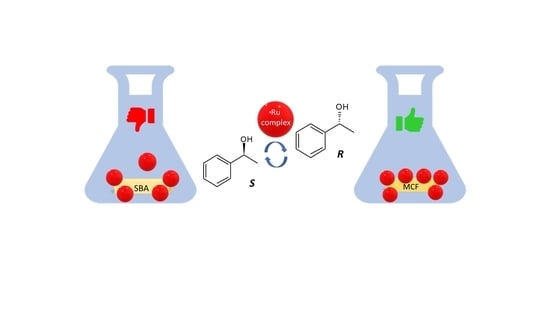
Engineering and Performance of Ruthenium Complexes Immobilized on Mesoporous Siliceous Materials as Racemization Catalysts
Abstract
1. Introduction
2. Results and Discussion
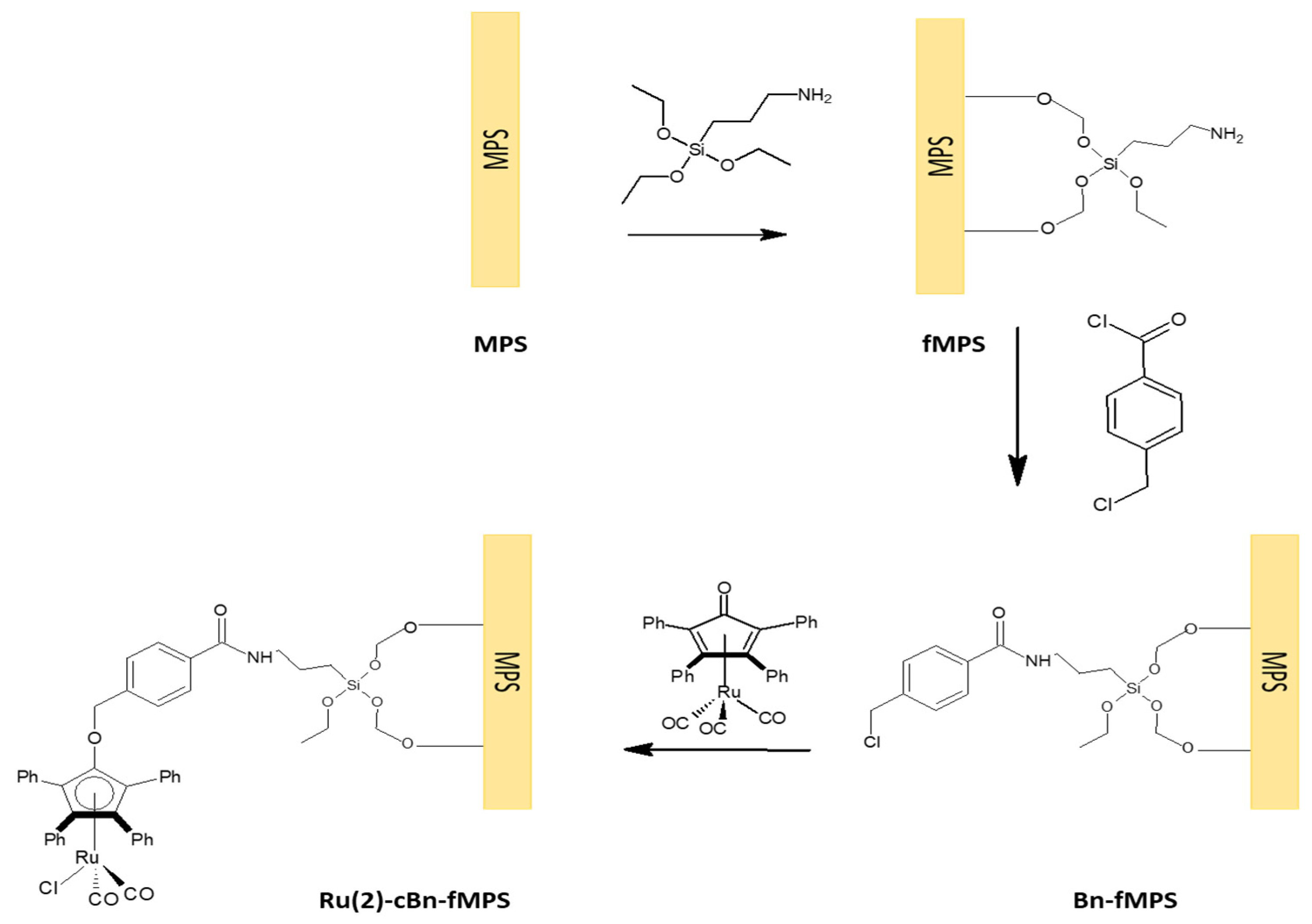
2.1. Immobilization of Ru complexes on MCFs and SBA-15
2.2. Characteristics of MCF, SBA, and the Immobilized Complexes
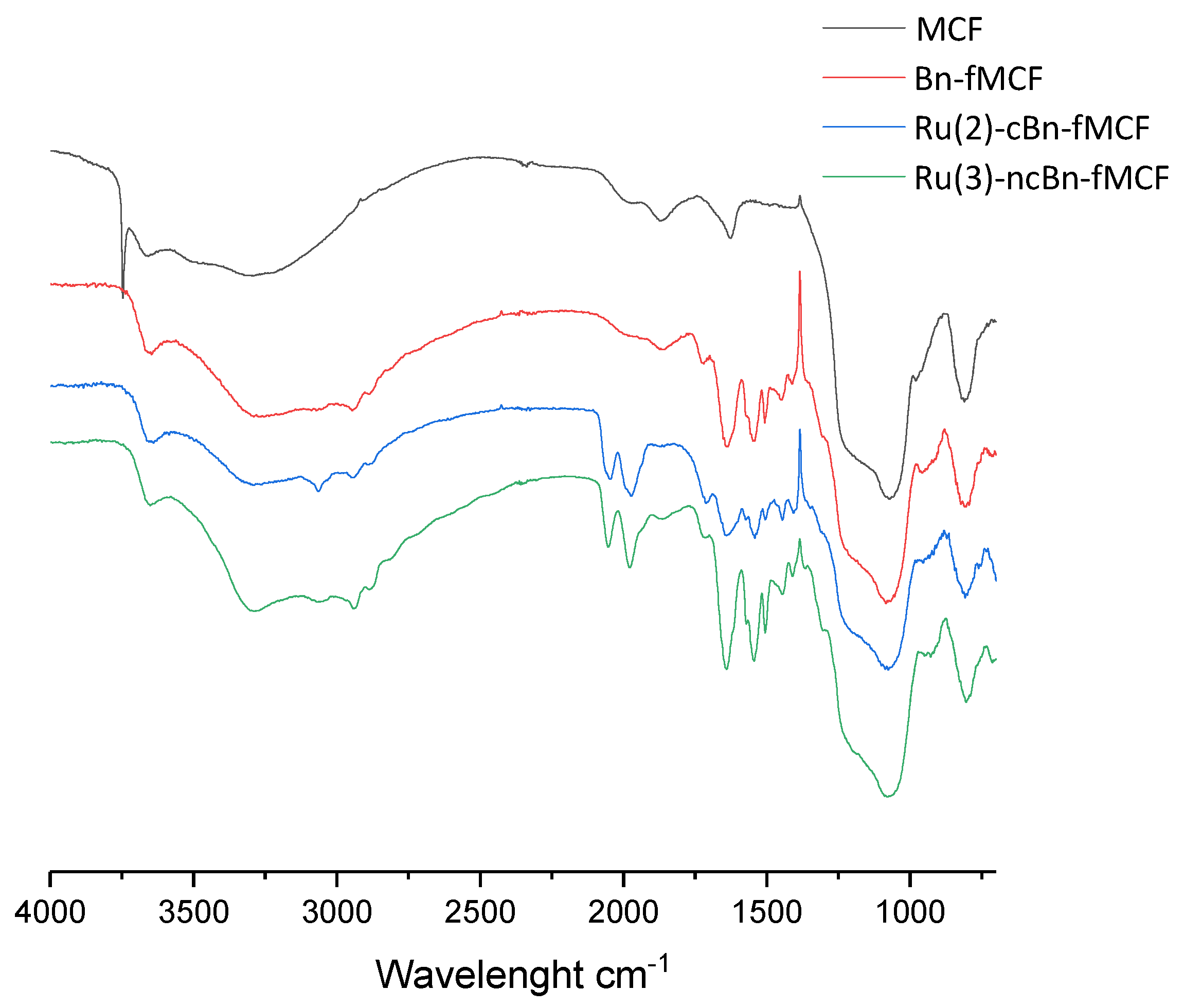
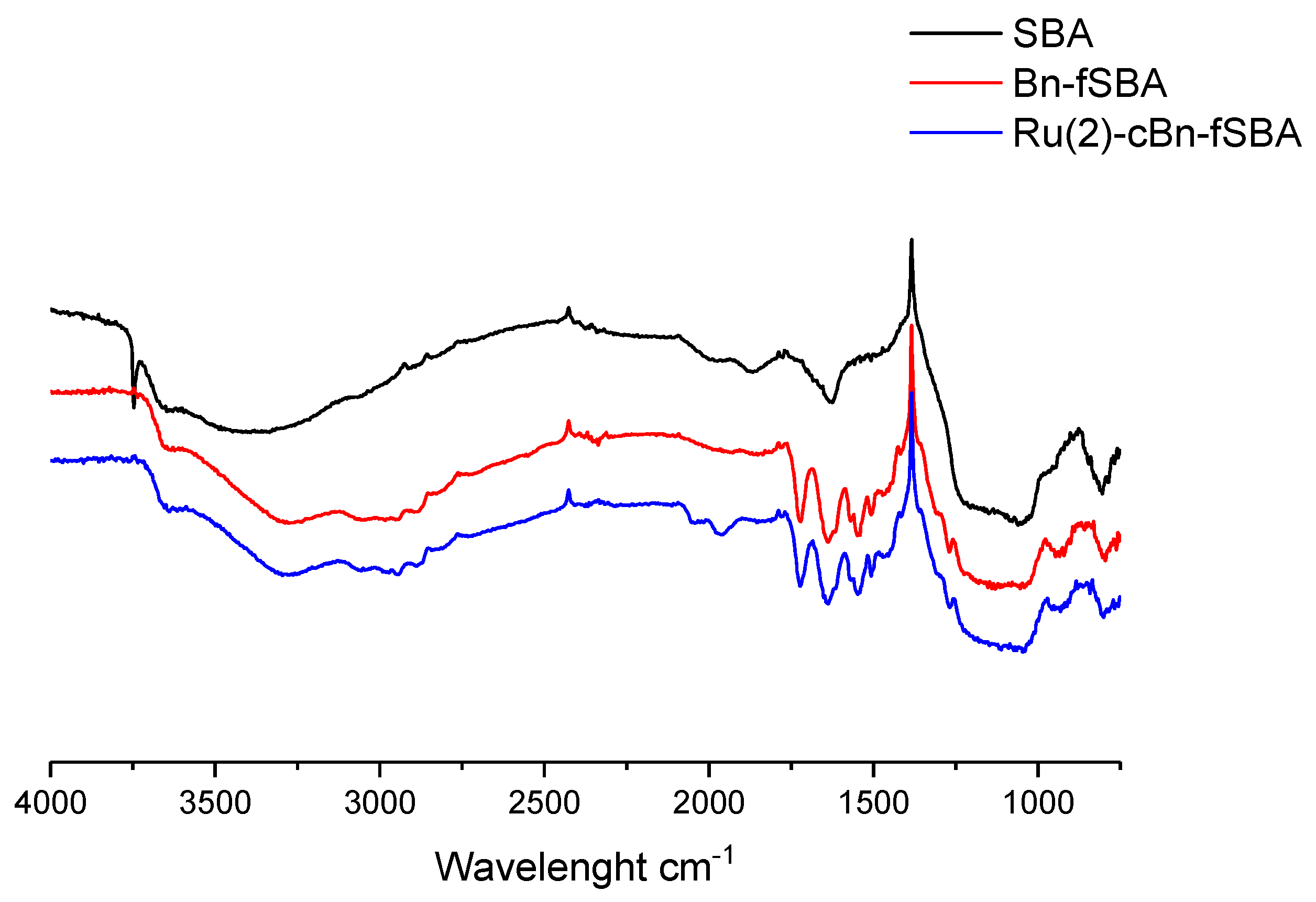
2.2.1. Fourier-Transform Infrared Spectroscopy Analysis
2.2.2. Atmospheric Solid Analysis Probe
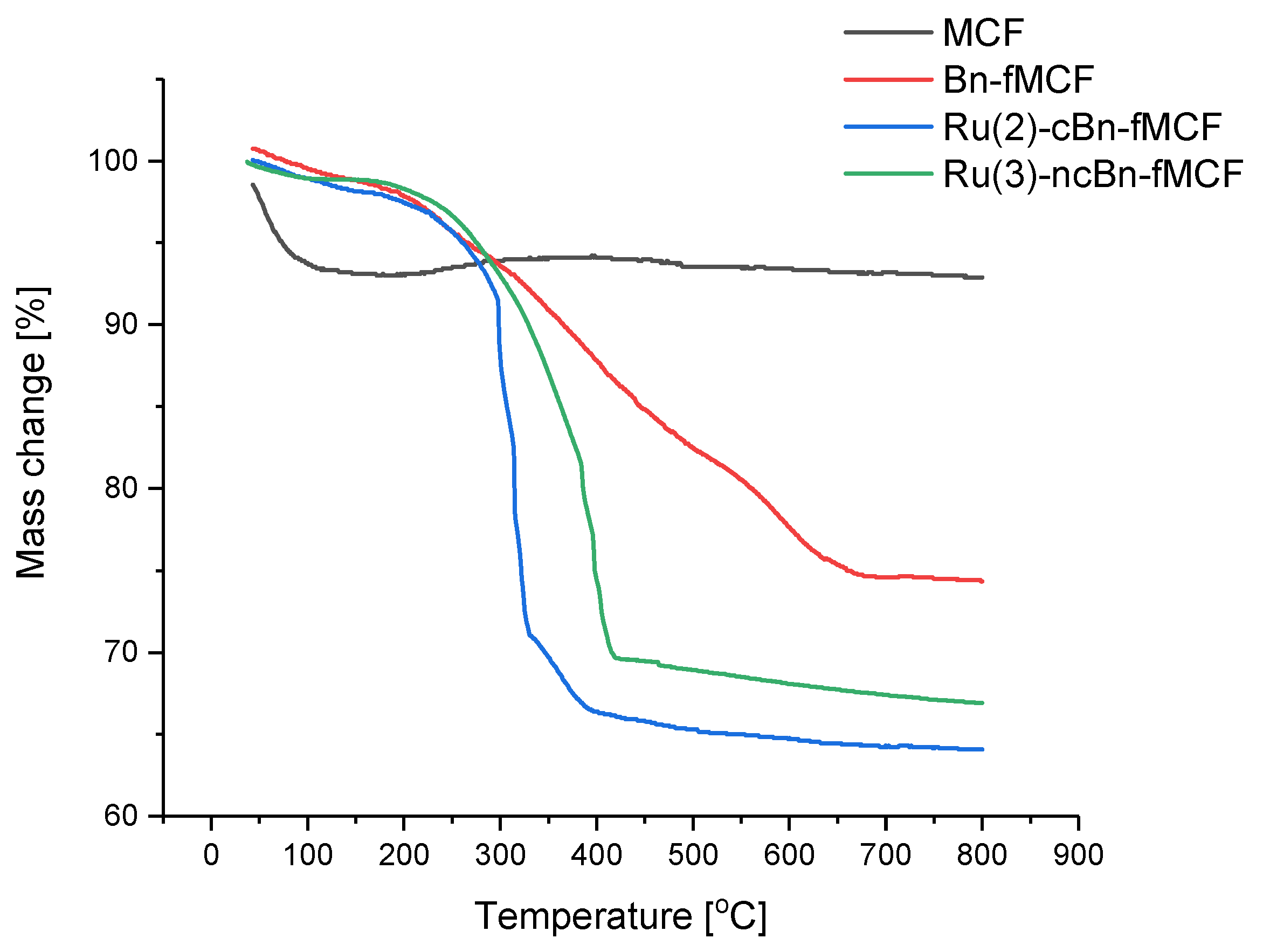
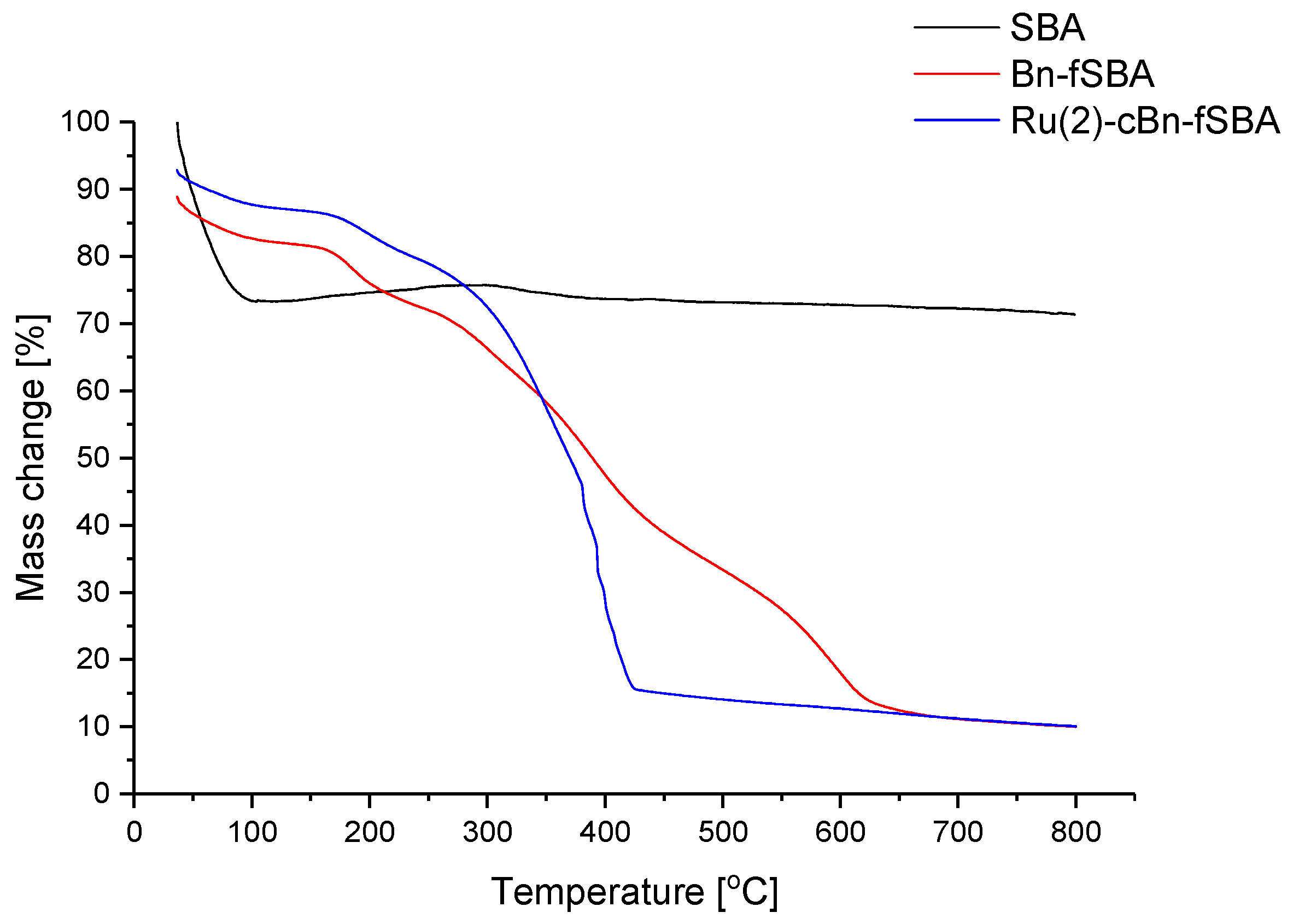
2.2.3. Thermogravimetry Analysis
2.2.4. ICP-OES Analysis
3. Experimental
3.1. General Procedure of MCF and SBA Functionalization
3.2. General Procedure of Immobilization Ru(2)-cBn-fMCF
3.3. General Procedure of Immobilization Ru(3)-ncBn-fMCF
3.4. General Procedure of Immobilization Ru(2)-cBn-fSBA
3.5. General Procedure of Synthesis Ru(1)
3.6. General Synthesis of Ru(2)
3.7. General Synthesis of Ru(3)
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bhardwaj, K.K.; Gupta, R. Synthesis of Chirally Pure Enantiomers by Lipase. J. Oleo Sci. 2017, 66, 1073–1084. [Google Scholar] [CrossRef] [PubMed]
- Pellissier, H. Recent developments in dynamic kinetic resolution. Tetrahedron 2008, 64, 1563–1601. [Google Scholar] [CrossRef]
- Wu, D.; Pan, F.; Tan, W.; Gao, L.; Tao, Y.; Kong, Y. Recent progress of enantioseparation under scale production (2014–2019). J. Sep. Sci. 2020, 43, 337–347. [Google Scholar] [CrossRef]
- Xiouras, C.; Fytopoulos, A.; Jordens, J.; Boudouvis, A.G.; Van Gerven, T.; Stefanidis, G.D. Applications of ultrasound to chiral crystallization, resolution and deracemization. Ultrason. Sonochem. 2018, 43, 184–192. [Google Scholar] [CrossRef]
- Ozkan, S.A.; Chankvetadze, B. Analytical and Preparative Scale Separation of Enantiomers of Chiral Drugs by Chromatography and Related Methods. Curr. Med. Chem. 2018, 25, 4152–4188. [Google Scholar] [CrossRef]
- Engström, K.; Johnston, E.V.; Verho, O.; Gustafson, K.P.J.; Shakeri, M.; Tai, C.-W.; Bäckvall, J.-E. Co-immobilization of an Enzyme and a Metal into the Compartments of Mesoporous Silica for Cooperative Tandem Catalysis: An Artificial Metalloenzyme. Angew. Chem. Int. Ed. 2013, 52, 14006–14010. [Google Scholar] [CrossRef] [PubMed]
- De Miranda, A.S.; Miranda, L.S.; De Souza, R.O. Lipases: Valuable catalysts for dynamic kinetic resolutions. Biotechnol. Adv. 2015, 33, 372–393. [Google Scholar] [CrossRef]
- Verho, O.; Bäckvall, J.-E. Chemoenzymatic Dynamic Kinetic Resolution: A Powerful Tool for the Preparation of Enantiomerically Pure Alcohols and Amines. J. Am. Chem. Soc. 2015, 137, 3996–4009. [Google Scholar] [CrossRef] [PubMed]
- Coldham, I.; Dufour, S.; Haxell, T.F.N.; Patel, J.J.; Sanchez-Jimenez, G. Dynamic Thermodynamic and Dynamic Kinetic Resolution of 2-Lithiopyrrolidines. J. Am. Chem. Soc. 2006, 128, 10943–10951. [Google Scholar] [CrossRef]
- Veum, L.; Hanefeld, U. Enantioselective formation of mandelonitrile acetate: Investigation of a dynamic kinetic resolution II. Tetrahedron Asymmetry 2004, 15, 3707–3709. [Google Scholar] [CrossRef]
- Huerta, F.F.; Minidis, A.B.E.; Bäckvall, J.E. Racemisation in asymmetric synthesis. Dynamic kinetic resolution and related processes in enzyme and metal catalysis. Chem. Soc. Rev. 2001, 30, 321–331. [Google Scholar] [CrossRef]
- Pàmies, O.; Bäckvall, J.-E. Combination of Enzymes and Metal Catalysts. A Powerful Approach in Asymmetric Catalysis. Chem. Rev. 2003, 103, 3247–3262. [Google Scholar] [CrossRef] [PubMed]
- Karvembu, R.; Prabhakaran, R.; Tamizh, M.M.; Natarajan, K. Ruthenium and enzyme-catalyzed dynamic kinetic resolution of alcohols. Comptes Rendus Chim. 2009, 12, 951–962. [Google Scholar] [CrossRef]
- Cao, H.; Zhu, X.-H.; Wang, N.; Sun, Z.; Deng, Y.; Hou, X.-F.; Zhao, D. Selectivity Enhancement in Dynamic Kinetic Resolution of Secondary Alcohols through Adjusting the Micro-Environment of Metal Complex Confined in Nanochannels: A Promising Strategy for Tandem Reactions. ACS Catal. 2014, 5, 27–33. [Google Scholar] [CrossRef]
- Magner, E. Immobilisation of enzymes on mesoporous silicate materials. Chem. Soc. Rev. 2013, 42, 6213–6222. [Google Scholar] [CrossRef] [PubMed]
- Jia, A.Q.; Shi, L.M.; Wu, F.L.; Xin, Z.F.; Zhang, Q.F. Syntheses, structures and immobilization of ruthenium complexes bearing N,O-Schiff-base or N,N′-diamine ligands functionalized with alkoxysilyl groups. J. Organomet. Chem. 2018, 855, 33–43. [Google Scholar] [CrossRef]
- Szymańska, K.; Bryjak, J.; Mrowiec-Białoń, J.; Jarzebski, A.B. Application and properties of siliceous mesostructured cellular foams as enzymes carriers to obtain efficient biocatalysts. Microporous Mesoporous Mater. 2007, 99, 167–175. [Google Scholar] [CrossRef]
- Rekuć, A.; Bryjak, J.; Szymańska, K.; Jarzębski, A.B. Laccase immobilization on mesostructured cellular foams affords preparations with ultra high activity. Process. Biochem. 2009, 44, 191–198. [Google Scholar] [CrossRef]
- Van Der Helm, M.P.; Bracco, P.; Busch, H.; Szymańska, K.; Jarzębski, A.B.; Hanefeld, U. Hydroxynitrile lyases covalently immobilized in continuous flow microreactors. Catal. Sci. Technol. 2019, 9, 1189–1200. [Google Scholar] [CrossRef]
- Choi, J.H.; Choi, Y.K.; Kim, Y.H.; Park, E.S.; Kim, E.J.; Kim, M.-J.; Park, J. Aminocyclopentadienyl Ruthenium Complexes as Racemization Catalysts for Dynamic Kinetic Resolution of Secondary Alcohols at Ambient Temperature. J. Org. Chem. 2004, 69, 1972–1977. [Google Scholar] [CrossRef] [PubMed]
- Kim, N.; Ko, S.-B.; Kwon, M.S.; Kim, M.-J.; Park, J. Air-Stable Racemization Catalyst for Dynamic Kinetic Resolution of Secondary Alcohols at Room Temperature. Org. Lett. 2005, 7, 4523–4526. [Google Scholar] [CrossRef]
- Martín-Matute, B.; Edin, M.; Bogár, K.; Bäckvall, J.-E. Highly Compatible Metal and Enzyme Catalysts for Efficient Dynamic Kinetic Resolution of Alcohols at Ambient Temperature. Angew. Chem. Int. Ed. 2004, 43, 6535–6539. [Google Scholar] [CrossRef] [PubMed]
- Martín-Matute, B.; Edin, M.; Bogár, K.; Kaynak, F.B.; Bäckvall, J.-E. Combined Ruthenium(II) and Lipase Catalysis for Efficient Dynamic Kinetic Resolution of Secondary Alcohols. Insight into the Racemization Mechanism. J. Am. Chem. Soc. 2005, 127, 8817–8825. [Google Scholar] [CrossRef] [PubMed]
- Crabtree, R.H. The Organometallic Chemistry of the Transition Metals, 2nd ed.; John Wiley & Sons: New York, NY, USA, 1994; p. 487. [Google Scholar]
- Shylesh, S.; Hanna, D.; Gomes, J.; Canlas, C.G.; Head-Gordon, M.; Bell, A.T. The Role of Hydroxyl Group Acidity on the Activity of Silica-Supported Secondary Amines for the Self-Condensation ofn-Butanal. ChemSusChem 2014, 8, 466–472. [Google Scholar] [CrossRef] [PubMed]
- Hu, L.; Ji, S.; Liu, Q.; Wu, P.; Zhu, J.; Li, C. Surface acidity and hydroxyl groups of SBA-15 mesoporous silica catalysts containing tungsten species. Chin. J. Catal. 2007, 11, 980–986. [Google Scholar] [CrossRef]
- Wouters, B.H.; Chen, T.; Dewilde, M.; Grobet, P.J. Reactivity of the surface hydroxyl groups of MCM-41 towards silylation with trimethylchlorosilane. Microporous Mesoporous Mater. 2001, 44–45, 453–457. [Google Scholar] [CrossRef]
- Mrowiec-Bialoń, J. Determination of hydroxyls density in the silica-mesostructured cellular foams by thermogravimetry. Thermochim. Acta 2006, 443, 49–52. [Google Scholar] [CrossRef]
- Gac, W.; Derylo-Marczewska, A.; Pasieczna-Patkowska, S.; Popivnyak, N.; Zukocinski, G. The influence of the preparation methods and pretreatment conditions on the properties of Ag-MCM-41 catalysts. J. Mol. Catal. A Chem. 2007, 268, 15–23. [Google Scholar] [CrossRef]
- Derylo-Marczewska, A.; Gac, W.; Popivnyak, N.; Zukocinski, G.; Pasieczna, S. The influence of preparation method on the structure and redox properties of mesoporous Mn-MCM-41 materials. Catal. Today 2006, 114, 293–306. [Google Scholar] [CrossRef]
- Zverev, S.A.; Andreev, S.; Zamilatskov, I.A.; Kurochkina, N.M.; Tyurin, V.S.; Senchikhin, I.N.; Ponomarev, G.V.; Erzina, D.R.; Chernyshev, V. Structure of ruthenium(II) complexes with coproporphyrin I tetraethyl ester. Russ. J. Phys. Chem. A 2017, 91, 1462–1467. [Google Scholar] [CrossRef]
- Mbese, J.Z.; Ajibade, P.A. Homonuclear tris-dithiocarbamato ruthenium(III) complexes as single-molecule precursors for the synthesis of ruthenium(III) sulfide nanoparticles. J. Sulfur Chem. 2016, 38, 173–187. [Google Scholar] [CrossRef]
- Zhao, D.; Huo, Q.; Feng, J.; Chmelka, B.F.; Stucky, G.D. Nonionic Triblock and Star Diblock Copolymer and Oligomeric Surfactant Syntheses of Highly Ordered, Hydrothermally Stable, Mesoporous Silica Structures. J. Am. Chem. Soc. 1998, 120, 6024–6036. [Google Scholar] [CrossRef]
- Haak, E. Ruthenium Complexes of Electronically Coupled Cyclopentadienone Ligands—Catalysts for Transformations of Propargyl Alcohols. Eur. J. Org. Chem. 2007, 2007, 2815–2824. [Google Scholar] [CrossRef]
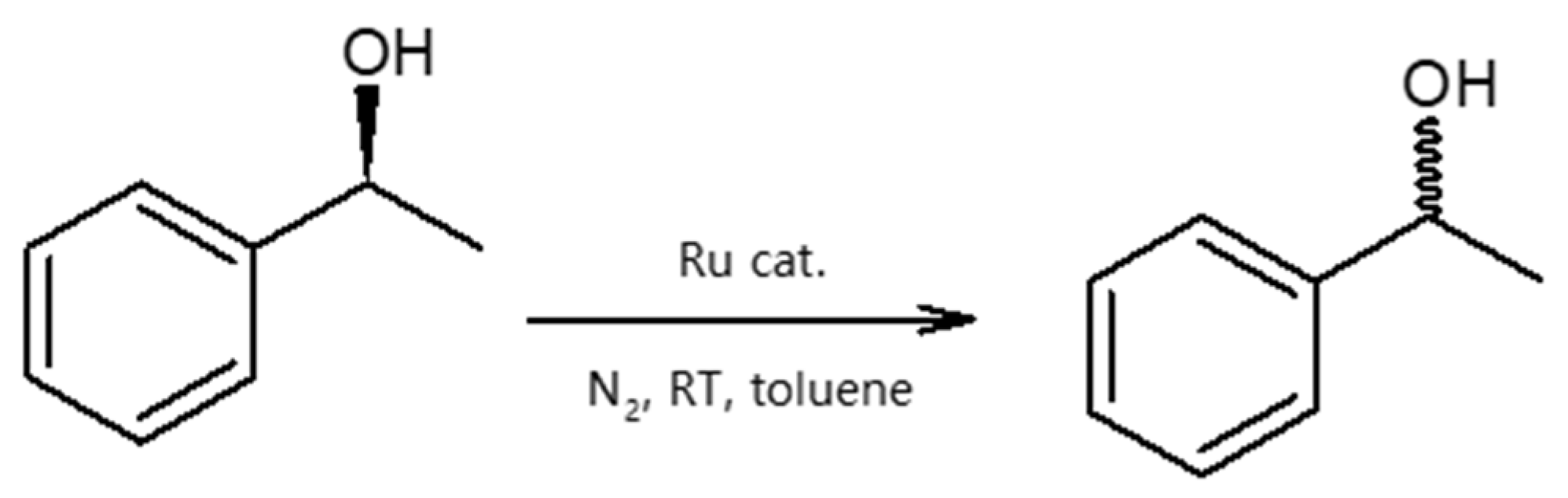

| Entry | Catalyst | Alcohol | ee [%] after 2 h |
|---|---|---|---|
| 1 | Ru(2) | 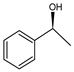 | <1 |
| 2 | Ru(2) |  | 16 |
| 3 | Ru(3) |  | <1 |
| 4 | Ru(3) | 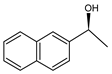 | <1 |
| Catalyst | Alcohol | ee [%] after 24 h |
|---|---|---|
| Ru(2)-cBn-fMCF | 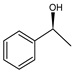 | <1 |
| Ru(2)-cBn-fMCF | 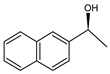 | 9 |
| Ru(3)-ncBn-fMCF | 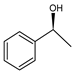 | 45 |
| Ru(3)-ncBn-fMCF |  | 69 |
| Catalyst | Number of Cycles | ee (%) after 48 h |
|---|---|---|
| Ru(2)-cBn-fMCF | 1 | <1 |
| 2 | <1 | |
| 3 | 60 |
| Catalyst | Alcohol | ee [%] after 24 h |
|---|---|---|
| SBA-125 | 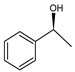 | 98 |
| SBA-125 | 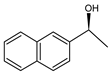 | 95 |
| Catalyst | Alcohol | Time [h] | ee [%] |
|---|---|---|---|
| Ru(2)-cBn-fSBA |  | 2 | >99 |
| 24 | 98 | ||
 | 2 | 97 | |
| 24 | 95 | ||
| Ru(2)-cBn-fMCF | 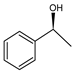 | 2 | 30 |
| 24 | <1 | ||
 | 2 | 27 | |
| 24 | 12 |
| SBET [m2/g] | Vp [cm3/g] | dp [nm] | dw [nm] | |
|---|---|---|---|---|
| MCF | 566 | 2.5 | 29 | 18 |
| Bn-fMCF | 258 | 1.3 | 27 | 18 |
| Ru(2)-cBn-fMCF | 220 | 0.99 | 25 | 15 |
| Ru(3)-ncBn-fMCF | 206 | 0.98 | 24 | 14 |
| SBA-15 | 527 | 0.65 | 7.6 | - |
| Bn-fSBA-15 | 19.9 | 0.28 | 4.49 | - |
| Ru(2)-cBn-fSBA-15 | 13.5 | 0.21 | 4.49 | - |
| Catalyst | Ruthenium Content on the Carrier [%] |
|---|---|
| Ru(2)-cBn-fMCF | 3.6 |
| Ru(3)-ncBn-fMCF | 0.7 |
| Ru(2)-cBn-fSBA | 1.7 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Heba, M.; Stradomska, D.; Szymańska, K.; Jarzębski, A.; Ambroziak, K.; Masternak, M.; Kolanowska, A.; Pudło, W.; Kuźnik, N. Engineering and Performance of Ruthenium Complexes Immobilized on Mesoporous Siliceous Materials as Racemization Catalysts. Catalysts 2021, 11, 316. https://doi.org/10.3390/catal11030316
Heba M, Stradomska D, Szymańska K, Jarzębski A, Ambroziak K, Masternak M, Kolanowska A, Pudło W, Kuźnik N. Engineering and Performance of Ruthenium Complexes Immobilized on Mesoporous Siliceous Materials as Racemization Catalysts. Catalysts. 2021; 11(3):316. https://doi.org/10.3390/catal11030316
Chicago/Turabian StyleHeba, Monika, Dominika Stradomska, Katarzyna Szymańska, Andrzej Jarzębski, Krzysztof Ambroziak, Monika Masternak, Anna Kolanowska, Wojciech Pudło, and Nikodem Kuźnik. 2021. "Engineering and Performance of Ruthenium Complexes Immobilized on Mesoporous Siliceous Materials as Racemization Catalysts" Catalysts 11, no. 3: 316. https://doi.org/10.3390/catal11030316
APA StyleHeba, M., Stradomska, D., Szymańska, K., Jarzębski, A., Ambroziak, K., Masternak, M., Kolanowska, A., Pudło, W., & Kuźnik, N. (2021). Engineering and Performance of Ruthenium Complexes Immobilized on Mesoporous Siliceous Materials as Racemization Catalysts. Catalysts, 11(3), 316. https://doi.org/10.3390/catal11030316