liu, G.; Sun, T.; Wu, H.; Hou, H.; Liu, X.; Zhang, Y.; Ji, X. Anatase inverse opal TiO2-x@N-doped C induced the dominant pseudocapacitive effect for durable and fast lithium/sodium storage. Electrochim. Acta 2019, 299, 540–548. [Google Scholar] [CrossRef]
- Simon, P.; Gogotsi, Y. Perspectives for electrochemical capacitors and related devices. Nat. Mater. 2020, 19, 1151–1163. [Google Scholar] [CrossRef] [PubMed]
- Sui, D.; Xie, Y.; Zhao, W.; Zhang, H.; Zhou, Y.; Qin, X.; Ma, Y.; Yang, Y.; Chen, Y. A high-performance ternary Si composite anode material with crystal graphite core and amorphous carbon shell. J. Power Sources 2018, 384, 328–333. [Google Scholar] [CrossRef]
- Guo, D.; Yang, M.; Zhang, L.; Li, Y.; Wang, J.; Liu, G.; Wu, N.; Kim, J.-K.; Liu, X. Cr2O3 nanosheet/carbon cloth anode with strong interaction and fast charge transfer for pseudocapacitive energy storage in lithium-ion batteries. RSC Adv. 2019, 9, 33446–33453. [Google Scholar] [CrossRef]
- Zhao, Z.; Yi, Z.; Li, H.; Pathak, R.; Yang, Z.; Wang, X.; Qiao, Q. Synergetic effect of spatially separated dual co-catalyst for accelerating multiple conversion reaction in advanced lithium sulfur batteries. Nano Energy 2021, 81, 105621. [Google Scholar] [CrossRef]
- Pathak, R.; Zhou, Y.; Qiao, Q. Recent Advances in Lithiophilic Porous Framework toward Dendrite-Free Lithium Metal Anode. Appl. Sci. 2020, 10, 4185. [Google Scholar] [CrossRef]
- Zhou, F.; Li, Z.; Lu, Y.-Y.; Shen, B.; Guan, Y.; Wang, X.-X.; Yin, Y.-C.; Zhu, B.-S.; Lu, L.-L.; Ni, Y.; et al. Diatomite derived hierarchical hybrid anode for high performance all-solid-state lithium metal batteries. Nat. Commun. 2019, 10, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Sui, D.; Wu, M.; Liu, Y.; Yang, Y.; Zhang, H.; Ma, Y.; Zhang, L.; Chen, Y. High performance Li-ion capacitor fabricated with dual graphene-based materials. Nanotechnology 2020, 32, 015403. [Google Scholar] [CrossRef]
- Wang, F.; Liu, Y.; Wei, H.-J.; Li, T.-F.; Xiong, X.-H.; Wei, S.-Z.; Ren, F.-Z.; Volinsky, A.A. Recent advances and perspective in metal coordination materials-based electrode materials for potassium-ion batteries. Rare Met. 2021, 40, 448–470. [Google Scholar] [CrossRef]
- Liu, G.; Li, M.; Wu, N.; Cui, L.; Huang, X.; Liu, X.; Zhao, Y.; Chen, H.; Yuan, W.; Bai, Y. Single-Crystalline Particles: An Effective Way to Ameliorate the Intragranular Cracking, Thermal Stability, and Capacity Fading of the LiNi0.6Co0.2Mn0.2O2 Electrodes. J. Electrochem. Soc. 2018, 165, A3040–A3047. [Google Scholar] [CrossRef]
- Wu, N.; Wu, H.; Kim, J.-K.; Liu, X. Zhang, Y. Restoration of Degraded Nickel-Rich Cathode Materials for Long-Life Lith-ium-Ion Batteries. ChemElectroChem 2018, 5, 78–83. [Google Scholar] [CrossRef]
- Zhou, Z.; Ma, L.; Tan, C. Preparation of Layered (NH4)2V6O16·H2O Nanosheets as an Anode for Li-ion Batteries. Chem. J. Chin. Univ. 2021, 42, 662–670. [Google Scholar]
- Sheberla, D.; Bachman, J.C.; Elias, J.S.; Sun, C.-J.; Shao-Horn, Y.; Dincă, M. Conductive MOF electrodes for stable superca-pacitors with high areal capacitance. Nat. Mater. 2017, 16, 220–224. [Google Scholar] [CrossRef] [PubMed]
- Sui, D.; Xu, L.; Zhang, H.; Sun, Z.; Kan, B.; Ma, Y.; Chen, Y. A 3D cross-linked graphene-based honeycomb carbon composite with excellent confinement effect of organic cathode material for lithium-ion batteries. Carbon 2020, 157, 656–662. [Google Scholar] [CrossRef]
- Xu, C.; Fang, X.; Zhan, J.; Chen, J.; Liang, F. Progress for Metal-CO2 Batteries: Mechanism and Advanced Materials. Prog. Chem. 2020, 32, 836–850. [Google Scholar]
- Licht, S.; Douglas, A.; Ren, J.; Carter, R.; Lefler, M.; Pint, C.L. Carbon Nanotubes Produced from Ambient Carbon Dioxide for Environmentally Sustainable Lithium-Ion and Sodium-Ion Battery Anodes. ACS Central Sci. 2016, 2, 162–168. [Google Scholar] [CrossRef]
- Xie, Z.; Zhang, X.; Zhang, Z.; Zhou, Z. Metal-CO2 Batteries on the Road: CO2 from Contamination Gas to Energy Source. Adv. Mater. 2017, 29, 1605891. [Google Scholar] [CrossRef]
- Chen, H.; Liu, P.; Liu, J.; Feng, X.; Zhou, S. Mechanochemical in-situ incorporation of Ni on MgO/MgH2 surface for the selective O-/C-terminal catalytic hydrogenation of CO2 to CH4. J. Catal. 2021, 394, 397–405. [Google Scholar] [CrossRef]
- Xie, J.; Zhou, Z.; Wang, Y. Metal–CO2 Batteries at the Crossroad to Practical Energy Storage and CO2 Recycle. Adv. Funct. Mater. 2020, 30, 1908285. [Google Scholar] [CrossRef]
- Chang, S.; Liang, F.; Yao, Y.; Ma, W.; Yang, B.; Dai, Y. Research Progress of Metallic Carbon Dioxide Batteries. Acta Chim. Sin. 2018, 76, 515–525. [Google Scholar] [CrossRef]
- Appaturi, J.N.; Ramalingam, R.J.; Gnanamani, M.K.; Periyasami, G.; Arunachalam, P.; Adnan, R.; Adam, F.; Wasmiah, M.D.; Al-Lohedan, H.A. Review on Carbon Dioxide Utilization for Cycloaddition of Epoxides by Ionic Liquid-Modified Hybrid Catalysts: Effect of Influential Parameters and Mechanisms Insight. Catalysts 2020, 11, 4. [Google Scholar] [CrossRef]
- Maina, J.W.; Pringle, J.M.; Razal, J.M.; Nunes, S.; Vega, L.; Gallucci, F.; Dumée, L.F. Strategies for Integrated Capture and Conversion of CO2 from Dilute Flue Gases and the Atmosphere. ChemSusChem 2021, 14, 1805–1820. [Google Scholar] [CrossRef] [PubMed]
- Zurrer, T.; Wong, K.; Horlyck, J.; Lovell, E.C.; Wright, J.; Bedford, N.M.; Han, Z.; Liang, K.; Scott, J.; Amal, R. Mixed-Metal MOF-74 Templated Catalysts for Efficient Carbon Dioxide Capture and Methanation. Adv. Funct. Mater. 2021, 31, 2007624. [Google Scholar] [CrossRef]
- Bai, S.-T.; De Smet, G.; Liao, Y.; Sun, R.; Zhou, C.; Beller, M.; Maes, B.U.W.; Sels, B.F. Homogeneous and heterogeneous catalysts for hydrogenation of CO2 to methanol under mild conditions. Chem. Soc. Rev. 2021, 50. [Google Scholar] [CrossRef]
- Klinkenberg, N.; Kraft, S.; Polarz, S. Great Location: About Effects of Surface Bound Neighboring Groups for Passive and Active Fine-Tuning of CO2 Adsorption Properties in Model Carbon Capture Materials. Adv. Mater. 2021, 33, 2007734. [Google Scholar] [CrossRef]
- Mgolombane, M.; Majodina, S.; Bankole, O.; Ferg, E.; Ogunlaja, A. Influence of surface modification of zinc oxide–based nanomaterials on the photocatalytic reduction of carbon dioxide. Mater. Today Chem. 2021, 20, 100446. [Google Scholar] [CrossRef]
- Keith, D.W.; Holmes, G.; Angelo, D.S.; Heidel, K. A Process for Capturing CO2 from the Atmosphere. Joule 2018, 2, 1573–1594. [Google Scholar] [CrossRef]
- Chen, H.; Liu, J.; Liu, P.; Wang, Y.; Xiao, H.; Yang, Q.; Feng, X.; Zhou, S. Carbon-confined magnesium hydride nano-lamellae for catalytic hydrogenation of carbon dioxide to lower olefins. J. Catal. 2019, 379, 121–128. [Google Scholar] [CrossRef]
- Chen, H.; Yang, M.; Liu, J.; Lu, G.; Feng, X. Insight into the effects of electronegativity on the H2 catalytic activation for CO2 hydrogenation: Four transition metal cases from a DFT study. Catal. Sci. Technol. 2020, 10, 5641–5647. [Google Scholar] [CrossRef]
- Cai, F.; Hu, Z.; Chou, S.-L. Progress and Future Perspectives on Li(Na)-CO2 Batteries. Adv. Sustain. Syst. 2018, 2, 1800060. [Google Scholar] [CrossRef]
- Xu, S.; Das, S.K.; Archer, L.A. The Li-CO2 battery: A novel method for CO2 capture and utilization. RSC Adv. 2013, 3, 6656–6660. [Google Scholar] [CrossRef]
- Zhang, Z.; Zhang, Q.; Chen, Y.; Bao, J.; Zhou, X.; Xie, Z.; Wei, J.; Zhou, Z. The First Introduction of Graphene to Rechargeable Li-CO2Batteries. Angew. Chem. Int. Ed. 2015, 54, 6550–6553. [Google Scholar] [CrossRef]
- Liu, B.; Sun, Y.; Liu, L.; Chen, J.; Yang, B.; Xu, S.; Yan, X. Recent advances in understanding Li-CO2 electrochemistry. Energy Environ. Sci. 2019, 12, 887–922. [Google Scholar] [CrossRef]
- Xie, J.; Wang, Y. Recent Development of CO2 Electrochemistry from Li-CO2 Batteries to Zn-CO2 Batteries. Acc. Chem. Res. 2019, 52, 1721–1729. [Google Scholar] [CrossRef]
- Zhang, Z.; Bai, W.-L.; Wang, K.-X.; Chen, J.-S. Electrocatalyst design for aprotic Li-CO2 batteries. Energy Environ. Sci. 2020, 13, 4717–4737. [Google Scholar] [CrossRef]
- Hu, X.; Sun, J.; Li, Z.; Zhao, Q.; Chen, C.; Chen, J. Rechargeable Room-Temperature Na-CO2 Batteries. Angew. Chem. Int. Ed. 2016, 55, 6482–6486. [Google Scholar] [CrossRef]
- Hu, X.; Li, Z.; Chen, J. Flexible Li-CO2Batteries with Liquid-Free Electrolyte. Angew. Chem. Int. Ed. 2017, 56, 5785–5789. [Google Scholar] [CrossRef]
- Ma, W.; Liu, X.; Li, C.; Yin, H.; Xi, W.; Liu, R.; He, G.; Zhao, X.; Luo, J.; Ding, Y. Rechargeable Al-CO2 Batteries for Reversible Utilization of CO2. Adv. Mater. 2018, 30, e1801152. [Google Scholar] [CrossRef]
- Kim, J.; Seong, A.; Yang, Y.; Joo, S.; Kim, C.; Jeon, D.H.; Dai, L.; Kim, G. Indirect surpassing CO2 utilization in membrane-free CO2 battery. Nano Energy 2021, 82, 105741. [Google Scholar] [CrossRef]
- Wang, Y.; Wang, Y.; Feng, X.; Chen, W.; Ai, X.; Yang, H.; Cao, Y. Developments and Perspectives on Emerging High-Energy-Density Sodium-Metal Batteries. Chem 2019, 5, 2547–2570. [Google Scholar] [CrossRef]
- Zheng, Z.; Wu, C.; Gu, Q.; Konstantinov, K.; Wang, J. Research Progress and Future Perspectives on Rechargeable Na-O2 and Na-CO2 Batteries. Energy Environ. Mater. 2020, 4. [Google Scholar] [CrossRef]
- Okoshi, M.; Yamada, Y.; Yamada, A.; Nakai, H. Theoretical Analysis on De-Solvation of Lithium, Sodium, and Magnesium Cations to Organic Electrolyte Solvents. J. Electrochem. Soc. 2013, 160, A2160–A2165. [Google Scholar] [CrossRef]
- Das, S.K.; Xu, S.; Archer, L.A. Carbon dioxide assist for non-aqueous sodium–oxygen batteries. Electrochem. Commun. 2013, 27, 59–62. [Google Scholar] [CrossRef]
- Mu, X.; Pan, H.; He, P.; Zhou, H. Li-CO2 and Na-CO2 Batteries: Toward Greener and Sustainable Electrical Energy Storage. Adv. Mater. 2020, 32, 1903790. [Google Scholar]
- Jena, A.; Tong, Z.; Chang, H.; Hu, S.; Liu, R. Capturing carbon dioxide in Na-CO2 batteries: A route for green energy. J. Chin. Chem. Soc. 2021, 68, 421–428. [Google Scholar] [CrossRef]
- Hu, A.; Shu, C.; Xu, C.; Liang, R.; Li, J.; Zheng, R.; Li, M.; Long, J. Design strategies toward catalytic materials and cathode structures for emerging Li–CO2 batteries. J. Mater. Chem. A 2019, 7, 21605–21633. [Google Scholar] [CrossRef]
- Zhang, Z.; Wang, X.-G.; Zhang, X.; Xie, Z.; Chen, Y.-N.; Ma, L.; Peng, Z.; Zhou, Z. Verifying the Rechargeability of Li-CO2 Batteries on Working Cathodes of Ni Nanoparticles Highly Dispersed on N-Doped Graphene. Adv. Sci. 2018, 5, 1700567. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Zhang, Q.; Zhang, Z.; Chen, Y.; Xie, Z.; Wei, J.; Zhou, Z. Rechargeable Li–CO2 batteries with carbon nanotubes as air cathodes. Chem. Commun. 2015, 51, 14636–14639. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Wang, R.; Lyu, Y.; Li, H.; Chen, L. Rechargeable Li/CO2–O2 (2:1) battery and Li/CO2 battery. Energy Environ. Sci. 2014, 7, 677–681. [Google Scholar] [CrossRef]
- Wang, R.; Yu, X.; Bai, J.; Li, H.; Huang, X.; Chen, L.; Yang, X. Electrochemical decomposition of Li2CO3 in NiO–Li2CO3 nanocomposite thin film and powder electrodes. J. Power Sources 2012, 218, 113–118. [Google Scholar] [CrossRef]
- Liu, Q.; Tang, Y.; Sun, H.; Yang, T.; Sun, Y.; Du, C.; Jia, P.; Ye, H.; Chen, J.; Peng, Q.; et al. In Situ Electrochemical Study of Na-O2/CO2 Batteries in an Environmental Transmission Electron Microscope. ACS Nano 2020, 14, 13232–13245. [Google Scholar] [CrossRef]
- Meini, S.; Tsiouvaras, N.; Schwenke, K.U.; Piana, M.; Beyer, H.; Lange, L.; Gasteiger, H.A. Rechargeability of Li–air cathodes pre-filled with discharge products using an ether-based electrolyte solution: Implications for cycle-life of Li–air cells. Phys. Chem. Chem. Phys. 2013, 15, 11478–11493. [Google Scholar] [CrossRef] [PubMed]
- Zhou, H.; Liu, M.; Gao, H.; Hou, D.; Yu, C.; Liu, C.; Zhang, D.; Wu, J.-C.; Yang, J.; Chen, D. Dense integration of solvent-free electrodes for Li-ion supercabattery with boosted low temperature performance. J. Power Sources 2020, 473, 228553. [Google Scholar] [CrossRef]
- Lu, Y.; Cai, Y.; Zhang, Q.; Liu, L.; Niu, Z.; Chen, J. A compatible anode/succinonitrile-based electrolyte interface in all-solid-state Na–CO2 batteries. Chem. Sci. 2019, 10, 4306–4312. [Google Scholar] [CrossRef]
- Chi, X.; Liang, Y.; Hao, F.; Zhang, Y.; Whiteley, J.; Dong, H.; Hu, P.; Lee, S.; Yao, Y. Tailored Organic Electrode Material Compatible with Sulfide Electrolyte for Stable All-Solid-State Sodium Batteries. Angew. Chem. Int. Ed. 2018, 57, 2630–2634. [Google Scholar] [CrossRef] [PubMed]
- Tang, H.; Deng, Z.; Lin, Z.; Wang, Z.; Chu, I.-H.; Chen, C.; Zhu, Z.; Zheng, C.; Ong, S.P. Probing Solid–Solid Interfacial Re-actions in All-Solid-State Sodium-Ion Batteries with First-Principles Calculations. Chem. Mater. 2018, 30, 163–173. [Google Scholar] [CrossRef]
- Zhao, S.; Wang, C.; Du, D.; Li, L.; Chou, S.; Li, F.; Chen, J. Bifunctional Effects of Cation Additive on Na-O2 Batteries. Angew. Chem. Int. Ed. 2021, 60, 3205–3211. [Google Scholar] [CrossRef] [PubMed]
- Lin, X.; Sun, Y.; Sun, Q.; Luo, J.; Zhao, Y.; Zhao, C.; Yang, X.; Wang, C.; Huo, H.; Li, R.; et al. Reviving Anode Protection Layer in Na-O2 Batteries: Failure Mechanism and Resolving Strategy. Adv. Energy Mater. 2021, 11, 2003789. [Google Scholar] [CrossRef]
- Fan, L.; Li, X. Recent advances in effective protection of sodium metal anode. Nano Energy 2018, 53, 630–642. [Google Scholar] [CrossRef]
- Luo, W.; Lin, C.-F.; Zhao, O.; Noked, M.; Zhang, Y.; Rubloff, G.W.; Hu, L. Ultrathin Surface Coating Enables the Stable Sodium Metal Anode. Adv. Energy Mater. 2017, 7, 1601526. [Google Scholar] [CrossRef]
- Xu, Y.; Wang, C.; Matios, E.; Luo, J.; Hu, X.; Yue, Q.; Kang, Y.; Li, W. Sodium Deposition with a Controlled Location and Orientation for Dendrite-Free Sodium Metal Batteries. Adv. Energy Mater. 2020, 10, 2002308. [Google Scholar] [CrossRef]
- Hu, X.; Li, Z.; Zhao, Y.; Sun, J.; Zhao, Q.; Wang, J.; Tao, Z.; Chen, J. Quasi–solid state rechargeable Na-CO2 batteries with reduced graphene oxide Na anodes. Sci. Adv. 2017, 3, e1602396. [Google Scholar] [CrossRef] [PubMed]
- Xu, S.; Lu, Y.; Wang, H.; Abruña, H.D.; Archer, L.A. A rechargeable Na-CO2/O2 battery enabled by stable nanoparticle hybrid electrolytes. J. Mater. Chem. A 2014, 2, 17723–17729. [Google Scholar] [CrossRef]
- Hu, X.; Joo, P.H.; Matios, E.; Wang, C.; Luo, J.; Yang, K.; Li, W. Designing an All-Solid-State Sodium-Carbon Dioxide Battery Enabled by Nitrogen-Doped Nanocarbon. Nano Lett. 2020, 20, 3620–3626. [Google Scholar] [CrossRef]
- Wang, X.; Zhang, X.; Lu, Y.; Yan, Z.; Tao, Z.; Jia, D.; Chen, J. Flexible and Tailorable Na-CO2 Batteries Based on an All-Solid-State Polymer Electrolyte. ChemElectroChem 2018, 5, 3628–3632. [Google Scholar] [CrossRef]
- Xu, C.; Zhan, J.; Wang, Z.; Fang, X.; Chen, J.; Liang, F.; Zhao, H.; Lei, Y. Biomass-derived highly dispersed Co/Co9S8 nanoparticles encapsulated in S, N-co-doped hierarchically porous carbon as an efficient catalyst for hybrid Na-CO2 batteries. Mater. Today Energy 2021, 19, 100594. [Google Scholar] [CrossRef]
- Hu, C.; Gong, L.; Xiao, Y.; Yuan, Y.; Bedford, N.M.; Xia, Z.; Ma, L.; Wu, T.; Lin, Y.; Connell, J.W.; et al. High-Performance, Long-Life, Rechargeable Li-CO2 Batteries based on a 3D Holey Graphene Cathode Im-planted with Single Iron Atoms. Adv. Mater. 2020, 32, 1907436. [Google Scholar] [CrossRef]
- Li, X.; Zhou, J.; Zhang, J.; Li, M.; Bi, X.; Liu, T.; He, T.; Cheng, J.; Zhang, F.; Li, Y.; et al. Bamboo-Like Nitro-gen-Doped Carbon Nanotube Forests as Durable Metal-Free Catalysts for Self-Powered Flexible Li-CO2 Batteries. Adv. Mater. 2019, 31, 1903852. [Google Scholar] [CrossRef]
- Wu, N.; Shen, J.; Sun, L.; Yuan, M.; Shao, Y.; Ma, J.; Liu, G.; Guo, D.; Liu, X.; He, Y.-B. Hierarchical N-doped graphene coated 1D cobalt oxide microrods for robust and fast lithium storage at elevated temperature. Electrochim. Acta 2019, 310, 70–77. [Google Scholar] [CrossRef]
- Zhu, Y.; Zhang, Z.; Li, W.; Lei, Z.; Cheng, N.; Tan, Y.; Mu, S.; Sun, X. Highly Exposed Active Sites of Defect-Enriched Derived MOFs for Enhanced Oxygen Reduction Reaction. ACS Sustain. Chem. Eng. 2019, 7, 17855–17862. [Google Scholar] [CrossRef]
- Wu, S.; Qiao, Y.; Yang, S.; Ishida, M.; He, P.; Zhou, H. Organic hydrogen peroxide-driven low charge potentials for high-performance lithium-oxygen batteries with carbon cathodes. Nat. Commun. 2017, 8, 15607. [Google Scholar] [CrossRef]
- Sun, J.; Zhao, N.; Li, Y.; Guo, X.; Feng, X.; Liu, X.; Liu, Z.; Cui, G.; Zheng, H.; Gu, L.; et al. A Rechargeable Li-Air Fuel Cell Battery Based on Garnet Solid Electrolytes. Sci. Rep. 2017, 7, 41217. [Google Scholar] [CrossRef]
- Qie, L.; Lin, Y.; Connell, J.W.; Xu, J.; Dai, L. Highly Rechargeable Lithium-CO2 Batteries with a Boron- and Nitrogen-Codoped Holey-Graphene Cathode. Angew. Chem. Int. Ed. 2017, 56, 6970–6974. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.-J.; Park, J.E.; Kim, S.; Lim, M.S.; Jin, A.; Kim, O.-H.; Kim, M.J.; Lee, K.-S.; Kim, J.; Kim, S.-S.; et al. Biomass-Derived Air Cathode Materials: Pore-Controlled S,N-Co-doped Carbon for Fuel Cells and Metal–Air Batteries. ACS Catal. 2019, 9, 3389–3398. [Google Scholar] [CrossRef]
- Sun, J.; Lu, Y.; Yang, H.; Han, M.; Shao, L.; Chen, J. Rechargeable Na-CO2 Batteries Starting from Cathode of Na2CO3 and Carbon Nanotubes. Research 2018, 1, 6914626. [Google Scholar] [CrossRef] [PubMed]
- Shen, J.; Wu, H.; Sun, W.; Wu, Q.; Zhen, S.; Wang, Z.; Sun, K.; Wang, S. Biomass-derived hierarchically porous carbon skeletons with in situ decorated IrCo nanoparticles as high-performance cathode catalysts for Li-O2 batteries. J. Mater. Chem. A 2019, 7, 10662–10671. [Google Scholar] [CrossRef]
- Tong, Z.; Wang, S.-B.; Fang, M.-H.; Lin, Y.-T.; Tsai, K.-T.; Tsai, S.-Y.; Yin, L.-C.; Hu, S.-F.; Liu, R.-S. Na-CO2 battery with NASICON-structured solid-state electrolyte. Nano Energy 2021, 85, 105972. [Google Scholar] [CrossRef]
- Guo, L.; Li, B.; Thirumal, V.; Song, J. Advanced rechargeable Na-CO2 batteries enabled by a ruthenium@porous carbon composite cathode with enhanced Na2CO3 reversibility. Chem. Commun. 2019, 55, 7946–7949. [Google Scholar] [CrossRef] [PubMed]
- Thoka, S.; Tsai, C.-M.; Tong, Z.; Jena, A.; Wang, F.-M.; Hsu, C.-C.; Chang, H.; Hu, S.-F.; Liu, R.-S. Comparative Study of Li-CO2 and Na–CO2 Batteries with Ru@CNT as a Cathode Catalyst. ACS Appl. Mater. Interfaces 2021, 13, 480–490. [Google Scholar] [CrossRef]
- Li, W.; Hu, Z.-Y.; Zhang, Z.; Wei, P.; Zhang, J.; Pu, Z.; Zhu, J.; He, D.; Mu, S.; Van Tendeloo, G. Nano-single crystal coalesced PtCu nanospheres as robust bifunctional catalyst for hydrogen evolution and oxygen reduction reactions. J. Catal. 2019, 375, 164–170. [Google Scholar] [CrossRef]
- Dong, H.; Tang, P.; Wang, X.; Li, K.; Wang, Y.; Wang, D.; Liu, H.; Yang, S.-T.; Wu, C. Pt/NiO Microsphere Composite as Efficient Multifunctional Catalysts for Nonaqueous Lithium–Oxygen Batteries and Alkaline Fuel Cells: The Synergistic Effect of Pt and Ni. ACS Appl. Mater. Interfaces 2019, 11, 39789–39797. [Google Scholar] [CrossRef]
- Bui, H.T.; Kim, D.Y.; Kim, Y.Y.; Le, N.H.; Kim, D.W.; Suk, J.; Kang, Y. Macroporous carbon nanofiber decorated with platinum nanorods as free-standing cathodes for high-performance Li-O2 batteries. Carbon 2019, 154, 448–456. [Google Scholar] [CrossRef]
- Zhu, Y.; Feng, S.; Zhang, P.; Guo, M.; Wang, Q.; Wu, D.; Zhang, L.; Li, H.; Wang, H.; Chen, L.; et al. Probing the electrochemical evolutions of Na-CO2 nanobatteries on Pt@NCNT cathodes using in-situ environmental TEM. Energy Storage Mater. 2020, 33, 88–94. [Google Scholar] [CrossRef]
- Zhang, Z.; Zhang, Z.; Liu, P.; Xie, Y.; Cao, K.; Zhou, Z. Identification of cathode stability in Li-CO2 batteries with Cu na-noparticles highly dispersed on N-doped graphene. J. Mater. Chem. A 2018, 6, 3218–3223. [Google Scholar] [CrossRef]
- Li, S.; Liu, Y.; Zhou, J.; Hong, S.; Dong, Y.; Wang, J.; Gao, X.; Qi, P.; Han, Y.; Wang, B. Monodispersed MnO nanoparticles in graphene-an interconnected N-doped 3D carbon framework as a highly efficient gas cathode in Li-CO2 batteries. Energy Environ. Sci. 2019, 12, 1046–1054. [Google Scholar] [CrossRef]
- Zhu, J.; Qu, T.; Su, F.; Wu, Y.; Kang, Y.; Chen, K.; Yao, Y.; Ma, W.; Yang, B.; Dai, Y.; et al. Highly dispersed Co nanoparticles decorated on a N-doped defective carbon nano-framework for a hybrid Na–air battery. Dalton Trans. 2020, 49, 1811–1821. [Google Scholar] [CrossRef]
- Wang, P.; Pu, Z.; Li, W.; Zhu, J.; Zhang, C.; Zhao, Y.; Mu, S. Coupling NiSe2-Ni2P heterostructure nanowrinkles for highly efficient overall water splitting. J. Catal. 2019, 377, 600–608. [Google Scholar] [CrossRef]
- Fang, C.; Luo, J.; Jin, C.; Yuan, H.; Sheng, O.; Huang, H.; Gan, Y.; Xia, Y.; Liang, C.; Zhang, J.; et al. Enhancing Catalyzed Decomposition of Na2CO3 with Co2MnOx Nanowire-Decorated Carbon Fibers for Advanced Na-CO2 Batteries. ACS Appl. Mater. Interfaces 2018, 10, 17240–17248. [Google Scholar] [CrossRef]
- Thoka, S.; Tong, Z.; Jena, A.; Hung, T.-F.; Wu, C.-C.; Chang, W.-S.; Wang, F.-M.; Wang, X.-C.; Yin, L.-C.; Chang, H.; et al. High-performance Na-CO2 batteries with ZnCo2O4@CNT as the cathode catalyst. J. Mater. Chem. A 2020, 8, 23974–23982. [Google Scholar] [CrossRef]
- Xu, C.; Zhang, K.; Zhang, D.; Chang, S.; Liang, F.; Yan, P.; Yao, Y.; Qu, T.; Zhan, J.; Ma, W.; et al. Reversible hybrid sodium-CO2 batteries with low charging voltage and long-life. Nano Energy 2020, 68, 104318. [Google Scholar] [CrossRef]
- Zhang, W.; Hu, C.; Guo, Z.; Dai, L. High-Performance K-CO2 Batteries Based on Metal-Free Carbon Electrocatalysts. Angew. Chem. Int. Ed. 2020, 59, 3470–3474. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Tang, Y.; Liu, Q.; Yang, T.; Du, C.; Jia, P.; Wang, Z.; Tang, Y.; Li, Y.; Shen, T.; et al. Probing the charging and discharging behavior of K-CO2 nanobatteries in an aberration corrected environmental transmission electron microscope. Nano Energy 2018, 53, 544–549. [Google Scholar] [CrossRef]
- Xie, J.; Wang, X.; Lv, J.; Huang, Y.; Wu, M.; Wang, Y.; Yao, J. Reversible Aqueous Zinc-CO2 Batteries Based on CO2 -HCOOH Interconversion. Angew. Chem. Int. Ed. 2018, 57, 16996–17001. [Google Scholar] [CrossRef]
- Wang, K.; Wu, Y.; Cao, X.; Gu, L.; Hu, J. A Zn-CO2 Flow Battery Generating Electricity and Methane. Adv. Funct. Mater. 2019, 30, 1908965. [Google Scholar] [CrossRef]
| Cathode | Overpotential (V) | Discharge Capacity (mAh g−1) | Cycling Stability | Ref. |
|---|---|---|---|---|
| t-MWCNT | 0.6 (1 A g−1) | 60,000 (1 A g−1) | 200 cycles (2000 mAh g−1) | [37] |
| MWCNTs | 1.53 (50 mA g−1) | 7624 (50 mA g−1) | 50 cycles (1000 mAh g−1) | [55] |
| a-MCNT | 0.74 (100 mA g−1) | 5000 (50 mA g−1) | 400 cycles (1000 mAh g−1) | [63] |
| N-doped nanocarbon | 1.5 (200 mA g−1) | 10,500 (300 mA g−1) | 100 cycles (400 mAh g−1) | [65] |
| Ru@KB | 1.5 (200 mA g−1) | 11,537 (100 mA g−1) | 130 cycles (1000 mAh g−1) | [79] |
| Ru@CNT | ≈1.6 (100 mA g−1) | 19,533 (100 mA g−1) | 100 cycles (500 mAh g−1) | [80] |
| Ru/CNT | 1.4 (100 mA g−1) | 28,830 (100 mA g−1) | 70 cycles (500 mAh g−1) | [78] |
| CMO@CF | 1.47 (100 mA g−1) | 8448 (200 mA g−1) | 75 cycles (500 mAh g−1) | [89] |
| ZnCo2O4@CNT | 1.5 (100 mA g−1) | 12,475 (100 mA g−1) | 150 cycles (500 mAh g−1) | [90] |
| Co/Co9S8@SNHC | ≈0.32 V (0.2 mA cm−2) | ≈18.9 mAh cm−2 (0.5 mA cm−2) | 200 cycles (0.1 mA cm−2) | [67] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sui, D.; Chang, M.; Wang, H.; Qian, H.; Yang, Y.; Li, S.; Zhang, Y.; Song, Y. A Brief Review of Catalytic Cathode Materials for Na-CO2 Batteries. Catalysts 2021, 11, 603. https://doi.org/10.3390/catal11050603
Sui D, Chang M, Wang H, Qian H, Yang Y, Li S, Zhang Y, Song Y. A Brief Review of Catalytic Cathode Materials for Na-CO2 Batteries. Catalysts. 2021; 11(5):603. https://doi.org/10.3390/catal11050603
Chicago/Turabian StyleSui, Dong, Meijia Chang, Haiyu Wang, Hang Qian, Yanliang Yang, Shan Li, Yongsheng Zhang, and Yingze Song. 2021. "A Brief Review of Catalytic Cathode Materials for Na-CO2 Batteries" Catalysts 11, no. 5: 603. https://doi.org/10.3390/catal11050603
APA StyleSui, D., Chang, M., Wang, H., Qian, H., Yang, Y., Li, S., Zhang, Y., & Song, Y. (2021). A Brief Review of Catalytic Cathode Materials for Na-CO2 Batteries. Catalysts, 11(5), 603. https://doi.org/10.3390/catal11050603








