Methanation of CO2 Using MIL-53-Based Catalysts: Ni/MIL-53–Al2O3 versus Ni/MIL-53
Abstract
:1. Introduction
2. Results and Discussion
2.1. Structural Characteristics of Catalysts
2.2. Catalytic Activity Tests
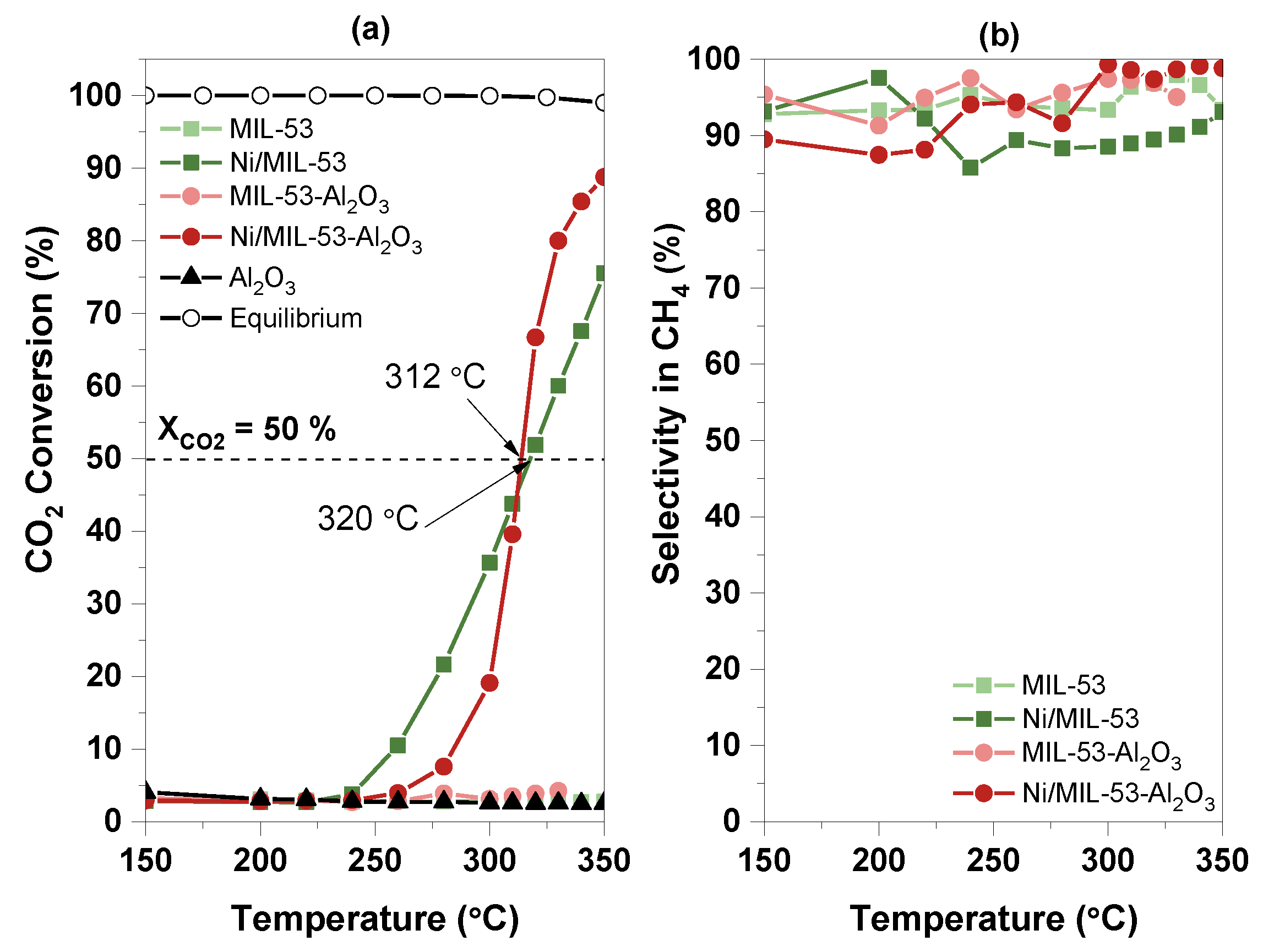
2.2.1. Temperature Influence
2.2.2. Stability Tests
3. Materials and Methods
3.1. Chemicals and Gases
3.2. MIL-53 and MIL-53–Al2O3 Synthesis
3.3. Preparation of Ni/MIL-53 and Ni/MIL-53–Al2O3 Catalysts
3.4. Characterization Techniques
3.5. Catalytic Measurements
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Vogt, C.; Groeneveld, E.; Kamsma, G.; Nachtegaal, M.; Lu, L.; Kiely, C.; Berben, P.H.; Meirer, F.; Weckhuysen, B.M. Unravelling structure sensitivity in CO2 hydrogenation over nickel. Nat. Catal. 2018, 1, 127–134. [Google Scholar] [CrossRef]
- Aresta, M.; Dibenedetto, A.; Angelini, A. Catalysis for the Valorization of Exhaust Carbon: From CO2 to Chemicals, Materials, and Fuels. Technological Use of CO2. Chem. Rev. 2013, 114, 1709–1742. [Google Scholar] [CrossRef] [PubMed]
- Ghaib, K.; Ben-Fares, F.-Z. Power-to-Methane: A state-of-the-art review. Renew. Sustain. Energy Rev. 2018, 81, 433–446. [Google Scholar] [CrossRef]
- Meylan, F.D.; Moreau, V.; Erkman, S. CO2 utilization in the perspective of industrial ecology, an overview. J. CO2 Util. 2015, 12, 101–108. [Google Scholar] [CrossRef]
- Najafabadi, A.T. CO2 chemical conversion to useful products: An engineering insight to the latest advances toward sustainability. Int. J. Energy Res. 2013, 37, 485–499. [Google Scholar] [CrossRef]
- Bahari, N.A.; Isahak, W.N.R.W.; Masdar, M.S.; Yaakob, Z. Clean hydrogen generation and storage strategies via CO2 utilization into chemicals and fuels: A review. Int. J. Energy Res. 2019, 43, 5128–5150. [Google Scholar] [CrossRef]
- Murena, F.; Esposito, S.; Deorsola, F.; Galletti, C.; Prati, M. CO2 abatement and CH4 recovery at vehicle exhausts: Comparison and characterization of Ru powder and pellet catalysts. Int. J. Hydrogen Energy 2020, 45, 8640–8648. [Google Scholar] [CrossRef]
- Fechete, I.; Vedrine, J.C. Nanoporous Materials as New Engineered Catalysts for the Synthesis of Green Fuels. Molecules 2015, 20, 5638–5666. [Google Scholar] [CrossRef] [Green Version]
- Lee, W.J.; Li, C.; Prajitno, H.; Yoo, J.; Patel, J.; Yang, Y.; Lim, S. Recent trend in thermal catalytic low temperature CO2 methanation: A critical review. Catal. Today 2020, 368, 2–19. [Google Scholar] [CrossRef]
- Frontera, P.; Macario, A.; Ferraro, M.; Antonucci, P. Supported Catalysts for CO2 Methanation: A Review. Catalysts 2017, 7, 59. [Google Scholar] [CrossRef]
- Kim, A.; Sanchez, C.; Haye, B.; Boissière, C.; Sassoye, C.; Debecker, D.P. Mesoporous TiO2 Support Materials for Ru-Based CO2 Methanation Catalysts. ACS Appl. Nano Mater. 2019, 2, 3220–3230. [Google Scholar] [CrossRef]
- Karam, L.; Bacariza, M.C.; Lopes, J.M.; Henriques, C.; Reboul, J.; El Hassan, N.; Massiani, P. Mesoporous nickel-alumina catalysts derived from MIL-53(Al) metal-organic framework: A new promising path for synthesizing CO2 methanation catalysts. J. CO2 Util. 2021, 51, 101651. [Google Scholar] [CrossRef]
- Liang, C.; Hu, X.; Wei, T.; Jia, P.; Zhang, Z.; Dong, D.; Zhang, S.; Liu, Q.; Hu, G. Methanation of CO2 over Ni/Al2O3 modified with alkaline earth metals: Impacts of oxygen vacancies on catalytic activity. Int. J. Hydrogen Energy 2019, 44, 8197–8213. [Google Scholar] [CrossRef]
- Renda, S.; Ricca, A.; Palma, V. Study of the effect of noble metal promotion in Ni-based catalyst for the Sabatier reaction. Int. J. Hydrogen Energy 2020, 46, 12117–12127. [Google Scholar] [CrossRef]
- Mihet, M.; Lazar, M.D. Methanation of CO2 on Ni/γ-Al2O3: Influence of Pt, Pd or Rh promotion. Catal. Today 2018, 306, 294–299. [Google Scholar] [CrossRef]
- Mutz, B.; Carvalho, H.W.; Mangold, S.; Kleist, W.; Grunwaldt, J.-D. Methanation of CO2: Structural response of a Ni-based catalyst under fluctuating reaction conditions unraveled by operando spectroscopy. J. Catal. 2015, 327, 48–53. [Google Scholar] [CrossRef]
- Kim, H.Y.; Lee, H.M.; Park, J.-N. Bifunctional Mechanism of CO2 Methanation on Pd-MgO/SiO2 Catalyst: Independent Roles of MgO and Pd on CO2 Methanation. J. Phys. Chem. C 2010, 114, 7128–7131. [Google Scholar] [CrossRef]
- Duyar, M.; Treviño, M.A.; Farrauto, R.J. Dual function materials for CO2 capture and conversion using renewable H2. Appl. Catal. B Environ. 2015, 168-169, 370–376. [Google Scholar] [CrossRef]
- Mihet, M.; Dan, M.; Barbu-Tudoran, L.; Lazar, M.D. CO2 Methanation Using Multimodal Ni/SiO2 Catalysts: Effect of Support Modification by MgO, CeO2, and La2O3. Catalysts 2021, 11, 443. [Google Scholar] [CrossRef]
- Moon, H.R.; Lim, D.-W.; Suh, M.P. Fabrication of metal nanoparticles in metal–organic frameworks. Chem. Soc. Rev. 2012, 42, 1807–1824. [Google Scholar] [CrossRef]
- Cui, W.-G.; Zhang, G.-Y.; Hu, T.-L.; Bu, X.-H. Metal-organic framework-based heterogeneous catalysts for the conversion of C1 chemistry: CO, CO2 and CH4. Coord. Chem. Rev. 2019, 387, 79–120. [Google Scholar] [CrossRef]
- Zhen, W.; Gao, F.; Tian, B.; Ding, P.; Deng, Y.; Li, Z.; Gao, H.; Lu, G. Enhancing activity for carbon dioxide methanation by encapsulating (1 1 1) facet Ni particle in metal–organic frameworks at low temperature. J. Catal. 2017, 348, 200–211. [Google Scholar] [CrossRef]
- Wang, H.; Zhu, Q.-L.; Zou, R.; Xu, Q. Metal-Organic Frameworks for Energy Applications. Chem 2017, 2, 52–80. [Google Scholar] [CrossRef] [Green Version]
- Chen, Y.-Z.; Zhang, R.; Jiao, L.; Jiang, H.-L. Metal–organic framework-derived porous materials for catalysis. Coord. Chem. Rev. 2018, 362, 1–23. [Google Scholar] [CrossRef]
- Gascon, J.; Corma, A.; Kapteijn, F.; Llabres i Xamena, F.X. Metal Organic Framework Catalysis: Quo vadis? ACS Catal. 2014, 4, 361–378. [Google Scholar] [CrossRef]
- Falcaro, P.; Ricco, R.; Yazdi, A.; Imaz, I.; Furukawa, S.; Maspoch, D.; Ameloot, R.; Evans, J.D.; Doonan, C.J. Application of metal and metal oxide nanoparticles@MOFs. Coord. Chem. Rev. 2016, 307, 237–254. [Google Scholar] [CrossRef]
- Zhen, W.; Li, B.; Lu, G.; Ma, J. Enhancing catalytic activity and stability for CO2 methanation on Ni@MOF-5 via control of active species dispersion. Chem. Commun. 2014, 51, 1728–1731. [Google Scholar] [CrossRef]
- Mihet, M.; Grad, O.; Blanita, G.; Radu, T.; Lazar, M.D. Effective encapsulation of Ni nanoparticles in metal-organic frameworks and their application for CO2 methanation. Int. J. Hydrogen Energy 2019, 44, 13383–13396. [Google Scholar] [CrossRef]
- Zhao, Z.-W.; Zhou, X.; Liu, Y.-N.; Shen, C.-C.; Yuan, C.-Z.; Jiang, Y.-F.; Zhao, S.-J.; Ma, L.-B.; Cheang, T.-Y.; Xu, A.-W. Ultrasmall Ni nanoparticles embedded in Zr-based MOFs provide high selectivity for CO2 hydrogenation to methane at low temperatures. Catal. Sci. Technol. 2018, 8, 3160–3165. [Google Scholar] [CrossRef]
- Mihet, M.; Blanita, G.; Dan, M.; Barbu-Tudoran, L.; Lazar, M.D. Pt/UiO-66 Nanocomposites as Catalysts for CO2 Methanation Process. J. Nanosci. Nanotechnol. 2019, 19, 3187–3196. [Google Scholar] [CrossRef]
- Xie, Z.; Xu, W.; Cui, X.; Wang, Y. Recent Progress in Metal-Organic Frameworks and Their Derived Nanostructures for Energy and Environmental Applications. ChemSusChem 2017, 10, 1645–1663. [Google Scholar] [CrossRef]
- Bazer-Bachi, D.; Assié, L.; Lecocq, V.; Harbuzaru, B.; Falk, V. Towards industrial use of metal-organic framework: Impact of shaping on the MOF properties. Powder Technol. 2014, 255, 52–59. [Google Scholar] [CrossRef]
- Li, Z.; Zeng, H.C. Armored MOFs: Enforcing Soft Microporous MOF Nanocrystals with Hard Mesoporous Silica. J. Am. Chem. Soc. 2014, 136, 5631–5639. [Google Scholar] [CrossRef] [PubMed]
- Blanita, G.; Mihet, M.; Borodi, G.; Misan, I.; Coldea, I.; Lupu, D. Ball milling and compression effects on hydrogen adsorption by MOF:Pt/carbon mixtures. Microporous Mesoporous Mater. 2015, 203, 195–201. [Google Scholar] [CrossRef]
- Ramos-Fernandez, E.V.; Garcia-Domingos, M.; Juan-Alcañiz, J.; Gascon, J.; Kapteijn, F. MOFs meet monoliths: Hierarchical structuring metal organic framework catalysts. Appl. Catal. A Gen. 2011, 391, 261–267. [Google Scholar] [CrossRef]
- Furukawa, S.; Reboul, J.; Diring, S.; Sumida, K.; Kitagawa, S. Structuring of metal–organic frameworks at the mesoscopic/macroscopic scale. Chem. Soc. Rev. 2014, 43, 5700–5734. [Google Scholar] [CrossRef] [PubMed]
- Song, X.; Guan, Q.; Cheng, Z.; Li, W. Eco-friendly controllable synthesis of highly dispersed ZIF-8 embedded in porous Al2O3 and its hydrogenation properties after encapsulating Pt nanoparticles. Appl. Catal. B Environ. 2018, 227, 13–23. [Google Scholar] [CrossRef]
- Górka, J.; Fulvio, P.F.; Pikus, S.; Jaroniec, M. Mesoporous metal organic framework–boehmite and silica composites. Chem. Commun. 2010, 46, 6798–6800. [Google Scholar] [CrossRef]
- Hermes, S.; Zacher, D.; Baunemann, A.; Wöll, C.; Fischer, R.A. Selective Growth and MOCVD Loading of Small Single Crystals of MOF-5 at Alumina and Silica Surfaces Modified with Organic Self-Assembled Monolayers†. Chem. Mater. 2007, 19, 2168–2173. [Google Scholar] [CrossRef]
- Buso, D.; Nairn, K.M.; Gimona, M.; Hill, A.J.; Falcaro, P. Fast Synthesis of MOF-5 Microcrystals Using Sol−Gel SiO2 Nanoparticles. Chem. Mater. 2011, 23, 929–934. [Google Scholar] [CrossRef]
- Karimi, Z.; Morsali, A. Modulated formation of metal-organic frameworks by oriented growth over mesoporous silica. J. Mater. Chem. A 2013, 1, 3047–3054. [Google Scholar] [CrossRef]
- Furtado, A.M.B.; Liu, J.; Wang, Y.; LeVan, M.D. Mesoporous silica–metal organic composite: Synthesis, characterization, and ammonia adsorption. J. Mater. Chem. 2011, 21, 6698–6706. [Google Scholar] [CrossRef]
- Ameloot, R.; Liekens, A.; Alaerts, L.; Maes, M.; Galarneau, A.; Coq, B.; Desmet, G.; Sels, B.F.; Denayer, J.; De Vos, D.E. Silica-MOF Composites as a Stationary Phase in Liquid Chromatography. Eur. J. Inorg. Chem. 2010, 2010, 3735–3738. [Google Scholar] [CrossRef]
- Liu, Y.; Ng, Z.; Khan, E.A.; Jeong, H.-K.; Ching, C.-B.; Lai, Z. Synthesis of continuous MOF-5 membranes on porous α-alumina substrates. Microporous Mesoporous Mater. 2009, 118, 296–301. [Google Scholar] [CrossRef]
- Grad, O.; Mihet, M.; Blanita, G.; Dan, M.; Barbu-Tudoran, L.; Lazar, M.D. MIL-101-Al2O3 as catalytic support in the methanation of CO2—Comparative study between Ni/MIL-101 and Ni/MIL-101-Al2O3 catalysts. Catal. Today 2020, 366, 114–122. [Google Scholar] [CrossRef]
- Cao, N.; Su, J.; Luo, W.; Cheng, G. Ni–Pt nanoparticles supported on MIL-101 as highly efficient catalysts for hydrogen generation from aqueous alkaline solution of hydrazine for chemical hydrogen storage. Int. J. Hydrogen Energy 2014, 39, 9726–9734. [Google Scholar] [CrossRef]
- Serre, C.; Millange, F.; Thouvenot, C.; Nogues, M.; Louer, D.; Ferey, G. Very Large Breathing Effect in the First Nanoporous Chromium (III)-Based Solids: MIL-53 or CrIII(OH)‚{O2C-C6H4-CO2}‚{HO2C-C6H4-CO2H}x*H2Oy. J. Am. Chem. Soc. 2002, 124, 13519–13526. [Google Scholar] [CrossRef]
- Bourrelly, S.; Llewellyn, P.L.; Serre, C.; Millange, F.; Loiseau, T.; Férey, G. Different adsorption behaviors of methane and carbon dioxide in the isotypic nanoporous metal terephthalates MIL-53 and MIL-47. J. Am. Chem. Soc. 2005, 127, 13519–13521. [Google Scholar] [CrossRef]
- Llewellyn, P.L.; Bourrelly, S.; Serre, C.; Filinchuk, Y.; Férey, G. How Hydration Drastically Improves Adsorption Selectivity for CO2 over CH4 in the Flexible Chromium Terephthalate MIL-53. Angew. Chem. 2006, 118, 7915–7918. [Google Scholar] [CrossRef]
- Zhao, X.; Jin, Y.; Zhang, F.; Zhong, Y.; Zhu, W. Catalytic hydrogenation of 2,3,5-trimethylbenzoquinone over Pd nanoparticles confined in the cages of MIL-101(Cr). Chem. Eng. J. 2014, 239, 33–41. [Google Scholar] [CrossRef]
- Thommes, M.; Kaneko, K.; Neimark, A.V.; Olivier, J.P.; Rodriguez-Reinoso, F.; Rouquerol, J.; Sing, K.S.W. Physisorption of gases, with special reference to the evaluation of surface area and pore size distribution (IUPAC Technical Report). Pure Appl. Chem. 2015, 87, 1051–1069. [Google Scholar] [CrossRef] [Green Version]
- Khan, N.A.; Jun, J.W.; Jhung, S.H. Effect of Water Concentration and Acidity on the Synthesis of Porous Chromium Benzenedicarboxylates. Eur. J. Inorg. Chem. 2010, 2010, 1043–1048. [Google Scholar] [CrossRef]
- Han, L.; Zhang, J.; Mao, Y.; Zhou, W.; Xu, W.; Sun, Y. Facile and Green Synthesis of MIL-53(Cr) and Its Excellent Adsorptive Desulfurization Performance. Ind. Eng. Chem. Res. 2019, 58, 15489–15496. [Google Scholar] [CrossRef]
- Zhou, X.; Huang, W.; Liu, J.; Wang, H.; Li, Z. Quenched breathing effect, enhanced CO2 uptake and improved CO2/CH4 selectivity of MIL-53(Cr)/graphene oxide composites. Chem. Eng. Sci. 2017, 167, 98–104. [Google Scholar] [CrossRef]
- Aijaz, A.; Karkamkar, A.; Choi, Y.J.; Tsumori, N.; Ronnebro, E.; Autrey, T.; Shioyama, H.; Xu, Q. Immobilizing Highly Cata-lytically Active Pt Nanoparticles inside the Pores of Metal—Organic Framework: A Double Solvents Approach. J. Am. Chem. Soc. 2012, 134, 13926–13929. [Google Scholar] [CrossRef] [PubMed]
- Swalus, C.; Jacquemin, M.; Poleunis, C.; Bertrand, P.; Ruiz, P. CO2 methanation on Rh/γ-Al2O3 catalyst at low temperature: “In situ” supply of hydrogen by Ni/activated carbon catalyst. Appl. Catal. B Environ. 2012, 125, 41–50. [Google Scholar] [CrossRef]






| Sample | SBET (m2/g) | Vp (cm3/g) | Vμ * (cm3/g) | Dm (nm) |
|---|---|---|---|---|
| MIL-53 | 933.2 | 0.48 | 0.34 | 0.4–2 |
| Ni(10%)/MIL-53 | 519.9 | 0.31 | 0.18 | 0.4–2 |
| Al2O3 | 110.0 | 0.21 | - | 10–25 |
| MIL-53–Al2O3 | 489.4 | 0.50 | 0.07 | 0.6–2.2; 3–6; 10–25 |
| Ni(10%)/MIL-53–Al2O3 | 277.9 | 0.42 | 0.03 | 0.6–2.8; 3–6; 12–25 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Grad, O.; Blanita, G.; Lazar, M.D.; Mihet, M. Methanation of CO2 Using MIL-53-Based Catalysts: Ni/MIL-53–Al2O3 versus Ni/MIL-53. Catalysts 2021, 11, 1412. https://doi.org/10.3390/catal11111412
Grad O, Blanita G, Lazar MD, Mihet M. Methanation of CO2 Using MIL-53-Based Catalysts: Ni/MIL-53–Al2O3 versus Ni/MIL-53. Catalysts. 2021; 11(11):1412. https://doi.org/10.3390/catal11111412
Chicago/Turabian StyleGrad, Oana, Gabriela Blanita, Mihaela D. Lazar, and Maria Mihet. 2021. "Methanation of CO2 Using MIL-53-Based Catalysts: Ni/MIL-53–Al2O3 versus Ni/MIL-53" Catalysts 11, no. 11: 1412. https://doi.org/10.3390/catal11111412








