An Accurate Growth Mechanism and Photocatalytic Degradation Rhodamine B of Crystalline Nb2O5 Nanotube Arrays
Abstract
1. Introduction
2. Results and Discussion
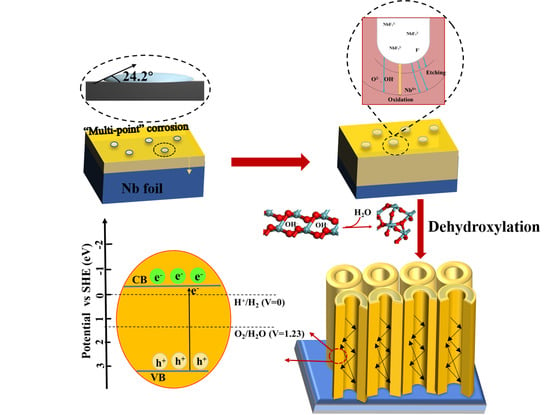
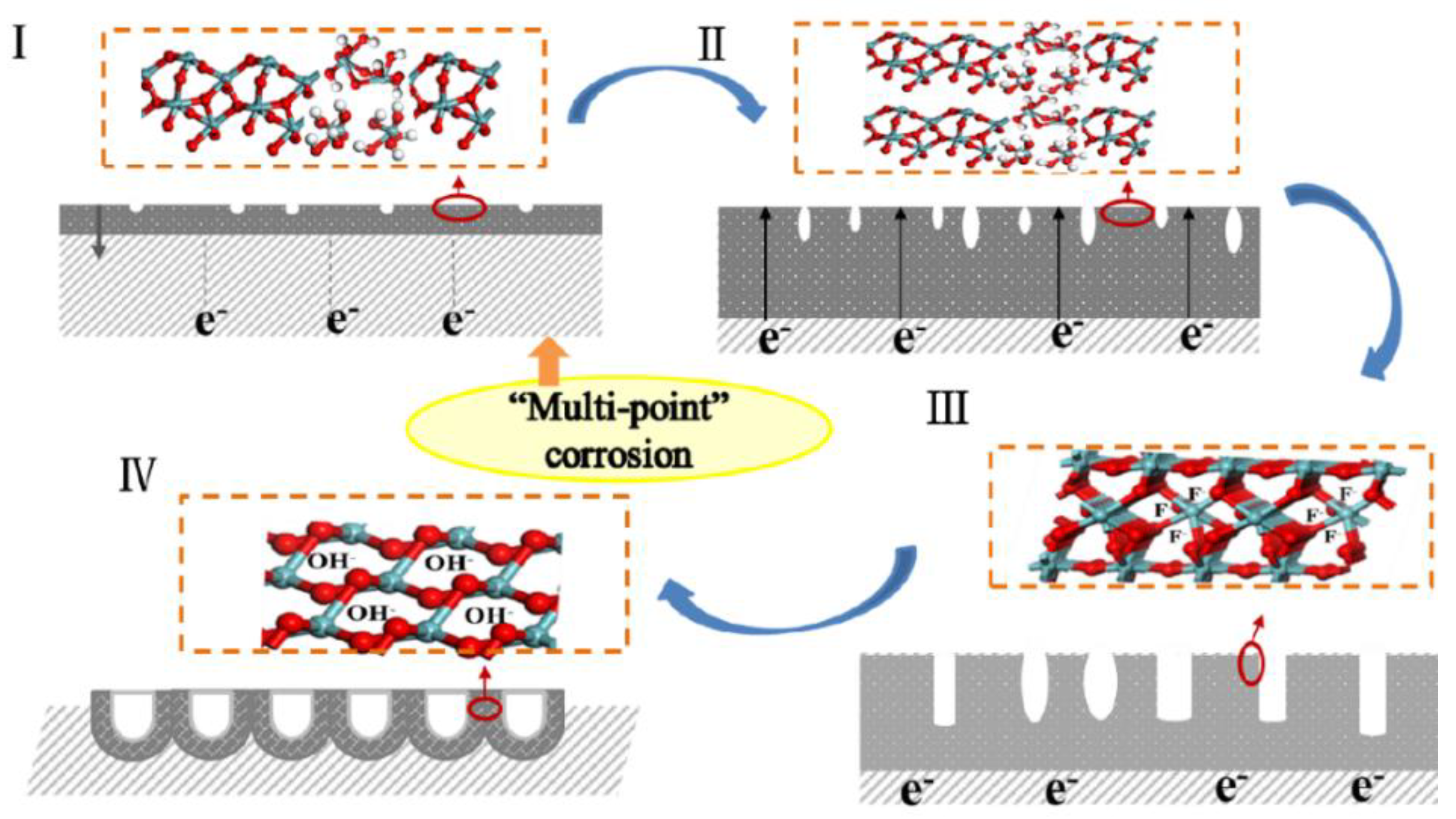
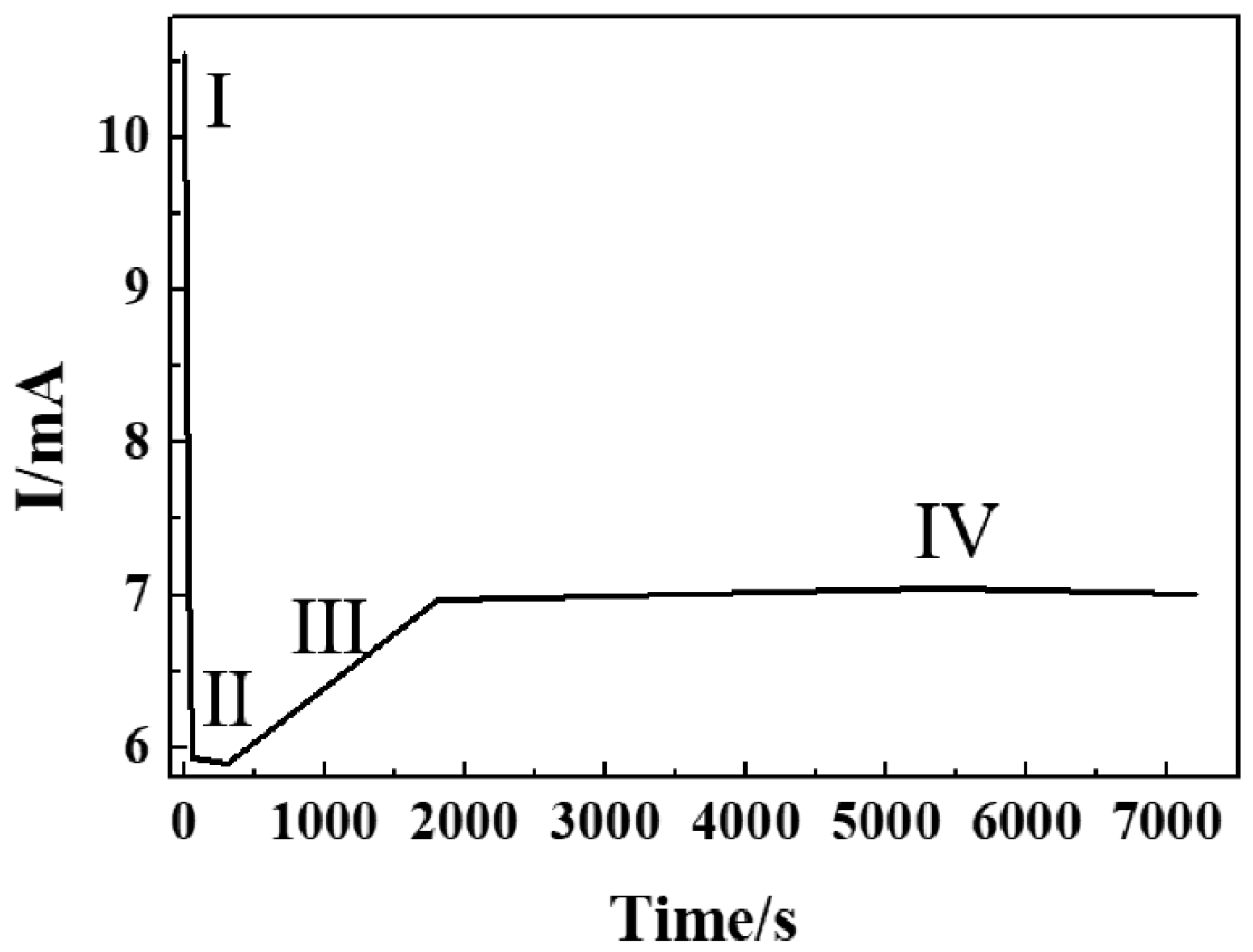
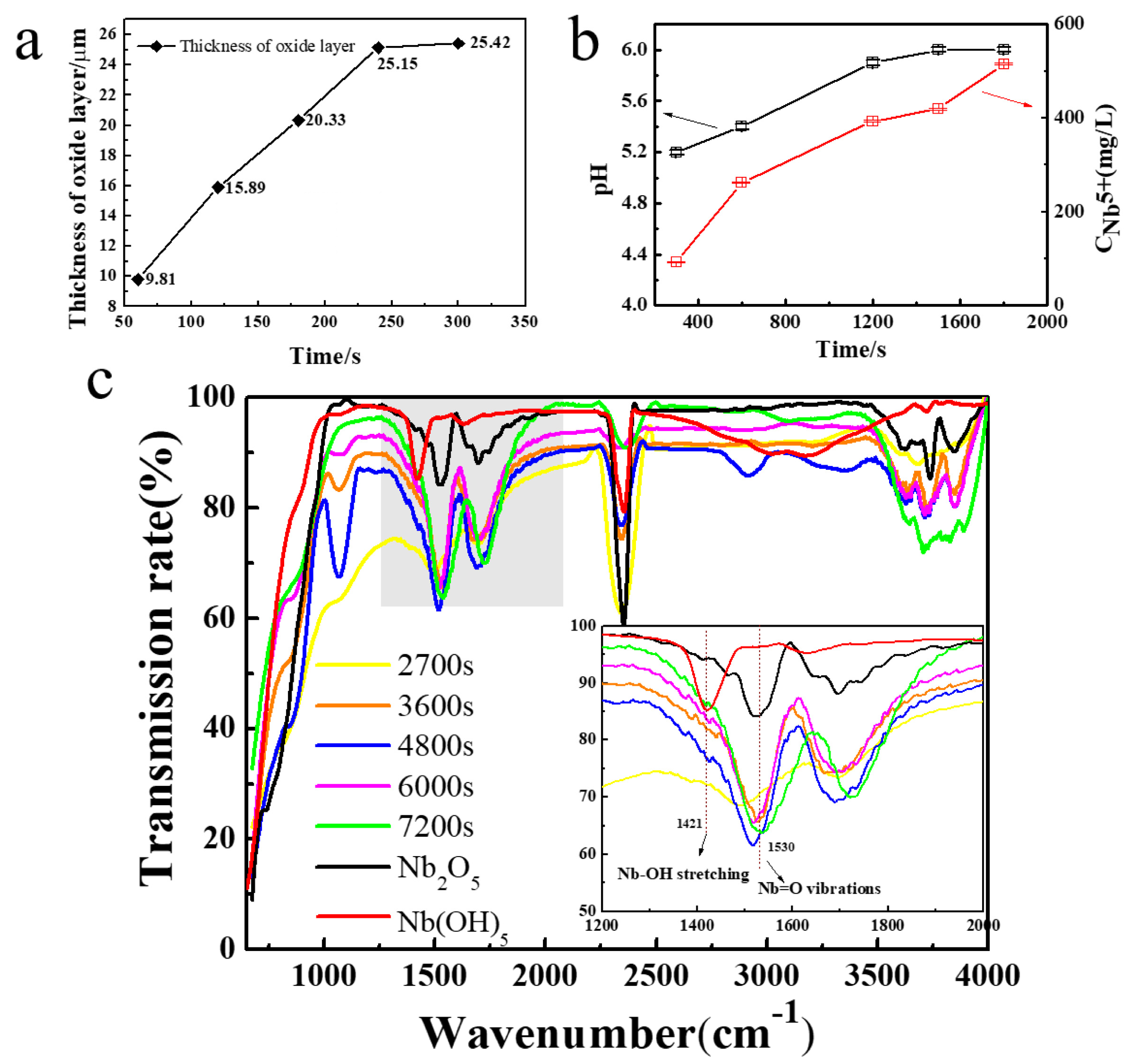
2.1. Growth Mechanism
2.2. Effect of Essential Ions on Formation of Nb2O5 NAs
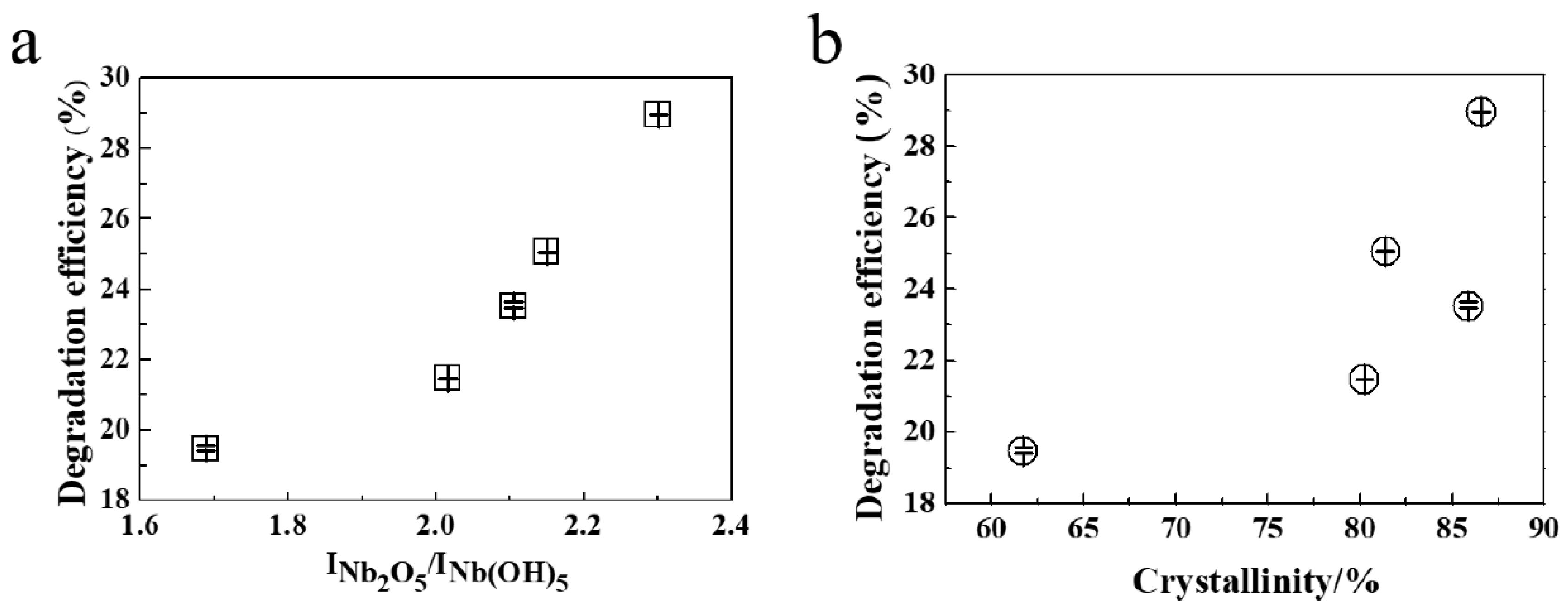
2.3. Catalytic Performance and Mechanism
3. Materials and Methods
3.1. Synthesis of Nb2O5 Nanotube Arrays
3.2. Characterization of Catalysts
3.3. Photocatalysis Activity Test
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Zhu, Y.; Chen, Z.; Gao, T.; Huang, Q.; Niu, F.; Qin, L.; Tang, P.; Huang, Y.; Sha, Z.; Wang, Y. Construction of hybrid Z-scheme Pt/CdS–TNTAs with enhanced visible-light photocatalytic performance. Appl. Catal. B Environ. 2015, 163, 16–22. [Google Scholar] [CrossRef]
- Yang, J.; Hao, J.; Xu, S.; Dai, J.; Wang, Y.; Pang, X. Visible-light-driven photocatalytic degradation of 4-CP and the synergistic reduction of Cr(VI) on one-pot synthesized amorphous Nb2O5 nanorods/graphene heterostructured composites. Chem. Eng. J. 2018, 353, 100–114. [Google Scholar] [CrossRef]
- Zhu, Y.; Wang, Y.; Chen, Z.; Qin, L.; Yang, L.; Zhu, L.; Tang, P.; Gao, T.; Huang, Y.; Sha, Z.; et al. Visible light induced photocatalysis on CdS quantum dots decorated TiO2 nanotube arrays. Appl. Catal. A Gen. 2015, 498, 159–166. [Google Scholar] [CrossRef]
- Lin, H.; Long, X.; Yang, S.; Zhou, D.; Yang, S. Three-Dimensional Decoupling Co-Catalyst from a Photoabsorbing Semiconductor as a New Strategy To Boost Photoelectrochemical Water Splitting. Nano Lett. 2018, 19, 455–460. [Google Scholar] [CrossRef] [PubMed]
- Ghicov, A.; Schmuki, P. Self-ordering electrochemistry: A review on growth and functionality of TiO2 nanotubes and other self-aligned MOx structures. Chem. Commun. 2009, 20, 2791–2808. [Google Scholar] [CrossRef]
- Zhang, N.; Quan, Q.; Qi, M.-Y.; Tang, Z.-R.; Xu, Y.-J. Hierarchically tailorable double-array film hybrids with enhanced photocatalytic and photoelectrochemical performances. Appl. Catal. B Environ. 2019, 259, 118086. [Google Scholar] [CrossRef]
- Furukawa, S.; Ohno, Y.; Shishido, T.; Teramura, K.; Tanaka, T. Selective Amine Oxidation Using Nb2O5 Photocatalyst and O2. ACS Catal. 2011, 1, 1150–1153. [Google Scholar] [CrossRef]
- Rao, W.-F.; Zhang, Y.; Wang, Z.; Yu, Q.; Yang, Y. Electric switching process of aligned Nb2O5 nanorods arrays. Mater. Lett. 2018, 211, 168–170. [Google Scholar] [CrossRef]
- Maček, M.; Orel, B. Electrochromism of sol–gel derived niobium oxide films. Sol. Energy Mater. Sol. Cells 1998, 54, 121–130. [Google Scholar] [CrossRef][Green Version]
- Mozalev, A.; Sakairi, M.; Saeki, I.; Takahashi, H. Nucleation and growth of the nanostructured anodic oxides on tantalum and niobium under the porous alumina film. Electrochim. Acta 2003, 48, 3155–3170. [Google Scholar] [CrossRef]
- Aagard, R.L. Optical waveguide characteristics of reactive dc-sputtered niobium pentoxide films. Appl. Phys. Lett. 1975, 27, 605–607. [Google Scholar] [CrossRef]
- Karlinsey, R.L. Preparation of self-organized niobium oxide microstructures via potentiostatic anodization. Electrochem. Commun. 2005, 7, 1190–1194. [Google Scholar] [CrossRef]
- Yang, M.; Zhao, X.; Zheng, S.; Liu, X.; Jin, B.; Li, H.; Gan, Y. A new electrochemical platform for ultrasensitive detection of atrazine based on modified self-ordered Nb2O5 nanotube arrays. J. Electroanal. Chem. 2017, 791, 17–22. [Google Scholar] [CrossRef]
- Liu, X.; Zheng, R.; Yuan, R.; Peng, L.; Liu, Y.; Lin, J. Released Defective Nb2O5with Optimized Solar Photocatalytic Activity. ECS J. Solid State Sci. Technol. 2017, 6, P665–P670. [Google Scholar] [CrossRef]
- Wen, H.; Liu, Z.; Wang, J.; Yang, Q.; Li, Y.; Yu, J. Facile synthesis of Nb2O5 nanorod array films and their electrochemical properties. Appl. Surf. Sci. 2011, 257, 10084–10088. [Google Scholar] [CrossRef]
- Shi, C.; Xiang, K.; Zhu, Y.; Zhou, W.; Chen, X.; Chen, H. Box-implanted Nb2O5 nanorods as superior anode materials in lithium ion batteries. Ceram. Int. 2017, 43, 12388–12395. [Google Scholar] [CrossRef]
- Lucas-Granados, B.; Sánchez-Tovar, R.; Fernández-Domene, R.M.; Estívalis-Martínez, J.M.; García-Antón, J. How does anodization time affect morphological and photocatalytic properties of iron oxide nanostructures? J. Mater. Sci. Technol. 2020, 38, 159–169. [Google Scholar] [CrossRef]
- Guan, D.; Wang, Y. Synthesis and growth mechanism of multilayer TiO2 nanotube arrays. Nanoscale 2012, 4, 2968–2977. [Google Scholar] [CrossRef]
- Guan, D.; Hymel, P.J.; Wang, Y. Growth mechanism and morphology control of double-layer and bamboo-type TiO2 nanotube arrays by anodic oxidation. Electrochim. Acta 2012, 83, 420–429. [Google Scholar] [CrossRef]
- Kumaravel, V.; Mathew, S.; Bartlett, J.; Pillai, S.C. Photocatalytic hydrogen production using metal doped TiO2: A review of recent advances. Appl. Catal. B Environ. 2019, 244, 1021–1064. [Google Scholar] [CrossRef]
- Xu, X.; Fang, X.; Zhai, T.; Zeng, H.; Liu, B.; Hu, X.; Bando, Y.; Golberg, D.V. Tube-in-Tube TiO2 Nanotubes with Porous Walls: Fabrication, Formation Mechanism, and Photocatalytic Properties. Small 2010, 7, 445–449. [Google Scholar] [CrossRef] [PubMed]
- Wei, W.; Macak, J.M.; Schmuki, P. High aspect ratio ordered nanoporous Ta2O5 films by anodization of Ta. Electrochem. Commun. 2008, 10, 428–432. [Google Scholar] [CrossRef]
- Guo, L.; Zhao, J.; Wang, X.; Xu, R.; Li, Y. Synthesis and growth mechanism of zirconia nanotubes by anodization in electrolyte containing Cl−. J. Solid State Electrochem. 2008, 13, 1321–1326. [Google Scholar] [CrossRef]
- O’Sullivan, J.P.; Wood, G.C. The morphology and mechanism of formation of porous anodic films on aluminium. Proc. R. Soc. London. Ser. A. Math. Phys. Sci. 1970, 317, 511–543. [Google Scholar] [CrossRef]
- Macak, J.M.; Tsuchiya, H.; Ghicov, A.; Yasuda, K.; Hahn, R.; Bauer, S.; Schmuki, P. TiO2 nanotubes: Self-organized electrochemical formation, properties and applications. Curr. Opin. Solid State Mat. Sci. 2007, 11, 3–18. [Google Scholar] [CrossRef]
- Su, Z.; Zhou, W.; Jiang, F.; Hong, M. Anodic formation of nanoporous and nanotubular metal oxides. J. Mater. Chem. 2012, 22, 535–544. [Google Scholar] [CrossRef]
- Garcia-Vergara, S.; Skeldon, P.; Thompson, G.; Habazaki, H. A flow model of porous anodic film growth on aluminium. Electrochim. Acta 2006, 52, 681–687. [Google Scholar] [CrossRef]
- Garcia-Vergara, S.; Iglesias-Rubianes, L.; Blanco-Pinzon, C.; Skeldon, P.; Thompson, G.; Campestrini, P. Mechanical instability and pore generation in anodic alumina. Proc. R. Soc. A Math. Phys. Eng. Sci. 2006, 462, 2345–2358. [Google Scholar] [CrossRef]
- Garcia-Vergara, S.; Habazaki, H.; Skeldon, P.; Thompson, G. Tracer studies relating to alloying element behaviour in porous anodic alumina formed in phosphoric acid. Electrochim. Acta 2010, 55, 3175–3184. [Google Scholar] [CrossRef]
- Lee, W.; Schwirn, K.; Steinhart, M.; Pippel, E.; Scholz, R.; Gösele, U. Structural engineering of nanoporous anodic aluminium oxide by pulse anodization of aluminium. Nat. Nanotechnol. 2008, 3, 234–239. [Google Scholar] [CrossRef]
- Li, G.-Z.; Tang, H.-P.; Zhang, W.-Y.; Li, G.; Yu, L.-L.; Li, Y.-N. Fabrication of multilayer Nb2O5 nanoporous film by anodization of niobium foils. Rare Met. 2013, 34, 77–80. [Google Scholar] [CrossRef]
- Zhao, J.; Wang, X.; Xu, R.; Mi, Y.; Li, Y. Preparation and Growth Mechanism of Niobium Oxide Microcones by the Anodization Method. Electrochem. Solid State Lett. 2007, 10, C31–C33. [Google Scholar] [CrossRef]
- Wei, M.; Qi, Z.-M.; Ichihara, M.; Zhou, H. Synthesis of single-crystal niobium pentoxide nanobelts. Acta Mater. 2008, 56, 2488–2494. [Google Scholar] [CrossRef]
- Sieber, I.; Hildebrand, H.; Friedrich, A.; Schmuki, P. Formation of self-organized niobium porous oxide on niobium. Electrochem. Commun. 2005, 7, 97–100. [Google Scholar] [CrossRef]
- Upadhyay, K.K.; Cha, G.; Hildebrand, H.; Schmuki, P.; Silva, T.M.; Montemor, M.F.; Altomare, M. Capacitance response in an aqueous electrolyte of Nb2O5 nanochannel layers anodically grown in pure molten σ-H3PO4. Electrochim. Acta 2018, 281, 725–737. [Google Scholar] [CrossRef]
- Nakajima, K.; Baba, Y.; Noma, R.; Kitano, M.; Kondo, J.N.; Hayashi, S.; Hara, M. Nb2O5·nH2O as a Heterogeneous Catalyst with Water-Tolerant Lewis Acid Sites. J. Am. Chem. Soc. 2011, 133, 4224–4227. [Google Scholar] [CrossRef]
- Jin, X.; Liu, C.; Xu, J.; Wang, Q.; Chen, D. Size-controlled synthesis of mesoporous Nb2O5 microspheres for dye sensitized solar cells. RSC Adv. 2014, 4, 35546–35553. [Google Scholar] [CrossRef]
- Amaravathy, P.; Sowndarya, S.; Sathyanarayanan, S.; Rajendran, N. Novel sol gel coating of Nb2O5 on magnesium alloy for biomedical applications. Surf. Coatings Technol. 2014, 244, 131–141. [Google Scholar] [CrossRef]
- Tsuchiya, H.; Macak, J.M.; Sieber, I.; Schmuki, P. Self-Organized High-Aspect-Ratio Nanoporous Zirconium Oxides Prepared by Electrochemical Anodization. Small 2005, 1, 722–725. [Google Scholar] [CrossRef]
- Karlinsey, R.L. Self-assembled Nb2O5 microcones with tailored crystallinity. J. Mater. Sci. 2006, 41, 5017–5020. [Google Scholar] [CrossRef]
- Preethi, L.K.; Antony, R.P.; Mathews, T.; Walczak, L.; Gopinath, C.S. A Study on Doped Heterojunctions in TiO2 Nanotubes: An Efficient Photocatalyst for Solar Water Splitting. Sci. Rep. 2017, 7, 14314. [Google Scholar] [CrossRef] [PubMed]
- Paulose, M.; Shankar, K.; Yoriya, S.; Prakasam, H.E.; Varghese, O.K.; Mor, G.K.; LaTempa, T.A.; Fitzgerald, A.; Grimes, C.A. Anodic Growth of Highly Ordered TiO2 Nanotube Arrays to 134 μm in Length. J. Phys. Chem. B 2006, 110, 16179–16184. [Google Scholar] [CrossRef] [PubMed]
- Galstyan, V.; Comini, E.; Faglia, G.; Sberveglieri, G. Synthesis of self-ordered and well-aligned Nb2O5 nanotubes. CrystEngComm 2014, 16, 10273–10279. [Google Scholar] [CrossRef]
- Wei, W.; Lee, K.; Shaw, S.; Schmuki, P. Anodic formation of high aspect ratio, self-ordered Nb2O5 nanotubes. Chem. Commun. 2012, 48, 4244–4246. [Google Scholar] [CrossRef] [PubMed]

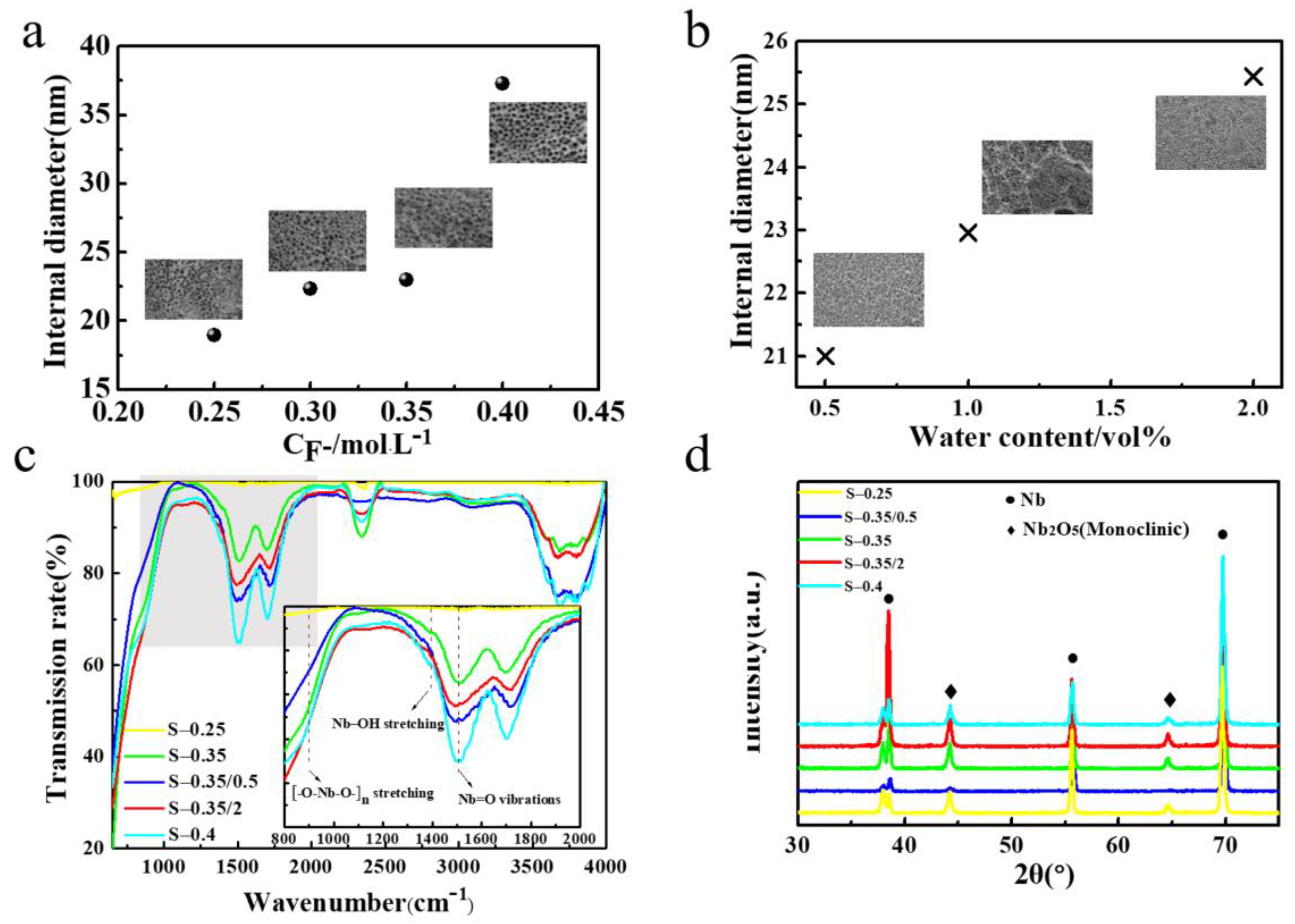


| Intensity | INb(OH)5 | INb2O5 | INb2O5/INb(OH)5 | |
|---|---|---|---|---|
| Time (s) | ||||
| 2700 | 29.14 | 32.88 | 1.13 | |
| 3600 | 19.71 | 32.68 | 1.66 | |
| 4800 | 22.89 | 38.38 | 1.68 | |
| 6000 | 18.30 | 34.52 | 1.89 | |
| 7200 | 18.09 | 42.54 | 2.35 | |
| No. | Internal Diameter (nm) | INb2O5/INb(OH)5 | Crystallinity (%) |
|---|---|---|---|
| S-0.25 | 18.96 | 2.15 | 81.39 |
| S-0.35/0.5 | 21.00 | 2.02 | 80.24 |
| S-0.35 | 23.00 | 2.30 | 86.59 |
| S-0.35/2 | 25.44 | 1.69 | 61.72 |
| S-0.4 | 37.31 | 2.11 | 85.88 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Guo, W.; Yang, L.; Lu, J.; Gao, P.; Li, W.; Feng, Z. An Accurate Growth Mechanism and Photocatalytic Degradation Rhodamine B of Crystalline Nb2O5 Nanotube Arrays. Catalysts 2020, 10, 1480. https://doi.org/10.3390/catal10121480
Guo W, Yang L, Lu J, Gao P, Li W, Feng Z. An Accurate Growth Mechanism and Photocatalytic Degradation Rhodamine B of Crystalline Nb2O5 Nanotube Arrays. Catalysts. 2020; 10(12):1480. https://doi.org/10.3390/catal10121480
Chicago/Turabian StyleGuo, Wei, Libin Yang, Jinghao Lu, Peng Gao, Wenjing Li, and Zhiying Feng. 2020. "An Accurate Growth Mechanism and Photocatalytic Degradation Rhodamine B of Crystalline Nb2O5 Nanotube Arrays" Catalysts 10, no. 12: 1480. https://doi.org/10.3390/catal10121480
APA StyleGuo, W., Yang, L., Lu, J., Gao, P., Li, W., & Feng, Z. (2020). An Accurate Growth Mechanism and Photocatalytic Degradation Rhodamine B of Crystalline Nb2O5 Nanotube Arrays. Catalysts, 10(12), 1480. https://doi.org/10.3390/catal10121480