Organocatalysis: A Tool of Choice for the Enantioselective Nucleophilic Dearomatization of Electron-Deficient Six-Membered Ring Azaarenium Salts
Abstract
:1. Introduction
2. Anion-Binding Catalysis
2.1. Activation Mode of Azaarenium Salts in Anion-Binding Catalysis
2.2. Isoquinolinium Salts
2.3. Quinolinium Salts
2.4. Pyridinium Salts
2.5. Others
3. Aminocatalysis
3.1. Activation Mode of Azaarenium Salts in Aminocatalysis
3.2. Isoquinolinium Salts
3.3. Quinolinium Salts
3.4. Pyridinium Salts
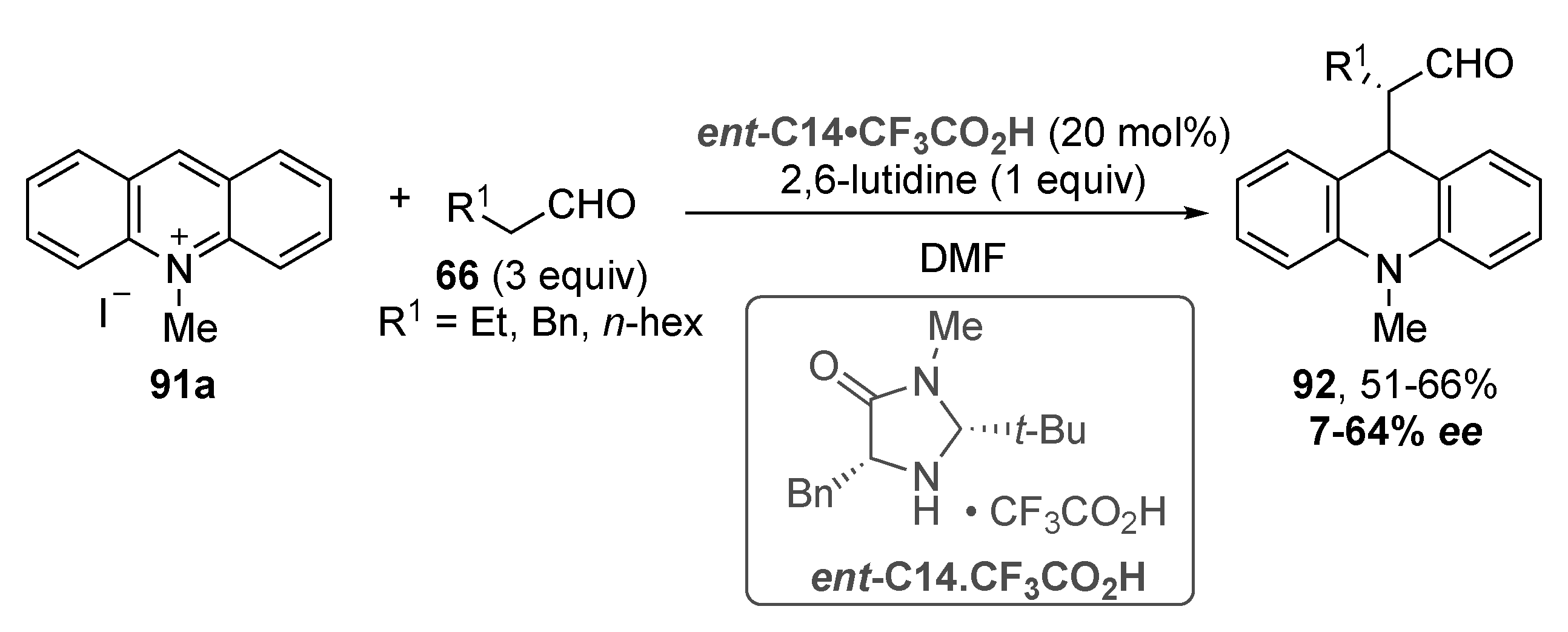
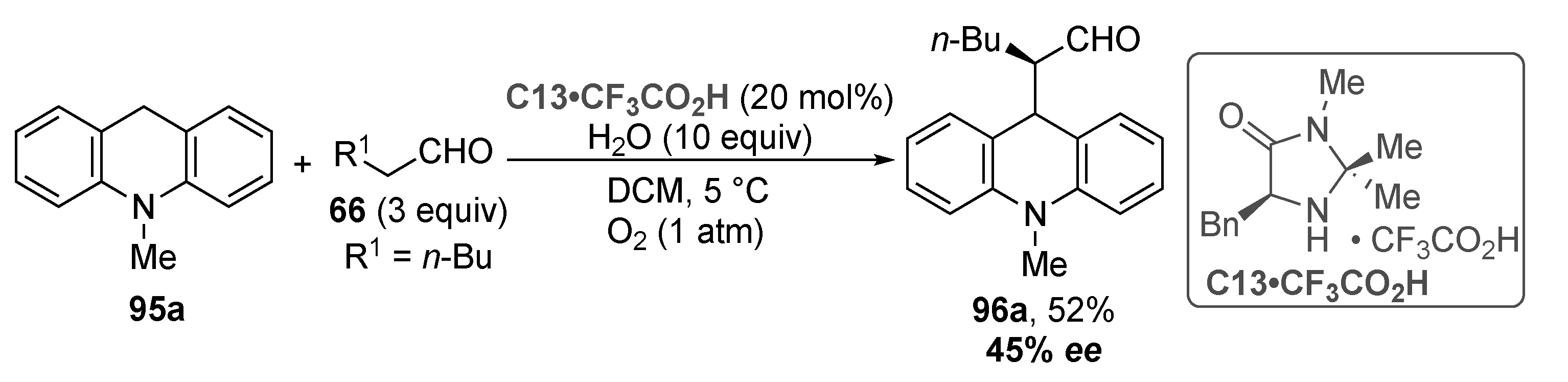
3.5. Acridinium Salts
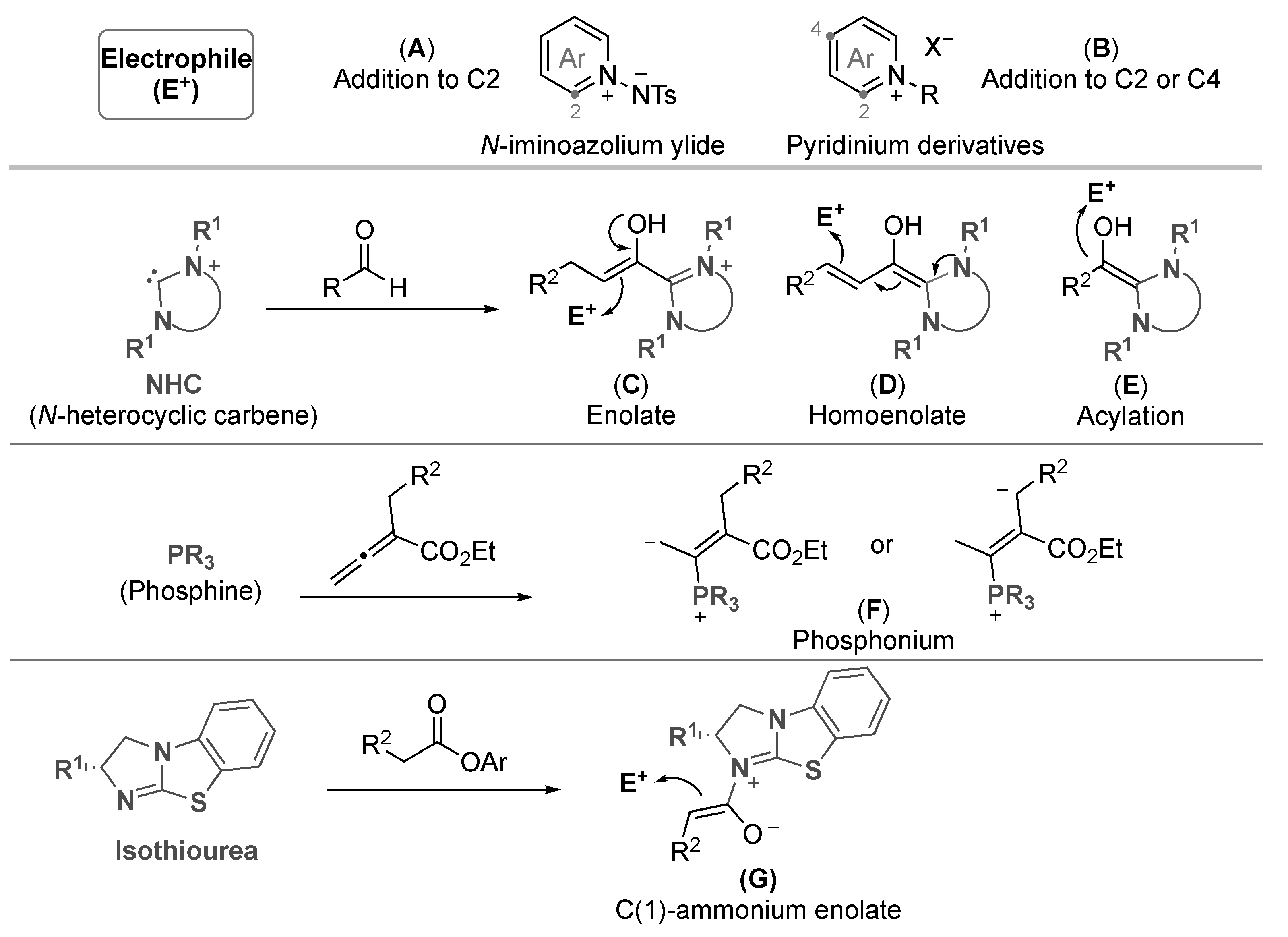
4. Nucleophilic Catalysis
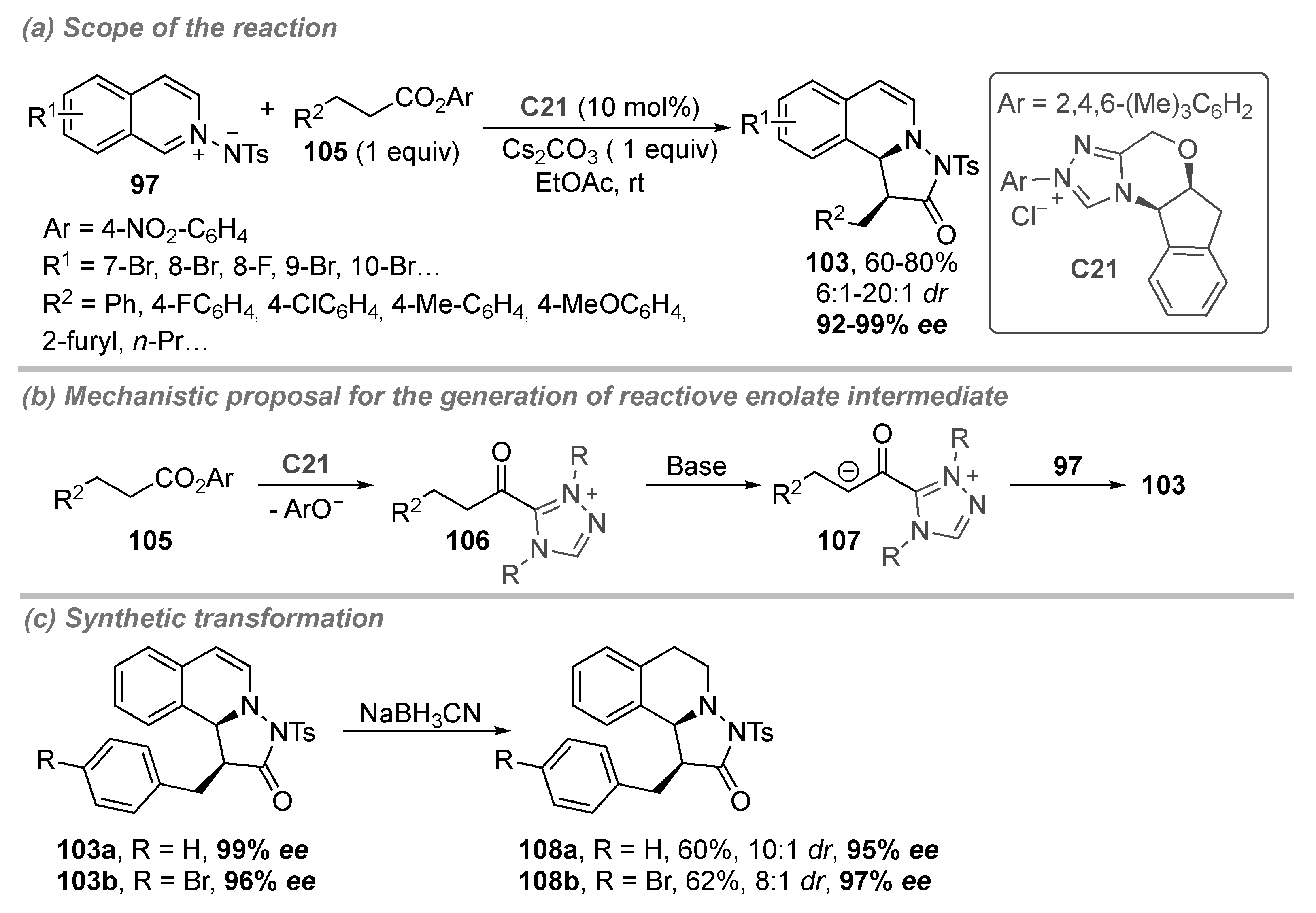
4.1. Isoquinolinium Salts
4.2. Pyridinium Salts
4.3. Others
5. Miscellaneous
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations and Symbols
| Cbz | benzyloxycarbonyl |
| CSA | camphorsulfonic acid |
| Boc | tert-butyloxycarbonyl |
| DCM | dichloromethane |
| DMAP | 4-dimethylaminopyridine |
| MMPP | magnesium monoperoxyphtalate hexahydrate |
| MTBE | methyl tert-butyl ether |
| NHC | N-heterocyclic carbene |
| PIFA | [bis(trifluoroacetoxy)-iodo]benzene |
| TBS | tert-butyl(dimethyl)silyl |
| TFA | triluoroacetic acid |
| Troc | 2,2,2-trichloroethoxycarbony |
| Ts | tosyl |
References
- Roche, S.P.; Porco, J.A. Dearomatization Strategies in the Synthesis of Complex Natural Products. Angew. Chem. Int. Ed. 2011, 50, 4068–4093. [Google Scholar] [CrossRef] [PubMed]
- Satoh, N.; Akiba, T.; Yokoshima, S.; Fukuyama, T. A practical synthesis of (-)-oseltamivir. Angew. Chem. Int. Ed. 2007, 46, 5734–5736. [Google Scholar] [CrossRef]
- Lavilla, R. Recent developments in the chemistry of dihydropyridines. J. Chem. Soc. Perkin Trans. 2002, 1, 1141–1156. [Google Scholar] [CrossRef]
- Auria-Luna, F.; Marqués-López, E.; Herrera, R.P. Organocatalytic Enantioselective Synthesis of 1,4-Dihydropyridines. Adv. Synth. Catal. 2017, 359, 2161–2175. [Google Scholar] [CrossRef]
- Thu Pham, H.; Chataigner, I.; Renaud, J.-L. New Approaches to Nitrogen Containing Heterocycles: Enantioselective Organocatalyzed Synthesis of Dihydropyridines (DHP’s), Quinolizidine Derivatives and Dihydropyrimidines (DHPM’s). Curr. Org. Chem. 2012, 16, 1754–1775. [Google Scholar] [CrossRef]
- Rucins, M.; Plotniece, A.; Bernotiene, E.; Tsai, W.-B.; Sobolev, A. Recent Approaches to Chiral 1,4-Dihydropyridines and their Fused Analogues. Catalysts 2020, 10, 1019. [Google Scholar] [CrossRef]
- Faisca Phillips, A.M.; Pombeiro, A.J. Recent advances in organocatalytic enantioselective transfer hydrogenation. Org. Biomol. Chem. 2017, 15, 2307–2340. [Google Scholar] [CrossRef]
- Kim, A.N.; Stoltz, B.M. Recent Advances in Homogeneous Catalysts for the Asymmetric Hydrogenation of Heteroarenes. ACS Catal. 2020, 10, 13834–13851. [Google Scholar] [CrossRef] [PubMed]
- Xia, Z.L.; Xu-Xu, Q.F.; Zheng, C.; You, S.L. Chiral phosphoric acid-catalyzed asymmetric dearomatization reactions. Chem. Soc. Rev. 2020, 49, 286–300. [Google Scholar] [CrossRef] [PubMed]
- Poddubnyi, I.S. Regioselectivity of the reactions of pyridinium and quinolinium salts with various nucleophiles (Review). Chem. Heterocycl. Comp. 1995, 31, 682–714. [Google Scholar] [CrossRef]
- Ahamed, M.; Todd, M.H. Catalytic Asymmetric Additions of Carbon-Centered Nucleophiles to Nitrogen-Containing Aromatic Heterocycles. Eur. J. Org. Chem. 2010, 2010, 5935–5942. [Google Scholar] [CrossRef]
- Bull, J.A.; Mousseau, J.J.; Pelletier, G.; Charette, A.B. Synthesis of pyridine and dihydropyridine derivatives by regio- and stereoselective addition to N-activated pyridines. Chem. Rev. 2012, 112, 2642–2713. [Google Scholar] [CrossRef] [PubMed]
- Zhuo, C.X.; Zhang, W.; You, S.L. Catalytic asymmetric dearomatization reactions. Angew. Chem. Int. Ed. 2012, 51, 12662–12686. [Google Scholar] [CrossRef] [PubMed]
- Ding, Q.; Zhou, X.; Fan, R. Recent advances in dearomatization of heteroaromatic compounds. Org. Biomol. Chem. 2014, 12, 4807–4815. [Google Scholar] [CrossRef]
- Gualandi, A.; Mengozzi, L.; Manoni, E.; Cozzi, P.G. Stereoselective Organocatalytic Addition of Nucleophiles to Isoquinolinium and 3,4-dihydroisoquinolinium Ions: A Simple Approach for the Synthesis of Isoquinoline Alkaloids. Catal. Lett. 2014, 145, 398–419. [Google Scholar] [CrossRef]
- Ramachandran, G.; Sathiyanarayanan, K. Dearomatization Strategies of Heteroaromatic Compounds. Curr. Organocatalysis 2015, 2, 14–26. [Google Scholar] [CrossRef]
- Liu, W.; Liu, S.; Jin, R.; Guo, H.; Zhao, J. Novel strategies for catalytic asymmetric synthesis of C1-chiral 1,2,3,4-tetrahydroisoquinolines and 3,4-dihydrotetrahydroisoquinolines. Org. Chem. Front. 2015, 2, 288–299. [Google Scholar] [CrossRef]
- Bertuzzi, G.; Bernardi, L.; Fochi, M. Nucleophilic Dearomatization of Activated Pyridines. Catalysts 2018, 8, 632. [Google Scholar] [CrossRef]
- Sharma, U.K.; Ranjan, P.; Van der Eycken, E.V.; You, S.L. Sequential and direct multicomponent reaction (MCR)-based dearomatization strategies. Chem. Soc. Rev. 2020, 49, 8721–8748. [Google Scholar] [CrossRef]
- Xia, Y.; Hu, F.; Jia, J. Transition-Metal-Catalyzed Nucleophilic Dearomatization of Electron-Deficient Heteroarenes. Synthesis 2021. [Google Scholar] [CrossRef]
- Dalko, P.I.; Moisan, L. Enantioselective Organocatalysis. Angew. Chem. Int. Ed. 2001, 40, 3726–3748. [Google Scholar] [CrossRef]
- Dalko, P.I.; Moisan, L. In the golden age of organocatalysis. Angew. Chem. Int. Ed. 2004, 43, 5138–5175. [Google Scholar] [CrossRef]
- Ooi, T.; Maruoka, K. Asymmetric organocatalysis of structurally well-defined chiral quaternary ammonium fluorides. Acc. Chem. Res. 2004, 37, 526–533. [Google Scholar] [CrossRef] [PubMed]
- Berkessel, A.; Gröger, H. Asymmetric Organocatalysis; Wiley-VCH Verlag GmbH: Weinheim, Germany, 2005. [Google Scholar]
- Seayad, J.; List, B. Asymmetric organocatalysis. Org. Biomol. Chem. 2005, 3, 719–724. [Google Scholar] [CrossRef] [PubMed]
- Beeson, T.D.; Mastracchio, A.; Hong, J.B.; Ashton, K.; Macmillan, D.W. Enantioselective organocatalysis using SOMO activation. Science 2007, 316, 582–585. [Google Scholar] [CrossRef]
- Enders, D.; Niemeier, O.; Henseler, A. Organocatalysis by N-heterocyclic carbenes. Chem. Rev. 2007, 107, 5606–5655. [Google Scholar] [CrossRef] [PubMed]
- Gaunt, M.J.; Johansson, C.C.; McNally, A.; Vo, N.T. Enantioselective organocatalysis. Drug Discov. Today 2007, 12, 8–27. [Google Scholar] [CrossRef] [PubMed]
- MacMillan, D.W. The advent and development of organocatalysis. Nature 2008, 455, 304–308. [Google Scholar] [CrossRef] [PubMed]
- Bertelsen, S.; Jorgensen, K.A. Organocatalysis-after the gold rush. Chem. Soc. Rev. 2009, 38, 2178–2189. [Google Scholar] [CrossRef] [PubMed]
- Briere, J.F.; Oudeyer, S.; Dalla, V.; Levacher, V. Recent advances in cooperative ion pairing in asymmetric organocatalysis. Chem. Soc. Rev. 2012, 41, 1696–1707. [Google Scholar] [CrossRef]
- Dalko, P.I. Comprehensive Enantioselective Organocatalysis; Wiley-VCH Verlag GbmH & Co. KGaA: Weinheim, Germany, 2013. [Google Scholar]
- Serdyuk, O.V.; Heckel, C.M.; Tsogoeva, S.B. Bifunctional primary amine-thioureas in asymmetric organocatalysis. Org. Biomol. Chem. 2013, 11, 7051–7071. [Google Scholar] [CrossRef] [PubMed]
- Legros, F.; Oudeyer, S.; Levacher, V. New Developments in Chiral Cooperative Ion Pairing Organocatalysis by Means of Ammonium Oxyanions and Fluorides: From Protonation to Deprotonation Reactions. Chem. Rec. 2017, 17, 429–440. [Google Scholar] [CrossRef]
- Oudeyer, S.; Levacher, V.; Brière, J.-F. Chiral Quaternary Ammonium Salts in Organocatalysis. In Quaternary Ammonium Salts in Organocatalysis; Elsevier: Amsterdam, The Netherlands, 2017; pp. 87–173. [Google Scholar]
- Guo, H.; Fan, Y.C.; Sun, Z.; Wu, Y.; Kwon, O. Phosphine Organocatalysis. Chem. Rev. 2018, 118, 10049–10293. [Google Scholar] [CrossRef] [PubMed]
- Lopez, S.S.; Nimmagadda, S.K.; Antilla, J.C. Organocatalytic Asymmetric Dearomatization Reactions. In Asymmetric Dearomatization Reactions; Wiley-VCH Verlag GmbH: Weinheim, Germany, 2016; pp. 175–206. [Google Scholar]
- Schifferer, L.; Stinglhamer, M.; Kaur, K.; Macheño, O.G. Halides as versatile anions in asymmetric anion-binding organocatalysis. Beilstein J. Org. Chem. 2021, 17, 2270–2286. [Google Scholar] [CrossRef] [PubMed]
- Gandhi, S.; Sivadas, V.; Baire, B. Thiourea–Tertiary Amine Promoted Cascade Catalysis: A Tool for Complexity Generation. Eur. J. Org. Chem. 2020, 2021, 220–234. [Google Scholar] [CrossRef]
- Doyle, A.G.; Jacobsen, E.N. Small-molecule H-bond donors in asymmetric catalysis. Chem. Rev. 2007, 107, 5713–5743. [Google Scholar] [CrossRef]
- Taylor, M.S.; Tokunaga, N.; Jacobsen, E.N. Enantioselective thiourea-catalyzed acyl-mannich reactions of isoquinolines. Angew. Chem. Int. Ed. 2005, 44, 6700–6704. [Google Scholar] [CrossRef]
- Schafer, A.G.; Wieting, J.M.; Fisher, T.J.; Mattson, A.E. Chiral silanediols in anion-binding catalysis. Angew. Chem. Int. Ed. 2013, 52, 11321–11324. [Google Scholar] [CrossRef]
- Chandrasekhar, V.; Boomishankar, R.; Nagendran, S. Recent developments in the synthesis and structure of organosilanols. Chem. Rev. 2004, 104, 5847–5910. [Google Scholar] [CrossRef]
- Zurro, M.; Asmus, S.; Bamberger, J.; Beckendorf, S.; Garcia Mancheno, O. Chiral Triazoles in Anion-Binding Catalysis: New Entry to Enantioselective Reissert-Type Reactions. Chemistry 2016, 22, 3785–3793. [Google Scholar] [CrossRef]
- Ray Choudhury, A.; Mukherjee, S. Enantioselective dearomatization of isoquinolines by anion-binding catalysis en route to cyclic alpha-aminophosphonates. Chem. Sci. 2016, 7, 6940–6945. [Google Scholar] [CrossRef] [PubMed]
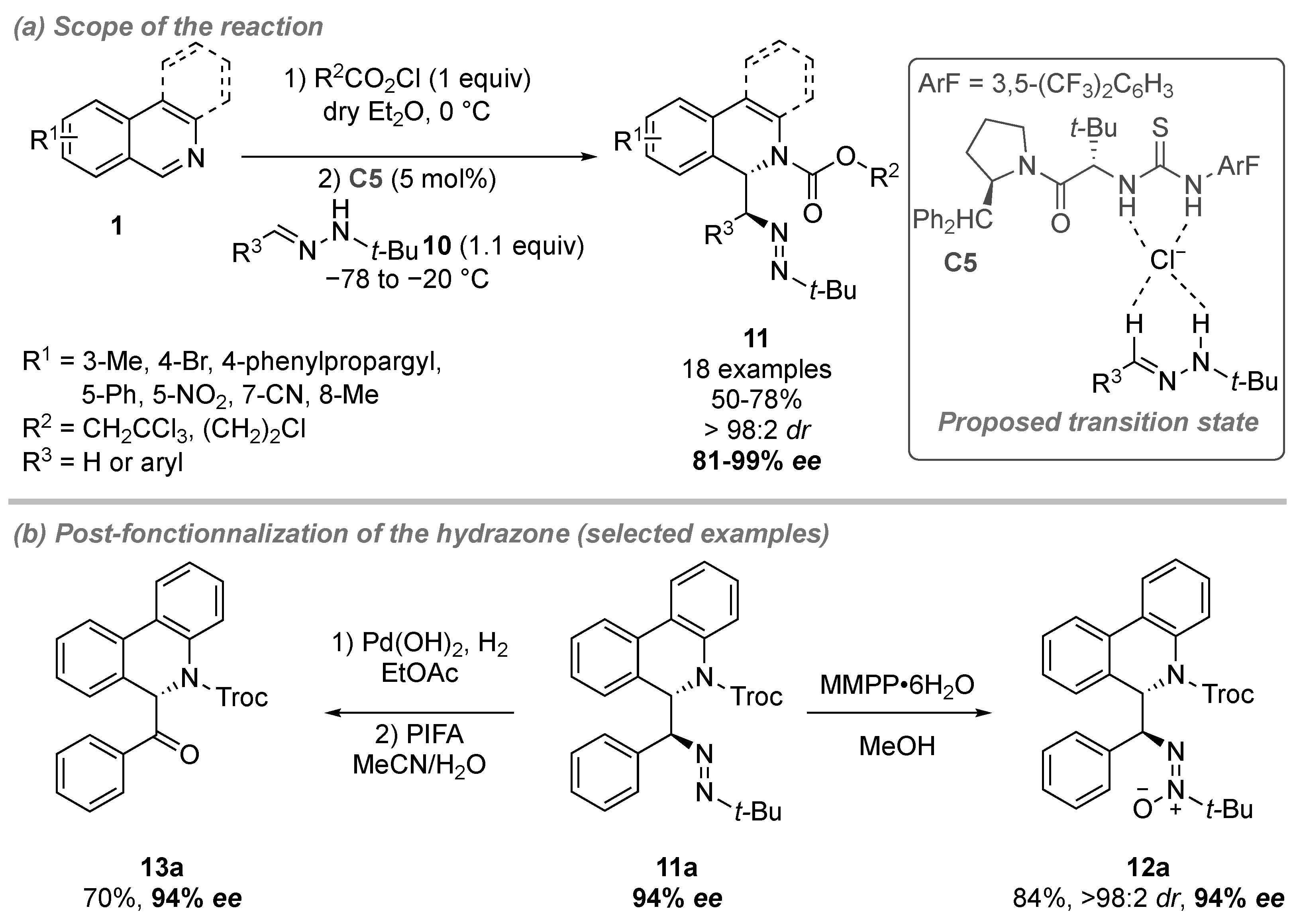
- Matador, E.; Iglesias-Siguenza, J.; Monge, D.; Merino, P.; Fernandez, R.; Lassaletta, J.M. Enantio- and Diastereoselective Nucleophilic Addition of N-tert-Butylhydrazones to Isoquinolinium Ions through Anion-Binding Catalysis. Angew. Chem. Int. Ed. 2021, 60, 5096–5101. [Google Scholar] [CrossRef] [PubMed]
- De, C.K.; Mittal, N.; Seidel, D. A dual-catalysis approach to the asymmetric Steglich rearrangement and catalytic enantioselective addition of O-acylated azlactones to isoquinolines. J. Am. Chem. Soc. 2011, 133, 16802–16805. [Google Scholar] [CrossRef] [PubMed]
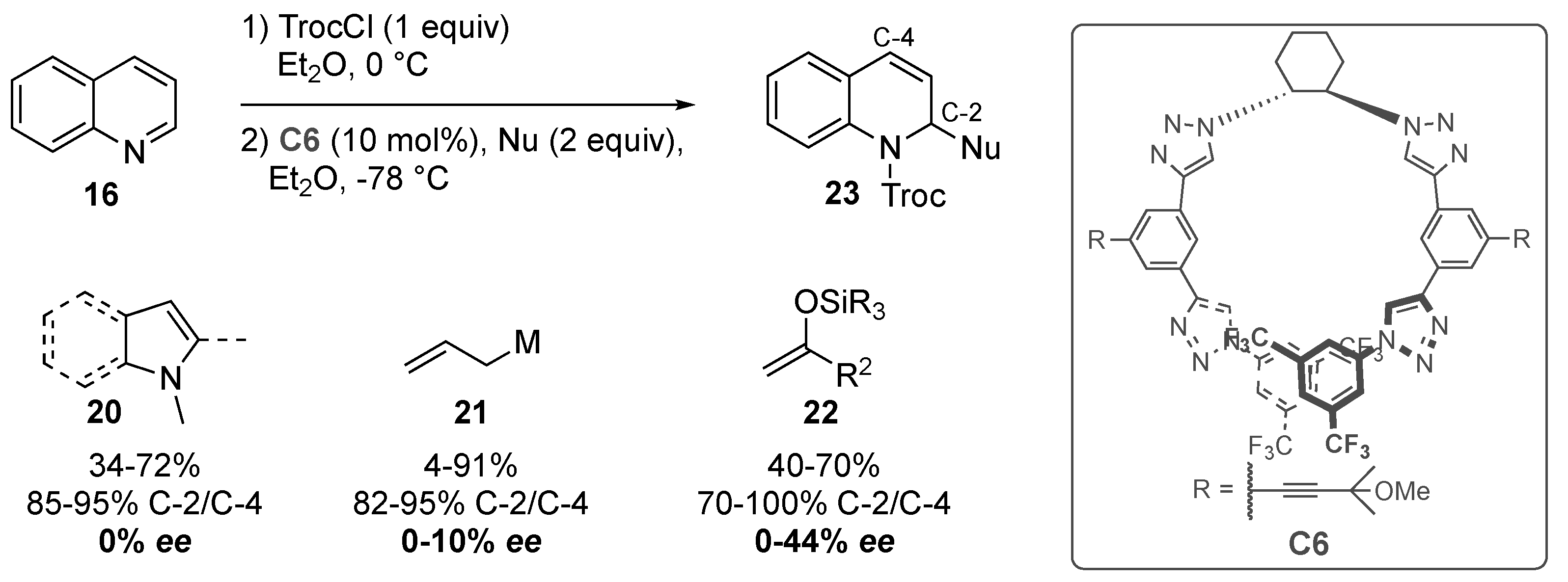
- Zurro, M.; Asmus, S.; Beckendorf, S.; Muck-Lichtenfeld, C.; Mancheno, O.G. Chiral helical oligotriazoles: New class of anion-binding catalysts for the asymmetric dearomatization of electron-deficient N-heteroarenes. J. Am. Chem. Soc. 2014, 136, 13999–14002. [Google Scholar] [CrossRef]
- Fischer, T.; Duong, Q.N.; Garcia Mancheno, O. Triazole-Based Anion-Binding Catalysis for the Enantioselective Dearomatization of N-Heteroarenes with Phosphorus Nucleophiles. Chemistry 2017, 23, 5983–5987. [Google Scholar] [CrossRef]
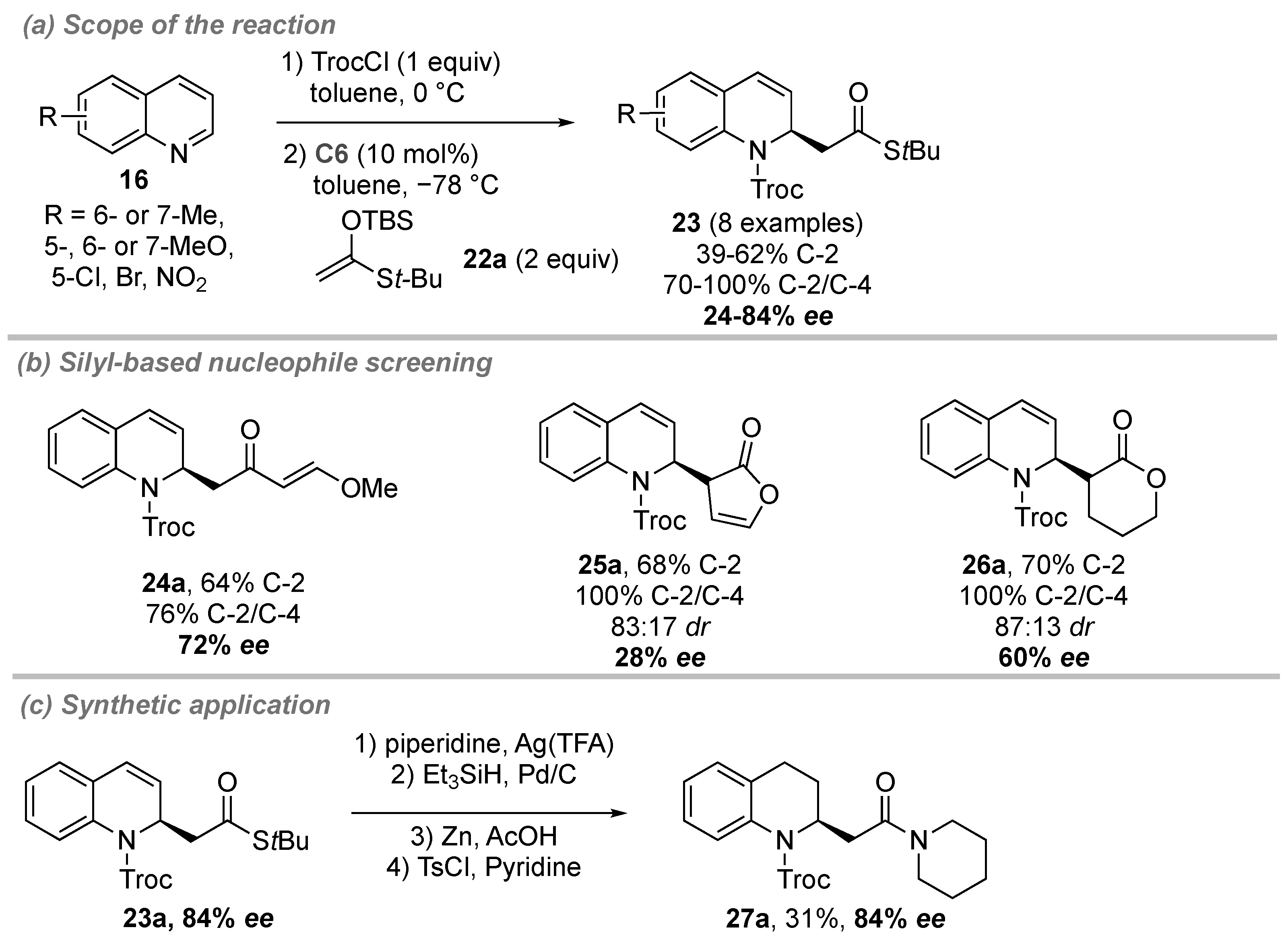
- Duong, Q.-N.; Schifferer, L.; García Mancheño, O. Nucleophile Screening in Anion-Binding Reissert-Type Reactions of Quinolines with Chiral Tetrakis(triazole) Catalysts. Eur. J. Org. Chem. 2019, 2019, 5452–5461. [Google Scholar] [CrossRef]
- Gomez-Martinez, M.; Del Carmen Perez-Aguilar, M.; Piekarski, D.G.; Daniliuc, C.G.; Garcia Mancheno, O. N,N-Dialkylhydrazones as Versatile Umpolung Reagents in Enantioselective Anion-Binding Catalysis. Angew. Chem. Int. Ed. 2021, 60, 5102–5107. [Google Scholar] [CrossRef]
- Yamaoka, Y.; Miyabe, H.; Takemoto, Y. Catalytic enantioselective petasis-type reaction of quinolines catalyzed by a newly designed thiourea catalyst. J. Am. Chem. Soc. 2007, 129, 6686–6687. [Google Scholar] [CrossRef]
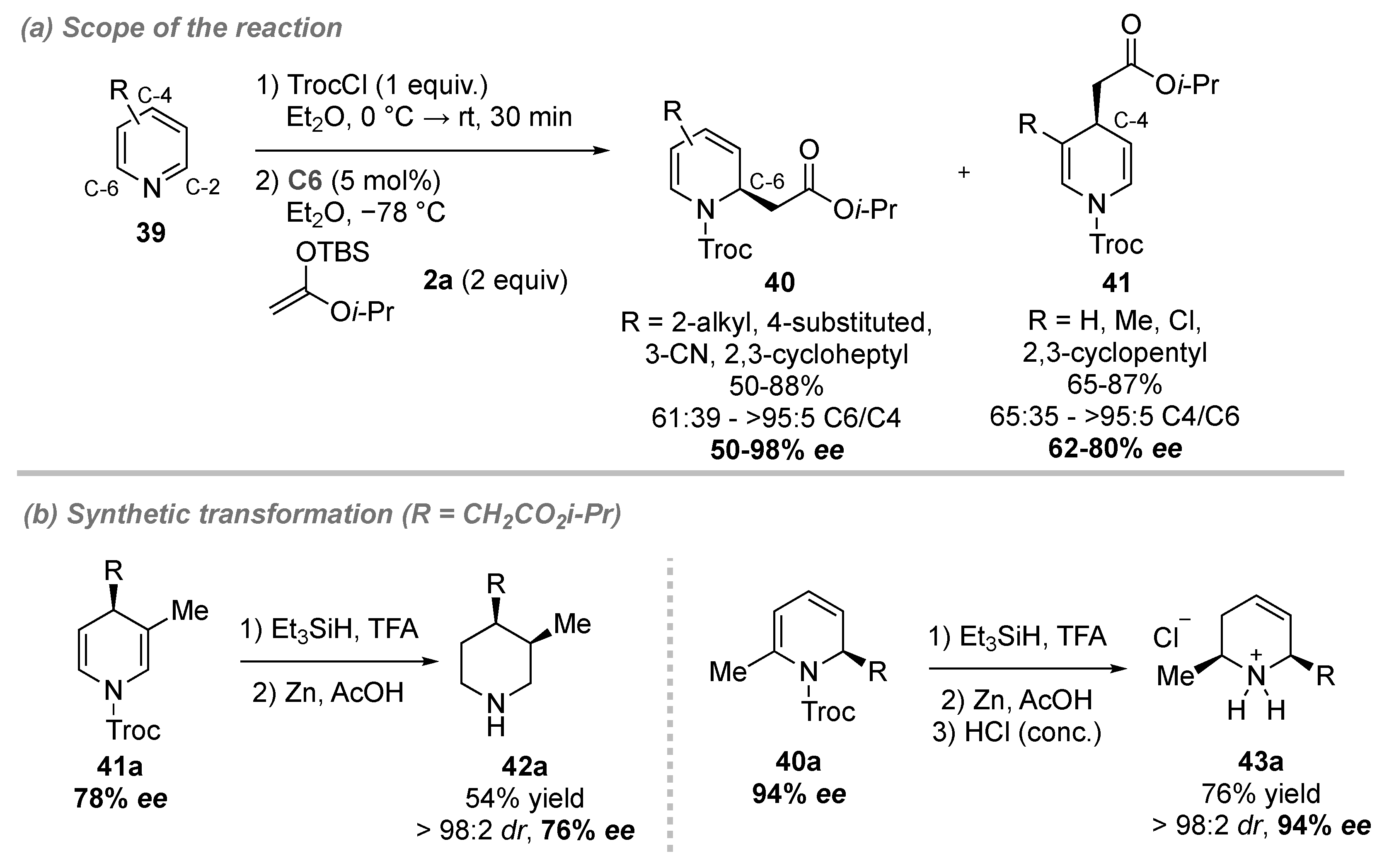
- Garcia Mancheno, O.; Asmus, S.; Zurro, M.; Fischer, T. Highly Enantioselective Nucleophilic Dearomatization of Pyridines by Anion-Binding Catalysis. Angew. Chem. Int. Ed. 2015, 54, 8823–8827. [Google Scholar] [CrossRef]
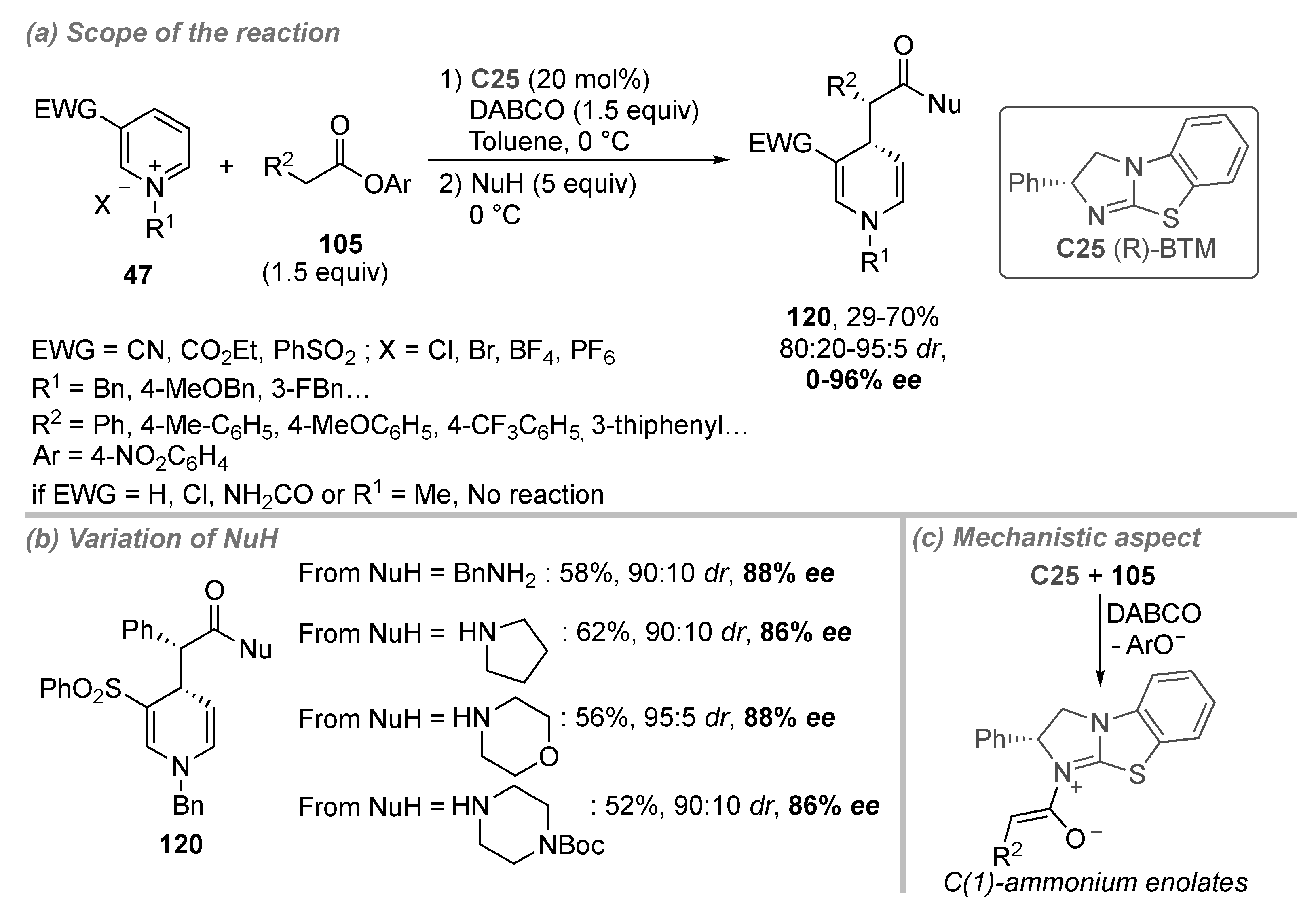
- Bertuzzi, G.; Sinisi, A.; Caruana, L.; Mazzanti, A.; Fochi, M.; Bernardi, L. Catalytic Enantioselective Addition of Indoles to Activated N-Benzylpyridinium Salts: Nucleophilic Dearomatization of Pyridines with Unusual C-4 Regioselectivity. ACS Catal. 2016, 6, 6473–6477. [Google Scholar] [CrossRef]
- Fischer, T.; Bamberger, J.; Mancheno, O.G. Asymmetric nucleophilic dearomatization of diazarenes by anion-binding catalysis. Org. Biomol. Chem. 2016, 14, 5794–5802. [Google Scholar] [CrossRef]
- Kaur, K.; Humbrias-Martin, J.; Hoppmann, L.; Fernandez-Salas, J.A.; Daniliuc, C.G.; Aleman, J.; Mancheno, O.G. Enantioselective vinylogous-Mukaiyama-type dearomatisation by anion-binding catalysis. Chem. Commun. 2021, 57, 9244–9247. [Google Scholar] [CrossRef]
- Melchiorre, P.; Marigo, M.; Carlone, A.; Bartoli, G. Asymmetric aminocatalysis--gold rush in organic chemistry. Angew. Chem. Int. Ed. 2008, 47, 6138–6171. [Google Scholar] [CrossRef]
- Frisch, K.; Landa, A.; Saaby, S.; Jorgensen, K.A. Organocatalytic diastereo- and enantioselective annulation reactions--construction of optically active 1,2-dihydroisoquinoline and 1,2-dihydrophthalazine derivatives. Angew. Chem. Int. Ed. 2005, 44, 6058–6063. [Google Scholar] [CrossRef] [PubMed]
- Mengozzi, L.; Gualandi, A.; Cozzi, P.G. A highly enantioselective acyl-Mannich reaction of isoquinolines with aldehydes promoted by proline derivatives: An approach to 13-alkyl-tetrahydroprotoberberine alkaloids. Chem. Sci. 2014, 5, 3915–3921. [Google Scholar] [CrossRef]
- Berti, F.; Malossi, F.; Marchetti, F.; Pineschi, M. A highly enantioselective Mannich reaction of aldehydes with cyclic N-acyliminium ions by synergistic catalysis. Chem. Commun. 2015, 51, 13694–13697. [Google Scholar] [CrossRef] [PubMed]
- Sun, S.; Mao, Y.; Lou, H.; Liu, L. Copper(II)/amine synergistically catalyzed enantioselective alkylation of cyclic N-acyl hemiaminals with aldehydes. Chem. Commun. 2015, 51, 10691–10694. [Google Scholar] [CrossRef] [PubMed]
- Volla, C.M.; Fava, E.; Atodiresei, I.; Rueping, M. Dual metal and Lewis base catalysis approach for asymmetric synthesis of dihydroquinolines and the alpha-arylation of aldehydes via N-acyliminium ions. Chem. Commun. 2015, 51, 15788–15791. [Google Scholar] [CrossRef]
- Mengozzi, L.; Gualandi, A.; Cozzi, P.G. Organocatalytic Stereoselective Addition of Aldehydes to Acylquinolinium Ions. Eur. J. Org. Chem. 2016, 2016, 3200–3207. [Google Scholar] [CrossRef]
- Song, X.; Yan, R.J.; Du, W.; Chen, Y.C. Asymmetric Dearomative Cascade Multiple Functionalizations of Activated N-Alkylpyridinium and N-Alkylquinolinium Salts. Org. Lett. 2020, 22, 7617–7621. [Google Scholar] [CrossRef]
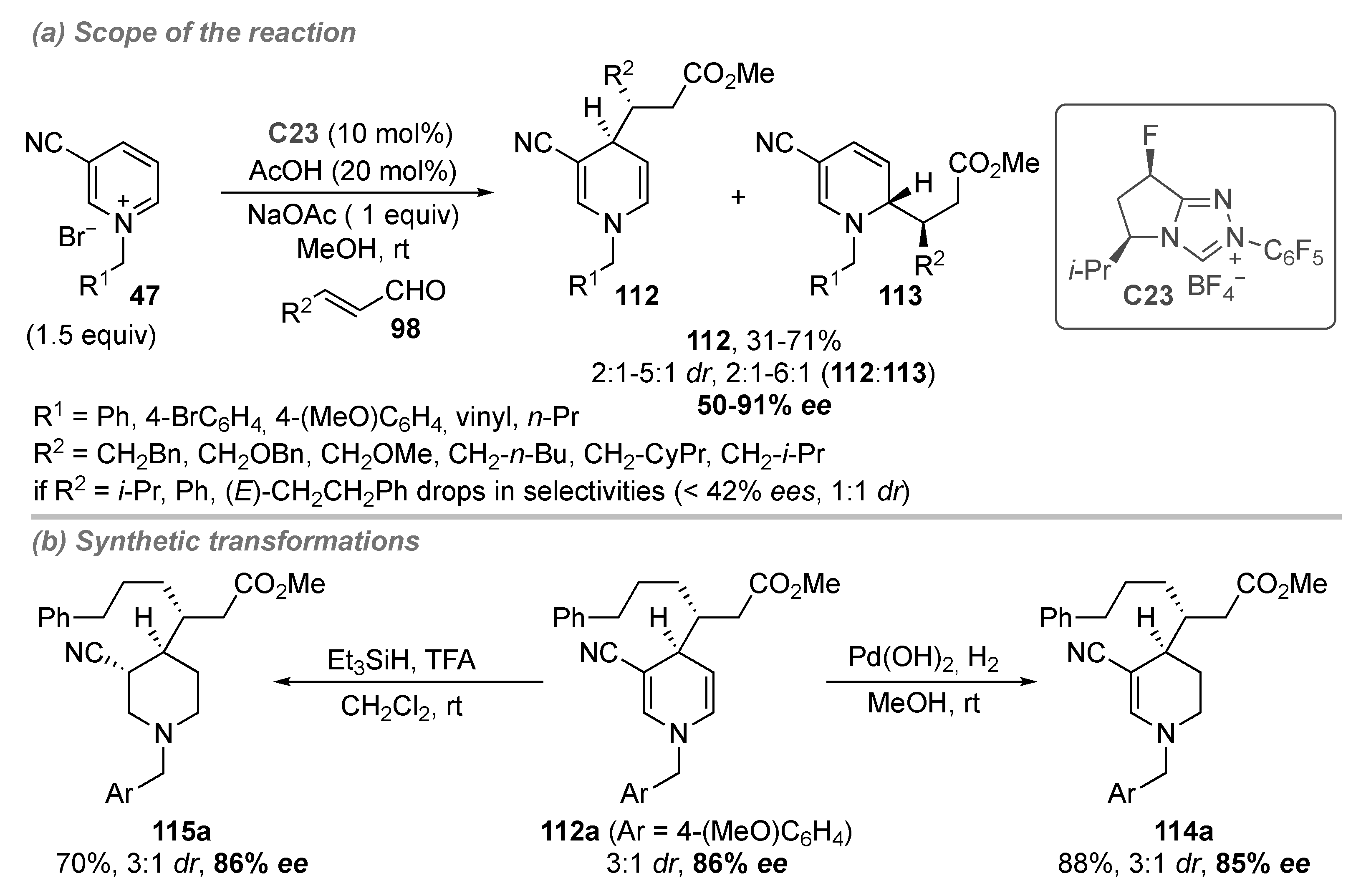
- Bertuzzi, G.; Sinisi, A.; Pecorari, D.; Caruana, L.; Mazzanti, A.; Bernardi, L.; Fochi, M. Nucleophilic Dearomatization of Pyridines under Enamine Catalysis: Regio-, Diastereo-, and Enantioselective Addition of Aldehydes to Activated N-Alkylpyridinium Salts. Org. Lett. 2017, 19, 834–837. [Google Scholar] [CrossRef]
- Yan, R.J.; Xiao, B.X.; Ouyang, Q.; Liang, H.P.; Du, W.; Chen, Y.C. Asymmetric Dearomative Formal [4 + 2] Cycloadditions of N,4-Dialkylpyridinium Salts and Enones To Construct Azaspiro[5.5]undecane Frameworks. Org. Lett. 2018, 20, 8000–8003. [Google Scholar] [CrossRef]
- Benfatti, F.; Benedetto, E.; Cozzi, P.G. Organocatalytic stereoselective alpha-alkylation of aldehydes with stable carbocations. Chem. Asian J. 2010, 5, 2047–2052. [Google Scholar] [CrossRef]
- Liang, T.; Xiao, J.; Xiong, Z.; Li, X. Organocatalytic asymmetric 1,4-addition of aldehydes to acridiniums catalyzed by a diarylprolinol silyl ether. J. Org. Chem. 2012, 77, 3583–3588. [Google Scholar] [CrossRef]
- Zhang, B.; Xiang, S.K.; Zhang, L.H.; Cui, Y.; Jiao, N. Organocatalytic asymmetric intermolecular dehydrogenative alpha-alkylation of aldehydes using molecular oxygen as oxidant. Org. Lett. 2011, 13, 5212–5215. [Google Scholar] [CrossRef]
- Guo, C.; Fleige, M.; Janssen-Muller, D.; Daniliuc, C.G.; Glorius, F. Switchable selectivity in an NHC-catalysed dearomatizing annulation reaction. Nat. Chem. 2015, 7, 842–847. [Google Scholar] [CrossRef]
- Wang, Y.; Qu, L.B.; Wei, D. Prediction on the Origin of Selectivities in Base-controlled Switchable NHC-catalyzed Transformations. Chem. Asian J. 2019, 14, 293–300. [Google Scholar] [CrossRef] [PubMed]
- Zhang, P.; Zhou, Y.; Han, X.; Xu, J.; Liu, H. N-Heterocyclic Carbene Catalyzed Enantioselective [3 + 2] Dearomatizing Annulation of Saturated Carboxylic Esters with N-Iminoisoquinolinium Ylides. J. Org. Chem. 2018, 83, 3879–3888. [Google Scholar] [CrossRef]
- Xu, J.H.; Zheng, S.C.; Zhang, J.W.; Liu, X.Y.; Tan, B. Construction of Tropane Derivatives by the Organocatalytic Asymmetric Dearomatization of Isoquinolines. Angew. Chem. Int. Ed. 2016, 55, 11834–11839. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Wu, Q.-Y.; Lai, T.-H.; Zheng, K.-J.; Qu, L.-B.; Wei, D. Prediction on the origin of selectivities of NHC-catalyzed asymmetric dearomatization (CADA) reactions. Catal. Sci. Technol. 2019, 9, 465–476. [Google Scholar] [CrossRef]
- Flanigan, D.M.; Rovis, T. Enantioselective N-heterocyclic carbene-catalyzed nucleophilic dearomatization of alkyl pyridiniums. Chem. Sci. 2017, 8, 6566–6569. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Qu, L.B.; Lan, Y.; Wei, D. Origin of Regio- and Stereoselectivity in the NHC-catalyzed Reaction of Alkyl Pyridinium with Aliphatic Enal. ChemCatChem 2019, 12, 1068–1074. [Google Scholar] [CrossRef]
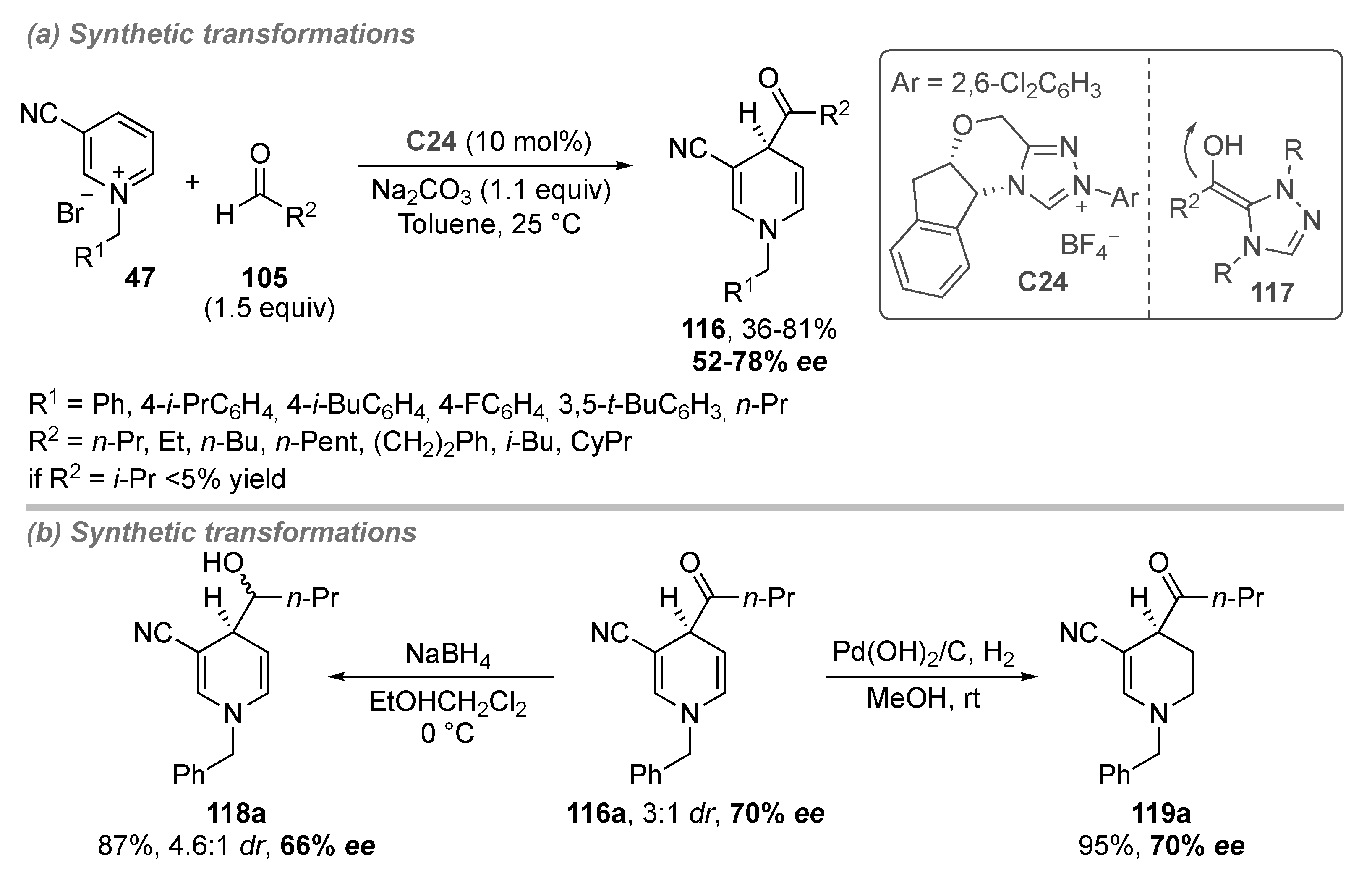
- Di Carmine, G.; Ragno, D.; Bortolini, O.; Giovannini, P.P.; Mazzanti, A.; Massi, A.; Fogagnolo, M. Enantioselective Dearomatization of Alkylpyridiniums by N-Heterocyclic Carbene-Catalyzed Nucleophilic Acylation. J. Org. Chem. 2018, 83, 2050–2057. [Google Scholar] [CrossRef]
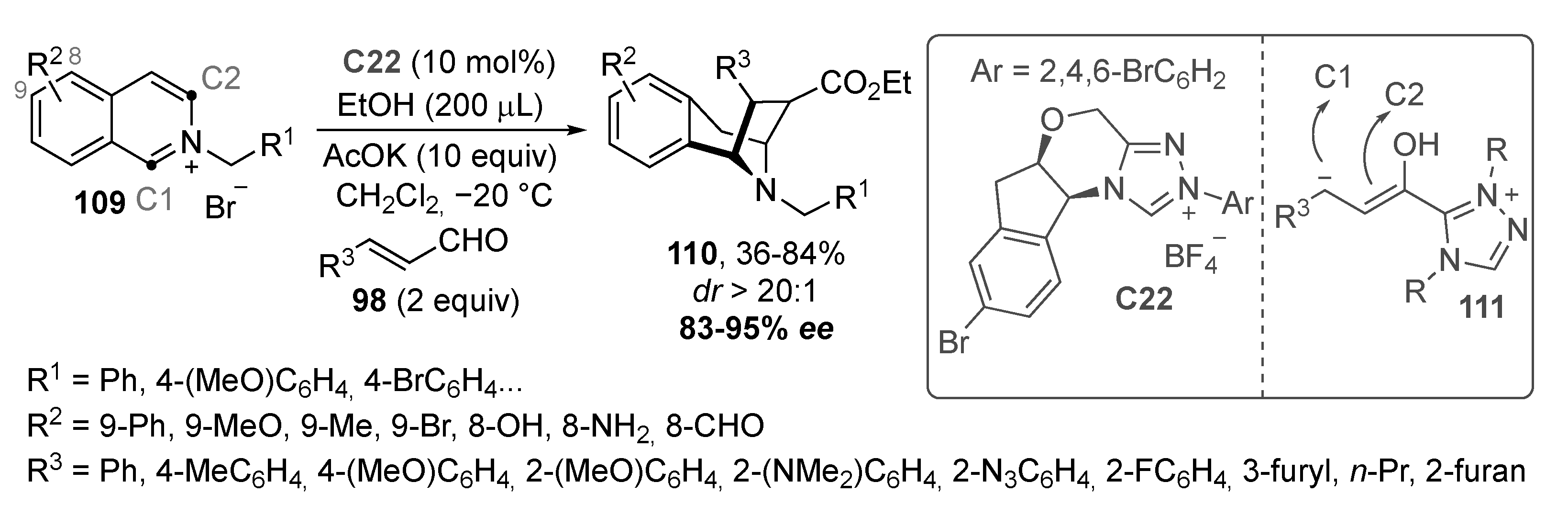
- McLaughlin, C.; Bitai, J.; Barber, L.J.; Slawin, A.M.; Smith, A.D. Catalytic enantioselective synthesis of 1,4-dihydropyridines via the addition of C(1)-ammonium enolates to pyridinium salts. Chem. Sci. 2021, 12, 12001–12011. [Google Scholar] [CrossRef]
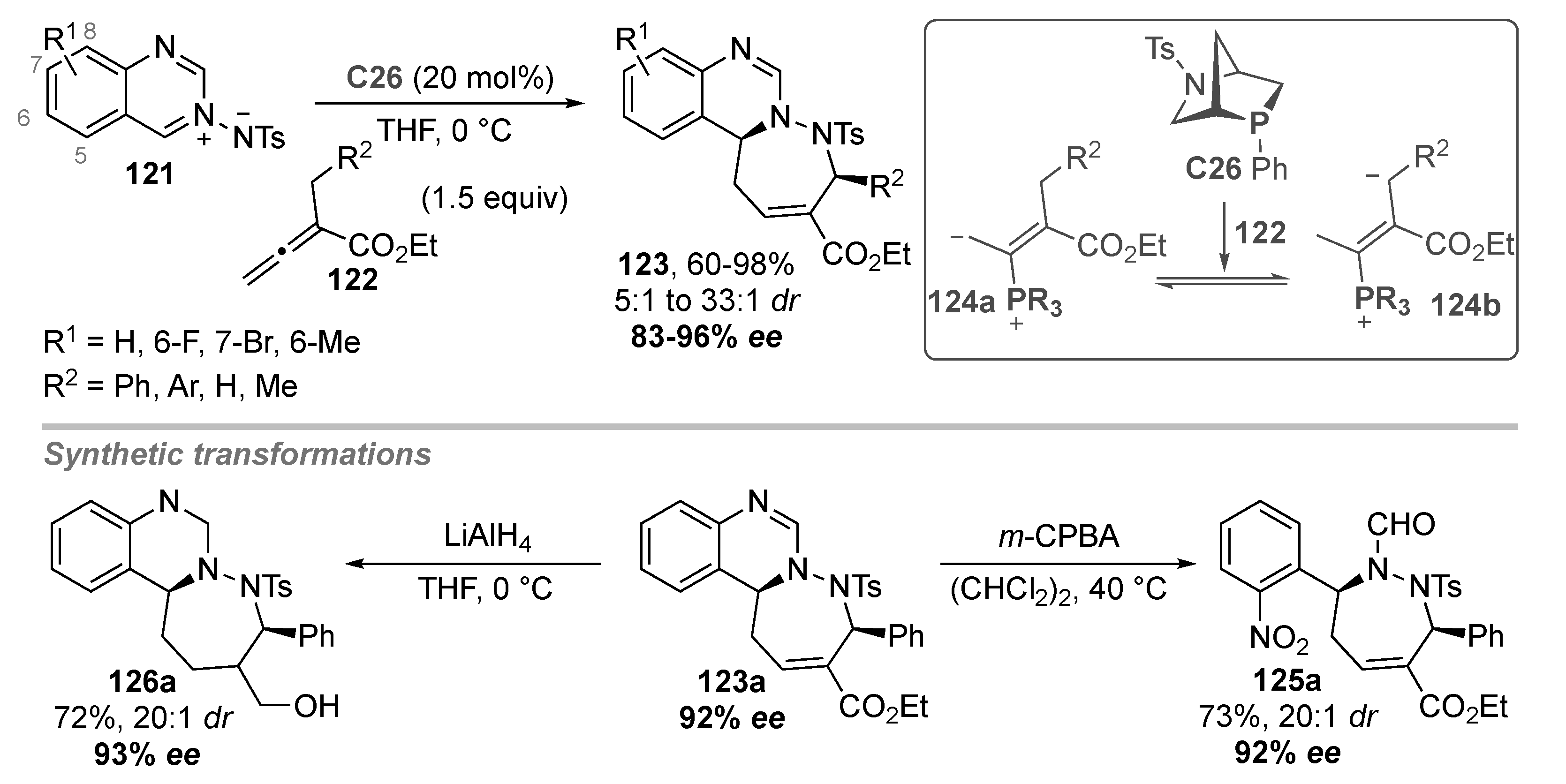
- Yuan, C.; Zhou, L.; Xia, M.; Sun, Z.; Wang, D.; Guo, H. Phosphine-Catalyzed Enantioselective [4 + 3] Annulation of Allenoates with C,N-Cyclic Azomethine Imines: Synthesis of Quinazoline-Based Tricyclic Heterocycles. Org. Lett. 2016, 18, 5644–5647. [Google Scholar] [CrossRef] [PubMed]
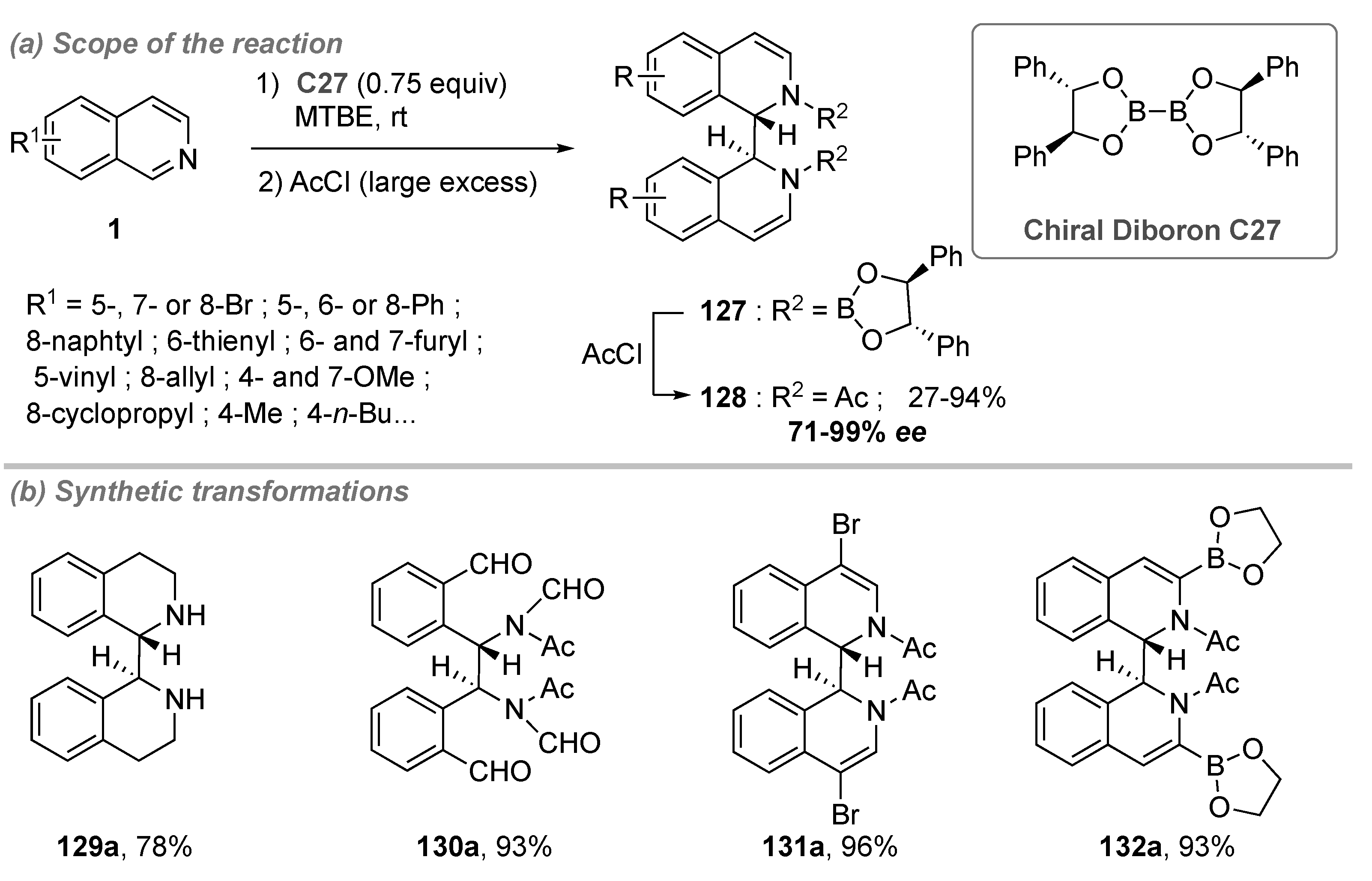
- Chen, D.; Xu, G.; Zhou, Q.; Chung, L.W.; Tang, W. Practical and Asymmetric Reductive Coupling of Isoquinolines Templated by Chiral Diborons. J. Am. Chem. Soc. 2017, 139, 9767–9770. [Google Scholar] [CrossRef]













Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Segovia, C.; Nocquet, P.-A.; Levacher, V.; Brière, J.-F.; Oudeyer, S. Organocatalysis: A Tool of Choice for the Enantioselective Nucleophilic Dearomatization of Electron-Deficient Six-Membered Ring Azaarenium Salts. Catalysts 2021, 11, 1249. https://doi.org/10.3390/catal11101249
Segovia C, Nocquet P-A, Levacher V, Brière J-F, Oudeyer S. Organocatalysis: A Tool of Choice for the Enantioselective Nucleophilic Dearomatization of Electron-Deficient Six-Membered Ring Azaarenium Salts. Catalysts. 2021; 11(10):1249. https://doi.org/10.3390/catal11101249
Chicago/Turabian StyleSegovia, Claire, Pierre-Antoine Nocquet, Vincent Levacher, Jean-François Brière, and Sylvain Oudeyer. 2021. "Organocatalysis: A Tool of Choice for the Enantioselective Nucleophilic Dearomatization of Electron-Deficient Six-Membered Ring Azaarenium Salts" Catalysts 11, no. 10: 1249. https://doi.org/10.3390/catal11101249
APA StyleSegovia, C., Nocquet, P.-A., Levacher, V., Brière, J.-F., & Oudeyer, S. (2021). Organocatalysis: A Tool of Choice for the Enantioselective Nucleophilic Dearomatization of Electron-Deficient Six-Membered Ring Azaarenium Salts. Catalysts, 11(10), 1249. https://doi.org/10.3390/catal11101249








