Oxidation of Thiol Using Ionic Liquid-Supported Organotelluride as a Recyclable Catalyst
Abstract
1. Introduction
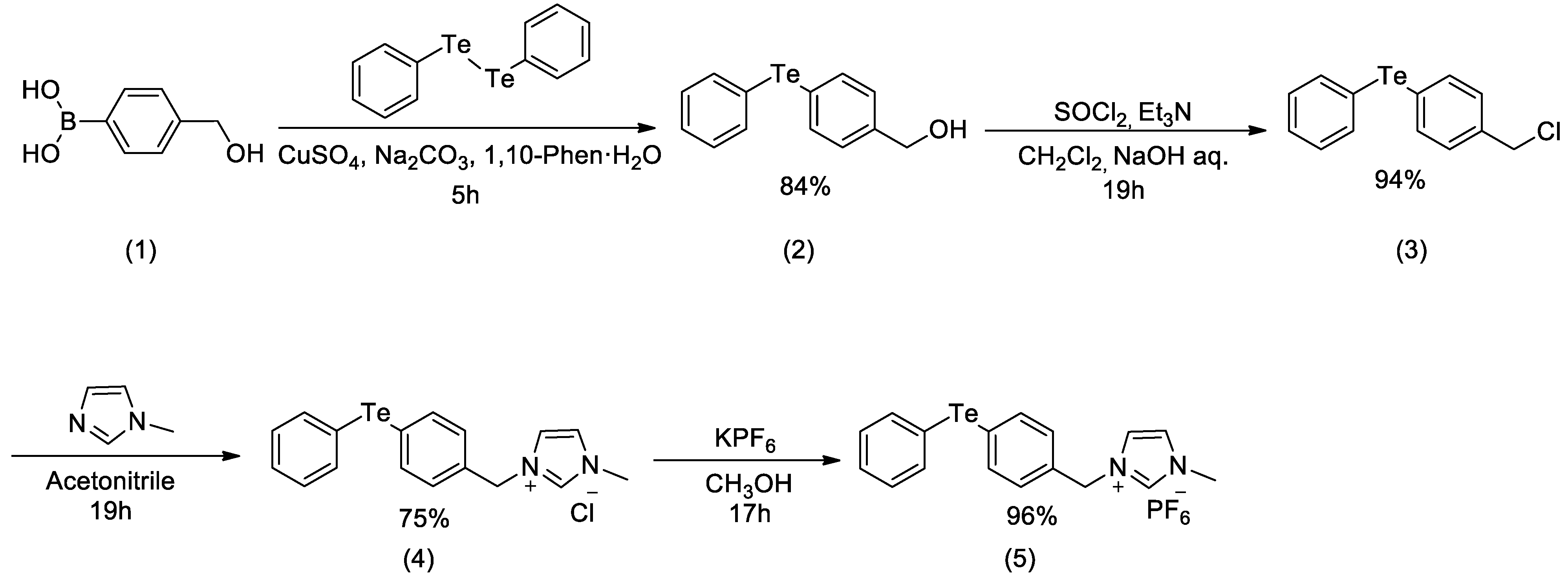
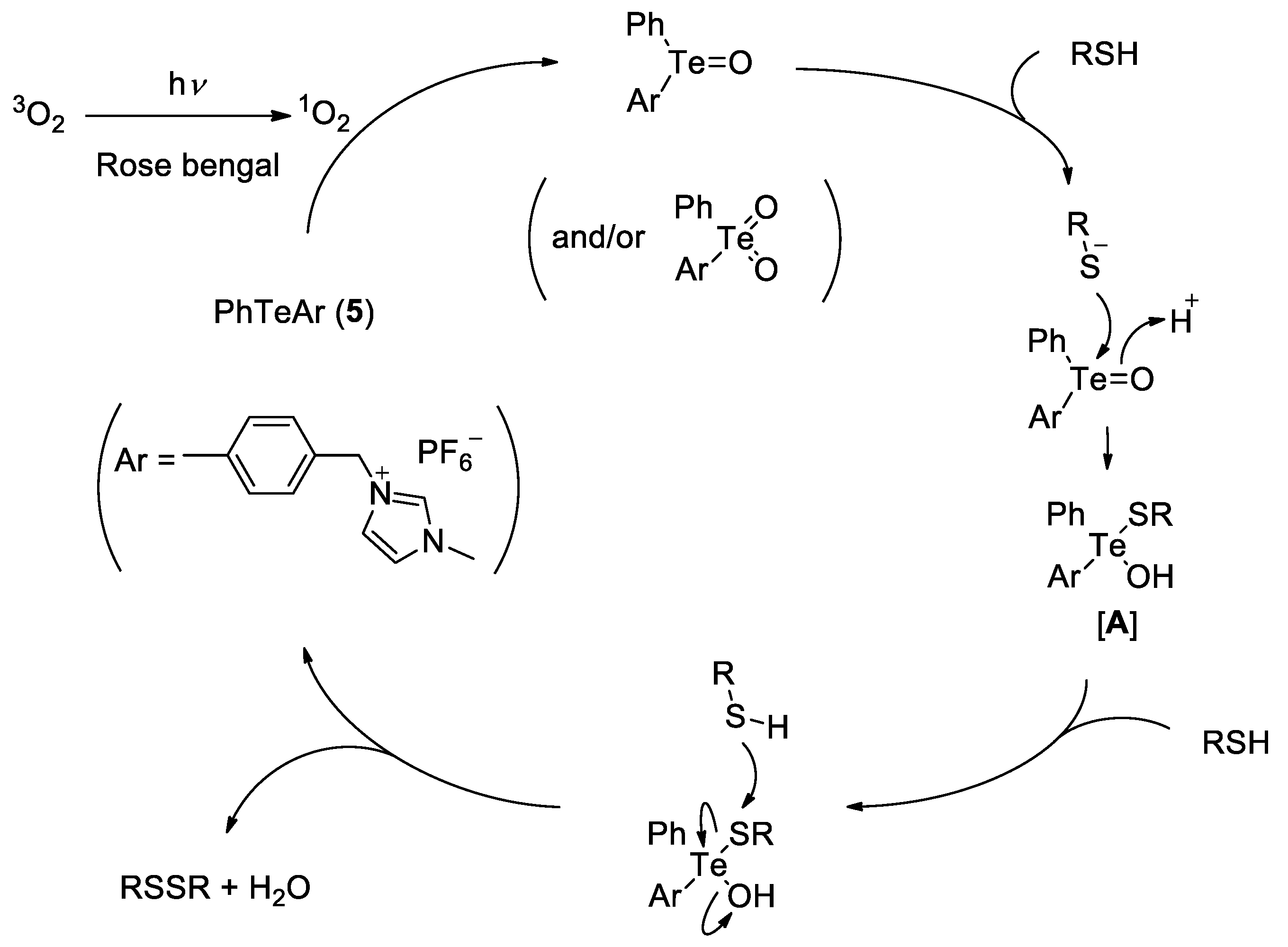
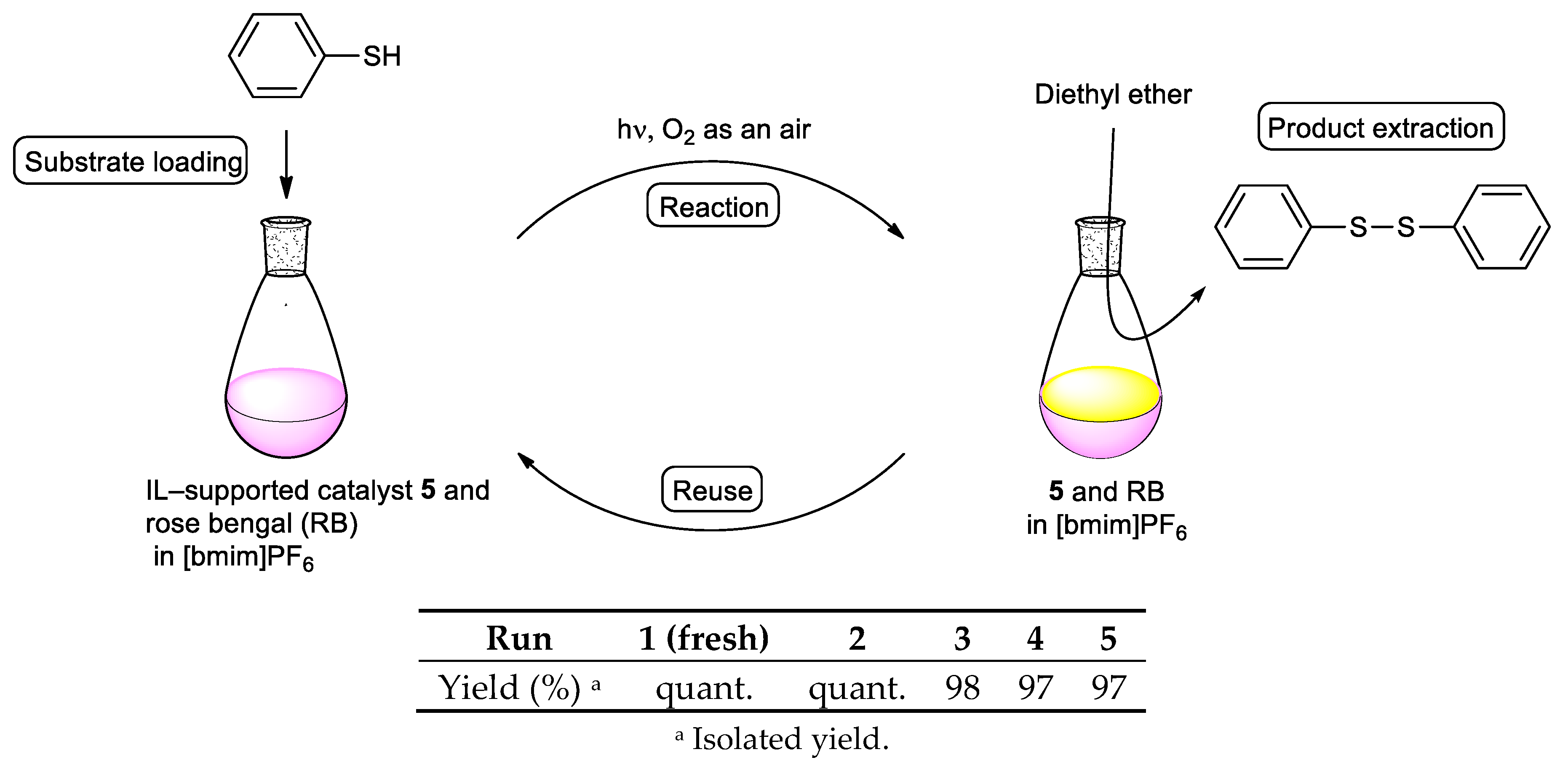
2. Results and Discussion
3. Materials and Methods
3.1. General
3.2. (4-(Hydroxymethyl)Phenyl)(Phenyl)Telluride (2)
3.3. (4-(Chloromethyl)Phenyl)(Phenyl)Telluride (3)
3.4. 1-Methyl-3-(4-(Phenyltellanyl)Benzyl)-1H-Imidazol-3-Ium Chloride (4)
3.5. 1-Methyl-3-(4-(Phenyltellanyl)Benzyl)-1H-Imidazol-3-Ium Hexafluorophosphate (5)
3.6. Oxidation of Thiols
3.6.1. Diphenyl Disulfide
3.6.2. Didodecyl Disulfide
3.6.3. Dicyclohexyl Disulfide
3.6.4. Di-Tert-Butyl Disulfide
3.6.5. 2-Hydroxyethyl Disulfide
3.6.6. Dimethyl 3,3′-Dithiodipropionate
3.6.7. Dibenzyl Disulfide
3.6.8. 1,2-Bis(4-Nitrophenyl)Disulfane
3.6.9. 1,2-Bis(4-Chlorophenyl)Disulfane
3.6.10. 4,4′-Dipyridyl Disulfide
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Petragnani, N.; Stefani, H.A. Tellurium in Organic Synthesis, 2nd ed.; Academic Press: London, UK, 2007; pp. 162–183. [Google Scholar]
- Barton, D.H.R.; Ley, S.V.; Meerholz, C.A. Bis(p-methoxyphenyl) telluroxide: A new, mild oxidising agent. Chem. Commun. 1979, 755–756. [Google Scholar] [CrossRef]
- Ley, S.V.; Meerholz, C.A.; Barton, D.H.R. Catalytic oxidation of thiocarbonyl compounds involving the use of 1,2-dibromotetrachloroethane as a brominating reagent for diaryl TeII species. Tetrahedron Lett. 1980, 21, 1785–1788. [Google Scholar] [CrossRef]
- Ley, S.V.; Meerholz, C.A.; Barton, D.H.R. Diaryl telluroxides as new mild oxidising reagents. Tetrahedron 1981, 37, 213–223. [Google Scholar] [CrossRef]
- Barton, D.H.R.; Finet, J.-P.; Thomas, M. Organotellurinic acid anhydrides as selective oxidants in organic synthesis. Tetrahedron 1986, 42, 2319–2324. [Google Scholar] [CrossRef]
- Hu, N.X.; Aso, Y.; Otsubo, T.; Ogura, F. Polymer-supported diaryl selenoxide and telluroxide as mild and selective oxidizing agents. Bull. Chem. Soc. Jpn. 1986, 59, 879–884. [Google Scholar] [CrossRef]
- Hu, N.X.; Aso, Y.; Otsubo, T.; Ogura, F. Novel oxidizing properties of p-methoxybenzenetellurinic acid anhydride. Tetrahedron Lett. 1986, 27, 6099–6102. [Google Scholar] [CrossRef]
- Fukumoto, T.; Matsuki, T.; Hu, N.X.; Aso, Y.; Otsubo, T.; Ogura, F. Benzenetellurinic mixed anhydrides as mild oxidizing agents. Chem. Lett. 1990, 19, 2269–2272. [Google Scholar] [CrossRef]
- Kambe, N.; Tsukamoto, T.; Miyoshi, N.; Murai, S.; Sonoda, N. Oxidation of olefins with benzenetellurinic anhydride. Chem. Lett. 1987, 16, 269–272. [Google Scholar] [CrossRef]
- Oba, M.; Endo, M.; Nishiyama, K.; Ouchi, A.; Ando, W. Photosensitized oxygenation of diaryl tellurides to telluroxides and their oxidizing properties. Chem. Commun. 2004, 14, 1672–1673. [Google Scholar] [CrossRef]
- Oba, M.; Okada, Y.; Nishiyama, K.; Shimada, S.; Ando, W. Synthesis, characterization and oxidizing properties of a diorgano tellurone carrying bulky aromatic substituents. Chem. Commun. 2008, 5378–5380. [Google Scholar] [CrossRef]
- Oba, M.; Okada, Y.; Nishiyama, K.; Ando, W. Aerobic photooxidation of phosphite esters using diorganotelluride catalysts. Org. Lett. 2009, 11, 1879–1881. [Google Scholar] [CrossRef] [PubMed]
- Okada, Y.; Oba, M.; Arai, A.; Tanaka, K.; Nishiyama, K.; Ando, W. Diorganotelluride-catalyzed oxidation of silanes to silanols under atmospheric oxygen. Inorg. Chem. 2010, 49, 383–385. [Google Scholar] [CrossRef] [PubMed]
- Oba, M.; Tanaka, K.; Nishiyama, K.; Ando, W. Aerobic oxidation of thiols to disulfides catalyzed by diaryl tellurides under photosensitized conditions. J. Org. Chem. 2011, 76, 4173–4177. [Google Scholar] [CrossRef] [PubMed]
- Miao, W.; Chan, T.H. Ionic–liquid–supported synthesis: A novel liquid–phase strategy for organic synthesis. Acc. Chem. Res. 2006, 39, 897–908. [Google Scholar] [CrossRef]
- Koguchi, S. An ionic–liquid–supported 18-crown-6 ether: Recyclable catalyst for acetylation and fluorination in anionic liquid. Trans. Mater. Res. Soc. Jpn. 2013, 38, 35–36. [Google Scholar] [CrossRef][Green Version]
- Koguchi, S.; Nakamura, K. Ascorbic acid based ionic liquid: Recyclable and efficient catalytic systems for the Huisgen cycloaddition. Synlett 2013, 24, 2305–2309. [Google Scholar] [CrossRef]
- Koguchi, S.; Izawa, K. Ionic liquid-phase synthesis of 1,5-disubstituted 1,2,3-triazoles. ACS Comb. Sci. 2014, 16, 381–385. [Google Scholar] [CrossRef]
- Koguchi, S.; Mihoya, A.; Mimura, M. Alcohol oxidation via recyclable hydrophobic ionic liquid-supported IBX. Tetrahedron 2016, 72, 7633–7637. [Google Scholar] [CrossRef]
- Koguchi, S.; Shibuya, Y.; Igarashi, Y.; Takemura, H. Ionic liquid–supported 1,3-dimethylimidazolidin-2-one: Application as a reusable halogenation reagent. Synlett 2019, 30, 943–946. [Google Scholar] [CrossRef]
- Dethe, D.H.; Srivastava, A.; Dherange, B.D.; Kumar, B.V. Unsymmetrical disulfide synthesis through photoredox catalysis. Adv. Synth. Catal. 2018, 360, 3020–3025. [Google Scholar] [CrossRef]
- Kumar, V.; Kaushik, M.P. Efficient oxidative coupling of thiols into disulfides using N-tert-butyl-N-chlorocyanamide. Bull. Chem. Soc. Jpn. 2008, 81, 160–162. [Google Scholar] [CrossRef]
- Kosobokov, M.; Cui, B.; Balia, A.; Matsuzaki, K.; Tokunaga, E.; Saito, N.; Shibata, N. Importance of a fluorine substituent for the preparation of meta- and para-pentafluoro-λ6-sulfanyl-substituted pyridines. Angew. Chem. Int. Ed. 2016, 55, 10781–10785. [Google Scholar] [CrossRef] [PubMed]

| Entry | Tellurium Catalyst | Solvent | Yield (%) b |
|---|---|---|---|
| 1 | 5 | [bmim](CF3SO2)2N | 78 |
| 2 | 5 | [bmim]MeSO4 | 90 |
| 3 | 5 | [bmim]PF6 | quant. |
| 4 | 5 | [bmim]BF4 | quant. |
| 5 | 5 | [bmim]Br | 79 |
| 6 | 4 | [bmim]PF6 | quant. |
| 7 | PhTePh | [bmim]PF6 | 84 |
| 8 | none | [bmim]PF6 | 47 |
| 9 c | 5 | [bmim]PF6 | 23 |

| Entry | Substrate | Product | Yield (5) b |
|---|---|---|---|
| 1 | 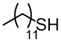 | 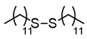 | quant. |
| 2 |  | 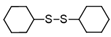 | 94 |
| 3 |  |  | 73 |
| 4 | 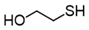 | 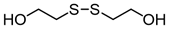 | 98 |
| 5 | 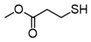 | 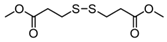 | quant. |
| 6 | 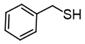 |  | 91 |
| 7 | 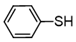 | 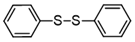 | quant. |
| 8 | 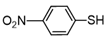 | 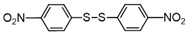 | quant. |
| 9 | 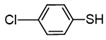 | 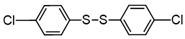 | 71 |
| 10 | 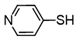 |  | quant. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mihoya, A.; Koguchi, S.; Shibuya, Y.; Mimura, M.; Oba, M. Oxidation of Thiol Using Ionic Liquid-Supported Organotelluride as a Recyclable Catalyst. Catalysts 2020, 10, 398. https://doi.org/10.3390/catal10040398
Mihoya A, Koguchi S, Shibuya Y, Mimura M, Oba M. Oxidation of Thiol Using Ionic Liquid-Supported Organotelluride as a Recyclable Catalyst. Catalysts. 2020; 10(4):398. https://doi.org/10.3390/catal10040398
Chicago/Turabian StyleMihoya, Aya, Shinichi Koguchi, Yuga Shibuya, Minato Mimura, and Makoto Oba. 2020. "Oxidation of Thiol Using Ionic Liquid-Supported Organotelluride as a Recyclable Catalyst" Catalysts 10, no. 4: 398. https://doi.org/10.3390/catal10040398
APA StyleMihoya, A., Koguchi, S., Shibuya, Y., Mimura, M., & Oba, M. (2020). Oxidation of Thiol Using Ionic Liquid-Supported Organotelluride as a Recyclable Catalyst. Catalysts, 10(4), 398. https://doi.org/10.3390/catal10040398




