1. Introduction
Millions of cars on the road are a source of air pollution. This is very important, especially in large cities and urban agglomerations due to the spread of vehicle exhaust gases in high concentrations at low altitudes in the immediate vicinity of people. Therefore, they are more harmful than industrial pollution [
1,
2]. To reduce the emission of harmful compounds, i.e., nitrogen oxides (NOx), hydrocarbons (HC) and carbon oxides (CO), car manufacturers were required to equip each newly produced vehicle with catalysts that significantly reduce the amount of hazardous chemicals in the air. Catalytic functions in car catalysts are performed by platinum group metals (PGMs) such as platinum, palladium and rhodium. The significant increase in the consumption of these metals in recent years is due to the increase in the number of vehicles equipped with catalysts. The annual supply of platinum is 169,400 kg and the demand is 229,400 kg, including 56,000 kg of recycled platinum [
3]. Therefore, the situation is not easy, and what is more there is no diversity in production—almost 70% of platinum production belongs to one country (South Africa), in which in the next two or three years is expected to decline this production is expected to decline [
4]. The situation is similar with palladium and rhodium; their supply is, respectively, 187,020 kg and 21,260 kg, whereas the demand is much higher: 287,800 kg (Pd) and 29,710 kg (Rh). Moreover, these metals are obtained from recycling at the following level: 82,950 kg of Pd and 9,180 kg of Rh [
3]. The data indicate that the amount of recycled PGMs is insufficient to cover the growing demand for these raw materials for the needs of the automotive industry; recycled materials do not cover even half (in the case of platinum) or one third (in the case of palladium) of the demand. This causes the existing reserves of these valuable raw materials to shrink.
Platinum recycling is influenced by many factors, such as the secondary materials market, legal regulations, labor costs, landfill fees, processing technologies used, as well as the quality and professionalism of the collection and dismantling system [
5]. That’s why recycling varies considerably from country to country. The largest portion of the recovered platinum group metals comes from the United States of America and Canada. This is mainly since emissions regulations were introduced very early in those countries. Japan was the next country to introduce emission regulations at the same time as the USA. In the 1980s, similar regulations were also introduced in the European Union, Australia and some Asian countries. The very restrictive legal regulations regarding the obligation to obtain specific recovery rates for end-of-life (ELV) vehicles and the mandatory removal of catalysts (these are waste intended for further segregation and treatment) have improved the situation of the European Union in the area of PGM recovery. Additional arguments in favor of recovering these precious metals are mainly their limited resources, scarcity, expensive and energy-intensive mining process and the significant amount of waste generated during this process. Platinum group metal ores contain very small amounts of these metals; less than 10 g/t (0.001%) [
6], whereas the catalytic converter of a catalyst contains approximately 2000 g/t of PGMs [
7,
8]. If comparing these two methods, it is possible to obtain the same number of platinum (1 kg) by recycling of 2 tons of spent autocatalysts or by processing 150 tons of ore [
9,
10,
11]. Assuming the recycling becomes a necessity, it is therefore important to find a rich source of these metals which seems to be auto catalysts.
2. Auto Catalyst Characteristics
Modern and well-functioning catalysts, fitted as standard in cars, should work for a maximum of 80,000–90,000 km. In practice, however, they are used much longer, although their effectiveness then drops by about 25%. The release of platinum as a result of mechanical abrasion and high temperature is the reason for the decrease in the efficiency of auto catalysts. Platinum emissions in exhaust gas reach 7–123 ng/m
3, which corresponds to 9–124 ng Pt/km and it is closely related to the speed of the car [
12,
13]. In addition, solid particles are released, whose granulometric composition is as follows: 62–67%—large grains (>10 μm, contain most of the platinum species), 21%—medium-sized particles (3.1–10 μm), 13%—small particles (<3.1 μm). The emission of platinum particles from used auto catalysts is independent of the degree of catalyst wear [
14].
The content of platinum metals in car catalysts varies considerably from 1.42 to 1.76 g/dm
3 of carrier capacity, depending on the vehicle manufacturer, model, engine capacity and even the catalyst serial number. Some cars use catalysts in which the catalytic layer is made only of palladium, only platinum, a mixture of platinum and palladium and finally platinum, palladium and rhodium together. Other catalysts are used in gasoline engines and other in diesel engines [
9,
15,
16].
Currently, most car catalysts are produced on ceramic carriers (approximately 96%), but the percentage share in the market of new generation catalysts based on metal carriers (MSC, Metal Substrate Converters), used initially in sports and racing cars, is still growing [
17]. The basic material of the ceramic carrier is a crystalline mass consisting of Al
2O
3 with the addition of other oxides, e.g., CeO
2 (15–30 wt.%) or ZrO
2 [
18], which play the role of oxygen storage promoters. Other materials used to build ceramic carriers include cordierite 2MgO·2Al
2O
3·5SiO
2, mullite 3Al
2O
3·2SiO
2, cordierite–mullite 2MgO·2Al
2O
3·5SiO
2-3Al
2O
3·2SiO
2, spinel MgO·Al
2O
3, mullite–aluminum titanate 3Al
2O
3·2SiO
2-Al
2O
3·TiO
2, [
19]. Platinum, palladium or rhodium are applied to a properly prepared carrier surface. As a consequence, this design increases the active surface, and thus the contact zone, of the catalytic substances (Pt, Pd, Rd) with the exhaust gases that flow through the carrier channels. A small addition (about 0.01%) of rare earth elements is also applied, such as lanthanum, cerium, praseodymium, neodymium, samarium, gadolinium, dysprosium, yttrium and europium [
20,
21] or barium, as stabilizers of the alumina surface area [
22]. The basic material of the metal carrier is ferritic stainless steel with chromium (17–22%) and aluminum (4–6%). The heat-resistant Fe-Cr-Al foil is coated with catalytic substances such as platinum, rolled up in a way that maximally increases the contact area with platinum. The typical structure of the ceramic carrier hole is square, while for the metal carrier the holes are triangular—see
Figure 1a,b, respectively. Both carriers have some advantages and disadvantages that are listed in
Table 1 (review based on [
17,
19,
21]).
In addition to platinum group metals, a catalyst weighing about 5 kg is also a source of steel scrap or ceramic materials, depending on the type of catalyst. The material composition of a typical ceramic and metal carrier is shown in
Table 2.
Generally, in most cases, recycled catalysts are ceramic supported catalysts that are ground and prepared properly (see
Figure 1c). The first very important stage includes homogenization. If the content of platinum and other precious metals in the input material is below 30%, it is necessary to concentrate the charge. For this purpose, various methods can be used, e.g., incineration, pyrometallurgical or hydrometallurgical processes. Some methods, however, are based on treatment of the whole catalytic carrier—in this case it is possible to encounter a carrier with a very low platinum content (below 0.02%) due to fouling (deposition of coke), poisoning (caused by strong chemical bonds between charge components, e.g., sulfur or catalytic reaction products with active places on the catalyst surface) or thermal degradation/sintering (migration of PGM located on the surface to the carrier oxides) [
23,
24]. Generally, losses of platinum group metals can occur in every stage of recycling from disassembly and collecting, through processing to PGM purification [
16,
25].
3. Methods of Auto Catalyst Treatment
Pyrometallurgical, hydrometallurgical, or mixed methods for the treatment of used catalysts are known and used in the world, but none of these methods are free from disadvantages. Generally, the selection of the recycling technique depends on catalytic carrier, PGM concentration and base metals involved in the process [
9,
26].
Hydrometallurgical methods are based on acid dissolution, chemical separation and finally purification. First, the dissolution step is conducted using different media such as aqua regia, acids like hydrochloric acid, nitric acid, sulfuric acid, along with oxygen, bromine, iodine, chlorine and hydrogen peroxide [
26]. However, due to the fact that PGMs have similar structures and chemical behavior, it is extremely difficult to separate and purify them [
27]. Therefore, such methods as precipitation, ion exchange, and solvent extraction [
28,
29,
30,
31,
32,
33,
34,
35] are often applied for the PGM separation. These methods are necessary to concentrate PGMs from leaching solutions with a relatively low level of PGM concentrations. Classical precipitation methods (using, for example, aluminum as a reducing agent) are characterized by poor selectivity, numerous unit operations, recycle streams and refining steps [
26]. Solvent extraction (liquid–liquid extraction), applying a variety of extractants such as Alamine 336, LIX 841, Cyanex 921, TBP, Aliquat 336, oximes, amines and phosphoric acid, is more appreciated because of higher selectivity, and involves a scrubbing step to achieve high metal purity and complete removal of metals by a multi-stage extraction step [
26,
30]. Generally, solvent extraction is one of the most versatile methods in terms of separation efficiency and environmental impact [
29,
30]. Hydrometallurgical methods result in the formation of waste solutions that are hazardous to the environment and require proper management. For example, the commercially used leachate reagent aqua regia generates NOx, chlorine and acid fumes during leaching [
36]. Additionally, there is generation of fumes and Cl
2 due to a high concentration of HCl in the leaching solution. In order to solve this problem, sulfuric acid and salts of chloride ions (AlCl
3 and NaCl) were used to form a stable chloro-complex; the efficiency of platinum recovery is at the same level, whereas this consumes a large amount of acid and the alkali used, and what is more, it requires a high temperature and pressure [
36]. There are still studies being condcuted to improve these processes before launching commercially (e.g., the leaching time could be reduced by using microwave-assisted heating [
4,
37]) and they should be evaluated regarding the environmental impact.
Pyrometallurgical methods are commonly used for commercial purposes. They can be divided into three groups: smelting using various collector metals, chlorination and sintering. The sintering method is conducted in the presence of plasma (T > 1200 °C, 2–3 h) in a constant flow of nitrogen as the central plasma gas. Of all pyrometallurgical processes, sintering is considered to be the least polluting one [
23,
38]; however, it is rather used for reforming of the spent catalyst. The chlorination method is based on volatilization of PGMs from the spent catalyst by selective chlorination with chlorine or chlorides at ahigher temperature (from 250 to 700 °C), and then condensation in a cooler zone, and adsorption on an activated carbon bed [
39]. The process, however, has some serious disadvantages such as: strong corrosion of furnace and apparatus, and health and environmental risks. Hence, application of sintering and chlorination methods is rather limited. Therefore, smelting processes are popularly used. They can be also divided into three groups: using collector metals such as lead, copper, nickel, iron, copper matte; using PCB (Printed Circuit Boards) collection; and using collector metals such as zinc, calcium, magnesium in the gaseous form [
40,
41,
42,
43,
44,
45,
46,
47,
48,
49,
50]. Generally, the use of metal vapors is considered as a pretreatment method to reduce the use of chemical agents and the environmental load involved in the recovery process [
40]. Calcium [
42] and magnesium [
43] vapors form compounds with a catalytic carrier containing alumina and silicon oxide, whereas zinc reacts with PGMs selectively [
40,
41]. The process relies on the strong affinity of PGMs with alkaline earth elements; what is more, PGMs in compounds with calcium or magnesium dissolve at an ambient temperature more than in their pure form [
42]. Considering only the recovery process, the efficiency of the process using magnesium, calcium and zinc is rather low (below 80%); therefore, using them is justified for pretreatment [
8] because they are effective in increasing the rate of dissolution [
42]. One of the last methods in this group is based on iron chloride vapour treatment [
44], in which PGMs were successfully concentrated by magnetic separation after the iron chloride vapor treatment [
44].
The classical collector metal method, which uses liquid metal for collecting PGMs, is effective however not suitable for extracting platinum directly from solid scrap. The platinum and other precious metals are transferred into collector metal, whereas the ceramic part of the carrier goes to the slag. Usually, slag is easily removed from the collector metal with PGMs. Subsequently, alloy with PGMs is dissolved in an aqueous solution and this is consequently followed by separation and purification processes [
44]; the same process is used after hydrometallurgical treatment [
26]. This process is commonly used in many plants such as Umicore, Johnson Mathhey, Badische Anilin-und-Soda Fabrik, MultimetcoInc (USA), Techmet (USA), Nippon PGM Co (Japan) [
23,
26,
45,
46]. When choosing the collector metal, the following parameters must be taken into consideration: melting points, chemical properties of the collector metal, mutual solubility. Lead, copper, nickel, iron, matte and other materials containing the mentioned metals (for example PCB) can all be used as collector metals. There was stated two different mechanisms of this process: a) wetting of micro-dispersed particulates of Platinum Group Metals in the slag by molten metal, creation of metal droplets with attracted microparticles of PGMs, settling of the molten droplets of the created alloy through the slag at the bottom of the furnace; and b) settling of solid Platinum Group Metal microparticles through the molten slag, creation of a solid solution with the molten metal at the bottom of the furnace [
46]. Kolliopoulous et al. [
46] stated that platinum recovery is rather conducted according to wetting mechanism, whereas palladium and rhodium are recovered basing on a balanced mixture of both mechanisms. In the case of different metals used as collector metal, lead was used as the oldest one, however, their biggest disadvantage is the detrimental impact on the environment. Short characteristics of different collector metals based on literature review [
23,
26,
45,
47,
48,
49] are presented in
Table 3.
The new trend in collector metal method is using PCB metal components (copper, tin, iron) as the collector metal, at the same time applying spent auto catalyst as a slag formative. Kim et al. [
50] stated that the process is simple and economical compared to the conventional process. The additional advantage is recovery of valuable metals from spent PCB.
Despite numerous methods applied for recovery of Platinum Group Metals from auto catalysts, finding an effective method is nowadays an important and extensively investigated problem. The conducted scientific research was aimed to develop a relatively simple and comprehensive technology for the recovery of platinum from spent auto catalysts based on a modified collector metal method that could be used in Polish scrap processing plants.
4. Method of Collector Metal with mhd Pump
One of the interesting and effective solutions can be the collector metal method using a magneto-hydrodynamic (mhd) pump applied for intensification of this process. This solution has been applied to our previous experience in the recovery of PGMs from auto catalysts [
2,
9,
51,
52,
53]. This method enables the processing of all types of auto catalysts available on the market, without having to send them abroad to specialized plants.
Preliminary tests enable the selection of the optimal metal collectors that can be successfully used in research aimed at modifying this method using magnetohydrodynamic phenomena. Due to the most promising results of platinum recovery, two metals were selected in preliminary tests: copper and lead. Copper is a more environmentally friendly metal than lead, however, it has a higher melting point.
This process involves the accumulation of platinum in the liquid lead. In the analyzed process, the content of platinum in the liquid lead is low. The Pb–Pt alloy below 2042 K is a double alloy that has limited liquid solubility and also limited solid solubility. Analyzing the Pb–Pt solubility charts available in the literature [
54,
55], it can be concluded that, at the temperature at which the process is carried out (673 K), nearly 15% by weight of platinum can be dissolved in the liquid lead.
In the discussed solution, the same metal is used to flush a large number of catalysts, which guarantees the appearance of platinum in the liquid metal solution, and then an increase in the concentration of platinum to values that ensure the profitability of their extraction from liquid metal. This allows the use of the device for a very long time before it is necessary to remove the platinum from the lead circulating in the device channel. The use of closed loop liquid lead guarantees a reduction in the environmental impact of the process. The liquid metal is placed in a closed-loop annular channel in which the spent catalysts are completely immersed (see
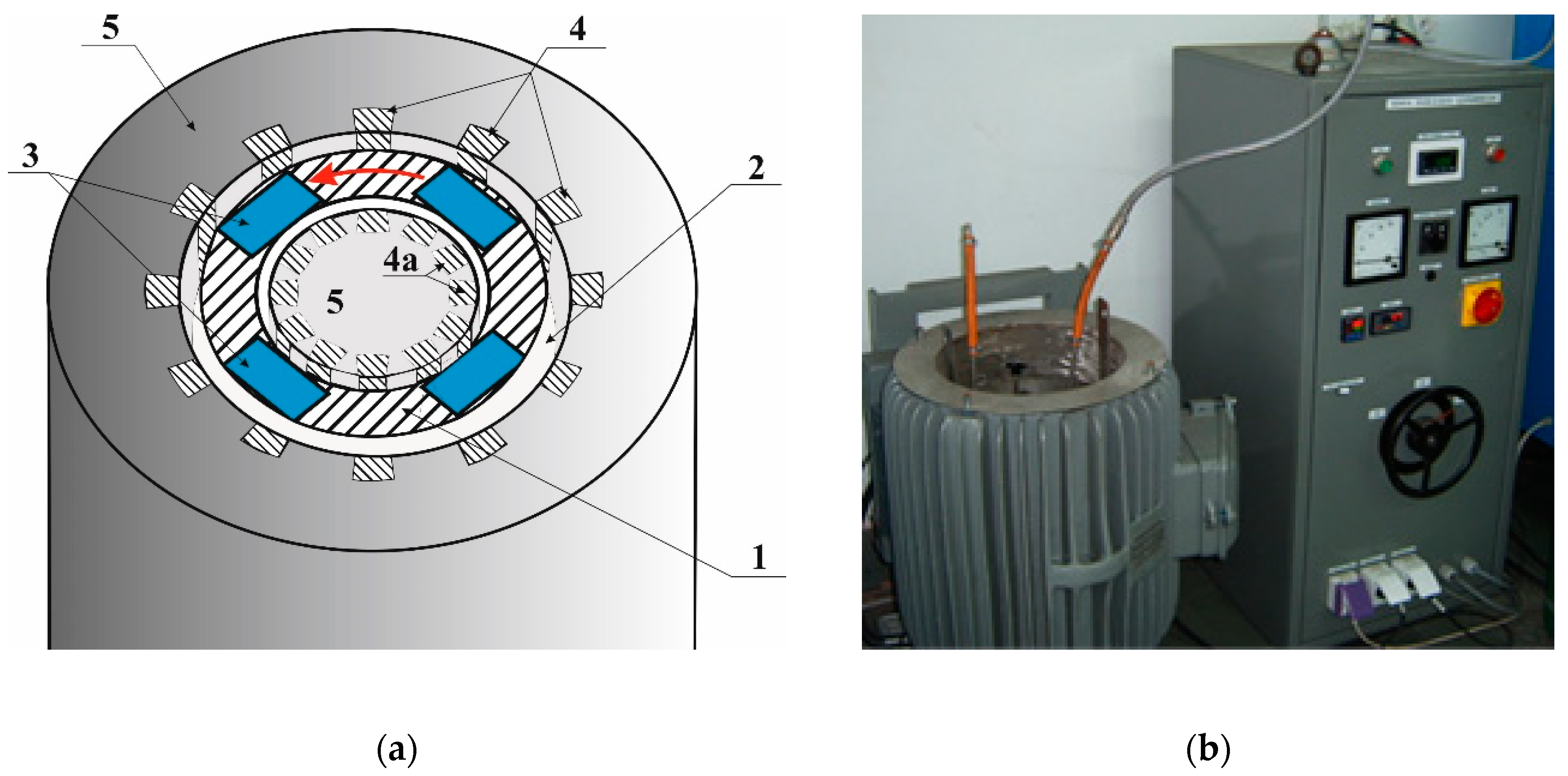
Figure 2). The liquid metal is set in motion using a rotating electromagnetic field generated in the inductor and it flows through the car catalytic converter channels accumulating platinum.
Appropriate effectiveness of the device is ensured by proper selection of operating parameters, which required the analysis of a complex of issues related to the electromagnetic field (EM field) and flow field calculations. It was important to consider the coupling of the electromagnetic field and the hydrodynamic field. Considering the fact that in the studied process there are low values of magnetic Reynolds number—and also that phase separation surface, which is moving during the infiltration of the catalyst carrier, occurs only in the area of this carrier and does not affect the distribution of the electromagnetic field (since both the air and metal phases have the same zero conductivity)—it was possible to use weak, one-way field coupling. This made it possible to split the simulation into two separate stages—a quasi-static electromagnetic field simulation providing the distribution of electromagnetic forces acting on the liquid metal, and a stage of non-stationary hydrodynamic simulation [
56,
57].
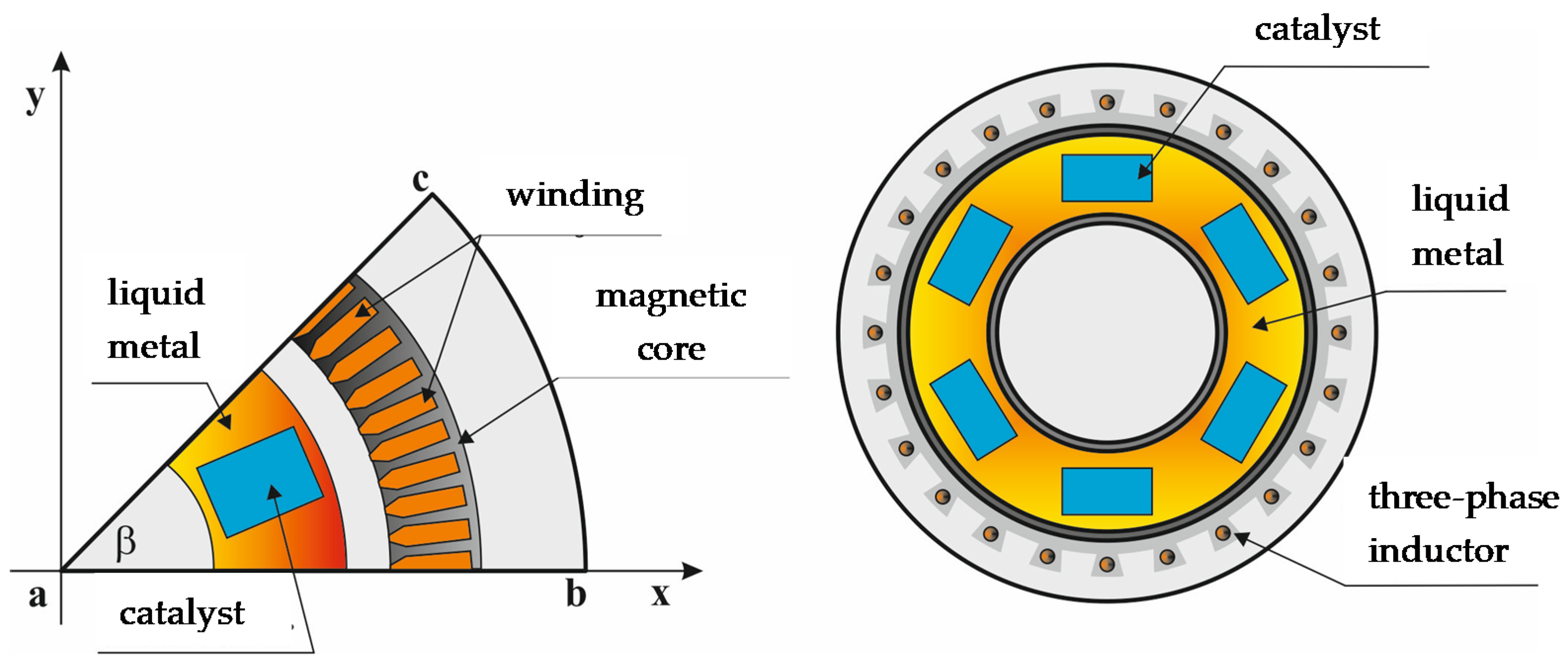
The main purpose of modeling was to determine process parameters such as: power frequency, inductor current, gaps between the inductor and the liquid metal channel, and their impact on flow parameters. Due to the geometric and electrical symmetry of the system, the model was limited to 1/8 of the entire device (see
Figure 3).
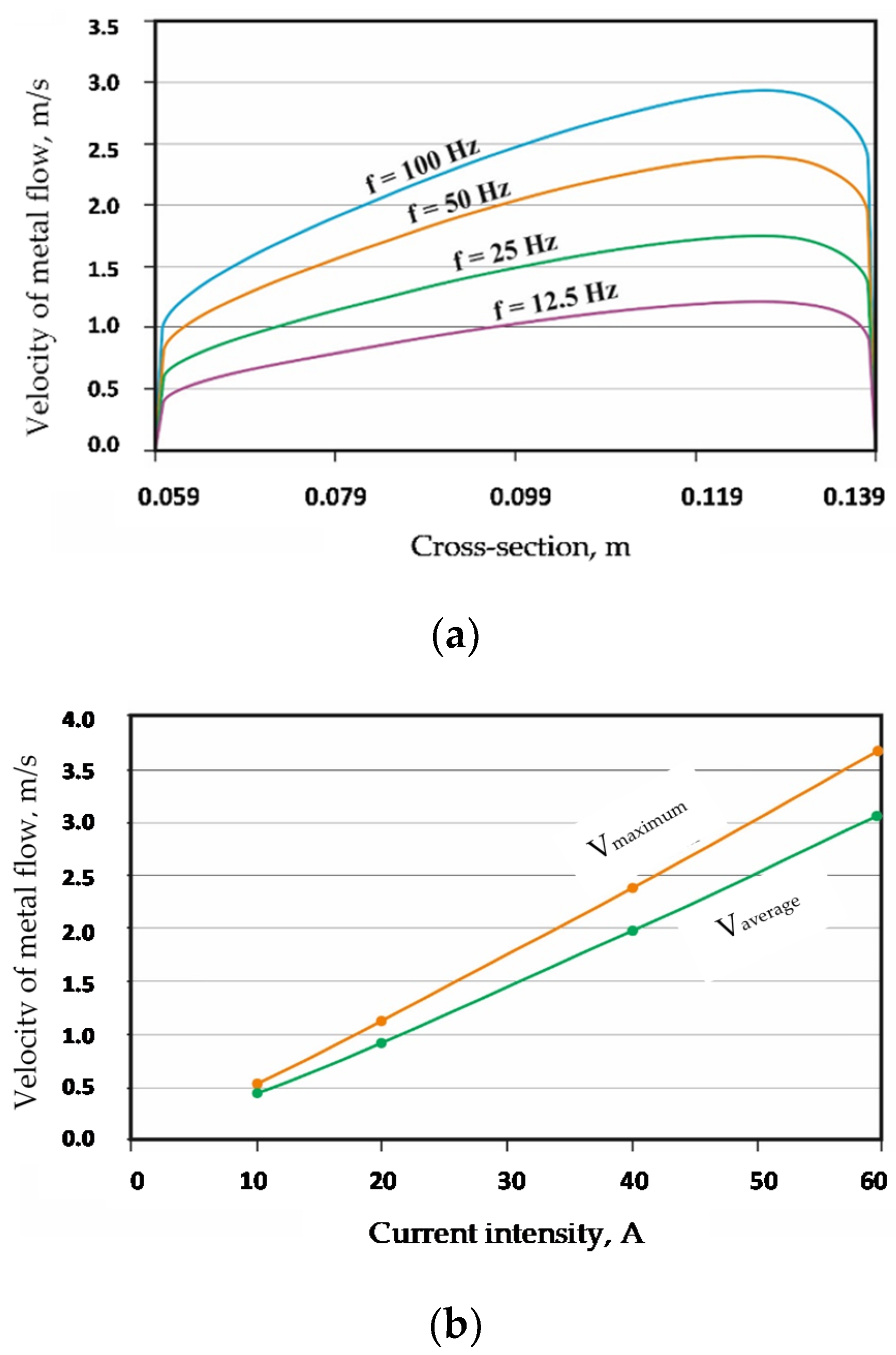
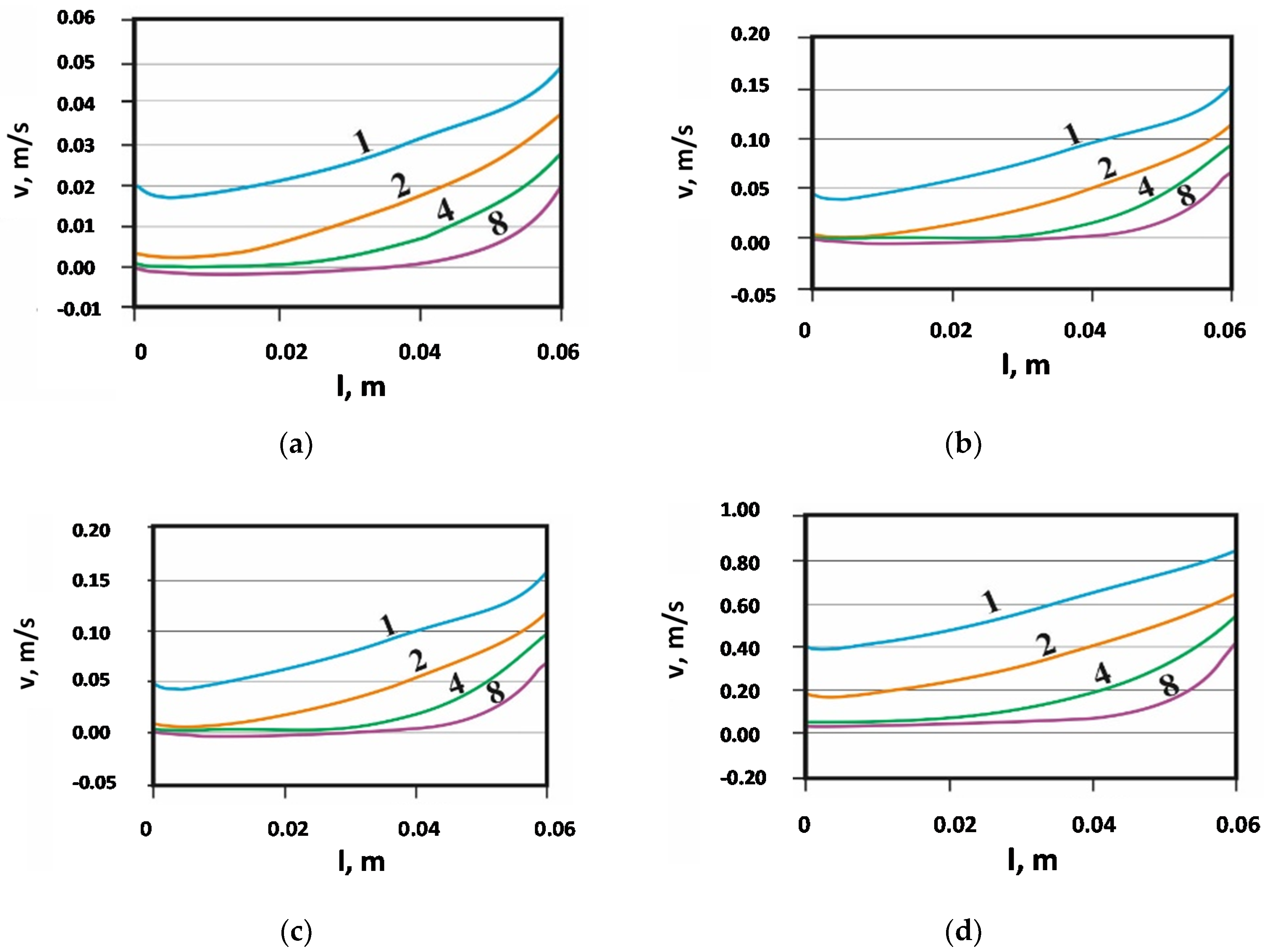
The analysis of the electromagnetic field and the flow field showed that, among the tested options, the best way to change the metal velocity in the channel is to change the inductor current, while the effect of frequency was smaller (see
Figure 4). The change in the inductor current had little effect on the liquid metal velocity profile in the device channel. This means that, in such a system, viscosity and friction have a greater impact on the distribution of velocity in the channel than the distribution of magnetic forces.
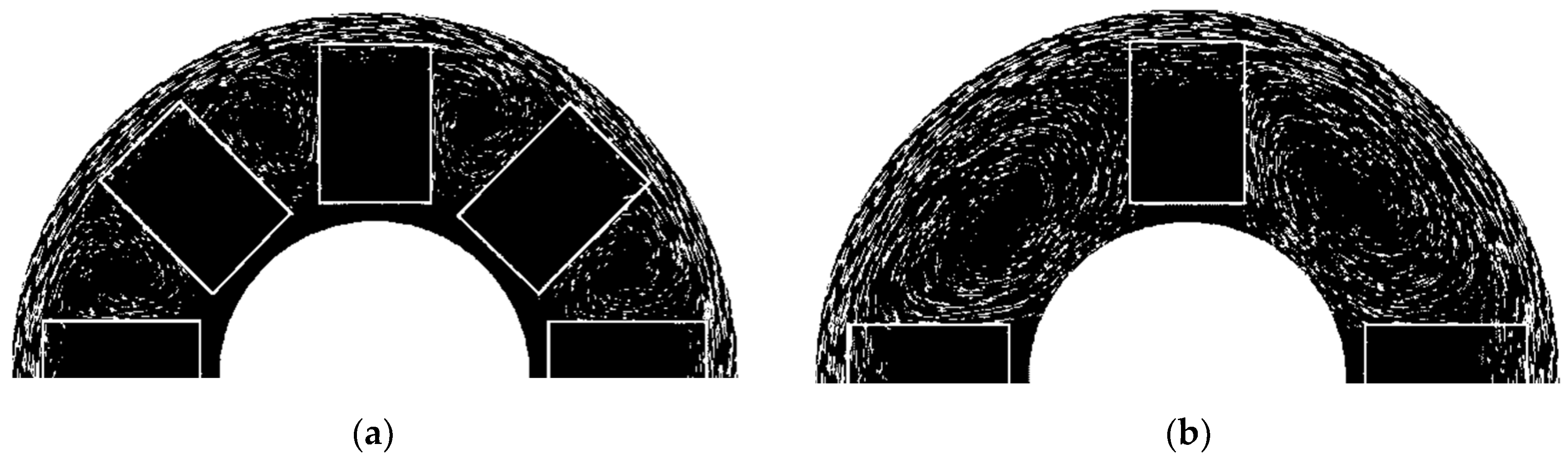
Results of calculations have shown that in the case of too many catalysts in the channel, and thus too small distances between them, the metal did not obtain sufficient momentum to overcome the capillary pressure and then the flow resistance through the catalyst carrier channels (see
Figure 5). In the case of four catalysts, a backflow phenomenon was observed, which also provided washing out of platinum from the carrier (see
Figure 5b). Further reduction in the number of catalysts caused a loss of backflow, and additionally, for such a small number of catalysts in the channel, it made this process unprofitable. This situation was improved by increasing the inductor supply current, however at the expense of increased energy consumption. Energy parameters such as frequency and current practically did not change the structure of the flow of liquid metal in the channel of the device—see
Figure 6. The change in current did not cause a significant change in the velocity and structure of the liquid metal, which can be seen in
Figure 6b,c. Changing the frequency did not affect the structure—for all variants regarding the number of catalysts in the channel, the shape of the curves and the intersection with the horizontal axis were identical, only the metal velocity decreased as the frequency decreased.
From the cost-effectiveness of the process, the duration of washing out catalysts with liquid lead is also important; it was found that about twenty seconds after switching on the inductor power supply, the average metal velocity, and thus the flow structure in the channel, stabilized and the final saturation of the catalyst structure was obtained.
5. Modification of Collector Metal Method with mhd Pump
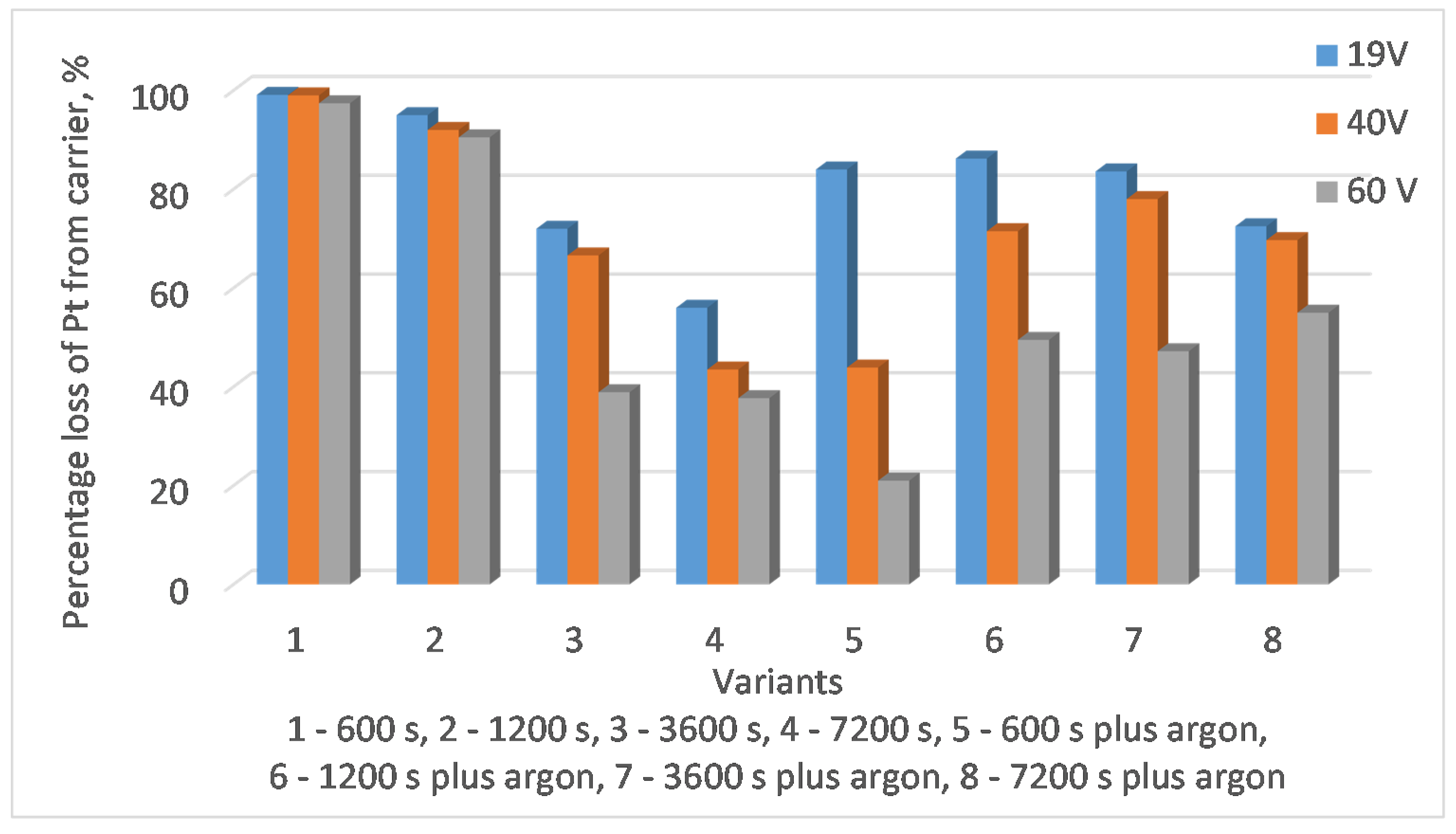
The next step was to conduct experimental research to test the device and modify it. Investigations of platinum recovery from used auto catalysts using magnetohydrodynamics included tests carried out at a temperature of 673 K. When the lead in the channel of the device reached this temperature, the actual experimental tests were started. The duration of the tests ranged from 600 to 7200 s. Initial tests showed that less than 600 s was not sufficient for lead to penetrate the catalyst channels. In addition, carrying out the tests in less than 600 s was logistically difficult (loading catalysts, obtaining full lead flow, changing the direction of lead flow, catalyst replacement). Therefore, this time was used as a reference point in the research. Each washing test was performed with liquid lead flowing clockwise and counterclockwise (i.e., a washing test of 600 s—it consisted of a spinning test left for 300 s and right for 300 s). Experiments were also carried out with argon blowing to avoid the formation of oxides on the surface of the liquid lead. The tests were carried out for three values of secondary voltage on transformer covers, equal to 19, 40 and 60 V, respectively.
Figure 7 presents results obtained during the research, the desired result is the reduction in the platinum content in washed carriers. The presented research results confirm the achievement of such an effect. It is worthless to estimate the increase in platinum content in the collector metal due to the fact that a huge number of carriers would have to be washed to see a real effect. At the device operating temperature, there was no risk of platinum being lost by evaporation of lead during the process. The formation temperatures of volatile platinum oxides are higher than the process temperature, similar to pure lead—its evaporation temperature is 2022 K. To prevent lead oxidation, an argon and nitrogen purge was used.
In some tests, the effect of secondary platinum deposition could be observed when the capillaries were washed through by the collector metal. This effect was much more visible for longer processing times and higher secondary voltage. Therefore, the optimal solution is a shorter time for washing the catalysts, this prevents the platinum from being re-applied to the capillary structures of the catalyst, and also improves the cost-effectiveness of the process. Much better results were obtained for tests with a secondary voltage of 19 V (than for 40 V or 60 V), this is due to the fact that in this case the lead centrifugation speed was lower, it did not disturb the metal flow, and the efficiency of metal collector penetration into the capillary structures of the carrier was higher.
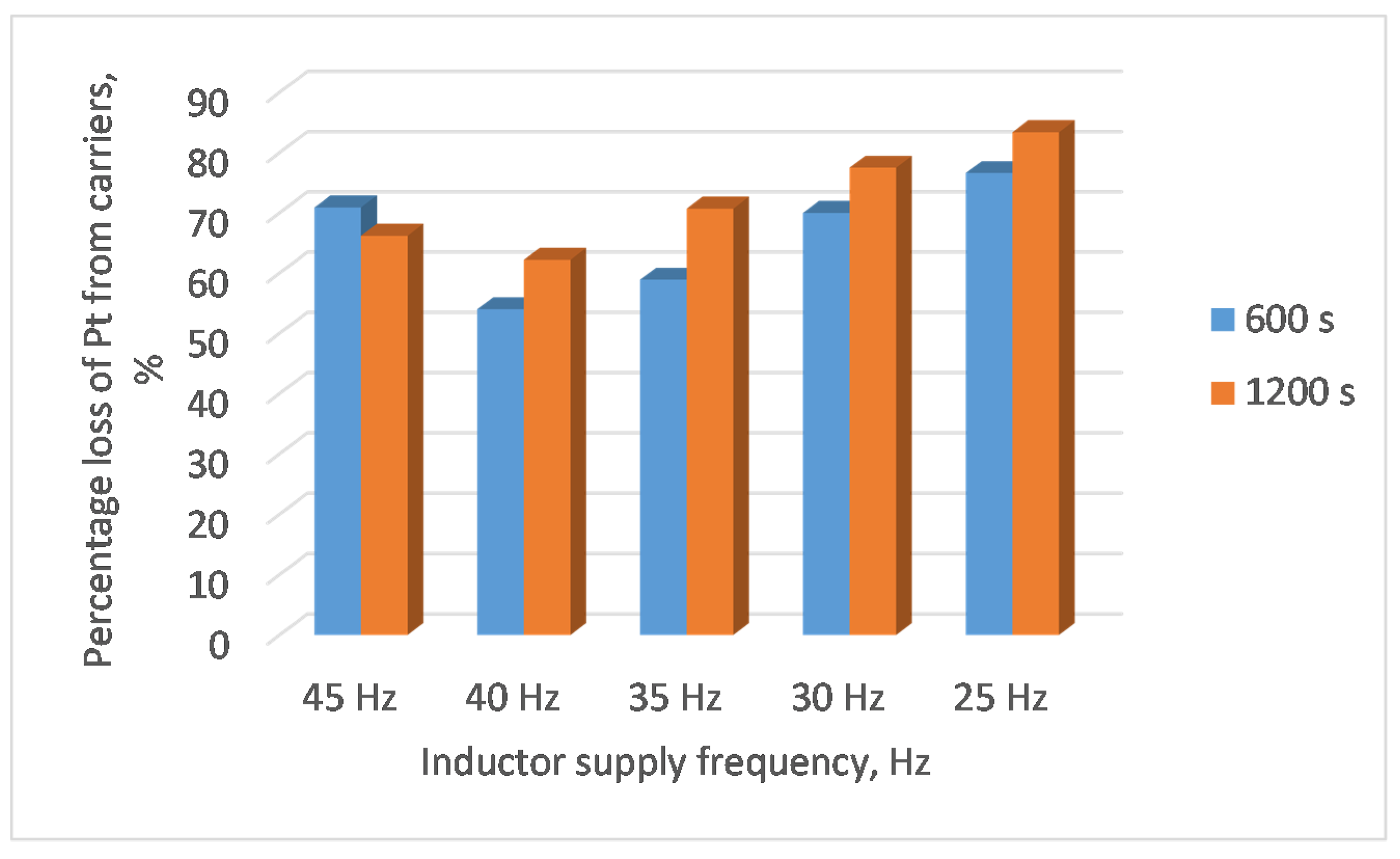
A research test was conducted with different inductor supply frequencies. Changing the frequency practically does not affect the flow structure of the liquid metal in the channel. This was also confirmed by the distribution (
Figure 4) of the velocity of the liquid metal in the channel over the entire width of the catalyst (l = 0–0.06 m). However, an increase in frequency caused an increase in the maximum values of the forces acting on liquid lead at the device walls for both types of field, i.e., transverse and longitudinal.
Figure 4 is the result of numerical research, which allowed the selection of experimental research. Therefore, in laboratory tests, the frequency of 12.5 Hz was rejected and not included in the tests. At the same time, the frequency value of the device (50–100 Hz) generates too high a liquid metal velocity in the channel, this is disadvantageous due to the difficulty of wetting the catalyst capillaries and problems with liquid lead splashing.
Figure 8 shows the obtained results.
The increase in the frequency of the current supplying the device increased the efficiency of the system (greater energy was generated by the collector metal rotating), this forced an increase in the mixing speed of the liquid metal, which made it difficult to wet the capillary carriers with lead. Therefore, in the presented experiment it is advisable to use lower frequencies, although they worsen the coupling between the charge and the supply system.
6. Improvements of Collector Metal Method with mhd Pump
In order to improve the profitability of the presented process, attempts were also made to optimize the operation of the device using heat balance. The process was powered in the laboratory version by electricity. The total power supplied to the system was a maximum of 2900 W. After reaching the operating temperature, the power of the heating system was limited to 1100 W, and the supplied stream of electricity was used to heat a new portion of the processed batch and to maintain the set operating temperature. The mass of molten lead in the reactor was not only used to dissolve the platinum contained in the ceramic matrix of the spent catalyst but also as a heat accumulator.
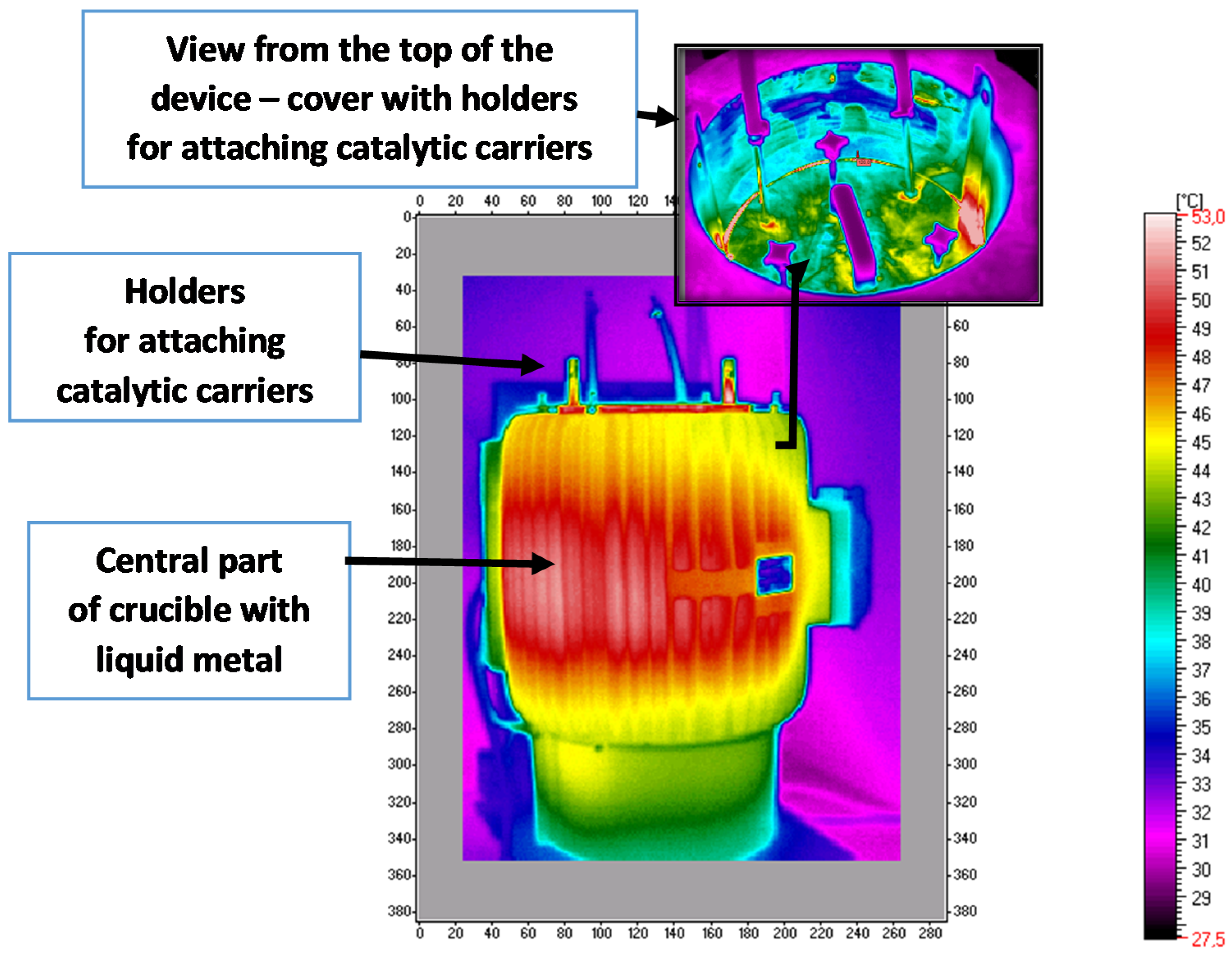
Taking into account the duration of the periods between the replacement of a new portion of the charge and the energy stream needed to heat the crucible with the bath to maintain the set temperature, the efficiency of the heating process was determined from the energy balance, which was about 28% on average. The remaining part of the supplied energy stream was lost to the environment. The aggregate efficiency determined was comparable to the efficiency of heating processes in typical industrial technologies. Heat losses to the environment took place mainly through the liquid metal mirror by convection and radiation during the loading of a new batch of catalyst. During the determination of heat losses to the environment from the working aggregate [
58], a thermographic analysis of the temperature distribution was used, and an example image is shown in
Figure 9 (based on [
58]).
The most intense heat losses in the discussed device occurred through the top cover, and it is also a place where it was possible to intervene to increase the energy efficiency of the device. A high-temperature ceramic fiber plate was used as the insulation material, which is characterized by high thermal and chemical resistance. Based on thermodynamic calculations, it was shown that the maximum insulation thickness to which it pays to insulate the device cover was 0.03 m, and the maximum efficiency that the device can achieve was a limit of 36%. The above procedures allowed an increase in the device’s efficiency by 8% compared to the initial state, while further increasing the thickness of the insulation layer on the cover was unprofitable.
The efficiency of the collector metal method with the mhd pump could be improved by using a lead with the addition of a light metal, for example lithium, for intensifying the extraction process of platinum and palladium from the surface of catalyst carrier capillaries. An important aspect of the research should, therefore, be the determination of Pt-Li, Pd-Li, because the thermodynamics properties of such a system are extremely limited and, particularly, phase diagram of Li-Pt and Li-Pd has not yet been studied in detail. Since that, thermodynamic investigations such as calorimetry and electromotive investigation are necessary to determine the reliable phase diagram in the future. As a consequence, the determination of thermodynamic properties of ternary alloys (Pb-Pt-Li, Pd-Pb-Li) will be necessary.
Lead alloys with the addition of lithium increase the extraction of platinum and palladium from thin catalytic layers as a result of reducing the surface tension of the extraction liquid (Pb-Li), improving its penetration deep into capillaries with catalytic layers and accelerating the dissolution of platinum in the Pb-Li alloy, as a result of the greater affinity of lithium for Pt and Pd compared to lead.
Figure 10 shows the preliminary results of the research. The calculated surface area of Pb-Li alloy droplets with different amounts of Li: 4%, 8%, 12% and 16% in all wetting tests of the platinum coated surface was 3.261 mm
2, 3.466 mm
2, 3.422 mm
2, 2.356 mm
2, respectively, compared to pure lead with a value of 3.133 mm
2. The size of Pb-Li alloy drop surface area was calculated as the arithmetic mean of three attempts to determine the surface area. The obtained test samples were photographed along with the scale, and then placed in the Fusion 360 graphic program. The software allows the determine of the surface of the drop by tracing its contour. The drops were also processed on a stereoscopic optical microscope to determine their dimensions. Both results, both the droplet dimensions and the plotted area with the calculated surface area are presented in
Figure 10.
Such studies are at an early stage but the obtained results are satisfactory; however, only further research will be decisive in this respect.
7. Conclusions
The developed method is a universal method (it can be used for both types of catalyst carriers available on the market), and the operating temperature of the device is much lower than for conventional methods. This solution does not require the milling of spent catalysts before washing out of platinum. This enables the elimination of the costs associated with their grinding, and also does not require dust removal, which eliminates the loss of PGMs. Due to the small amount of platinum in a single catalyst, it is reasonable to wash platinum from multiple catalysts simultaneously in one process using the same liquid metal stream in a closed loop.
The use of the same metal to wash out a large number of catalysts allowed the platinum group metals to accumulate in liquid lead, followed by an increase in the PGM concentration to values that guarantee the profitability of their extraction from collector metal. The use of a closed liquid metal loop has reduced the environmental impact of the process.
Experimental studies on platinum recovery using an mhd pump with the use of mains frequency current gave satisfactory results, both in terms of mixing intensity and maintaining the temperature of the metal bath in the tank at the required level during mixing. In some tests, the effect of secondary deposition of platinum was observed when the capillaries were washed out by the metal collector. This effect was more visible during longer times of the conducted process. Therefore, the time has been shortened to prevent the re-application of platinum on the capillary structures of the catalyst, and thus improve the cost-effectiveness of the process.
Much better results were obtained for tests with lower voltage, which is explained by the fact that, in this case, the lead centrifugation speed was the smallest, it did not disturb the metal flow, and the efficiency of the collector metal penetration into the capillary structures of the support was higher. In tests of the recovery of platinum from used auto catalysts with a different operating frequency of the device, it was found that the frequencies that generates too high a liquid metal velocity in the channel, which is unfavorable due to the difficulty of wetting the catalyst capillaries and problems with liquid lead splashing.
Further research is to be conducted to improve the process energetically and improve its efficiency, e.g., by modifying the collector’s metal with lithium to obtain better wettability of this alloy.