Manipulatable Interface Electric Field and Charge Transfer in a 2D/2D Heterojunction Photocatalyst via Oxygen Intercalation
Abstract
1. Introduction
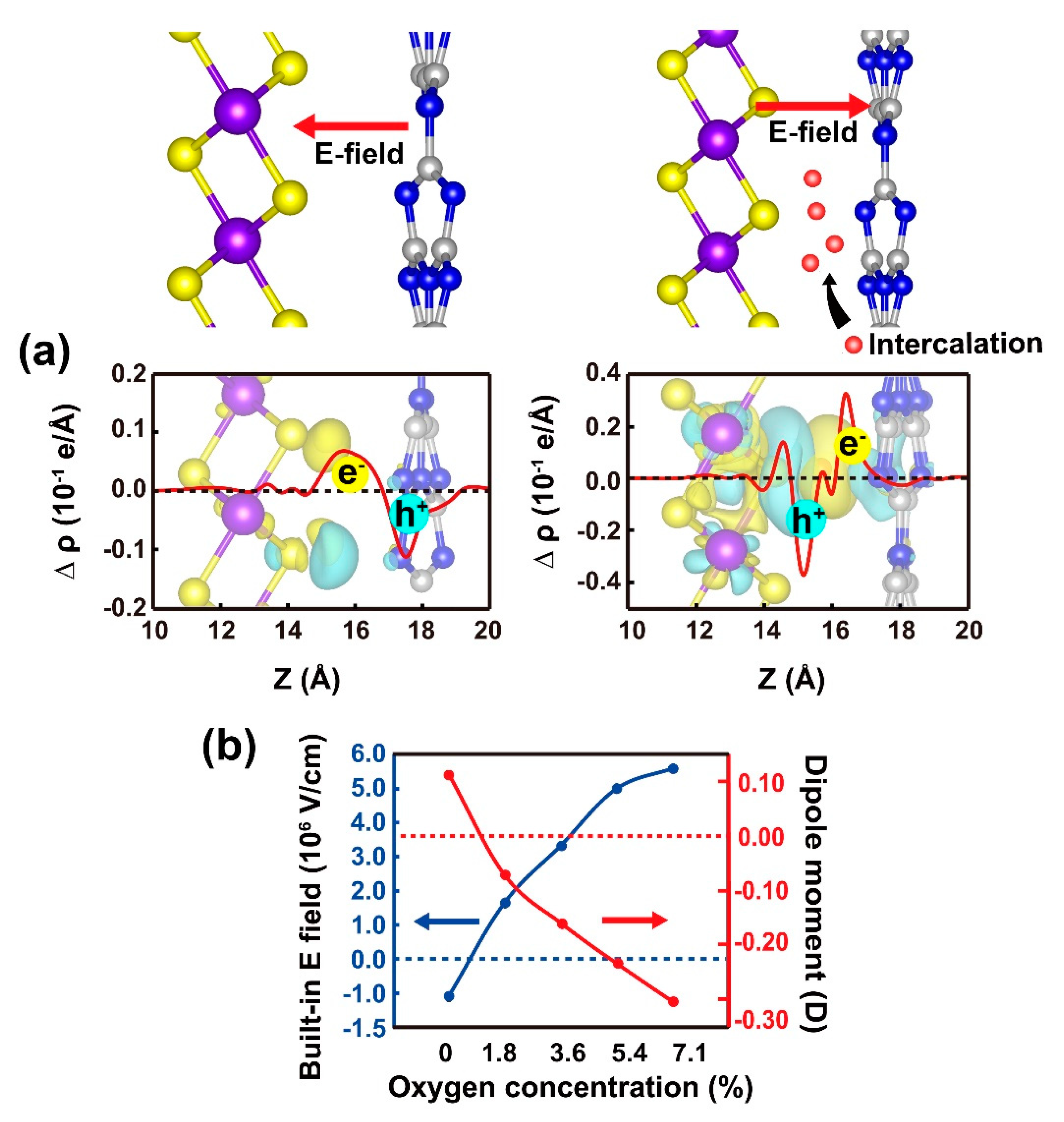
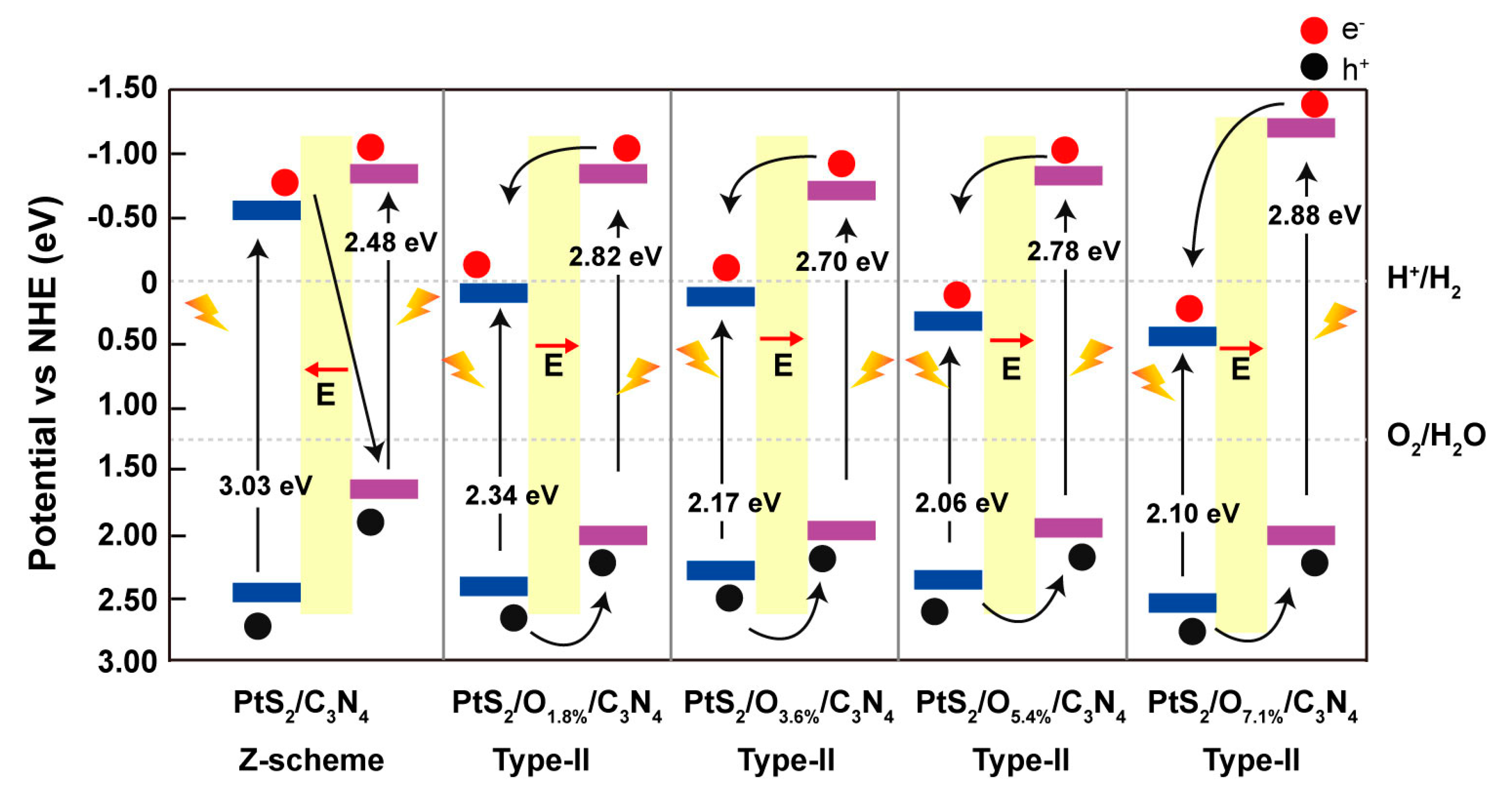
2. Results and Discussion
3. Calculation Method
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Toroker, M.C.; Kanan, D.K.; Alidoust, N.; Isseroff, L.Y.; Liao, P.; Carter, E.A. First principles scheme to evaluate band edge positions in potential transition metal oxide photocatalysts and photoelectrodes. Phys. Chem. Chem. Phys. 2011, 13, 16644–16654. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Hu, H.; Bao, C.; Guo, F.; Zhang, X.; Liu, X.; Hua, J.; Tan, J.; Wang, A.; Zhou, H. Forming heterojunction: An effective strategy to enhance the photocatalytic efficiency of a new metal-free organic photocatalyst for water splitting. Sci. Rep. 2016, 6, 29327. [Google Scholar] [CrossRef] [PubMed]
- Hisatomi, T.; Kubota, J.; Domen, K. Recent advances in semiconductors for photocatalytic and photoelectrochemical water splitting. Chem. Soc. Rev. 2014, 43, 7520–7535. [Google Scholar] [CrossRef] [PubMed]
- Esswein, A.J.; Nocera, D.G. Hydrogen Production by Molecular Photocatalysis. Chem. Rev. 2007, 107, 4022–4047. [Google Scholar] [CrossRef]
- Ngoh, S.K.; Njomo, D. An overview of hydrogen gas production from solar energy. Renew. Sustain. Energy Rev. 2012, 16, 6782–6792. [Google Scholar] [CrossRef]
- Saraf, S.; Giraldo, M.; Paudel, H.P.; Sakthivel, T.S.; Shepard, C.; Gupta, A.; Leuenberger, M.N.; Seal, S. Photoelectrochemical analysis of band gap modulated TiO2 for photocatalytic water splitting. Int. J. Hydrog. Energy 2017, 42, 9938–9944. [Google Scholar] [CrossRef]
- Ikeda, S.; Fujikawa, S.; Harada, T.; Nguyen, T.H.; Nakanishi, S.; Takayama, T.; Iwase, A.; Kudo, A. Photocathode Characteristics of a Spray-Deposited Cu2ZnGeS4 Thin Film for CO2 Reduction in a CO2-Saturated Aqueous Solution. ACS Appl. Energy Mater. 2019, 2, 6911–6918. [Google Scholar] [CrossRef]
- Zhou, R.; Guzman, M.I. CO2 Reduction under Periodic Illumination of ZnS. J. Phys. Chem. C 2014, 118, 11649–11656. [Google Scholar] [CrossRef]
- Zhu, Z.; Kao, C.T.; Tang, B.H.; Chang, W.C.; Wu, R.J. Efficient hydrogen production by photocatalytic water-splitting using Pt-doped TiO2 hollow spheres under visible light. Ceram. Int. 2016, 42, 6749–6754. [Google Scholar] [CrossRef]
- Zhang, J.; Zhou, P.; Liu, J.; Yu, J. New understanding of the difference of photocatalytic activity among anatase, rutile and brookite TiO2. Phys. Chem. Chem. Phys. 2014, 16, 20382–20386. [Google Scholar] [CrossRef]
- Yan, J.; Wu, H.; Chen, H.; Pang, L.; Zhang, Y.; Jiang, R.; Li, L.; Liu, S.F. One-pot hydrothermal fabrication of layered β-Ni(OH)2/g-C3N4 nanohybrids for enhanced photocatalytic water splitting. Appl. Catal. B 2016, 194, 74–83. [Google Scholar] [CrossRef]
- Hoque, M.D.A.; Guzman, M.I. Photocatalytic Activity: Experimental Features to Report in Heterogeneous Photocatalysis. Materials 2018, 11, 1990. [Google Scholar] [CrossRef] [PubMed]
- Opoku, F.; Govender, K.K.; Sittert, C.G.C.E.V.; Govender, P.P. Understanding the mechanism of enhanced charge separation and visible light photocatalytic activity of modified wurtzite ZnO with nanoclusters of ZnS and graphene oxide: From a hybrid density functional study. New. J. Chem. 2017, 41, 8140–8155. [Google Scholar] [CrossRef]
- Lahiri, J.; Batzill, M. Surface Functionalization of ZnO Photocatalysts with Monolayer ZnS. J. Phys. Chem. C 2008, 112, 4304–4307. [Google Scholar] [CrossRef]
- Tahir, M.; Cao, C.; Mahmood, N.; Butt, F.K.; Mahmood, A.; Idrees, F.; Hussain, S.; Tanveer, M.; Ali, Z.; Aslam, I. Multifunctional g-C3N4 Nanofibers: A Template-Free Fabrication and Enhanced Optical, Electrochemical, and Photocatalyst Properties. ACS Appl. Mater. Interfaces 2014, 6, 1258–1265. [Google Scholar] [CrossRef]
- Xu, Y.; Fu, Z.C.; Cao, S.; Chen, Y.; Fu, W.F. Highly selective oxidation of sulfides on a CdS/C3N4 catalyst with dioxygen under visible-light irradiation. Catal. Sci. Technol. 2017, 7, 587–595. [Google Scholar] [CrossRef]
- Wang, X.C.; Maeda, K.; Thomas, A.; Takanabe, K.; Xin, G.; Carlsson, J.M.; Domen, K.; Antonietti, M. A metal-free polymeric photocatalyst for hydrogen production from water under visible light. Nat. Mater. 2009, 8, 76–80. [Google Scholar] [CrossRef]
- Zhang, W.; Sun, Y.; Dong, F.; Zhang, W.; Duan, S.; Zhang, Q. Facile synthesis of organic–inorganic layered nanojunctions of g-C3N4/(BiO)2CO3 as efficient visible light photocatalyst. Dalton Trans. 2014, 43, 12026–12036. [Google Scholar] [CrossRef]
- Zhang, G.; Lan, Z.A.; Lin, L.; Lin, S.; Wang, X. Overall water splitting by Pt/g-C3N4 photocatalysts without using sacrificial agents. Chem. Sci. 2016, 7, 3062–3066. [Google Scholar] [CrossRef]
- Liu, J.; Cheng, B.; Yu, J. A new understanding of the photocatalytic mechanism of the direct Z-scheme g-C3N4/TiO2 heterostructure. Phys. Chem. Chem. Phys. 2018, 18, 31175–31183. [Google Scholar] [CrossRef]
- Liu, J.; Jia, Q.; Long, J.; Wang, X.; Gao, Z.; Gu, Q. Amorphous NiO as co-catalyst for enhanced visible-light-driven hydrogen generation over g-C3N4 photocatalyst. Appl. Catal. B 2018, 222, 35–43. [Google Scholar] [CrossRef]
- Su, T.; Shao, Q.; Qin, Z.; Guo, Z.; Wu, Z. Role of Interfaces in Two-Dimensional Photocatalyst for Water Splitting. ACS Catal. 2018, 8, 2253–2276. [Google Scholar] [CrossRef]
- Li, H.; Yu, H.; Quan, X.; Chen, S.; Zhang, Y. Uncovering the Key Role of the Fermi Level of the Electron Mediator in a Z-Scheme Photocatalyst by Detecting the Charge Transfer Process of WO3-metal-gC3N4 (Metal = Cu, Ag, Au). ACS Appl. Mater. Interfaces 2016, 8, 2111–2119. [Google Scholar] [CrossRef] [PubMed]
- Cai, X.; Zhang, J.; Fujitsuka, M.; Majima, T. Graphitic-C3N4 hybridized N-doped La2Ti2O7 two-dimensional layered composites as efficient visible-light-driven photocatalyst. Appl. Catal. B 2017, 202, 191–198. [Google Scholar] [CrossRef]
- Scanlon, D.O.; Dunnill, C.W.; Buckeridge, J.; Shevlin, S.A.; Logsdail, A.J.; Woodley, S.M.; Catlow, C.R.A.; Powell, M.J.; Palgrave, R.G.; Parkin, I.; et al. Band alignment of rutile and anatase TiO₂. Nat. Mater. 2013, 12, 798–801. [Google Scholar] [CrossRef]
- Khan, S.; Cho, H.; Kim, D.; Han, S.S.; Lee, K.H.; Cho, S.H.; Song, T.; Choi, H. Defect engineering toward strong photocatalysis of Nb-doped anatase TiO2: Computational predictions and experimental verifications. Appl. Catal. B 2017, 206, 520–530. [Google Scholar] [CrossRef]
- Zhang, J.; Zhang, M.; Sun, R.Q.; Wang, X. A facile band alignment of polymeric carbon nitride semiconductors to construct isotype heterojunctions. Angew. Chem. 2012, 51, 10145–10149. [Google Scholar] [CrossRef]
- Wang, X.; Xu, Q.; Li, M.; Shen, S.; Wang, X.; Wang, Y.; Feng, Z.; Shi, J.; Han, H.; Li, C. Photocatalytic overall water splitting promoted by an α-β phase junction on Ga2O3. Angew. Chem. 2012, 51, 13089–13092. [Google Scholar] [CrossRef]
- Hu, S.; Ma, L.; Li, F.; Fan, Z.; Wang, Q.; Bai, J.; Kang, X.; Wu, G. Construction of g-C3N4/S-g-C3N4 metal-free isotype heterojunctions with an enhanced charge driving force and their photocatalytic performance under anoxic conditions. RSC Adv. 2015, 5, 90750–90756. [Google Scholar] [CrossRef]
- Li, J.; Zhang, Z.; Cui, W.; Wang, H.; Cen, W.; Johnson, G.; Jiang, G.; Zhang, S.; Dong, F. The Spatially Oriented Charge Flow and Photocatalysis Mechanism on Internal van der Waals Heterostructures Enhanced g-C3N4. ACS Catal. 2018, 8, 8376–8385. [Google Scholar] [CrossRef]
- Chen, L.; Shi, G.; Shen, J.; Peng, B.; Zhang, B.; Wang, Y.; Bian, F.; Wang, J.; Li, D.; Qian, Z.; et al. Ion sieving in graphene oxide membranes via cationic control of interlayer spacing. Nature 2017, 550, 380–383. [Google Scholar] [CrossRef] [PubMed]
- Ditto, J.; Merrill, D.R.; Mitchson, G.; Gabriel, J.J.; Mathew, K.; Hennig, R.G.; Medlin, D.L.; Browning, N.D.; Johnson, D.C. Interface-Driven Structural Distortions and Composition Segregation in Two-Dimensional Heterostructure. Angew. Chem. 2017, 56, 14448–14452. [Google Scholar] [CrossRef] [PubMed]
- Xiong, T.; Cen, W.; Zhang, Y.; Dong, F. Bridging the g-C3N4 Interlayers for Enhanced Photocatalysis. ACS Catal. 2016, 6, 2462–2472. [Google Scholar] [CrossRef]
- Qu, X.; Hu, S.; Bai, J.; Lu, G.; Kang, X. A facile approach to synthesize oxygen doped g-C3N4 with enhanced visible light activity under anoxic conditions via oxygen-plasma treatment. New J. Chem. 2018, 42, 4998–5004. [Google Scholar] [CrossRef]
- Li, J.H.; Shen, B.; Hong, Z.; Lin, B.; Gao, B.; Chen, Y. A facile approach to synthesize novel oxygen-doped g-C3N4 with superior visible-light photoreactivity. Chem. Commum. 2012, 48, 12017–12019. [Google Scholar] [CrossRef] [PubMed]
- Han, B.; Hu, Y.H. MoS2 as a co-catalyst for photocatalytic hydrogen production from water. Energy Sci. Eng. 2016, 4, 285–304. [Google Scholar] [CrossRef]
- Wei, L.; Chen, Y.; Lin, Y.; Wu, H.; Yuan, R.; Li, Z. MoS2 as non-noble-metal co-catalyst for photocatalytic hydrogen evolution over hexagonal ZnIn2S4 under visible light irradiations. Appl. Catal. B 2014, 144, 521–527. [Google Scholar] [CrossRef]
- Zhang, W.; Xiao, X.; Zeng, X.; Li, Y.; Zheng, L.; Wan, C. Enhanced photocatalytic activity of TiO2 nanoparticles using SnS2/RGO hybrid as co-catalyst: DFT study and photocatalytic mechanism. J. Alloys Comd. 2016, 685, 774–783. [Google Scholar] [CrossRef]
- Akple, M.S.; Low, J.; Wageh, S.; Al-Ghamdi, A.A.; Yu, J.; Zhang, J. Enhanced visible light photocatalytic H2-production of g-C3N4/WS2 composite heterostructures. Appl. Surf. Sci. 2015, 358, 196–203. [Google Scholar] [CrossRef]
- Yan, J.; Chen, Z.; Ji, H.; Liu, Z.; Wang, X.; Xu, Y.; She, X.; Huang, L.; Xu, L.; Xu, H.; et al. Construction of a 2D Graphene-Like MoS2/C3N4 Heterojunction with Enhanced Visible-Light Photocatalytic Activity and Photoelectrochemical Activity. Chemistry 2016, 22, 4764–4773. [Google Scholar] [CrossRef]
- Wang, J.; Guan, Z.; Huang, J.; Li, Q.; Yang, J. Enhanced photocatalytic mechanism for the hybrid g-C3N4/MoS2 nanocomposite. J. Mater. Chem. A 2014, 2, 7960–7966. [Google Scholar] [CrossRef]
- Liu, J.; Hua, E. High Photocatalytic Activity of Heptazine-Based g-C3N4/SnS2 Heterojunction and Its Origin: Insights from Hybrid DFT. J. Phys. Chem. C 2017, 121, 25827–25835. [Google Scholar] [CrossRef]
- Mir, S.H.; Chakraborty, S.; Wärnå, J.; Narayan, S.; Jha, P.C.; Jha, P.K.; Ahuja, R. A comparative study of hydrogen evolution reaction on pseudo-monolayer WS2 and PtS2: Insights based on the density functional theory. Catal. Sci. Technol. 2017, 7, 687–692. [Google Scholar] [CrossRef]
- Chia, X.; Adriano, A.; Lazar, P.; Sofer, Z.; Luxa, J.; Pumera, M. Layered Platinum Dichalcogenides (PtS2, PtSe2, and PtTe2) Electrocatalysis: Monotonic Dependence on the Chalcogen Size. Adv. Funct. Mater. 2016, 26, 4306–4318. [Google Scholar] [CrossRef]
- Li, L.; Wang, W.; Chai, Y.; Li, H.; Tian, M.; Zhai, T. Few-Layered PtS2 Phototransistor on h-BN with High Gain. Adv. Funct. Mater. 2017, 27, 1701011. [Google Scholar] [CrossRef]
- Zhang, K.; Hu, S.; Zhang, Y.; Zhang, T.; Zhou, X.; Sun, Y.; Li, T.X.; Fan, H.J.; Shen, G.; Chen, X.; et al. Self-Induced Uniaxial Strain in MoS2 Monolayers with Local van der Waals-Stacked Interlayer Interactions. ACS Nano 2015, 9, 2704–2710. [Google Scholar] [CrossRef]
- Jiang, J.W.; Park, H. Mechanical properties of MoS2/graphene heterostructure. Appl. Phys. Lett. 2014, 105, 033108. [Google Scholar] [CrossRef]
- Huang, Z.F.; Song, J.; Pan, L.; Wang, Z.; Zhang, X.; Zou, J.J.; Mi, W.; Zhang, X.; Wang, L. Carbon nitride with simultaneous porous network and O-doping for efficient solar-energy-driven hydrogen evolution. Nano Energy 2015, 12, 646–656. [Google Scholar] [CrossRef]
- Björkman, T.; Gulans, A.; Krasheninnikov, A.V.; Nieminen, R.M. van der Waals Bonding in Layered Compounds from Advanced Density-Functional First-Principles Calculations. Phys. Rev. Lett. 2012, 108, 235502. [Google Scholar] [CrossRef]
- Liao, J.; Sa, B.; Zhou, J.; Ahuja, R.; Sun, Z. Design of High-Efficiency Visible-Light Photocatalysts for Water Splitting: MoS2/AlN(GaN) Heterostructures. J. Phys. Chem. C 2014, 118, 17594–17599. [Google Scholar] [CrossRef]
- Huang, H.; Song, Z.; Wei, N.; Shi, L.; Mao, Y.; Ying, Y.; Sun, L.; Xu, Z.; Peng, X. Ultrafast viscous water flow through nanostrand-channelled graphene oxide membranes. Nat. Commun. 2013, 4, 2979. [Google Scholar] [CrossRef] [PubMed]
- Baxter, J.B.; Richter, C.; Schmuttenmaer, C. Ultrafast Carrier Dynamics in Nanostructures for Solar Fuels. Annu. Rev. Phys. Chem. 2014, 65, 423–447. [Google Scholar] [CrossRef] [PubMed]
- Takanabe, K. Solar Water Splitting Using Semiconductor Photocatalyst Powders. In Solar Energy for Fuels; Springer: Cham, Switzerland, 2015; pp. 73–103. [Google Scholar]
- Li, M.; Dai, Y.; Ma, X.; Li, Z.; Huang, B. The synergistic effect between effective mass and built-in electric field for the transfer of carriers in nonlinear optical materials. Phys. Chem. Chem. Phys. 2015, 17, 17710–17717. [Google Scholar] [CrossRef] [PubMed]
- Fang, Q.; Zhao, X.; Huang, Y.; Xu, K.; Min, T.; Chu, P.K.; Ma, F. Interfacial electronic states and self-formed p–n junctions in hydrogenated MoS2/SiC heterostructure. J. Mater. Chem. C 2018, 6, 4523–4530. [Google Scholar] [CrossRef]
- Zhang, X.; Shen, J.X.; Wang, W.; Van de Walle, C.G. First-Principles Analysis of Radiative Recombination in Lead-Halide Perovskites. ACS Eng. Lett. 2018, 3, 2329–2334. [Google Scholar] [CrossRef]
- Gao, G.; Jiao, Y.; Ma, F.; Jiao, Y.; Waclawik, E.; Du, A. Carbon nanodot decorated graphitic carbon nitride: New insights into the enhanced photocatalytic water splitting from ab initio studies. Phys. Chem. Chem. Phys. 2015, 17, 31140–31144. [Google Scholar] [CrossRef]
- Mo, Z.; She, X.; Li, Y.; Liu, L.; Huang, L.; Chen, Z.; Zhang, Q.; Xu, H.; Li, H. Synthesis of g-C3N4 at different temperatures for superior visible/UV photocatalytic performance and photoelectrochemical sensing of MB solution. RSC. Adv. 2015, 5, 101552–101562. [Google Scholar] [CrossRef]
- Kresse, G.; Furthmüller, J. Efficiency of ab-initio total energy calculations for metals and semiconductors using a plane-wave basis set. Comput. Mater. Sci. 1996, 6, 15–50. [Google Scholar] [CrossRef]
- Kresse, G.; Furthmüller, J. Efficient iterative schemes for ab initio total-energy calculations using a plane-wave basis set. Phys. Rev. B 1996, 54, 11169. [Google Scholar] [CrossRef]
- Kresse, G.; Joubert, D. From ultrasoft pseudopotentials to the projector augmented-wave method. Phys. Rev. B 1999, 59, 1758. [Google Scholar] [CrossRef]
- Perdew, J.P.; Chevary, J.A.; Vosko, S.H.; Jackson, K.A.; Pederson, M.R.; Singh, D.J.; Fiolhais, C. Atoms, molecules, solids, and surfaces: Applications of the generalized gradient approximation for exchange and correlation. Phys. Rev. B 1992, 46, 6671. [Google Scholar] [CrossRef] [PubMed]
- Perdew, J.P.; Burke, K.; Ernzerhof, M. Generalized Gradient Approximation Made Simple. Phys. Rev. Lett. 1996, 77, 3865. [Google Scholar] [CrossRef]
- Monkhorst, H.J.; Pack, J.D. Special points for Brillouin-zone integrations. Phys. Rev. B 1976, 13, 5188. [Google Scholar] [CrossRef]
- Methfessel, M.; Paxton, A. High-precision sampling for Brillouin-zone integration in metals. Phys. Rev. B 1989, 40, 3616. [Google Scholar] [CrossRef] [PubMed]
- Grimme, S.; Ehrlich, S.; Goerigk, L. Effect of the damping function in dispersion corrected density functional theory. J. Comput. Chem. 2011, 32, 1456–1465. [Google Scholar] [CrossRef]
- Heyd, J.; Scuseria, G.E.; Ernzerhof, M. Hybrid functionals based on a screened Coulomb potential. J. Chem. Phys. 2003, 118, 8207–8215. [Google Scholar] [CrossRef]


© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Je, M.; Sim, E.S.; Woo, J.; Choi, H.; Chung, Y.-C. Manipulatable Interface Electric Field and Charge Transfer in a 2D/2D Heterojunction Photocatalyst via Oxygen Intercalation. Catalysts 2020, 10, 469. https://doi.org/10.3390/catal10050469
Je M, Sim ES, Woo J, Choi H, Chung Y-C. Manipulatable Interface Electric Field and Charge Transfer in a 2D/2D Heterojunction Photocatalyst via Oxygen Intercalation. Catalysts. 2020; 10(5):469. https://doi.org/10.3390/catal10050469
Chicago/Turabian StyleJe, Minyeong, Eun Seob Sim, Jungwook Woo, Heechae Choi, and Yong-Chae Chung. 2020. "Manipulatable Interface Electric Field and Charge Transfer in a 2D/2D Heterojunction Photocatalyst via Oxygen Intercalation" Catalysts 10, no. 5: 469. https://doi.org/10.3390/catal10050469
APA StyleJe, M., Sim, E. S., Woo, J., Choi, H., & Chung, Y.-C. (2020). Manipulatable Interface Electric Field and Charge Transfer in a 2D/2D Heterojunction Photocatalyst via Oxygen Intercalation. Catalysts, 10(5), 469. https://doi.org/10.3390/catal10050469





