Potential Cycling Effects on Activities of Nickel-Mediated Benzyl Alcohol and Glycine Electrooxidation in Alkaline Solutions
Abstract
1. Introduction
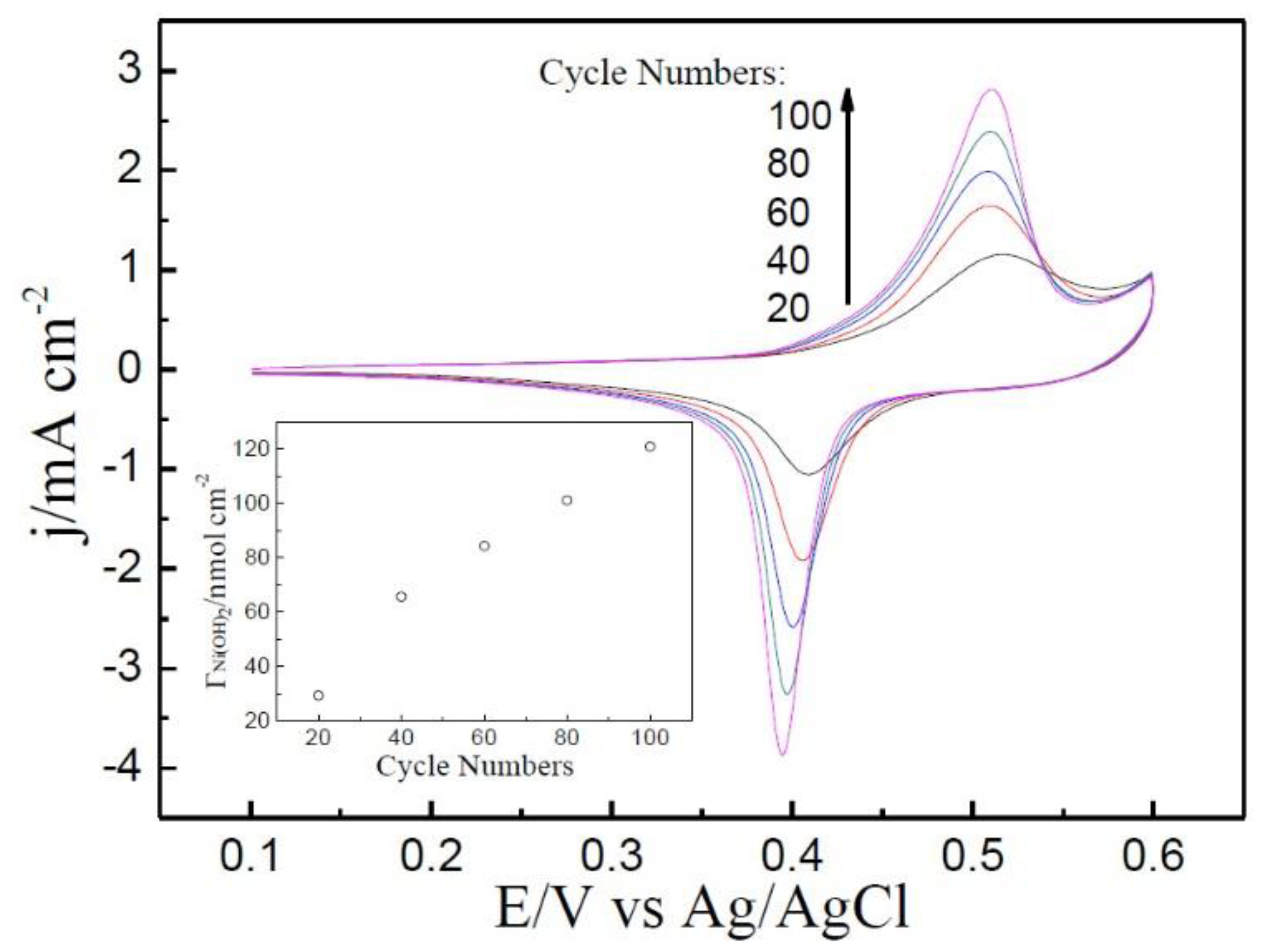
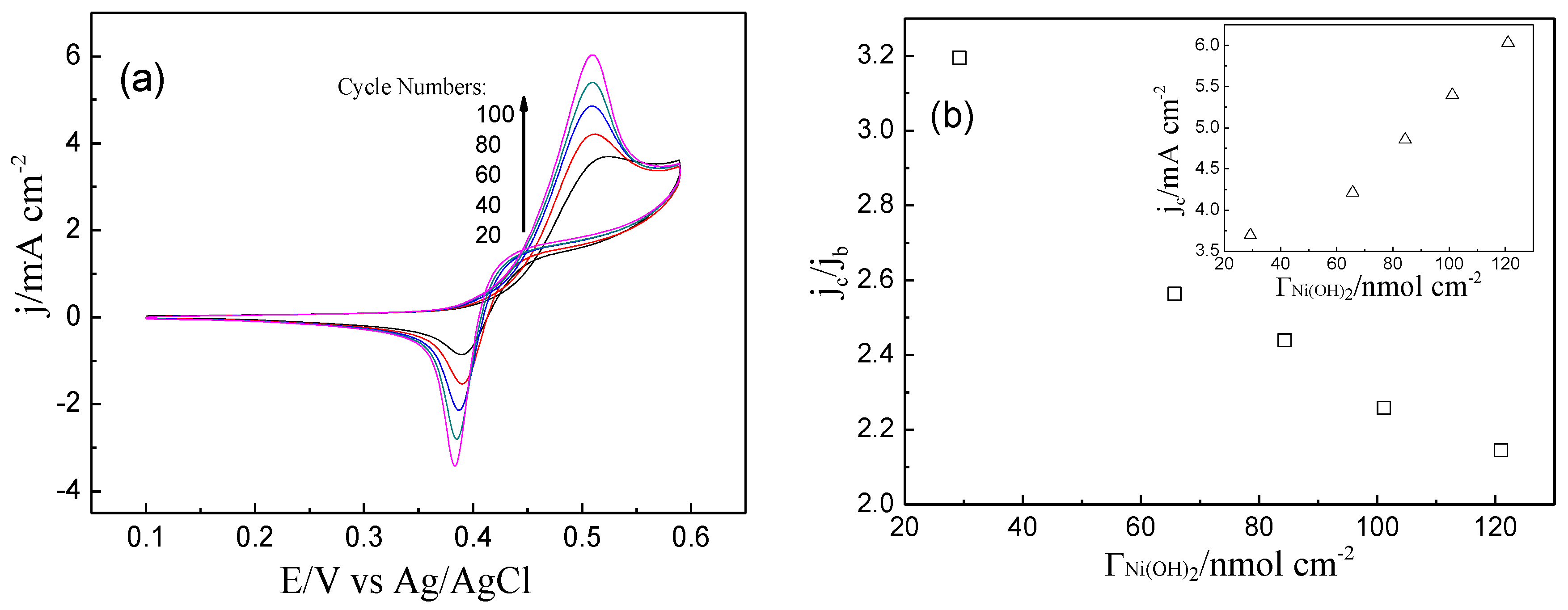
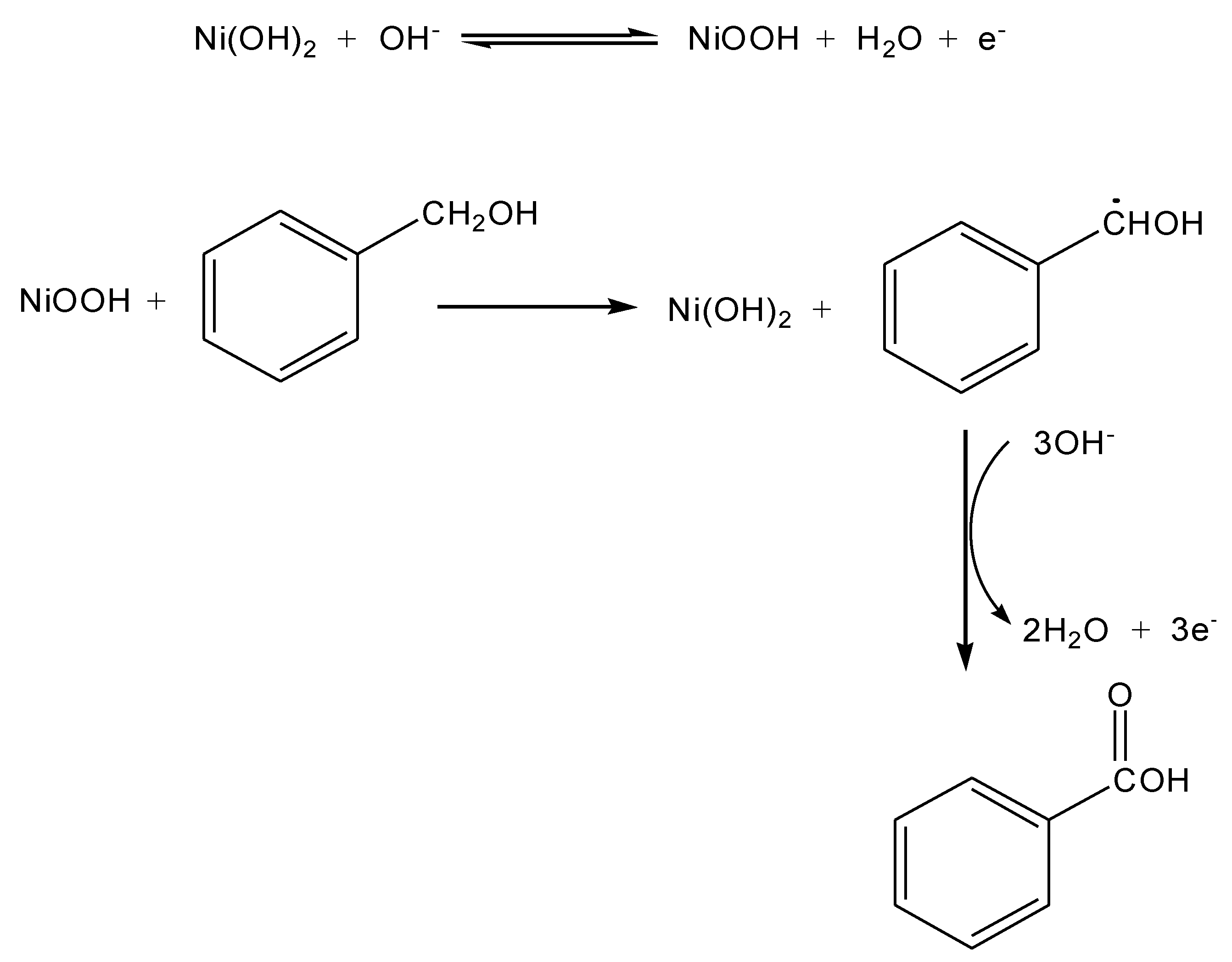
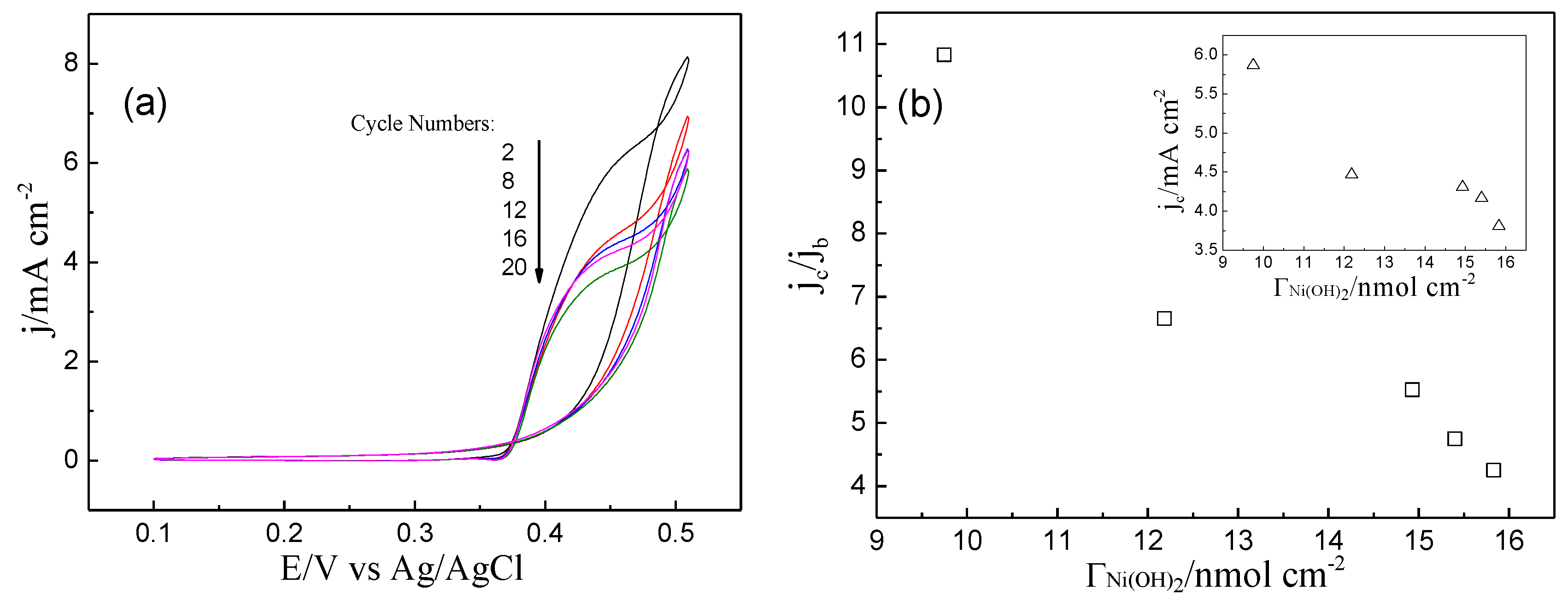
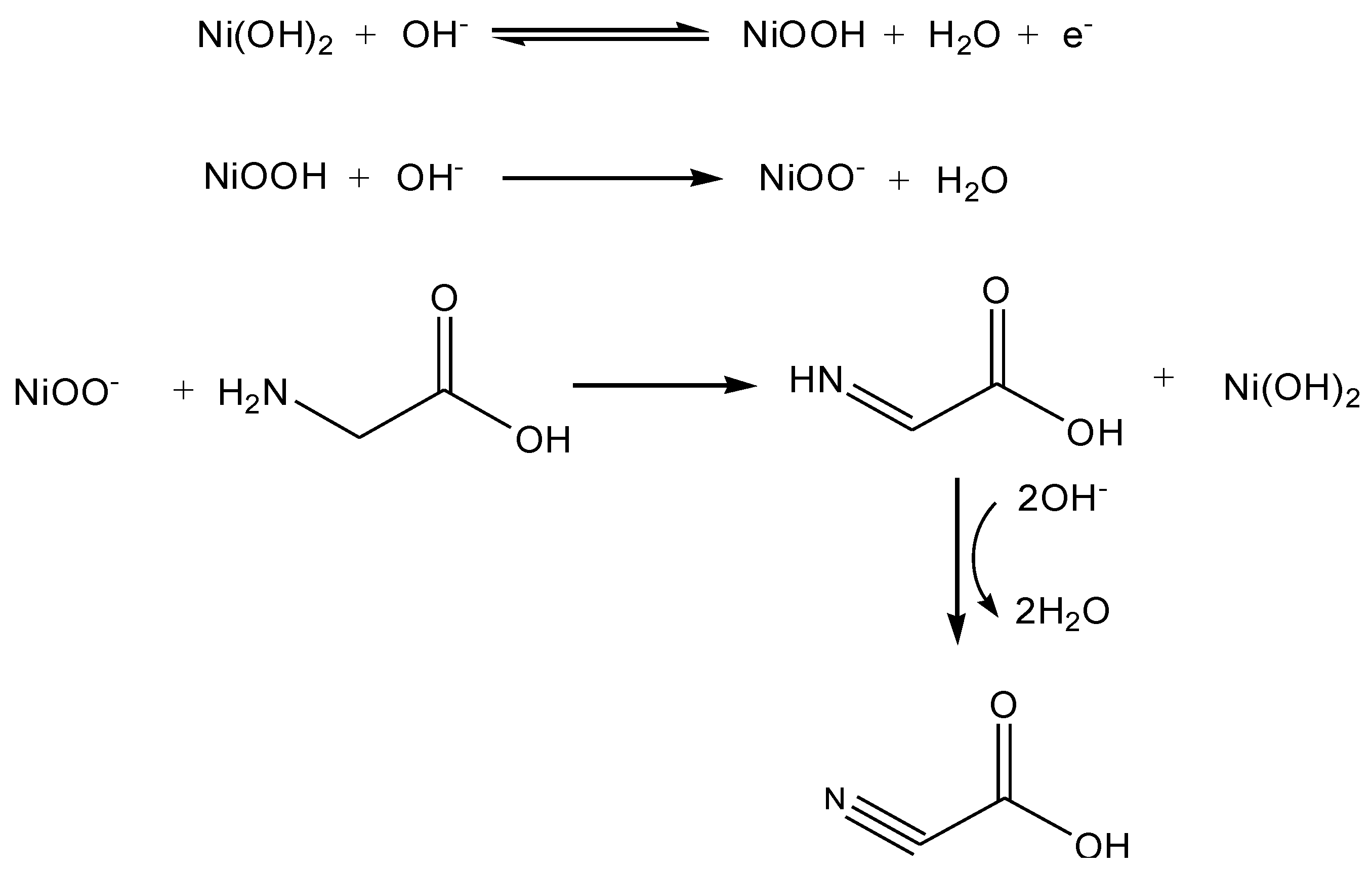
2. Results and Discussion
3. Materials and Methods
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Vij, V.; Sultan, S.; Harzandi, A.M.; Meena, A.; Tiwari, J.N.; Lee, W.-G.; Yoon, T.; Kim, K.S. Nickel-Based Electrocatalysts for Energy-Related Applications: Oxygen Reduction, Oxygen Evolution, and Hydrogen Evolution Reactions. ACS Catal. 2017, 7, 7196–7225. [Google Scholar] [CrossRef]
- Miao, Y.; Ouyang, L.; Zhou, S.; Xu, L.; Yang, Z.; Xiao, M.; Ouyang, R. Electrocatalysis and electroanalysis of nickel, its oxides, hydroxides and oxyhydroxides toward small molecules. Biosens. Bioelectron. 2014, 53, 428–439. [Google Scholar] [CrossRef] [PubMed]
- Gong, M.; Dai, H. A mini review of NiFe-based materials as highly active oxygen evolution reaction electrocatalysts. Nano Res. 2015, 8, 23–39. [Google Scholar] [CrossRef]
- Louie, M.W.; Bell, A.T. An investigation of thin-film Ni-Fe oxide catalysts for the electrochemical evolution of oxygen. J. Am. Chem. Soc. 2013, 135, 12329–12337. [Google Scholar] [CrossRef] [PubMed]
- Goerlin, M.; Araújo, J.; Schmies, H.; Bernsmeier, D.; Dresp, S.; Gliech, M.; Jusys, Z.; Chernev, P.; Kraehnert, R.; Dau, H.; et al. Tracking catalyst redox states and reaction dynamics in Ni-Fe oxyhydroxide oxygen evolution reaction (OER) electrocatalysts: The role of catalyst support and electrolyte pH. J. Am. Chem. Soc. 2017, 139, 2070–2082. [Google Scholar] [CrossRef] [PubMed]
- Fleischmann, M.; Korinek, K.; Pletcher, D. The oxidation of organic compounds at a nickel anode in alkaline solution. J. Electroanal. Chem. Interfacial Electrochem. 1971, 31, 39–49. [Google Scholar] [CrossRef]
- Fleischmann, M.; Korinek, K.; Pletcher, D. The kinetics and mechanism of the oxidation of amines and alcohols at oxide-covered nickel, silver, copper, and cobalt electrodes. J. Chem. Soc. Perkin Trans. 1972, 2, 1396–1403. [Google Scholar] [CrossRef]
- Motheo, A.J.; Tremiliosi-filho, G.; Gonzalez, E.R.; Kokoh, K.B.; Le’ger, J.M.; Lamy, C. Electrooxidation of benzyl alcohol and benzaldehyde on a nickel oxy-hydroxide electrode in a filter-press type cell. J. Appl. Electrochem. 2006, 36, 1035–1061. [Google Scholar] [CrossRef]
- Ureta-Zañartu, M.S.; González, T.; Fernández, F.; Báez, D.; Berríos, C.; Gutiérrez, C. Electro-Oxidation of Benzyl and Aliphatic Alcohols on polyNiTSPc- and Ni(OH)2-Modified Glassy-Carbon and Gold Electrodes. Int. J. Electrochem. Sci. 2012, 7, 8794–8812. [Google Scholar]
- Vidotti, M.; Córdoba deTorresi, S.I.; Kubota, L.T. Electrochemical oxidation of glycine by doped nickel hydroxide modified electrode. Sens. Actuators B Chem. 2008, 135, 245–249. [Google Scholar] [CrossRef]
- Mohan, R.; Modak, A.; Schechter, A.; Cahan, R.; Gedanken, A.; Subramanian, P.; Sivakumar, P. Electrochemical oxidation of glycine by bimetallic nickel-manganese oxide catalysts. ChemElectroChem 2019. [Google Scholar] [CrossRef]
- Zhu, X.; Dou, X.; Dai, J.; An, X.; Guo, Y.; Zhang, L.; Tao, S.; Zhao, J.Y.; Chu, W.S.; Cheng, Z.; et al. Metallic Nickel Hydroxide Nanosheets Give Superior Electrocatalytic Oxidation of Urea for Fuel Cells. Angew. Chem. Int. Ed. 2016, 55, 12465–12469. [Google Scholar] [CrossRef] [PubMed]
- Boggs, B.K.; King, R.L.; Botte, G.G. Urea electrolysis: Direct hydrogen production from urine. Chem. Commun. 2009, 32, 4859–4861. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Yan, W.; Vijapur, S.H.; Botte, G.G. Enhanced electrocatalytic oxidation of urea based on nickel hydroxide nanoribbons. J. Power Sources 2012, 217, 498–502. [Google Scholar] [CrossRef]
- Raoof, J.B.; Ojani, R.; Abdi, S.; Hosseini, S.R. Highly improved electrooxidation of formaldehyde on nickel/poly (o-toluidine)/Triton X-100 film modified carbon nanotube paste electrode. Int. J. Hydrogen Energy 2012, 37, 2137–2146. [Google Scholar] [CrossRef]
- Giovanelli, D.; Lawrence, N.S.; Jiang, L.; Jones, T.; Compton, R.G. Electrochemical determination of sulphide at nickel electrodes in alkaline media: A new electrochemical sensor. Sens. Actuators B Chem. 2003, 88, 320–328. [Google Scholar] [CrossRef]
- Marshall, A.T.; Golovko, V.; Padayachee, D. Influence of gold nanoparticle loading in Au/C on the activity towards electrocatalytic glycerol oxidation. Electrochim. Acta 2015, 153, 370–378. [Google Scholar] [CrossRef]
- Quiñonero, J.; Gómez, R. Controlling the amount of co-catalyst as a critical factor in determining the efficiency of photoelectrodes: The case of nickel (II) hydroxide on vanadate photoanodes. Appl. Catal. B Environ. 2017, 217, 437–447. [Google Scholar] [CrossRef]
- Chen, Z.; Jaramillo, T.F.; Deutsch, T.G.; Kleiman-Shwarsctein, A.; Forman, A.; Gaillard, N.; Garland, R.; Takanabe, K.; Heske, C.; Sunkara, M.; et al. Accelerating materials development for photoelectrochemical hydrogen production: Standards for methods, definitions, and reporting protocols. J. Mater. Res. 2010, 25, 3–16. [Google Scholar] [CrossRef]
- Hoque, M.A.; Guzman, M.I. Photocatalytic Activity: Experimental Features to Report in Heterogeneous Photocatalysis. Materials 2018, 11, 1990. [Google Scholar] [CrossRef]
- Batchellor, A.S.; Boettcher, S.W. Pulse-Electrodeposited Ni-Fe (Oxy)hydroxide Oxygen Evolution Electrocatalysts with High Geometric and Intrinsic Activities at Large Mass Loadings. ACS Catal. 2015, 5, 6680–9668. [Google Scholar] [CrossRef]
- Hall, D.S.; Lockwood, D.J.; Bock, C.; MacDougall, B.R. Nickel hydroxides and related materials: A review of their structures, synthesis and properties. Proc. Math Phys. Eng. Sci. 2015, 471, 20140792. [Google Scholar] [CrossRef] [PubMed]
- Kowal, A.; Niewiara, R.; Perończyk, B.; Haber, J. In Situ Atomic Force Microscopy Observation of Change in Thickness of Nickel Hydroxide Layer on Ni Electrode. Langmuir 1996, 12, 2332–2333. [Google Scholar] [CrossRef]
- Delahaye-Vidal, A.; Beaudoin, B.; Sac-Epée, N.; Tekaia-Elhsissen, K.; Audemer, A.; Figlarz, M. Structural and textural investigations of the nickel hydroxide electrode. Solid State Ion. 1996, 84, 239–248. [Google Scholar] [CrossRef]
- Yeo, B.S.; Bell, A.T. In Situ Raman Study of Nickel Oxide and Gold-Supported Nickel Oxide Catalysts for the Electrochemical Evolution of Oxygen. J. Phys. Chem. C 2012, 116, 8394–8400. [Google Scholar] [CrossRef]
- Sharel, P.E.; Liu, D.; Lazenby, R.A.; Sloan, J.; Vidotti, M.; Patrick, P.R.; Macpherson, J.V. Electrodeposition of Nickel Hydroxide Nanoparticles on Carbon Nanotube Electrodes: Correlation of Particle Crystallography with Electrocatalytic Properties. J. Phys. Chem. C 2016, 120, 16059–16068. [Google Scholar]
- de Souza, L.M.M.; Kong, F.P.; McLarnon, F.R.; Muller, R.H. Spectroscopic Ellipsometry Study of Nickel Oxidation in Alkaline Solution. Electrochim. Acta 1997, 42, 1253–1267. [Google Scholar] [CrossRef]
- Casella, I.G.; Guascito, M.R.; Sannazzaro, M.G. Voltammetric and XPS investigations of nickel hydroxide electrochemically dispersed on gold surface electrodes. J. Electroanal. Chem. 1999, 462, 202–210. [Google Scholar] [CrossRef]
- Grdeñ, M.; Klimek, K. EQCM studies on oxidation of metallic nickel electrode in basic solutions. J. Electroanal. Chem. 2005, 581, 122–131. [Google Scholar] [CrossRef]
- Tucceri, R. Surface resistance change during nickel hydroxide phase transformations. J. Electroanal. Chem. 2017, 792, 104–109. [Google Scholar] [CrossRef]
- Yoshida, M.; Onishi, S.; Mitsutomi, Y.; Yamamoto, F.; Nagasaka, M.; Yuzawa, H.; Kosugi, N.; Kondoh, H. Integration of Active Nickel Oxide Clusters by Amino Acids for Water Oxidation. J. Phys. Chem. C 2017, 121, 255–260. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wei, Y.; Zhang, W. Potential Cycling Effects on Activities of Nickel-Mediated Benzyl Alcohol and Glycine Electrooxidation in Alkaline Solutions. Catalysts 2020, 10, 119. https://doi.org/10.3390/catal10010119
Wei Y, Zhang W. Potential Cycling Effects on Activities of Nickel-Mediated Benzyl Alcohol and Glycine Electrooxidation in Alkaline Solutions. Catalysts. 2020; 10(1):119. https://doi.org/10.3390/catal10010119
Chicago/Turabian StyleWei, Yuyang, and Wenbin Zhang. 2020. "Potential Cycling Effects on Activities of Nickel-Mediated Benzyl Alcohol and Glycine Electrooxidation in Alkaline Solutions" Catalysts 10, no. 1: 119. https://doi.org/10.3390/catal10010119
APA StyleWei, Y., & Zhang, W. (2020). Potential Cycling Effects on Activities of Nickel-Mediated Benzyl Alcohol and Glycine Electrooxidation in Alkaline Solutions. Catalysts, 10(1), 119. https://doi.org/10.3390/catal10010119



