Glycerol Acetylation Mediated by Thermally Hydrolysed Biosolids-Based Material
Abstract
1. Introduction
2. Results
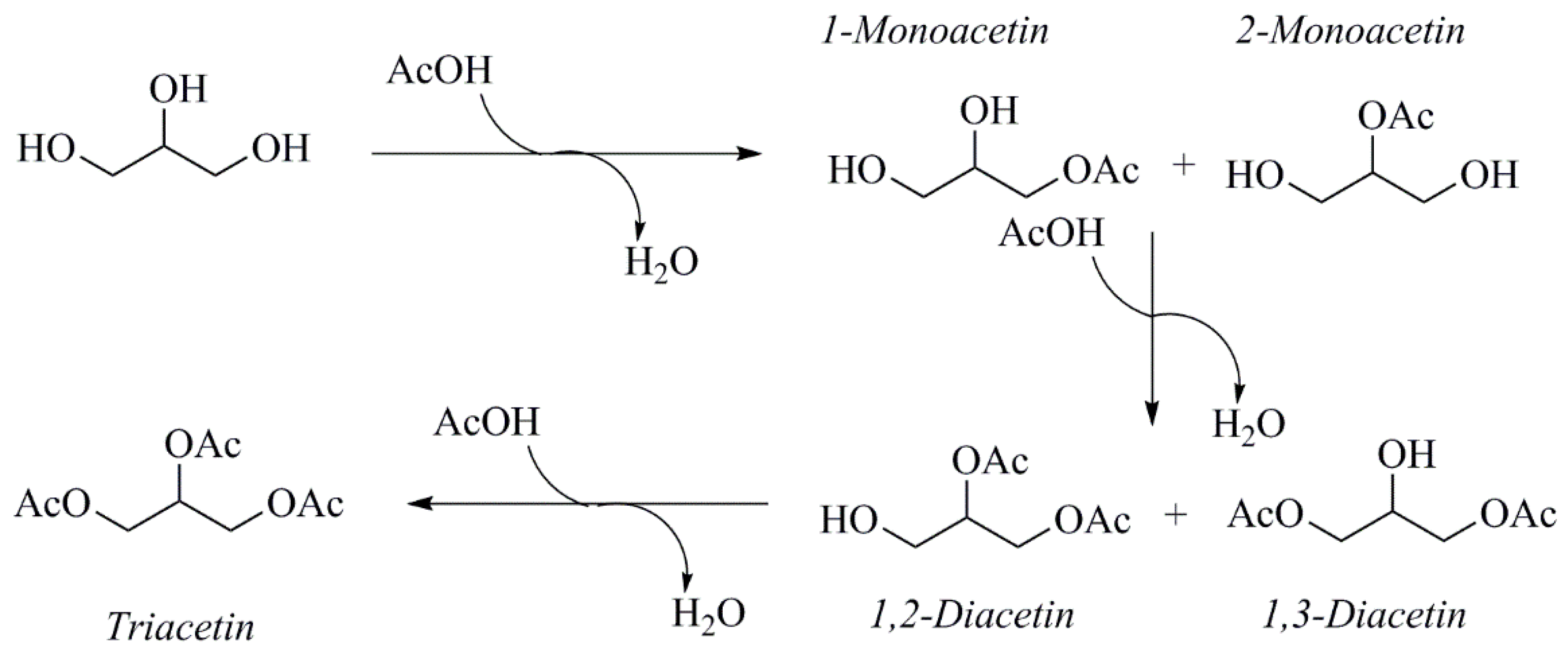
2.1. Preliminary Considerations Regarding Catalyst-Induced Reactivity and Composition
2.2. Influence of Temperature on Glycerol Acetylation Conversion Rate and Selectivity
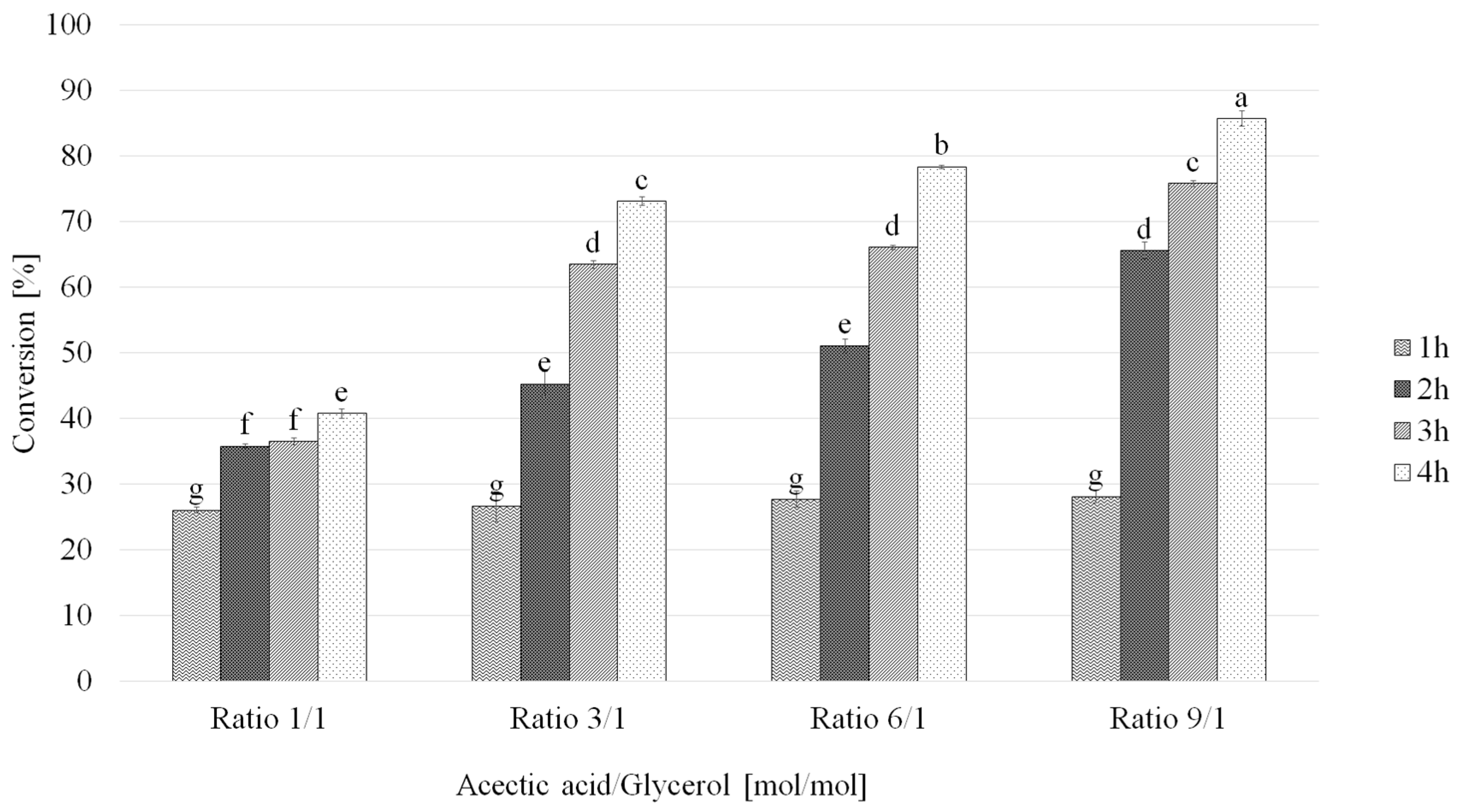
2.3. Influence of the Acetic Acid:Glycerol Ratio on Glycerol Acetylation Conversion Rate and Selectivity
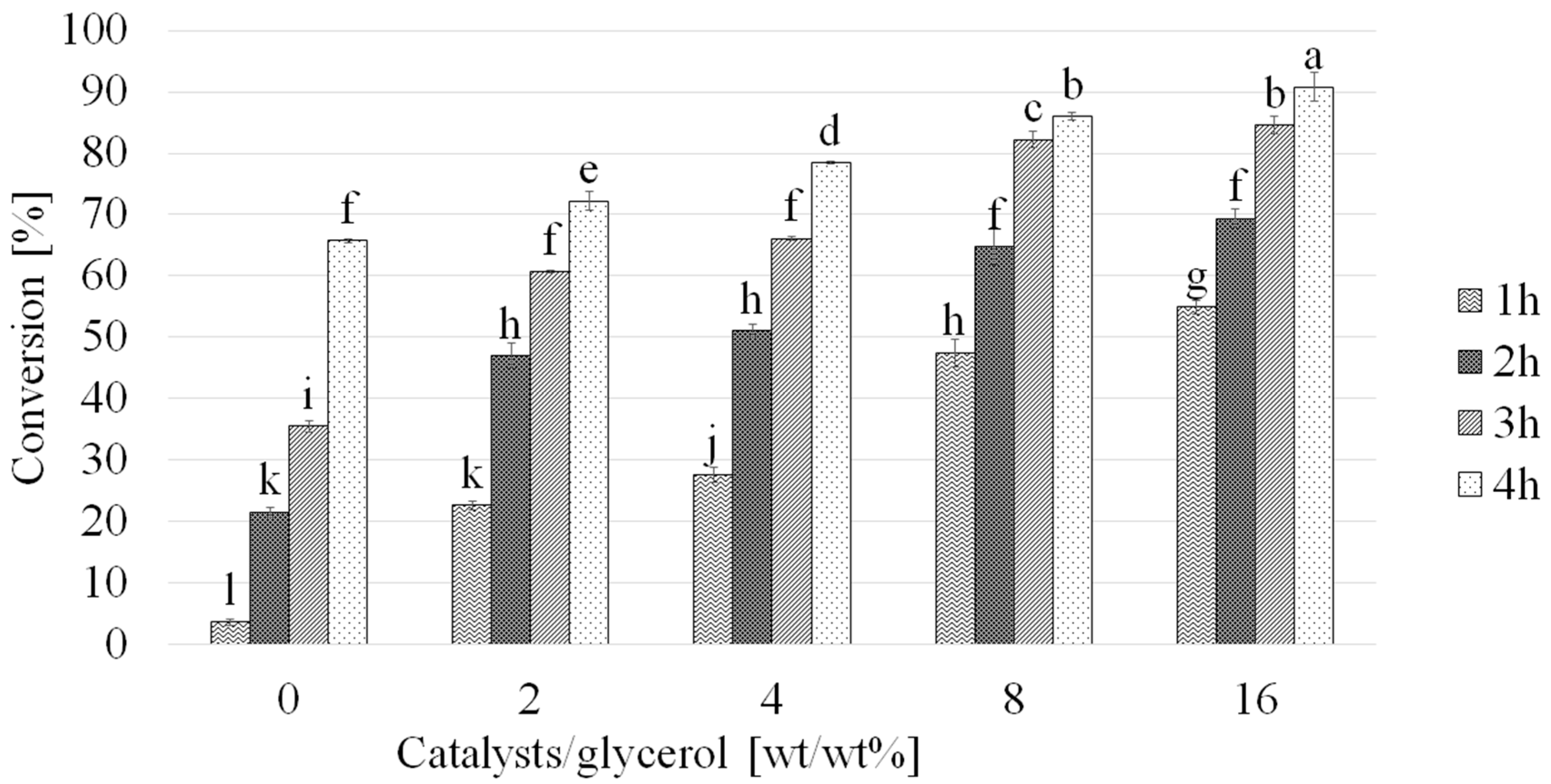
2.4. Influence of Catalyst Loading on Glycerol Acetylation Conversion Rate and Selectivity
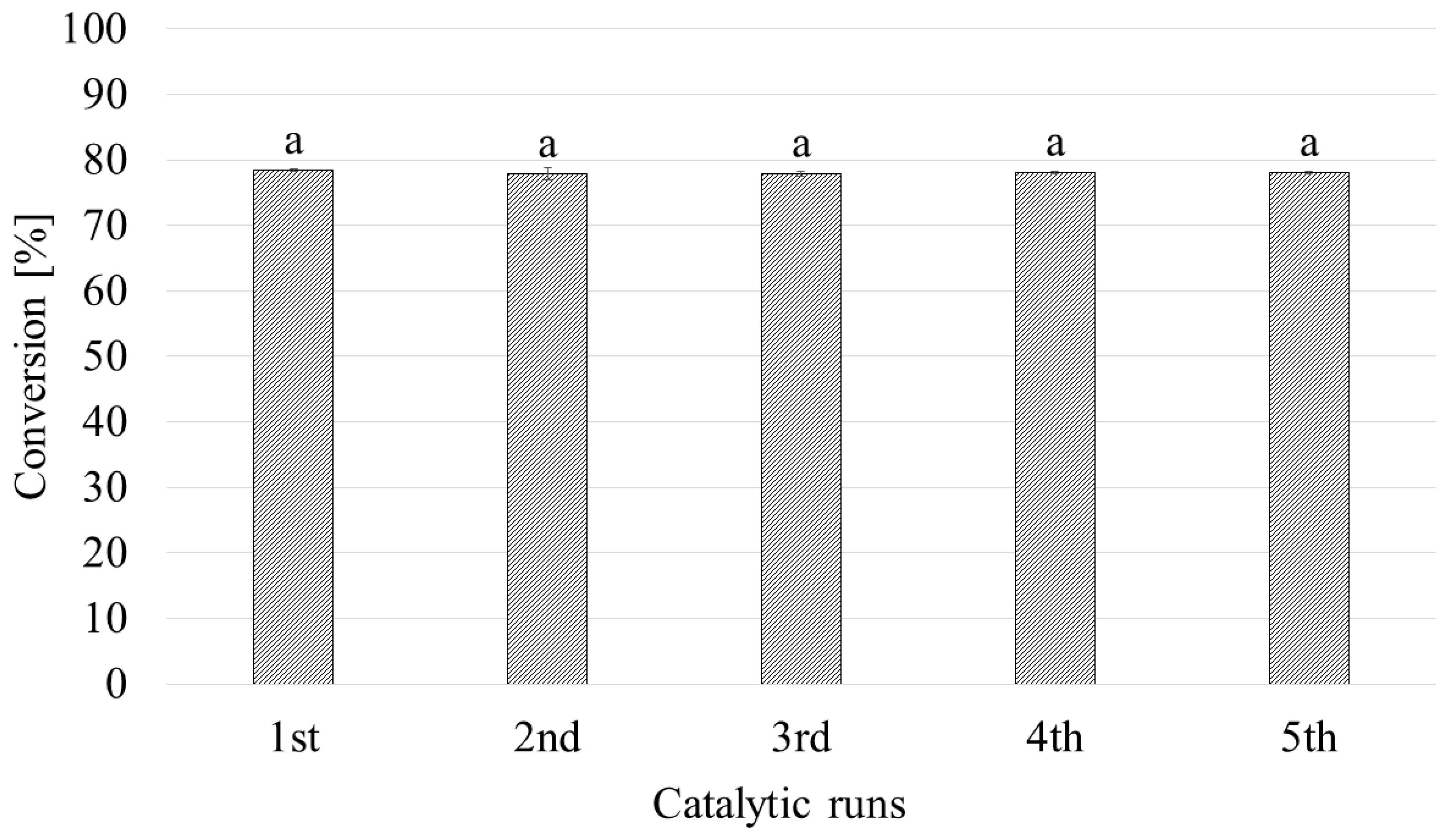
2.5. Influence of Catalyst Recycling on Glycerol Acetylation Conversion Rate and Selectivity
2.6. Considerations on Catalytic Performance of Thermally Hydrolysed Biosolids and Available Catalysts
3. Materials and Methods
3.1. Materials
3.2. Methods
3.2.1. Catalytic Acetylation of Glycerol
3.2.2. Sample Analysis
3.2.3. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Gasparatos, A.; Doll, C.N.H.; Esteban, M.; Ahmed, A.; Olang, T.A. Renewable Energy and Biodiversity: Implications for Transitioning to A Green Economy. Renew. Sustain. Energy Rev. 2017, 70, 161–184. [Google Scholar] [CrossRef]
- Mahmudul, H.; Hagos, F.; Mamat, R.; Adam, A.A.; Ishak, W.; Alenezi, R. Production, Characterization and Performance of Biodiesel as An Alternative Fuel in Diesel Engines—A Review. Renew. Sustain. Energy Rev. 2017, 72, 497–509. [Google Scholar] [CrossRef]
- Mardhiah, H.H.; Ong, H.C.; Masjuki, H.; Lim, S.; Lee, H. A Review on Latest Developments and Future Prospects of Heterogeneous Catalyst in Biodiesel Production from Non-Edible Oils. Renew. Sustain. Energy Rev. 2017, 67, 1225–1236. [Google Scholar] [CrossRef]
- Espinosa-Gonzalez, I.; Parashar, A.; Chae, M.; Bressler, D.C. Cultivation of Oleaginous Yeast Using Aqueous Fractions Derived from Hydrothermal Pretreatments of Biomass. Bioresour. Technol. 2014, 170, 413–420. [Google Scholar] [CrossRef] [PubMed]
- Asomaning, J.; Mussone, P.; Bressler, D.C. Two-Stage Thermal Conversion of Inedible Lipid Feedstocks to Renewable Chemicals and Fuels. Bioresour. Technol. 2014, 158, 55–62. [Google Scholar] [CrossRef]
- Espinosa-Gonzalez, I.; Asomaning, J.; Mussone, P.; Bressler, D.C. Two-Step Thermal Conversion of Oleaginous Microalgae into Renewable Hydrocarbons. Bioresour. Technol. 2014, 158, 91–97. [Google Scholar] [CrossRef]
- OCED. OECD-FAO Agricultural Outlook 2018–2027; OECD Publishing: Paris, France, 2018. [Google Scholar]
- Monteiro, M.R.; Kugelmeier, C.L.; Pinheiro, R.S.; Batalha, M.O.; Da Silva César, A. Glycerol fom Biodiesel Production: Technological Paths for Sustainability. Renew. Sustain. Energy Rev. 2018, 88, 109–122. [Google Scholar] [CrossRef]
- França, R.G.; Souza, P.A.; Lima, E.R.; Costa, A.L. An Extended Techno-Economic Analysis of the Utilization of Glycerol as An Alternative Feedstock for Methanol Production. Clean Technol. Environ. Policy 2017, 19, 1855–1865. [Google Scholar] [CrossRef]
- Ardi, M.S.; Aroua, M.K.; Hashim, N.A. Progress, Prospect and Challenges in Glycerol Purification Process: A Review. Renew. Sustain. Energy Rev. 2015, 42, 1164–1173. [Google Scholar] [CrossRef]
- Ciriminna, R.; Pina, C.D.; Rossi, M.; Pagliaro, M. Understanding the Glycerol Market. Eur. J. Lipid Sci. Technol. 2014, 116, 1432–1439. [Google Scholar] [CrossRef]
- Bartoli, M.; Rosi, L.; Frediani, M. Introductory Chapter: A Brief Insight about Glycerol. In Glycerine Production And Transformation—An Innovative Platform For Sustainable Biorefinery And Energy; Intechopen: London, UK, 2019. [Google Scholar]
- Dunphy, P.J.; Meyers, A.J.; Rigg, R.T. Cosmetic Water-In-Oil Emulsion Lipstick Comprising A Phospholipid and Glycerol Fatty Acid Esters Emulsifying System. U.S. Patent 5,085,856, 2 April 1992. [Google Scholar]
- Suh, Y.; Kil, D.; Chung, K.; Abdullayev, E.; Lvov, Y.; Mongayt, D. Natural Nanocontainer for the Controlled Delivery of Glycerol as A Moisturizing Agent. J. Nanosci. Nanotechnol. 2011, 11, 661–665. [Google Scholar] [CrossRef] [PubMed]
- Tamarkin, D.; Besonov, A. Glycerol Ethers Vehicle and Pharmaceutical Compositions Thereof. U.S. Patent 20090130029A1, 21 May 2009. [Google Scholar]
- Van Cleef, E.H.C.B.; Ezequiel, J.M.B.; D’aurea, A.P.; Fávaro, V.R.; Sancanari, J.B.D. Crude Glycerin in Diets for Feedlot Nellore Cattle. Rev. Bras. Zootec. 2014, 43, 86–91. [Google Scholar] [CrossRef]
- Meale, S.; Chaves, A.; Ding, S.; Bush, R.; Mcallister, T. Effects of Crude Glycerin Supplementation on Wool Production, Feeding Behavior, and Body Condition of Merino Ewes. J. Anim. Sci. 2013, 91, 878–885. [Google Scholar] [CrossRef] [PubMed]
- Razali, N.; Abdullah, A.Z. Production of Lactic Acid from Glycerol Via Chemical Conversion Using Solid Catalyst: A Review. Appl. Catal. A Gen. 2017, 543, 234–246. [Google Scholar] [CrossRef]
- Oberhauser, W.; Evangelisti, C.; Jumde, R.P.; Psaro, R.; Vizza, F.; Bevilacqua, M.; Filippi, J.; Machado, B.F.; Serp, P. Platinum on Carbonaceous Supports for Glycerol Hydrogenolysis: Support Effect. J. Catal. 2015, 325, 111–117. [Google Scholar] [CrossRef]
- Magar, S.; Kamble, S.; Mohanraj, G.T.; Jana, S.K.; Rode, C. Solid-Acid-Catalyzed Etherification of Glycerol to Potential Fuel Additives. Energy Fuels 2017, 31, 12272–12277. [Google Scholar] [CrossRef]
- Shukla, K.; Srivastava, V.C. Synthesis of Organic Carbonates from Alcoholysis of Urea: A Review. Catal. Rev. 2017, 59, 1–43. [Google Scholar] [CrossRef]
- Oliverio, M.; Costanzo, P.; Nardi, M.; Calandruccio, C.; Salerno, R.; Procopio, A. Tunable Microwave-Assisted Method for the Solvent-Free and Catalyst-Free Peracetylation of Natural Products. Beilstein J. Org. Chem. 2016, 12, 2222–2233. [Google Scholar] [CrossRef]
- Bartoli, M.; Zhu, C.; Chae, M.; Bressler, D. Value-Added Products from Urea Glycerolysis Using A Heterogeneous Biosolids-Based Catalyst. Catalysts 2018, 8, 373. [Google Scholar] [CrossRef]
- Amaral, P.F.F.; Ferreira, T.F.; Fontes, G.C.; Coelho, M.A.Z. Glycerol Valorization: New Biotechnological Routes. Food Bioprod. Process. 2009, 87, 179–186. [Google Scholar] [CrossRef]
- Costanzo, P.; Calandruccio, C.; Di Gioia, M.L.; Nardi, M.; Oliverio, M.; Procopio, A. First Multicomponent Reaction Exploiting Glycerol Carbonate Synthesis. J. Clean. Prod. 2018, 202, 504–509. [Google Scholar] [CrossRef]
- Mufrodi, Z.; Rochmadi, R.; Sutijan, S.; Budiman, A. Synthesis Acetylation of Glycerol Using Batch Reactor and Continuous Reactive Distillation Column. Eng. J. 2014, 18, 29–40. [Google Scholar] [CrossRef]
- Rastegari, H.; Ghaziaskar, H.S. From Glycerol as the by-Product Of Biodiesel Production to Value-Added Monoacetin by Continuous and Selective Esterification in Acetic Acid. J. Ind. Eng. Chem. 2015, 21, 856–861. [Google Scholar] [CrossRef]
- Odibi, C.; Babaie, M.; Zare, A.; Nabi, M.N.; Bodisco, T.A.; Brown, R.J. Exergy Analysis of A Diesel Engine with Waste Cooking Biodiesel and Triacetin. Energy Convers. Manag. 2019, 198. [Google Scholar] [CrossRef]
- Nda-Umar, U.I.; Ramli, I.; Taufiq-Yap, Y.H.; Muhamad, E.N. An Overview of Recent Research in the Conversion of Glycerol into Biofuels, Fuel Additives and Other Bio-Based Chemicals. Catalysts 2019, 9, 15. [Google Scholar] [CrossRef]
- Gama, N.; Santos, R.; Godinho, B.; Silva, R.; Ferreira, A. Triacetin as A Secondary Pvc Plasticizer. J. Polym. Environ. 2019, 27, 1294–1301. [Google Scholar] [CrossRef]
- Okoye, P.; Abdullah, A.; Hameed, B. A Review on Recent Developments and Progress in the Kinetics and Deactivation of Catalytic Acetylation of Glycerol—A Byproduct of Biodiesel. Renew. Sustain. Energy Rev. 2017, 74, 387–401. [Google Scholar] [CrossRef]
- Liao, X.; Zhu, Y.; Wang, S.-G.; Chen, H.; Li, Y. Theoretical Elucidation of Acetylating Glycerol with Acetic Acid and Acetic Anhydride. Appl. Catal. B Environ. 2010, 94, 64–70. [Google Scholar] [CrossRef]
- Melero, J.A.; Van Grieken, R.; Morales, G.; Paniagua, M. Acidic Mesoporous Silica for the Acetylation of Glycerol: Synthesis of Bioadditives to Petrol Fuel. Energy Fuels 2007, 21, 1782–1791. [Google Scholar] [CrossRef]
- Herrada-Vidales, J.A.; García-González, J.M.; Martínez-Palou, R.; Guzmán-Pantoja, J. Integral Process for Obtaining Acetins from Crude Glycerol and Their Effect on the Octane Index. Chem. Eng. Commun. 2019, 207, 1–11. [Google Scholar] [CrossRef]
- Gorji, Y.M.; Ghaziaskar, H.S. Optimization of Solketalacetin Synthesis as A Green Fuel Additive from Ketalization of Monoacetin with Acetone. Ind. Eng. Chem. Res. 2016, 55, 6904–6910. [Google Scholar] [CrossRef]
- Lachman, L.; Drubulis, A. Factors Influencing the Properties of Films Used for Tablet Coating I. Effects of Plasticizers on the Water Vapor Transmission Of Cellulose Acetate Phthalate Films. J. Pharm. Sci. 1964, 53, 639–643. [Google Scholar] [CrossRef] [PubMed]
- Weber, F. Pharmaceutical Composition. U.S. Patent US7074412B2, 11 July 2006. [Google Scholar]
- Knowles, M.; Pridgen, H.S. Solvents for Fat and Oil Antioxidants. U.S. Patent 2944908A, 12 July 1960. [Google Scholar]
- Deline, G.D. Homogeneous, Free-Flowing Liquid Black Pepper Oleoresin Composition. European Patent 0137082A1, 11 October 1983. [Google Scholar]
- Dosuna-Rodríguez, I.; Gaigneaux, E.M. Glycerol Acetylation Catalysed by Ion Exchange Resins. Catal. Today 2012, 195, 14–21. [Google Scholar] [CrossRef]
- Caballero, K.V.; Guerrero-Amaya, H.; Baldovino-Medrano, V.G. Revisiting Glycerol Esterification with Acetic Acid over Amberlyst-35 Via Statistically Designed Experiments: Overcoming Transport Limitations. Chem. Eng. Sci. 2019. [Google Scholar] [CrossRef]
- De Abreu Dessimoni, A.L.; De Oliveira Pereira, L.; Penido, E.S.; Lima Abreu Veiga, T.R.; De Barros Fernandes, R.V.; Teixeira, M.L.; Bonésio, M.D.R.; Bianchi, M.L. Characterization of Catalysts for Glycerol Ester Production with Various Acetylating Agents. Anal. Lett. 2017. [Google Scholar] [CrossRef]
- Gonçalves, V.L.C.; Pinto, B.P.; Silva, J.C.; Mota, C.J.A. Acetylation of Glycerol Catalyzed by Different Solid Acids. Catal. Today 2008, 133–135, 673–677. [Google Scholar]
- Dill, L.P.; Kochepka, D.M.; Melinski, A.; Wypych, F.; Cordeiro, C.S. Microwave-Irradiated Acetylation of Glycerol Catalyzed by Acid Activated Clays. React. Kinet. Mech. Catal. 2019, 127, 1–14. [Google Scholar] [CrossRef]
- Okoye, P.; Abdullah, A.; Hameed, B. Synthesis of Oxygenated Fuel Additives Via Glycerol Esterification with Acetic Acid over Bio-Derived Carbon Catalyst. Fuel 2017, 209, 538–544. [Google Scholar] [CrossRef]
- Okoye, P.U.; Hameed, B.H. Review On Recent Progress in Catalytic Carboxylation and Acetylation of Glycerol as A Byproduct of Biodiesel Production. Renew. Sustain. Energy Rev. 2016, 53, 558–574. [Google Scholar] [CrossRef]
- Silva, L.N.; Gonçalves, V.L.C.; Mota, C.J.A. Catalytic Acetylation of Glycerol with Acetic Anhydride. Catal. Commun. 2010, 11, 1036–1039. [Google Scholar] [CrossRef]
- Cunningham, J.K.; Liu, L.-M.; Callaghan, R.C. Essential (“Precursor”) Chemical Control for Heroin: Impact of Acetic Anhydride Regulation on Us Heroin Availability. Drug Alcohol Depend. 2013, 133, 520–528. [Google Scholar] [CrossRef] [PubMed]
- Len, C.; Luque, R. Continuous Flow Transformations of Glycerol to Valuable Products: An Overview. Sustain. Chem. Process. 2014, 2, 1. [Google Scholar] [CrossRef]
- Mufrodi, Z.; Budiman, A. Continuous Process of Reactive Distillation to Produce Bio-Additive Triacetin from Glycerol. Mod. Appl. Sci. 2013, 7, 70. [Google Scholar] [CrossRef][Green Version]
- Costa, I.C.; Itabaiana, I., Jr.; Flores, M.C.; Lourenço, A.C.; Leite, S.G.; De, M.E.; Miranda, L.S.; Leal, I.C.; De Souza, R.O. Biocatalyzed Acetins Production under Continuous-Flow Conditions: Valorization of Glycerol Derived from Biodiesel Industry. J. Flow Chem. 2013, 3, 41–45. [Google Scholar] [CrossRef]
- Varma, R.S.; Len, C. Glycerol Valorization under Continuous Flow Conditions-Recent Advances. Curr. Opin. Green Sustain. Chem. 2018, 15, 83–90. [Google Scholar] [CrossRef]
- Chae, M.; Xia, L.; Zhu, C.; Bressler, D.C. Accelerating Settling Rates of Biosolids Lagoons through Thermal Hydrolysis. J. Environ. Manag. 2018, 220, 227–232. [Google Scholar] [CrossRef]
- Zhou, L.; Nguyen, T.-H.; Adesina, A.A. The Acetylation of Glycerol over Amberlyst-15: Kinetic and Product Distribution. Fuel Process. Technol. 2012, 104, 310–318. [Google Scholar] [CrossRef]
- Patel, A.; Singh, S. A Green and Sustainable Approach for Esterification of Glycerol Using 12-Tungstophosphoric Acid Anchored to Different Supports: Kinetics and Effect of Support. Fuel 2014, 118, 358–364. [Google Scholar] [CrossRef]
- Reddy, P.S.; Sudarsanam, P.; Raju, G.; Reddy, B.M. Selective Acetylation of Glycerol over Ceo2–M and So42−/Ceo2–M (M = Zro2 And Al2o3) Catalysts for Synthesis of Bioadditives. J. Ind. Eng. Chem. 2012, 18, 648–654. [Google Scholar] [CrossRef]
- Arizona, U.O. Rruff™ Project. Available online: Http://Rruff.Info/ (accessed on 21 October 2019).
- Jaeger, A.O.; Selden Co, Assignee. Catalytic Esterification. U.S. Patent 1,819,818, 18 August 1931. [Google Scholar]
- Greenwood, N.N.; Earnshaw, A. Chemistry of the Elements; Elsevier: Amsterdam, The Netherlands, 2012. [Google Scholar]
- Keogh, J.; Tiwari, M.S.; Manyar, H. Esterification of Glycerol with Acetic Acid Using Nitrogen-Based Brønsted-Acidic Ionic Liquids. Ind. Eng. Chem. Res. 2019, 58, 17235–17243. [Google Scholar] [CrossRef]
- Testa, M.L.; La Parola, V.; Mesrar, F.; Ouanji, F.; Kacimi, M.; Ziyad, M.; Liotta, L.F. Use of Zirconium Phosphate-Sulphate as Acid Catalyst for Synthesis of Glycerol-Based Fuel Additives. Catalysts 2019, 9, 148. [Google Scholar] [CrossRef]
- Liu, J.; Wang, Z.; Sun, Y.; Jian, R.; Jian, P.; Wang, D. Selective Synthesis of Triacetin fom Glycerol Catalyzed by Hzsm-5/Mcm-41 Micro/Mesoporous Molecular Sieve. Chin. J. Chem. Eng. 2019, 27, 1073–1078. [Google Scholar] [CrossRef]
- Bandyopadhyay, M.; Tsunoji, N.; Bandyopadhyay, R.; Sano, T. Comparison of Sulfonic Acid Loaded Mesoporous Silica in Transesterification of Triacetin. React. Kinet. Mech. Catal. 2019, 126, 167–179. [Google Scholar] [CrossRef]
- Marwan, M.; Indarti, E.; Darmadi, D.; Rinaldi, W.; Hamzah, D.; Rinaldi, T. Production of Triacetin by Microwave Assisted Esterification of Glycerol Using Activated Natural Zeolite. Bull. Chem. React. Eng. Catal. 2019, 14, 672. [Google Scholar] [CrossRef]
- Huang, M.-Y.; Han, X.-X.; Hung, C.-T.; Lin, J.-C.; Wu, P.-H.; Wu, J.-C.; Liu, S.-B. Heteropolyacid-Based Ionic Liquids as Efficient Homogeneous Catalysts for Acetylation of Glycerol. J. Catal. 2014, 320, 42–51. [Google Scholar] [CrossRef]
- Omidghane, M.; Jenab, E.; Chae, M.; Bressler, D.C. Production of renewable hydrocarbons by thermal cracking of oleic acid in the presence of water. Energ. Fuel. 2017, 31, 9446–9454. [Google Scholar] [CrossRef]
- Boehm, H. Acidic and Basic Properties of Hydroxylated Metal Oxide Surfaces. Discuss. Faraday Soc. 1971, 52, 264–275. [Google Scholar] [CrossRef]


| Catalyst | Concentration (mg/g) | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Cr | Fe | Mn | Sr | Al | Cu | Zn | Pb | Ti | Ref. | |
| Initial | 0.4 ± 0.2 | 26.5 ± 1.3 | 0.58 ± 0.03 | 0.4 ± 0.1 | 26.9 ± 1.3 | 0.7 ± 0.1 | 1.1 ± 0.1 | 0.1 ± 0.1 | 3.2 ± 0.2 | [23] |
| After 5 reactions | 0.4 ± 0.2 | 28.7 ± 1.7 | 0.36 ± 0.02 | 0.4 ± 0.1 | 25.2 ± 1.6 | 0.2 ± 0.01 | 0.7 ± 0.1 | 0.2 ± 0.1 | 0.1 ± 0.1 | Current study |
| T [h] | T [°C] | Conversion a (%) | Selectivity b (%) | ||
|---|---|---|---|---|---|
| Monoacetins | Diacetins | Triacetin | |||
| 1 | 60 | 8.3 ± 1.7 | >99 | Not detected | Not detected |
| 2 | 20.0 ± 3.4 | >99 | Not detected | Not detected | |
| 3 | 26.8 ± 0.5 | >99 | Not detected | Not detected | |
| 4 | 32.1 ± 1.6 | 88.4 ± 0.8 | 11.4 ± 0.9 | Not detected | |
| 1 | 80 | 8.9 ± 1.3 | 95.7 ± 0.6 | 3.6 ± 0.3 | 0.7 ± 0.2 |
| 2 | 22.8 ± 1.2 | 93.7 ± 0.5 | 6.1 ± 0.5 | 0.5 ± 0.2 | |
| 3 | 29.3 ± 0.8 | 91.0 ± 0.7 | 8.6 ± 0.4 | 0.5 ± 0.3 | |
| 4 | 41.1 ± 2.5 | 87.5 ± 1.0 | 11.3 ± 0.6 | 1.2 ± 0.4 | |
| 1 | 100 | 8.9 ± 0.7 | 86.6 ± 0.7 | 12.5 ± 0.2 | 0.8 ± 0.2 |
| 2 | 37.3 ± 1.2 | 73.9 ± 0.3 | 25.3 ± 0.6 | 0.9 ± 0.3 | |
| 3 | 51.1 ± 0.7 | 62.7 ± 0.9 | 33.5 ± 0.3 | 3.8 ± 0.9 | |
| 4 | 63.2 ± 1.5 | 53.3 ± 0.7 | 41.3 ± 0.4 | 5.4 ± 0.8 | |
| 1 | 120 | 27.7 ± 1.2 | 70.9 ± 1.9 | 23.9 ± 2.0 | 1.5 ± 0.1 |
| 2 | 51.1 ± 1.1 | 64.7 ± 0.5 | 33.1 ± 1.7 | 2.0 ± 0.7 | |
| 3 | 66.1 ± 0.3 | 54.2 ± 1.4 | 40.8 ± 3.0 | 4.6 ± 0.1 | |
| 4 | 78.4 ± 0.2 | 45.5 ± 1.4 | 46.6 ± 1.5 | 6.6 ± 0.8 | |
| T [h] | Acetic Acid: Glycerol (mol/mol) | Conversion a (%) | Selectivity b (%) | ||
|---|---|---|---|---|---|
| Monoacetins | Diacetins | Triacetin | |||
| 1 | 1 | 26.0 ± 0.5 | 74.3 ± 3.2 | 24.5 ± 1.8 | 1.2 ± 0.3 |
| 2 | 35.8 ± 0.3 | 59.7 ± 2.6 | 34.6 ± 1.1 | 4.1 ± 0.9 | |
| 3 | 36.5 ± 0.5 | 51.7 ± 0.9 | 42.5 ± 2.6 | 5.5 ± 0.5 | |
| 4 | 40.8 ± 0.7 | 43.8 ± 2.0 | 49.0 ± 2.8 | 6.3 ± 0.1 | |
| 1 | 3 | 26.6 ± 0.3 | 70.9 ± 1.9 | 23.9 ± 2.0 | 1.5 ± 0.1 |
| 2 | 45.3 ± 1.9 | 64.7 ± 0.5 | 33.1 ± 1.7 | 2.0 ± 0.7 | |
| 3 | 63.5 ± 0.6 | 54.2 ± 1.4 | 40.8 ± 3.0 | 4.6 ± 0.1 | |
| 4 | 73.1 ± 0.7 | 45.5 ± 1.4 | 46.6 ± 1.5 | 6.6 ± 0.8 | |
| 1 | 6 | 27.7 ± 0.7 | 70.9 ± 1.9 | 23.9 ± 2.0 | 1.5 ± 0.1 |
| 2 | 51.1 ± 1.1 | 64.7 ± 0.5 | 33.1 ± 1.7 | 2.0 ± 0.7 | |
| 3 | 66.1 ± 0.3 | 54.2 ± 1.4 | 40.8 ± 3.0 | 4.6 ± 0.1 | |
| 4 | 78.4 ± 0.2 | 45.5 ± 1.4 | 46.6 ± 1.5 | 6.6 ± 0.8 | |
| 1 | 9 | 28.1 ± 0.9 | 80.4 ± 3.8 | 17.0 ± 0.5 | 0.9 ± 0.1 |
| 2 | 65.6 ± 1.2 | 62.1 ± 2.6 | 34.2 ± 1.5 | 2.4 ± 0.4 | |
| 3 | 75.9 ± 0.5 | 49.7 ± 2.1 | 44.1 ± 0.7 | 3.9 ± 0.3 | |
| 4 | 85.8 ± 1.2 | 46.9 ± 2.0 | 48.4 ± 0.3 | 6.3 ± 0.4 | |
| T [h] | Catalyst [wt%] | Conversion a (%) | Selectivity b (%) | ||
|---|---|---|---|---|---|
| Monoacetins | Diacetins | Triacetin | |||
| 1 | 0 | 3.6 ± 0.4 | 65.2 ± 2.6 | 34.5 ± 2.6 | Not detected |
| 2 | 21.5 ± 0.9 | 56.6 ± 1.2 | 42.9 ± 2.8 | Not detected | |
| 3 | 35.5 ± 0.9 | 49.0 ± 3.7 | 52.3 ± 1.9 | Not detected | |
| 4 | 65.7 ± 0.4 | 44.4 ± 1.0 | 55.8 ± 2.6 | Not detected | |
| 1 | 2 | 22.6 ± 0.3 | 74.3 ± 3.2 | 24.5 ± 3.0 | 1.2 ± 0.3 |
| 2 | 47.0 ± 2.0 | 59.7 ± 2.6 | 34.6 ± 1.5 | 4.1 ± 0.9 | |
| 3 | 60.7 ± 0.3 | 51.7 ± 0.9 | 42.5 ± 1.8 | 5.5 ± 0.5 | |
| 4 | 72.2 ± 1.5 | 43.8 ± 2.0 | 49.0 ± 1.1 | 6.3 ± 0.1 | |
| 1 | 4 | 27.7 ± 1.2 | 70.9 ± 1.9 | 23.9 ± 2.0 | 1.5 ± 0.1 |
| 2 | 51.1 ± 1.1 | 64.7 ± 0.5 | 33.1 ± 1.7 | 2.0 ± 0.7 | |
| 3 | 66.1 ± 0.3 | 54.2 ± 1.4 | 40.8 ± 3.0 | 4.6 ± 0.1 | |
| 4 | 78.4 ± 0.2 | 45.5 ± 1.4 | 46.6 ± 1.5 | 6.6 ± 0.8 | |
| 1 | 8 | 47.5 ± 2.2 | 78.6 ± 2.2 | 20.3 ± 2.3 | 1.0 ± 0.3 |
| 2 | 64.8 ± 3.1 | 65.2 ± 2.5 | 33.0 ± 1.9 | 2.5 ± 0.4 | |
| 3 | 82.2 ± 1.3 | 54.1 ± 1.2 | 41.4 ± 1.1 | 4.6 ± 0.4 | |
| 4 | 86.0 ± 0.5 | 46.1 ± 1.3 | 47.2 ± 0.3 | 6.8 ± 0.7 | |
| 1 | 16 | 54.9 ± 1.1 | 80.4 ± 3.8 | 17.0 ± 0.5 | 0.9 ± 0.1 |
| 2 | 69.3 ± 1.6 | 62.1 ± 2.6 | 34.2 ± 1.5 | 2.4 ± 0.4 | |
| 3 | 84.6 ± 1.3 | 49.7 ± 2.1 | 44.1 ± 0.7 | 3.9 ± 0.3 | |
| 4 | 90.8 ± 2.3 | 46.9 ± 2.0 | 48.4 ± 0.3 | 6.3 ± 0.4 | |
| Catalytic Run | Conversion a (%) | Selectivity b (%) | ||
|---|---|---|---|---|
| Monoacetins | Diacetins | Triacetin | ||
| 1st | 78.4 ± 0.2 | 45.5 ± 1.4 | 46.6 ± 1.5 | 6.6 ± 0.8 |
| 2nd | 77.9 ± 0.9 | 44.9 ± 0.5 | 45.9 ± 1.1 | 6.3 ± 0.6 |
| 3rd | 77.9 ± 0.4 | 44.6 ± 0.3 | 46.3 ± 1.3 | 6.7 ± 0.4 |
| 4th | 78.1 ± 0.2 | 46.6 ± 0.8 | 45.8 ± 0.8 | 6.0 ± 0.9 |
| 5th | 78.1 ± 0.3 | 42.0 ± 0.7 | 46.2 ± 1.1 | 6.9 ± 0.5 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bartoli, M.; Zhu, C.; Chae, M.; Bressler, D.C. Glycerol Acetylation Mediated by Thermally Hydrolysed Biosolids-Based Material. Catalysts 2020, 10, 5. https://doi.org/10.3390/catal10010005
Bartoli M, Zhu C, Chae M, Bressler DC. Glycerol Acetylation Mediated by Thermally Hydrolysed Biosolids-Based Material. Catalysts. 2020; 10(1):5. https://doi.org/10.3390/catal10010005
Chicago/Turabian StyleBartoli, Mattia, Chengyong Zhu, Michael Chae, and David C. Bressler. 2020. "Glycerol Acetylation Mediated by Thermally Hydrolysed Biosolids-Based Material" Catalysts 10, no. 1: 5. https://doi.org/10.3390/catal10010005
APA StyleBartoli, M., Zhu, C., Chae, M., & Bressler, D. C. (2020). Glycerol Acetylation Mediated by Thermally Hydrolysed Biosolids-Based Material. Catalysts, 10(1), 5. https://doi.org/10.3390/catal10010005






