Modification of MCM-22 Zeolite and Its Derivatives with Iron for the Application in N2O Decomposition
Abstract
:1. Introduction
2. Results
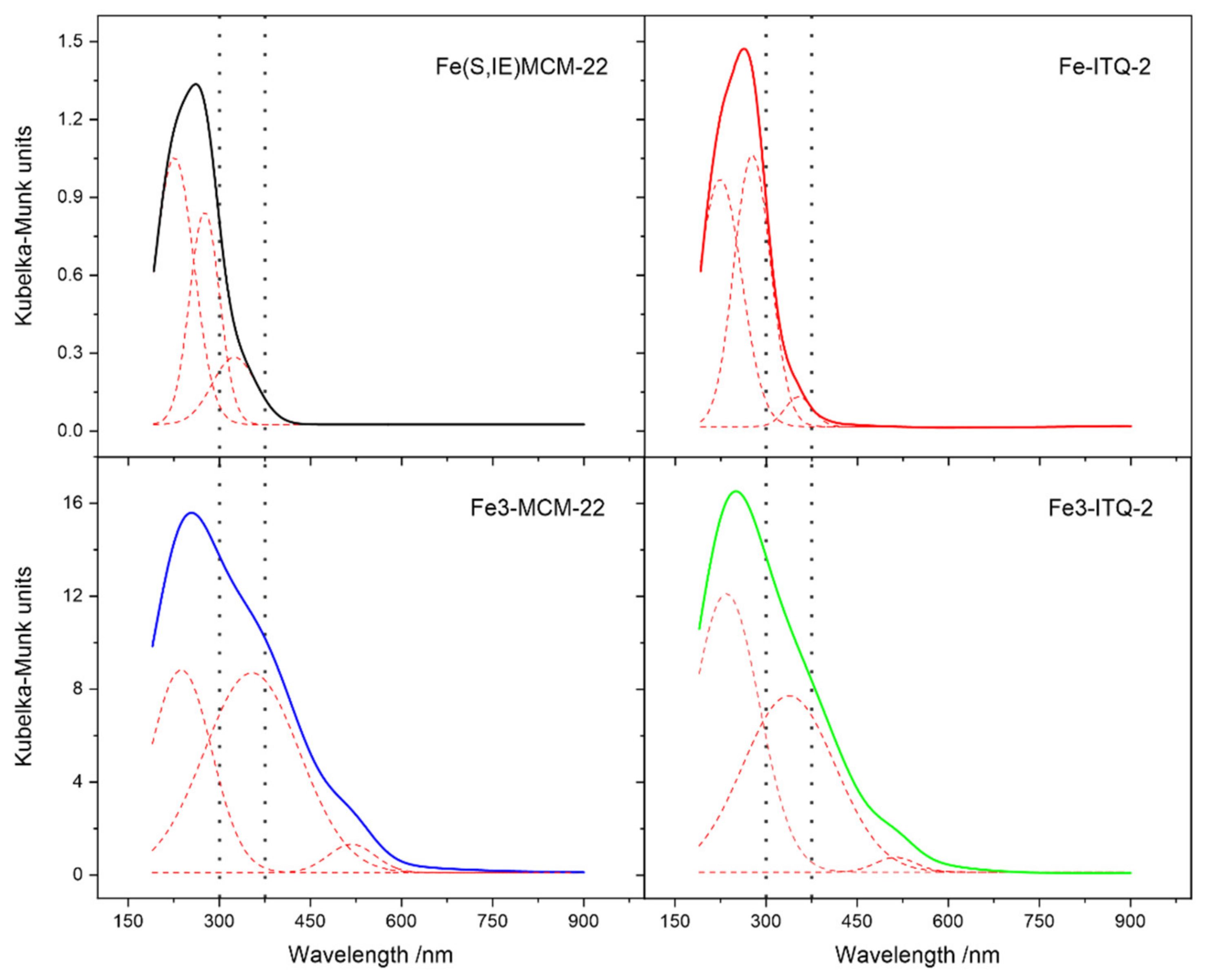
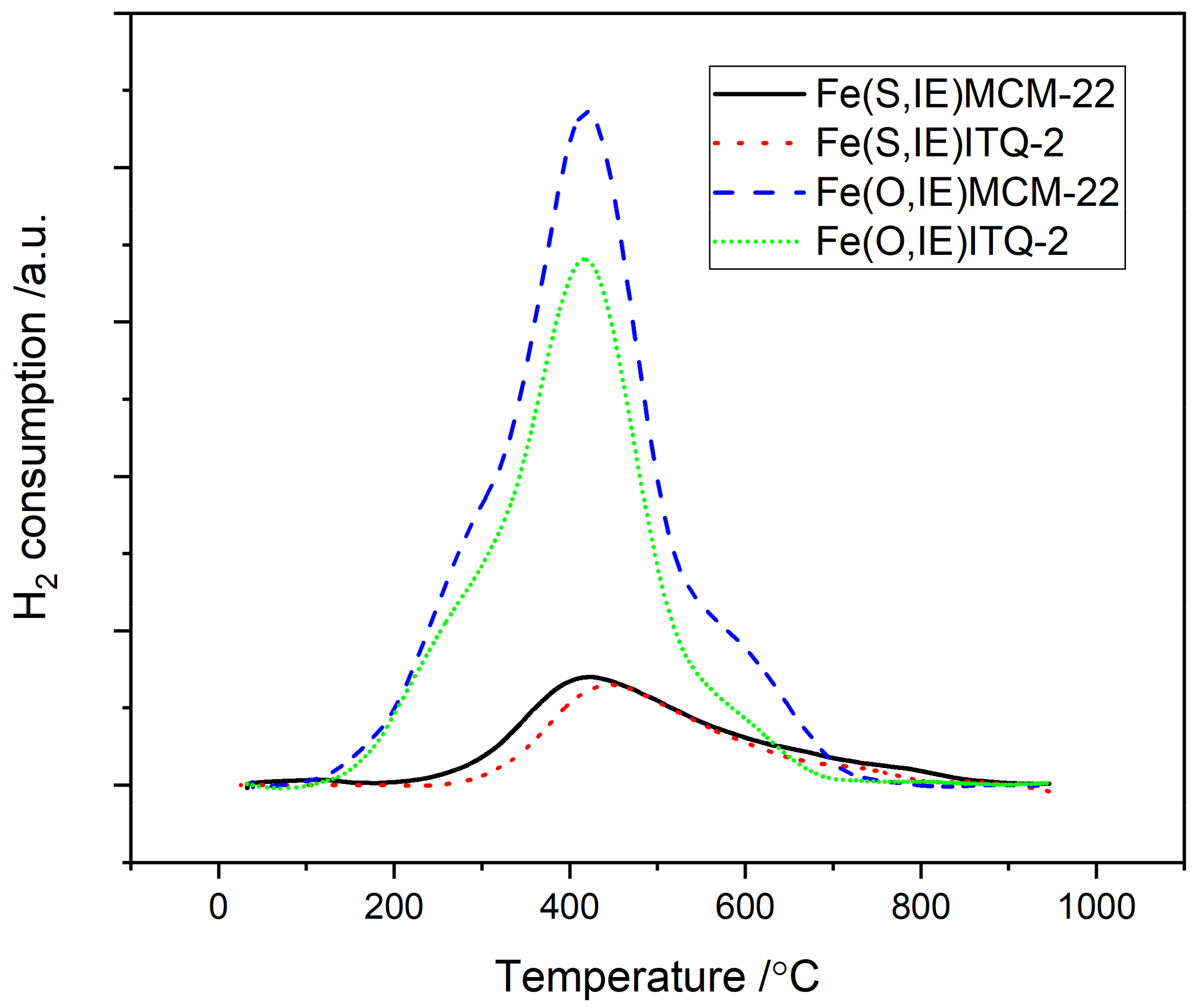
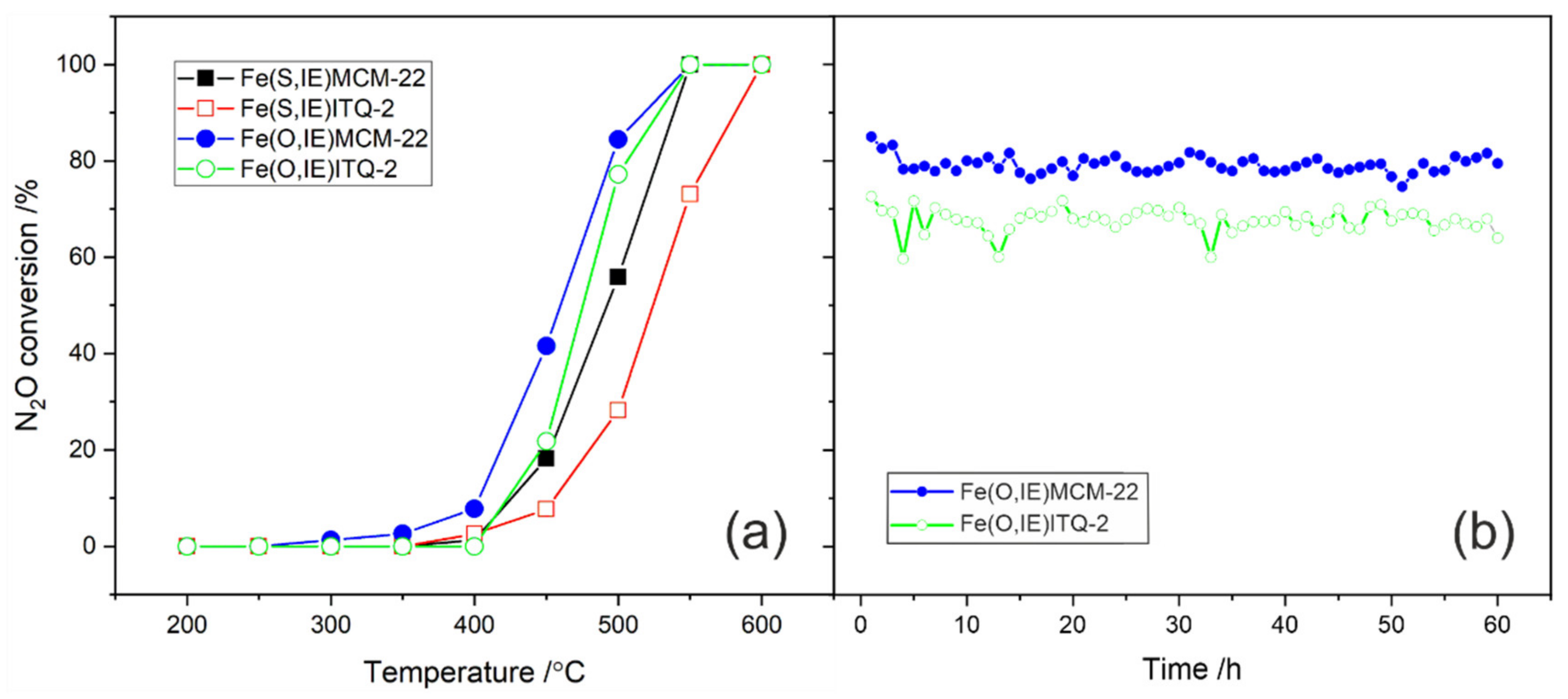
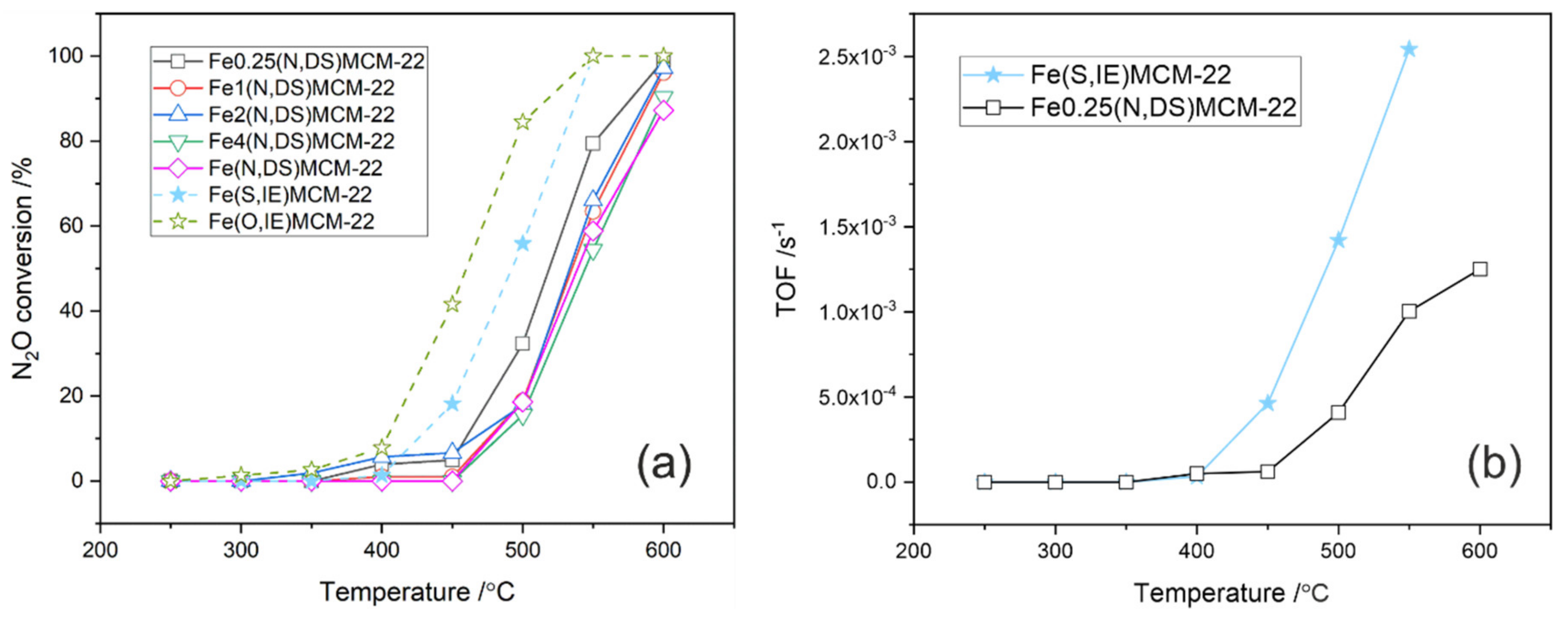
2.1. MCM-22 and ITQ-2 Modified with FeSO4∙7H2O and [Fe3(OCOCH3)7∙OH∙2H2O]NO3 by Ion-Exchange
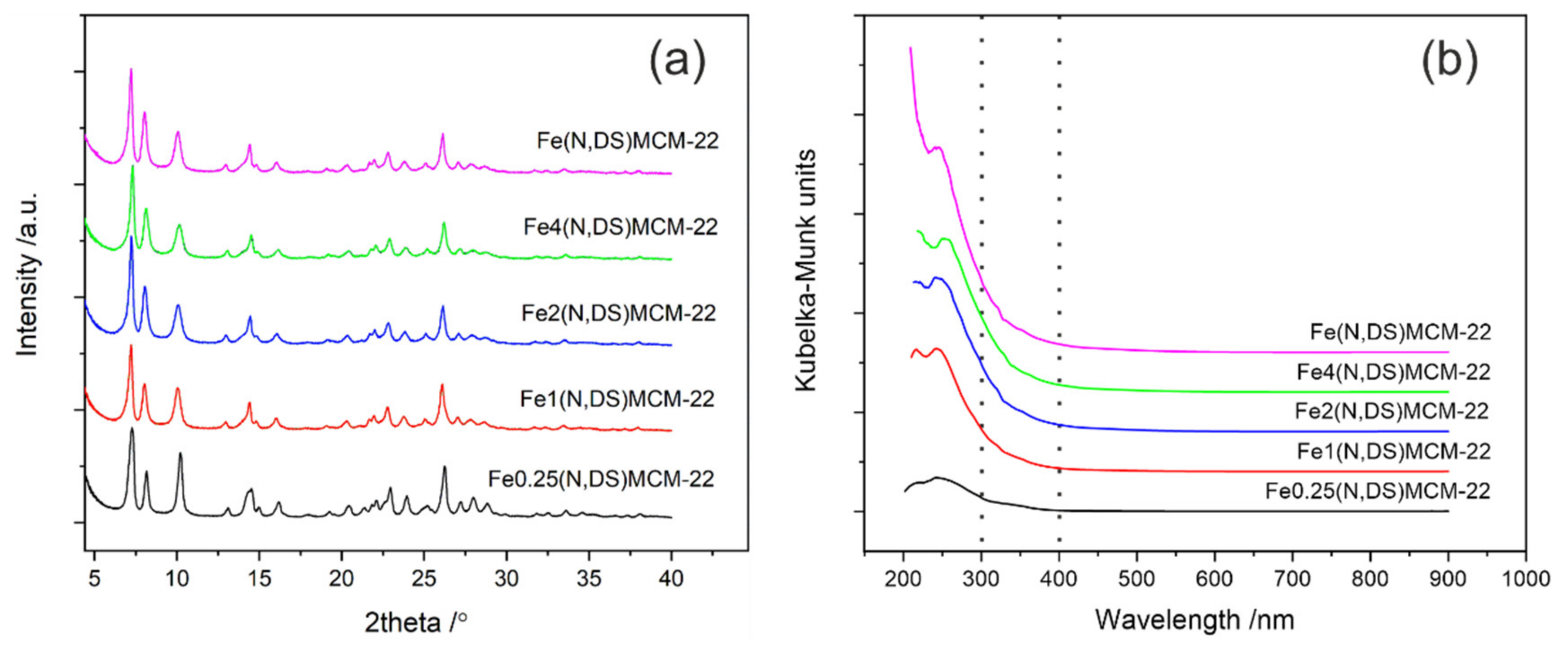
2.2. MCM-22 Zeolite Modified with Fe(NO3)3∙9H2O by Direct Synthesis
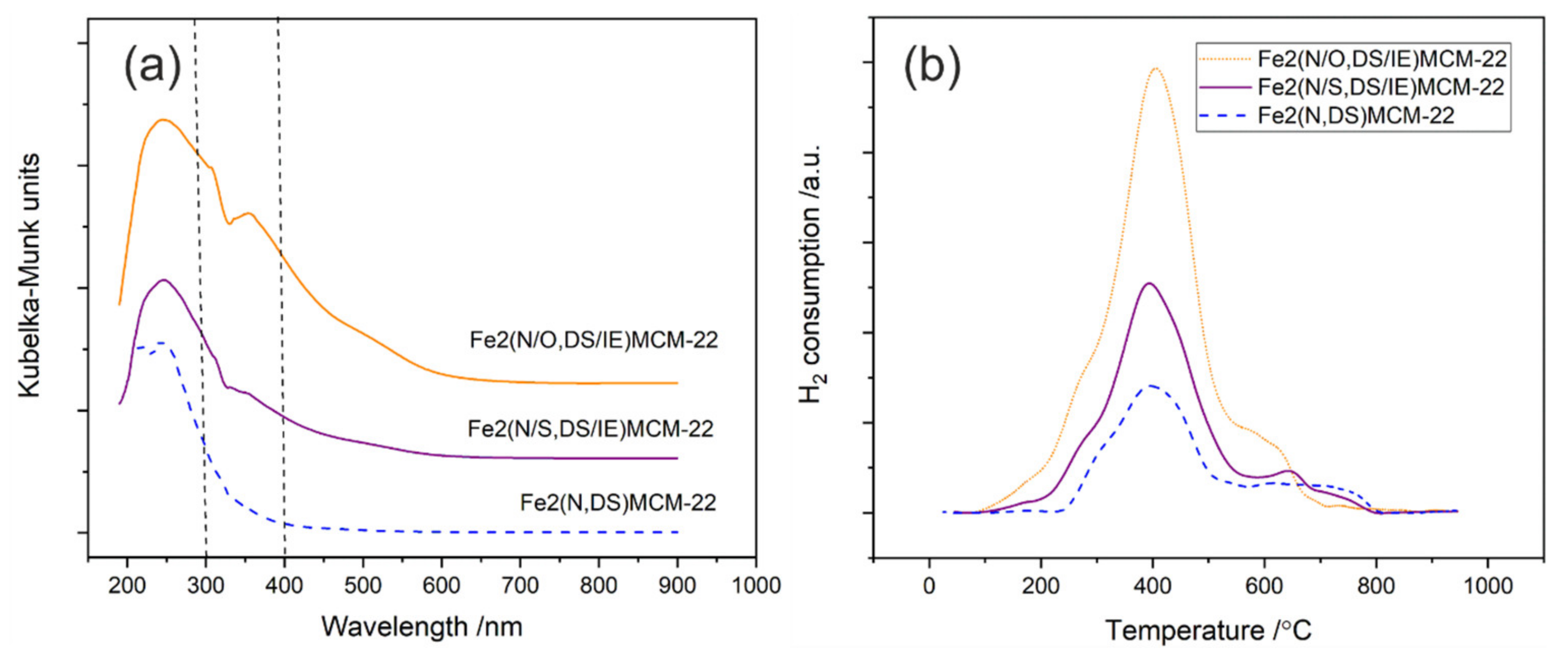
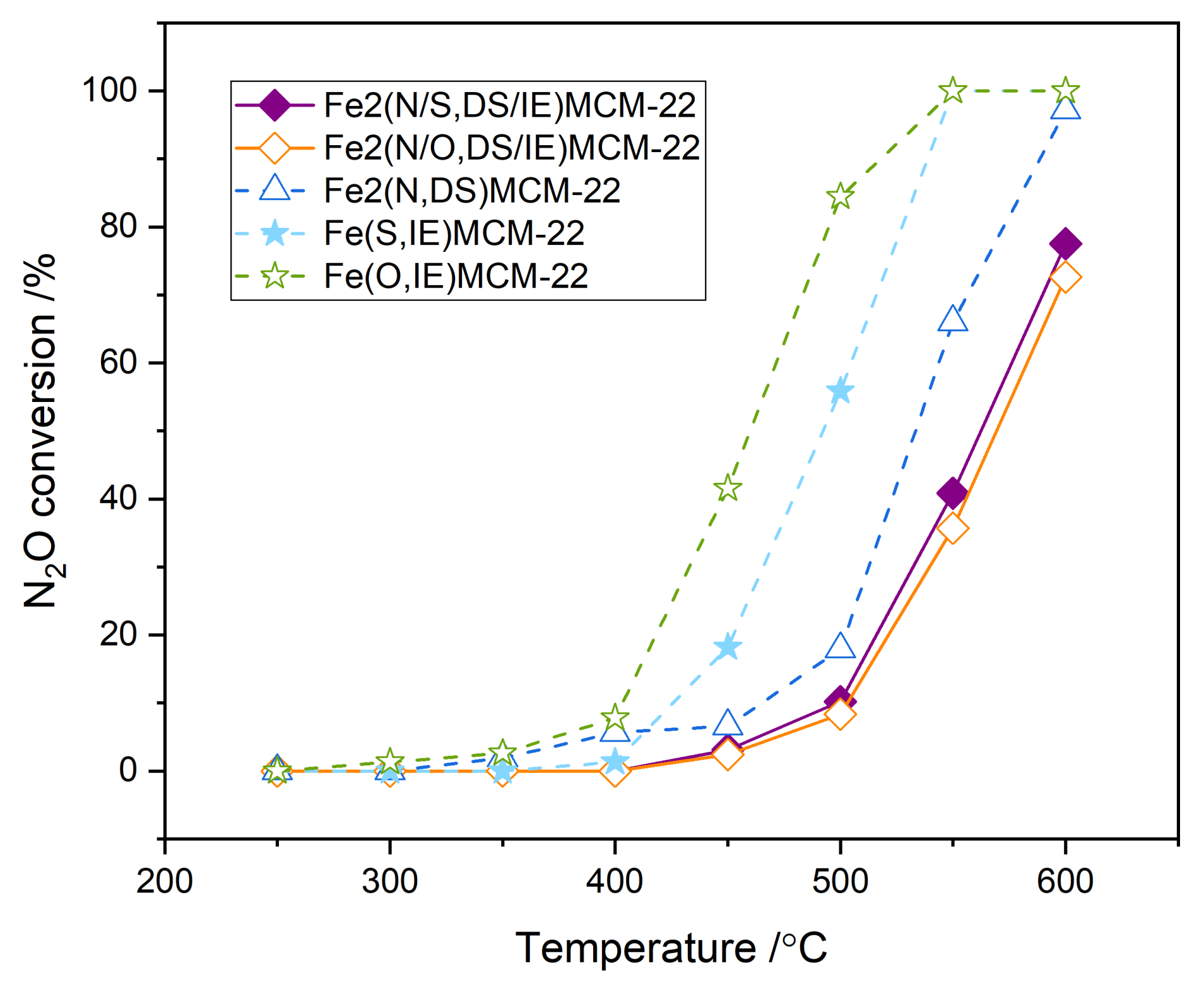
2.3. Fe2(N,DS)MCM-22 Zeolite Modified with Different Iron Precursors by Post-Synthesis Ion-Exchange
3. Materials and Methods
3.1. Catalysts Synthesis
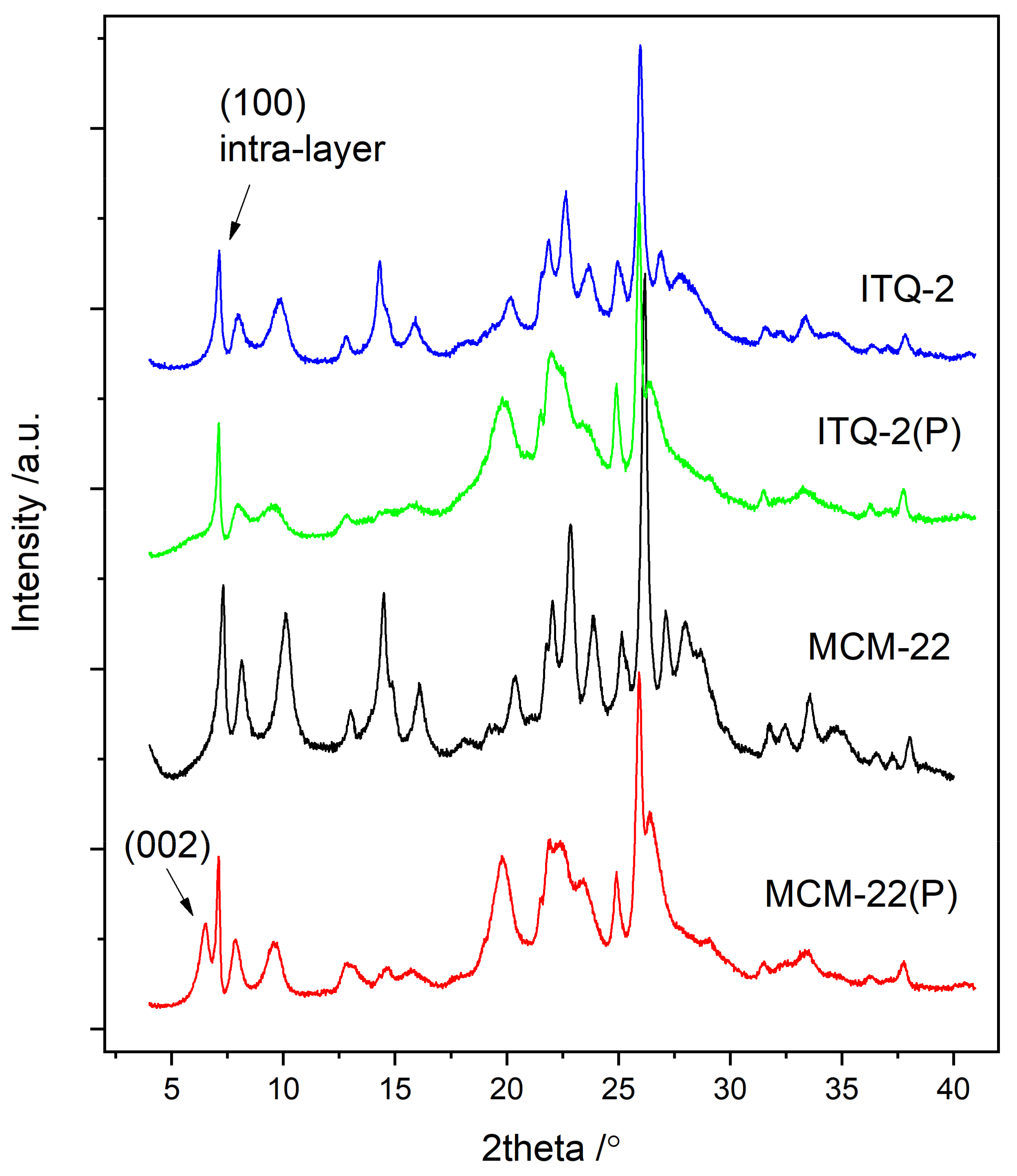
3.1.1. MCM-22 and ITQ-2
3.1.2. FeX(N,DS)MCM-22 Modified with Fe(III) by Direct Synthesis
3.1.3. Ion-Exchange
3.2. Catalysts Characterization
3.3. Catalytic Tests
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Rubin, M.K.; Chu, P. Composition of Synthetic Porous Crystalline Material, Its Synthesis and Use. U.S. Patent No 4954325, 06 November 1990. [Google Scholar]
- Osman, M.; Al-Khattaf, S.; Díaz, U.; Martínez, C.; Corma, A. Influencing the activity and selectivity of alkylaromatic catalytic transformations by varying the degree of delamination in MWW zeolites. Catal. Sci. Technol. 2016, 6, 3166–3181. [Google Scholar] [CrossRef] [Green Version]
- Xing, E.; Shi, Y.; Xie, W.; Zhang, F.; Mu, X.; Shu, X. Temperature-controlled phase-transfer hydrothermal synthesis of MWW zeolites and their alkylation performances. RSC Adv. 2016, 6, 29707–29717. [Google Scholar] [CrossRef]
- Gallego, E.M.; Paris, C.; Martínez, C.; Moliner, M.; Corma, A. Nanosized MCM-22 zeolite using simple nonsurfactant organic growth modifiers: Synthesis and catalytic applications. Chem. Commun. 2018, 54, 9989–9992. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Gao, Y.; Xie, S.; Liu, S.; Chen, F.; Xin, W.; Zhu, X.; Li, X.; Jiang, N.; Xu, L. Adjustment of the Al siting in MCM-22 zeolite and its effect on alkylation performance of ethylene with benzene. Catal. Today 2018, 316, 71–77. [Google Scholar] [CrossRef]
- Lim, T.H.; Nam, K.; Song, I.K.; Lee, K.-Y.; Kim, D.H. Effect of Si/Al2 ratios in Mo/H-MCM-22 on methane dehydroaromatization. Appl. Catal. A Gen. 2018, 552, 11–20. [Google Scholar] [CrossRef]
- Lee, W.; Lee, T.; Jang, H.-G.; Cho, S.J.; Choi, J.; Ha, K.-S. Effects of hierarchical zeolites on aromatization of acetylene. Catal. Today 2018, 303, 177–184. [Google Scholar] [CrossRef]
- Zhang, L.; Wang, H.; Liu, G.; Gao, K.; Wu, J. Methanol-to-olefin conversion over H-MCM-22 catalyst. J. Mol. Catal. A Chem. 2016, 411, 311–316. [Google Scholar] [CrossRef]
- Chen, J.; Liang, T.; Li, J.; Wang, S.; Qin, Z.; Wang, P.; Huang, L.; Fan, W.; Wang, J. Regulation of framework aluminum siting and acid distribution in H-MCM-22 by boron incorporation and its effect on the catalytic performance in methanol to hydrocarbons. ACS Catal. 2016, 6, 2299–2313. [Google Scholar] [CrossRef]
- Carriço, C.S.; Cruz, F.T.; Dos Santos, M.B.; Oliveira, D.S.; Pastore, H.O.; Andrade, H.M.C.; Mascarenhas, A.J.S. MWW-type catalysts for gas phase glycerol dehydration to acrolein. J. Catal. 2016, 334, 34–41. [Google Scholar] [CrossRef]
- Dos Santos, M.B.; Andrade, H.M.C.; Mascarenhas, A.J.S. Oxidative dehydration of glycerol over alternative H,Fe-MCM-22 catalysts: Sustainable production of acrylic acid. Microporous Mesoporous Mater. 2019, 278, 366–377. [Google Scholar] [CrossRef]
- Wang, Y.; Yokoi, T.; Namba, S.; Kondo, J.N.; Tatsumi, T. Improvement of catalytic performance of MCM-22 in the cracking of n-hexane by controlling the acidic property. J. Catal. 2016, 333, 17–28. [Google Scholar] [CrossRef]
- Hussain, A.I.; Aitani, A.M.; Kubů, M.; Čejka, J.; Al-Khattaf, S. Catalytic cracking of Arabian Light VGO over novel zeolites as FCC catalyst additives for maximizing propylene yield. Fuel 2016, 167, 226–239. [Google Scholar] [CrossRef]
- Hao, Q.-Q.; Lei, C.-Y.; Song, Y.-H.; Liu, Z.-T.; Liu, Z.-W. The delaminating and pillaring of MCM-22 for Fischer–Tropsch synthesis over cobalt. Catal. Today 2016, 274, 109–115. [Google Scholar] [CrossRef]
- Wojtaszek-Gurdak, A.; Calvino-Casilda, V.; Grzesinska, A.; Martin-Aranda, R.; Ziolek, M. Impact of Brønsted acid sites in MWW zeolites modified with cesium and amine species on Knoevenagel condensation. Microporous Mesoporous Mater. 2019, 280, 288–296. [Google Scholar] [CrossRef]
- Ma, X.; Zhou, D.; Chu, X.; Li, D.; Wang, J.; Song, W.; Xia, Q. Highly selective isomerization of biomass β-pinene over hierarchically acidic MCM-22 catalyst. Microporous Mesoporous Mater. 2017, 237, 180–188. [Google Scholar] [CrossRef]
- Štekrová, M.; Kubů, M.; Shamzhy, M.; Musilová, Z.; Čejka, J. α-Pinene oxide isomerization: Role of zeolite structure and acidity in the selective synthesis of campholenic aldehyde. Catal. Sci. Technol. 2018, 8, 2488–2501. [Google Scholar] [CrossRef]
- Rutkowska, M.; Díaz, U.; Palomares, A.E.; Chmielarz, L. Cu and Fe modified derivatives of 2D MWW-type zeolites (MCM-22, ITQ-2 and MCM-36) as new catalysts for DeNOx proces. Appl. Catal. B Environ. 2015, 168–169, 531–539. [Google Scholar] [CrossRef]
- Wojtaszek-Gurdak, A.; Sobczak, I.; Grzelak, K.; Ziolek, M.; Hartfelder, U.; Van Bokhoven, J.A. The role of pillaring in MCM-22 on the dispersion of noble metals and catalytic activity. Mater. Res. Bull. 2016, 76, 169–178. [Google Scholar] [CrossRef]
- Roth, W.J.; Kresge, C.T.; Vartuli, J.C.; Leonowicz, M.E.; Fung, A.S.; McCullen, S.B. MCM-36: The first pillared molecular sieve with zeoliteproperties. Stud. Surf. Sci. Catal. 1995, 94, 301–308. [Google Scholar]
- Corma, A.; Fornés, V.; Pergher, S.; Maesen, T.L.M.; Buglass, J.G. Delaminated zeolite precursors as selective acidic catalysts. Nature 1998, 396, 353–356. [Google Scholar] [CrossRef]
- Corma, A.; Fornés, V.; Guil, J.M.; Pergher, S.; Maesen, T.L.M.; Buglass, J.G. Preparation, characterisation and catalytic activity of ITQ-2, a delaminated zeolite. Microporous Mesoporous Mater. 2000, 38, 301–309. [Google Scholar] [CrossRef]
- Corma, A.; Díaz, U.; Fornés, V.; Guil, J.M.; Martínez-Triguero, J.; Creyghton, E.J. characterization and catalytic activity of MCM-22 and MCM-56 compared with ITQ-2. J. Catal. 2000, 191, 218–224. [Google Scholar] [CrossRef]
- Kapteijn, F.; Rodriguez-Mirasol, J.; Moulijn, J.A. Heterogeneous catalytic decomposition of nitrous oxide. Appl. Catal. B Environ. 1996, 9, 25–64. [Google Scholar] [CrossRef]
- Pérez-Ramírez, J.; Kapteijn, F.; Schöffel, K.; Moulijn, J.A. Formation and control of N2O in nitric acid production: Where do we stand today? Appl. Catal. B Environ. 2003, 44, 117–151. [Google Scholar] [CrossRef]
- Pérez-Ramírez, J.; Kapteijn, F.; Brückner, A. Active site structure sensitivity in N2O conversion over FeMFI zeolites. J. Catal. 2003, 218, 234–238. [Google Scholar] [CrossRef]
- Pirngruber, G.D.; Luechinger, M.; Roy, P.K.; Cecchetto, A.; Smirniotis, P. N2O decomposition over iron-containing zeolites prepared by different methods: A comparison of the reaction mechanism. J. Catal. 2004, 224, 429–440. [Google Scholar] [CrossRef]
- Jíša, K.; Nováková, J.; Schwarze, M.; Vondrová, A.; Sklenák, S.; Sobalik, Z. Role of the Fe-zeolite structure and iron state in the N2O decomposition: Comparison of Fe-FER, Fe-BEA, and Fe-MFI catalysts. J. Catal. 2009, 262, 27–34. [Google Scholar] [CrossRef]
- Sádovská, G.; Bernauera, M.; Bernauer, B.; Tabor, E.; Vondrová, A.; Sobalík, Z. On the mechanism of high-temperature N2O decomposition over Fe-FER in the presence of NO. Catal. Commun. 2018, 112, 58–62. [Google Scholar] [CrossRef]
- Stelmachowski, P.; Maniak, G.; Kotarba, A.; Sojka, Z. Strong electronic promotion of Co3O4 towards N2O decomposition by surface alkali dopants. Catal. Commun. 2009, 10, 1062–1065. [Google Scholar] [CrossRef]
- Maniak, G.; Stelmachowski, P.; Stanek, J.J.; Kotarba, A.; Sojka, Z. Catalytic properties in N2O decomposition of mixed cobalt–iron spinels. Catal. Commun. 2011, 15, 127–131. [Google Scholar] [CrossRef]
- Abu-Zied, B.M.; Soliman, S.A.; Abdellah, S.E. Enhanced direct N2O decomposition over CuxCo1-xCo2O4 (0.0 ≤ x ≤ 1.0) spinel-oxide catalysts. J. Ind. Eng. Chem. 2015, 21, 814–821. [Google Scholar] [CrossRef]
- Wójcik, S.; Thersleff, T.; Gębska, K.; Grzybek, G.; Kotarba, A. Atomic-Level Dispersion of Bismuth over Co3O4 Nanocrystals—Outstanding Promotional Eect in Catalytic DeN2O. Catalysts 2020, 10, 351. [Google Scholar] [CrossRef] [Green Version]
- Wójcik, S.; Grzybek, G.; Stelmachowski, P.; Sojka, Z.; Kotarba, A. Bulk, Surface and Interface Promotion of Co3O4 for the Low-Temperature N2O Decomposition Catalysis. Catalysts 2019, 10, 41. [Google Scholar] [CrossRef] [Green Version]
- Pacultová, K.; Karásková, K.; Kovanda, F.; Jirátová, K.; Šrámek, J.; Kustrowski, P.; Kotarba, A.; Chromčáková, Z.; Kočí, K.; Obalová, L. K-Doped Co-Mn-Al Mixed Oxide Catalyst for N2O Abatement from Nitric Acid Plant Waste Gases: Pilot Plant Studies. Ind. Eng. Chem. Res. 2016, 55, 7076–7084. [Google Scholar] [CrossRef]
- Pérez-Ramírez, J.; Kapteijn, F.; Moulijn, J.A. High activity and stability of the Rh-free Co-based ex-hydrotalcite containing Pd in the catalytic decomposition of N2O. Catal. Lett. 1999, 60, 133–138. [Google Scholar] [CrossRef]
- Kovanda, F.; Rojka, T.; Dobešová, J.; Machovič, V.; Bezdičkac, P.; Obalová, L.; Jirátová, K.; Grygar, T. Mixed oxides obtained from Co and Mn containing layered double hydroxides: Preparation, characterization, and catalytic properties. J. Solid State Chem. 2006, 179, 812–823. [Google Scholar] [CrossRef]
- Obalová, L.; Karásková, K.; Jirátová, K.; Kovanda, F. Effect of potassium in calcined Co–Mn–Al layered double hydroxide on the catalytic decomposition of N2O. Appl. Catal. B Environ. 2009, 90, 132–140. [Google Scholar] [CrossRef]
- Richards, N.; Nowicka, E.; Carter, J.H.; Morgan, D.J.; Dummer, N.F.; Golunski, S.; Hutchings, G.J. Investigating the Influence of Fe Speciation on N2O Decomposition Over Fe-ZSM-5 Catalysts. Top. Catal. 2018, 61, 1983–1992. [Google Scholar] [CrossRef] [Green Version]
- Dubkov, K.A.; Ovanesyan, N.S.; Shteinman, A.A.; Starokon, E.V.; Panov, G.I. Evolution of Iron States and Formation of α-Sites upon Activation of FeZSM-5 Zeolites. J. Catal. 2002, 207, 341–352. [Google Scholar] [CrossRef]
- Pirngruber, G.D. The surface chemistry of N2O decomposition on iron containing zeolites (I). J. Catal. 2003, 219, 456–463. [Google Scholar] [CrossRef]
- Pérez-Ramírez, J.; Kapteijn, F.; Groen, J.C.; Doménech, A.; Mul, G.; Moulijn, J.A. Steam-activated FeMFI zeolites. Evolution of iron species and activity in direct N2O decomposition. J. Catal. 2003, 214, 33–45. [Google Scholar] [CrossRef]
- Roth, W.J.; Douglas, L.D. Expanded view of zeolite structures and their varriability based on layerd nature of 3-D frameworks. Microporous Mesoporous Mater. 2011, 142, 32–36. [Google Scholar] [CrossRef]
- Roth, W.J.; Douglas, L.D.; Kennedy, G.J. Discovery of new MWW family zeolite EMM-10: Identification of EMM-10P as the missing MWW precursor with disordered layers. Microporous Mesoporous Mater. 2011, 142, 168–177. [Google Scholar] [CrossRef]
- Rutkowska, M.; Borcuch, A.; Marzec, A.; Kowalczyk, A.; Samojeden, B.; Moreno, J.M.; Díaz, U.; Chmielarz, L. Influence of iron aggregation on the catalytic performance of desilicated MFI in the DeNOx proces. Microporous Mesoporous Mater. 2020, 304, 109114–109122. [Google Scholar] [CrossRef]
- Pan, H.; Guo, Y.; Bi, H.T. NOx adsorption and reduction with C3H6 over Fe/zeolite catalysts: Effect of catalyst suport. Chem. Eng. J. 2015, 280, 66–73. [Google Scholar] [CrossRef]
- Romero-Sáez, M.; Divakar, D.; Aranzabal, A.; González-Velasco, J.R.; González-Marcos, J.A. Catalytic oxidation of trichloroethylene over Fe-ZSM-5: Influence of the preparation method on the iron species and the catalytic activity. Appl. Catal. B Environ. 2016, 180, 210–218. [Google Scholar] [CrossRef]
- Gao, Y.; Zheng, B.; Wu, G.; Ma, F.; Liu, C. Effect of the Si/Al ratio on the performance of hierarchical ZSM-5 zeolites for methanol aromatization. RSC Adv. 2016, 6, 83581–83588. [Google Scholar] [CrossRef]
- Testa, F.; Crea, F.; Diodati, G.D.; Pasqua, L.; Aiello, R.; Terwagne, G.; Lentz, P.; Nagy, J.B. Synthesis and characterization of Fe- and [Fe,Al]-MCM-22 zeolites. Microporous Mesoporous Mater. 1999, 30, 187–197. [Google Scholar] [CrossRef]
- Macina, D.; Piwowarska, Z.; Góra-Marek, K.; Tarach, K.; Rutkowska, M.; Girman, V.; Błachowski, A.; Chmielarz, L. SBA-15 loaded with iron by various methods as catalyst for DeNOx proces. Mater. Res. Bull. 2016, 78, 72–82. [Google Scholar] [CrossRef]
- Macina, D.; Opioła, A.; Rutkowska, M.; Basąg, S.; Piwowarska, Z.; Michalik, M.; Chmielarz, L. Mesoporous silica materials modified with aggregated transition metal species (Cr, Fe and Cr-Fe) in the role of catalysts for selective catalytic oxidation of ammonia to dinitrogen. Mater. Chem. Phys. 2017, 187, 60–71. [Google Scholar] [CrossRef]
- Rouquerol, J.; Llewellyn, P.; Rouquerol, F. Is the BET equation applicable to microporous adsorbents? Stud. Surf. Sci. Catal. 2007, 160, 49–56. [Google Scholar]
- Rutkowska, M.; Duda, M.; Macina, D.; Górecka, S.; Dębek, R.; Moreno, J.M.; Díaz, U.; Chmielarz, L. Mesoporous Beta zeolite functionalisation with FexCry oligocations; catalytic activity in the NH3-SCO proces. Microporous Mesoporous Mater. 2019, 278, 1–13. [Google Scholar] [CrossRef]
- Borcuch, A.; Rutkowska, M.; Marzec, A.; Kowalczyk, A.; Michalik, M.; Moreno, J.M.; Díaz, U.; Chmielarz, L. Selective ammonia oxidation over ZSM-5 zeolite: Impact of catalyst’s suport and type of deposited iron species. Catal. Today 2020, 348, 223–229. [Google Scholar] [CrossRef]

| Sample Code. | Iron Source | Modification Method | SBET/ m2/g | SEXT/ m2/g | VMIC /cm3/g | VMES/ cm3/g | Fe (%) | H2/Fe |
|---|---|---|---|---|---|---|---|---|
| MCM-22 | ---- | ---- | 622 | 117 | 0.202 | 0.281 | --- | --- |
| ITQ-2 | ---- | ---- | 608 | 186 | 0.178 | 0.384 | --- | --- |
| Fe(S,IE)MCM-22 | S | IE | 611 | 117 | 0.198 | 0.287 | 0.82 | 1.44 |
| Fe(S,IE)ITQ-2 | S | IE | 586 | 173 | 0.173 | 0.359 | 0.80 | 1.29 |
| Fe(O,IE)MCM-22 | O | IE | 519 | 131 | 0.156 | 0.285 | 12.67 | 0.45 |
| Fe(O,IE)ITQ-2 | O | IE | 519 | 173 | 0.145 | 0.353 | 9.79 | 0.43 |
| Fe0.25(N,DS)MCM-22 | N | DS | 614 | 53 | 0.223 | 0.125 | 1.65 | 0.83 |
| Fe1(N,DS)MCM-22 | N | DS | 514 | 69 | 0.177 | 0.171 | 3.40 | 0.56 |
| Fe2(N,DS)MCM-22 | N | DS | 563 | 79 | 0.193 | 0.200 | 4.50 | 0.56 |
| Fe4(N,DS)MCM-22 | N | DS | 531 | 74 | 0.183 | 0.177 | 5.28 | 0.43 |
| Fe(N,DS)MCM-22 | N | DS | 561 | 74 | 0.195 | 0.178 | 6.51 | 0.51 |
| Fe2(N/S,DS/IE)MCM-22 | N/S | DS/IE | 542 | 92 | 0.180 | 0.219 | 6.70 | 0.48 |
| Fe2(N/O,DS/IE)MCM-22 | N/O | DS/IE | 473 | 100 | 0.149 | 0.217 | 13.50 | 0.43 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rutkowska, M.; Jankowska, A.; Różycka-Dudek, E.; Dubiel, W.; Kowalczyk, A.; Piwowarska, Z.; Llopis, S.; Díaz, U.; Chmielarz, L. Modification of MCM-22 Zeolite and Its Derivatives with Iron for the Application in N2O Decomposition. Catalysts 2020, 10, 1139. https://doi.org/10.3390/catal10101139
Rutkowska M, Jankowska A, Różycka-Dudek E, Dubiel W, Kowalczyk A, Piwowarska Z, Llopis S, Díaz U, Chmielarz L. Modification of MCM-22 Zeolite and Its Derivatives with Iron for the Application in N2O Decomposition. Catalysts. 2020; 10(10):1139. https://doi.org/10.3390/catal10101139
Chicago/Turabian StyleRutkowska, Małgorzata, Aleksandra Jankowska, Ewelina Różycka-Dudek, Wiktoria Dubiel, Andrzej Kowalczyk, Zofia Piwowarska, Sebastián Llopis, Urbano Díaz, and Lucjan Chmielarz. 2020. "Modification of MCM-22 Zeolite and Its Derivatives with Iron for the Application in N2O Decomposition" Catalysts 10, no. 10: 1139. https://doi.org/10.3390/catal10101139
APA StyleRutkowska, M., Jankowska, A., Różycka-Dudek, E., Dubiel, W., Kowalczyk, A., Piwowarska, Z., Llopis, S., Díaz, U., & Chmielarz, L. (2020). Modification of MCM-22 Zeolite and Its Derivatives with Iron for the Application in N2O Decomposition. Catalysts, 10(10), 1139. https://doi.org/10.3390/catal10101139








