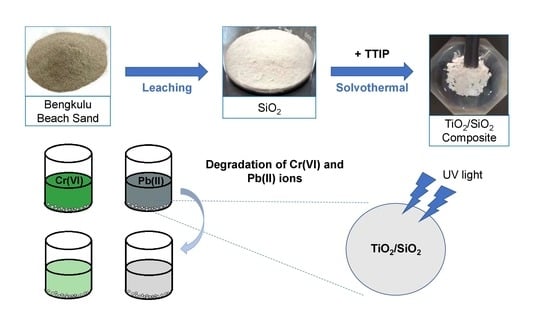
Synthesis of Titanium Dioxide/Silicon Dioxide from Beach Sand as Photocatalyst for Cr and Pb Remediation
Abstract
1. Introduction
2. Results and Discussion
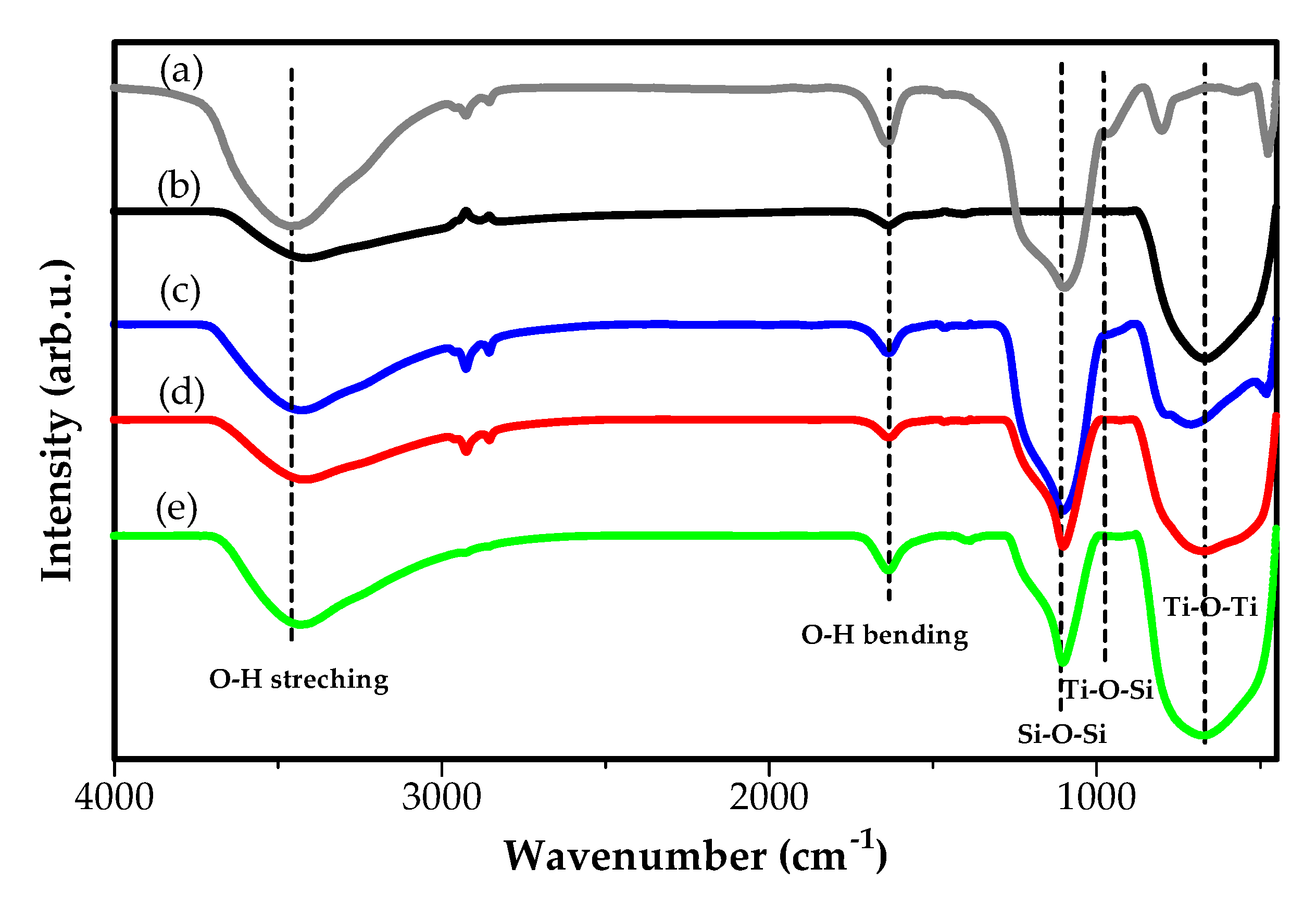
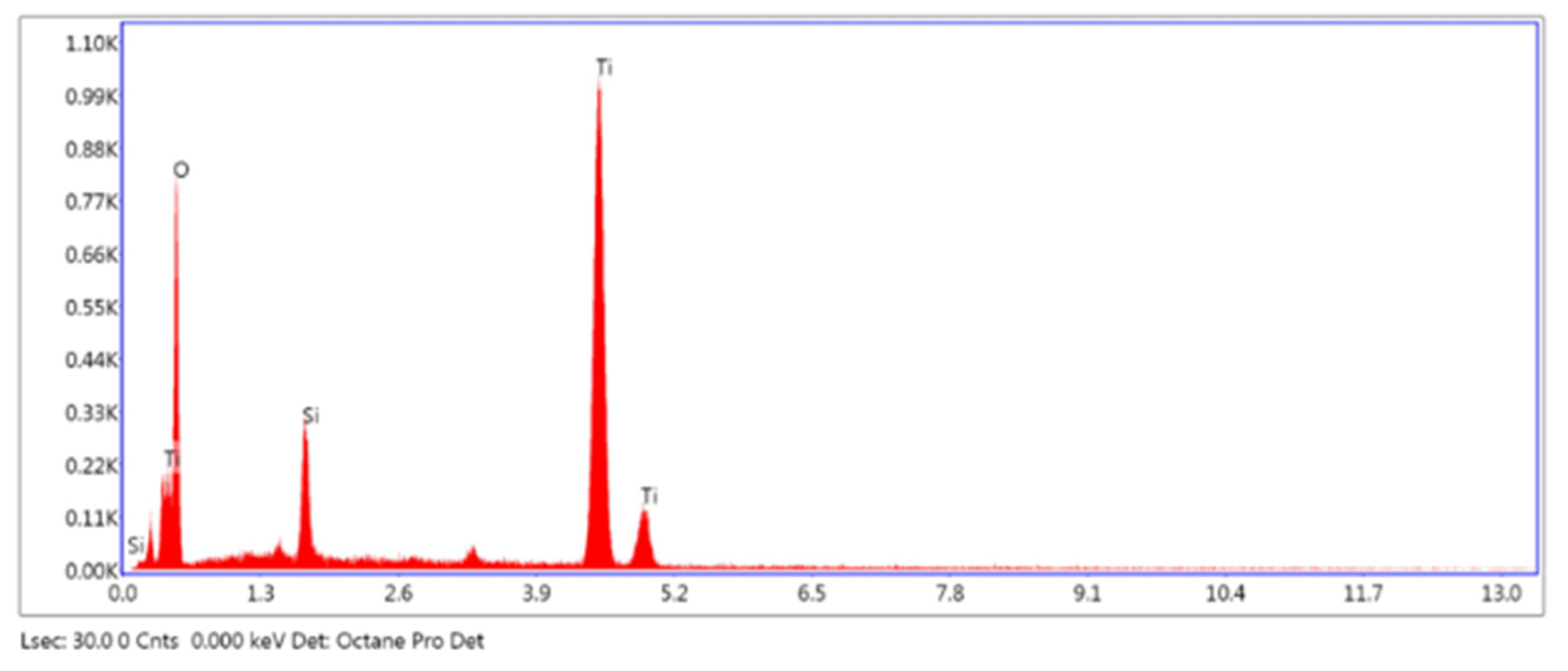
2.1. Characterization of Catalyst
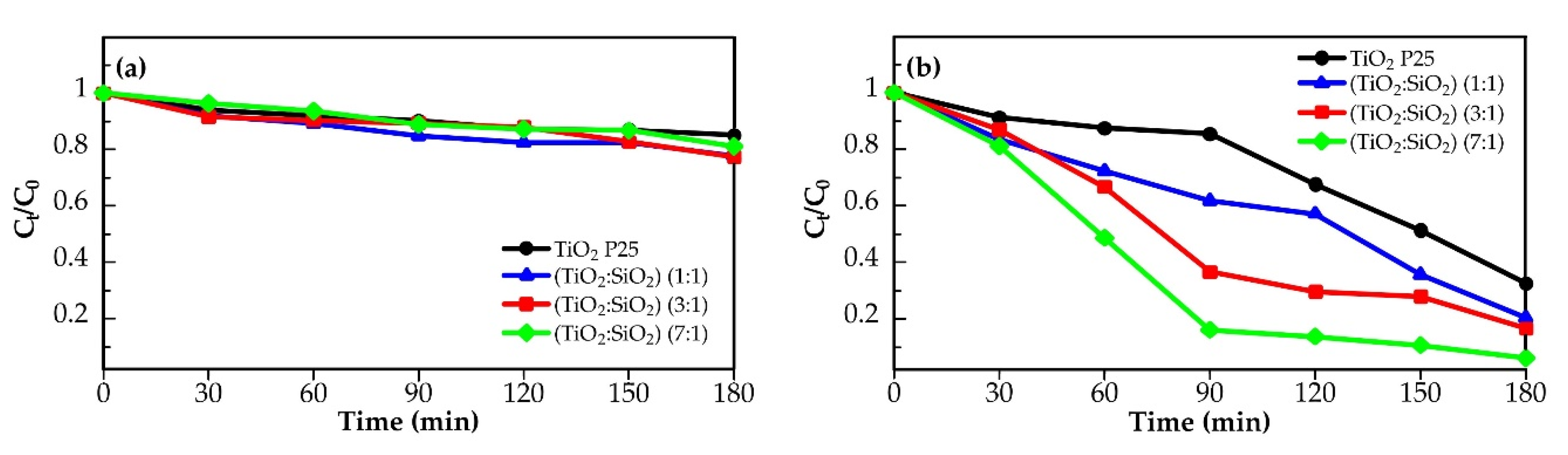
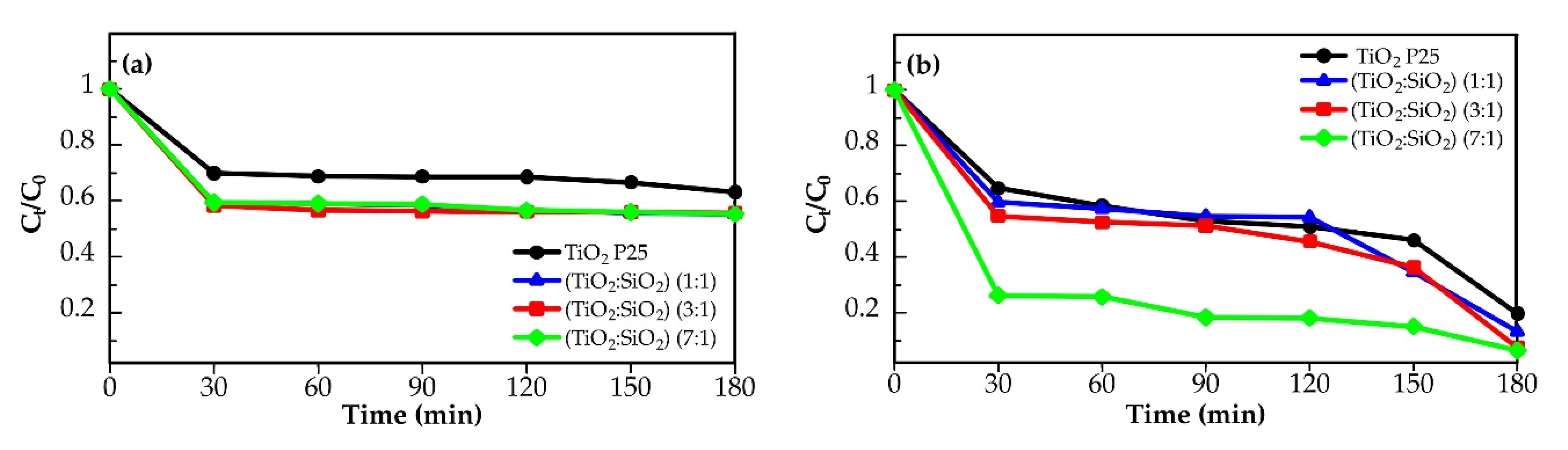
2.2. Photocatalytic Activity
3. Materials and Methods
3.1. Materials
3.2. Extraction of Silica
3.3. The Synthesis of TiO2/SiO2 Composite
3.4. Characterization of Catalysis
3.5. Photocatalytic Activity Test
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Sane, P.; Chaudhari, S.; Nemade, P.; Sontakke, S. Photocatalytic reduction of chromium(VI) using combustion synthesized TiO2. J. Environ. Chem. Eng. 2018, 6, 68–73. [Google Scholar] [CrossRef]
- Ojemaye, M.O.; Okoh, O.O.; Okoh, A.I. Performance of NiFe2O4-SiO2-TiO2 Magnetic Photocatalyst for the Effective Photocatalytic Reduction of Cr(VI) in Aqueous Solutions. J. Nanomater. 2017, 2017, 5264910. [Google Scholar] [CrossRef]
- Kotz, J.C.; Treichel, P.M.; Townsend, J.R. Chemistry and Chemical Reactivity, 7th ed.; Thomson Brooks/Cole: Pacific Grove, CA, USA, 2009. [Google Scholar]
- Said, N.I. Pengolahan Air Limbah Domestik di DKI Jakarta “Tinjauan Permasalahan, Strategi dan Teknologi Pengolahan”, 1st ed.; Badan Pengkajian dan Penerapan Teknologi: Jakarta, Indonesia, 2008; 606p.
- Eddy, D.R.; Rahayu, I.; Hartati, Y.W.; Firdaus, M.L.; Bakti, H.H. Photocatalytic activity of gadolinium doped TiO2 particles for decreasing heavy metal chromium concentration. J. Phys. Conf. Ser. 2018, 1080, 012013. [Google Scholar] [CrossRef]
- Bi, J.; Wang, J.; Huang, X.; Tao, Q.; Chen, M.; Wang, T.; Hao, H. Enhanced removal of Pb (II) and organics by titanate in a designed simultaneous process. Sep. Purif. Technol. 2020, 251, 117339. [Google Scholar] [CrossRef]
- Besançon, M.; Michelin, L.; Josien, L.; Vidal, L.; Assaker, K.; Bonne, M.; Lebeau, B.; Blin, J.L. Influence of the porous texture of SBA-15 mesoporous silica on the anatase formation in TiO2-SiO2 nanocomposites. New J. Chem. 2016, 40, 4386–4397. [Google Scholar] [CrossRef]
- Cheng, Y.; Luo, F.; Jiang, Y.; Li, F.; Wei, C. The effect of calcination temperature on the structure and activity of TiO2/SiO2 composite catalysts derived from titanium sulfate and fly ash acid sludge. Colloids Surf. A Physicochem. Eng. Asp. 2018, 554, 81–85. [Google Scholar] [CrossRef]
- Eddy, D.R.; Puri, F.N.; Noviyanti, A.R. Synthesis and Photocatalytic Activity of Silica-based Sand Quartz as the Supporting TiO2 Photocatalyst. Procedia Chem. 2015, 17, 55–58. [Google Scholar] [CrossRef]
- Munasir; Sulton, A.; Triwikantoro; Zainuri, M.; Darminto. Synthesis of silica nanopowder produced from Indonesian natural sand via alkalifussion route. AIP Conf. Proc. 2013, 1555, 28–31. [Google Scholar]
- Ishmah, S.N.; Permana, M.D.; Firdaus, M.L.; Eddy, D.R. Extraction of Silica from Bengkulu Beach Sand using Alkali Fusion Method. PENDIPA J. Sci. Edu. 2020, 4, 1–5. [Google Scholar] [CrossRef]
- Firdaus, M.L.; Madina, F.E.; Yulia, F.S.; Elvia, R.; Soraya, N.I.; Eddy, D.R.; Cid-Andres, A.P. Silica Extraction from Beach Sand for Dyes Removal: Isotherms, Kinetics and Thermodynamics. Rasayan J. Chem. 2020, 13, 249–254. [Google Scholar] [CrossRef]
- Zhang, Y.; Weidenkaff, A.; Reller, A. Mesoporous Structure and Phase Transition of Nanosrystalline TiO2. Mater. Lett. 2002, 54, 375–381. [Google Scholar] [CrossRef]
- Sikong, L.; Damchan, J.; Kooptarnond, K.; Niyomwas, S. Effect of doped SiO2 and calcinations temperature on phase transformation of TiO2 photocatalyst prepared by sol-gel method. Songklanakarin J. Sci. Technol. 2008, 30, 385–391. [Google Scholar]
- Devi, L.G.; Rajashekhar, K.E. A kinetic model based on non-linear regression analysis is proposed for the degradation of phenol under UV/solar light using nitrogen doped TiO2. J. Mol Catal. A Chem. 2011, 334, 65–76. [Google Scholar] [CrossRef]
- Lin, L.; Zheng, R.Y.; Xie, J.L.; Zhu, Y.X.; Xie, Y.C. Synthesis and characterization of phosphor and nitrogen co-doped titania. Appl. Catal. B Environ. 2007, 76, 196–202. [Google Scholar] [CrossRef]
- Sirimahachai, U.; Ndiege, N.; Chandrasekharan, R.; Wongnawa, S.; Shannon, M.A. Nanosized TiO2 particles decorated on SiO2 spheres: Synthesis and photocatalytic activities. J. Sol-Gel Sci. Technol. 2010, 56, 3–6. [Google Scholar] [CrossRef]
- Sellapan, R. Mechanisms of Enhanced Activity of Model TiO2/Carbon and TiO2/Metal Nanocomposite Photocatalysts; Department of Applied Physics Chalmers University: Gotebrog, Sweden, 2013. [Google Scholar]
- Pu, S.; Hou, Y.; Chen, H.; Deng, D.; Yang, Z.; Xue, S.; Zhu, R.; Diao, Z.; Chu, W. An efficient photocatalyst for fast reduction of Cr(VI) by ultra-trace silver enhanced titania in aqueous solution. Catalysts 2018, 8, 251. [Google Scholar] [CrossRef]
- Zaccariello, G.; Moretti, E.; Storaro, L.; Riello, P.; Canton, P.; Gombac, V.; Montini, T.; Rodríguez-Castellón, E.; Benedetti, A. TiO2-mesoporous silica nanocomposites: Cooperative effect in the photocatalytic degradation of dyes and drugs. RSC Adv. 2014, 4, 37826–37837. [Google Scholar] [CrossRef]
- Gao, M.; Zhu, L.; Ong, W.L.; Wang, J.; Ho, G.W. Structural design of TiO2-based photocatalyst for H2 production and degradation applications. Catal. Sci. Technol. 2015, 5, 4703–4726. [Google Scholar] [CrossRef]
- Pal, A.; Jana, T.K.; Chatterjee, K. Silica supported TiO2 nanostructures for highly efficient photocatalytic application under visible light irradiation. Mat. Res. Bull. 2016, 76, 353–357. [Google Scholar] [CrossRef]



| Photocatalysts | Phase (%) | ||
|---|---|---|---|
| Anatase | Rutile | Amorphous SiO2 | |
| TiO2 P25 | 85.7 | 14.3 | - |
| TiO2:SiO2 (1:1) | 49.0 | 6.5 | 44.4 |
| TiO2:SiO2 (3:1) | 69.0 | 8.3 | 22.6 |
| TiO2:SiO2 (7:1) | 97.9 | 0.9 | 1.2 |
| Photocatalysts | Crystallite Size (nm) | ||
|---|---|---|---|
| Anatase | Rutile | Amorphous SiO2 | |
| TiO2 P25 | 32.84 | 22.59 | - |
| TiO2:SiO2 (1:1) | 8.79 | 9.01 | 2.57 |
| TiO2:SiO2 (3:1) | 11.79 | 7.16 | 2.59 |
| TiO2:SiO2 (7:1) | 11.58 | - | - |
| Bond Type | SiO2 | TiO2 P25 | TiO2:SiO2 (1:1) | TiO2:SiO2 (3:1) | TiO2:SiO2 (7:1) |
|---|---|---|---|---|---|
| O–H streching | 3467 cm−1 | 3411 cm−1 | 3429 cm−1 | 3411 cm−1 | 3429 cm−1 |
| O–H bending | 1638 cm−1 | 1635 cm−1 | 1634 cm−1 | 1631 cm−1 | 1636 cm−1 |
| Si–O–Si | 1091 cm−1 | - | 1100 cm−1 | 1105 cm−1 | 1102 cm−1 |
| Ti–O–Ti | - | 667 cm−1 | 711 cm−1 | 671 cm−1 | 667 cm−1 |
| Ti–O–Si | - | - | 942 cm−1 | 958 cm−1 | 942 cm−1 |
| Particle Size | TiO2/SiO2 Ratio | ||
|---|---|---|---|
| 1:1 | 3:1 | 7:1 | |
| Median (nm) | 797.7 | 628.9 | 719.0 |
| Mode (nm) | 698.5 | 620.1 | 696.4 |
| Z-Average (nm) | 557.4 | 616.9 | 683.9 |
| Photocatalysts | k (min−1) | r0 (mg/L·min) | R2 |
|---|---|---|---|
| Photocatalysis of Cr(VI): | |||
| TiO2 P25 | 0.0060 | 0.0180 | 0.9301 |
| TiO2:SiO2 (1:1) | 0.0084 | 0.0252 | 0.9462 |
| TiO2:SiO2 (3:1) | 0.0101 | 0.0303 | 0.9645 |
| TiO2:SiO2 (7:1) | 0.0162 | 0.0486 | 0.9559 |
| Photocatalysis of Pb(II): | |||
| TiO2 P25 | 0.0085 | 0.3400 | 0.9626 |
| TiO2:SiO2 (1:1) | 0.0105 | 0.4200 | 0.9224 |
| TiO2:SiO2 (3:1) | 0.0135 | 0.5400 | 0.9060 |
| TiO2:SiO2 (7:1) | 0.0143 | 0.5720 | 0.9443 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Eddy, D.R.; Ishmah, S.N.; Permana, M.D.; Firdaus, M.L. Synthesis of Titanium Dioxide/Silicon Dioxide from Beach Sand as Photocatalyst for Cr and Pb Remediation. Catalysts 2020, 10, 1248. https://doi.org/10.3390/catal10111248
Eddy DR, Ishmah SN, Permana MD, Firdaus ML. Synthesis of Titanium Dioxide/Silicon Dioxide from Beach Sand as Photocatalyst for Cr and Pb Remediation. Catalysts. 2020; 10(11):1248. https://doi.org/10.3390/catal10111248
Chicago/Turabian StyleEddy, Diana Rakhmawaty, Soraya Nur Ishmah, Muhamad Diki Permana, and M. Lutfi Firdaus. 2020. "Synthesis of Titanium Dioxide/Silicon Dioxide from Beach Sand as Photocatalyst for Cr and Pb Remediation" Catalysts 10, no. 11: 1248. https://doi.org/10.3390/catal10111248
APA StyleEddy, D. R., Ishmah, S. N., Permana, M. D., & Firdaus, M. L. (2020). Synthesis of Titanium Dioxide/Silicon Dioxide from Beach Sand as Photocatalyst for Cr and Pb Remediation. Catalysts, 10(11), 1248. https://doi.org/10.3390/catal10111248