Synthesis and Surface Modification of TiO2-Based Photocatalysts for the Conversion of CO2
Abstract
1. Introduction
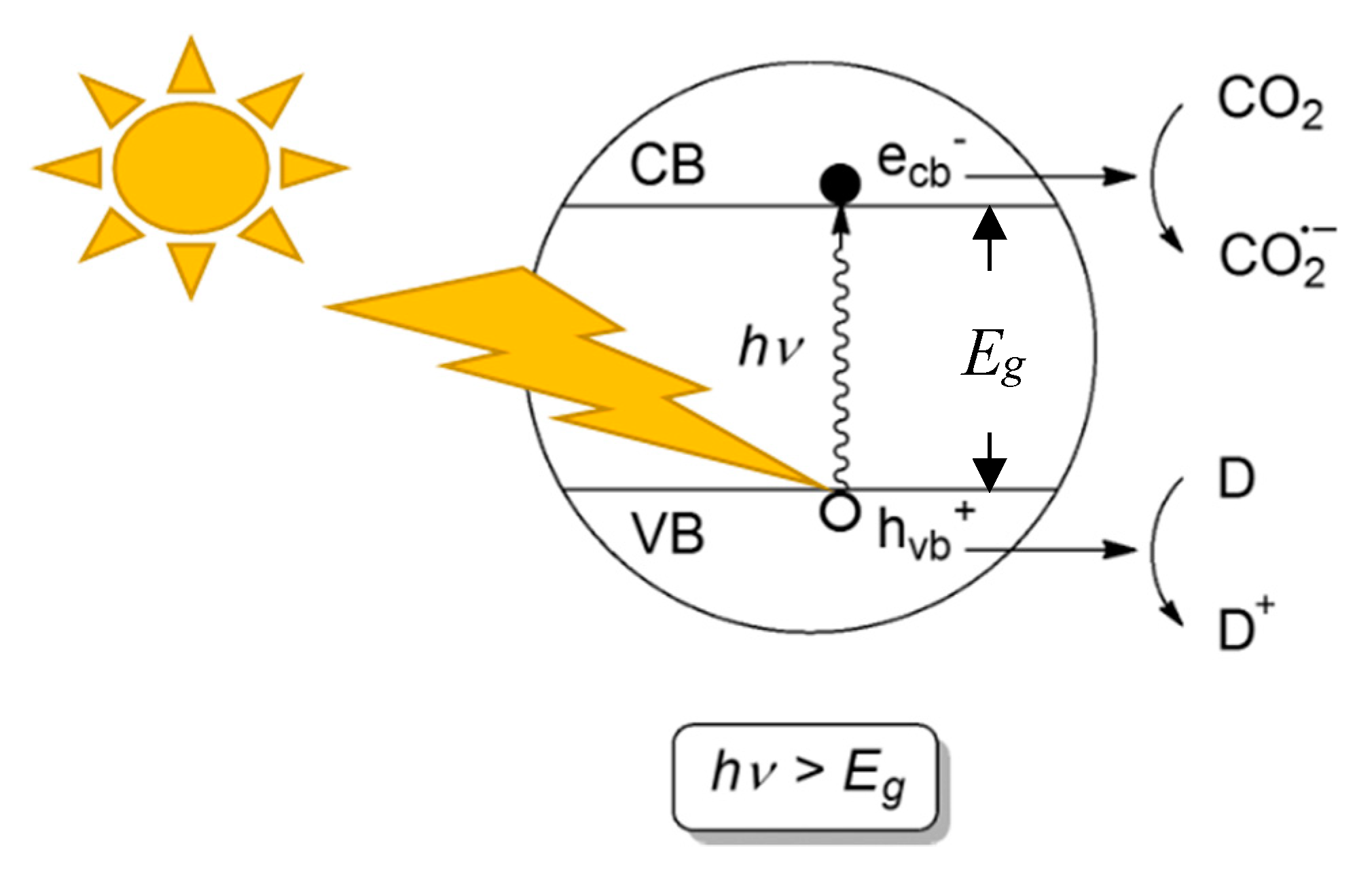
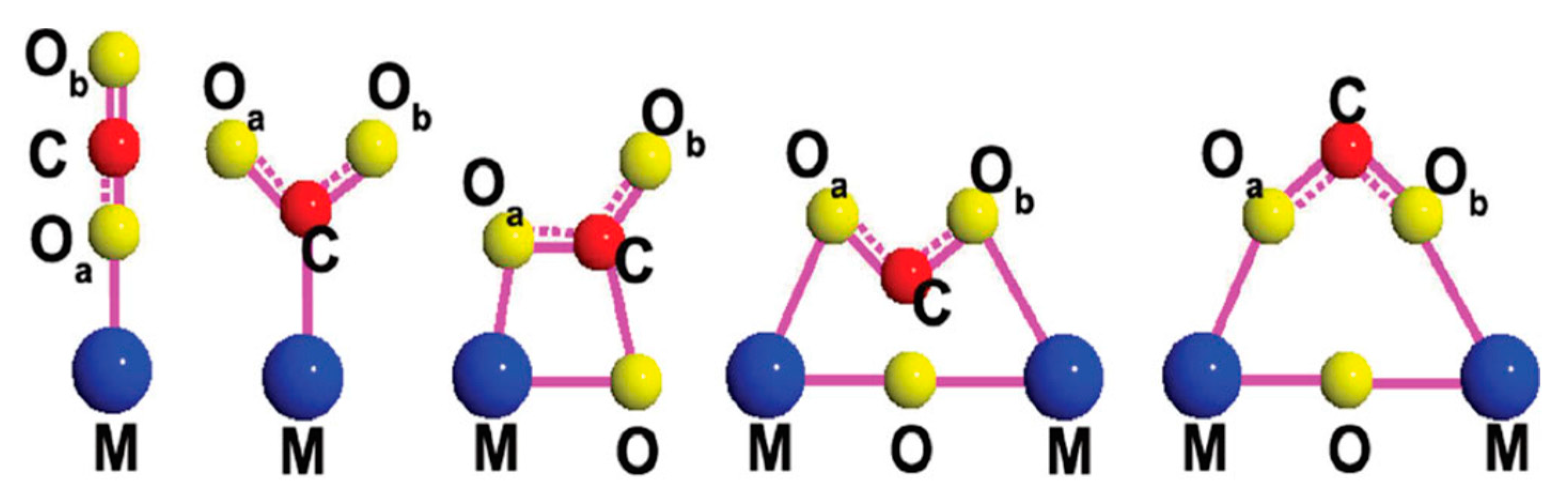
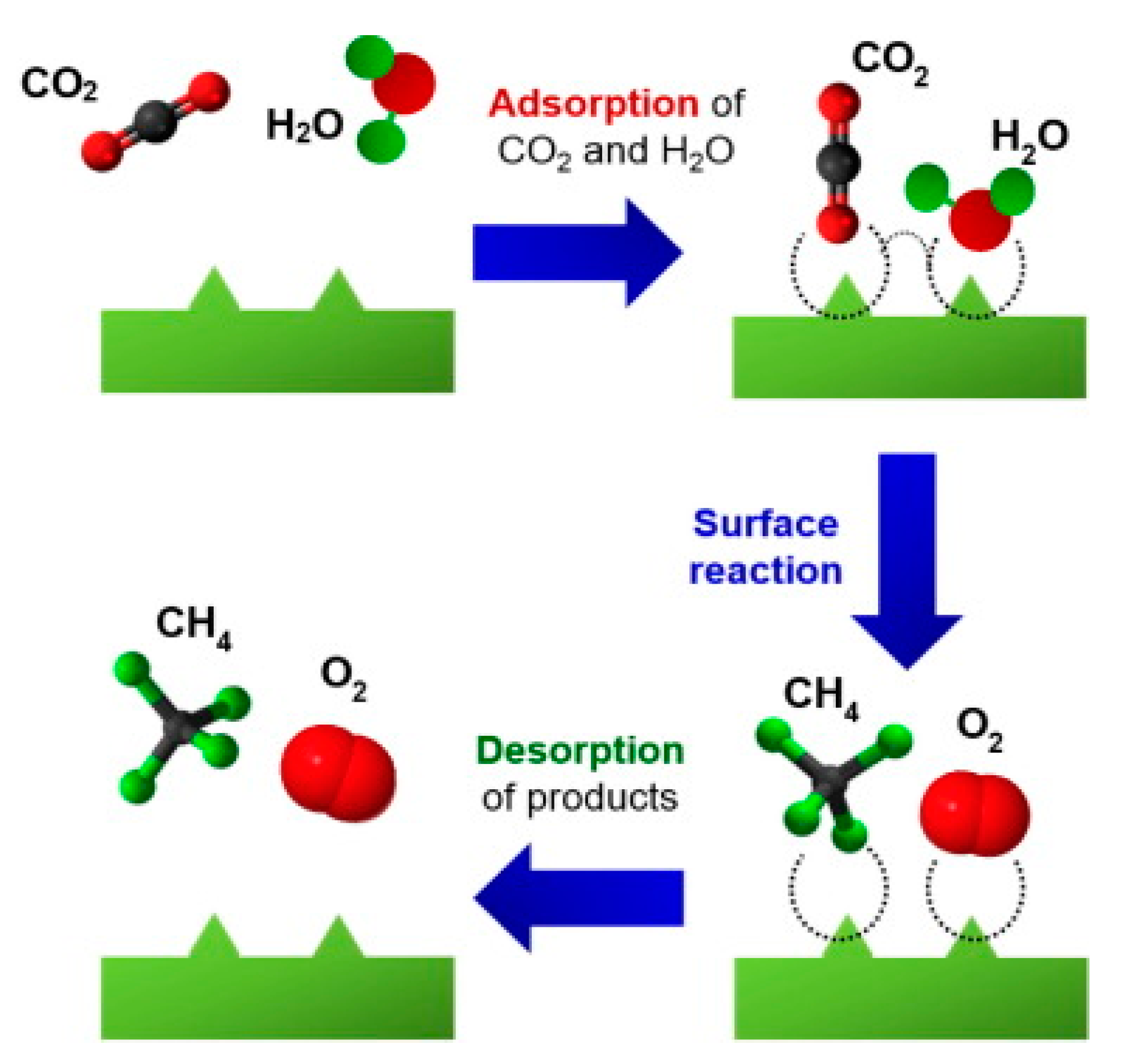
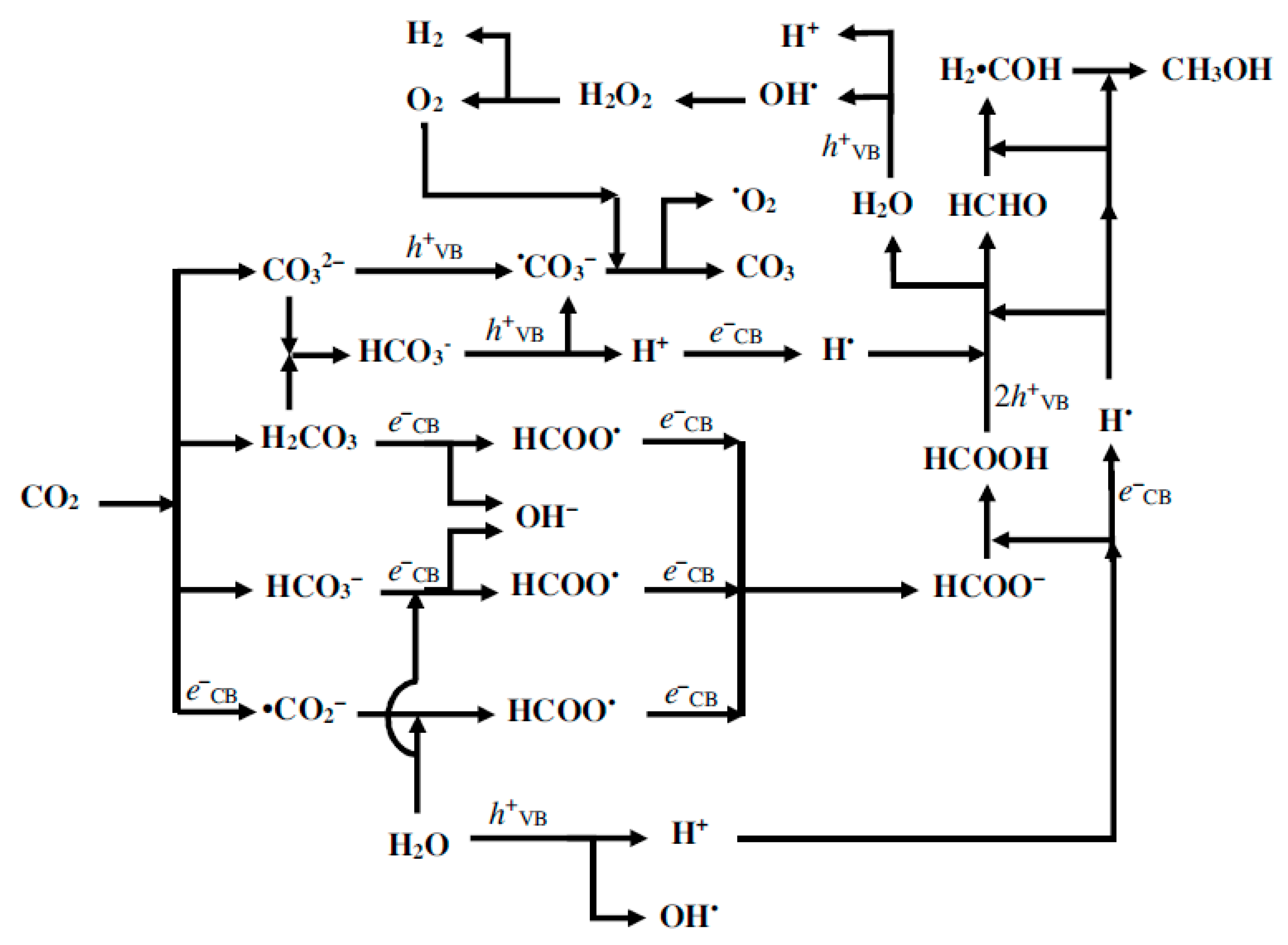
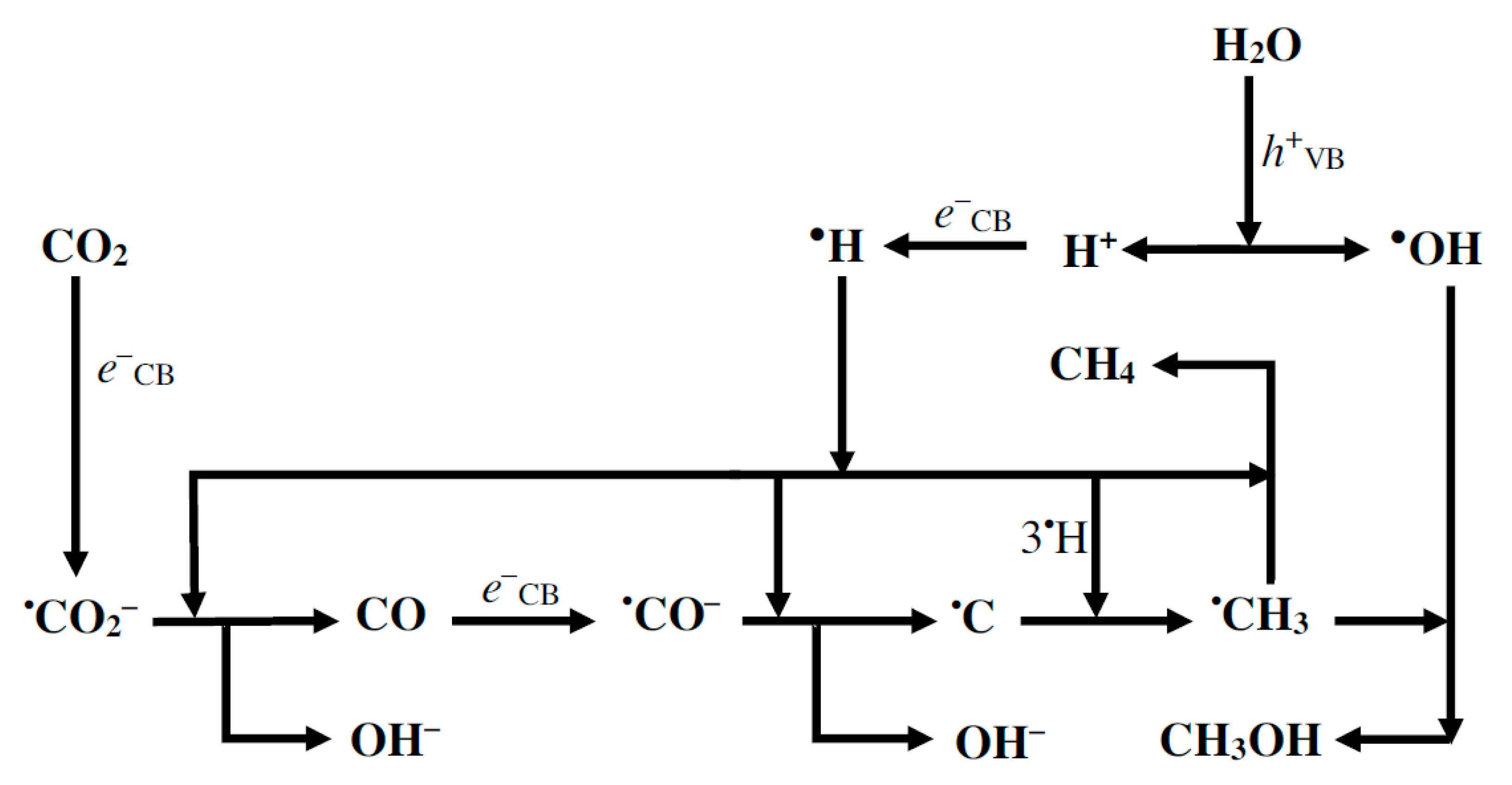
2. Photocatalytic Conversion of CO2: Thermodynamics and Kinetics
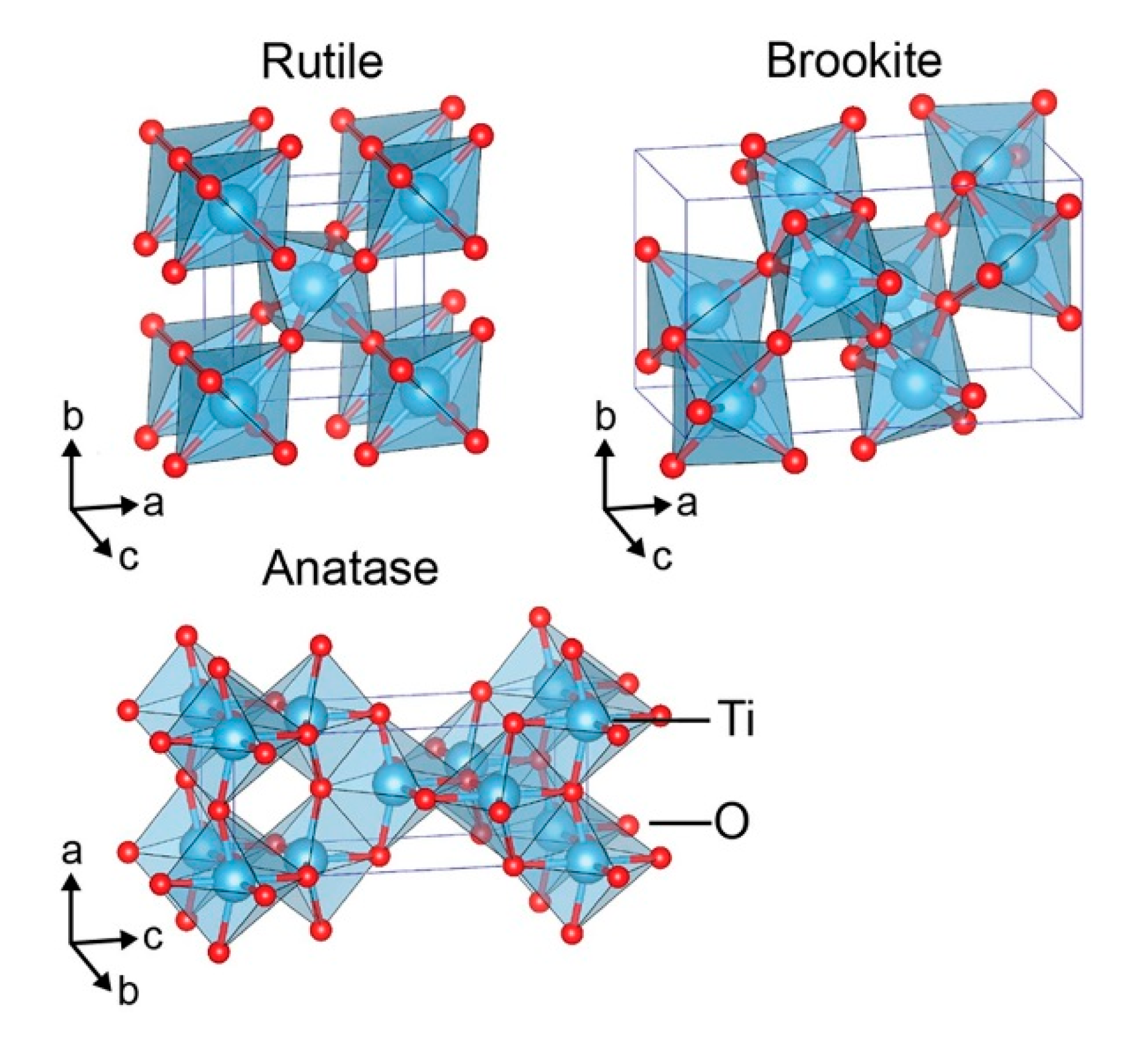
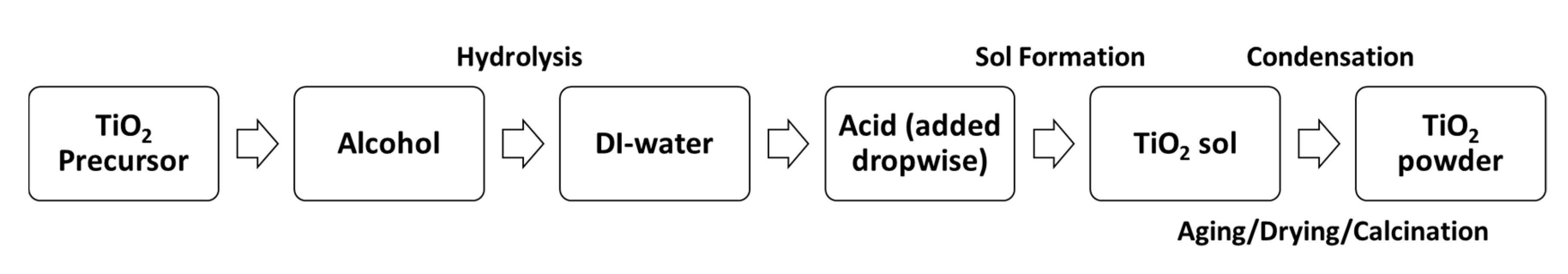
3. Titanium Dioxide: Synthesis and Surface Modification Strategies for Enhanced CO2 Photoreduction
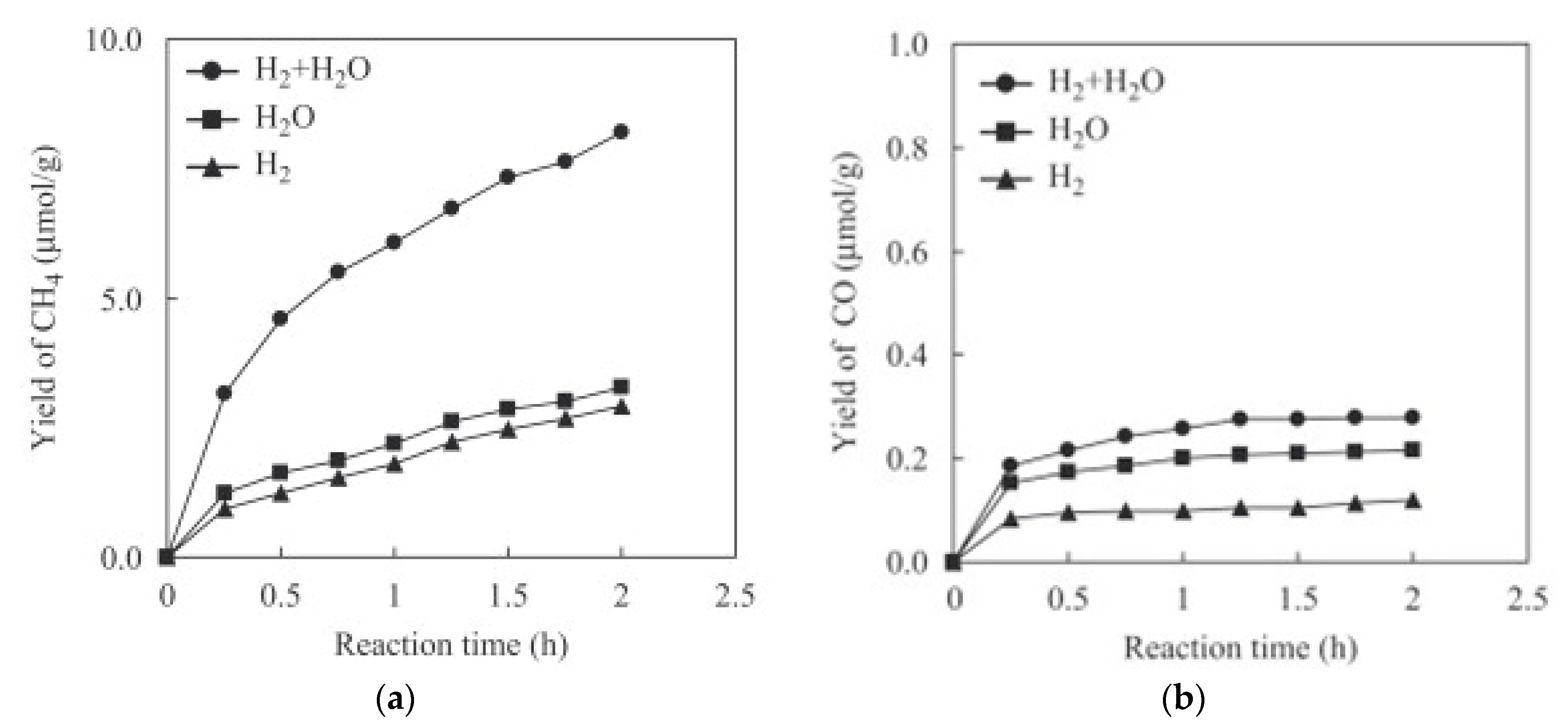
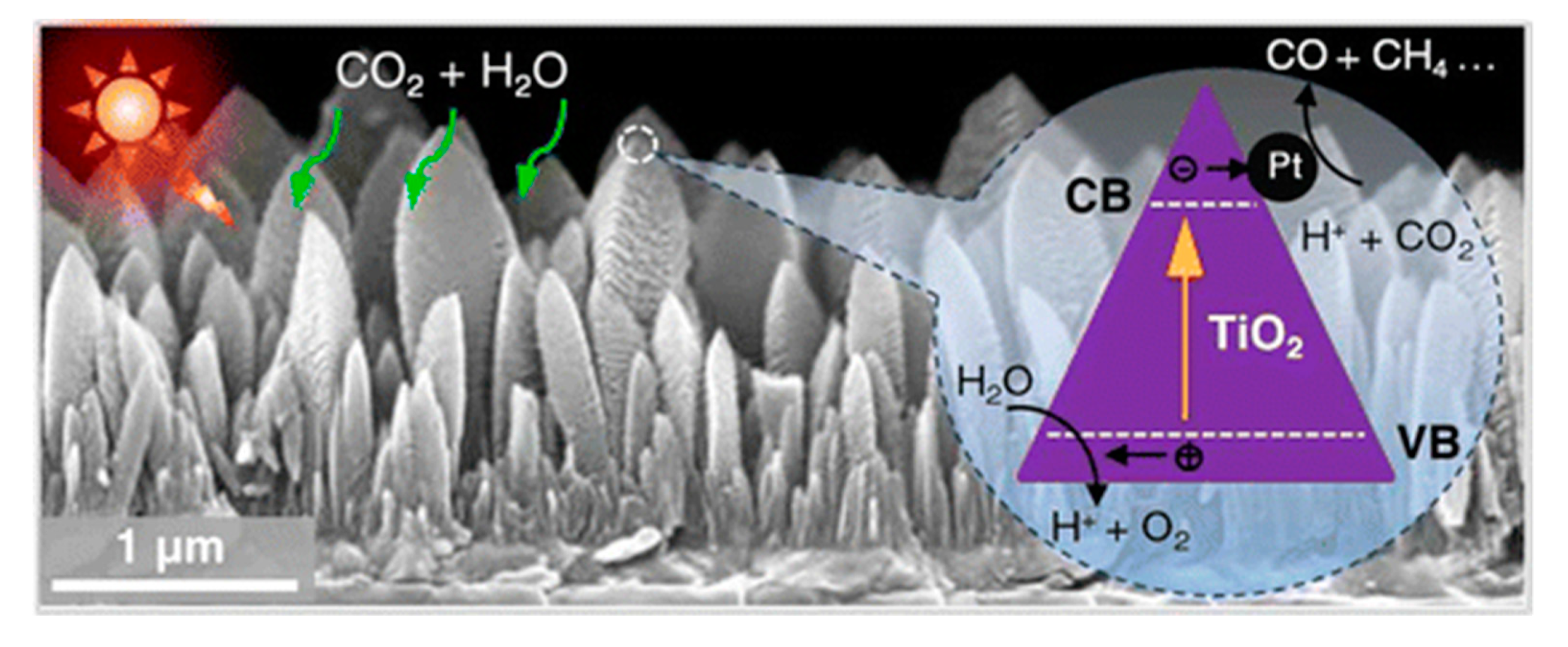
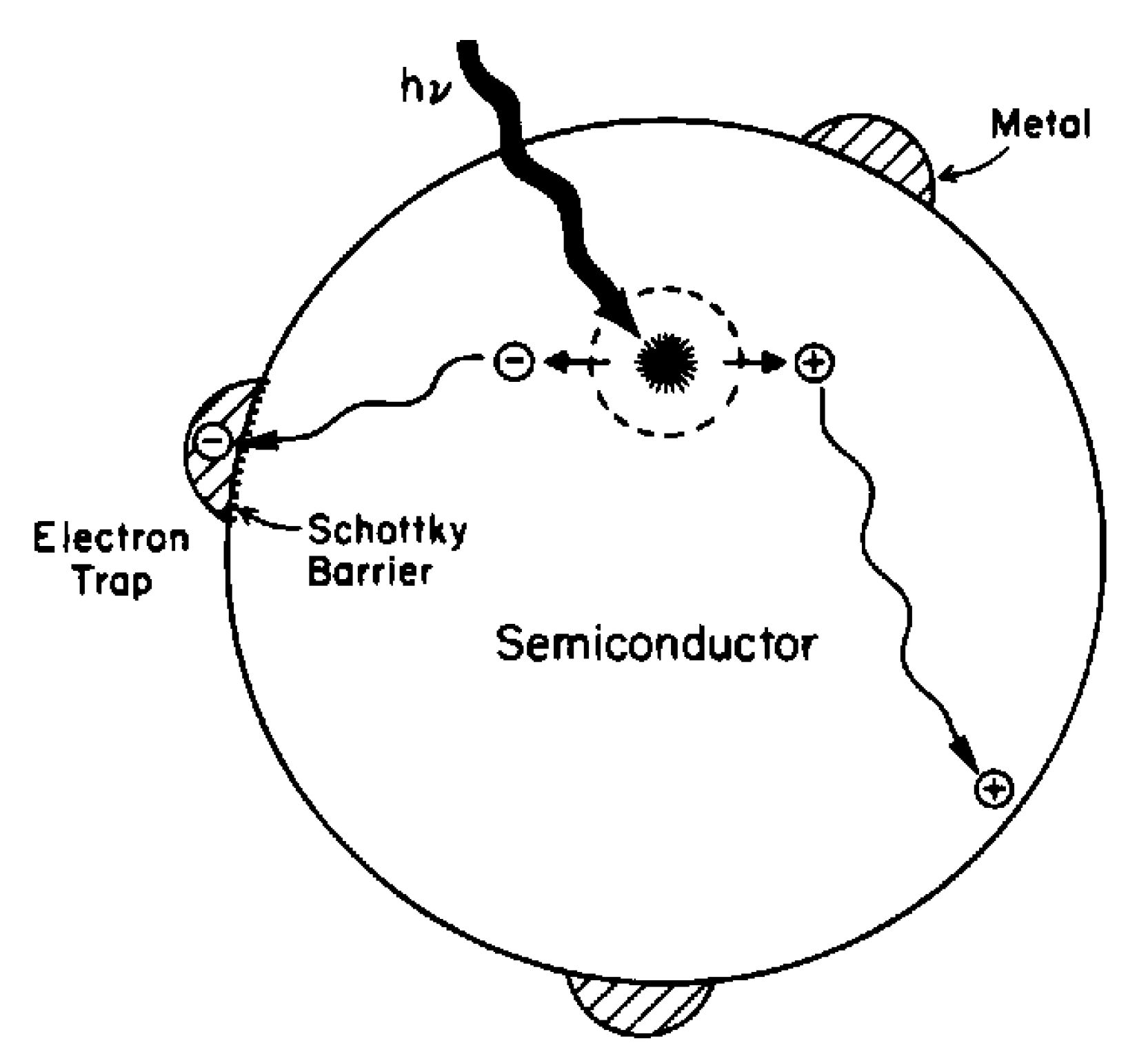
3.1. Metal Deposition
3.2. Doping
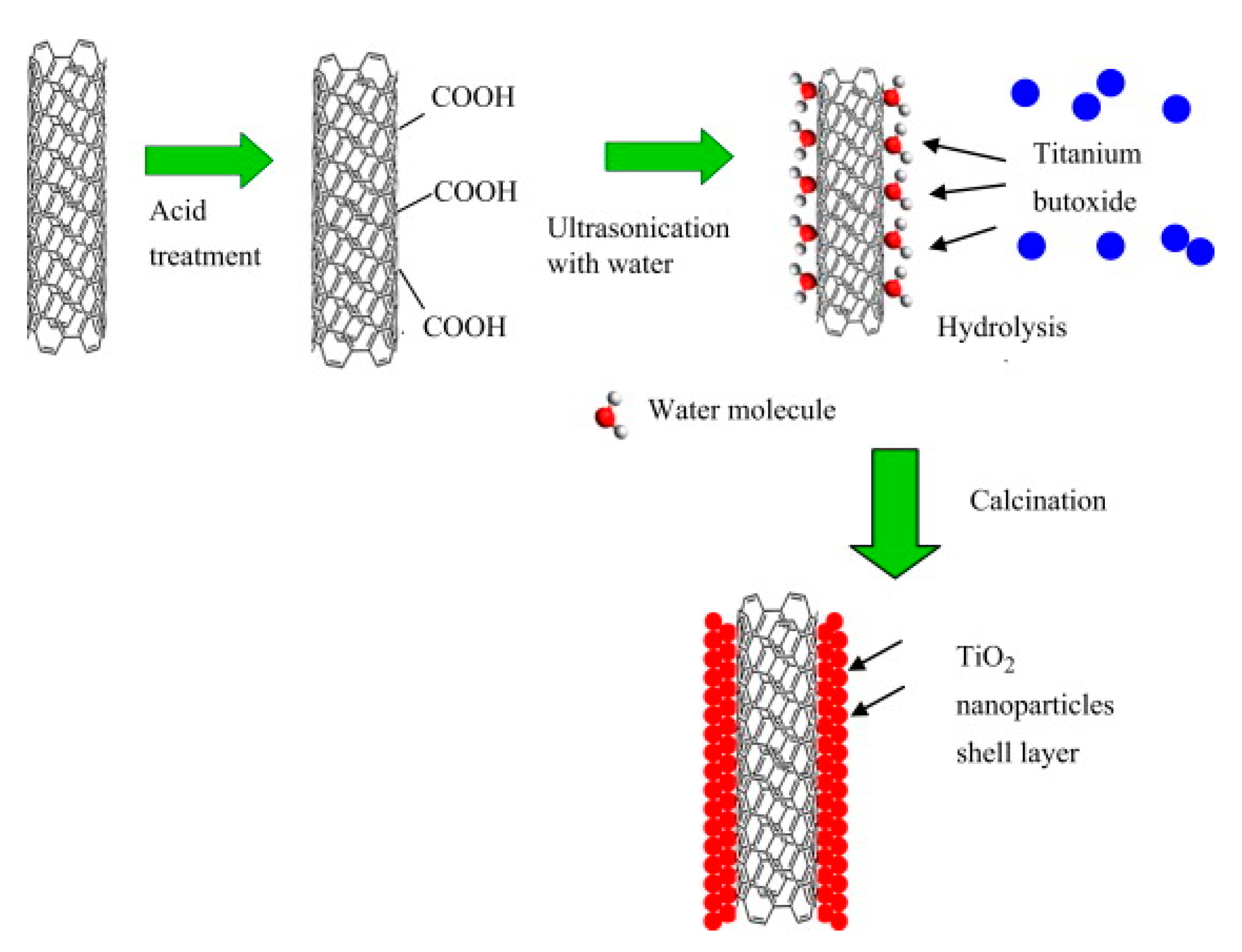
3.3. Carbon-Based Material Loading
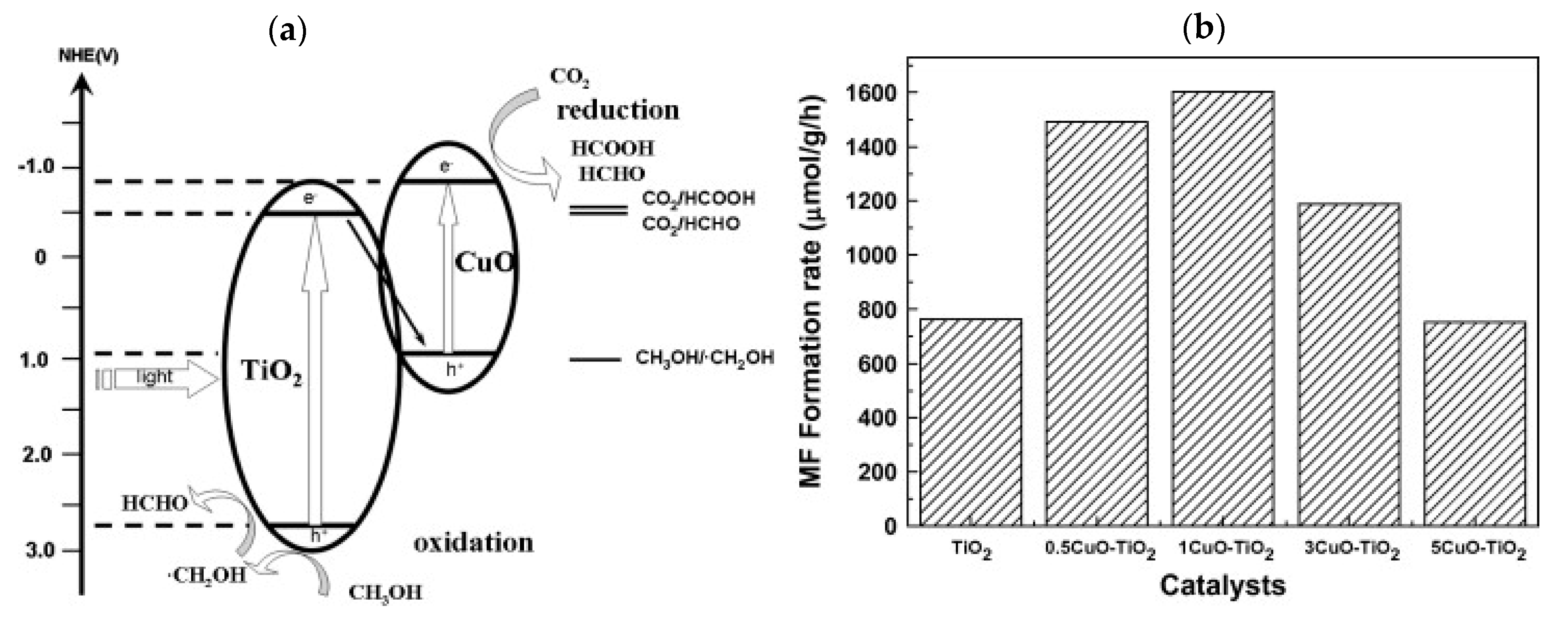
3.4. Heterostructures
3.5. Dispersion on Supports
4. Conclusions and Future Perspective
Funding
Conflicts of Interest
References
- Ryaboshapko, A.G.; Revokatova, A.P. A potential role of the negative emission of carbon dioxide in solving the climate problem. Russ. Meteorol. Hydrol. 2015, 40, 443–455. [Google Scholar] [CrossRef]
- Nordhaus, W.D. The Climate Casino: Risk, Uncertainty, and Economics for a Warming World; Yale University Press: New Haven, CT, USA, 2013. [Google Scholar]
- Gore, A. The Crisis Comes Ashore. New Repub. 2010, 241, 10–12. [Google Scholar]
- Shin, D.Y.; Jo, J.H.; Lee, J.-Y.; Lim, D.-H. Understanding mechanisms of carbon dioxide conversion into methane for designing enhanced catalysts from first-principles. Comput. Chem. 2016, 1083, 31–37. [Google Scholar] [CrossRef]
- Holdren, J.P. Population and the energy problem. Popul. Environ. 1991, 12, 231–255. [Google Scholar] [CrossRef]
- Sanchez, J.A. Carbon Dioxide Capture: Processes, Technology and Environmental Implications; Nova Science Publishers, Inc.: Hauppauge, NY, USA, 2016. [Google Scholar]
- Spigarelli, B.P.; Kawatra, S.K. Opportunities and challenges in carbon dioxide capture. J. Co2 Util. 2013, 1, 69–87. [Google Scholar] [CrossRef]
- Cheah, W.Y.; Ling, T.C.; Juan, J.C.; Lee, D.-J.; Chang, J.-S.; Show, P.L. Biorefineries of carbon dioxide: From carbon capture and storage (CCS) to bioenergies production. Bioresour. Technol. 2016, 215, 346–356. [Google Scholar] [CrossRef]
- Koytsoumpa, E.I.; Bergins, C.; Kakaras, E. The CO2 economy: Review of CO2 capture and reuse technologies. J. Supercrit. Fluids 2018, 132, 3–16. [Google Scholar] [CrossRef]
- Sahebdelfar, S.; Takht Ravanchi, M. Carbon dioxide utilization for methane production: A thermodynamic analysis. J. Pet. Sci. Eng. 2015, 134, 14–22. [Google Scholar] [CrossRef]
- Centi, G.; Perathoner, S. Opportunities and prospects in the chemical recycling of carbon dioxide to fuels. Catal. Today 2009, 148, 191–205. [Google Scholar] [CrossRef]
- Muradov, N. Industrial Utilization of CO2: A Win–Win Solution. In Liberating Energy from Carbon: Introduction to Decarbonization; Springer: New York, NY, USA, 2014; pp. 325–383. [Google Scholar] [CrossRef]
- Ganesh, I. Conversion of carbon dioxide into methanol—A potential liquid fuel: Fundamental challenges and opportunities (a review). Renew. Sustain. Energy Rev. 2014, 31, 221–257. [Google Scholar] [CrossRef]
- Arakawa, H.; Aresta, M.; Armor, J.N.; Barteau, M.A.; Beckman, E.J.; Bell, A.T.; Bercaw, J.E.; Creutz, C.; Dinjus, E.; Dixon, D.A.; et al. Catalysis Research of Relevance to Carbon Management: Progress, Challenges, and Opportunities. Chem. Rev. 2001, 101, 953–996. [Google Scholar] [CrossRef]
- Liu, X.-M.; Lu, G.Q.; Yan, Z.-F.; Beltramini, J. Recent Advances in Catalysts for Methanol Synthesis via Hydrogenation of CO and CO2. Ind. Eng. Chem. Res. 2003, 42, 6518–6530. [Google Scholar] [CrossRef]
- Farrelly, D.J.; Everard, C.D.; Fagan, C.C.; McDonnell, K.P. Carbon sequestration and the role of biological carbon mitigation: A review. Renew. Sustain. Energy Rev. 2013, 21, 712–727. [Google Scholar] [CrossRef]
- Alaswad, A.; Dassisti, M.; Prescott, T.; Olabi, A.G. Technologies and developments of third generation biofuel production. Renew. Sustain. Energy Rev. 2015, 51, 1446–1460. [Google Scholar] [CrossRef]
- Ramachandriya, K.D.; Kundiyana, D.K.; Wilkins, M.R.; Terrill, J.B.; Atiyeh, H.K.; Huhnke, R.L. Carbon dioxide conversion to fuels and chemicals using a hybrid green process. Appl. Energy 2013, 112, 289–299. [Google Scholar] [CrossRef]
- de_Richter, R.K.; Ming, T.; Caillol, S. Fighting global warming by photocatalytic reduction of CO2 using giant photocatalytic reactors. Renew. Sustain. Energy Rev. 2013, 19, 82–106. [Google Scholar] [CrossRef]
- Hoffmann, M.R.; Moss, J.A.; Baum, M.M. Artificial photosynthesis: Semiconductor photocatalytic fixation of CO2 to afford higher organic compounds. Dalton Trans. 2011, 40, 5151–5158. [Google Scholar] [CrossRef]
- Kočí, K.; Obalová, L.; Lacný, Z. Photocatalytic reduction of CO2 over TiO2 based catalysts. Chem. Pap. 2008, 62, 1–9. [Google Scholar] [CrossRef]
- Habisreutinger, S.N.; Schmidt-Mende, L.; Stolarczyk, J.K. Photocatalytic reduction of CO2 on TiO2 and other semiconductors. Angew. Chem. Int. Ed. 2013, 52, 7372–7408. [Google Scholar] [CrossRef]
- Ola, O.; Maroto-Valer, M.M. Review of material design and reactor engineering on TiO2 photocatalysis for CO2 reduction. J. Photochem. Photobiol. C 2015, 24, 16–42. [Google Scholar] [CrossRef]
- Kumar, A.; Ashok, A.; Shurair, M.S.; Yussuf, K.; van den Broeke, L.J. Carbon Management: Regional Solutions Based on Carbon Dioxide Utilization and Process Integration. In Proceedings of the 4th International Gas Processing Symposium, Doha, Qatar, 26–27 October 2014; Elsevier: Oxford, UK, 2015. [Google Scholar]
- Li, K.; An, X.; Park, K.H.; Khraisheh, M.; Tang, J. A critical review of CO2 photoconversion: Catalysts and reactors. Catal. Today 2014, 224, 3–12. [Google Scholar] [CrossRef]
- Meryem, S.S.; Nasreen, S.; Siddique, M.; Khan, R. An overview of the reaction conditions for an efficient photoconversion of CO2. Rev. Chem. Eng. 2018, 34, 409–425. [Google Scholar] [CrossRef]
- Mallouk, T.E. The Emerging Technology of Solar Fuels. J. Phys. Chem. Lett. 2010, 1, 2738–2739. [Google Scholar] [CrossRef]
- Shi, R.; Waterhouse, G.I.N.; Zhang, T. Recent Progress in Photocatalytic CO2 Reduction Over Perovskite Oxides. Sol. Rrl 2017, 1, 1700126. [Google Scholar] [CrossRef]
- AlOtaibi, B.; Fan, S.; Wang, D.; Ye, J.; Mi, Z. Wafer-level artificial photosynthesis for CO2 reduction into CH4 and CO using GaN nanowires. ACS Catal. 2015, 5, 5342–5348. [Google Scholar] [CrossRef]
- Zhang, Y.; Xu, C.; Chen, J.; Zhang, X.; Wang, Z.; Zhou, J.; Cen, K. A novel photo-thermochemical cycle for the dissociation of CO2 using solar energy. Appl. Energy 2015, 156, 223–229. [Google Scholar] [CrossRef]
- Wang, P.; Wang, S.; Wang, H.; Wu, Z.; Wang, L. Recent Progress on Photo-Electrocatalytic Reduction of Carbon Dioxide. Part. Part. Syst. Char. 2018, 35, 1700371. [Google Scholar] [CrossRef]
- Halmann, M. Photoelectrochemical reduction of aqueous carbon dioxide on p-type gallium phosphide in liquid junction solar cells. Nature 1978, 275, 115–116. [Google Scholar] [CrossRef]
- Kumar, S.G.; Devi, L.G. Review on Modified TiO2 Photocatalysis under UV/Visible Light: Selected Results and Related Mechanisms on Interfacial Charge Carrier Transfer Dynamics. J. Phys. Chem. A 2011, 115, 13211–13241. [Google Scholar] [CrossRef]
- Daghrir, R.; Drogui, P.; Robert, D. Modified TiO2 For Environmental Photocatalytic Applications: A Review. Ind. Eng. Chem. Res. 2013, 52, 3581–3599. [Google Scholar] [CrossRef]
- Wen, J.; Li, X.; Liu, W.; Fang, Y.; Xie, J.; Xu, Y. Photocatalysis fundamentals and surface modification of TiO2 nanomaterials. Chin. J. Catal. 2015, 36, 2049–2070. [Google Scholar] [CrossRef]
- Li, X.; Zhuang, Z.; Li, W.; Pan, H. Photocatalytic reduction of CO2 over noble metal-loaded and nitrogen-doped mesoporous TiO2. Appl. Catal. A 2012, 429–430, 31–38. [Google Scholar] [CrossRef]
- Corma, A.; Garcia, H. Photocatalytic reduction of CO2 for fuel production: Possibilities and challenges. J. Catal. 2013, 308, 168–175. [Google Scholar] [CrossRef]
- Qin, S.; Xin, F.; Liu, Y.; Yin, X.; Ma, W. Photocatalytic reduction of CO2 in methanol to methyl formate over CuO–TiO2 composite catalysts. J. Colloid Interface Sci. 2011, 356, 257–261. [Google Scholar] [CrossRef]
- Karamian, E.; Sharifnia, S. On the general mechanism of photocatalytic reduction of CO2. J. Co2 Util. 2016, 16, 194–203. [Google Scholar] [CrossRef]
- Low, J.; Cheng, B.; Yu, J. Surface modification and enhanced photocatalytic CO2 reduction performance of TiO2: A review. Appl. Surf. Sci. 2017, 392, 658–686. [Google Scholar] [CrossRef]
- Dilla, M.; Schlögl, R.; Strunk, J. Photocatalytic CO2 Reduction Under Continuous Flow High-Purity Conditions: Quantitative Evaluation of CH4 Formation in the Steady-State. ChemCatChem 2017, 9, 696–704. [Google Scholar] [CrossRef]
- Mino, L.; Spoto, G.; Ferrari, A.M. CO2 capture by TiO2 anatase surfaces: A combined DFT and FTIR study. J. Phys. Chem. C 2014, 118, 25016–25026. [Google Scholar] [CrossRef]
- Sorescu, D.C.; Al-Saidi, W.A.; Jordan, K.D. CO2 adsorption on TiO2 (101) anatase: A dispersion-corrected density functional theory study. J. Chem. Phys. 2011, 135, 124701. [Google Scholar] [CrossRef]
- Tu, W.; Zhou, Y.; Zou, Z. Photocatalytic conversion of CO2 into renewable hydrocarbon fuels: State-of-the-art accomplishment, challenges, and prospects. Adv. Mater. 2014, 26, 4607–4626. [Google Scholar] [CrossRef]
- Yuan, L.; Xu, Y.-J. Photocatalytic conversion of CO2 into value-added and renewable fuels. Appl. Surf. Sci. 2015, 342, 154–167. [Google Scholar] [CrossRef]
- Halmann, M. 15—Photochemical Fixation of Carbon Dioxide. In Energy Resources Through Photochemistry and Catalysis; Grätzel, M., Ed.; Academic Press: Cambridge, MA, USA, 1983; pp. 507–534. Available online: https://linkinghub.elsevier.com/retrieve/pii/B9780122957208500193 (accessed on 1 December 2019). [CrossRef]
- Tan, L.-L.; Ong, W.-J.; Chai, S.-P.; Mohamed, A.R. Photocatalytic reduction of CO2 with H2O over graphene oxide-supported oxygen-rich TiO2 hybrid photocatalyst under visible light irradiation: Process and kinetic studies. Chem. Eng. J. 2017, 308, 248–255. [Google Scholar] [CrossRef]
- Liu, L.; Li, Y. Understanding the reaction mechanism of photocatalytic reduction of CO2 with H2O on TiO2-based photocatalysts: A review. Aerosol Air Qual. Res. 2014, 14, 453–469. [Google Scholar] [CrossRef]
- Uner, D.; Oymak, M.M. On the mechanism of photocatalytic CO2 reduction with water in the gas phase. Catal. Today 2012, 181, 82–88. [Google Scholar] [CrossRef]
- Saladin, F.; Alxneit, I. Temperature dependence of the photochemical reduction of CO2 in the presence of H2 Oat the solid/gas interface of TiO2. J. Chem. Soc. Faraday Trans. 1997, 93, 4159–4163. [Google Scholar] [CrossRef]
- Handoko, A.D.; Li, K.; Tang, J. Recent progress in artificial photosynthesis: CO2 photoreduction to valuable chemicals in a heterogeneous system. Curr. Opin. Chem. Eng. 2013, 2, 200–206. [Google Scholar] [CrossRef]
- Subrahmanyam, M.; Kaneco, S.; Alonso-Vante, N. A screening for the photo reduction of carbon dioxide supported on metal oxide catalysts for C1–C3 selectivity. Appl. Catal. B 1999, 23, 169–174. [Google Scholar] [CrossRef]
- Sasirekha, N.; Basha, S.J.S.; Shanthi, K. Photocatalytic performance of Ru doped anatase mounted on silica for reduction of carbon dioxide. Appl. Catal. B 2006, 62, 169–180. [Google Scholar] [CrossRef]
- Akhter, P.; Hussain, M.; Saracco, G.; Russo, N. Novel nanostructured-TiO2 materials for the photocatalytic reduction of CO2 greenhouse gas to hydrocarbons and syngas. Fuel 2015, 149, 55–65. [Google Scholar] [CrossRef]
- Klyukin, K.; Alexandrov, V. CO2 Adsorption and Reactivity on Rutile TiO2(110) in Water: An Ab Initio Molecular Dynamics Study. J. Phys. Chem. C 2017, 121, 10476–10483. [Google Scholar] [CrossRef]
- Liu, B.-J.; Torimoto, T.; Matsumoto, H.; Yoneyama, H. Effect of solvents on photocatalytic reduction of carbon dioxide using TiO2 nanocrystal photocatalyst embedded in SiO2 matrices. J. Photochem. Photobiol. A 1997, 108, 187–192. [Google Scholar] [CrossRef]
- Kočí, K.; Obalová, L.; Matějová, L.; Plachá, D.; Lacný, Z.; Jirkovský, J.; Šolcová, O. Effect of TiO2 particle size on the photocatalytic reduction of CO2. Appl. Catal. B 2009, 89, 494–502. [Google Scholar] [CrossRef]
- Tseng, I.-H.; Chang, W.-C.; Wu, J.C. Photoreduction of CO2 using sol–gel derived titania and titania-supported copper catalysts. Appl. Catal. B 2002, 37, 37–48. [Google Scholar] [CrossRef]
- Sastre, F.; Corma, A.; García, H. 185 nm Photoreduction of CO2 to Methane by Water. Influence of the Presence of a Basic Catalyst. J. Am. Chem. Soc. 2012, 134, 14137–14141. [Google Scholar] [CrossRef] [PubMed]
- Lo, C.-C.; Hung, C.-H.; Yuan, C.-S.; Wu, J.-F. Photoreduction of carbon dioxide with H2 and H2O over TiO2 and ZrO2 in a circulated photocatalytic reactor. Sol. Energy Mater. Sol. Cells 2007, 91, 1765–1774. [Google Scholar] [CrossRef]
- Grills, D.C.; Fujita, E. New directions for the photocatalytic reduction of CO2: Supramolecular, scCO2 or biphasic ionic liquid—scCO2 systems. J. Phys. Chem. Lett. 2010, 1, 2709–2718. [Google Scholar] [CrossRef]
- Kaneco, S.; Kurimoto, H.; Ohta, K.; Mizuno, T.; Saji, A. Photocatalytic reduction of CO2 using TiO2 powders in liquid CO2 medium. J. Photochem. Photobiol. A 1997, 109, 59–63. [Google Scholar] [CrossRef]
- Kaneco, S.; Kurimoto, H.; Shimizu, Y.; Ohta, K.; Mizuno, T. Photocatalytic reduction of CO2 using TiO2 powders in supercritical fluid CO2. Energy 1999, 24, 21–30. [Google Scholar] [CrossRef]
- Kometani, N.; Hirata, S.; Chikada, M. Photocatalytic reduction of CO2 by Pt-loaded TiO2 in the mixture of sub-and supercritical water and CO2. J. Supercrit. Fluids 2017, 120, 443–447. [Google Scholar] [CrossRef]
- Liu, L.; Zhao, H.; Andino, J.M.; Li, Y. Photocatalytic CO2 reduction with H2O on TiO2 nanocrystals: Comparison of anatase, rutile, and brookite polymorphs and exploration of surface chemistry. ACS Catal. 2012, 2, 1817–1828. [Google Scholar] [CrossRef]
- Chen, L.; Graham, M.E.; Li, G.; Gentner, D.R.; Dimitrijevic, N.M.; Gray, K.A. Photoreduction of CO2 by TiO2 nanocomposites synthesized through reactive direct current magnetron sputter deposition. Thin Solid Film. 2009, 517, 5641–5645. [Google Scholar] [CrossRef]
- Haggerty, J.E.S.; Schelhas, L.T.; Kitchaev, D.A.; Mangum, J.S.; Garten, L.M.; Sun, W.; Stone, K.H.; Perkins, J.D.; Toney, M.F.; Ceder, G.; et al. High-fraction brookite films from amorphous precursors. Sci. Rep. 2017, 7, 15232. [Google Scholar] [CrossRef] [PubMed]
- Zhang, P.; Tang, B.; Xia, W.; Wang, F.; Wei, X.; Xiang, J.; Li, J. Preparation and Characterizations of TiO2 Nanoparticles by Sol-Gel Process using DMAC Solvent. MMECEB 2015, 2016, 892–895. [Google Scholar]
- Akpan, U.; Hameed, B. The advancements in sol–gel method of doped-TiO2 photocatalysts. Appl. Catal. A 2010, 375, 1–11. [Google Scholar] [CrossRef]
- Wang, Y.; He, Y.; Lai, Q.; Fan, M. Review of the progress in preparing nano TiO2: An important environmental engineering material. J. Environ. Sci. 2014, 26, 2139–2177. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Lunsford, S.K.; Song, Y.; Ju, H.; Falaras, P.; likodimos, V.; Kontos, A.G.; Dionysiou, D.D. Synthesis, characterization and electrochemical properties of mesoporous zirconia nanomaterials prepared by self-assembling sol–gel method with Tween 20 as a template. Chem. Eng. J. 2011, 170, 518–524. [Google Scholar] [CrossRef]
- Sun, Y.; Seo, J.H.; Takacs, C.J.; Seifter, J.; Heeger, A.J. Inverted polymer solar cells integrated with a low-temperature-annealed sol-gel-derived ZnO film as an electron transport layer. Adv. Mater. 2011, 23, 1679–1683. [Google Scholar] [CrossRef]
- Djaoued, Y.; Balaji, S.; Beaudoin, N. Sol–gel synthesis of mesoporous WO3–TiO2 composite thin films for photochromic devices. J. Sol-Gel Sci. Technol. 2013, 65, 374–383. [Google Scholar] [CrossRef]
- Chen, C.; Cai, W.; Long, M.; Zhang, J.; Zhou, B.; Wu, Y.; Wu, D. Template-free sol–gel preparation and characterization of free-standing visible light responsive C, N-modified porous monolithic TiO2. J. Hazard. Mater. 2010, 178, 560–565. [Google Scholar] [CrossRef]
- Tseng, T.K.; Lin, Y.S.; Chen, Y.J.; Chu, H. A review of photocatalysts prepared by sol-gel method for VOCs removal. Int. J. Mol. Sci. 2010, 11, 2336–2361. [Google Scholar] [CrossRef]
- Lu, J.; Li, L.; Wang, Z.; Wen, B.; Cao, J. Synthesis of visible-light-active TiO2-based photo-catalysts by a modified sol–gel method. Mater. Lett. 2013, 94, 147–149. [Google Scholar] [CrossRef]
- You, Y.; Zhang, S.; Wan, L.; Xu, D. Preparation of continuous TiO2 fibers by sol–gel method and its photocatalytic degradation on formaldehyde. Appl. Surf. Sci. 2012, 258, 3469–3474. [Google Scholar] [CrossRef]
- Xie, Y.; Zhao, Q.; Zhao, X.J.; Li, Y. Low temperature preparation and characterization of N-doped and NS-codoped TiO 2 by sol–gel route. Catal. Lett. 2007, 118, 231–237. [Google Scholar] [CrossRef]
- Šegota, S.; Ćurković, L.; Ljubas, D.; Svetličić, V.; Houra, I.F.; Tomašić, N. Synthesis, characterization and photocatalytic properties of sol–gel TiO2 films. Ceram. Int. 2011, 37, 1153–1160. [Google Scholar] [CrossRef]
- Kim, M.-S.; Liu, G.; Nam, W.K.; Kim, B.-W. Preparation of porous carbon-doped TiO2 film by sol–gel method and its application for the removal of gaseous toluene in the optical fiber reactor. J. Ind. Eng. Chem. 2011, 17, 223–228. [Google Scholar] [CrossRef]
- Macwan, D.; Dave, P.N.; Chaturvedi, S. A review on nano-TiO 2 sol–gel type syntheses and its applications. Asian J. Mater. Sci. 2011, 46, 3669–3686. [Google Scholar] [CrossRef]
- Payakgul, W.; Mekasuwandumrong, O.; Pavarajarn, V.; Praserthdam, P. Effects of reaction medium on the synthesis of TiO2 nanocrystals by thermal decomposition of titanium (IV) n-butoxide. Ceram. Int. 2005, 31, 391–397. [Google Scholar] [CrossRef]
- Liu, Z.; Sun, D.D.; Guo, P.; Leckie, J.O. One-step fabrication and high photocatalytic activity of porous TiO2 hollow aggregates by using a low-temperature hydrothermal method without templates. Chem. Eur. J. 2007, 13, 1851–1855. [Google Scholar] [CrossRef]
- Ou, H.-H.; Lo, S.-L. Review of titania nanotubes synthesized via the hydrothermal treatment: Fabrication, modification, and application. Sep. Purif. Technol. 2007, 58, 179–191. [Google Scholar] [CrossRef]
- Fumin, W.; Zhansheng, S.; Feng, G.; Jinting, J.; Adachi, M. Morphology control of anatase TiO2 by surfactant-assisted hydrothermal method. Chin. J. Chem. Eng. 2007, 15, 754–759. [Google Scholar]
- Camarillo, R.; Tostón, S.; Martínez, F.; Jiménez, C.; Rincón, J. Preparation of TiO2-based catalysts with supercritical fluid technology: Characterization and photocatalytic activity in CO2 reduction. J. Chem. Technol. Biotechnol. 2017, 92, 1710–1720. [Google Scholar] [CrossRef]
- Bellardita, M.; Di Paola, A.; García-López, E.; Loddo, V.; Marcì, G.; Palmisano, L. Photocatalytic CO2 reduction in gas-solid regime in the presence of bare, SiO2 supported or Cu-loaded TiO2 samples. Curr. Org. Chem. 2013, 17, 2440–2448. [Google Scholar] [CrossRef]
- Maeda, K. Photocatalytic water splitting using semiconductor particles: History and recent developments. J. Photochem. Photobiol. C 2011, 12, 237–268. [Google Scholar] [CrossRef]
- Humayun, M.; Raziq, F.; Khan, A.; Luo, W. Modification strategies of TiO2 for potential applications in photocatalysis: A critical review. Green Chem. Lett. Rev. 2018, 11, 86–102. [Google Scholar] [CrossRef]
- Meng, X.; Ouyang, S.; Kako, T.; Li, P.; Yu, Q.; Wang, T.; Ye, J. Photocatalytic CO2 conversion over alkali modified TiO2 without loading noble metal cocatalyst. Chem. Commun. 2014, 50, 11517–11519. [Google Scholar] [CrossRef]
- Wang, W.-N.; An, W.-J.; Ramalingam, B.; Mukherjee, S.; Niedzwiedzki, D.M.; Gangopadhyay, S.; Biswas, P. Size and structure matter: Enhanced CO2 photoreduction efficiency by size-resolved ultrafine Pt nanoparticles on TiO2 single crystals. J. Am. Chem. Soc. 2012, 134, 11276–11281. [Google Scholar] [CrossRef]
- Tahir, M.; Tahir, B.; Amin, N.A.S. Gold-nanoparticle-modified TiO2 nanowires for plasmon-enhanced photocatalytic CO2 reduction with H2 under visible light irradiation. Appl. Surf. Sci. 2015, 356, 1289–1299. [Google Scholar] [CrossRef]
- Kong, D.; Tan, J.Z.Y.; Yang, F.; Zeng, J.; Zhang, X. Electrodeposited Ag nanoparticles on TiO2 nanorods for enhanced UV visible light photoreduction CO2 to CH4. Appl. Surf. Sci. 2013, 277, 105–110. [Google Scholar] [CrossRef]
- Zhai, Q.; Xie, S.; Fan, W.; Zhang, Q.; Wang, Y.; Deng, W.; Wang, Y. Photocatalytic conversion of carbon dioxide with water into methane: Platinum and copper (I) oxide co-catalysts with a core–shell structure. Angew. Chem. 2013, 125, 5888–5891. [Google Scholar] [CrossRef]
- Yan, Y.; Yu, Y.; Cao, C.; Huang, S.; Yang, Y.; Yang, X.; Cao, Y. Enhanced photocatalytic activity of TiO2–Cu/C with regulation and matching of energy levels by carbon and copper for photoreduction of CO2 into CH4. CrystEngComm 2016, 18, 2956–2964. [Google Scholar] [CrossRef]
- Wu, J.C.S.; Lin, H.-M.; Lai, C.-L. Photo reduction of CO2 to methanol using optical-fiber photoreactor. Appl. Catal. A 2005, 296, 194–200. [Google Scholar] [CrossRef]
- Nasution, H.W.; Purnama, E.; Kosela, S.; Gunlazuardi, J. Photocatalytic reduction of CO2 on copper-doped Titania catalysts prepared by improved-impregnation method. Catal. Commun. 2005, 6, 313–319. [Google Scholar] [CrossRef]
- Kwak, B.S.; Vignesh, K.; Park, N.-K.; Ryu, H.-J.; Baek, J.-I.; Kang, M. Methane formation from photoreduction of CO2 with water using TiO2 including Ni ingredient. Fuel 2015, 143, 570–576. [Google Scholar] [CrossRef]
- Matějová, L.; Kočí, K.; Reli, M.; Čapek, L.; Hospodková, A.; Peikertová, P.; Matěj, Z.; Obalová, L.; Wach, A.; Kuśtrowski, P.; et al. Preparation, characterization and photocatalytic properties of cerium doped TiO2: On the effect of Ce loading on the photocatalytic reduction of carbon dioxide. Appl. Catal. B 2014, 152–153, 172–183. [Google Scholar] [CrossRef]
- Zhao, Z.; Fan, J.; Wang, J.; Li, R. Effect of heating temperature on photocatalytic reduction of CO2 by N–TiO2 nanotube catalyst. Catal. Commun. 2012, 21, 32–37. [Google Scholar] [CrossRef]
- Xue, L.M.; Zhang, F.H.; Fan, H.J.; Bai, X.F. Preparation of C Doped TiO2 Photocatalysts and their Photocatalytic Reduction of Carbon Dioxide. Adv. Mater. Res. 2011, 183–185, 1842–1846. [Google Scholar] [CrossRef]
- Gui, M.M.; Chai, S.-P.; Xu, B.-Q.; Mohamed, A.R. Enhanced visible light responsive MWCNT/TiO2 core–shell nanocomposites as the potential photocatalyst for reduction of CO2 into methane. Sol. Energy Mater. Sol. Cells 2014, 122, 183–189. [Google Scholar] [CrossRef]
- Xia, X.-H.; Jia, Z.-J.; Yu, Y.; Liang, Y.; Wang, Z.; Ma, L.-L. Preparation of multi-walled carbon nanotube supported TiO2 and its photocatalytic activity in the reduction of CO2 with H2O. Carbon 2007, 45, 717–721. [Google Scholar] [CrossRef]
- Ong, W.-J.; Gui, M.M.; Chai, S.-P.; Mohamed, A.R. Direct growth of carbon nanotubes on Ni/TiO2 as next generation catalysts for photoreduction of CO2 to methane by water under visible light irradiation. RSC Adv. 2013, 3, 4505–4509. [Google Scholar] [CrossRef]
- Ong, W.-J.; Tan, L.-L.; Chai, S.-P.; Yong, S.-T.; Mohamed, A.R. Self-assembly of nitrogen-doped TiO2 with exposed {001} facets on a graphene scaffold as photo-active hybrid nanostructures for reduction of carbon dioxide to methane. Nano Res. 2014, 7, 1528–1547. [Google Scholar] [CrossRef]
- Li, Y.; Wang, W.-N.; Zhan, Z.; Woo, M.-H.; Wu, C.-Y.; Biswas, P. Photocatalytic reduction of CO2 with H2O on mesoporous silica supported Cu/TiO2 catalysts. Appl. Catal. B 2010, 100, 386–392. [Google Scholar] [CrossRef]
- Sakthivel, S.; Shankar, M.V.; Palanichamy, M.; Arabindoo, B.; Bahnemann, D.W.; Murugesan, V. Enhancement of photocatalytic activity by metal deposition: Characterisation and photonic efficiency of Pt, Au and Pd deposited on TiO2 catalyst. Wat. Res. 2004, 38, 3001–3008. [Google Scholar] [CrossRef] [PubMed]
- Tostón, S.; Camarillo, R.; Martínez, F.; Jiménez, C.; Rincón, J. Supercritical synthesis of platinum-modified titanium dioxide for solar fuel production from carbon dioxide. Chin. J. Catal. 2017, 38, 636–650. [Google Scholar] [CrossRef]
- Khan, M.R.; Chuan, T.W.; Yousuf, A.; Chowdhury, M.; Cheng, C.K. Schottky barrier and surface plasmonic resonance phenomena towards the photocatalytic reaction: Study of their mechanisms to enhance photocatalytic activity. Catal. Sci. Technol. 2015, 5, 2522–2531. [Google Scholar] [CrossRef]
- Murakami, N.; Saruwatari, D.; Tsubota, T.; Ohno, T. Photocatalytic reduction of carbon dioxide over shape-controlled titanium (IV) oxide nanoparticles with co-catalyst loading. Curr. Org. Chem. 2013, 17, 2449–2453. [Google Scholar] [CrossRef]
- Zaleska, A. Doped-TiO2: A review. Recent Pat. Eng. 2008, 2, 157–164. [Google Scholar] [CrossRef]
- Linsebigler, A.L.; Lu, G.; Yates, J.T. Photocatalysis on TiO2 Surfaces: Principles, Mechanisms, and Selected Results. Chem. Rev. 1995, 95, 735–758. [Google Scholar] [CrossRef]
- Liu, E.; Qi, L.; Bian, J.; Chen, Y.; Hu, X.; Fan, J.; Liu, H.; Zhu, C.; Wang, Q. A facile strategy to fabricate plasmonic Cu modified TiO2 nano-flower films for photocatalytic reduction of CO2 to methanol. Mater. Res. Bull. 2015, 68, 203–209. [Google Scholar] [CrossRef]
- Kemp, T.J.; McIntyre, R.A. Transition metal-doped titanium (IV) dioxide: Characterisation and influence on photodegradation of poly (vinyl chloride). Polym. Degrad. Stab. 2006, 91, 165–194. [Google Scholar] [CrossRef]
- Feng, H.; Zhang, M.-H.; Yu, L.E. Hydrothermal synthesis and photocatalytic performance of metal-ions doped TiO2. Appl. Catal. A 2012, 413–414, 238–244. [Google Scholar] [CrossRef]
- Di Valentin, C.; Pacchioni, G. Trends in non-metal doping of anatase TiO2: B, C, N and F. Catal. Today 2013, 206, 12–18. [Google Scholar] [CrossRef]
- Marschall, R.; Wang, L. Non-metal doping of transition metal oxides for visible-light photocatalysis. Catal. Today 2014, 225, 111–135. [Google Scholar] [CrossRef]
- Sun, L.; Zhao, X.; Cheng, X.; Sun, H.; Li, Y.; Li, P.; Fan, W. Evaluating the C, N, and F pairwise codoping effect on the enhanced photoactivity of ZnWO4: The charge compensation mechanism in donor–acceptor pairs. J. Phys. Chem. C 2011, 115, 15516–15524. [Google Scholar] [CrossRef]
- Wang, W.; Serp, P.; Kalck, P.; Silva, C.G.; Faria, J.L. Preparation and characterization of nanostructured MWCNT-TiO2 composite materials for photocatalytic water treatment applications. Mater. Res. Bull. 2008, 43, 958–967. [Google Scholar] [CrossRef]
- Oh, W.-C.; Zhang, F.-J.; Chen, M.-L. Preparation of MWCNT/TiO2 composites by using MWCNTs and Titanium (IV) alkoxide precursors in Benzene and their photocatalytic effect and bactericidal activity. Bull. Korean Chem. Soc. 2009, 30, 2637–2642. [Google Scholar]
- Kuo, C.-Y. Prevenient dye-degradation mechanisms using UV/TiO2/carbon nanotubes process. J. Hazard. Mater. 2009, 163, 239–244. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Serp, P.; Kalck, P.; Faria, J.L. Photocatalytic degradation of phenol on MWNT and titania composite catalysts prepared by a modified sol–gel method. Appl. Catal. B 2005, 56, 305–312. [Google Scholar] [CrossRef]
- Hu, G.; Meng, X.; Feng, X.; Ding, Y.; Zhang, S.; Yang, M. Anatase TiO2 nanoparticles/carbon nanotubes nanofibers: Preparation, characterization and photocatalytic properties. Asian J. Mater. Sci. 2007, 42, 7162–7170. [Google Scholar] [CrossRef]
- Aryal, S.; Kim, C.K.; Kim, K.-W.; Khil, M.S.; Kim, H.Y. Multi-walled carbon nanotubes/TiO2 composite nanofiber by electrospinning. Mater. Sci. Eng. C 2008, 28, 75–79. [Google Scholar] [CrossRef]
- Yu, H.; Quan, X.; Chen, S.; Zhao, H. TiO2− multiwalled carbon nanotube heterojunction arrays and their charge separation capability. J. Phys. Chem. C 2007, 111, 12987–12991. [Google Scholar] [CrossRef]
- Chen, M.-L.; Zhang, F.-J.; Oh, W.-C. Synthesis, characterization, and photocatalytic analysis of CNT/TiO2 composites derived from MWCNTs and titanium sources. New Carbon Mater. 2009, 24, 159–166. [Google Scholar] [CrossRef]
- Tang, B.; Chen, H.; Peng, H.; Wang, Z.; Huang, W. Graphene modified TiO2 composite photocatalysts: Mechanism, progress and perspective. Nanomaterials 2018, 8, 105. [Google Scholar] [CrossRef]
- Xiang, Q.; Yu, J.; Jaroniec, M. Graphene-based semiconductor photocatalysts. Chem. Soc. Rev. 2012, 41, 782–796. [Google Scholar] [CrossRef] [PubMed]
- Chowdhury, S.; Balasubramanian, R. Graphene/semiconductor nanocomposites (GSNs) for heterogeneous photocatalytic decolorization of wastewaters contaminated with synthetic dyes: A review. Appl. Catal. B 2014, 160, 307–324. [Google Scholar] [CrossRef]
- Jianwei, C.; Jianwen, S.; Xu, W.; Haojie, C.; Minglai, F. Recent progress in the preparation and application of semiconductor/graphene composite photocatalysts. Chin. J. Catal. 2013, 34, 621–640. [Google Scholar]
- Singh, V.; Joung, D.; Zhai, L.; Das, S.; Khondaker, S.I.; Seal, S. Graphene based materials: Past, present and future. Prog. Mater Sci. 2011, 56, 1178–1271. [Google Scholar] [CrossRef]
- Hu, C.; Lu, T.; Chen, F.; Zhang, R. A brief review of graphene–metal oxide composites synthesis and applications in photocatalysis. J. Chin. Adv. Mater. Soc. 2013, 1, 21–39. [Google Scholar] [CrossRef]
- Ozer, L.Y.; Garlisi, C.; Oladipo, H.; Pagliaro, M.; Sharief, S.A.; Yusuf, A.; Almheiri, S.; Palmisano, G. Inorganic semiconductors-graphene composites in photo(electro)catalysis: Synthetic strategies, interaction mechanisms and applications. J. Photochem. Photobiol. C 2017, 33, 132–164. [Google Scholar] [CrossRef]
- Wang, L.-Q.; Wang, D.; Liu, J.; Exarhos, G.J. Probing Porosity and Pore Interconnectivity in Self-Assembled TiO2–Graphene Hybrid Nanostructures Using Hyperpolarized 129Xe NMR. J. Phys. Chem. C 2012, 116, 22–29. [Google Scholar] [CrossRef]
- Ying, J.; Yang, X.-Y.; Tian, G.; Janiak, C.; Su, B.-L. Self-assembly: An option to nanoporous metal nanocrystals. Nanoscale 2014, 6, 13370–13382. [Google Scholar] [CrossRef]
- Yu, X.; Lin, D.; Li, P.; Su, Z. Recent advances in the synthesis and energy applications of TiO2-graphene nanohybrids. Sol. Energy Mater. Sol. Cells 2017, 172, 252–269. [Google Scholar] [CrossRef]
- Kim, I.Y.; Lee, J.M.; Kim, T.W.; Kim, H.N.; Kim, H.I.; Choi, W.; Hwang, S.J. A Strong Electronic Coupling between Graphene Nanosheets and Layered Titanate Nanoplates: A Soft-Chemical Route to Highly Porous Nanocomposites with Improved Photocatalytic Activity. Small 2012, 8, 1038–1048. [Google Scholar] [CrossRef] [PubMed]
- Tu, W.; Zhou, Y.; Liu, Q.; Yan, S.; Bao, S.; Wang, X.; Xiao, M.; Zou, Z. An In Situ Simultaneous Reduction-Hydrolysis Technique for Fabrication of TiO2-Graphene 2D Sandwich-Like Hybrid Nanosheets: Graphene-Promoted Selectivity of Photocatalytic-Driven Hydrogenation and Coupling of CO2 into Methane and Ethane. Adv. Funct. Mater. 2013, 23, 1743–1749. [Google Scholar] [CrossRef]
- Tian, J.; Zhao, Z.; Kumar, A.; Boughton, R.I.; Liu, H. Recent progress in design, synthesis, and applications of one-dimensional TiO2 nanostructured surface heterostructures: A review. Chem. Soc. Rev. 2014, 43, 6920–6937. [Google Scholar] [CrossRef] [PubMed]
- Beigi, A.A.; Fatemi, S.; Salehi, Z. Synthesis of nanocomposite CdS/TiO2 and investigation of its photocatalytic activity for CO2 reduction to CO and CH4 under visible light irradiation. J. Co2 Util. 2014, 7, 23–29. [Google Scholar] [CrossRef]
- Li, X.; Liu, H.; Luo, D.; Li, J.; Huang, Y.; Li, H.; Fang, Y.; Xu, Y.; Zhu, L. Adsorption of CO2 on heterostructure CdS(Bi2S3)/TiO2 nanotube photocatalysts and their photocatalytic activities in the reduction of CO2 to methanol under visible light irradiation. Chem. Eng. J. 2012, 180, 151–158. [Google Scholar] [CrossRef]
- Wang, C.; Thompson, R.L.; Baltrus, J.; Matranga, C. Visible light photoreduction of CO2 using CdSe/Pt/TiO2 heterostructured catalysts. J. Phys. Chem. Lett. 2009, 1, 48–53. [Google Scholar] [CrossRef]
- Wang, C.; Thompson, R.L.; Ohodnicki, P.; Baltrus, J.; Matranga, C. Size-dependent photocatalytic reduction of CO2 with PbS quantum dot sensitized TiO2 heterostructured photocatalysts. J. Mater. Chem. 2011, 21, 13452–13457. [Google Scholar] [CrossRef]
- Asi, M.A.; He, C.; Su, M.; Xia, D.; Lin, L.; Deng, H.; Xiong, Y.; Qiu, R.; Li, X.-Z. Photocatalytic reduction of CO2 to hydrocarbons using AgBr/TiO2 nanocomposites under visible light. Catal. Today 2011, 175, 256–263. [Google Scholar] [CrossRef]
- Kumar, S.; Isaacs, M.A.; Trofimovaite, R.; Durndell, L.; Parlett, C.M.; Douthwaite, R.E.; Coulson, B.; Cockett, M.C.; Wilson, K.; Lee, A.F. P25@ CoAl layered double hydroxide heterojunction nanocomposites for CO2 photocatalytic reduction. Appl. Catal. B 2017, 209, 394–404. [Google Scholar] [CrossRef]
- Low, J.; Yu, J.; Jaroniec, M.; Wageh, S.; Al-Ghamdi, A.A. Heterojunction photocatalysts. Adv. Mater. 2017, 29, 1601694. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Zhou, Y.; Tu, W.; Ye, J.; Zou, Z. State-of-the-art progress in diverse heterostructured photocatalysts toward promoting photocatalytic performance. Adv. Funct. Mater. 2015, 25, 998–1013. [Google Scholar] [CrossRef]
- George, S.M. Atomic layer deposition: An overview. Chem. Rev. 2010, 110, 111–131. [Google Scholar] [CrossRef] [PubMed]
- Parsons, G.N.; George, S.M.; Knez, M. Progress and future directions for atomic layer deposition and ALD-based chemistry. MRS Bull. 2011, 36, 865–871. [Google Scholar] [CrossRef]
- Lin, Y.; Xu, Y.; Mayer, M.T.; Simpson, Z.I.; McMahon, G.; Zhou, S.; Wang, D. Growth of p-type hematite by atomic layer deposition and its utilization for improved solar water splitting. J. Am. Chem. Soc. 2012, 134, 5508–5511. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.; Luo, Z.; Li, C.; Gong, J. Controllable fabrication of nanostructured materials for photoelectrochemical water splitting via atomic layer deposition. Chem. Soc. Rev. 2014, 43, 7469–7484. [Google Scholar] [CrossRef]
- Son, D.H.; Hughes, S.M.; Yin, Y.; Paul Alivisatos, A. Cation exchange reactions in ionic nanocrystals. Sci. (New York N.Y.) 2004, 306, 1009–1012. [Google Scholar] [CrossRef]
- Rivest, J.B.; Jain, P.K. Cation exchange on the nanoscale: An emerging technique for new material synthesis, device fabrication, and chemical sensing. Chem. Soc. Rev. 2013, 42, 89–96. [Google Scholar] [CrossRef]
- Nguyen, T.-V.; Wu, J.C.S.; Chiou, C.-H. Photoreduction of CO2 over Ruthenium dye-sensitized TiO2-based catalysts under concentrated natural sunlight. Catal. Commun. 2008, 9, 2073–2076. [Google Scholar] [CrossRef]
- Srikanth, B.; Goutham, R.; Badri Narayan, R.; Ramprasath, A.; Gopinath, K.P.; Sankaranarayanan, A.R. Recent advancements in supporting materials for immobilised photocatalytic applications in waste water treatment. J. Environ. Manag. 2017, 200, 60–78. [Google Scholar] [CrossRef]
- Suárez, S. Immobilised Photocatalysts. In Design of Advanced Photocatalytic Materials for Energy and Environmental Applications; Coronado, J.M., Fresno, F., Hernández-Alonso, M.D., Portela, R., Eds.; Springer: London, UK, 2013; pp. 245–267. [Google Scholar] [CrossRef]
- Wu, L.; Yang, D.; Fei, L.; Huang, Y.; Wu, F.; Sun, Y.; Shi, J.; Xiang, Y. Dip-Coating Process Engineering and Performance Optimization for Three-State Electrochromic Devices. Nanoscale Res. Lett. 2017, 12, 390. [Google Scholar] [CrossRef] [PubMed]
- Kyesmen, P.I.; Nombona, N.; Diale, M. Influence of coating techniques on the optical and structural properties of hematite thin films. Surf. Interfaces 2019, 17, 100384. [Google Scholar] [CrossRef]
- Deepa, M.; Saxena, T.K.; Singh, D.P.; Sood, K.N.; Agnihotry, S.A. Spin coated versus dip coated electrochromic tungsten oxide films: Structure, morphology, optical and electrochemical properties. Electrochim. Acta 2006, 51, 1974–1989. [Google Scholar] [CrossRef]
- Matsushima, T.; Inoue, M.; Fujihara, T.; Terakawa, S.; Qin, C.; Sandanayaka, A.S.D.; Adachi, C. High-coverage organic-inorganic perovskite film fabricated by double spin coating for improved solar power conversion and amplified spontaneous emission. Chem. Phys. Lett. 2016, 661, 131–135. [Google Scholar] [CrossRef]
- Pitoniak, E.; Wu, C.-Y.; Londeree, D.; Mazyck, D.; Bonzongo, J.-C.; Powers, K.; Sigmund, W. Nanostructured silica-gel doped with TiO2 for mercury vapor control. J. Nanopart. Res. 2003, 5, 281–292. [Google Scholar] [CrossRef]

| Modification Strategy | Advantages | Disadvantages |
|---|---|---|
| Metal Deposition | -Enhances electron–hole separation | -Expensive -Rare |
| Metal Doping | -Extends light absorption into visible range -Enhances electron–hole separation | -Act as recombination centers -Leads to structural defects -Possibility of metal leaching |
| Non-Metal Doping | -Extends light absorption into visible range -Enhances electron–hole separation | -Act as recombination centers |
| Carbon-based Material Loading | -Extends light absorption into visible range -Enhances electron–hole separation -Increases electron concentration on TiO2 surface -Improves CO2 adsorption on catalytic surface -Reduces agglomeration of TiO2 nanoparticles | -Forbids light absorption by TiO2 |
| Heterostructures | -Extends light absorption into visible range -Enhances electron–hole separation -Separates reduction and oxidation sites | -Complex synthesis method -Instability |
| Dispersion on Supports | -Enhances the catalyst’s product selectivity, pore structure and electronic properties -Eliminates need for post treatment separation -Provides high surface area -Enhances dispersion of TiO2 nanoparticles -Improves adsorption of reactants on catalytic surface | -Low light utilization efficiency |
| Alkali Modification | -Enhances electron–hole separation -Enhances the chemisorption of CO2 | -Encapsulation of TiO2 nanoparticles |
| Modification Strategy | Photocatalyst | Synthesis Method | Reductant | Light Source | Main Product Yield (μmol/g-cat/h) | Ref. |
|---|---|---|---|---|---|---|
| Metal Deposition Platinum | One-dimensional TiO2 single crystals coated with ultrafine Pt NPs | CVD | Water | UV | Methane: 1361 | [91] |
| 0.2 wt.% Pt on mesoporous TiO2 | Soft Template | Water | UV | Methane: 2.85 | [36] | |
| Metal Deposition Gold | 0.5 wt.% Au on TiO2 NWs | HT | Hydrogen | Visible | Carbon monoxide: 1237 Methanol: 12.65 | [92] |
| Metal Deposition Silver | Ag-electrodeposited on TiO2 NRs | HT | Water | UV | Methane: 2.64 | [93] |
| Metal Deposition Copper | 1.7 wt.% Cu and 0.9 wt.% Pt NPs on TiO2 (Evonik P-25) | - | Water | UV + Visible | Methane: 33 | [94] |
| Cu/C–TiO2 | Sol–gel | Water | UV | Methane: 2.526 | [95] | |
| Metal Doping Copper | 1.2 wt.% Cu-TiO2 | HT | Water | UV | Methanol: 0.45 | [96] |
| 2 wt.% Cu-TiO2 | Sol–gel | NaOH | UV | Methanol: 12.5 | [58] | |
| 3 wt.% Cu-TiO2 (Evonik P-25) | - | KHCO3 | UV | Methanol: 194 | [97] | |
| Metal Doping Nickel | 0.1 mol % Ni-TiO2 | ST | Water | UV | Methane: 14 | [98] |
| Metal Doping Cerium | 0.28 mol % Ce-TiO2 | Sol–gel | NaOH | UV | Methane: 0.889 | [99] |
| Non-Metal Doping Nitrogen | N-TiO2 NTs | HT | NaOH | Visible | Formic acid: 1039 Methanol: 94.4 Formaldehyde: 76.8 | [100] |
| Non-Metal Doping Carbon | C–TiO2 | IMM | Na2SO3 | Solar | Formic acid: 439 | [101] |
| Carbon-based Material Loading CNTs | MWCNT/TiO2 nanocomposite | - | Water | Visible | Methane: 0.17 | [102] |
| Anatase-TiO2 NPs/MWCNT | Sol–gel | Water | UV | Ethanol: 29.872 | [103] | |
| Rutile-TiO2 NRs/MWCNT | HT | Water | UV | Formic acid: 25.02 | [103] | |
| CNT-Ni/TiO2 nanocomposites | CVD | Water | Visible | Methane: 0.145 | [104] | |
| Carbon-based Material Loading Graphene | Graphene/ N-TiO2 | ST | Water | Visible | Methane: 0.37 | [105] |
| Hetero-structures | 1 wt.% CuO-TiO2 composite | - | Methanol | UV | Methyl formate: 1602 | [38] |
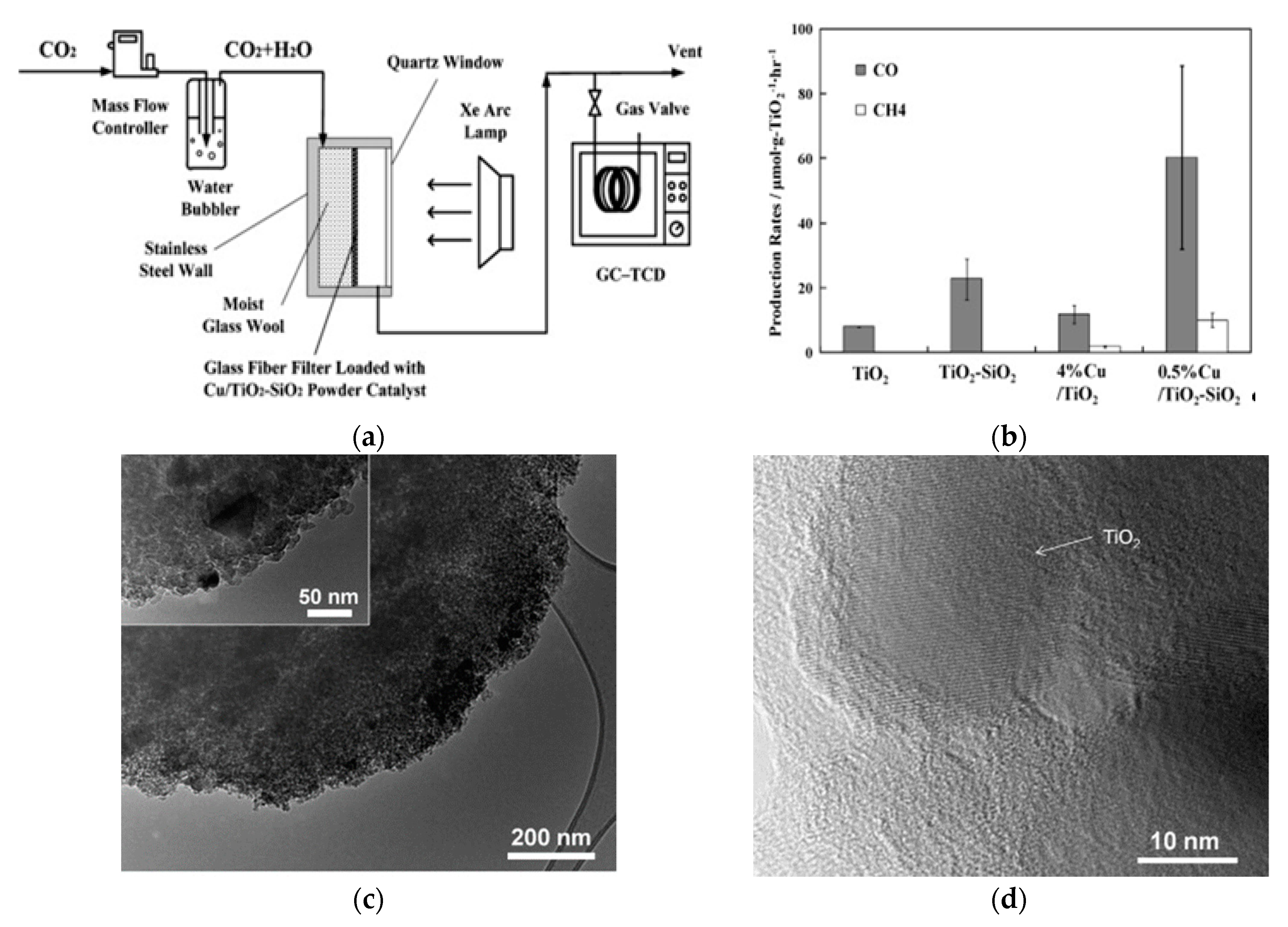
| Dispersion on Supports | 0.5 wt.% Cu/TiO2-silica nanocomposite | Sol–gel | Water | UV | Carbon monoxide: 60 Methane: 10 | [106] |
| Alkali Modification | 3 wt.% NaOH-TiO2 | IMP | Water | UV | Methane: 8.667 | [90] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Al Jitan, S.; Palmisano, G.; Garlisi, C. Synthesis and Surface Modification of TiO2-Based Photocatalysts for the Conversion of CO2. Catalysts 2020, 10, 227. https://doi.org/10.3390/catal10020227
Al Jitan S, Palmisano G, Garlisi C. Synthesis and Surface Modification of TiO2-Based Photocatalysts for the Conversion of CO2. Catalysts. 2020; 10(2):227. https://doi.org/10.3390/catal10020227
Chicago/Turabian StyleAl Jitan, Samar, Giovanni Palmisano, and Corrado Garlisi. 2020. "Synthesis and Surface Modification of TiO2-Based Photocatalysts for the Conversion of CO2" Catalysts 10, no. 2: 227. https://doi.org/10.3390/catal10020227
APA StyleAl Jitan, S., Palmisano, G., & Garlisi, C. (2020). Synthesis and Surface Modification of TiO2-Based Photocatalysts for the Conversion of CO2. Catalysts, 10(2), 227. https://doi.org/10.3390/catal10020227





