The Application of Catalytic Processes on the Production of Algae-Based Biofuels: A Review
Abstract
1. Introduction
2. Algae-Based Biofuels
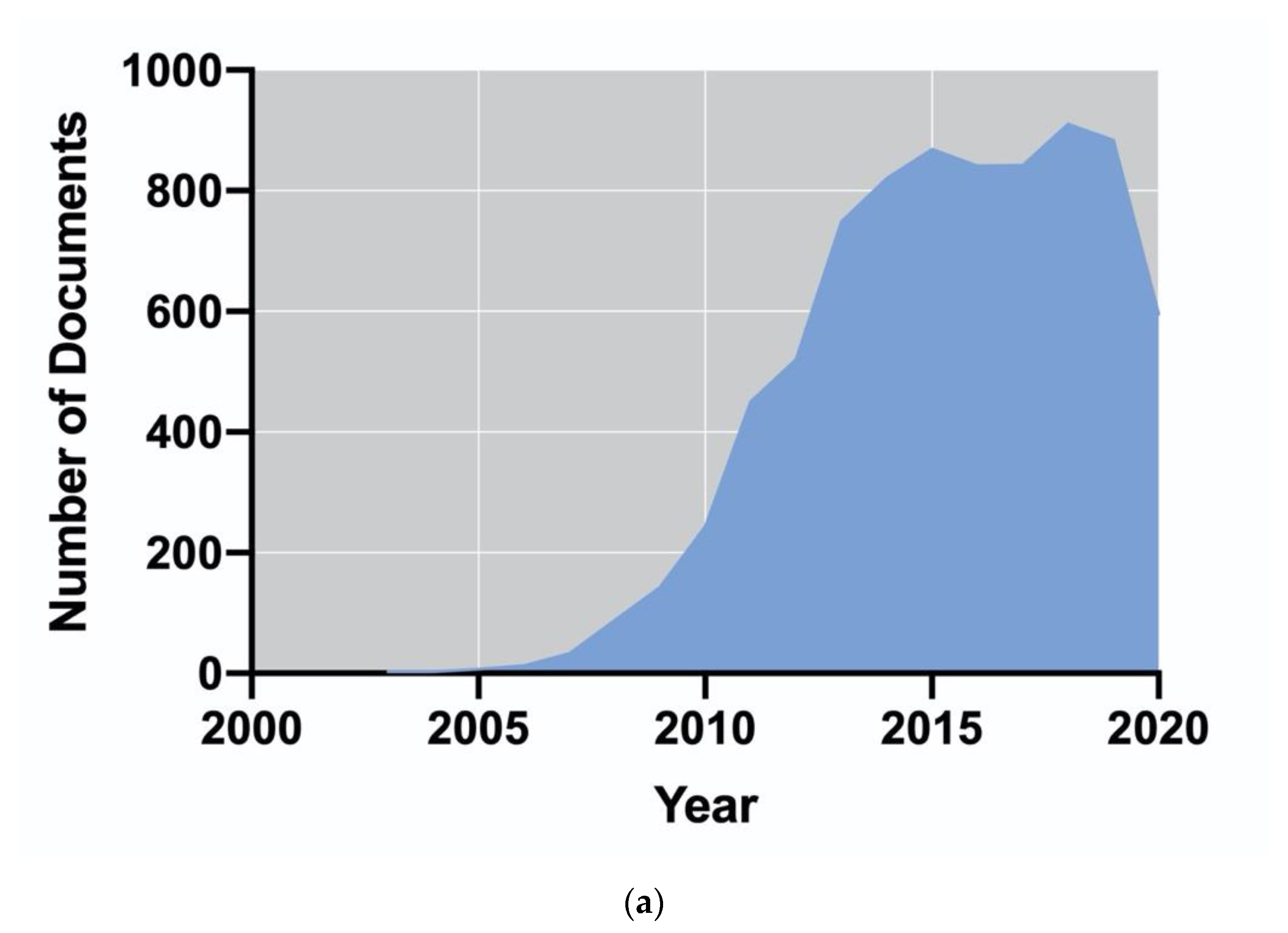
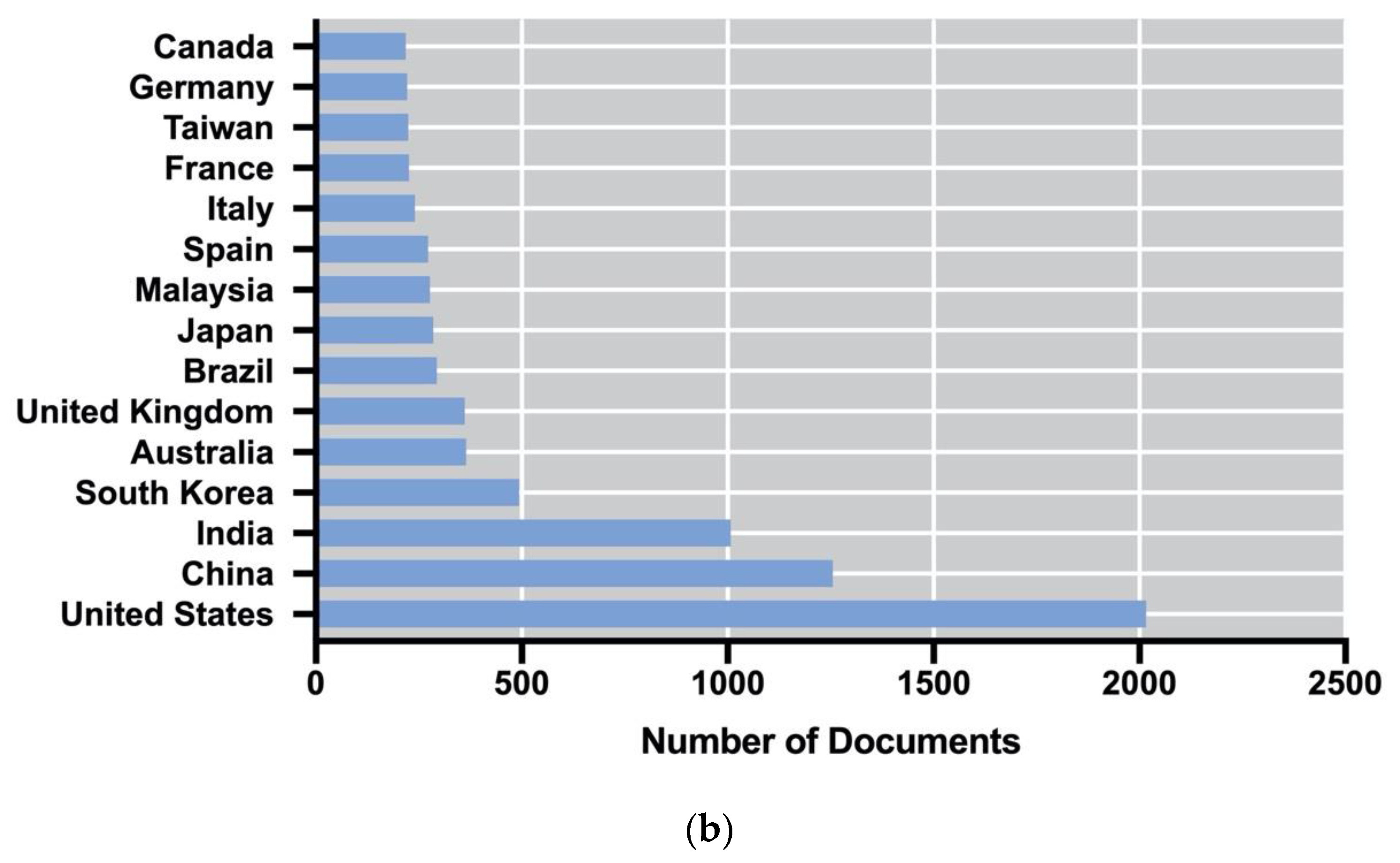
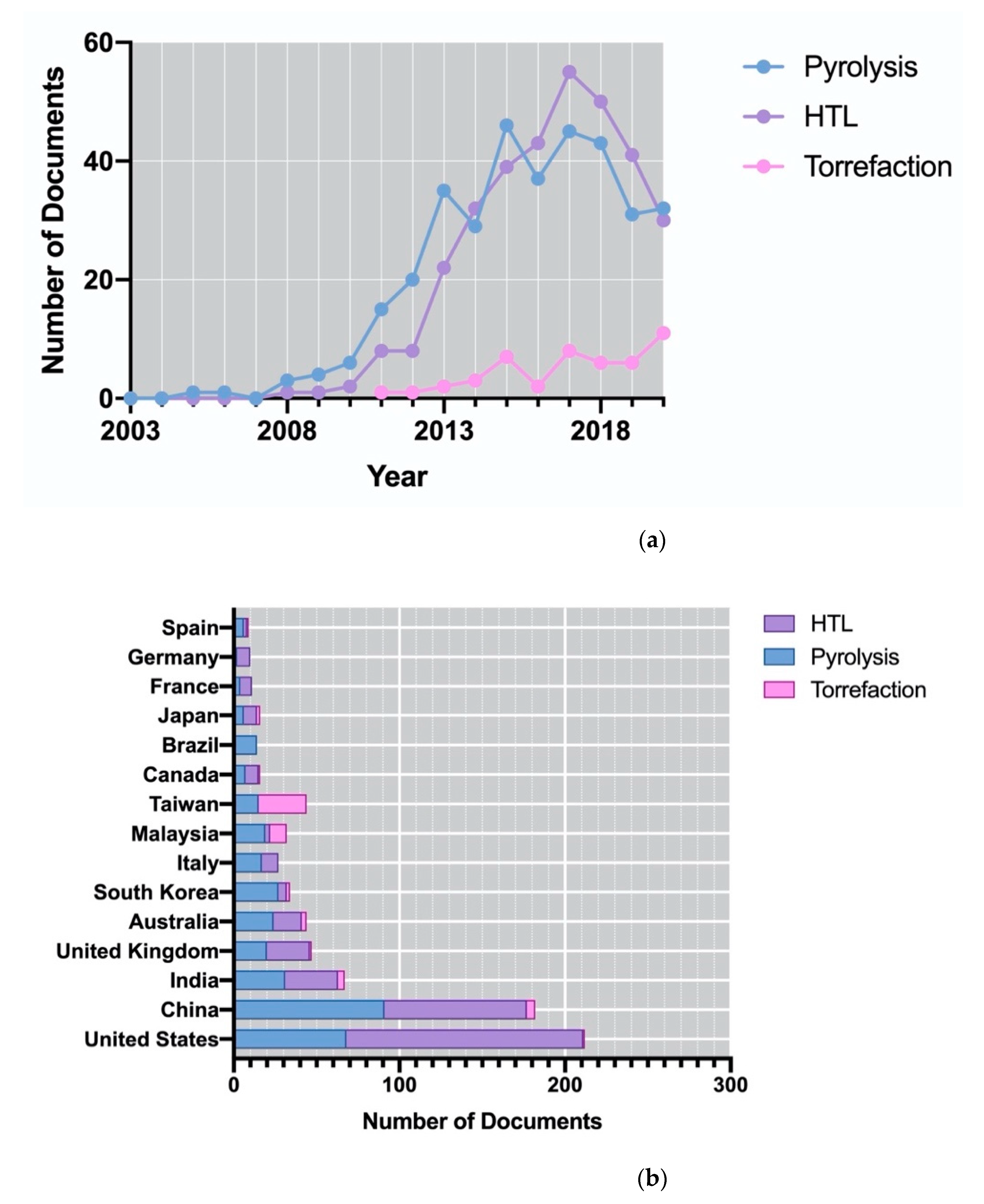
2.1. How the Production of Algae-Based Biofuels Changed over Time
2.2. Biochemical Conversion for Third-Generation Biofuel
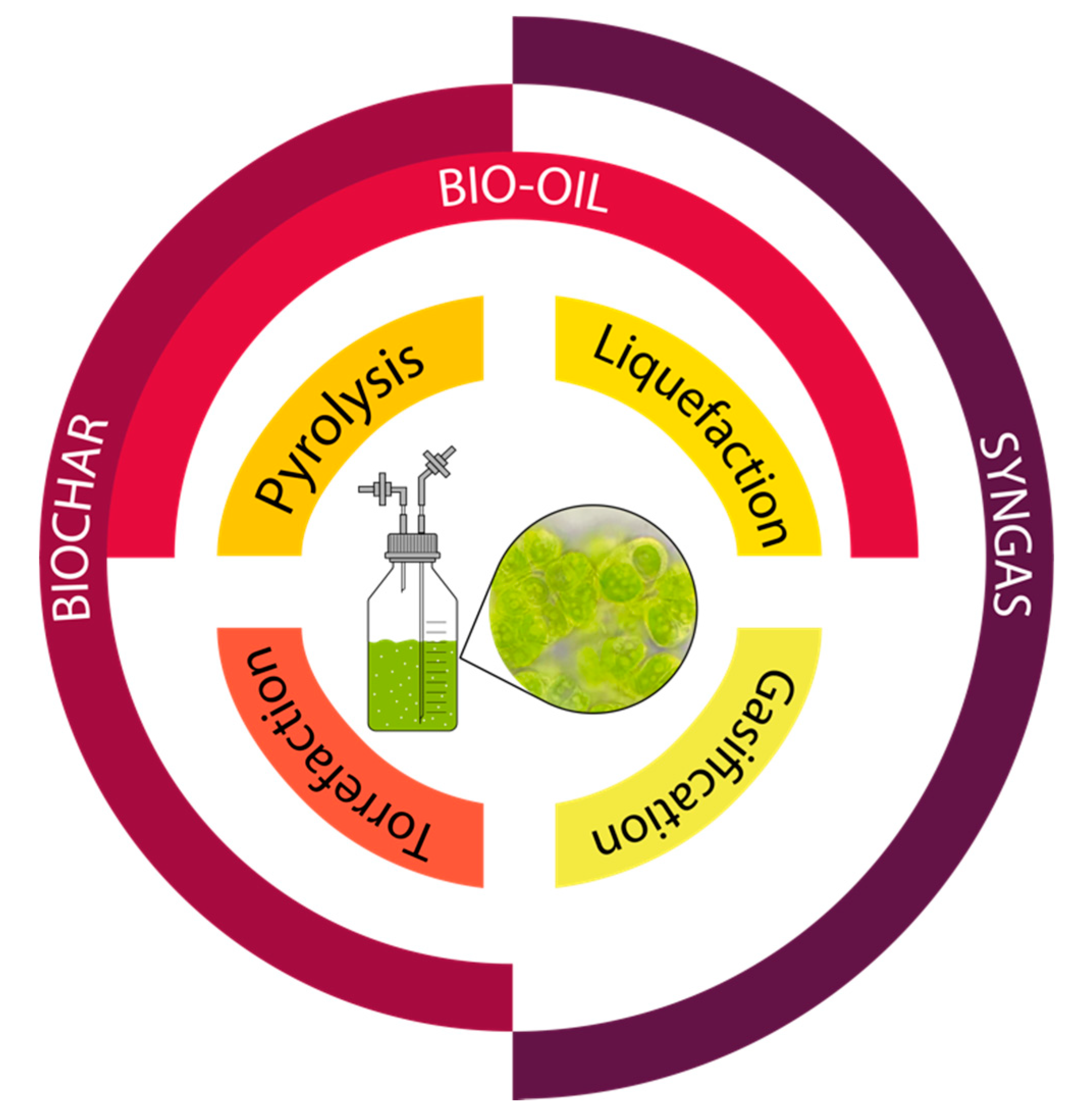
3. Thermochemical Conversion of Algal Biomass
3.1. Microalgal Torrefaction
3.2. Pyrolysis of Microalgae
3.3. Hydrothermal Liquefaction of Algal Biomass
3.3.1. Homogeneous Catalysis
3.3.2. Heterogeneous Catalysis
| Strain | HHV Biomass (MJ/kg) | Catalyst | Temperature (°C) | Residence Time (min) | Catalyst Type | Bio-oil Yield (wt%) | HHV Bio-oil (MJ/kg) | Reference |
|---|---|---|---|---|---|---|---|---|
| Chlorella vulgaris | 23.2 | HCOOH | 320 | 30 | homogeneous catalysts | 28 | 33.2 | [151] |
| Na2CO3 | 28 | 37.1 | ||||||
| 23.2 | 300–350 | 60 | 27.3 | 37.2 | [152] | |||
| KOH | 22.4 | 35.7 | ||||||
| CH3COOH | 20.4 | 34.1 | ||||||
| HCOOH | 19.1 | 34.7 | ||||||
| n/a | HCOOH | 275 | 50 | 29.39 | 36.03 | [153] | ||
| Na2CO3 | 12.5 | 31.8 | ||||||
| C. pyrenoidosa | n/a | NaOH | 240–280 | 20–50 | 41–47.5 | n/a | [154] | |
| Na2CO3 | 31–45 | |||||||
| Cyanidioschyzon merolae | 18.11 | CH3COOH | 300 | 30 | 21.23 | 33.36 | [155] | |
| NaOH | 21.78 | 32.89 | ||||||
| KOH | 22.67 | 33.6 | ||||||
| H2SO4 | 16.98 | 33.76 | ||||||
| Dunaliella tertiolecta | n/a | Na2CO3 | 340 | 60 | 42.0 | n/a | [156] | |
| Enteromorpha prolifera | 13.4 | 290 | 20 | 23.0 | 29.5 | [157] | ||
| Isochrysis sp. | 22.97 | 300 | 60 | 42.5 | 35.61 | [158] | ||
| Laminaria saccharina | 14.46 | KOH | 350 | 15 | 63 | 34.18 | [159] | |
| Microcystis viridis | n/a | Na2CO3 | 300–340 | 30–60 | 33 | 28–30 | [160] | |
| Nannochloropsis sp. | 24.02 | 350 | 60 | 48.67 | 33.71 | [158] | ||
| 17.9 | HCOOH | 320 | 30 | 28 | 39 | [151] | ||
| Na2CO3 | 28 | 35.5 | ||||||
| 23.88 | 250 | 60 | 24.2 | 38.31 | [161] | |||
| Pavlova sp. | 22.69 | 350 | 60 | 47.7 | 36.93 | [158] | ||
| Porphyridium cruentum | 14.7 | 320 | 30 | 27.1 | 22.8 | [151] | ||
| Spirulina sp. | 21.2 | KOH | 300–350 | 60 | 15.2 | 35.7 | [152] | |
| Na2CO3 | 20 | 37.8 | ||||||
| CH3COOH | 16.6 | 34.1 | ||||||
| HCOOH | 14.2 | 34.7 | ||||||
| S. platensis | 20.52 | Ca3(PO4)2 | 350 | 60 | 34.5 | 35.07 | [162] | |
| NiO | 30.2 | 38.41 | ||||||
| Na2CO3 | 51.6 | 36.29 | ||||||
| n/a | Na2CO3 | 250–350 | 30 | 35 | 38.65 | [163] | ||
| Tetraselmis sp. | 40 | 35.58 | ||||||
| Ulva prolifera | KOH | 290 | 30 | 26.7 | 33.6 | [164] | ||
| NaOH | 25.2 | 29.8 | ||||||
| Na2CO3 | 19 | 29.2 | ||||||
| Green macroalgal blooms | 9.45 | 270 | 45 | 20.1 | 25.59 | [165] | ||
| CaO | 14.6 | 23.8 | ||||||
| TiO2 | 17.3 | 25.37 | ||||||
| Chlorella sp. | n/a | CuO/Al-SBA-15 | 170–350 | 30 | heterogeneous catalysts | 45.1 | n/a | [168] |
| 17.31 | Pt/C | 350 | 30 | 37.9 | 33.2 | [169] | ||
| C. pyrenoidosa | n/a | HZSM-5 | 250–300 | 60 | 73 | n/a | [170] | |
| NaY | 68 | |||||||
| USY | 66 | |||||||
| HY | 64 | |||||||
| Ce/H-ZSM−5 | 300 | 20 | 49.87 | 26.09 | [171] | |||
| HZSM-5 | 34.02 | 21.77 | ||||||
| Pd/Al2O3 | 240–280 | 30 | 27.5–48 | n/a | [154] | |||
| Pd/C | 30–42.5 | |||||||
| Pt/Al2O3 | 34–46 | |||||||
| Pt/C | 33–45 | |||||||
| Raney Ni | 33–50 | |||||||
| C. vulgaris | Pt/Al2O3 | 350 | 60 | 38.9 | n/a | [168] | ||
| Ni/Al2O3 | 30 | |||||||
| Co/Mo/Al2O3 | 38.7 | |||||||
| D. tertiolecta | 17.81 | KtB | 360 | 30 | 49.09 | 32.36 | [172] | |
| ZrO2/SO42- | 29 | 33.24 | ||||||
| HZSM-5 | 31.1 | 33.67 | ||||||
| MgO/MCM-41 | 36 | 33.17 | ||||||
| D. salina | 18.47 | Ni/REHY | 200 | 60 | 72 | 30.11 | [173] | |
| REHY | 51.6 | 26.88 | ||||||
| N. oculata | n/a | Pt/Al2O3 | 350 | 60 | 30.2 | n/a | [168] | |
| Ni/Al2O3 | 18.1 | |||||||
| Co/Mo/Al2O3 | 25.5 | |||||||
| Nannochloropsis sp. | 18.5 | Pd/C | 350 | 60 | 57 | 38.9 | [174] | |
| Pt/C | 49 | 40.1 | ||||||
| Ru/C | 50 | 38.4 | ||||||
| Ni/SiO2-Al2O3 | 50 | 39.4 | ||||||
| CoMo/Al2O3 | 55 | 38.6 | ||||||
| Zeolite | 48 | 38.5 | ||||||
| n/a | Pd/C | 350 | 60 | 48 | n/a | [175] | ||
| Fe/HZSM-5 | 365 | 60 | 38.1 | n/a | [176] | |||
| Spirulina sp. | HZSM-5 | 380 | 120 | 30.63 | 28.32 | [177] | ||
| HZSM-5@MS | 32.45 | 29.51 | ||||||
| Pd/HZSM-5 | 34.9 | 29.43 | ||||||
| Pd/HZSM-5@MS | 35.62 | 29.21 | ||||||
| S. platensis | CeO2 | 250 | 30 | 34 | 39.21 | [178] | ||
| Fe3O4 | 272 | 37 | 27.6 | 30.98 | [179] | |||
| Ulva prolifera | ZSM-5 | 280 | 15 | 29.3 | 34.8 | [180] | ||
| Microalgae consortium | H-ZSM−5 | 350 | 120 | 16.0 | 37.7–41.6 | [181] |
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Kosmela:, P.; Kazimierski, P.; Formela, K.; Haponiuk, J.; Piszczyk, Ł. Liquefaction of Macroalgae Enteromorpha Biomass for the Preparation of Biopolyols by Using Crude Glycerol. J. Ind. Eng. Chem. 2017, 56, 399–406. [Google Scholar] [CrossRef]
- Chowdhury, H.; Loganathan, B. Third-Generation Biofuels from Microalgae: A Review. Curr. Opin. Green Sustain. Chem. 2019, 20, 39–44. [Google Scholar] [CrossRef]
- Garcia-Moscoso, J.L.; Obeid, W.; Kumar, S.; Hatcher, P.G. Flash Hydrolysis of Microalgae (Scenedesmus sp.) for Protein Extraction and Production of Biofuels Intermediates. J. Supercrit. Fluids 2013, 82, 183–190. [Google Scholar] [CrossRef]
- Ansah, E.; Wang, L.; Zhang, B.; Shahbazi, A. Catalytic Pyrolysis of Raw and Hydrothermally Carbonized Chlamydomonas Debaryana Microalgae for Denitrogenation and Production of Aromatic Hydrocarbons. Fuel 2018, 228, 234–242. [Google Scholar] [CrossRef]
- Costa, J.A.V.; de Freitas, B.C.B.; Lisboa, C.R.; Santos, T.D.; de Fraga Brusch, L.R.; de Morais, M.G. Microalgal Biorefinery from CO2 and the Effects under the Blue Economy. Renew. Sustain. Energy Rev. 2019, 99, 58–65. [Google Scholar] [CrossRef]
- Quintero-Dallos, V.; García-Martínez, J.B.; Contreras-Ropero, J.E.; Barajas-Solano, A.F.; Barajas-Ferrerira, C.; Lavecchia, R.; Zuorro, A. Vinasse as a Sustainable Medium for the Production of Chlorella vulgaris UTEX 1803. Water 2019, 11, 1526. [Google Scholar] [CrossRef]
- Lu, W.; Asraful Alam, M.; Liu, S.; Xu, J.; Parra Saldivar, R. Critical Processes and Variables in Microalgae Biomass Production Coupled with Bioremediation of Nutrients and CO2 from Livestock Farms: A Review. Sci. Total Environ. 2020, 716, 135247. [Google Scholar] [CrossRef]
- Zuorro, A.; Lavecchia, R.; Maffei, G.; Marra, F.; Miglietta, S.; Petrangeli, A.; Familiari, G.; Valente, T. Enhanced lipid extraction from unbroken microalgal cells using enzymes. Chem. Eng. Trans. 2015, 43, 211–216. [Google Scholar] [CrossRef]
- Ananthi, V.; Brindhadevi, K.; Pugazhendhi, A.; Arun, A. Impact of Abiotic Factors on Biodiesel Production by Microalgae. Fuel 2021, 284, 118962. [Google Scholar] [CrossRef]
- Castellanos-Estupiñan, M.; Sanchez-Galvis, M.; Garcia-Martinez, J.B.; Barajas-Ferreira, C.; Zuorro, A.; Barajas-Solano, A.F. Design of an Electroflotation System for the Concentration and Harvesting of Freshwater Microalgae. Chem. Eng. Trans. 2018, 64, 1–6. [Google Scholar] [CrossRef]
- Zabed, H.M.; Akter, S.; Yun, J.; Zhang, G.; Zhang, Y.; Qi, X. Biogas from Microalgae: Technologies, Challenges and Opportunities. Renew. Sustain. Energy Rev. 2020, 117, 109503. [Google Scholar] [CrossRef]
- Raheem, A.; Wan Azlina, W.A.K.G.; Taufiq Yap, Y.H.; Danquah, M.K.; Harun, R. Thermochemical Conversion of Microalgal Biomass for Biofuel Production. Renew. Sustain. Energy Rev. 2015, 49, 990–999. [Google Scholar] [CrossRef]
- Chen, W.-H.; Lin, B.-J.; Huang, M.-Y.; Chang, J.-S. Thermochemical Conversion of Microalgal Biomass into Biofuels: A Review. Bioresour. Technol. 2015, 184, 314–327. [Google Scholar] [CrossRef] [PubMed]
- Ong, H.C.; Chen, W.-H.; Farooq, A.; Gan, Y.Y.; Lee, K.T.; Ashokkumar, V. Catalytic Thermochemical Conversion of Biomass for Biofuel Production: A Comprehensive Review. Renew. Sustain. Energy Rev. 2019, 113, 109266. [Google Scholar] [CrossRef]
- Kargbo, H.; Harris, J.S.; Phan, A.N. “Drop-in” Fuel Production from Biomass: Critical Review on Techno-Economic Feasibility and Sustainability. Renew. Sustain. Energy Rev. 2021, 135, 110168. [Google Scholar] [CrossRef]
- Huang, C.-H.; Tan, C.-S. A Review: CO2 Utilization. Aerosol Air Qual. Res. 2014, 14, 480–499. [Google Scholar] [CrossRef]
- Hena, S.; Znad, H.; Heong, K.T.; Judd, S. Dairy Farm Wastewater Treatment and Lipid Accumulation by Arthrospira platensis. Water Res. 2018, 128, 267–277. [Google Scholar] [CrossRef]
- Polat, E.; Yüksel, E.; Altınbaş, M. Mutual Effect of Sodium and Magnesium on the Cultivation of Microalgae Auxenochlorella protothecoides. Biomass Bioenergy 2020, 132, 105441. [Google Scholar] [CrossRef]
- Gouveia, J.D.; Ruiz, J.; van den Broek, L.A.M.; Hesselink, T.; Peters, S.; Kleinegris, D.M.M.; Smith, A.G.; van der Veen, D.; Barbosa, M.J.; Wijffels, R.H. Botryococcus braunii Strains Compared for Biomass Productivity, Hydrocarbon and Carbohydrate Content. J. Biotechnol. 2017, 248, 77–86. [Google Scholar] [CrossRef]
- Barajas-Solano, A.F.; Guzmán-Monsalve, A.; Kafarov, V. Effect of Carbon-Nitrogen Ratio for the Biomass Production, Hydrocarbons and Lipids on Botryoccus braunii UIS 003. Chem. Eng. Trans. 2016, 49, 247–252. [Google Scholar] [CrossRef]
- Banerjee, S.; Ray, A.; Das, D. Optimization of Chlamydomonas reinhardtii Cultivation with Simultaneous CO2 Sequestration and Biofuels Production in a Biorefinery Framework. Sci. Total Environ. 2020, 143080. [Google Scholar] [CrossRef] [PubMed]
- Kao, P.-H.; Ng, I.-S. CRISPRi Mediated Phosphoenolpyruvate Carboxylase Regulation to Enhance the Production of Lipid in Chlamydomonas reinhardtii. Bioresour. Technol. 2017, 245, 1527–1537. [Google Scholar] [CrossRef] [PubMed]
- Shin, Y.S.; Jeong, J.; Nguyen, T.H.T.; Kim, J.Y.H.; Jin, E.; Sim, S.J. Targeted Knockout of Phospholipase A2 to Increase Lipid Productivity in Chlamydomonas reinhardtii for Biodiesel Production. Bioresour. Technol. 2019, 271, 368–374. [Google Scholar] [CrossRef] [PubMed]
- Gao, F.; Yang, H.-L.; Li, C.; Peng, Y.-Y.; Lu, M.-M.; Jin, W.-H.; Bao, J.-J.; Guo, Y.-M. Effect of Organic Carbon to Nitrogen Ratio in Wastewater on Growth, Nutrient Uptake and Lipid Accumulation of a Mixotrophic Microalgae Chlorella sp. Bioresour. Technol. 2019, 282, 118–124. [Google Scholar] [CrossRef]
- Bauer, L.M.; Costa, J.A.V.; da Rosa, A.P.C.; Santos, L.O. Growth Stimulation and Synthesis of Lipids, Pigments and Antioxidants with Magnetic Fields in Chlorella kessleri Cultivations. Bioresour. Technol. 2017, 244, 1425–1432. [Google Scholar] [CrossRef]
- Cheng, P.; Chu, R.; Zhang, X.; Song, L.; Chen, D.; Zhou, C.; Yan, X.; Cheng, J.J.; Ruan, R. Screening of the Dominant Chlorella pyrenoidosa for Biofilm Attached Culture and Feed Production While Treating Swine Wastewater. Bioresour. Technol. 2020, 318, 124054. [Google Scholar] [CrossRef]
- Alavijeh, R.S.; Karimi, K.; Wijffels, R.H.; van den Berg, C.; Eppink, M. Combined Bead Milling and Enzymatic Hydrolysis for Efficient Fractionation of Lipids, Proteins, and Carbohydrates of Chlorella vulgaris Microalgae. Bioresour. Technol. 2020, 309, 123321. [Google Scholar] [CrossRef]
- Estévez-Landazábal, L.L.; Barajas-Solano, A.F.; Barajas-Ferreira, C.; Kafarov, V. Improvement of lipid productivity on Chlorella vulgaris using waste glycerol and sodium acetate. CTF Cienc. Tecnol. Futuro 2013, 5, 113–126. Available online: http://www.scielo.org.co/scielo.php?script=sci_arttext&pid=S0122-53832013000100009 (accessed on 29 November 2020).
- Sarayloo, E.; Simsek, S.; Unlu, Y.S.; Cevahir, G.; Erkey, C.; Kavakli, I.H. Enhancement of the Lipid Productivity and Fatty Acid Methyl Ester Profile of Chlorella vulgaris by Two Rounds of Mutagenesis. Bioresour. Technol. 2018, 250, 764–769. [Google Scholar] [CrossRef]
- Del Río, E.; García-Gómez, E.; Moreno, J.; Guerrero, M.G.; García-González, M. Microalgae for Oil. Assessment of Fatty Acid Productivity in Continuous Culture by Two High-Yield Strains, Chlorococcum oleofaciens and Pseudokirchneriella subcapitata. Algal Res. 2017, 23, 37–42. [Google Scholar] [CrossRef]
- Chinnasamy, S.; Bhatnagar, A.; Hunt, R.W.; Das, K.C. Microalgae Cultivation in a Wastewater Dominated by Carpet Mill Effluents for Biofuel Applications. Bioresour. Technol. 2010, 101, 3097–3105. [Google Scholar] [CrossRef] [PubMed]
- Sanchez-Silva, L.; López-González, D.; Garcia-Minguillan, A.M.; Valverde, J.L. Pyrolysis, Combustion and Gasification Characteristics of Nannochloropsis gaditana Microalgae. Bioresour. Technol. 2013, 130, 321–331. [Google Scholar] [CrossRef] [PubMed]
- Xue, J.; Balamurugan, S.; Li, D.-W.; Liu, Y.-H.; Zeng, H.; Wang, L.; Yang, W.-D.; Liu, J.-S.; Li, H.-Y. Glucose-6-Phosphate Dehydrogenase as a Target for Highly Efficient Fatty Acid Biosynthesis in Microalgae by Enhancing NADPH Supply. Metab. Eng. 2017, 41, 212–221. [Google Scholar] [CrossRef] [PubMed]
- Priharto, N.; Ronsse, F.; Prins, W.; Carleer, R.; Heeres, H.J. Experimental Studies on a Two-Step Fast Pyrolysis-Catalytic Hydrotreatment Process for Hydrocarbons from Microalgae (Nannochloropsis gaditana and Scenedesmus almeriensis). Fuel Process. Technol. 2020, 206, 106466. [Google Scholar] [CrossRef]
- Gupta, S.; Pawar, S.B. An Integrated Approach for Microalgae Cultivation Using Raw and Anaerobic Digested Wastewaters from Food Processing Industry. Bioresour. Technol. 2018, 269, 571–576. [Google Scholar] [CrossRef]
- Girard, J.-M.; Roy, M.-L.; Hafsa, M.B.; Gagnon, J.; Faucheux, N.; Heitz, M.; Tremblay, R.; Deschênes, J.-S. Mixotrophic Cultivation of Green Microalgae Scenedesmus Obliquus on Cheese Whey Permeate for Biodiesel Production. Algal Res. 2014, 5, 241–248. [Google Scholar] [CrossRef]
- Cuéllar-García, D.J.; Rangel-Basto, Y.A.; Urbina-Suarez, N.A.; Barajas-Solano, A.F.; Muñoz-Peñaloza, Y.A. Lipids production from Scenedesmus obliquus through carbon/nitrogen ratio optimization. J. Phys. Conf. Ser. 2019, 1388, 012043. [Google Scholar] [CrossRef]
- Cuéllar-García, D.J.; Rangel-Basto, Y.A.; Barajas-Solano, A.F.; Muñoz-Peñaloza, Y.A.; Urbina-Suarez, N.A. Towards the production of microalgae biofuels: The effect of the culture medium on lipid deposition. BioTechnologia 2019, 100, 273–278. [Google Scholar] [CrossRef]
- Andreotti, V.; Solimeno, A.; Rossi, S.; Ficara, E.; Marazzi, F.; Mezzanotte, V.; García, J. Bioremediation of Aquaculture Wastewater with the Microalgae Tetraselmis suecica: Semi-Continuous Experiments, Simulation and Photo-Respirometric Tests. Sci. Total Environ. 2020, 738, 139859. [Google Scholar] [CrossRef]
- Srivatsa, S.C.; Li, F.; Bhattacharya, S. Optimization of Reaction Parameters for Bio-Oil Production by Catalytic Pyrolysis of Microalga Tetraselmis suecica: Influence of Ni-Loading on the Bio-Oil Composition. Renew. Energy 2019, 142, 426–436. [Google Scholar] [CrossRef]
- Guiza-Franco, L.; Orozco-Rojas, L.G.; Sanchez-Galvis, M.; Garcia-Martinez, J.B.; Barajas-Ferreira, C.; Zuorro, A.; Barajas-Solano, A.F. Production of Chlorella vulgaris Biomass on UV-Treated Wastewater as an Alternative for Environmental Sustainability on High-Mountain Fisheries. Chem. Eng. Trans. 2018, 64, 517–522. [Google Scholar] [CrossRef]
- Merchuk, J.C. Chapter 5—Photobioreactor Design; Jacob-Lopes, E., Maroneze, M.M., Queiroz, M.I., Zepka, L.Q., Eds.; Academic Press: Cambridge, MA, USA, 2020; pp. 101–126. [Google Scholar] [CrossRef]
- Sanchez-Galvis, E.M.; Cardenas-Gutierrez, I.Y.; Contreras-Ropero, J.E.; García-Martínez, J.B.; Barajas-Solano, A.F.; Zuorro, A. An Innovative Low-Cost Equipment for Electro-Concentration of Microalgal Biomass. Appl. Sci. 2020, 10, 4841. [Google Scholar] [CrossRef]
- Garcia-Martinez, B.; Ayala-Torres, E.; Reyes-Gomez, O.; Zuorro, A.; Barajas-Solano, A.; Barajas-Ferreira, C. Evaluation of a Two-Phase Extraction System of Carbohydrates and Proteins from Chlorella vulgaris UTEX 1803. Chem. Eng. Trans. 2016, 49, 355–360. [Google Scholar] [CrossRef]
- Kumar, R.; Ghosh, A.K.; Pal, P. Synergy of Biofuel Production with Waste Remediation along with Value-Added Co-Products Recovery through Microalgae Cultivation: A Review of Membrane-Integrated Green Approach. Sci. Total Environ. 2020, 698, 134169. [Google Scholar] [CrossRef] [PubMed]
- Zuorro, A.; Malavasi, V.; Cao, G.; Lavecchia, R. Use of cell wall degrading enzymes to improve the recovery of lipids from Chlorella sorokiniana. Chem. Eng. J. 2019, 377, 120325. [Google Scholar] [CrossRef]
- Arun, J.; Gopinath, K.P.; SundarRajan, P.; Felix, V.; JoselynMonica, M.; Malolan, R. A Conceptual Review on Microalgae Biorefinery through Thermochemical and Biological Pathways: Bio-Circular Approach on Carbon Capture and Wastewater Treatment. Bioresour. Technol. Rep. 2020, 11, 100477. [Google Scholar] [CrossRef]
- Rangel-Basto, Y.A.; García-Ochoa, I.E.; Suarez-Gelvez, J.H.; Zuorro, A.; Barajas-Solano, A.F.; Urbina-Suarez, N.A. The Effect of Temperature and Enzyme Concentration in the Transesterification Process of Synthetic Microalgae Oil. Chem. Eng. Trans. 2018, 64, 331–336. [Google Scholar] [CrossRef]
- Kumar, M.; Sun, Y.; Rathour, R.; Pandey, A.; Thakur, I.S.; Tsang, D.C.W. Algae as Potential Feedstock for the Production of Biofuels and Value-Added Products: Opportunities and Challenges. Sci. Total Environ. 2020, 716, 137116. [Google Scholar] [CrossRef]
- Demirbas, A. Use of Algae as Biofuel Sources. Energy Convers. Manag. 2010, 51, 2738–2749. [Google Scholar] [CrossRef]
- Tripathi, R.; Singh, J.; Thakur, I.S. Characterization of Microalga Scenedesmus sp. ISTGA1 for Potential CO2 Sequestration and Biodiesel Production. Renew. Energy 2015, 74, 774–781. [Google Scholar] [CrossRef]
- Kumar, M.; Thakur, I.S. Municipal Secondary Sludge as Carbon Source for Production and Characterization of Biodiesel from Oleaginous Bacteria. Bioresour. Technol. Rep. 2018, 4, 106–113. [Google Scholar] [CrossRef]
- De Farias Silva, C.E.; Bertucco, A. Bioethanol from Microalgae and Cyanobacteria: A Review and Technological Outlook. Process Biochem. 2016, 51, 1833–1842. [Google Scholar] [CrossRef]
- Dragone, G.; Fernandes, B.D.; Abreu, A.P.; Vicente, A.A.; Teixeira, J.A. Nutrient Limitation as a Strategy for Increasing Starch Accumulation in Microalgae. Appl. Energy 2011, 88, 3331–3335. [Google Scholar] [CrossRef]
- Ho, S.-H.; Huang, S.-W.; Chen, C.-Y.; Hasunuma, T.; Kondo, A.; Chang, J.-S. Bioethanol Production Using Carbohydrate-Rich Microalgae Biomass as Feedstock. Bioresour. Technol. 2013, 135, 191–198. [Google Scholar] [CrossRef] [PubMed]
- El-Dalatony, M.M.; Kurade, M.B.; Abou-Shanab, R.A.I.; Kim, H.; Salama, E.-S.; Jeon, B.-H. Long-Term Production of Bioethanol in Repeated-Batch Fermentation of Microalgal Biomass Using Immobilized Saccharomyces cerevisiae. Bioresour. Technol. 2016, 219, 98–105. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.-Y.; Zhao, X.-Q.; Yen, H.-W.; Ho, S.-H.; Cheng, C.-L.; Lee, D.-J.; Bai, F.-W.; Chang, J.-S. Microalgae-Based Carbohydrates for Biofuel Production. Biochem. Eng. J. 2013, 78, 1–10. [Google Scholar] [CrossRef]
- Wang, H.; Ji, C.; Bi, S.; Zhou, P.; Chen, L.; Liu, T. Joint Production of Biodiesel and Bioethanol from Filamentous Oleaginous Microalgae Tribonema sp. Bioresour. Technol. 2014, 172, 169–173. [Google Scholar] [CrossRef]
- Talebnia, F.; Karakashev, D.; Angelidaki, I. Production of Bioethanol from Wheat Straw: An Overview on Pretreatment, Hydrolysis and Fermentation. Bioresour. Technol. 2010, 101, 4744–4753. [Google Scholar] [CrossRef]
- Baeyens, J.; Kang, Q.; Appels, L.; Dewil, R.; Lv, Y.; Tan, T. Challenges and Opportunities in Improving the Production of Bio-Ethanol. Prog. Energy Combust. Sci. 2015, 47, 60–88. [Google Scholar] [CrossRef]
- Hernández, D.; Riaño, B.; Coca, M.; García-González, M.C. Saccharification of Carbohydrates in Microalgal Biomass by Physical, Chemical and Enzymatic Pre-Treatments as a Previous Step for Bioethanol Production. Chem. Eng. J. 2015, 262, 939–945. [Google Scholar] [CrossRef]
- Barajas-Solano, A.F.; Gonzalez-Delgado, A.D.; Kafarov, V. Effect of Thermal Pre-Treatment on Fermentable Sugar Production of Chlorella vulgaris. Chem. Eng. Trans. 2014, 37, 655–660. [Google Scholar] [CrossRef]
- Rizza, L.S.; Smachetti, M.E.S.; Do Nascimento, M.; Salerno, G.L.; Curatti, L. Bioprospecting for Native Microalgae as an Alternative Source of Sugars for the Production of Bioethanol. Algal Res. 2017, 22, 140–147. [Google Scholar] [CrossRef]
- Córdova, O.; Santis, J.; Ruiz-Fillipi, G.; Zuñiga, M.E.; Fermoso, F.G.; Chamy, R. Microalgae Digestive Pretreatment for Increasing Biogas Production. Renew. Sustain. Energy Rev. 2018, 82, 2806–2813. [Google Scholar] [CrossRef]
- Jankowska, E.; Sahu, A.K.; Oleskowicz-Popiel, P. Biogas from Microalgae: Review on Microalgae’s Cultivation, Harvesting and Pretreatment for Anaerobic Digestion. Renew. Sustain. Energy Rev. 2017, 75, 692–709. [Google Scholar] [CrossRef]
- González-Fernández, C.; Sialve, B.; Bernet, N.; Steyer, J.P. Thermal Pretreatment to Improve Methane Production of Scenedesmus Biomass. Biomass Bioenergy 2012, 40, 105–111. [Google Scholar] [CrossRef]
- Sialve, B.; Bernet, N.; Bernard, O. Anaerobic Digestion of Microalgae as a Necessary Step to Make Microalgal Biodiesel Sustainable. Biotechnol. Adv. 2009, 27, 409–416. [Google Scholar] [CrossRef]
- Anwar, M.; Lou, S.; Chen, L.; Li, H.; Hu, Z. Recent Advancement and Strategy on Bio-Hydrogen Production from Photosynthetic Microalgae. Bioresour. Technol. 2019, 292, 121972. [Google Scholar] [CrossRef]
- Jiménez-Llanos, J.; Ramírez-Carmona, M.; Rendón-Castrillón, L.; Ocampo-López, C. Sustainable Biohydrogen Production by Chlorella sp. Microalgae: A Review. Int. J. Hydrogen Energy 2020, 45, 8310–8328. [Google Scholar] [CrossRef]
- Gimpel, J.A.; Specht, E.A.; Georgianna, D.R.; Mayfield, S.P. Advances in Microalgae Engineering and Synthetic Biology Applications for Biofuel Production. Curr. Opin. Chem. Biol. 2013, 17, 489–495. [Google Scholar] [CrossRef]
- Mathimani, T.; Baldinelli, A.; Rajendran, K.; Prabakar, D.; Matheswaran, M.; Pieter van Leeuwen, R.; Pugazhendhi, A. Review on Cultivation and Thermochemical Conversion of Microalgae to Fuels and Chemicals: Process Evaluation and Knowledge Gaps. J. Clean. Prod. 2019, 208, 1053–1064. [Google Scholar] [CrossRef]
- Chen, W.-H.; Peng, J.; Bi, X.T. A State-of-the-Art Review of Biomass Torrefaction, Densification and Applications. Renew. Sustain. Energy Rev. 2015, 44, 847–866. [Google Scholar] [CrossRef]
- Wu, K.-T.; Tsai, C.-J.; Chen, C.-S.; Chen, H.-W. The Characteristics of Torrefied Microalgae. Appl. Energy 2012, 100, 52–57. [Google Scholar] [CrossRef]
- Cahyanti, M.N.; Doddapaneni, T.R.K.C.; Kikas, T. Biomass Torrefaction: An Overview on Process Parameters, Economic and Environmental Aspects and Recent Advancements. Bioresour. Technol. 2020, 301, 122737. [Google Scholar] [CrossRef] [PubMed]
- Bach, Q.-V.; Chen, W.-H.; Lin, S.-C.; Sheen, H.-K.; Chang, J.-S. Effect of Wet Torrefaction on Thermal Decomposition Behavior of Microalga Chlorella vulgaris ESP-31. Energy Procedia 2017, 105, 206–211. [Google Scholar] [CrossRef]
- Bach, Q.-V.; Chen, W.-H.; Sheen, H.-K.; Chang, J.-S. Gasification Kinetics of Raw and Wet-Torrefied Microalgae Chlorella vulgaris ESP-31 in Carbon Dioxide. Bioresour. Technol. 2017, 244, 1393–1399. [Google Scholar] [CrossRef] [PubMed]
- Uemura, Y.; Matsumoto, R.; Saadon, S.; Matsumura, Y. A Study on Torrefaction of Laminaria japonica. Fuel Process. Technol. 2015, 138, 133–138. [Google Scholar] [CrossRef]
- Chen, W.-H.; Huang, M.-Y.; Chang, J.-S.; Chen, C.-Y.; Lee, W.-J. An Energy Analysis of Torrefaction for Upgrading Microalga Residue as a Solid Fuel. Bioresour. Technol. 2015, 185, 285–293. [Google Scholar] [CrossRef]
- Yu, K.L.; Chen, W.-H.; Sheen, H.-K.; Chang, J.-S.; Lin, C.-S.; Ong, H.C.; Show, P.L.; Ng, E.-P.; Ling, T.C. Production of Microalgal Biochar and Reducing Sugar Using Wet Torrefaction with Microwave-Assisted Heating and Acid Hydrolysis Pretreatment. Renew. Energy 2020, 156, 349–360. [Google Scholar] [CrossRef]
- Yu, K.L.; Chen, W.-H.; Sheen, H.-K.; Chang, J.-S.; Lin, C.-S.; Ong, H.C.; Show, P.L.; Ling, T.C. Bioethanol Production from Acid Pretreated Microalgal Hydrolysate Using Microwave-Assisted Heating Wet Torrefaction. Fuel 2020, 279, 118435. [Google Scholar] [CrossRef]
- Brennan, L.; Owende, P. Biofuels from Microalgae—A Review of Technologies for Production, Processing, and Extractions of Biofuels and Co-Products. Renew. Sustain. Energy Rev. 2010, 14, 557–577. [Google Scholar] [CrossRef]
- Harman-Ware, A.E.; Morgan, T.; Wilson, M.; Crocker, M.; Zhang, J.; Liu, K.; Stork, J.; Debolt, S. Microalgae as a Renewable Fuel Source: Fast Pyrolysis of Scenedesmus sp. Renew. Energy 2013, 60, 625–632. [Google Scholar] [CrossRef]
- Babich, I.V.; van der Hulst, M.; Lefferts, L.; Moulijn, J.A.; O’Connor, P.; Seshan, K. Catalytic Pyrolysis of Microalgae to High-Quality Liquid Bio-Fuels. Biomass Bioenergy 2011, 35, 3199–3207. [Google Scholar] [CrossRef]
- Roberts, D.A.; Paul, N.A.; Dworjanyn, S.A.; Bird, M.I.; de Nys, R. Biochar from Commercially Cultivated Seaweed for Soil Amelioration. Sci. Rep. 2015, 5, 9665. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.F.; Lo, S.L. Predicting Heating Value of Lignocellulosic Biomass Based on Elemental Analysis. Energy 2020, 191, 116501. [Google Scholar] [CrossRef]
- Choi, J.H.; Kim, S.-S.; Suh, D.J.; Jang, E.-J.; Min, K.-I.; Woo, H.C. Characterization of the Bio-Oil and Bio-Char Produced by Fixed Bed Pyrolysis of the Brown Alga Saccharina japonica. Korean J. Chem. Eng. 2016, 33, 2691–2698. [Google Scholar] [CrossRef]
- Bae, Y.J.; Ryu, C.; Jeon, J.-K.; Park, J.; Suh, D.J.; Suh, Y.-W.; Chang, D.; Park, Y.-K. The Characteristics of Bio-Oil Produced from the Pyrolysis of Three Marine Macroalgae. Bioresour. Technol. 2011, 102, 3512–3520. [Google Scholar] [CrossRef]
- Maddi, B.; Viamajala, S.; Varanasi, S. Comparative Study of Pyrolysis of Algal Biomass from Natural Lake Blooms with Lignocellulosic Biomass. Bioresour. Technol. 2011, 102, 11018–11026. [Google Scholar] [CrossRef]
- Lee, X.J.; Ong, H.C.; Gan, Y.Y.; Chen, W.H.; Mahlia, T.M.I. State of Art Review on Conventional and Advanced Pyrolysis of Macroalgae and Microalgae for Biochar, Bio-Oil and Bio-Syngas Production. Energy Convers. Manag. 2020, 210, 112707. [Google Scholar] [CrossRef]
- Ahmed, A.; Abu Bakar, M.S.; Azad, A.K.; Sukri, R.S.; Phusunti, N. Intermediate Pyrolysis of Acacia cincinnata and Acacia holosericea Species for Bio-Oil and Biochar Production. Energy Convers. Manag. 2018, 176, 393–408. [Google Scholar] [CrossRef]
- Kebelmann, K.; Hornung, A.; Karsten, U.; Griffiths, G. Thermo-Chemical Behaviour and Chemical Product Formation from Polar Seaweeds during Intermediate Pyrolysis. J. Anal. Appl. Pyrolysis 2013, 104, 131–138. [Google Scholar] [CrossRef]
- Mahmood, A.S.N.; Brammer, J.G.; Hornung, A.; Steele, A.; Poulston, S. The Intermediate Pyrolysis and Catalytic Steam Reforming of Brewers Spent Grain. J. Anal. Appl. Pyrolysis 2013, 103, 328–342. [Google Scholar] [CrossRef]
- Yang, Y.; Zhang, Y.; Omairey, E.; Cai, J.; Gu, F.; Bridgwater, A.V. Intermediate Pyrolysis of Organic Fraction of Municipal Solid Waste and Rheological Study of the Pyrolysis Oil for Potential Use as Bio-Bitumen. J. Clean. Prod. 2018, 187, 390–399. [Google Scholar] [CrossRef]
- Mohammed, I.Y.; Abakr, Y.A.; Yusup, S.; Kazi, F.K. Valorization of Napier Grass via Intermediate Pyrolysis: Optimization Using Response Surface Methodology and Pyrolysis Products Characterization. J. Clean. Prod. 2017, 142, 1848–1866. [Google Scholar] [CrossRef]
- Kebelmann, K.; Hornung, A.; Karsten, U.; Griffiths, G. Intermediate Pyrolysis and Product Identification by TGA and Py-GC/MS of Green Microalgae and Their Extracted Protein and Lipid Components. Biomass Bioenergy 2013, 49, 38–48. [Google Scholar] [CrossRef]
- Yang, Y.; Brammer, J.G.; Mahmood, A.S.N.; Hornung, A. Intermediate Pyrolysis of Biomass Energy Pellets for Producing Sustainable Liquid, Gaseous and Solid Fuels. Bioresour. Technol. 2014, 169, 794–799. [Google Scholar] [CrossRef] [PubMed]
- Chang, Y.-M.; Tsai, W.-T.; Li, M.-H. Chemical Characterization of Char Derived from Slow Pyrolysis of Microalgal Residue. J. Anal. Appl. Pyrolysis 2015, 111, 88–93. [Google Scholar] [CrossRef]
- Wang, K.; Brown, R.C.; Homsy, S.; Martinez, L.; Sidhu, S.S. Fast Pyrolysis of Microalgae Remnants in a Fluidized Bed Reactor for Bio-Oil and Biochar Production. Bioresour. Technol. 2013, 127, 494–499. [Google Scholar] [CrossRef]
- Ashokkumar, V.; Chen, W.-H.; Kamyab, H.; Kumar, G.; Al-Muhtaseb, A.H.; Ngamcharussrivichai, C. Cultivation of Microalgae Chlorella sp. in Municipal Sewage for Biofuel Production and Utilization of Biochar Derived from Residue for the Conversion of Hematite Iron Ore (Fe2O3) to Iron (Fe)—Integrated Algal Biorefinery. Energy 2019, 189, 116128. [Google Scholar] [CrossRef]
- Woolf, D.; Amonette, J.E.; Street-Perrott, F.A.; Lehmann, J.; Joseph, S. Sustainable Biochar to Mitigate Global Climate Change. Nat. Commun. 2010, 1, 56. [Google Scholar] [CrossRef]
- Lavecchia, R.; Medici, F.; Patterer, M.S.; Zuorro, A. Lead removal from water by adsorption on spent coffee grounds. Chem. Eng. Trans. 2016, 47, 295–300. [Google Scholar] [CrossRef]
- Nejati, B.; Adami, P.; Bozorg, A.; Tavasoli, A.; Mirzahosseini, A.H. Catalytic Pyrolysis and Bio-Products Upgrading Derived from Chlorella vulgaris over Its Biochar and Activated Biochar-Supported Fe Catalysts. J. Anal. Appl. Pyrolysis 2020, 104799. [Google Scholar] [CrossRef]
- Jung, K.-W.; Jeong, T.-U.; Kang, H.-J.; Ahn, K.-H. Characteristics of Biochar Derived from Marine Macroalgae and Fabrication of Granular Biochar by Entrapment in Calcium-Alginate Beads for Phosphate Removal from Aqueous Solution. Bioresour. Technol. 2016, 211, 108–116. [Google Scholar] [CrossRef] [PubMed]
- Cai, J.; Wu, W.; Liu, R.; Huber, G.W. A Distributed Activation Energy Model for the Pyrolysis of Lignocellulosic Biomass. Green Chem. 2013, 15, 1331–1340. [Google Scholar] [CrossRef]
- Hertzog, J.; Carré, V.; Jia, L.; Mackay, C.L.; Pinard, L.; Dufour, A.; Mašek, O.; Aubriet, F. Catalytic Fast Pyrolysis of Biomass over Microporous and Hierarchical Zeolites: Characterization of Heavy Products. ACS Sustain. Chem. Eng. 2018, 6, 4717–4728. [Google Scholar] [CrossRef]
- Li, F.; Srivatsa, S.C.; Bhattacharya, S. A Review on Catalytic Pyrolysis of Microalgae to High-Quality Bio-Oil with Low Oxygeneous and Nitrogenous Compounds. Renew. Sustain. Energy Rev. 2019, 108, 481–497. [Google Scholar] [CrossRef]
- Yang, C.; Li, R.; Zhang, B.; Qiu, Q.; Wang, B.; Yang, H.; Ding, Y.; Wang, C. Pyrolysis of Microalgae: A Critical Review. Fuel Process. Technol. 2019, 186, 53–72. [Google Scholar] [CrossRef]
- Amin, M.; Chetpattananondh, P.; Ratanawilai, S. Application of Extracted Marine Chlorella Sp. Residue for Bio-Oil Production as the Biomass Feedstock and Microwave Absorber. Energy Convers. Manag. 2019, 195, 819–829. [Google Scholar] [CrossRef]
- Jafarian, S.; Tavasoli, A. A Comparative Study on the Quality of Bioproducts Derived from Catalytic Pyrolysis of Green Microalgae Spirulina (Arthrospira) plantensis over Transition Metals Supported on HMS-ZSM5 Composite. Int. J. Hydrogen Energy 2018, 43, 19902–19917. [Google Scholar] [CrossRef]
- Andrade, L.A.; Barrozo, M.A.S.; Vieira, L.G.M. Catalytic Solar Pyrolysis of Microalgae Chlamydomonas reinhardtii. Sol. Energy 2018, 173, 928–938. [Google Scholar] [CrossRef]
- Campanella, A.; Harold, M.P. Fast Pyrolysis of Microalgae in a Falling Solids Reactor: Effects of Process Variables and Zeolite Catalysts. Biomass Bioenergy 2012, 46, 218–232. [Google Scholar] [CrossRef]
- Huang, F.; Tahmasebi, A.; Maliutina, K.; Yu, J. Formation of Nitrogen-Containing Compounds during Microwave Pyrolysis of Microalgae: Product Distribution and Reaction Pathways. Bioresour. Technol. 2017, 245, 1067–1074. [Google Scholar] [CrossRef] [PubMed]
- Wang, K.; Brown, R.C. Catalytic Pyrolysis of Microalgae for Production of Aromatics and Ammonia. Green Chem. 2013, 15, 675–681. [Google Scholar] [CrossRef]
- Zainan, N.H.; Srivatsa, S.C.; Li, F.; Bhattacharya, S. Quality of Bio-Oil from Catalytic Pyrolysis of Microalgae Chlorella vulgaris. Fuel 2018, 223, 12–19. [Google Scholar] [CrossRef]
- Thangalazhy-Gopakumar, S.; Adhikari, S.; Chattanathan, S.A.; Gupta, R.B. Catalytic Pyrolysis of Green Algae for Hydrocarbon Production Using H+ZSM-5 Catalyst. Bioresour. Technol. 2012, 118, 150–157. [Google Scholar] [CrossRef] [PubMed]
- Conti, R.; Pezzolesi, L.; Pistocchi, R.; Torri, C.; Massoli, P.; Fabbri, D. Photobioreactor Cultivation and Catalytic Pyrolysis of the Microalga Desmodesmus communis (Chlorophyceae) for Hydrocarbons Production by HZSM-5 Zeolite Cracking. Bioresour. Technol. 2016, 222, 148–155. [Google Scholar] [CrossRef] [PubMed]
- Gong, Z.; Fang, P.; Wang, Z.; Li, Q.; Li, X.; Meng, F.; Zhang, H.; Liu, L. Catalytic Pyrolysis of Chemical Extraction Residue from Microalgae Biomass. Renew. Energy 2020, 148, 712–719. [Google Scholar] [CrossRef]
- Aysu, T.; Abd Rahman, N.A.; Sanna, A. Catalytic Pyrolysis of Tetraselmis and Isochrysis Microalgae by Nickel Ceria Based Catalysts for Hydrocarbon Production. Energy 2016, 103, 205–214. [Google Scholar] [CrossRef]
- Rahman, N.A.A.; Fermoso, J.; Sanna, A. Effect of Li-LSX-Zeolite on the in-Situ Catalytic Deoxygenation and Denitrogenation of Isochrysis sp. Microalgae Pyrolysis Vapours. Fuel Process. Technol. 2018, 173, 253–261. [Google Scholar] [CrossRef]
- Abd Rahman, N.A.; Fermoso, J.; Sanna, A. Stability of Li-LSX Zeolite in the Catalytic Pyrolysis of Non-Treated and Acid Pre-Treated Isochrysis sp. Microalgae. Energies 2020, 13, 959. [Google Scholar] [CrossRef]
- Pan, P.; Hu, C.; Yang, W.; Li, Y.; Dong, L.; Zhu, L.; Tong, D.; Qing, R.; Fan, Y. The Direct Pyrolysis and Catalytic Pyrolysis of Nannochloropsis sp. Residue for Renewable Bio-Oils. Bioresour. Technol. 2010, 101, 4593–4599. [Google Scholar] [CrossRef]
- Aysu, T.; Sanna, A. Nannochloropsis Algae Pyrolysis with Ceria-Based Catalysts for Production of High-Quality Bio-Oils. Bioresour. Technol. 2015, 194, 108–116. [Google Scholar] [CrossRef] [PubMed]
- Qi, P.; Chang, G.; Wang, H.; Zhang, X.; Guo, Q. Production of Aromatic Hydrocarbons by Catalytic Co-Pyrolysis of Microalgae and Polypropylene Using HZSM-5. J. Anal. Appl. Pyrolysis 2018, 136, 178–185. [Google Scholar] [CrossRef]
- Gautam, R.; Vinu, R. Non-Catalytic Fast Pyrolysis and Catalytic Fast Pyrolysis of Nannochloropsis Oculata Using Co-Mo/γ-Al2O3 Catalyst for Valuable Chemicals. Algal Res. 2018, 34, 12–24. [Google Scholar] [CrossRef]
- Kawale, H.D.; Kishore, N. Production of Hydrocarbons from a Green Algae (Oscillatoria) with Exploration of Its Fuel Characteristics over Different Reaction Atmospheres. Energy 2019, 178, 344–355. [Google Scholar] [CrossRef]
- Aysu, T.; Fermoso, J.; Sanna, A. Ceria on Alumina Support for Catalytic Pyrolysis of Pavlova sp. Microalgae to High-Quality Bio-Oils. J. Energy Chem. 2018, 27, 874–882. [Google Scholar] [CrossRef]
- Aysu, T.; Ola, O.; Maroto-Valer, M.M.; Sanna, A. Effects of Titania Based Catalysts on In-Situ Pyrolysis of Pavlova Microalgae. Fuel Process. Technol. 2017, 166, 291–298. [Google Scholar] [CrossRef]
- Anand, V.; Gautam, R.; Vinu, R. Non-Catalytic and Catalytic Fast Pyrolysis of Schizochytrium limacinum Microalga. Fuel 2017, 205, 1–10. [Google Scholar] [CrossRef]
- Mo, L.; Dai, H.; Feng, L.; Liu, B.; Li, X.; Chen, Y.; Khan, S. In-Situ Catalytic Pyrolysis Upgradation of Microalgae into Hydrocarbon Rich Bio-Oil: Effects of Nitrogen and Carbon Dioxide Environment. Bioresour. Technol. 2020, 314, 123758. [Google Scholar] [CrossRef]
- Xu, Y.; Hu, Y.; Peng, Y.; Yao, L.; Dong, Y.; Yang, B.; Song, R. Catalytic Pyrolysis and Liquefaction Behavior of Microalgae for Bio-Oil Production. Bioresour. Technol. 2020, 300, 122665. [Google Scholar] [CrossRef]
- Suali, E.; Sarbatly, R. Conversion of Microalgae to Biofuel. Renew. Sustain. Energy Rev. 2012, 16, 4316–4342. [Google Scholar] [CrossRef]
- Cheng, Y.-T.; Jae, J.; Shi, J.; Fan, W.; Huber, G.W. Production of Renewable Aromatic Compounds by Catalytic Fast Pyrolysis of Lignocellulosic Biomass with Bifunctional Ga/ZSM-5 Catalysts. Angew. Chem. Int. Ed. 2012, 51, 1387–1390. [Google Scholar] [CrossRef] [PubMed]
- Du, Z.; Ma, X.; Li, Y.; Chen, P.; Liu, Y.; Lin, X.; Lei, H.; Ruan, R. Production of Aromatic Hydrocarbons by Catalytic Pyrolysis of Microalgae with Zeolites: Catalyst Screening in a Pyroprobe. Bioresour. Technol. 2013, 139, 397–401. [Google Scholar] [CrossRef] [PubMed]
- Vichaphund, S.; Aht-ong, D.; Sricharoenchaikul, V.; Atong, D. Production of Aromatic Compounds from Catalytic Fast Pyrolysis of Jatropha Residues Using Metal/HZSM-5 Prepared by Ion-Exchange and Impregnation Methods. Renew. Energy 2015, 79, 28–37. [Google Scholar] [CrossRef]
- Naqvi, S.R.; Naqvi, M.; Noor, T.; Hussain, A.; Iqbal, N.; Uemura, Y.; Nishiyama, N. Catalytic Pyrolysis of Botryococcus braunii (Microalgae) Over Layered and Delaminated Zeolites for Aromatic Hydrocarbon Production. Energy Procedia 2017, 142, 381–385. [Google Scholar] [CrossRef]
- Belotti, G.; de Caprariis, B.; De Filippis, P.; Scarsella, M.; Verdone, N. Effect of Chlorella vulgaris Growing Conditions on Bio-Oil Production via Fast Pyrolysis. Biomass Bioenergy 2014, 61, 187–195. [Google Scholar] [CrossRef]
- Du, Z.; Hu, B.; Ma, X.; Cheng, Y.; Liu, Y.; Lin, X.; Wan, Y.; Lei, H.; Chen, P.; Ruan, R. Catalytic Pyrolysis of Microalgae and Their Three Major Components: Carbohydrates, Proteins, and Lipids. Bioresour. Technol. 2013, 130, 777–782. [Google Scholar] [CrossRef]
- Gao, L.; Sun, J.; Xu, W.; Xiao, G. Catalytic Pyrolysis of Natural Algae over Mg-Al Layered Double Oxides/ZSM-5 (MgAl-LDO/ZSM-5) for Producing Bio-Oil with Low Nitrogen Content. Bioresour. Technol. 2017, 225, 293–298. [Google Scholar] [CrossRef]
- Galadima, A.; Muraza, O. Hydrothermal Liquefaction of Algae and Bio-Oil Upgrading into Liquid Fuels: Role of Heterogeneous Catalysts. Renew. Sustain. Energy Rev. 2018, 81, 1037–1048. [Google Scholar] [CrossRef]
- Yang, J.; He, Q.; Yang, L. A Review on Hydrothermal Co-Liquefaction of Biomass. Appl. Energy 2019, 250, 926–945. [Google Scholar] [CrossRef]
- Ponnusamy, V.K.; Nagappan, S.; Bhosale, R.R.; Lay, C.-H.; Duc Nguyen, D.; Pugazhendhi, A.; Chang, S.W.; Kumar, G. Review on Sustainable Production of Biochar through Hydrothermal Liquefaction: Physico-Chemical Properties and Applications. Bioresour. Technol. 2020, 310, 123414. [Google Scholar] [CrossRef]
- Chaudry, S.; Bahri, P.A.; Moheimani, N.R. Pathways of Processing of Wet Microalgae for Liquid Fuel Production: A Critical Review. Renew. Sustain. Energy Rev. 2015, 52, 1240–1250. [Google Scholar] [CrossRef]
- Xu, D.; Lin, G.; Guo, S.; Wang, S.; Guo, Y.; Jing, Z. Catalytic Hydrothermal Liquefaction of Algae and Upgrading of Biocrude: A Critical Review. Renew. Sustain. Energy Rev. 2018, 97, 103–118. [Google Scholar] [CrossRef]
- Guo, Y.; Yeh, T.; Song, W.; Xu, D.; Wang, S. A Review of Bio-Oil Production from Hydrothermal Liquefaction of Algae. Renew. Sustain. Energy Rev. 2015, 48, 776–790. [Google Scholar] [CrossRef]
- Tekin, K.; Karagöz, S.; Bektaş, S. A Review of Hydrothermal Biomass Processing. Renew. Sustain. Energy Rev. 2014, 40, 673–687. [Google Scholar] [CrossRef]
- Pavlovič, I.; Knez, Ž.; Škerget, M. Hydrothermal Reactions of Agricultural and Food Processing Wastes in Sub- and Supercritical Water: A Review of Fundamentals, Mechanisms, and State of Research. J. Agric. Food Chem. 2013, 61, 8003–8025. [Google Scholar] [CrossRef]
- Hu, Y.; Gong, M.; Feng, S.; Xu, C.; Bassi, A. A Review of Recent Developments of Pre-Treatment Technologies and Hydrothermal Liquefaction of Microalgae for Bio-Crude Oil Production. Renew. Sustain. Energy Rev. 2019, 101, 476–492. [Google Scholar] [CrossRef]
- Eboibi, B.E.; Lewis, D.M.; Ashman, P.J.; Chinnasamy, S. Influence of Process Conditions on Pretreatment of Microalgae for Protein Extraction and Production of Biocrude during Hydrothermal Liquefaction of Pretreated Tetraselmis sp. RSC Adv. 2015, 5, 20193–20207. [Google Scholar] [CrossRef]
- Fu, J.; Yang, C.; Wu, J.; Zhuang, J.; Hou, Z.; Lu, X. Direct Production of Aviation Fuels from Microalgae Lipids in Water. Fuel 2015, 139, 678–683. [Google Scholar] [CrossRef]
- Mathimani, T.; Mallick, N. A Review on the Hydrothermal Processing of Microalgal Biomass to Bio-Oil—Knowledge Gaps and Recent Advances. J. Clean. Prod. 2019, 217, 69–84. [Google Scholar] [CrossRef]
- Biller, P.; Ross, A.B. Potential Yields and Properties of Oil from the Hydrothermal Liquefaction of Microalgae with Different Biochemical Content. Bioresour. Technol. 2011, 102, 215–225. [Google Scholar] [CrossRef]
- Ross, A.B.; Biller, P.; Kubacki, M.L.; Li, H.; Lea-Langton, A.; Jones, J.M. Hydrothermal Processing of Microalgae Using Alkali and Organic Acids. Fuel 2010, 89, 2234–2243. [Google Scholar] [CrossRef]
- Hu, Y.; Feng, S.; Yuan, Z.; Xu, C.; Bassi, A. Investigation of Aqueous Phase Recycling for Improving Bio-Crude Oil Yield in Hydrothermal Liquefaction of Algae. Bioresour. Technol. 2017, 239, 151–159. [Google Scholar] [CrossRef] [PubMed]
- Yu, G.; Zhang, Y.; Guo, B.; Funk, T.; Schideman, L. Nutrient Flows and Quality of Bio-Crude Oil Produced via Catalytic Hydrothermal Liquefaction of Low-Lipid Microalgae. BioEnergy Res. 2014, 7, 1317–1328. [Google Scholar] [CrossRef]
- Muppaneni, T.; Reddy, H.K.; Selvaratnam, T.; Dandamudi, K.P.R.; Dungan, B.; Nirmalakhandan, N.; Schaub, T.; Omar Holguin, F.; Voorhies, W.; Lammers, P.; et al. Hydrothermal Liquefaction of Cyanidioschyzon merolae and the Influence of Catalysts on Products. Bioresour. Technol. 2017, 223, 91–97. [Google Scholar] [CrossRef] [PubMed]
- Minowa, T.; Yokoyama, S.; Kishimoto, M.; Okakura, T. Oil Production from Algal Cells of Dunaliella tertiolecta by Direct Thermochemical Liquefaction. Fuel 1995, 74, 1735–1738. [Google Scholar] [CrossRef]
- Yang, W.; Li, X.; Liu, S.; Feng, L. Direct Hydrothermal Liquefaction of Undried Macroalgae Enteromorpha prolifera Using Acid Catalysts. Energy Convers. Manag. 2014, 87, 938–945. [Google Scholar] [CrossRef]
- Shakya, R.; Whelen, J.; Adhikari, S.; Mahadevan, R.; Neupane, S. Effect of Temperature and Na2CO3 Catalyst on Hydrothermal Liquefaction of Algae. Algal Res. 2015, 12, 80–90. [Google Scholar] [CrossRef]
- Bach, Q.-V.; Sillero, M.V.; Tran, K.-Q.; Skjermo, J. Fast Hydrothermal Liquefaction of a Norwegian Macro-Alga: Screening Tests. Algal Res. 2014, 6, 271–276. [Google Scholar] [CrossRef]
- Yang, Y.F.; Feng, C.P.; Inamori, Y.; Maekawa, T. Analysis of Energy Conversion Characteristics in Liquefaction of Algae. Resour. Conserv. Recycl. 2004, 43, 21–33. [Google Scholar] [CrossRef]
- Saber, M.; Golzary, A.; Hosseinpour, M.; Takahashi, F.; Yoshikawa, K. Catalytic Hydrothermal Liquefaction of Microalgae Using Nanocatalyst. Appl. Energy 2016, 183, 566–576. [Google Scholar] [CrossRef]
- Jena, U.; Das, K.C.; Kastner, J.R. Comparison of the Effects of Na2CO3, Ca3(PO4)2, and NiO Catalysts on the Thermochemical Liquefaction of Microalga Spirulina platensis. Appl. Energy 2012, 98, 368–375. [Google Scholar] [CrossRef]
- Lavanya, M.; Meenakshisundaram, A.; Renganathan, S.; Chinnasamy, S.; Lewis, D.M.; Nallasivam, J.; Bhaskar, S. Hydrothermal Liquefaction of Freshwater and Marine Algal Biomass: A Novel Approach to Produce Distillate Fuel Fractions through Blending and Co-Processing of Biocrude with Petrocrude. Bioresour. Technol. 2016, 203, 228–235. [Google Scholar] [CrossRef] [PubMed]
- Yan, L.; Wang, Y.; Li, J.; Zhang, Y.; Ma, L.; Fu, F.; Chen, B.; Liu, H. Hydrothermal Liquefaction of Ulva prolifera Macroalgae and the Influence of Base Catalysts on Products. Bioresour. Technol. 2019, 292, 121286. [Google Scholar] [CrossRef] [PubMed]
- Kumar, V.; Kumar, S.; Chauhan, P.K.; Verma, M.; Bahuguna, V.; Joshi, H.C.; Ahmad, W.; Negi, P.; Sharma, N.; Ramola, B.; et al. Low-Temperature Catalyst Based Hydrothermal Liquefaction of Harmful Macroalgal Blooms, and Aqueous Phase Nutrient Recycling by Microalgae. Sci. Rep. 2019, 9, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Zou, S.; Wu, Y.; Yang, M.; Li, C.; Tong, J. Thermochemical Catalytic Liquefaction of the Marine Microalgae Dunaliella tertiolecta and Characterization of Bio-Oils. Energy Fuels 2009, 23, 3753–3758. [Google Scholar] [CrossRef]
- Zhuang, Y.; Guo, J.; Chen, L.; Li, D.; Liu, J.; Ye, N. Microwave-Assisted Direct Liquefaction of Ulva prolifera for Bio-Oil Production by Acid Catalysis. Bioresour. Technol. 2012, 116, 133–139. [Google Scholar] [CrossRef]
- Li, J.; Fang, X.; Bian, J.; Guo, Y.; Li, C. Microalgae Hydrothermal Liquefaction and Derived Biocrude Upgrading with Modified SBA-15 Catalysts. Bioresour. Technol. 2018, 266, 541–547. [Google Scholar] [CrossRef]
- Xu, D.; Guo, S.; Liu, L.; Lin, G.; Wu, Z.; Guo, Y.; Wang, S. Heterogeneous Catalytic Effects on the Characteristics of Water-Soluble and Water-Insoluble Biocrudes in Chlorella Hydrothermal Liquefaction. Appl. Energy 2019, 243, 165–174. [Google Scholar] [CrossRef]
- Yang, L.; Ma, R.; Ma, Z.; Li, Y. Catalytic Conversion of Chlorella pyrenoidosa to Biofuels in Supercritical Alcohols over Zeolites. Bioresour. Technol. 2016, 209, 313–317. [Google Scholar] [CrossRef]
- Xu, Y.; Zheng, X.; Yu, H.; Hu, X. Hydrothermal Liquefaction of Chlorella pyrenoidosa for Bio-Oil Production over Ce/HZSM-5. Bioresour. Technol. 2014, 156, 1–5. [Google Scholar] [CrossRef]
- Chen, Y.; Wu, Y.; Ding, R.; Zhang, P.; Liu, J.; Yang, M.; Zhang, P. Catalytic Hydrothermal Liquefaction of D. tertiolecta for the Production of Bio-Oil over Different Acid/Base Catalysts. AIChE J. 2015, 61, 1118–1128. [Google Scholar] [CrossRef]
- Yang, C.; Jia, L.; Chen, C.; Liu, G.; Fang, W. Bio-Oil from Hydro-Liquefaction of Dunaliella salina over Ni/REHY Catalyst. Bioresour. Technol. 2011, 102, 4580–4584. [Google Scholar] [CrossRef] [PubMed]
- Duan, P.; Savage, P.E. Hydrothermal Liquefaction of a Microalga with Heterogeneous Catalysts. Ind. Eng. Chem. Res. 2011, 50, 52–61. [Google Scholar] [CrossRef]
- Yang, L.; Li, Y.; Savage, P.E. Catalytic Hydrothermal Liquefaction of a Microalga in a Two-Chamber Reactor. Ind. Eng. Chem. Res. 2014, 53, 11939–11944. [Google Scholar] [CrossRef]
- Liu, Z.; Li, H.; Zeng, J.; Liu, M.; Zhang, Y.; Liu, Z. Influence of Fe/HZSM-5 Catalyst on Elemental Distribution and Product Properties during Hydrothermal Liquefaction of Nannochloropsis sp. Algal Res. 2018, 35, 1–9. [Google Scholar] [CrossRef]
- Liu, C.; Kong, L.; Wang, Y.; Dai, L. Catalytic Hydrothermal Liquefaction of Spirulina to Bio-Oil in the Presence of Formic Acid over Palladium-Based Catalysts. Algal Res. 2018, 33, 156–164. [Google Scholar] [CrossRef]
- Kandasamy, S.; Zhang, B.; He, Z.; Chen, H.; Feng, H.; Wang, Q.; Wang, B.; Ashokkumar, V.; Siva, S.; Bhuvanendran, N.; et al. Effect of Low-Temperature Catalytic Hydrothermal Liquefaction of Spirulina platensis. Energy 2020, 190, 116236. [Google Scholar] [CrossRef]
- Kandasamy, S.; Zhang, B.; He, Z.; Chen, H.; Feng, H.; Wang, Q.; Wang, B.; Bhuvanendran, N.; Esakkimuthu, S.; Ashokkumar, V.; et al. Hydrothermal Liquefaction of Microalgae Using Fe3O4 Nanostructures as Efficient Catalyst for the Production of Bio-Oil: Optimization of Reaction Parameters by Response Surface Methodology. Biomass Bioenergy 2019, 131, 105417. [Google Scholar] [CrossRef]
- Ma, C.; Geng, J.; Zhang, D.; Ning, X. Hydrothermal Liquefaction of Macroalgae: Influence of Zeolites Based Catalyst on Products. J. Energy Inst. 2020, 93, 581–590. [Google Scholar] [CrossRef]
- Nava Bravo, I.; Velásquez-Orta, S.B.; Cuevas-García, R.; Monje-Ramírez, I.; Harvey, A.; Orta Ledesma, M.T. Bio-Crude Oil Production Using Catalytic Hydrothermal Liquefaction (HTL) from Native Microalgae Harvested by Ozone-Flotation. Fuel 2019, 241, 255–263. [Google Scholar] [CrossRef]
- Zhang, J.; Chen, W.-T.; Zhang, P.; Luo, Z.; Zhang, Y. Hydrothermal Liquefaction of Chlorella pyrenoidosa in Sub- and Supercritical Ethanol with Heterogeneous Catalysts. Bioresour. Technol. 2013, 133, 389–397. [Google Scholar] [CrossRef] [PubMed]
- López Barreiro, D.; Prins, W.; Ronsse, F.; Brilman, W. Hydrothermal Liquefaction (HTL) of Microalgae for Biofuel Production: State of the Art Review and Future Prospects. Biomass Bioenergy 2013, 53, 113–127. [Google Scholar] [CrossRef]
- Tian, C.; Li, B.; Liu, Z.; Zhang, Y.; Lu, H. Hydrothermal Liquefaction for Algal Biorefinery: A Critical Review. Renew. Sustain. Energy Rev. 2014, 38, 933–950. [Google Scholar] [CrossRef]
- Biller, P.; Riley, R.; Ross, A.B. Catalytic Hydrothermal Processing of Microalgae: Decomposition and Upgrading of Lipids. Bioresour. Technol. 2011, 102, 4841–4848. [Google Scholar] [CrossRef] [PubMed]
| Strain | Lipids (wt%) | Carbohydrates (wt%) | Proteins (wt%) | Reference |
|---|---|---|---|---|
| Arthrospira platensis | 30.23 | 31.89 | 16.81 | [17] |
| Auxenochlorella protothecoides | 42 | 26 | 30 | [18] |
| Botryococcus braunii | 45 | 10 | 44 | [19] |
| 60 | 20 | 18 | [20] | |
| Chlamydomonas reinhardtii | 22.11 | 52.2 | 23.69 | [21] |
| Ch. reinhardtii CC-400 | 28.5 | n/a | n/a | [22] |
| Ch. Reinhardtii CC-4349 | 64.25 | n/a | n/a | [23] |
| Chlorella sp G-9 | 36.5 | n/a | n/a | [24] |
| C. kessleri | 20 | 18.7 | 53.8 | [25] |
| C. pyrenoidosa | 19.8 | 14.8 | 57.3 | [26] |
| C. vulgaris UTEX 259 | 28 | 35 | 20 | [27] |
| C. vulgaris UTEX 1803 | 12 | 36 | 41 | [28] |
| C. vulgaris Mutant (UV715) | 41 | n/a | n/a | [29] |
| Chlorococcum oleofaciens | 20 | 42 | 35 | [30] |
| Dunaliella tertiolecta | 15 | 10 | 56 | [31] |
| Nannochloropsis gaditana | 17.6 | n/a | 24.1 | [32] |
| Pseudokirchneriella Subcapitata | 40 | 20 | 30 | [30] |
| Phaeodactylum tricornutum | 55.7 | 9 | 22 | [33] |
| Scenedesmus almeriensis | 13.1 | n/a | 30 | [34] |
| S. obliquus | 32.5 | n/a | n/a | [35] |
| 24.9 | n/a | n/a | [36] | |
| 35 | 22 | 32 | [37,38] | |
| Tetraselmis suecica | 9.03 | 20 | 37.27 | [39] |
| 25.07 | 17.52 | 42.05 | [40] |
| Strain | HHV (MJ/kg) | Heating Rate (°C/min) | Pyrolysis Time (min) | Pyrolysis Temperature (°C) | Catalyst | Bio-oil (wt%) | Bio-char (wt%) | Syngas (wt%) | Reference |
|---|---|---|---|---|---|---|---|---|---|
| Arthrospira plantensis | 21.45 | 100 | 30 | 400–700 | Ni/HMS-ZSM5 | 32.52 | 34.04 | 33.44 | [109] |
| Fe/HMS-ZSM5 | 30.01 | 31.84 | 38.15 | ||||||
| Ce/HMS-ZSM5 | 31.80 | 31.79 | 36.41 | ||||||
| Chlamydomonas reinhardtii | 20.47 | 150 | 10–34 | 500 | hydrotalcite | 54.84 | 37.59 | 7.57 | [110] |
| Ch. debaryana | 21.9 | >200 | 30 | 500–800 | β-zeolite | 23.5 | n/a | n/a | [4] |
| Activated charcoal | 43.8 | n/a | n/a | ||||||
| Chlorella sp. | 21.2 | n/a | 30 | 300–450 | Na2CO3 | 41.0 | 54.4 | 34.1 | [83] |
| 19.5 | n/a | 10 | 500 | Fe-ZSM-5 | 43.1 | 29.7 | 27.1 | [111] | |
| Cu-ZSM-5 | 46.9 | 27.9 | 24.6 | ||||||
| Ni-ZSM-5 | 45.1 | 30.1 | 25.4 | ||||||
| n/a | n/a | 50 | 350–650 | Magnetite | 53.8 | 27.4 | 22.8 | [112] | |
| Activated carbon | 49. 4 | 37.3 | 13.3 | ||||||
| C. vulgaris | 16.8 | 10 | 30 | 700 | H+ZSM-5 | 25 | 24 | n/a | [113] |
| n/a | 10 | 30 | 300–600 | Ni-ZSM-5 | 18.97 | n/a | n/a | [114] | |
| 18.6 | 48 | 30 | 500 | H+ZSM-5 | 52.7 | 25.7 | 21.6 | [115] | |
| Desmodesmus communis | n/a | n/a | 20 | 460 | HZSM-5 | 8 | 42 | n/a | [116] |
| Haematococcus pluvialis | 8.98 | 10 | n/a | 600 | KCl | 12 | 60 | 28 | [117] |
| KOH | 11 | 65 | 76 | ||||||
| K2CO3 | 13 | 64.8 | 22.2 | ||||||
| MgO | 12.5 | 62 | 25.5 | ||||||
| Al2O3 | 15 | 61 | 24 | ||||||
| CaO | 13 | 63 | 24 | ||||||
| Microalgae Residue | 15 | 60 | 25 | ||||||
| Isochrysis sp. | 12.38 | 100 | 60 | 500 | CeO3 | 23 | 30 | 47 | [118] |
| Ce/Al2O3 | 25 | 32 | 42 | ||||||
| NiCe/Al2O3 | 24 | 32 | 43 | ||||||
| MgCe/Al2O3 | 23 | 31 | 46 | ||||||
| Ce/ZnO2 | 25 | 29 | 54 | ||||||
| NiCe/ZnO2 | 23 | 27 | 50 | ||||||
| MgCe/ZnO2 | 23 | 28 | 49 | ||||||
| 15 | 100 | 20 | 500 | Li-LSX-zeolite | 29 | 35 | 36 | [119] | |
| 15 | 100 | 20 | 500 | 42.5 | 33 | 24.5 | [120] | ||
| Nannochloropsis sp. | n/a | 10 | 120 | 300–500 | HZSM-5 | 25 | 38 | n/a | [121] |
| 15.17 | 50 | 60 | 400–600 | Ni–Ce/Al2O3 | 23.3 | 30.9 | n/a | [122] | |
| n/a | n/a | 15 | 500–900 | HZSM-5 | 49 | 40 | 10 | [123] | |
| N. oculata | 18 | n/a | n/a | 400–600 | Co-Mo/γ-Al2O3 | 26 | 42 | n/a | [124] |
| Oscillatoria sp. | 14.26 | 20 | 120 | 550 | TiO2, ZnO | 33.33 | 43.05 | 26.25 | [125] |
| Pavlova sp. | 12.96 | 100 | 60 | 450–550 | CeO3 | 21.07 | 47.96 | 45.92 | [126] |
| TiO3 | 20.04 | 48.18 | 45.10 | ||||||
| Ce/TiO3 | 21.67 | 47.44 | 46.26 | ||||||
| Ni/TiO3 | 22.55 | 47.66 | 45.39 | ||||||
| Co/TiO3 | 20.4 | 48.28 | 44.61 | ||||||
| 12.96 | 100 | 60 | 450–550 | CeO2 | 21.07 | 37.86 | 41.07 | [127] | |
| TiO2 | 20.04 | 39.49 | 40.47 | ||||||
| Ce/TiO2 | 21.67 | 37.46 | 40.87 | ||||||
| Ni/TiO2 | 22.55 | 37.16 | 40.29 | ||||||
| Co/TiO2 | 20.41 | 38.85 | 40.74 | ||||||
| Schizochytrium limacinum | 25.8 | n/a | n/a | 350–800 | ZYNa | 26 | 9 | n/a | [128] |
| Tetraselmis sp. | 12.07 | 100 | 60 | 500 | CeO3 | 23 | 19 | 58 | [118] |
| Ce/Al2O3 | 25 | 17 | 58 | ||||||
| NiCe/Al2O3 | 25 | 17 | 58 | ||||||
| MgCe/Al2O3 | 23 | 16 | 51 | ||||||
| Mg/ZnO2 | 23 | 18 | 59 | ||||||
| Ce/ZnO2 | 23 | 17 | 58 | ||||||
| NiCe/ZnO2 | 23 | 16 | 51 | ||||||
| MgCe/ZnO2 | 23 | 17 | 58 | ||||||
| Spirulina sp. | n/a | n/a | 50 | 350–650 | Magnetite | 49.4 | 25.4 | 25.2 | [112] |
| Activated carbon | 46.4 | 33.2 | 20.4 | ||||||
| S. platensis | 17.6 | 15 | 60 | 350–500 | ZSM-5 | 44.8 | 21.1 | 34.1 | [129] |
| MgO | 46.2 | 29.5 | 24.3 | ||||||
| 18.6 | 10 | n/a | 400 | Ce(II)/HZSM-5 | 49.7 | 20 | 30.3 | [130] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zuorro, A.; García-Martínez, J.B.; Barajas-Solano, A.F. The Application of Catalytic Processes on the Production of Algae-Based Biofuels: A Review. Catalysts 2021, 11, 22. https://doi.org/10.3390/catal11010022
Zuorro A, García-Martínez JB, Barajas-Solano AF. The Application of Catalytic Processes on the Production of Algae-Based Biofuels: A Review. Catalysts. 2021; 11(1):22. https://doi.org/10.3390/catal11010022
Chicago/Turabian StyleZuorro, Antonio, Janet B. García-Martínez, and Andrés F. Barajas-Solano. 2021. "The Application of Catalytic Processes on the Production of Algae-Based Biofuels: A Review" Catalysts 11, no. 1: 22. https://doi.org/10.3390/catal11010022
APA StyleZuorro, A., García-Martínez, J. B., & Barajas-Solano, A. F. (2021). The Application of Catalytic Processes on the Production of Algae-Based Biofuels: A Review. Catalysts, 11(1), 22. https://doi.org/10.3390/catal11010022








