2.1. Characterization of the Prepared and Modified TiO2 Nanoparticles
The photocatalytic material development process and degradation for MB dye by TiO
2-NH
2-Cu
xO nanophotocatalyst is illustrated in
Figure 1. The preparation method includes three main steps: development of porous TiO
2 nanoparticles as a core material, modification of the surface of TiO
2 nanoparticles, and the bonding of amine groups to Cu
xO which resulted in a homogeneous distribution of Cu
xO on the TiO
2 surface to improve the photocatalytic degradation process.
The crystallinity phase of the nanophotocatalyst was studied using powder X-ray diffraction, and the results in
Figure 2a shows the XRD diffractograms of TiO
2, TiO
2-NH
2, and TiO
2-NH
2-Cu
xO nanophotocatalysts. The peaks revealed at 2 theta values are 25.26 (101), 37.90 (004), 47.91 (200), 54.63 (105), 62.21 (213), 69.21 (116), 75.015 (215), and 82.1 (303) represents the existence of the anatase phase TiO
2 according to the Joint Committee on Powder Diffraction Standards (JCPDS) card no. 21-1272. The X-ray diffraction (XRD) analysis demonstrates that the chemical modification of the TiO
2 nanoparticles did not change the anatase crystalline phase. This observation indicates that the surface modification and addition of copper did not change the TiO
2 crystalline phase, which could be due to the low concentration of copper (1.8 at %), and/or the phase intensity of TiO
2 and/or the high homogenous dispersion of copper on the TiO
2 surface. The calculated crystallite size of TiO
2, TiO
2-NH
2, and TiO
2-NH
2-Cu
xO nanophotocatalysts was about 8 ± 1 nm. No change in crystallite size was observed before and after surface modification. This is could be due to the significant interaction between copper and TiO
2 via the amine bonding that formed the copper imine groups and hydroxyl groups on TiO
2 surface, and this interaction controls or prevents the further growth of crystallization [
25]. The crystallite size in TiO
2, TiO
2-NH
2, and TiO
2-NH
2-Cu
xO nanophotocatalysts was measured according to the Scherrer Equation
where k is the Scherrer constant, λ is the wavelength (Cu; 1.5406), β is the line broadening of the XRD peak, and θ is the diffraction angle. The FWHM for the prepared TiO
2 nanoparticles was 1.332, and the crystallite size was 6.79 nm. However, when the TiO
2 was modified with 3-aminopropyltrimethoxysilane to form TiO
2-NH
2, the FWHM reduced to 1.135 and the crystallite size increased to 7.97 nm, while when the copper was attached to the surface to form TiO
2-NH
2-Cu
xO nanophotocatalyst, the FWHM was 1.177 and the crystallite size 8.92 nm. This variation is owing to the surface modification process as both TiO
2-NH
2 and TiO
2-NH
2-Cu
xO nanophotocatalysts show the increment in crystallite size of ~1 nm. Additionally, the Cu diffraction peaks are not observed in the diffraction pattern due to the low copper content of 1.8 at %, as well as the phase intensity of the TiO
2, or due to the similarity in the radius of Ti
4+ and Cu
2+ (0.068 and 0.073 nm).
For further understanding of the surface chemical modification and the formation of TiO
2-NH
2-Cu
xO nanophotocatalyst, Fourier-transform infrared spectroscopy (FTIR) spectra were recorded on freshly synthesized, surface modified, and Cu
xO added TiO
2 nanoparticles (
Figure 2b). The peaks of TiO-Ti for the anatase phase were observed at 752 and 1450 cm
−1. The peaks at 1650 and 3410 cm
−1 are attributed to the OH stretching vibration of the Titania [
26]. In the case of TiO
2-NH
2, several additional peaks are observed for CH
2, Si–O from the silane coupling agent, and N–H along with the TiO
2 peaks. In detail, the peaks at 2980 and 2888 cm
−1 are attributed to CH
2 in the silane coupling agent and the peak at 1040 cm
−1 is ascribed to Si–O from the silane coupling agent [
27]. The peaks at 1550, 3039, and 3450 cm
−1 are attributed to N–H. In the case of TiO
2-NH
2-Cu
xO nanophotocatalyst, the copper ions attached to the amine on the TiO
2 surface have peaks at 756 and 793 cm
−1 which belong to CH
2 and the peaks at 1047 and 1080 cm
−1 are attributed to Si–O. The peak at 1410 cm
−1 is ascribed to the OH group. The peaks at 1438, 2923, and 2970 cm
−1 are ascribed to CH
2 alkanes stretching. The observed bands at 3100 and 3410 cm
−1 belong to C–H and N–H, respectively [
28]. These results confirm the modification of TiO
2 with the silane coupling agent [
29]. Furthermore, when the copper was attached to the TiO
2 surface, the shift of some bands was observed, such as the band at 649 cm
−1, which was shifted to 643 cm
−1, the band at 1400 cm
−1 was shifted to 1410 cm
−1, and the band at around 1609 cm
−1, which belongs to N–H, was hidden due to the copper mask. These results showed that the chemical attachment of amine to the TiO
2 occurred and the interfacing between the amine group and Cu
xO succeeded. In addition to FTIR, micro-Raman spectroscopy was also applied to understand the molecular level of bonding in the chemically modified material, as shown in
Figure 2c. The peaks at 144, 197, 399, 515, 519, and 639 cm
−1 are ascribed to the symmetric anatase phase of TiO
2 particles [
30]. The band at 1050 cm
−1 in TiO
2-NH
2 and TiO
2-NH
2-Cu
xO nanophotocatalyst is attributed to C–C aliphatic chain vibrations, the band at 1380 cm
−1 is attributed to CH
3. Both FTIR and micro-Raman results confirm the modification of the TiO
2 surface with the amine silane coupling agent.
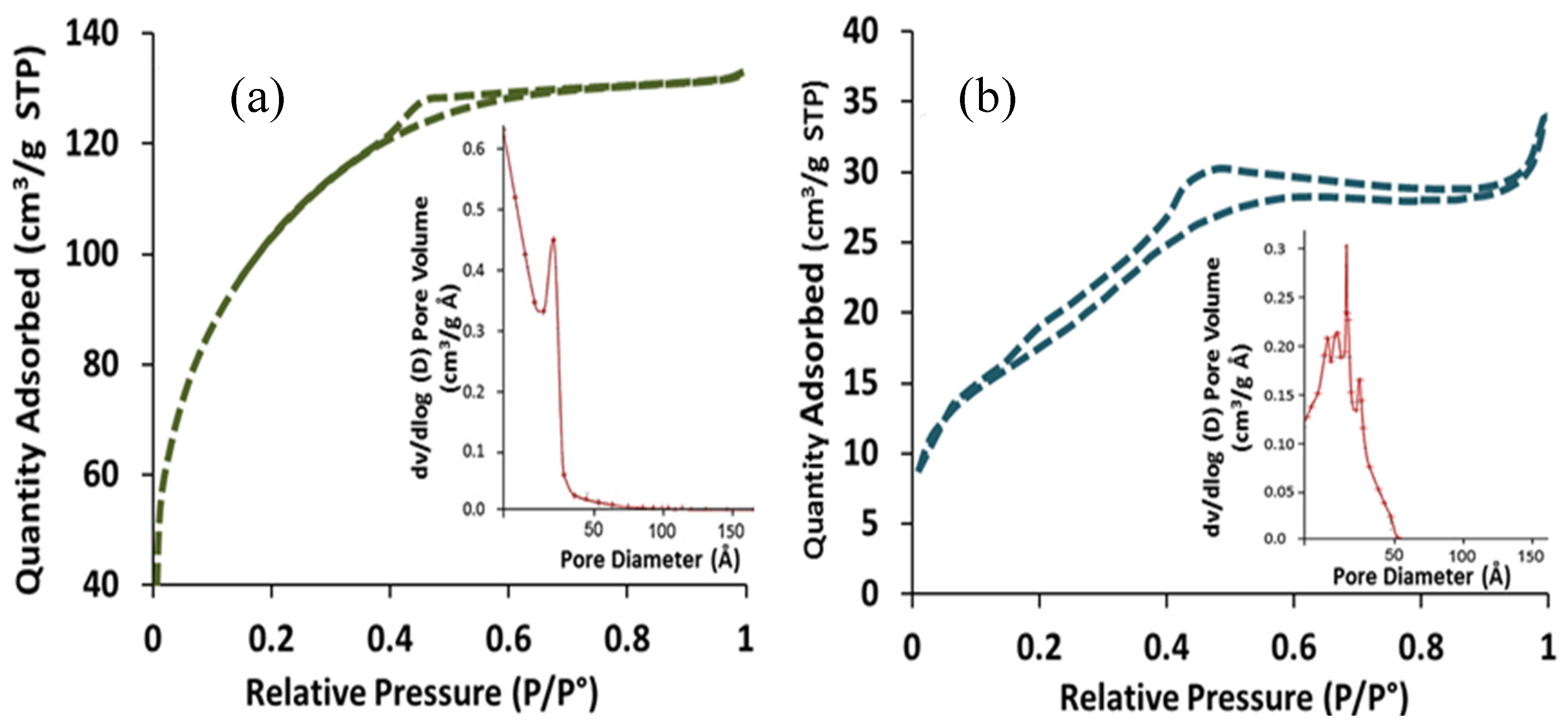
The Brunauer, Emmett and Teller (BET) specific surface area and pore size of the TiO
2 and TiO
2-NH
2-Cu
xO samples were examined using nitrogen adsorption–desorption isotherms at 77 K, and the calculated values are shown in
Table 1. The results revealed that the specific surface area of the as-synthesized TiO
2 nanoparticle was 337 m
2g
−1 which is comparatively higher than other reported TiO
2 nanoparticles [
31]. The isotherm of TiO
2 nanoparticles is illustrated in
Figure 3a and shows the multilayer adsorption pursue capillary condensation consequential type IV isotherm, which confirms the mesoporous phase of the synthesized materials. Also, the measured pore volume and pore diameter of the TiO
2 material was 0.322 cm
3g
−1 and 4.77 nm, respectively. Furthermore, the distribution of the pore size in
Figure 3a (inset) showed a very narrow distribution which indicates a uniform pore distribution. On the other hand, the surface area of TiO
2 nanoparticles decreased from 337 m
2g
−1 to 97 m
2g
−1 after surface modification with an amine (TiO
2-NH
2), and further attachment to Cu
xO (TiO
2-NH
2-Cu
xO). The reduction in surface area was mainly due to the deposition of surface functional groups, and attachment of Cu
xO on the surface of the TiO
2 nanoparticles which could cause a barrier and obstacles to the N
2 molecules entering through the pores and cause a non-uniform pore distribution, as shown in
Figure 3b.
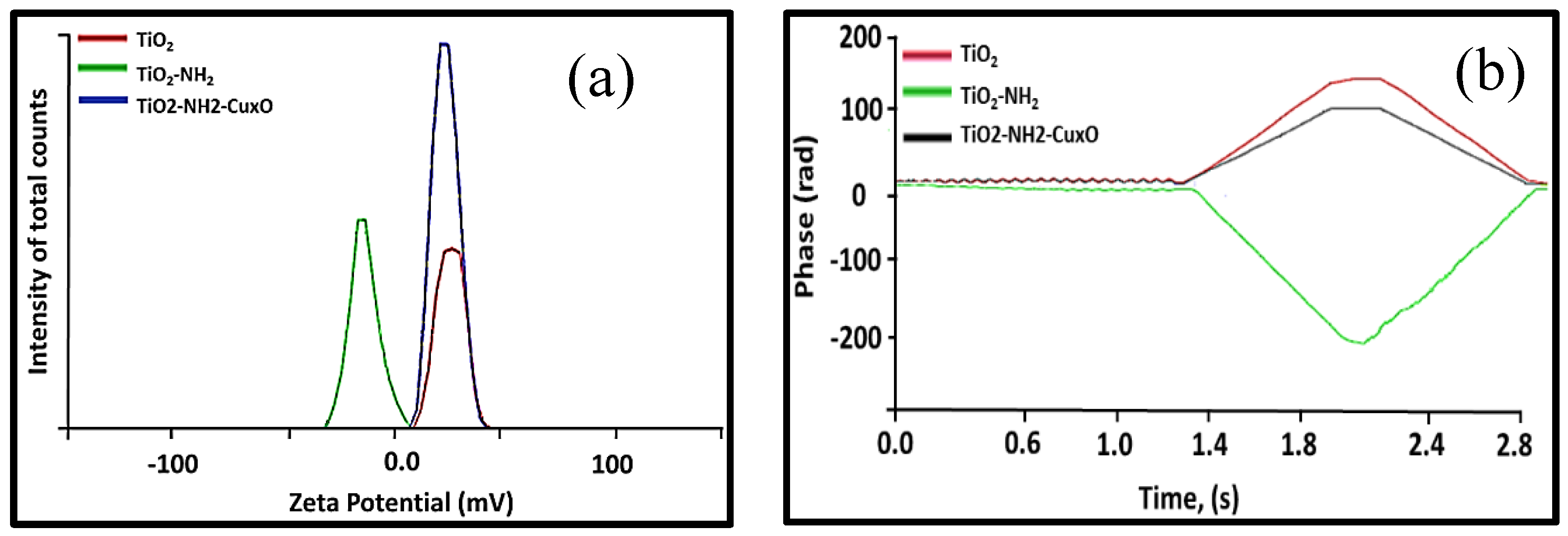
The surface charge of the TiO
2, TiO
2-NH
2, and TiO
2-NH
2-Cu
xO nanophotocatalysts were investigated by performing the zeta potential measurements and the results were presented in
Figure 4a. It is very clear that upon the attachment of the Cu
xO to the amine group of the TiO
2, the potential was almost equally shifted to the negative side due to the positive charge of copper, which was also confirmed by measuring the phase over time, as shown in
Figure 4b. The higher surface charge was measured for TiO
2 and TiO
2-NH
2 which indicates the stability due to the charge distribution on the particle surface, such that the negatively charged particle surfaces repulse each other in the solution. However, when the Cu
xO was attached (TiO
2-NH
2-Cu
xO) to the amine group, there was a possibility of a hydroxyl group formation on the surface upon dispersion in water, which led to a negative shift in the Zeta potential [
32,
33]. In all the cases, the nanoparticles were highly suspended in water which is impartment to have direct contact between the photocatalyst and pollutants for greater efficiency.
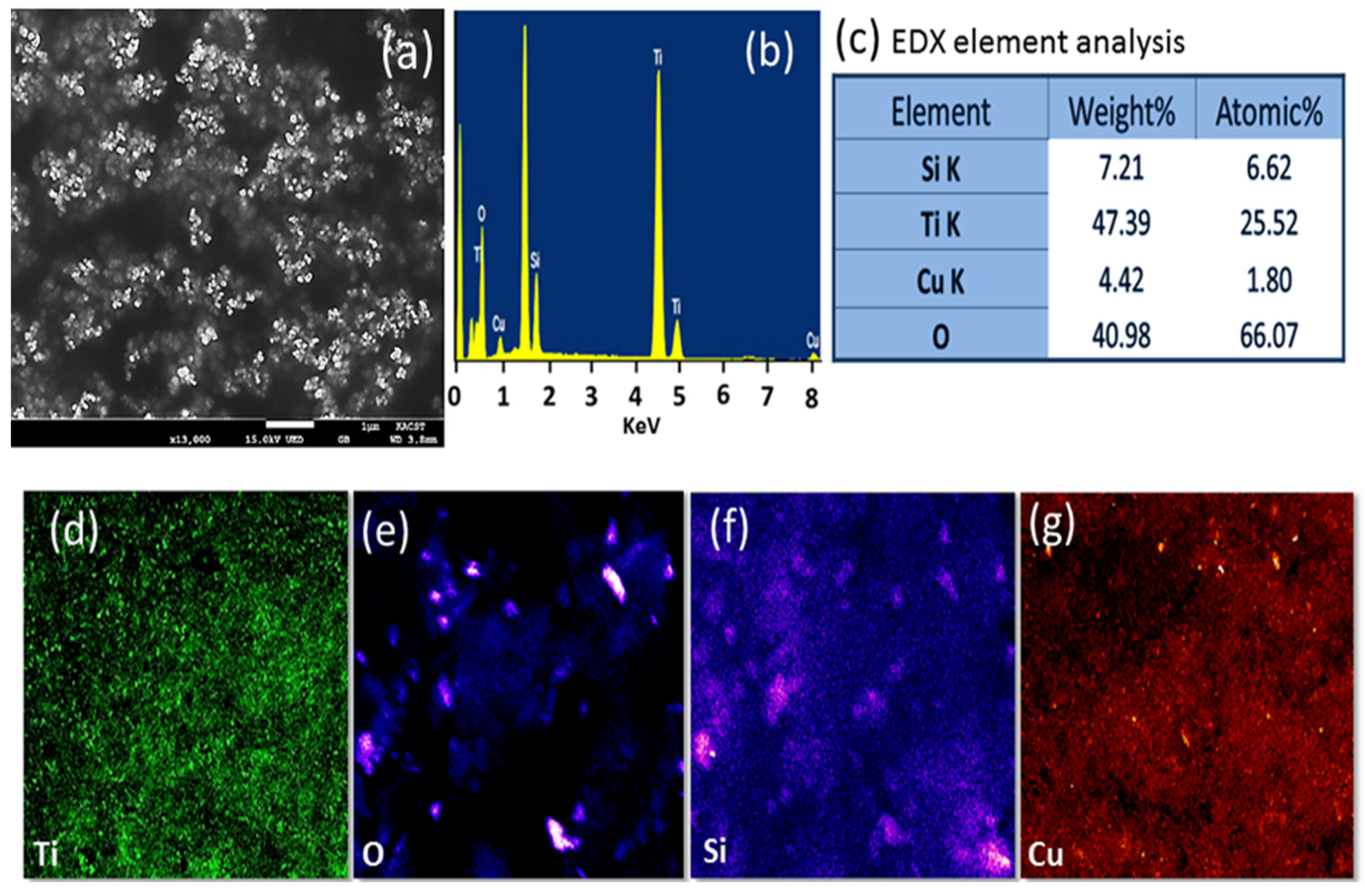
The surface morphological features and elemental analysis of TiO
2-NH
2-Cu
xO nanophotocatalyst were explored using high resolution scanning electron microscope- Energy dispersive X-ray spectroscopy (HR-SEM/EDX) and the results are shown in
Figure 5. The HR-SEM image showed a spherical particle of TiO
2 in the nanometer range as shown in
Figure 5a. The EDX analysis in
Figure 5b revealed the distinctive peaks for Ti (Lα 0.45 and, Kα 4.5 KeV), O (Kα 0.53 KeV), Cu (Lα 0.9 and Kα 8.1 KeV), C (Kα, 0.28 KeV), and Si (Kα, 1.8 KeV). The measured weight and atomic percentage of copper are 4.42 and 1.80, respectively. These values were expected as the copper was in the form of copper oxide. To understand the distribution of the elements located on the surface of TiO
2-NH
2-Cu
xO nanophotocatalyst, EDX-elemental mapping was performed, and the results are shown in
Figure 5c–f. The distribution of copper was proportional to the rest of the elements and the contrast of the color demonstrates that the distribution of copper was uniform on the titanium surface.
Further investigation on the morphological structure was performed on the TiO
2, and TiO
2- NH
2-Cu
xO nanophotocatalyst using HR-TEM, and selected area electron diffraction (SAED) as shown in
Figure 6. The HR-TEM images of TiO
2 (a) and its SAED pattern
Figure 6b in comparison with TiO
2-NH
2-Cu
xO nanophotocatalyst SAED pattern confirmed the formation of additional crystalline structures beside the TiO
2 crystalline structure, as compared between
Figure 6b and d. The additional diffraction pattern confirms the (1 1 1) and (2 2 0) phases which could be assigned to copper oxide. The intensity of the diffraction pattern Debye rings in
Figure 6d was clearly due to the loading of Cu
xO on the surface, which is consistent with the EDX results.
The thermal behavior of the TiO
2, TiO
2-NH
2, and TiO
2-NH
2-Cu
xO nanophotocatalysts was explored and the thermogravimetric analysis curves are shown in
Figure 7. The curves revealed that TiO
2 nanoparticles show three main areas of weight loss and the total weight loss was 12.4%. The first area of weight loss from 25 °C to 110 °C was due to the evaporation of solvent and water molecules physically adsorbed on the TiO
2 nanoparticles. The second weight loss from 110 °C to 298 °C is owed to the removal of the chemically bonded water molecules [
34,
35]. The weight loss from 298 °C to 850 °C could be attributed to the condensation of hydroxyl groups on the surface of TiO
2 nanoparticles. On the other hand, the Thermogravimetric analysis (TGA) curve of TiO
2-NH
2 shows a total weight loss of 19.4%, which is significantly higher compared to the as-synthesized TiO
2 mainly due to the amine loading at the surface of the TiO
2 nanoparticles upon modification. This is supported by the evidence of the higher weight loss from 462 °C to 850 °C (6.3%), which was attributed to the combustion of organic materials. The TiO
2-NH
2-Cu
xO nanophotocatalyst TGA curve shows many weight loss steps, and the total weight loss was 13.61%, less than the TiO
2-NH
2 nanoparticles. The less weight loss could be attributed to the presence of Cu
xO which improves the heat stability [
36].
The chemical analysis of TiO
2 and TiO
2-NH
2-Cu
xO nanophotocatalyst using X-ray photoelectron spectroscopy (XPS) which was performed to identify the chemical bonding relevant to the surface modification and the oxidation state of the copper attached to the TiO
2-NH
2-Cu
xO nanophotocatalyst. The recorded XPS spectra are fitted using peak fit software to deconvolute the peaks and the results are shown in
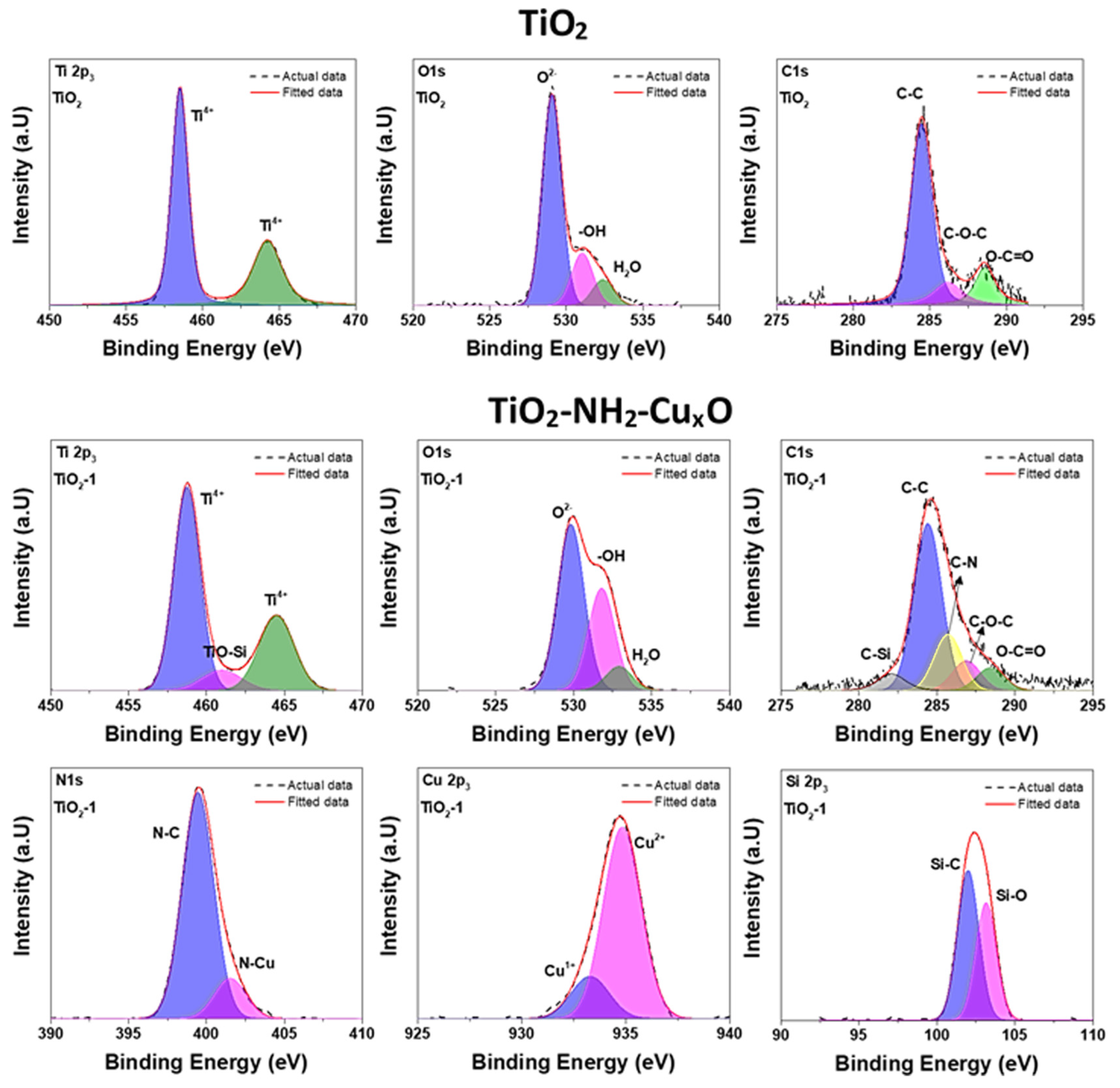
Figure 8.
The deconvolute Ti2p and O1s envelop of TiO
2 indicate that the synthesized material does not have any contamination before the surface functionalization and the presence of C1s in pure TiO
2 is due to the surface adsorbed atmospheric carbon. If the C1s peaks are compared for pure TiO
2 and TiO
2-NH
2-Cu
xO, we can observe the clear changes in the peak position of deconvoluted peaks. The peaks position of C–C, C–O–C, and O–C=O for pure TiO
2 are 284.46, 286.20, and 288.34 eV, respectively. In the case of TiO
2-NH
2-Cu
xO, the peaks of C–Si, C–C, C–N, C–O–C, and O–C=O are observed at 282.09, 284.42, 285.71, 286.89, and 288.68 eV, respectively The peaks at the higher binding energies (C–O–C and O–C=O) increased by 0.48 eV and the peak broadening of the same indicates the improvement of electronegativity due to the surface modification of (3-aminopropyl) trimethoxysilane. This result suggests that the electronegativity of TiO
2-NH
2-Cu
xO is stronger than that of pure TiO
2. This may be attributed to the electrostatic interaction between TiO
2 and (3-aminopropyl) trimethoxysilane, suggesting that surface modification is not a simple physical mixture. Cu2p3 envelop in TiO
2-NH
2-Cu
xO shows the peaks at 234.9 eV and 233.2 eV indicate the presence of Cu
2+ and Cu
1+, respectively [
36,
37].
The relative concentration of each cation state was calculated using the Equation
where Cu x is the Cu atomic concentration of the cation state x, IA of Cu {x} is the integrated area of the corresponding state x, and IA of Cu is the total integrated area of Cu peaks. Upon quantifying the percentage of two Cu states from the XPS spectra, it was found that the percentage of Cu
2+ and Cu
1+ was 81.91% and 18.09%, respectively. The capability of copper to donate and accept electrons explains its important character in oxidation–reduction reactions [
26]. Therefore, the presence of two valences of oxidation states (Cu
1+ and Cu
2+) could enhance photocatalytic activity due to the possibility of mid-level state formation [
38]. N1s spectrum of TiO
2-NH
2-Cu
xO shows two peaks centered at 399.2 eV and 400.1 eV. The lower binding energy at 399.2 eV belongs to N–C because the amine groups were chemically attached to the surface of the nanoparticles. The metal cations that combine with amine groups most likely appear at the higher binding energy [
39], and according to the structural representation, Cu cations were connected to the amine group. Therefore, it can be concluded that the higher binding energy peak was attributed to N–Cu bonding.
The recorded O1s peak of TiO
2 and TiO
2-NH
2-Cu
xO is broad with multiple overlapping components as shown in
Figure 8. The first peak centered at 529.5 eV marked for the samples belong to O
2 (metal oxide) and the second peak, marked around 531.7 eV, may be attributed to the OH
− species that were mostly present on the surface. The higher binding energy peak centered at around 532.6 eV belongs to surface adsorbed moisture; however, Si–O boding also appears in the same peak position. Hence, it is hard to differentiate the chemically attached Si-O bonding in surface modified TiO
2 nanoparticles. Besides, the Si 2p3 peak in TiO
2-NH
2-Cu
xO shows the Si–O bonding which confirms the presence of Si.
The XPS spectra of Ti 2p in TiO
2-NH
2-Cu
xO displayed two peaks centered at 458.7 eV (2p
3/2), and 464.5 eV (2p
1/2) which are the predominant state of Ti
4+. An additional small peak centered at 460 eV belongs to Ti–O–Si, according to the literature [
40], which also confirms the chemical attachment of Si with TiO
2 nanoparticles.
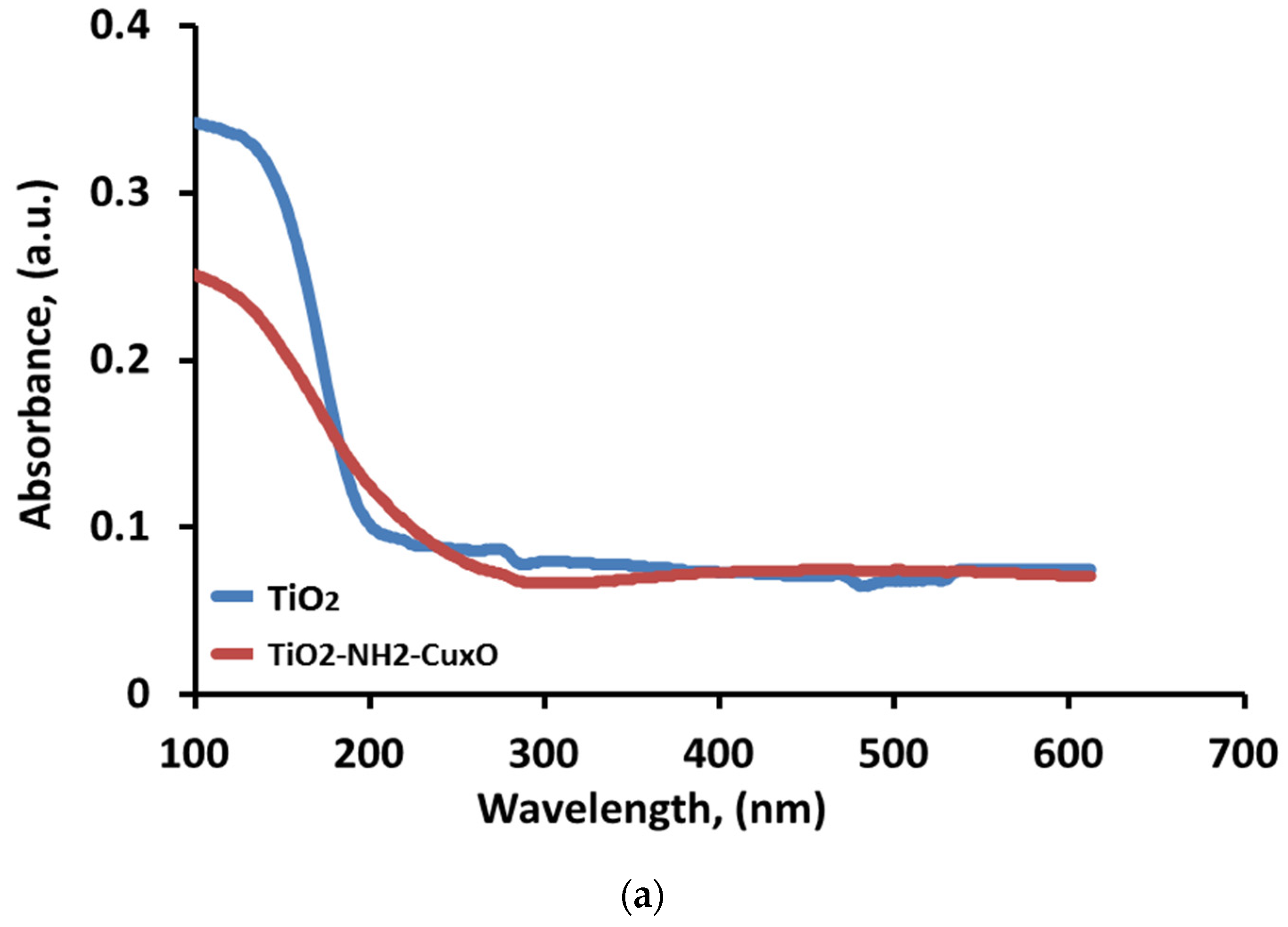
UV–vis diffuse reflectance spectra were measured for TiO
2 and TiO
2-NH
2-Cu
xO nanophotocatalyst to study the optical absorption properties of the samples, as shown in
Figure 9a. It is apparent that when Cu chemically attached to the TiO
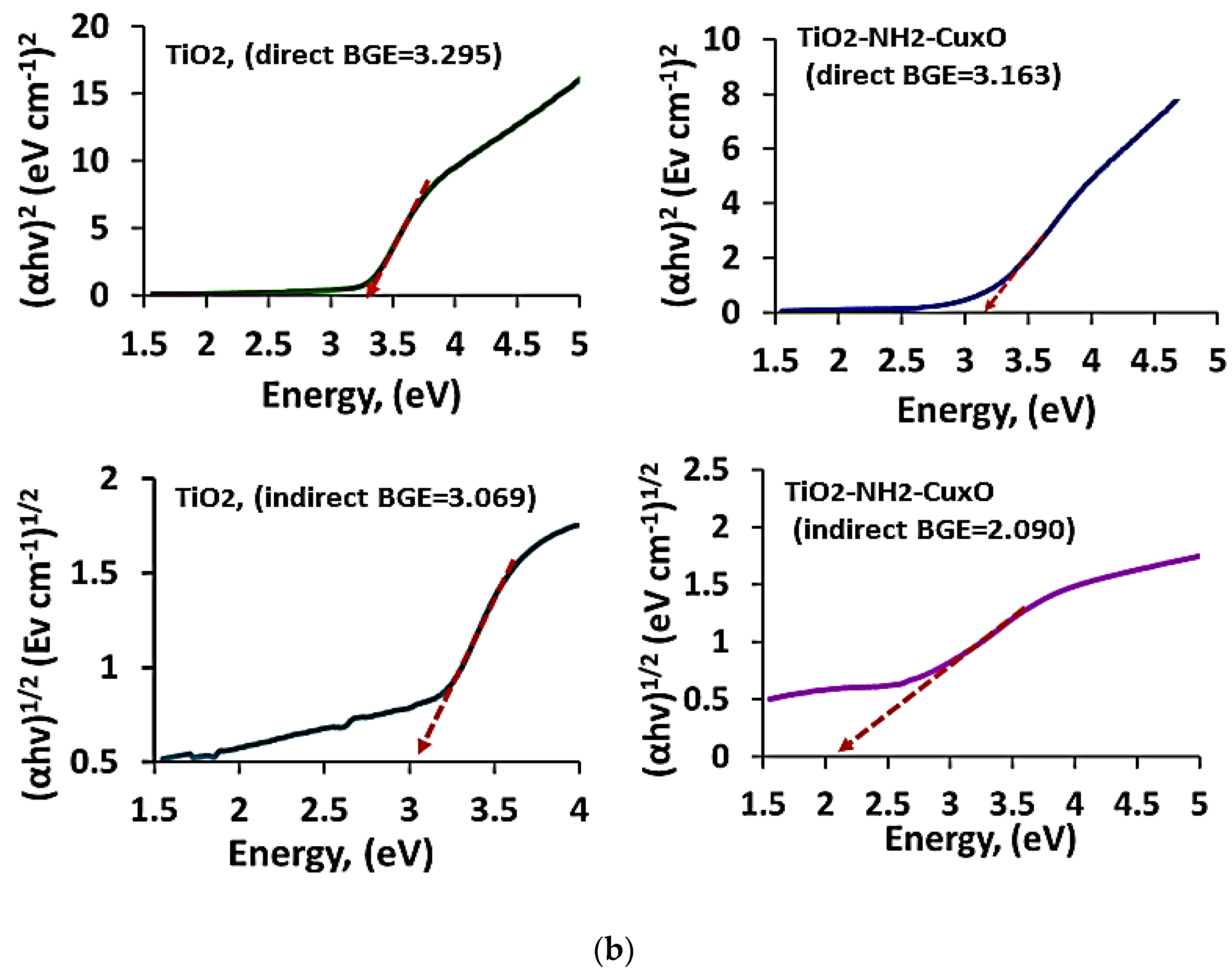
2 via amine complexation the phenomena of redshift are observed in the wavelength which led to a decrease in frequency. Accordingly, this led to a decrease in the value of bandgap energy (Eg). To obtain adequate information about the bandgap energy due to the addition of copper; the direct and indirect band gaps were calculated to infer from the relationship shown in the Equations (3) and (4), respectively.
where α is the absorption coefficient, (
hν) is the photon energy and A is absorbance. Tauc plots were performed by drawing the (
αhν)n vs. Photon energy (eV) and the direct and indirect bandgaps for both TiO
2 and TiO
2-NH
2-Cu
xO nanophotocatalyst were calculated as it is presented in
Figure 9b. Accordingly, the direct bandgap energy for TiO
2 nanoparticles was 3.24 eV, whereas the indirect bandgap was 3.10 eV, which is consistent with other obtained results [
41]. Moreover, when the TiO
2 chemically decorated with Cu the direct bandgap was shifted to 2.98 eV, as well as the indirect bandgap to 2.10 eV. The bandgap for TiO
2 is around 3.2 eV for the anatase phase but the observed variation in this study may be due to a difference in pore size and high surface area [
42]. Also, it can be seen that the modification of TiO
2 has an impact on the absorption range as it shifts to the visible wavelength after copper is attached to the TiO
2 surface. The shift in the bandgap towards lower energy is due to the chemically attached Cu on the TiO
2 surface which reduces the electron recombination, thus, the difference in bandgap energy between TiO
2 and TiO
2-NH
2-Cu
xO nanophotocatalyst is (0.58 eV). This result suggests that the modification of TiO
2 anatase phase (nanophotocatalyst) allows TiO
2 to absorb visible light and indicates that the presence of copper on the surface of the catalyst shifted the bandgap to the visible light (1.6–3.2 eV) which greatly enhanced the degradation of the MB dye molecule in visible light rather than UV light.
2.2. Photocatalytic Assessment
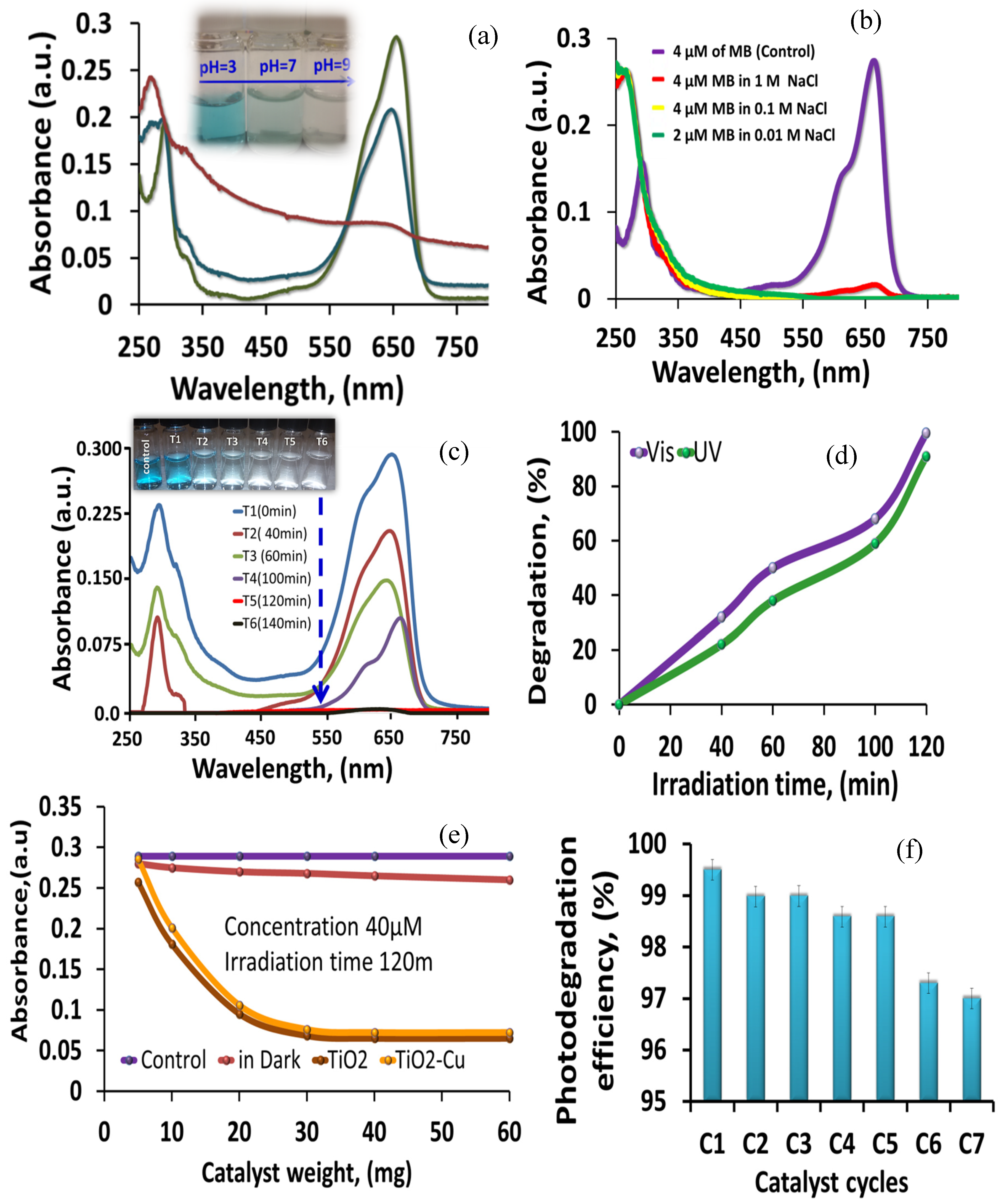
The photocatalytic degradation of the MB dye by the TiO
2-NH
2-Cu
xO nanophotocatalyst at different pH was investigated. As shown in
Figure 10a, the photocatalytic degradation of the MB dye was greatly enhanced by increasing the solution pH value and the faster degradation rate was observed above pH 7. The maximum degradation was obtained at pH 9, which is mainly due to the generation of hydroxyl radicals (·OH) at a pH higher than 7. At pH lower than 7, there was a decrease in degradation effectiveness due to the coulombic repulsion. Indeed, the surface charge of the TiO
2-NH
2-Cu
xO nanophotocatalyst changes, based on the pH variation, and so changes the catalyst reaction potential [
43]. By this potential variation, the interaction between the MB dye and surface of the TiO
2-NH
2-Cu
xO nanophotocatalysts vary and results in a difference of reaction velocity. Under acidic conditions, the TiO
2-NH
2-Cu
xO nanophotocatalyst surface could be pseudo-photon charged whereas, in alkaline conditions, the surface could be negatively charged. In alkaline media, the TiO
2-NH
2-Cu
xO nanophotocatalyst’s surface is negatively charged and the cationic MB can be simply adsorbed, consequently, the hydroxyl radicals generated from the surface of the TiO
2-NH
2-Cu
xO nanophotocatalyst help to degrade MB dye in the solution. On the other hand, in acid media, the adsorption of the cationic MB dye on the surface of the TiO
2-NH
2-Cu
xO nanophotocatalyst is reduced due to the electrostatic repulsion, thus, reduces the photocatalytic degradation rate. This is consistent with another study and indicates that the acid–base property of the synthesized TiO
2-NH
2-Cu
xO nanophotocatalyst surface has a significant role in the photocatalytic degradation activity by changing the pH. Therefore, detailed knowledge of the material’s surface properties are highly important to understand and predict their photocatalytic efficiency.
The effect of ionic strength is considered an important factor especially in highly saline water [
44]. Therefore, the ionic strength at different ionic concentrations was also investigated by changing the NaCl concentration (0.01, 0.1, and 1M) with/without a TiO
2-NH
2-Cu
xO nanophotocatalyst, as shown in
Figure 10b. The results reveal that the ionic strength does not have a significant impact on the catalyst activity at the mentioned concentration. From the degradation rate, it can also observe that when a high concentration of NaCl (1M) was applied, the efficiency was only decreased by about 5%. Therefore, this catalyst can work effectively for the photocatalytic applications at high ionic strength and ionic concentration.
To assess the appropriate time and photocatalytic activity for the TiO
2-NH
2-Cu
xO nanophotocatalyst to decompose MB dye, an experiment was carried out and the results were presented in
Figure 10c, which shows the absorption spectrum for the MB versus degradation time. The inset in
Figure 10c shows the photo of the samples taken at specific times after the TiO
2-NH
2-Cu
xO nanophotocatalyst was removed from solution using centrifugation. It is obvious that, after 2 h contact time between the TiO
2-NH
2-Cu
xO nanophotocatalyst and the MB dye, more than 95% of the MB dye was degraded. We also observed a slight shift in the adsorption spectra which is due to the demethylation of MB occurred at the catalyst surface [
45].
Separate experiments were carried out to compare the efficiency of the TiO
2-NH
2-Cu
xO nanophotocatalyst for the degradation of MB dye under ultraviolet and visible light and the results were presented in
Figure 10d, which indicates that more than over 95% degradation of 40 µM methylene blue under visible light was obtained and the same phenomenon was also observed under ultraviolet light. These results proved the successful tuning of the electronic band of TiO
2 toward visible light and showed the ability of the TiO
2-NH
2-Cu
xO nanophotocatalyst to work under both UV and visible light, which can show the maximum efficiency under sunlight, as the catalyst worked dually in UV and visible light. This is attributed to the reduction of the electron–hole recombination process by the addition of Cu to the surface of the TiO
2 nanoparticles. On the other hand, the high surface area material resulted in higher contact with the MB dye molecules, and Cu attachments to the surface of the TiO
2 nanoparticles reduce the electron–hole pair recombination. It is the combination of these two phenomena which improved the photocatalytic degradation efficiency in both UV and visible light.
It was reported that the charge recombination process in semiconducting materials takes place in femtoseconds or picoseconds, and electron trapping on the TiO
2 surface occurs in a few femtoseconds, thus, the lifetime of electron–hole pairs is a few nanoseconds [
39]. Thus, the rate of electron–hole recombination decreases as charge transfer occurs very fast on the TiO
2-NH
2-Cu
xO nanophotocatalyst surface. However, there are several other parameters such as particle size, defects, porosity, and impurities which can affect the electron–hole recombination rate. Therefore, the selection of suitable particle size with high surface area, proper chemical attachment agent and metallic ions are important for the reduction of electron–hole pair recombination and the improvement in photocatalytic reaction efficiency [
46].
To assess the efficiency of TiO
2-NH
2-Cu
xO nanophotocatalyst for the maximum degradation of the MB dye, various amounts of catalyst (0–60 mg) in 40 µM solution of MB dye were irradiated under visible light for 120 min. A similar experiment was carried out with TiO
2 NPs for the comparison and the maximum capacity of TiO
2-NH
2-CuxO nanophotocatalyst was calculated. Each experiment was repeated three times to get the standard deviation. As shown in
Figure 10e, it can be observed that the photocatalyst can break down certain concentrations of the MB dye until it reaches equilibrium with the species in the solution. Also, it is clear that TiO
2-NH
2-Cu
xO nanophotocatalyst has the highest degradation capacity and it reaches the equilibrium using 30 mg of the catalyst with the mentioned concentration, the maximum limit of the catalytic degradation capacity was 1.5 mM g
−1. This result confirms the high catalytic efficiency and the need for only small weight to conduct the photodegradation process.
The stability and reusability of the TiO
2-NH
2-Cu
xO nanophotocatalyst were studied and the results are shown in
Figure 10f. The catalyst has maintained its strength after seven cycles of application and the percentage difference between C1 (high recovery) and C7 (low recovery) was less than 5%, which indicates that the TiO
2-NH
2-Cu
xO nanophotocatalyst can be recycled several times without losing the activity. Furthermore, there are many reports available on commercial P25 TiO
2 material showing the lower photocatalytic degradation efficiency and not suitable for multiple recycling time [
47,
48,
49]. In our case, the degradation efficiency decreased by less than 5% with seven recycling times.
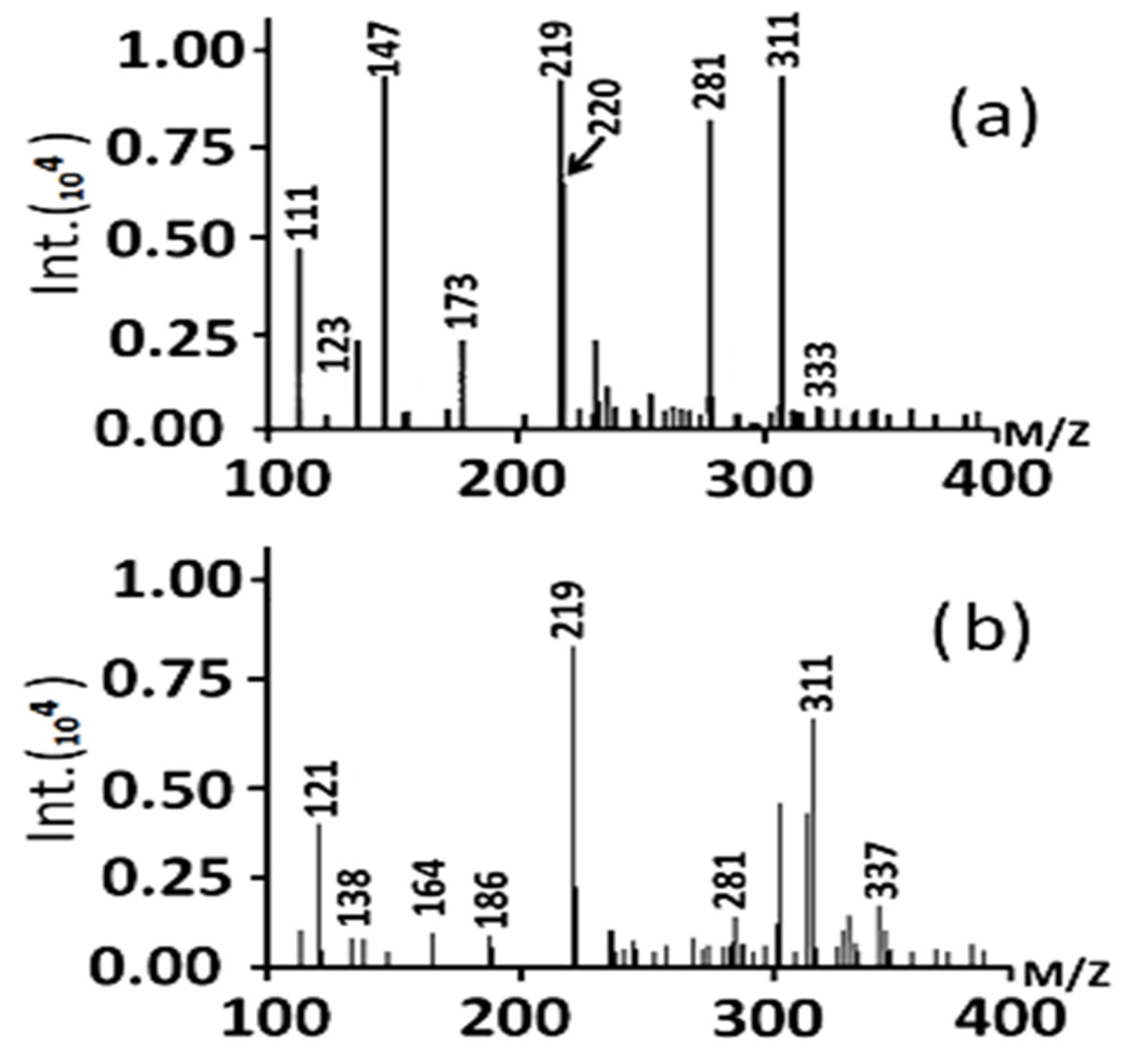
The degradation of the MB dye using the TiO
2-NH
2-Cu
xO nanophotocatalyst under visible light was investigated using Liquid chromatography-mass spectrometry (LC-MS) to elucidate the degradation pathway. The chromatograms of the degraded compounds after 40 min and 60 min of irradiation are shown in
Figure 11a,b, respectively. The TiO
2-NH
2-Cu
xO nanophotocatalyst can degrade MB dye in visible light through multiple steps as shown in
Figure 12, which illustrated that the hydroxyl free radical reacted MB dye molecule. After that, the produced molecules start to cleave through the multi-steps due to the free radical’s attack, and it ends with colorless compounds (H
2O, CO
2, NO
2, SO
42−).
Mechanism: the mechanism of the photocatalytic activity of the synthesized materials is illustrated in
Figure 13, which postulate that the TiO
2 within the TiO
2-NH
2-Cu
xO nanophotocatalyst releases the electrons from the valence band by exposure to the light and the Cu
xO works as a reservoir to collect the electrons underneath the conduction band, and therefore minimizes the bandgap energy which makes the catalyst very effective in visible light. Also, it is postulated that the high surface area anatase phase TiO
2 decorated with homogeneously distributed Cu
xO nanoparticles can cause a large surface area exposure to the MB dye and thus increase the photocatalytic degradation effectiveness. In general, the hydroxyl radical and peroxide ion can be initiated due to the existence of holes and electrons in the TiO
2 nanoparticle solution bypassing the UV light. These energetic molecules destroy the organic compound through a series of chemical reactions as the potential of ·OH is 2.8 V, which is higher than other oxidant agents such as hypochlorous acid (1.49 V) [
39]. Moreover, it is well known that TiO
2 is more suitable only for UV light range due to its bandgap, and to make the TiO
2 suitable for both the UV and vis ranges, a series of TiO
2-NH
2-Cu
xO nanophotocatalyst was developed to overcome this problem by shifting the energy band gap to the visible wavelength region.
The XPS characterization of the nanophotocatalyst showed that the Cu is present in two different oxidation states; Cu
2+ which is dominant and Cu
1+. It is believed that this changeable oxidation states in transition metals can improve the catalytic activity [
50]. It was speculated that when the electron skips from the conduction band, the chance for electron recombination would be very low due to the series which creates charge carriers, electrons, and holes; therefore, a series of chemical reactions will begin to degrade the MB dye as shown in
Figure 13.