Comprehensive Comparison of Hetero-Homogeneous Catalysts for Fatty Acid Methyl Ester Production from Non-Edible Jatropha curcas Oil
Abstract
:1. Introduction
2. Results and Discussion
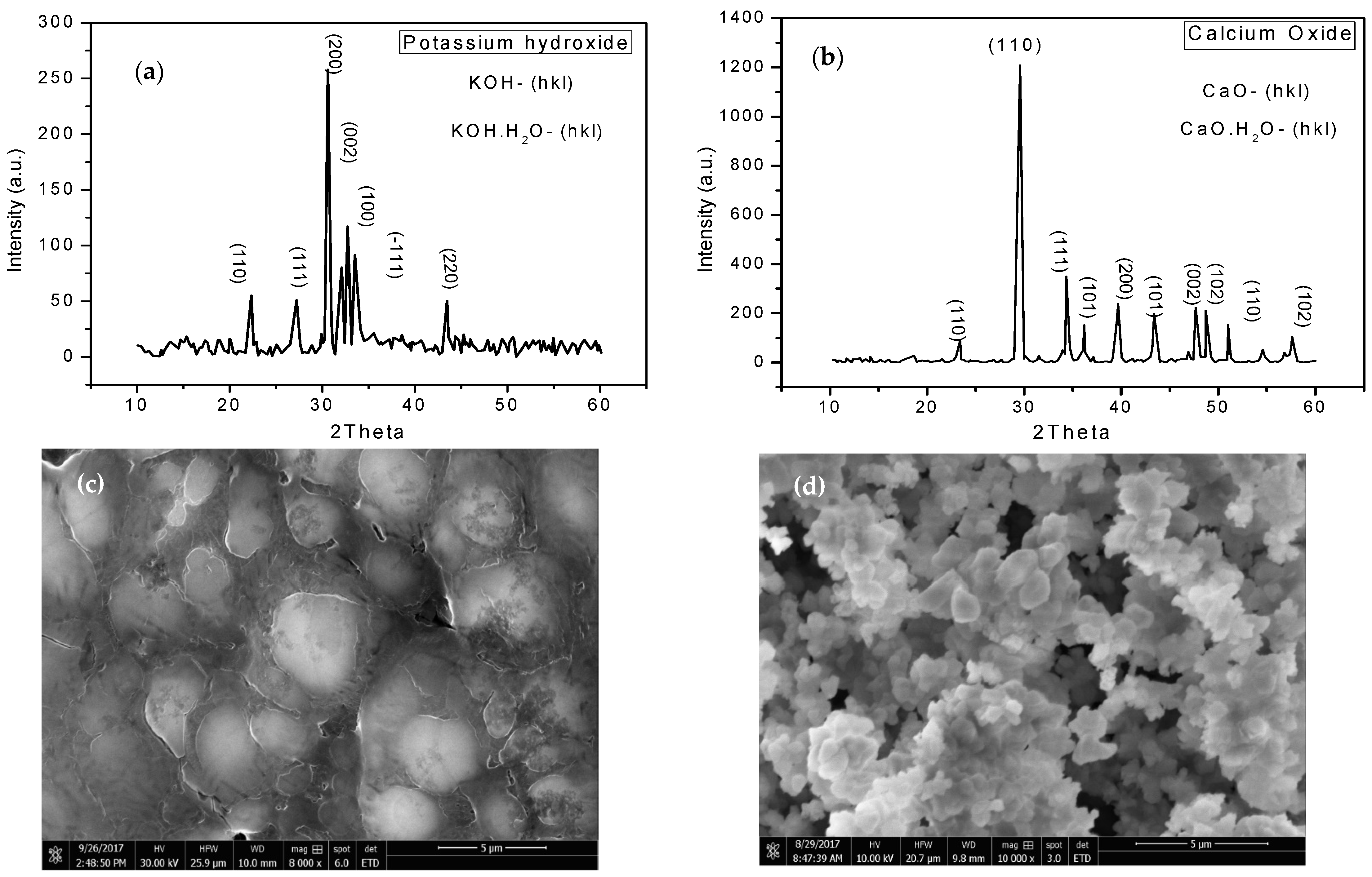
2.1. Catalyst Characterization
2.2. FTIR Analysis of Biodiesel
2.3. Yield of Biodiesel
2.4. Effect of Reaction Paramenters on Biodiesel Yield
2.4.1. Effect of Methanol-to-Oil Molar Ratio
2.4.2. Effect of Catalyst Load
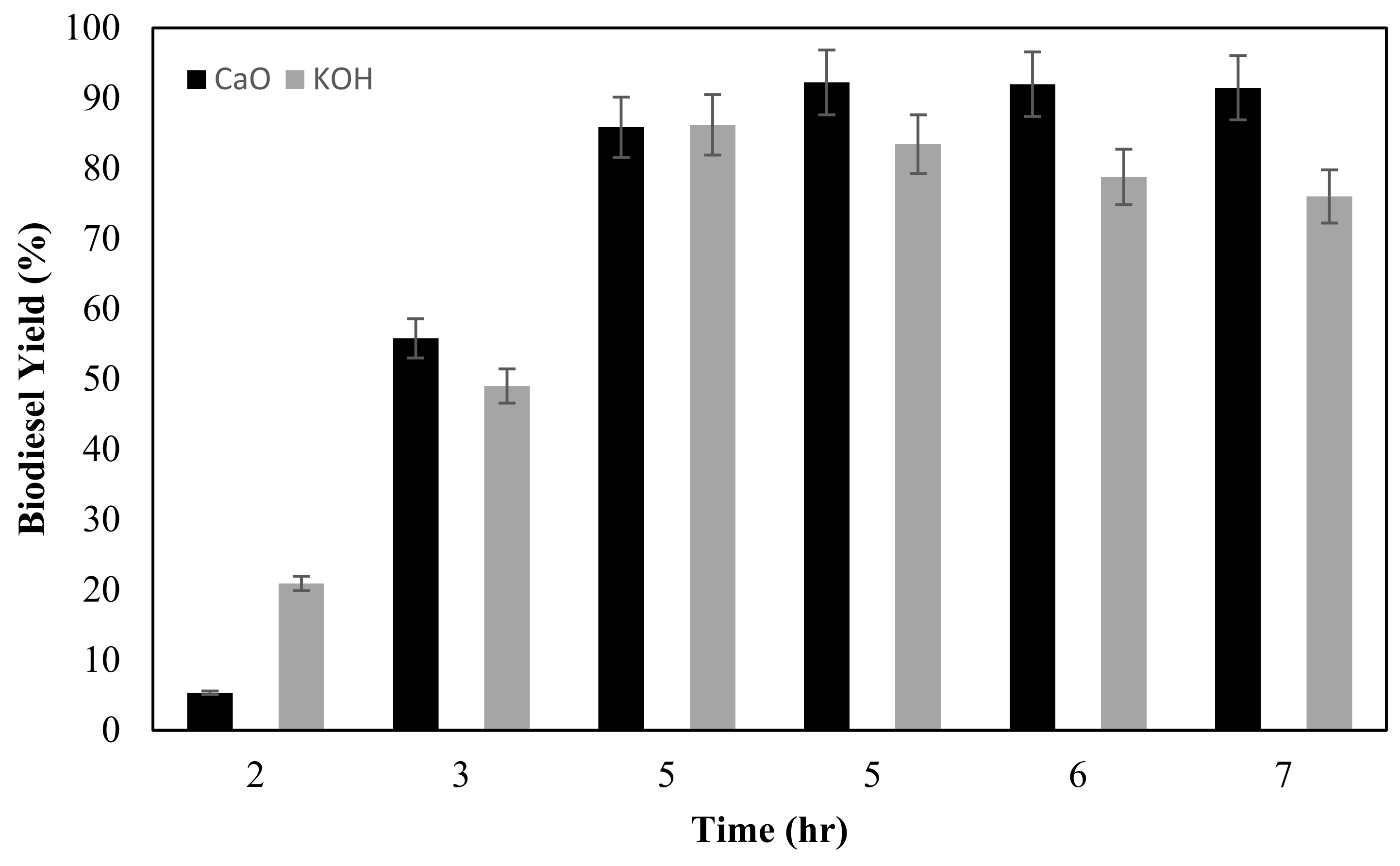
2.4.3. Effect of Reaction Time
2.5. Biodiesel Fuel Properties
3. Materials and Methods
3.1. Materials
3.2. Preparation of Catalysts
3.3. Characterization of Catalysts and Biodiesel
3.4. Pretreatment and Transesterification of Jatropha Oil
3.5. Fuel Properties and Yield of Jatropha Oil-Based Biodiesel
3.6. GC-MS Analysis of Biodiesel
3.7. Blending Biodiesel with Petrodiesel
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Disclaimer
References
- Agarwal, D.; Agarwal, A.K. Performance and emissions characteristics of Jatropha oil (preheated and blends) in a direct injection compression ignition engine. Appl. Therm. Eng. 2007, 27, 2314–2323. [Google Scholar] [CrossRef]
- Jain, S.; Sharma, M. Prospects of biodiesel from Jatropha in India: A review. Renew. Sustain. Energy Rev. 2010, 14, 763–771. [Google Scholar] [CrossRef]
- Jayed, M.; Masjuki, H.H.; Saidur, R.; Kalam, M.A.; Jahirul, M.I. Environmental aspects and challenges of oilseed produced biodiesel in Southeast Asia. Renew. Sustain. Energy Rev. 2009, 13, 2452–2462. [Google Scholar] [CrossRef]
- Robles-Medina, A.; González-Moreno, P.A.; Esteban-Cerdán, L.; Molina-Grima, E. Biocatalysis: Towards ever greener biodiesel production. Biotechnol. Adv. 2009, 27, 398–408. [Google Scholar] [CrossRef] [PubMed]
- El Diwani, G.; Attia, N.; Hawash, S. Development and evaluation of biodiesel fuel and by-products from jatropha oil. Int. J. Environ. Sci. Technol. 2009, 6, 219–224. [Google Scholar] [CrossRef] [Green Version]
- Kulkarni, M.G.; Dalai, A.K. Waste cooking oil an economical source for biodiesel: A review. Ind. Eng. Chem. Res. 2006, 45, 2901–2913. [Google Scholar] [CrossRef]
- Pahl, G. Biodiesel: Growing a New Energy Economy; Chelsea Green Publishing: Hartford, VT, USA, 2008. [Google Scholar]
- Su, E.; Wei, D. Improvement in lipase-catalyzed methanolysis of triacylglycerols for biodiesel production using a solvent engineering method. J. Mol. Catal. B Enzym. 2008, 55, 118–125. [Google Scholar] [CrossRef]
- Nie, K.; Xie, F.; Wang, F.; Tan, T. Lipase catalyzed methanolysis to produce biodiesel: Optimization of the biodiesel production. J. Mol. Catal. B Enzym. 2006, 43, 142–147. [Google Scholar] [CrossRef]
- Shah, S.; Sharma, S.; Gupta, M. Biodiesel preparation by lipase-catalyzed transesterification of Jatropha oil. Energy Fuels 2004, 18, 154–159. [Google Scholar] [CrossRef]
- Vieira, A.d.A.; Silva, M.D.; Langone, M. Biodiesel production via esterification reactions catalyzed by lipase. Lat. Am. Appl. Res. 2006, 36, 283–288. [Google Scholar]
- Hanif, M.A.; Nisar, S.; Rashid, U. Supported solid and heteropoly acid catalysts for production of biodiesel. Catal. Rev. Sci. Eng. 2017, 59, 165–188. [Google Scholar] [CrossRef]
- Helwani, Z.; Othman, M.R.; Aziz, N.; Kim, J.; Fernando, W.J.N. Solid heterogeneous catalysts for transesterification of triglycerides with methanol: A review. Appl. Catal. A Gen. 2009, 363, 1–10. [Google Scholar] [CrossRef]
- Leung, D.Y.; Wu, X.; Leung, M. A review on biodiesel production using catalyzed transesterification. Appl. Energy 2010, 87, 1083–1095. [Google Scholar] [CrossRef]
- Parawira, W. Biotechnological production of biodiesel fuel using biocatalysed transesterification: A review. Crit. Rev. Biotechnol. 2009, 29, 82–93. [Google Scholar] [CrossRef]
- Pinzi, S.; Garcia, I.L.; Lopez-Gimenez, F.J.; Luque de Castro, M.D.; Dorado, G.; Dorado, M.P. The ideal vegetable oil-based biodiesel composition: A review of social, economical and technical implications. Energy Fuels 2009, 23, 2325–2341. [Google Scholar] [CrossRef]
- Endalew, A.K.; Kiros, Y.; Zanzi, R. Heterogeneous catalysis for biodiesel production from Jatropha curcas oil (JCO). Energy 2011, 36, 2693–2700. [Google Scholar] [CrossRef]
- Elkady, M.F.; Zaatout, A.; Balbaa, O. Production of biodiesel from waste vegetable oil via KM micromixer. J. Chem. 2015, 2015, 630168. [Google Scholar] [CrossRef]
- Tyagi, O.S.; Atray, N.; Kumar, B.; Datta, A. Production, characterization and development of standards for biodiesel—A review. MAPAN 2010, 25, 197–218. [Google Scholar] [CrossRef]
- Mathiyazhagan, M.; Ganapathi, A. Factors affecting biodiesel production. Res. Plant Biol. 2011, 1, 1–5. [Google Scholar]
- Cao, F.; Chen, Y.; Zhai, F.; Li, J.; Wang, J.; Wang, X.; Wang, S.; Zhu, W. Biodiesel production from high acid value waste frying oil catalyzed by superacid heteropoly acid. Biotechnol. Bioeng. 2008, 101, 93–100. [Google Scholar] [CrossRef]
- Kartika, I.A.; Yani, M.; Ariono, D.; Evon, P.; Rigal, L. Biodiesel production from jatropha seeds: Solvent extraction and in situ transesterification in a single step. Fuel 2013, 106, 111–117. [Google Scholar] [CrossRef] [Green Version]
- Ngamcharussrivichai, C.; Wiwatnimit, W.; Wangnoi, S. Modified dolomites as catalysts for palm kernel oil transesterification. J. Mol. Catal. A Chem. 2007, 276, 24–33. [Google Scholar] [CrossRef]
- Hawash, S.; El Diwani, G.; Abdel Kader, E. Optimization of biodiesel production from jatropha oil by heterogeneous base catalysed transesterification. Int. J. Eng. Sci. Technol. 2011, 3, 5242–5251. [Google Scholar]
- Chen, G.-Y.; Shan, R.; Shi, J.-F.; Yan, B.-B. Transesterification of palm oil to biodiesel using rice husk ash-based catalysts. Fuel Process. Technol. 2015, 133, 8–13. [Google Scholar] [CrossRef]
- Sakai, T.; Kawashima, A.; Koshikawa, T. Economic assessment of batch biodiesel production processes using homogeneous and heterogeneous alkali catalysts. Bioresour. Technol. 2009, 100, 3268–3276. [Google Scholar] [CrossRef]
- Gangadhara, R.; Prasad, N. Studies on optimization of transesterification of certain oils to produce biodiesel. Chem. Int. 2016, 2, 59. [Google Scholar]
- Mumtaz, M.W.; Mukhtar, H.; Dilawer, U.A.; Hussain, S.M.; Hussain, M.; Iqbal, M.; Adnan, A.; Nisar, J. Biocatalytic transesterification of Eruca sativa oil for the production of biodiesel. Biocatal. Agric. Biotechnol. 2016, 5, 162–167. [Google Scholar] [CrossRef]
- Arias, E.L.M.; Martins, P.F.; Munhoz, A.L.J.; Gutierrez-Rivera, L.; Filho, R.M. Continuous synthesis and in situ monitoring of biodiesel production in different microfluidic devices. Ind. Eng. Chem. Res. 2012, 51, 10755–10767. [Google Scholar] [CrossRef]
- Hazmi, B.; Rashid, U.; Taufiq-Yap, Y.H.; Ibrahim, M.L.; Nehdi, I.A. Supermagnetic nano-bifunctional catalyst from rice husk: Synthesis, characterization and application for conversion of used cooking oil to biodiesel. Catalysts 2020, 10, 225. [Google Scholar] [CrossRef] [Green Version]


| Peak | Possible Functional Group | Type of Vibration | Absorption Range (cm−1) | BD-C | BD-K |
|---|---|---|---|---|---|
| 1 | –CH2– | Stretching | 2850–2950 | 2922 | 2921 |
| 2 | –CH2– | Stretching | 2850–2950 | 2853 | 2852 |
| 3 | C=O | Stretching | 1730–1750 | 1742 | 1742 |
| 4 | –CH3 | Stretching | 1350–1475 | 1462 | 1462 |
| 5 | C–O | Stretching | 1000–1300 | 1163 | 1162 |
| Biodiesel | Property | ASTM Method | B5 | B100 | ASTM D6751 | ASTM D975 |
|---|---|---|---|---|---|---|
| FP (°C) | D93 | 84 | 288 | 93 minimum | 52 minumum | |
| CP (°C) | D2500 | 1 | 7 | Not specified | Not specified | |
| BD-K | KV (40 °C; mm2/s) | D445 | 2.05 | 4.40 | 1.9–6.0 | 1.9–4.1 |
| CI | D976 | 53.7 | 47.3 | 47 minimum | 40 minimum | |
| SG (15.6 °C) | D1298 | 0.851 | 0.896 | Not specified | Not specified | |
| FP (°C) | D93 | 83 | 293 | 93 minimum | 52 minumum | |
| CP (°C) | D2500 | 4 | 9 | Not specified | Not specified | |
| BD-C | KV (40 °C; mm2/s) | D445 | 2.12 | 4.50 | 1.9–6.0 | 1.9–4.1 |
| CI | D976 | 53.9 | 49.7 | 47 minimum | 40 minimum | |
| SG (15.6 °C) | D1298 | 0.853 | 0.899 | Not specified | Not specified |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Khan, K.; Ul-Haq, N.; Rahman, W.U.; Ali, M.; Rashid, U.; Ul-Haq, A.; Jamil, F.; Ahmed, A.; Ahmed, F.; Moser, B.R.; et al. Comprehensive Comparison of Hetero-Homogeneous Catalysts for Fatty Acid Methyl Ester Production from Non-Edible Jatropha curcas Oil. Catalysts 2021, 11, 1420. https://doi.org/10.3390/catal11121420
Khan K, Ul-Haq N, Rahman WU, Ali M, Rashid U, Ul-Haq A, Jamil F, Ahmed A, Ahmed F, Moser BR, et al. Comprehensive Comparison of Hetero-Homogeneous Catalysts for Fatty Acid Methyl Ester Production from Non-Edible Jatropha curcas Oil. Catalysts. 2021; 11(12):1420. https://doi.org/10.3390/catal11121420
Chicago/Turabian StyleKhan, Khawer, Noaman Ul-Haq, Wajeeh Ur Rahman, Muzaffar Ali, Umer Rashid, Anwar Ul-Haq, Farrukh Jamil, Ashfaq Ahmed, Faisal Ahmed, Bryan R. Moser, and et al. 2021. "Comprehensive Comparison of Hetero-Homogeneous Catalysts for Fatty Acid Methyl Ester Production from Non-Edible Jatropha curcas Oil" Catalysts 11, no. 12: 1420. https://doi.org/10.3390/catal11121420
APA StyleKhan, K., Ul-Haq, N., Rahman, W. U., Ali, M., Rashid, U., Ul-Haq, A., Jamil, F., Ahmed, A., Ahmed, F., Moser, B. R., & Alsalme, A. (2021). Comprehensive Comparison of Hetero-Homogeneous Catalysts for Fatty Acid Methyl Ester Production from Non-Edible Jatropha curcas Oil. Catalysts, 11(12), 1420. https://doi.org/10.3390/catal11121420







