Abstract
The microenvironment of malignant gliomas is described according to its definition in the literature. Beside tumor cells, a series of stromal cells (microglia/macrophages, pericytes, fibroblasts, endothelial cells, normal and reactive astrocytes) represents the cell component, whereas a complex network of molecular signaling represents the functional component. Its most evident expressions are perivascular and perinecrotic niches that are believed to be the site of tumor stem cells or progenitors in the tumor. Phenotypically, both niches are not easily recognizable; here, they are described together with a critical revision of their concept. As for perinecrotic niches, an alternative interpretation is given about their origin that regards the tumor stem cells as the residue of those that populated hyperproliferating areas in which necroses develop. This is based on the concept that the stem-like is a status and not a cell type, depending on the microenvironment that regulates a conversion of tumor non-stem cells and tumor stem cells through a cell reprogramming.
With the term “tumor microenvironment” it is intended to indicate everything is active within the tumor except tumor cells. It includes, therefore, many cell types, such as endothelial cells, microglia/macrophages, reactive astrocytes, fibroblasts, pericytes, immune cells, etc., [1] and the relevant factors and molecular signaling addressed to promote tumor transformation, growth, invasion, therapeutic resistance [2] and defense from host immunity [3,4].
Any regulation of tumor cells towards regression, such as necrosis, or progression, such as proliferation, invasion and angiogenesis, finds its main driver in the microenvironment that mainly expresses itself in the so-called niches [3,4,5]. As a matter of fact, in glioblastoma multiforme (GBM), niches are regarded as the crucial points where microenvironment exerts influence, since they are the sites where glioblastoma stem cells (GSCs) are believed to reside, be maintained or originate and where the signaling arising in stromal and in tumor cells converges to regulate tumor features. Niches can be perivascular (PVN) or perinecrotic (PNN). The former have been conceived as simply represented by endothelial cells associated with Nestin+ and CD133+ stem cells, which condition angiogenesis and tumor growth [6], or as more complicated structures including, beside tumor stem cells, endothelial cells, astrocytes, fibroblasts, macrophages, pericytes, non-stem tumor cells, and microglia [3] (Figure 1). GSCs have been demonstrated to occur in PVN by CD133 positivity [7] or by side population signature genes, aspartate beta-hydroxylase domain containing 2 (ASPHD2) or nuclear factor erythroid 2-like 2 (NFE2L2) or hypoxia-inducible factor 2 (HIF-2) [8]. Positivity to stemness antigens increases with malignancy [9]. C6 glioma xenografts with a high content of GSCs exhibit an increased microvessel density and an increased recruitment of bone marrow (BM)-derived endothelia progenitors [10].
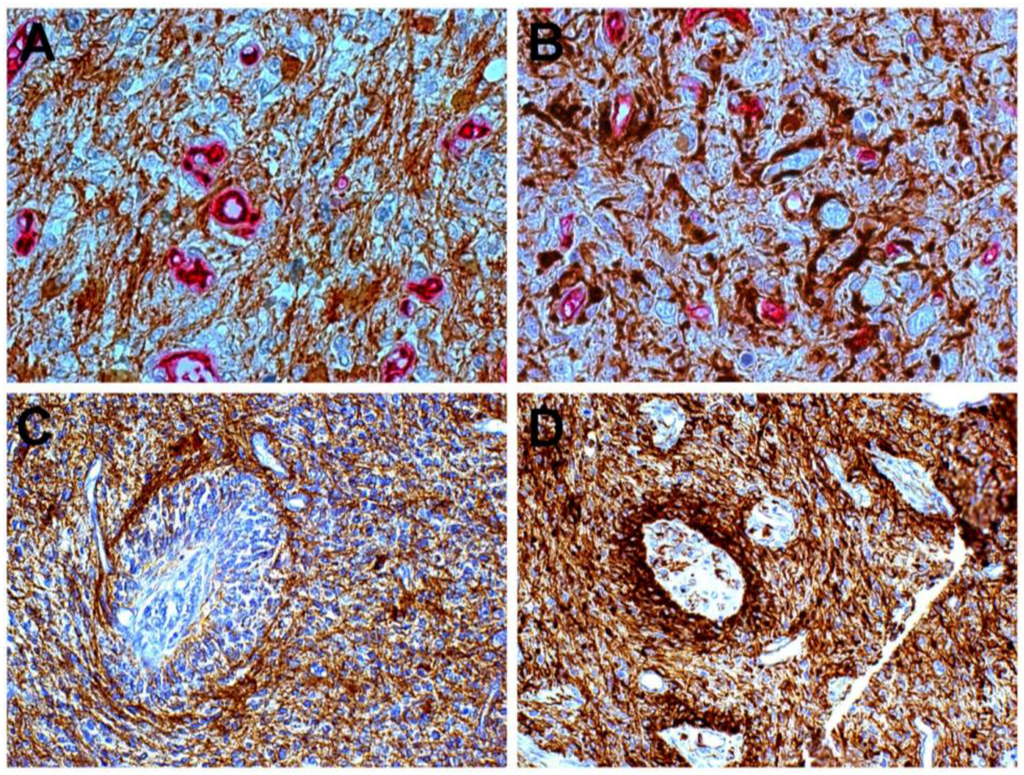
Figure 1.
Glioblastoma. Close relationship of Nestin+ cells with small vessels (A), but not with GFAP cells (B); DAB, ×400; Ring of tumor cells around a vessel: the inner cells are Nestin+ (C) and GFAP+ cells are external (D), DAB, ×200.
Figure 1.
Glioblastoma. Close relationship of Nestin+ cells with small vessels (A), but not with GFAP cells (B); DAB, ×400; Ring of tumor cells around a vessel: the inner cells are Nestin+ (C) and GFAP+ cells are external (D), DAB, ×200.
PNN have been described to develop around circumscribed necroses where hypoxia and HIF-1/2 [4] play a central role and to contain GSCs/progenitors as in PVN [7,8]. Therefore, in both niches a fundamental feature is, therefore, the occurrence of GSCs/progenitors that condition tumor aggressiveness (growth, proliferation, migration, resistance and recurrence).
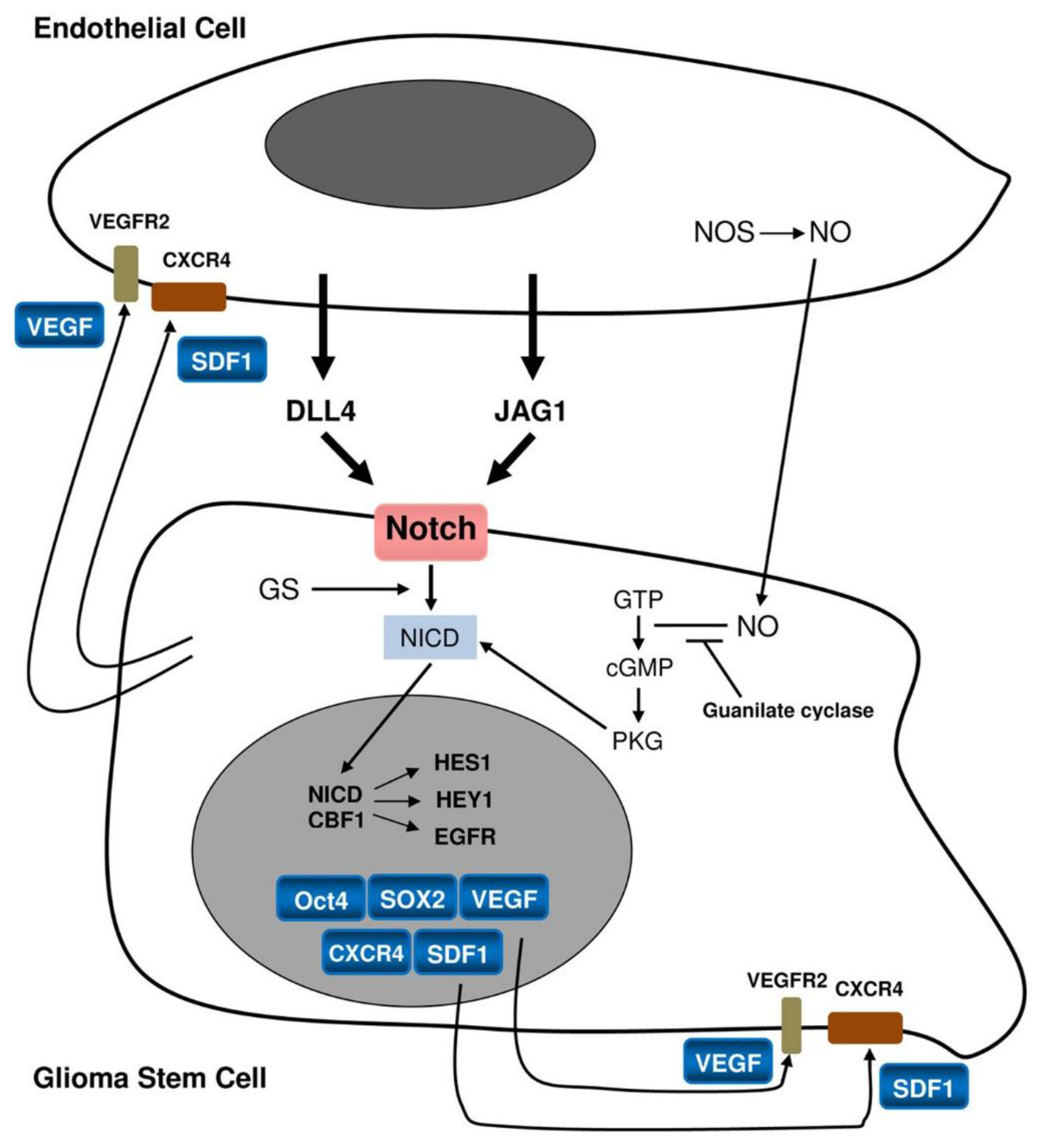
PVN are not merely repositories of stem cells [11], but they are dynamic entities that save stem cells from depletion and protect the host from their over-exuberant proliferation [12]. In GBM, vasculature is represented by simple endothelial cells, their hyperplasia or microvascular proliferations (MVP) to which not only endothelial cells, but mainly pericytes and muscle cells contribute [13,14]. The first inter-relation to be considered is the reciprocal influence between GSCs/progenitors and endothelial cells [11]. The stemness status of the former is maintained by endothelial cells via nitric oxide and Notch activation [3,6,15], whereas GSCs/progenitors would activate endothelial cells to proliferate, eliciting angiogenesis through vascular endothelial growth factor (VEGF) (Figure 2), to host at the tumor the BM-derived endothelial precursor cells (EPCs) promoting their differentiation into blood vessels inserted into the pre-existent vasculature [4]. In PVN, microenvironment includes the crosstalk with microglia/macrophages with their double pro-proliferation and pro-inflammatory exchanges [16,17,18,19], the function of pericytes [20,21,22,23], of reactive astrocytes, etc., [24].
Hypoxia, a mechanism of primary importance in the biology and aggressive behavior of malignant gliomas [25], is fundamental in PNN. It is critically involved in the regulation of GSCs [8] of which it promotes the expansion through the phosphatidylinositol 3 kinase (PI3K)/Akt and ERK1/2 pathways; the inhibition of the latter reduces the number of GSCs [26]. The mechanism of GSC promotion still consists in Notch activation through its ligands and the final activation of target genes Hes1 and Hey1 [8,27] (Figure 2). This has been confirmed by the blockade of Notch by γ-secretase inhibition that reduces the expression of stemness antigens such as Nestin, CD133, Bmi1 and inhibits in vitro neurosphere formation and xenographts [28]. Hypoxia is a feature of the entire GBM, but it is particularly evident where circumscribed necroses develop and where it induces key stem cells genes such as Nanog, Oct4 and c-Myc [29]. GSCs are in fact well demonstrable not only in perinecrotic palisadings, but also in cells scattered in the proliferating tumor [7,8]. In the signaling that occurs in niches all pathways that regulate tumor progression and transformation and other processes are included, such as epidermal growth factor receptor (EGFR) gene amplification, phosphatase and tensin homolog (PTEN) mutation, PI3K/Akt, bone morphogenetic proteins (BMP), loss of heterozygosity (LOH) on critical chromosomal regions, etc., as well as the intrinsic signaling such as Wnt/β-catenin, Bmi1, c-Myc, Oct4, OLIG2, Sonic Hedgehog (SHH), and Notch.
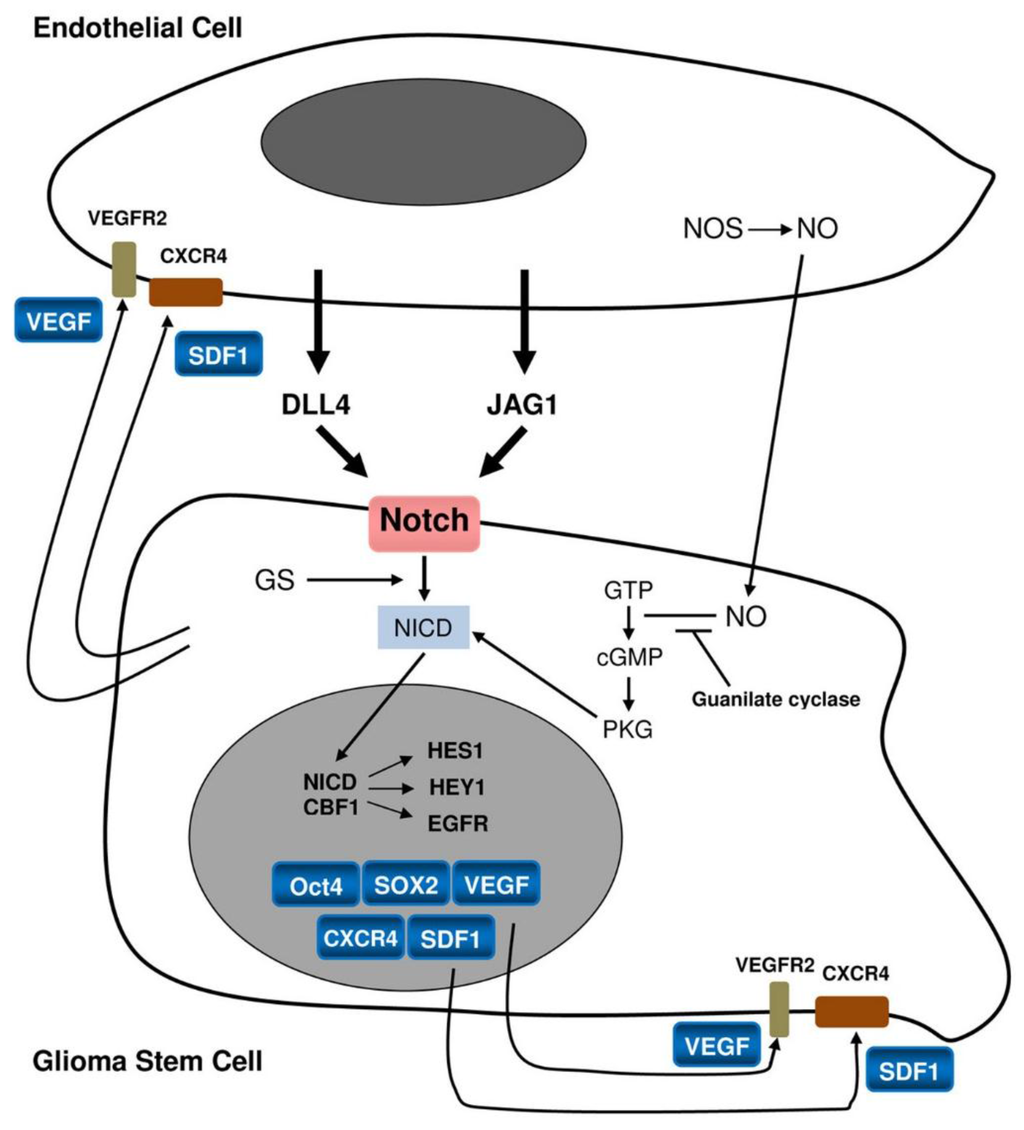
Figure 2.
Relationship between a stem cell/progenitor and an endothelial cell.
Figure 2.
Relationship between a stem cell/progenitor and an endothelial cell.
In the discussion of the tumor microenvironment, it is mandatory to consider the supposed origin of GSCs, i.e., by transformation of the normal neural stem cells (NSCs) and progenitors in relation with the so-called glioma initiating cells (GICs), the concept of which is just mentioned here. In line with the old belief that cancer cells are similar to embryonic stem cells [30], it has been established that glioma cells derive from immature glia [31,32], i.e., from primitive neuroepithelial cells or NSCs [33]. GICs share properties with NSCs [34,35,36], either in the sub-ventricular zone (SVZ) or during migration. GSCs, on the other hand, share genetic alterations with gliomas [37].
Other possibilities are the origin of GSCs from oligodendroglial precursor cells (OPCs) or NG2 cells [38,39,40,41] or by dedifferentiation either from normal glia through a multistep process [42,43,44] or through dedifferentiation of tumor cells that acquire stemness properties [45] (Scheme 1). This interpretation implies that GSCs are not a cell type, but they represent a functional status [46,47,48,49] that can be acquired or lost depending on the microenvironment. Glioma heterogeneity would depend on polyclonality, i.e., on genotypic and phenotypic differences acquired during proliferation, migration, also by epigenetic mechanisms [49,50] and, therefore, on the undifferentiation/differentiation status of its cell elements. The hypothesis is gaining consent that there is an equilibrium between tumor stem cells and tumor non-stem cells with the possibility of a conversion into one another regulated by the microenvironment [50]. This has been demonstrated to happen also in gliomas [1].
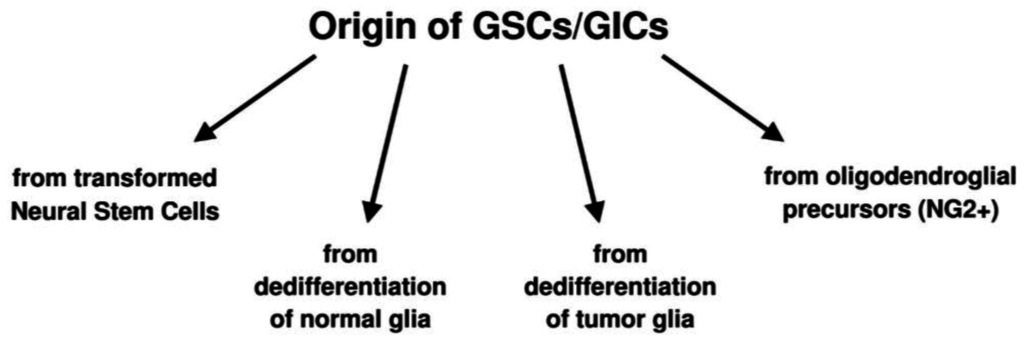
Scheme 1.
Hypotheses on the origin of glioblastoma stem cells (GSCs)/glioma initiating cells (GICs).
Scheme 1.
Hypotheses on the origin of glioblastoma stem cells (GSCs)/glioma initiating cells (GICs).
From the neuropathological point of view, the concept of microenvironment in gliomas materializes in its main expression sites, i.e., PVN and PNN. Theoretically and conceptually, the niches have been very well-defined [3,4,5], but, practically, except for the occurrence of GSCs/progenitors, it would be very difficult to establish when and how we can recognize them in the tumor phenotype. Either the occurrence of GSCs/progenitors is mandatory for their definition or each association between vessels/necroses and tumor falls within the definition of niche. A second point of uncertainty is that the term GSCs includes progenitors in different stages of differentiation, compared to the normal cytogenesis, until they keep the quantity of stemness that allow them to proliferate, to further differentiate, to give origin to neurospheres in culture and to be tumorigenic. It is not easy to ascertain this by immunohistochemistry, unless specific antigens are used [8] or by fluorescence-activated cell sorting (FACS), even though not even by the latter a certainty can be reached [51]. One of the two: either the definition of niches in the vessels/tumor and necroses/tumor relationship is uncertain or each such relationship is a niche. It would only depend on the degree of stemness of tumor cells, including the possibility that, hypothetically, NSCs could occur in such position. This impasse could be overcome with the hypothesis of a re-programming or a conversion of tumor cells into tumor stem cells by the activity of the microenvironment [1,52]. In a series of GBMs, the possible relationship between tumor stem and non-stem cells and vessels has been analyzed and the occurrence of cells expressing stemness antigens has been described [48].
As for PNN, we showed that, at variance with the activation of GSCs by hypoxia through HIF-1/2 [4,8], the tumor stem cells/progenitors around circumscribed necroses could represent the residues of those that populated hyperproliferating areas of GBM, that occur after MRI in the enhancing area around central necrosis in which necroses develop, regulated by the microenvironment [47]. Incidentally, this subject correlates with the relationship between tumor zone composition and heterogeneity with the extent of surgical removal and outcome [53]. Circumscribed necroses have been interpreted as due to a vessel pathology with consequent ischemia/hypoxia and activation of HIF-1/2 [54,55,56]. Another hypothesis has been put forward, i.e., that necrosis is due to the imbalance between the high proliferation rate of hyperproliferating areas of the tumor and the low one of endothelial cells [57]. Cell death is due either to necrosis or to apoptosis that occurs prevailingly in the palisading. The highly proliferating areas are populated by GSCs/progenitors deriving from dedifferentiated tumor cells that acquired stemness properties and that disappear after necrosis development, remaining to line the pseudopalisading as the cells spared by necrosis [1] (Figure 3 and Figure 4).
In conclusion, microenvironment by genetic and epigenetic mechanisms regulates the equilibrium between tumor stem cells and tumor non-stem cells so that the occurrence of GSCs would not imply the existence of a special type of cells, but it would be the consequence of an interplay that takes place in the microenvironment (Scheme 2). This would have therapeutic consequences, since the therapies directed to annihilate a fixed target such as GSCs as the responsible for growth, resistance and recurrence of the tumor should be converted into therapies aimed at molecular modifications of the microenvironment.
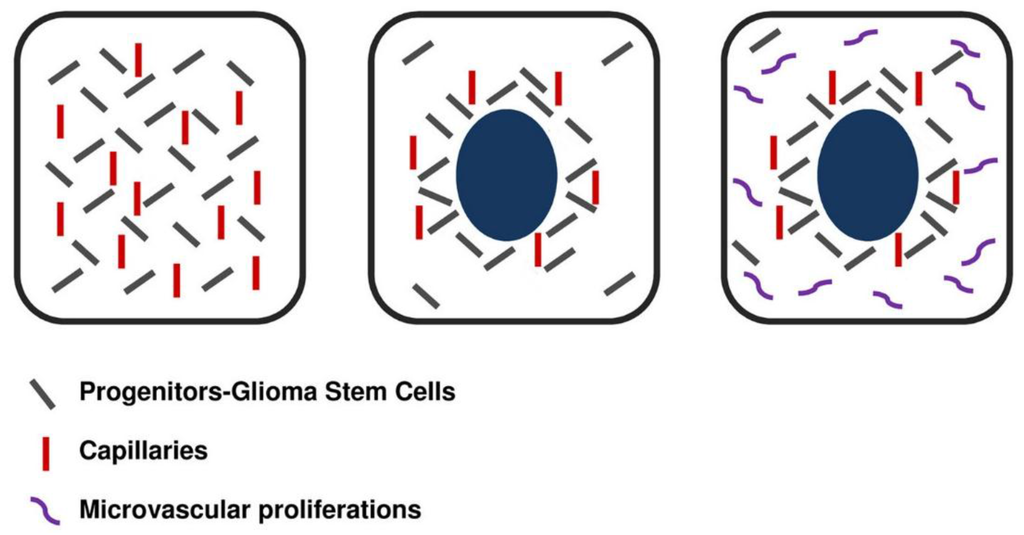
Figure 3.
Development of necrosis in a hyperproliferating area from ischemia due to the imbalance between proliferation rate of tumor and endothelial cells.
Figure 3.
Development of necrosis in a hyperproliferating area from ischemia due to the imbalance between proliferation rate of tumor and endothelial cells.
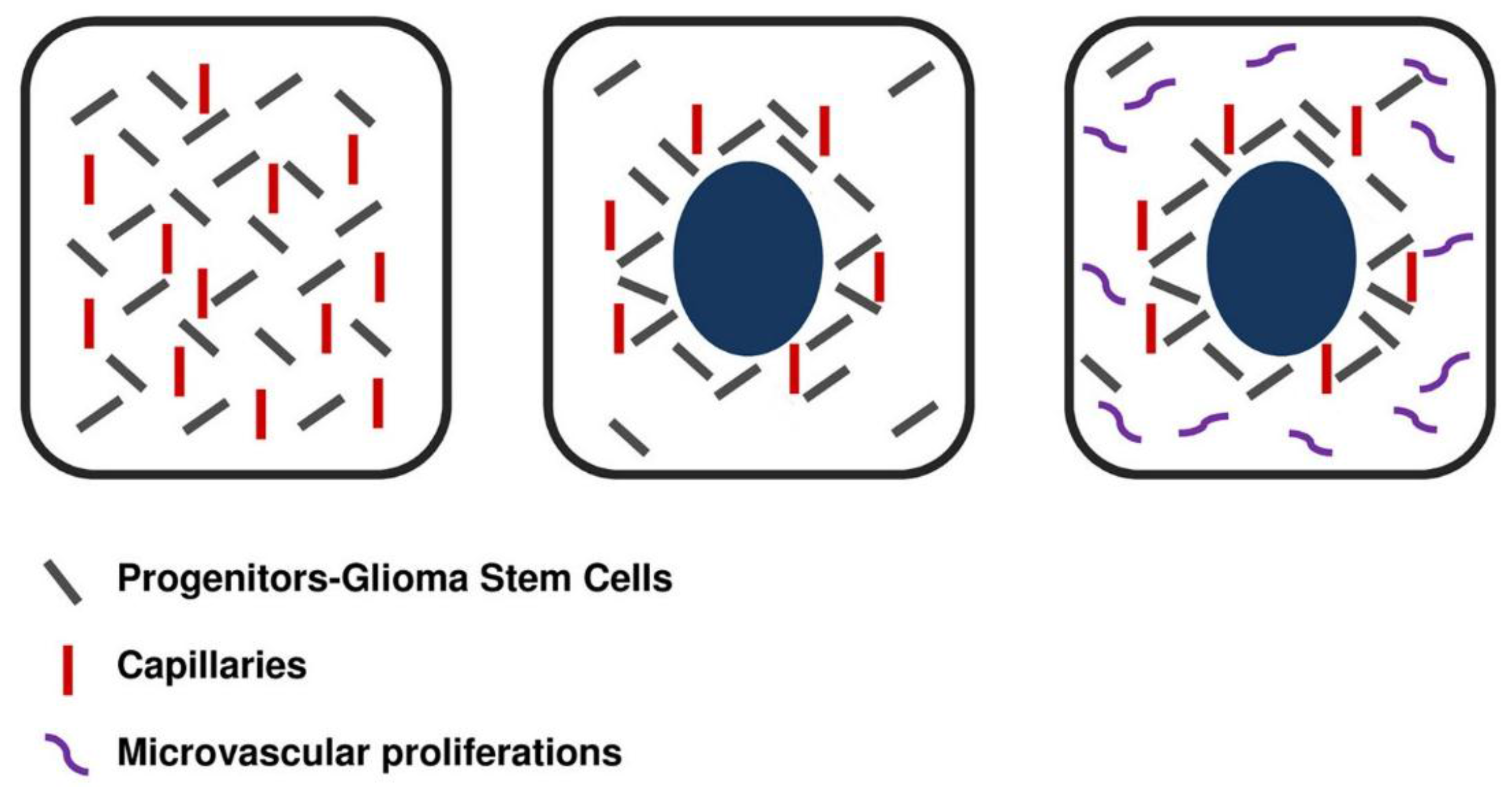
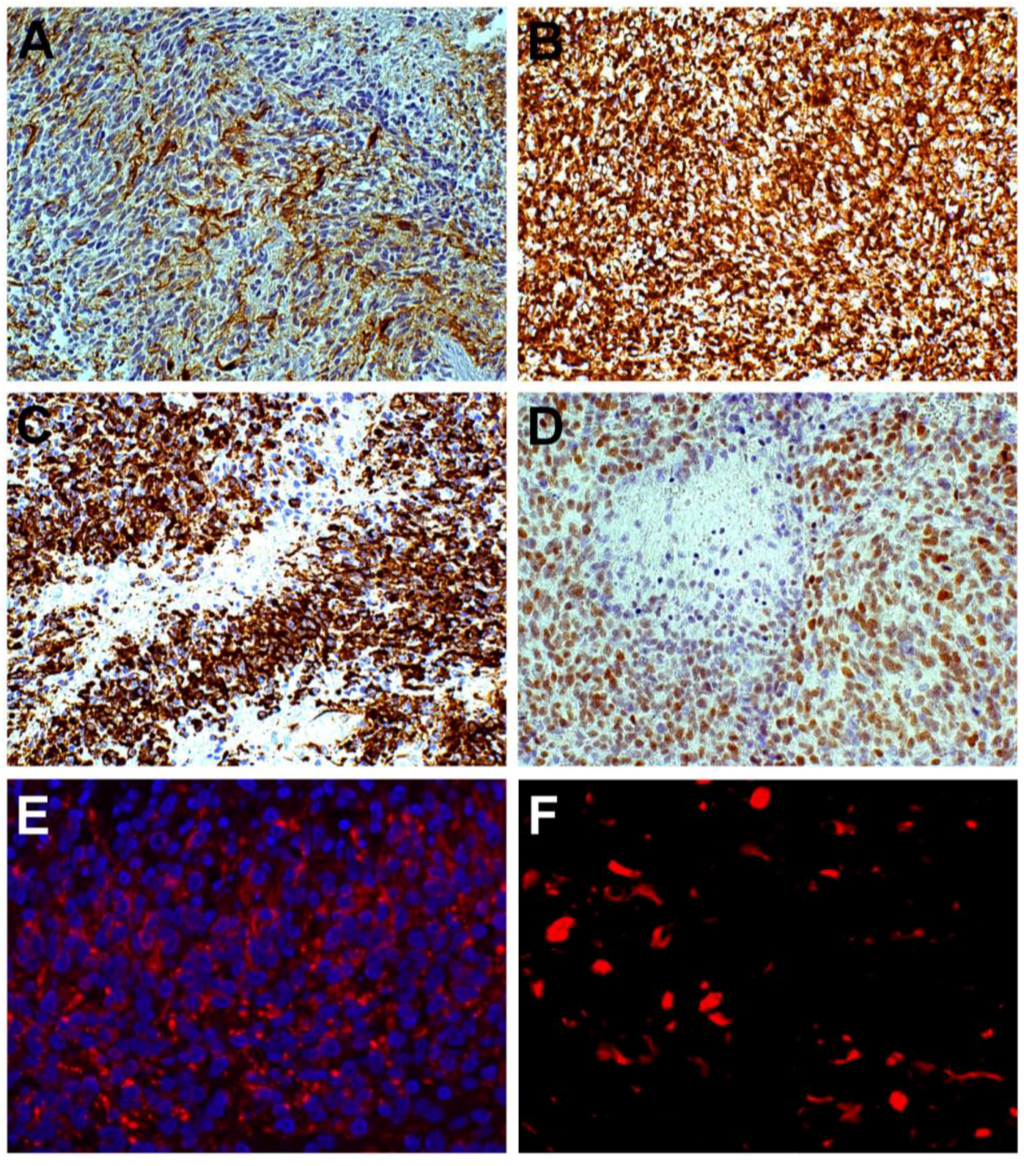
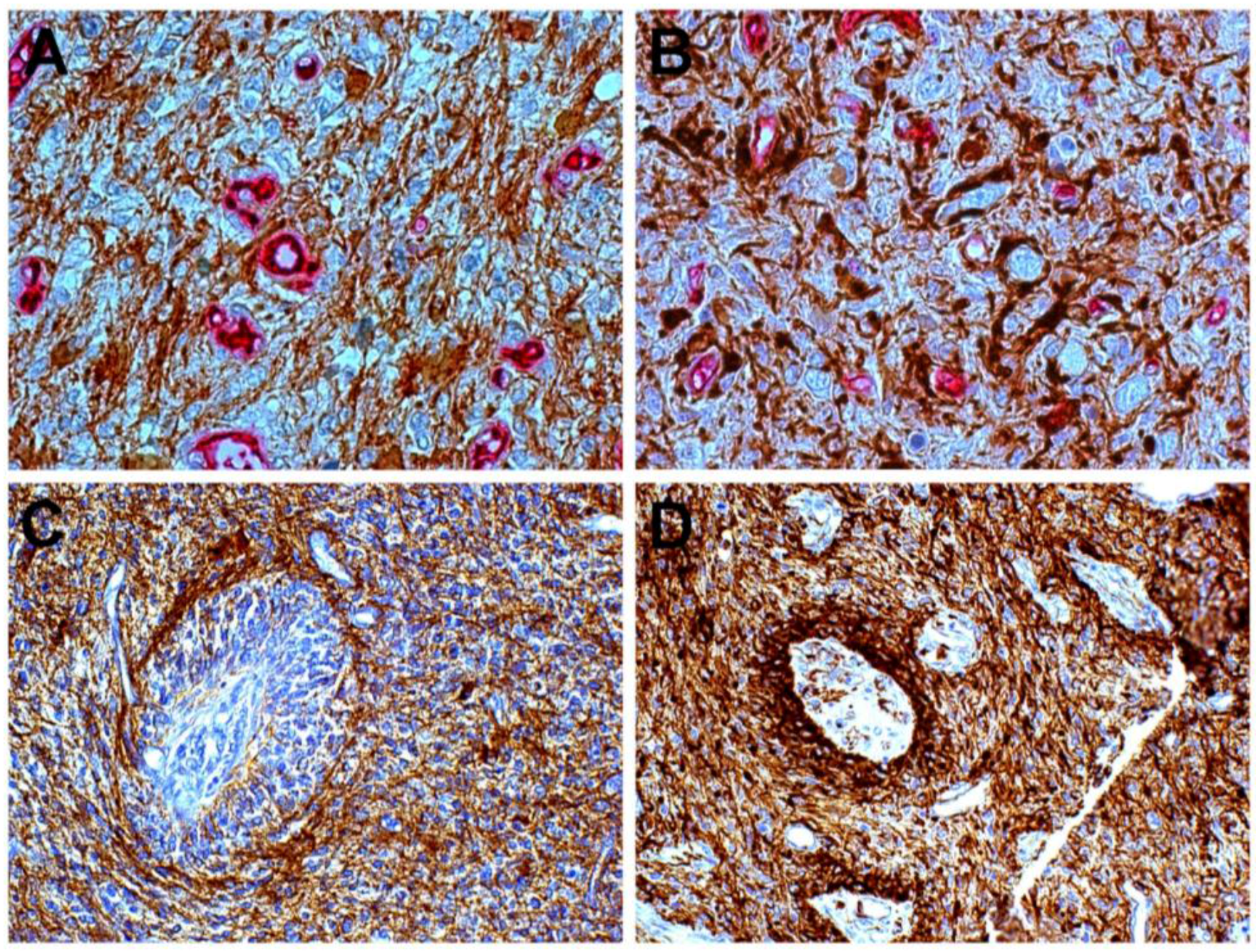
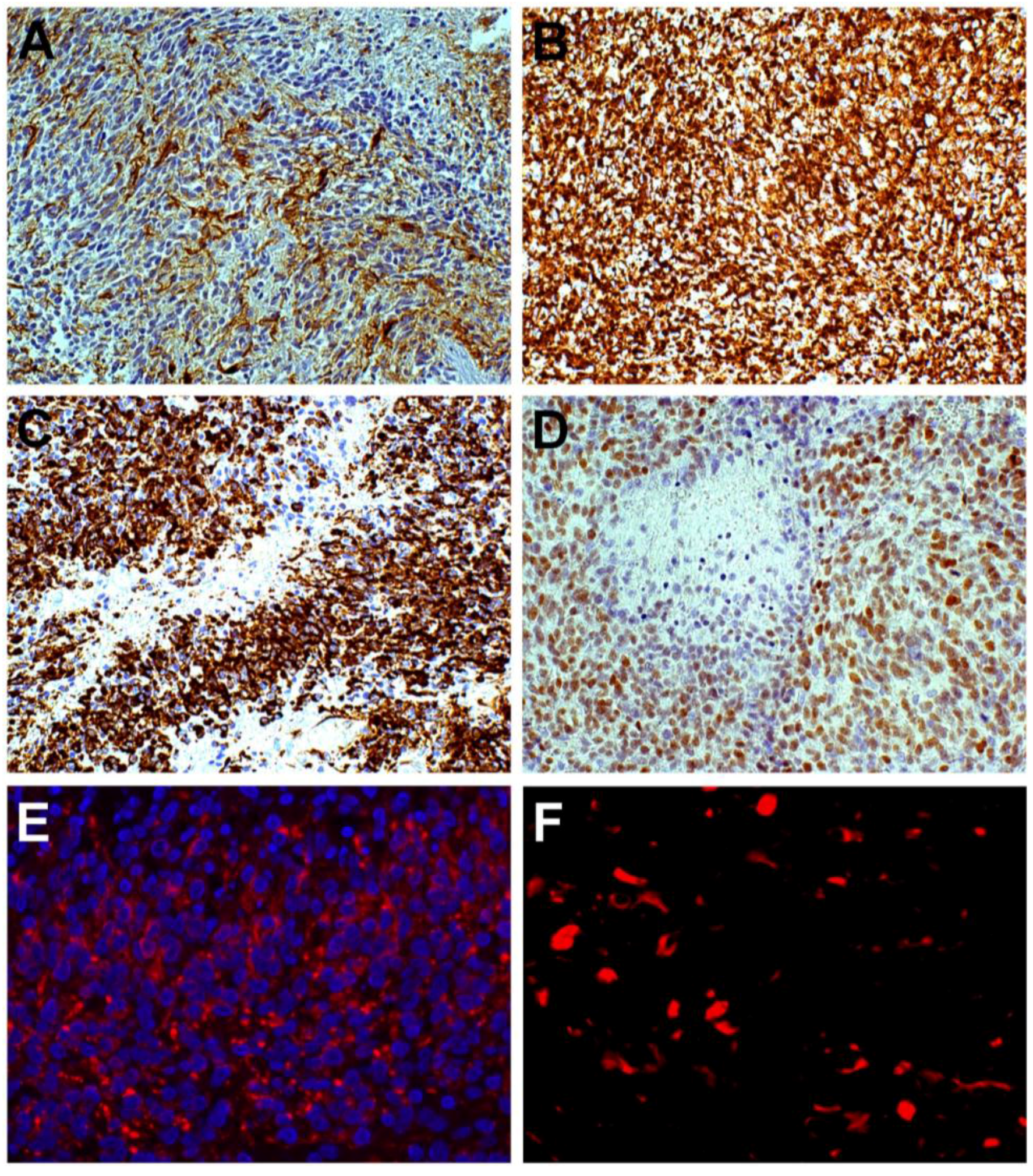
Figure 4.
Glioblastoma. (A) Hyperproliferating area with scarce GFAP-positive cells, DAB, ×200; (B) Id. with abundant Nestin+ cells, DAB, ×200; (C) Circumscribed necrosis developing in a Nestin-rich hyperproliferating area, DAB, ×200; (D) Circumscribed necrosis developed in a SOX2-rich hyperproliferating area, DAB, ×200; (E) Most cells are Nestin+ in a perinecrotic palisade, immunofluorescence, ×400; (F) Nestin+ cells around a circumscribed necrosis, immunofluorescence, ×400.
Figure 4.
Glioblastoma. (A) Hyperproliferating area with scarce GFAP-positive cells, DAB, ×200; (B) Id. with abundant Nestin+ cells, DAB, ×200; (C) Circumscribed necrosis developing in a Nestin-rich hyperproliferating area, DAB, ×200; (D) Circumscribed necrosis developed in a SOX2-rich hyperproliferating area, DAB, ×200; (E) Most cells are Nestin+ in a perinecrotic palisade, immunofluorescence, ×400; (F) Nestin+ cells around a circumscribed necrosis, immunofluorescence, ×400.
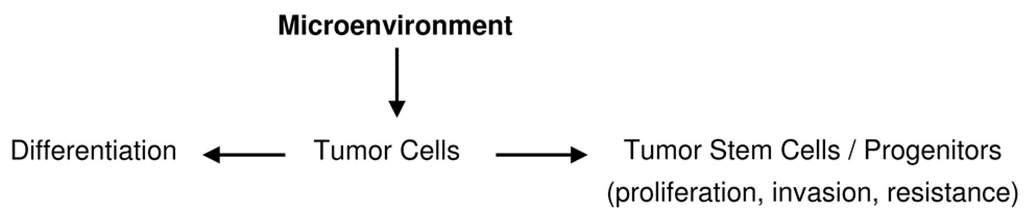
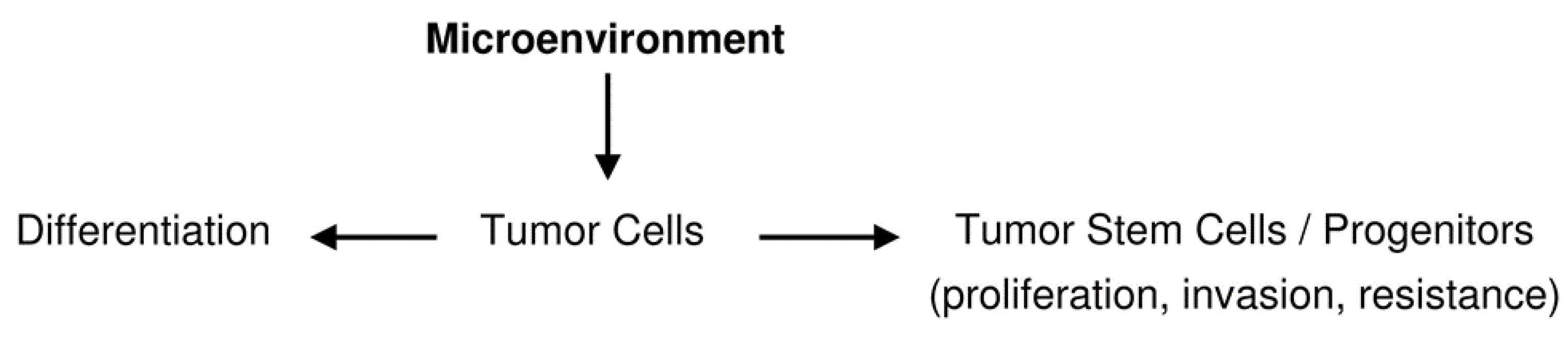
Scheme 2.
Possible dynamics of stemness and differentiation.
Scheme 2.
Possible dynamics of stemness and differentiation.
Author Contributions
Davide Schiffer, Marta Mellai and Laura Annovazzi conceived and designed the work. Marta Mazzucco performed the experiments.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Schiffer, D.; Annovazzi, L.; Cassoni, P.; Valentini, C.; Mazzucco, M.; Mellai, M. Glioblastoma stem cells: Conversion or reprogramming from tumor non-stem cells? J. Stem Cell Res. Ther. 2015, in press. [Google Scholar]
- Swartz, M.A.; Iida, N.; Roberts, E.W.; Sangaletti, S.; Wong, M.H.; Yull, F.E.; Coussens, L.M.; DeClerck, Y.A. Tumor microenvironment complexity: Emerging roles in cancer therapy. Cancer Res. 2012, 72, 2473–2480. [Google Scholar] [CrossRef] [PubMed]
- Charles, N.A.; Holland, E.C.; Gilbertson, R.; Glass, R.; Kettenmann, H. The brain tumor microenvironment. Glia 2011, 59, 1169–1180. [Google Scholar] [CrossRef] [PubMed]
- Filatova, A.; Acker, T.; Garvalov, B.K. The cancer stem cell niche(s): The crosstalk between glioma stem cells and their microenvironment. Biochim. Biophys. Acta 2013, 1830, 2496–2508. [Google Scholar] [CrossRef] [PubMed]
- Lorger, M. Tumor microenvironment in the brain. Cancers 2012, 4, 218–243. [Google Scholar] [CrossRef] [PubMed]
- Calabrese, C.; Poppleton, H.; Kocak, M.; Hogg, T.L.; Fuller, C.; Hamner, B.; Oh, E.Y.; Gaber, M.W.; Finklestein, D.; Allen, M.; et al. A perivascular niche for brain tumor stem cells. Cancer Cell 2007, 11, 69–82. [Google Scholar] [CrossRef] [PubMed]
- Christensen, K.; Schrøder, H.D.; Kristensen, B.W. CD133 identifies perivascular niches in grade II–IV astrocytomas. J. Neurooncol. 2008, 90, 157–170. [Google Scholar] [CrossRef] [PubMed]
- Seidel, S.; Garvalov, B.K.; Wirta, V.; von Stechow, L.; Schänzer, A.; Meletis, K.; Wolter, M.; Sommerlad, D.; Henze, A.T.; Nistér, M.; et al. A hypoxic niche regulates glioblastoma stem cells through hypoxia inducible factor 2 alpha. Brain 2010, 133, 983–995. [Google Scholar] [CrossRef] [PubMed]
- He, H.; Li, M.W.; Niu, C.S. The pathological characteristics of glioma stem cell niches. J. Clin. Neurosci. 2012, 19, 121–127. [Google Scholar] [CrossRef] [PubMed]
- Folkins, C.; Shaked, Y.; Man, S.; Tang, T.; Lee, C.R.; Zhu, Z.; Hoffman, R.M.; Kerbel, R.S. Glioma tumor stem-like cells promote tumor angiogenesis and vasculogenesis via vascular endothelial growth factor and stromal-derived factor 1. Cancer Res. 2009, 69, 7243–7251. [Google Scholar] [CrossRef] [PubMed]
- Gilbertson, R.J.; Rich, J.N. Making a tumour’s bed: Glioblastoma stem cells and the vascular niche. Nat. Rev. Cancer 2007, 7, 733–766. [Google Scholar] [CrossRef] [PubMed]
- Scadden, D.T. The stem-cell niche as an entity of action. Nature 2006, 441, 1075–1079. [Google Scholar] [CrossRef] [PubMed]
- Wesseling, P.; Schlingemann, R.O.; Rietveld, F.J.; Link, M.; Burger, P.C.; Ruiter, D.J. Early and extensive contribution of pericytes/vascular smooth muscle cells to microvascular proliferation in glioblastoma multiforme: An immuno-light and immuno-electron microscopic study. J. Neuropathol. Exp. Neurol. 1995, 54, 304–310. [Google Scholar] [CrossRef] [PubMed]
- Schiffer, D. Brain tumors. In Biology, Pathology and Clinical References, 2nd ed.; Springer-Verlag: Berlin, Germany, 1997. [Google Scholar]
- Hambardzumyan, D.; Becher, O.J.; Rosenblum, M.K.; Pandolfi, P.P.; Manova-Todorova, K.; Holland, E.C. PI3K pathway regulates survival of cancer stem cells residing in the perivascular niche following radiation in medulloblastoma in vivo. Genes Dev. 2008, 22, 436–448. [Google Scholar] [CrossRef] [PubMed]
- Zhai, H.; Heppner, F.L.; Tsirka, S.E. Microglia/macrophages promote glioma progression. Glia 2011, 59, 472–485. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Graeber, M.B. The molecular profile of microglia under the influence of glioma. Neuro-Oncology 2012, 14, 958–778. [Google Scholar] [CrossRef] [PubMed]
- Prinz, M. Microglia and monocytes: Molecularly defined. Acta Neuropathol. 2014, 128, 317–318. [Google Scholar] [CrossRef] [PubMed]
- Szulzewsky, F.; Pelz, A.; Feng, X.; Synowitz, M.; Markovic, D.; Langmann, T.; Holtman, I.R.; Wang, X.; Eggen, B.J.; Boddeke, H.W.; et al. Glioma-associated microglia/macrophages display an expression profile different from M1 and M2 polarization and highly express Gpnmb and Spp1. PLoS ONE 2015, 10, e0116644. [Google Scholar] [CrossRef] [PubMed]
- Bergers, G.; Song, S. The role of pericytes in blood-vessel formation and maintenance. Neuro-Oncology 2005, 7, 452–464. [Google Scholar] [CrossRef] [PubMed]
- Bexell, D.; Gunnarsson, S.; Tormin, A.; Darabi, A.; Gisselsson, D.; Roybon, L.; Scheding, S.; Bengzon, J. Bone marrow multipotent mesenchymal stroma cells act as pericyte-like migratory vehicles in experimental gliomas. Mol. Ther. 2009, 17, 183–190. [Google Scholar] [CrossRef] [PubMed]
- Armulik, A.; Genové, G.; Mäe, M.; Nisancioglu, M.H.; Wallgard, E.; Niaudet, C.; He, L.; Norlin, J.; Lindblom, P.; Strittmatter, K.; et al. Pericytes regulate the blood-brain barrier. Nature 2010, 468, 557–561. [Google Scholar] [CrossRef] [PubMed]
- Birnbaum, T.; Hildebrandt, J.; Nuebling, G.; Sostak, P.; Straube, A. Glioblastoma-dependent differentiation and angiogenic potential of human mesenchymal stem cells in vitro. J. Neurooncol. 2011, 105, 57–65. [Google Scholar] [CrossRef] [PubMed]
- Schiffer, D.; Mellai, M.; Annovazzi, L.; Casalone, C.; Cassoni, P. Tumor microenvironment—Perivascular and perinecrotic niches. In Tumors of the Central Nervous System; Lichtor, T., Ed.; InTech: Rijeka, Croatia, 2015; pp. 49–82. [Google Scholar]
- Evans, S.M.; Judy, K.D.; Dunphy, I.; Jenkins, W.T.; Hwang, W.T.; Nelson, P.T.; Lustig, R.A.; Jenkins, K.; Magarelli, D.P.; Hahn, S.M.; et al. Hypoxia is important in the biology and aggression of human glial brain tumors. Clin. Cancer Res. 2004, 10, 8177–8184. [Google Scholar] [CrossRef] [PubMed]
- Assanah, M.; Lochhead, R.; Ogden, A.; Bruce, J.; Goldman, J.; Canoll, P. Glial progenitors in adult white matter are driven to form malignant gliomas by platelet-derived growth factor-expressing retroviruses. J. Neurosci. 2006, 26, 6781–6790. [Google Scholar] [CrossRef] [PubMed]
- Bar, E.E.; Lin, A.; Mahairaki, V.; Matsui, W.; Eberhart, C.G. Hypoxia increases the expression of stem-cell markers and promotes clonogenicity in glioblastoma neurospheres. Am. J. Pathol. 2010, 177, 1491–1502. [Google Scholar] [CrossRef] [PubMed]
- Fan, X.; Khaki, L.; Zhu, T.S.; Soules, M.E.; Talsma, C.E.; Gul, N.; Koh, C.; Zhang, J.; Li, Y.M.; Maciaczyk, J.; et al. NOTCH pathway blockade depletes CD133-positive glioblastoma cells and inhibits growth of tumor neurospheres and xenografts. Stem Cells 2010, 28, 5–16. [Google Scholar] [CrossRef] [PubMed]
- Heddleston, J.M.; Li, Z.; McLendon, R.E.; Hjelmeland, A.B.; Rich, J.N. The hypoxic microenvironment maintains glioblastoma stem cells and promotes reprogramming towards a cancer stem cell phenotype. Cell Cycle 2009, 8, 3274–3284. [Google Scholar] [CrossRef] [PubMed]
- Virchow, R. Die krankhaften Geschwülste; Hirschwald: Berlin, Germany, 1865. [Google Scholar]
- Reynolds, B.A.; Weiss, S. Generation of neurons and astrocytes from isolated cells of the adult mammalian central nervous system. Science 1992, 255, 1707–1710. [Google Scholar] [CrossRef] [PubMed]
- Doetsch, F.; Caillé, I.; Lim, D.A.; García-Verdugo, J.M.; Alvarez-Buylla, A. Subventricular zone astrocytes are neural stem cells in the adult mammalian brain. Cell 1999, 97, 703–716. [Google Scholar] [CrossRef]
- Visvader, J.E.; Lindeman, G.J. Cancer stem cells in solid tumours: Accumulating evidence and unresolved questions. Nat. Rev. Cancer 2008, 8, 755–768. [Google Scholar] [CrossRef] [PubMed]
- Ignatova, T.N.; Kukekov, V.G.; Laywell, E.D.; Suslov, O.N.; Vrionis, F.D.; Steindler, D.A. Human cortical glial tumors contain neural stem-like cells expressing astroglial and neuronal markers in vitro. Glia 2002, 39, 193–206. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.K.; Clarke, I.D.; Hide, T.; Dirks, P.B. Cancer stem cells in nervous system tumors. Oncogene 2004, 23, 7267–7273. [Google Scholar] [CrossRef] [PubMed]
- Sanai, N.; Alvarez-Buylla, A.; Berger, M.S. Neural stem cells and the origin of gliomas. N. Engl. J. Med. 2005, 353, 811–822. [Google Scholar] [CrossRef] [PubMed]
- Caldera, V.; Mellai, M.; Annovazzi, L.; Piazzi, A.; Lanotte, M.; Cassoni, P.; Schiffer, D. Antigenic and Genotypic Similarity between Primary Glioblastomas and Their Derived Neurospheres. J. Oncol. 2011, 2011, 314962. [Google Scholar] [CrossRef] [PubMed]
- Lindberg, N.; Kastemar, M.; Olofsson, T.; Smits, A.; Uhrbom, L. Oligodendrocyte progenitor cells can act as cell of origin for experimental glioma. Oncogene 2009, 28, 2266–2275. [Google Scholar] [CrossRef] [PubMed]
- Persson, A.I.; Petritsch, C.; Swartling, F.J.; Itsara, M.; Sim, F.J.; Auvergne, R.; Goldenberg, D.D.; Vandenberg, S.R.; Nguyen, K.N.; Yakovenko, S.; et al. Non-stem cell origin for oligodendroglioma. Cancer Cell 2010, 18, 669–682. [Google Scholar] [CrossRef] [PubMed]
- Lathia, J.D.; Gallagher, J.; Myers, J.T.; Li, M.; Vasanji, A.; McLendon, R.E.; Hjelmeland, A.B.; Huang, A.Y.; Rich, J.N. Direct in vivo evidence for tumor propagation by glioblastoma cancer stem cells. PLoS ONE 2011, 6, e24807. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Y.; Uhrbom, L. On the origin of glioma. Upsala J. Med. Sci. 2012, 117, 113–121. [Google Scholar] [CrossRef] [PubMed]
- Holland, E.C.; Celestino, J.; Dai, C.; Schaefer, L.; Sawaya, R.E.; Fuller, G.N. Combined activation of Ras and Akt in neural progenitors induces glioblastoma formation in mice. Nat. Genet. 2000, 25, 55–57. [Google Scholar] [CrossRef] [PubMed]
- Bachoo, R.M.; Maher, E.A.; Ligon, K.L.; Sharpless, N.E.; Chan, S.S.; You, M.J.; Tang, Y.; DeFrances, J.; Stover, E.; Weissleder, R.; et al. Epidermal growth factor receptor and Ink4a/Arf: Convergent mechanisms governing terminal differentiation and transformation along the neural stem cell to astrocyte axis. Cancer Cell 2002, 1, 269–277. [Google Scholar] [CrossRef]
- Radke, J.; Bortolussi, G.; Pagenstecher, A. Akt and c-Myc induce stem-cell markers in mature primary p53⁻/⁻ astrocytes and render these cells gliomagenic in the brain of immunocompetent mice. PLoS ONE 2013, 8, e56691. [Google Scholar] [CrossRef] [PubMed]
- Schiffer, D.; Mellai, M.; Annovazzi, L.; Piazzi, A.; Monzeglio, O.; Caldera, V. Glioblastoma cancer stem cells: Basis for a functional hypothesis. Stem Cell Discov. 2012, 2, 122–131. [Google Scholar] [CrossRef]
- Zipori, D. The nature of stem cells: State rather than entity. Nat. Rev. Genet. 2004, 5, 873–878. [Google Scholar] [CrossRef] [PubMed]
- Vescovi, A.L.; Galli, R.; Reynolds, B.A. Brain tumour stem cells. Nat. Rev. Cancer 2006, 6, 425–236. [Google Scholar] [CrossRef] [PubMed]
- Schiffer, D.; Mellai, M.; Annovazzi, L.; Caldera, V.; Piazzi, A.; Denysenko, T.; Melcarne, A. Stem cell niches in glioblastoma: A neuropathological view. Biomed. Res. Int. 2014, 2014, 725921. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Neaves, W.B. Normal stem cells and cancer stem cells: The niche matters. Cancer Res. 2006, 66, 4553–4557. [Google Scholar] [CrossRef] [PubMed]
- Furnari, F.B.; Fenton, T.; Bachoo, R.M.; Mukasa, A.; Stommel, J.M.; Stegh, A.; Hahn, W.C.; Ligon, K.L.; Louis, D.N.; Brennan, C.; et al. Malignant astrocytic glioma: Genetics, biology, and paths to treatment. Genes Dev. 2007, 21, 2683–2710. [Google Scholar] [CrossRef] [PubMed]
- Golebiewska, A.; Brons, N.H.; Bjerkvig, R.; Niclou, S.P. Critical appraisal of the side population assay in stem cell and cancer stem cell research. Cell Stem Cell 2011, 8, 136–147. [Google Scholar] [CrossRef] [PubMed]
- Safa, A.R.; Saadatzadeh, M.R.; Cohen-Gadol, A.A.; Pollok, K.E.; Bijangi-Vishehsaraei, K. Glioblastoma stem cells (GSCs) epigenetic plasticity and interconversion between differentiated non-GSCs and GSCs. Genes Dis. 2015, 2, 152–163. [Google Scholar] [CrossRef] [PubMed]
- Eyüpoglu, I.Y.; Buchfelder, M.; Savaskan, N.E. Surgical resection of malignant gliomas-role in optimizing patient outcome. Nat. Rev. Neurol. 2013, 9, 141–151. [Google Scholar] [CrossRef] [PubMed]
- Rong, Y.; Durden, D.L.; van Meir, E.G.; Brat, D.J. “Pseudopalisading” necrosis in glioblastoma: A familiar morphologic feature that links vascular pathology, hypoxia, and angiogenesis. J. Neuropathol. Exp. Neurol. 2006, 65, 529–539. [Google Scholar] [CrossRef] [PubMed]
- Fischer, I.; Gagner, J.P.; Law, M.; Newcomb, E.W.; Zagzag, D. Angiogenesis in gliomas: Biology and molecular pathophysiology. Brain Pathol. 2005, 15, 297–310. [Google Scholar] [CrossRef] [PubMed]
- Hardee, M.E.; Zagzag, D. Mechanisms of glioma-associated neovascularization. Am. J. Pathol. 2012, 181, 1126–1141. [Google Scholar] [CrossRef] [PubMed]
- Schiffer, D.; Chiò, A.; Giordana, M.T.; Mauro, A.; Migheli, A.; Vigliani, M.C. The vascular response to tumor infiltration in malignant gliomas. Morphometric and reconstruction study. Acta Neuropathol. 1989, 77, 369–378. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
