Matrix Metalloproteinases: The Gene Expression Signatures of Head and Neck Cancer Progression
Abstract
:1. Introduction
2. MMP Expression in HNSCCs
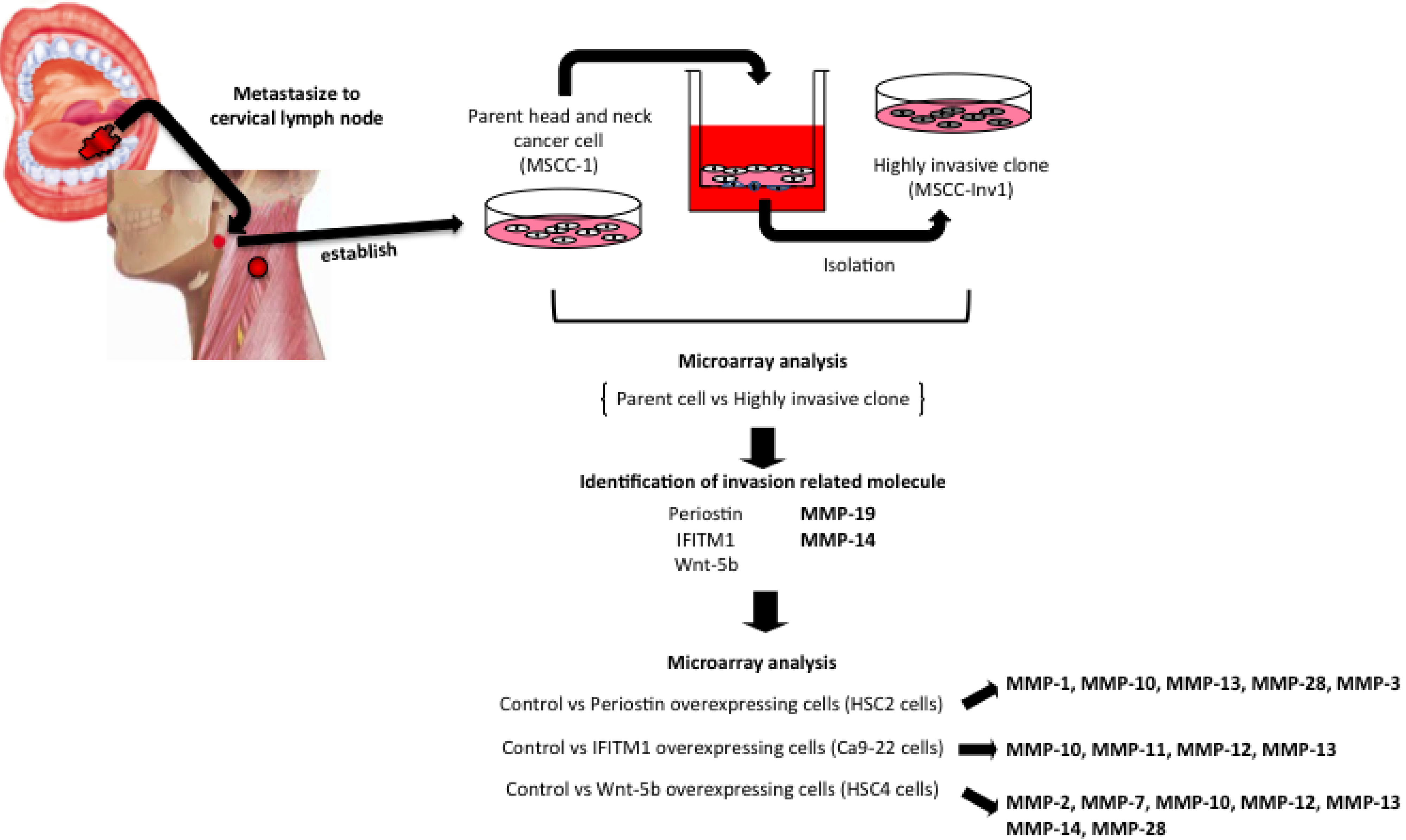
| Comparison (/Parent Cells) | Fold Change | Symbol | Other Designations |
|---|---|---|---|
| MSCC-Inv1 | 2.82 | MMP-19 | MMP-18, RASI-1 |
| 2.44 | MMP-14 | MT1-MMP | |
| Periostin overexpressing cells | 30.2 | MMP-1 | Interstitial collagenase |
| 17.4 | MMP-10 | Stromelysin 2 | |
| 14.7 | MMP-13 | Collagenase 3 | |
| 2.9 | MMP-28 | Epilysin | |
| 2.6 | MMP-3 | Stromelysin 1, Progelatinase | |
| IFITM1 overexpressing cells | 19.1 | MMP-13 | Collagenase 3 |
| 8.2 | MM-12 | Macrophage elastase | |
| 3.6 | MMP-10 | Stromelysin 2 | |
| 2.8 | MMP-11 | Stromelysin 3 | |
| Wnt-5B overexpressing cells | 7.9 | MMP-12 | Macrophage elastase |
| 7.8 | MMP-13 | Collagenase 3 | |
| 6.1 | MMP-7 | Matrilysin, uterine | |
| 5.8 | MMP-10 | Stromelysin 2 | |
| 2.8 | MMP-14 | MT1-MMP | |
| 2.5 | MMP-28 | Epilysin | |
| 2.2 | MMP-2 | 72 kDa gelatinase, type IV collagenase |
| Fold Change | Symbol | Other Designations |
|---|---|---|
| 13.9 | MMP-1 | Interstitial collagenase |
| 12.0 | MMP-12 | Macrophage elastase |
| 8.6 | MMP-10 | Stromelysin 2 |
| 8.1 | MMP-3 | Stromelysin 1 |
| 7.3 | MMP-7 | Matrilysin, uterine |
| 7.2 | MMP-9 | 92kDa gelatinase, type IV Collagenase |
| 5.7 | MMP-13 | Collagenase 3 |
| Symbol | Other Designations | p value |
|---|---|---|
| MMP-1 | Interstitial collaganase | ** |
| MMP-2 | 72 kDa gelatinase, type IV collagenase | ** |
| MMP-3 | Stromelysin 1 | ** |
| MMP-7 | Matrilysin, uterine | ** |
| MMP-8 | PMNL collagenase (MNL-CL) | * |
| MMP-9 | 92 kDa gelatinase, type IV Collagenase | ** |
| MMP-10 | Stromelysin 2 | ** |
| MMP-11 | Stromelysin 3 | ** |
| MMP-12 | Macrophage elastase | ** |
| MMP-13 | Collagenase 3 | ** |
| MMP-14 | MT1-MMP | ** |
| MMP-15 | MT2-MMP | NS |
| MMP-16 | MT3-MMP | ** |
| MMP-17 | MT4-MMP | ** |
| MMP-19 | MMP-18, RASI-1 | ** |
| MMP-23 | CA-MMP | ** |
| MMP-24 | MT5-MMP | ** |
| MMP-25 | MT6-MMP | ** |
| MMP-27 | MMP-27 | NS |
| MMP-28 | Epilysin | NS |
| Fold Change | Symbol | Other Designations |
|---|---|---|
| 57.6 | MMP-1 | Interstitial collaganase |
| 8.4 | MMP-10 | Stromelysin 2 |
| 8.4 | MMP-3 | Stromelysin 1 |
| 7.8 | MMP-12 | Macrophage elastase |
| 4.1 | MMP-9 | 92 kDa gelatinase, type IV Collagenase |
| 3.8 | MMP-13 | Collagenase 3 |
| 2.8 | MMP-7 | Matrilysin, uterine |
| 2.0 | MMP-11 | Stromelysin 3 |

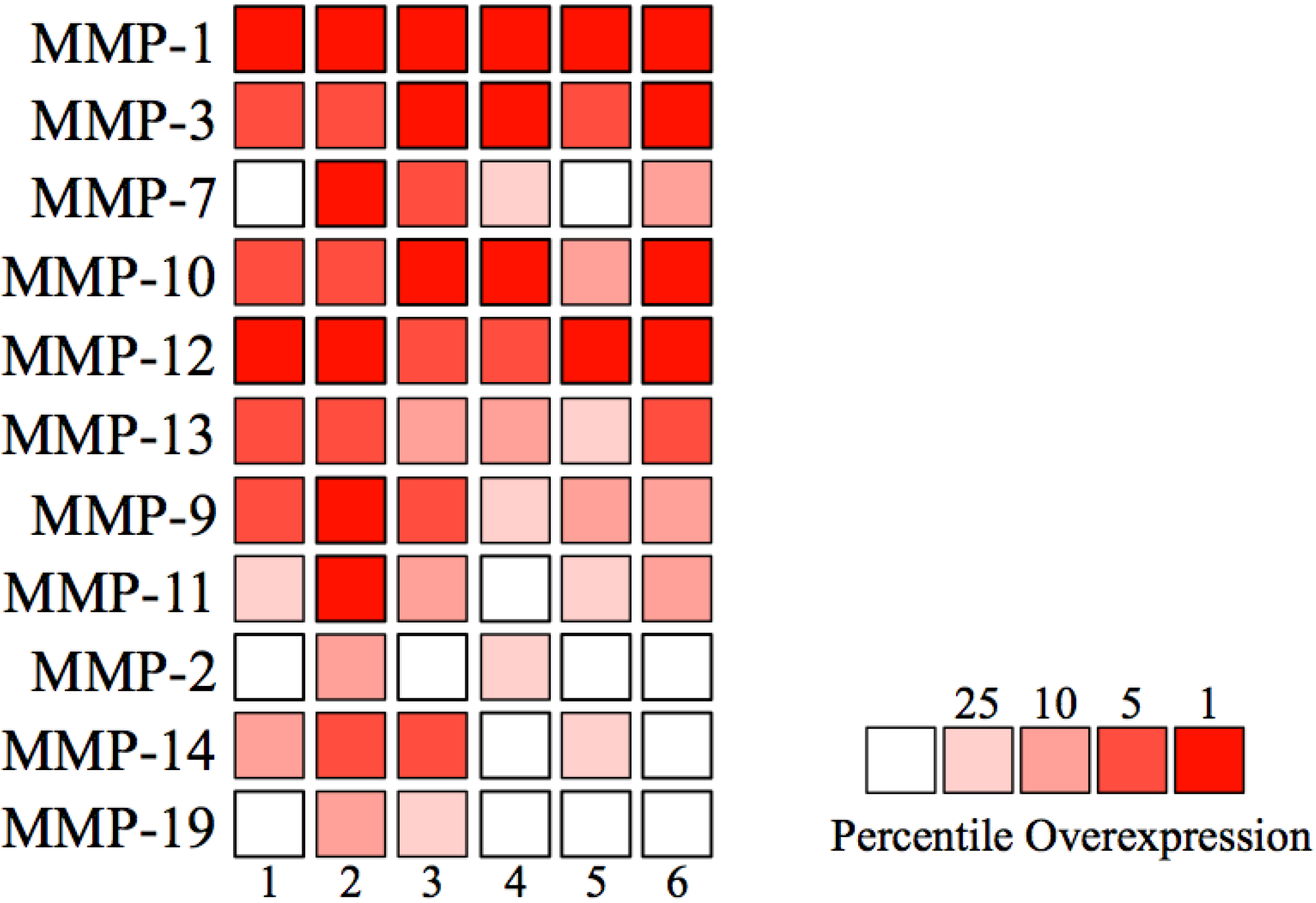
| Number | Author | Number of samples | MMP-1 | MMP-3 | MMP-7 | MMP-10 | MMP-12 | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Normal | HNSCC | Fold Change | p value | Fold Change | p value | Fold Change | p value | Fold Change | p value | Fold Change | p value | ||
| 1 | Estilo | 26 | 31 | 61.7 | * | 14.6 | * | 3.0 | * | 23.6 | * | 5.8 | * |
| 2 | Ginos | 13 | 41 | 221.8 | * | 11.9 | * | 7.5 | * | 14.9 | * | 15.6 | * |
| 3 | Peng | 22 | 57 | 86.3 | * | 29.9 | * | 4.8 | * | 25.6 | * | 17.3 | * |
| 4 | Pyeon | 10 | 15 | 137.5 | * | 19.0 | * | 3.8 | * | 24.1 | * | 27.6 | * |
| 5 | Talbot | 26 | 31 | 27.7 | * | 3.5 | * | 1.5 | 0.066 | 4.0 | * | 4.1 | * |
| 6 | Ye | 12 | 26 | 124.6 | * | 10.6 | * | 2.2 | * | 6.4 | * | 7.3 | * |
| Total | 109 | 201 | |||||||||||
| Number | Author | MMP-13 | MMP-9 | MMP-11 | MMP-2 | MMP-14 | MMP-19 | ||||||
| Fold Change | P value | Fold Change | p value | Fold Change | p value | Fold Change | p value | Fold Change | p value | Fold Change | p value | ||
| 1 | Estilo | 20.3 | * | 9.5 | * | 2.3 | * | 1.1 | 0.356 | 1.9 | * | -1.2 | 0.953 |
| 2 | Ginos | 15.2 | * | 11.8 | * | 5.3 | * | 2.2 | * | 1.8 | * | 8.8 | * |
| 3 | Peng | 9.2 | * | 3.8 | * | 2.3 | * | 1.4 | * | 2.1 | * | 1.2 | * |
| 4 | Pyeon | 4.3 | * | 8.1 | * | 1.4 | * | 1.8 | * | 1.0 | 0.257 | 1.1 | 0.286 |
| 5 | Talbot | 4.7 | * | 3.2 | * | 1.6 | * | 1.0 | 0.439 | 1.1 | * | -1.2 | 1 |
| 6 | Ye | 4.0 | * | 4.6 | * | 1.5 | * | 1.1 | 0.055 | 1.1 | 0.159 | 1.1 | 0.201 |
3. Roles of MMPs in HNSCCs
3.1. MMP-1 in HNSCCs
3.2. MMP-3 in HNSCCs
3.3. MMP-7 in HNSCCs
3.4. MMP-10 in HNSCCs
3.5. MMP-12 in HNSCCs
3.6. MMP-13 in HNSCCs
3.7. MMP-14 (MT1-MMP) in HNSCCs
3.8. MMP-19 in HNSCCs
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Jemal, A.; Siegel, R.; Xu, J.; Ward, E. Cancer statistics, 2010. CA Cancer J. Clin. 2010, 60, 277–300. [Google Scholar] [CrossRef]
- Siegel, R.; DeSantis, C.; Virgo, K.; Stein, K.; Mariotto, A.; Smith, T.; Cooper, D.; Gansler, T.; Lerro, C.; Fedewa, S.; et al. Cancer treatment and survivorship statistics, 2012. CA Cancer J. Clin. 2012, 62, 220–241. [Google Scholar] [CrossRef]
- Neville, B.W.; Damm, D.D.; Allen, C.M.; Bouquot, J.E. Epithelial Pathology. In Oral and Maxillofacial Pathology, 3rd ed.; Neville, B.W., Ed.; W B Saunders: Philadelphia, PA, USA, 1995; pp. 627–638. [Google Scholar]
- Kudo, Y.; Kitajima, S.; Ogawa, I.; Hiraoka, M.; Sargolzaei, S.; Keikhaee, R.; Sato, S.; Miyauchi, M.; Takashi, T. Invasion and metastasis of oral cancer cells require methylation of E-cadherin and/or degradation of membranous β-catenin. Clin. Cancer Res. 2004, 10, 5455–5463. [Google Scholar] [CrossRef]
- Werner, J.A.; Rathcke, I.O.; Mandic, R. The role of matrix metalloproteinases in squamous cell carcinomas of the head and neck. Clin. Exp. Metastasis 2002, 19, 25–282. [Google Scholar] [CrossRef]
- Milner, J.M.; Cawston, T.E. Matrix metalloproteinase knockout studies and the potential use of matrix metalloproteinase inhibitors in the rheumatic diseases. Curr. Drug Targets Inflamm. Allergy 2005, 4, 363–375. [Google Scholar] [CrossRef]
- Puente, X.S.; López-Otín, C. A genomic analysis of rat proteases and protease inhibotors. Genome Res. 2004, 14, 609–622. [Google Scholar] [CrossRef]
- Kurahara, S.; Shinohara, M.; Ikebe, T.; Nakamura, S.; Beppu, M.; Hiraki, A.; Takeuchi, H.; Shirasuna, K. Expression of MMPs, MT-MMP, and TIMPs in squamous cell carcinoma of oral cavity. Head Neck 1999, 21, 627–638. [Google Scholar] [CrossRef]
- Kawata, R.; Shimada, T.; Maruyama, S.; Hisa, Y.; Takenaka, H.; Murakami, M. Enhanced production of matrix metalloproteinase-2 in human head and neck carcinomas is correlated with lymph node metastasis. Acta Otolaryngol. 2002, 122, 101–106. [Google Scholar] [CrossRef]
- Kudo, Y.; Ogawa, I.; Kitajima, S.; Kitagawa, M.; Kawai, H.; Gaffney, P.M.; Miyauchi, M.; Takata, T. Periostin promotes invasion and anchorage-independent growth in the metastatic process of head and neck cancer. Cancer Res. 2006, 66, 6928–6935. [Google Scholar] [CrossRef]
- Hatano, H.; Kudo, Y.; Ogawa, I.; Tsunematsu, T.; Kikuchi, A.; Abiko, Y.; Takata, T. IFN-induced transmembrane protein 1 promotes invasion at early stage of head and neck cancer progression. Clin. Cancer Res. 2008, 14, 6097–6105. [Google Scholar] [CrossRef]
- Deraz, E.M.; Kudo, Y.; Yoshida, M.; Obayashi, M.; Tsunematsu, T.; Tani, H.; Siriwardena, S.B.; Keikhaee, M.R.; Qi, G.; Iizuka, S.; et al. MMP-10/stromelysin-2 promotes invasion of head and neck cancer. PLoS One 2011, 6, e25438. [Google Scholar] [CrossRef]
- Pyeon, D.; Michael, A.N.; Paul, F.; Lambert, P.F.; den Boon, J.A.; Sengupta, S.; Marsit, C.J.; Woodworth, C.D.; Connor, J.P.; Haugen, T.H.; et al. Fundamental differences in cell cycle deregulation in human papillomavirus-positive and human papillomavirus-negative head/neck and cervical cancers. Cancer Res. 2007, 67, 4605–4619. [Google Scholar] [CrossRef]
- Stokes, A.; Joutsa, J.; Ala-aho, R.; Pitchers, M.; Pennington, C.J.; Martin, C.; Premachandra, D.J.; Okada, Y.; Peltonen, J.; Grénman, R.; et al. Expression Profiles and clinical correlations of degradome components in the tumor microenvironment of head and neck squamous cell carcinoma. Clin. Cancer Res. 2010, 16, 2022–2035. [Google Scholar] [CrossRef]
- Ye, H.; Yu, T.; Temam, S.; Ziober, B.L.; Wang, J.; Schwartz, J.L.; Mao, L.; Wong, D.T.; Zhou, X. Transcriptomic dissection of tongue squamous cell carcinoma. BMC Genomics 2008, 9, 69. [Google Scholar] [CrossRef]
- Rhodes, D.R.; Yu, J.; Shanker, K.; Deshpande, N.; Varambally, R.; Ghosh, D.; Barrete, T.; Pandey, A.; Chinnaiyan, A.M. ONCOMINE: A cancer microarray database and integrated data-mining platform. Neoplasia 2004, 6, 1–6. [Google Scholar]
- Stetler-Stevenson, W.G.; Liotta, L.A.; Kleiner, D.E. Extracellular matrix 6: Role of matrix metalloproteinases in tumor invasion and metastasis. FASEB J. 1993, 7, 1434–1441. [Google Scholar]
- Nagase, H.; Woessner, J.F. Matrix metalloproteinases. Semin. Cancer Biol. 1999, 274, 21491–21494. [Google Scholar]
- Stetler-Stevenson, W.G.; Yu, A.E. Proteases in invasion: Matrix metalloproteinases. Semin. Cancer Biol. 2001, 11, 143–152. [Google Scholar] [CrossRef]
- Sternlicht, M.D.; Werb, Z. How matrix metalloproteinases regulate cell behavior. Annu. Rev. Cell Dev. Biol. 2001, 17, 463–516. [Google Scholar] [CrossRef]
- Visse, R.; Nagase, H. Matrix metalloproteinases and tissue inhibitors of metalloproteinases: Structure, function, and biochemistry. Circ. Res. 2003, 92, 827–839. [Google Scholar] [CrossRef]
- Amălinei, C.; Căruntu, I.D.; Giuşcă, S.E.; Bălan, R.A. Matrix metalloproteinases involvement in pathologic conditions. Rom. J. Morphol. Embryol. 2010, 51, 215–228. [Google Scholar]
- Freije, J.M.; Balbín, M.; Pendás, A.M.; Sánchez, L.M.; Puente, X.S.; López-Otín, C. Matrix metalloproteinases and progression. Adv. Exp. Biol. 2003, 532, 91–107. [Google Scholar] [CrossRef]
- D’Armiento, J.; DiColandrea, T.; Dalal, S.S.; Okada, Y.; Huang, M.T.; Conney, A.H.; Chada, K. Collagenase expression in transgenic mouse skin causes hyperkeratosis and acanthosis and increases susceptibility to tumorigenesis. Mol. Cell. Biol. 1995, 15, 5732–5739. [Google Scholar]
- Boire, A.; Covic, L.; Agarwal, A.; Jacques, S.; Sheriffi, S.; Kulippulos, K. PAR1 is a matrix metalloprotease-1 receptor that promotes invasion and tumorigenesis of breast cancer cells. Cell 2005, 120, 303–313. [Google Scholar] [CrossRef]
- Blackburn, J.S.; Brinckerhoff, C.E. Matrix metalloprotease-1 and thrombin differentially activate gene expression in endothelial cells via PAR-1 and promote angiogenesis. Am. J. Pathol. 2008, 173, 1736–1746. [Google Scholar] [CrossRef]
- Liu, H.; Kato, Y.; Erzinger, S.A.; Kiriakova, G.M.; Qian, Y.; Palmieri, D.; Steeg, P.S.; Price, J.E. The role of MMP-1 in breast cancer growth and metastasis to the brain in a xenograft model. BMC Cancer 2012, 12, 583. [Google Scholar] [CrossRef]
- Johansson, N.; Airola, K.; Grénman, R.; Kariniemi, A.L.; Saarialho-Kere, U.; Kähäri, V.M. Expression of collagenase-3 (Matrix metalloproteinase-13) in squamous cell carcinomas of the head and neck. Am. J. Pathol. 1997, 151, 499–508. [Google Scholar]
- Gray, S.T.; Wilkins, R.J.; Tyun, K. Interstitial collagenase gene expression in oral squamous cell carcinoma. Am. J. Pathol. 1992, 141, 301–306. [Google Scholar]
- Polette, M.; Clavel, C.; Muller, D.; Abecassis, J.; Binninger, I.; Birembaut, P. Detection o30mRNAs encoding collagenase 1 and stromelysin 2 in carcinomas of the head and neck by in situ hybridization. Invasion Metastasis 1991, 11, 76–83. [Google Scholar]
- O-Charoenrat, P.; Rhys-Evans, P.H.; Eccles, S.A. Expression of matrix metalloproteinases and their inhibitors correlates with invasion and metastasis in squamous cell carcinoma of the head and neck. Arch Otolaryngol. Head Neck Surg. 2001, 127, 813–820. [Google Scholar]
- Sun, Y.; Zeng, X.R.; Wenger, L.; Firestein, G.S.; Cheung, H.S. P53 down-regulates matrix metalloproteinase-1 by targeting the communications between AP-1 and the basal transcription complex. J. Cell. Biochem. 2004, 92, 258–269. [Google Scholar] [CrossRef]
- Sun, Y.; Sun, Y.I.; Wenger, L.; Rutter, J.L.; Brinckerhoff, C.E.; Cheung, H.S. P53 down-regulates human metalloproteinases-1 (collagenase-1) gene expression. J. Biol. Chem. 1999, 274, 11535–11540. [Google Scholar]
- Singh, P.K.; Behrens, M.E.; Eggers, J.P.; Cerny, R.L.; Bailery, J.M.; Shanmugam, K.; Gendler, S.J.; Bennett, E.P.; Hollingsworth, M.A. Phosphorylation of MUC1 by Met modulates interaction with p53 ad MMP1 expression. J. Biol. Chem. 2008, 283, 26985–26995. [Google Scholar] [CrossRef]
- Leemans, C.R.; Braakhuis, B.J.M.; Brakenhoff, R.H. The molecular biology of head and neck cancer. Nat. Rev. Cancer 2011, 11, 9–22. [Google Scholar] [CrossRef]
- Poeta, M.L.; Manola, J.; Goldwasser, M.A.; Forastiere, A.; Benoit, N.; Califano, J.A.; Ridge, J.A.; Goodwin, J.; Kenady, D.; Saunders, J.; et al. TP53 mutations and survival in squamous-cell carcinoma of the head and neck. N. Engl. J. Med. 2007, 357, 2552–2561. [Google Scholar] [CrossRef]
- Nagase, H. Stromelysins 1 and 2. In Matrix Metalloproteinases; Parks, W.C., Mecham, R.P., Eds.; Academic Press: San Diego, CA, USA, 1998; pp. 43–84. [Google Scholar]
- Abramson, S.R.; Conner, G.E.; Nagase, H.; Neuhaus, I.; Woessner, J.F., Jr. Characterization of rat uterine matrilysin and its cDNA relationship to human pump-1 and activation of procollagease. J. Biol. Chem. 1995, 270, 16016–16022. [Google Scholar]
- Bramhall, S.R.; Stamp, G.W.; Dunn, J.; Lemoine, N.R.; Neoptolemos, J.P. Expression of collagenase (MMP2), stromelysin (MMP3) and tissue inhibitor of the metalloproteinases (TIMP1) in pancreatic and ampullary disease. Br. J. Cancer 1996, 73, 972–978. [Google Scholar] [CrossRef] [Green Version]
- Nozaki, S.; Sledge, G.W., Jr.; Nakshatri, H. Cancer cell-derived interleukin 1alpha contributes to autocrine and paracrine induction of pro-metastatic genes in breast cancer. Biochem. Biophys. Res. Commun. 2000, 18, 60–62. [Google Scholar]
- Sternlicht, M.D.; Lochter, A.; Sympson, C.J.; Huey, B.; Rougier, J.P.; Gray, J.W.; Pinkel, D.; Bissell, M.J.; Werb, Z. The stromal proteinase MMP3/stromelysin-1 promotes mammary carcinogenesis. Cell 1999, 23, 137–146. [Google Scholar]
- Kessenbrock, K.; Dijkgraaf, G.J.; Lawson, D.A.; Littlepage, L.E.; Shahi, P.; Pieper, U.; Werb, Z. A role for matrix metalloproteinases in regulating mammary stem cell function via the Wnt signaling pathway. Cell Stem Cell 2013, 13, 300–313. [Google Scholar] [CrossRef]
- McCawley, L.J.; Crawford, H.C.; King, L.E., Jr.; Mudgett, J.; Matrisian, L.M. A protective role for matrix metalloproteinase-3 in squamous cell carcinoma. Cancer Res. 2004, 64, 6965–6972. [Google Scholar] [CrossRef]
- Mitra, D.; Malkoski, S.P.; Wang, X.J. Cancer stem cells in head and neck cancer. Cancers 2011, 3, 415–427. [Google Scholar] [CrossRef]
- Prince, M.E.; Sivanandan, R.; Kaczorowski, A.; Wolf, G.T.; Kaplan, J.J.; Dalerba, P.; Weissman, I.L.; Clarke, M.F.; Ailles, L.E. Identification of a subpopulation of cells with cancer stem cell properties in head and neck squamous cell carcinoma. Proc. Natl. Acad. Sci. USA 2007, 104, 973–978. [Google Scholar] [CrossRef]
- Wu, M.J.; Jan, C.I.; Tsay, Y.G.; Yu, Y.H.; Huang, C.Y.; Lin, S.C.; Liu, C.J.; Chen, Y.S.; Lo, J.F.; Yu, C.C. Elimination of head and neck cancer initiating cells through targeting glucose regulated protein78 signaling. Mol. Cancer 2010, 9, 283. [Google Scholar] [CrossRef]
- Samukange, V.; Yasukawa, K.; Inoue, K. Interaction of 8-anilinonaphthalene 1-sulphonate (ANS) and human matrix metalloproteinase 7 (MMP-7) as examined by MMP-7 activity and ANS fluorescence. J. Biochem. 2012, 151, 533–540. [Google Scholar] [CrossRef]
- Ougolkov, A.V.; Yamashita, K.; Mai, M.; Minamoto, T. Oncogenic beta-catenin and MMP-7 (matrilysin) cosegregate in late-stage clinical colon cancer. Gastroenterology 2002, 122, 60–71. [Google Scholar] [CrossRef] [Green Version]
- Pajouh, M.S.; Nagle, R.B.; Breathnach, R.; Finch, J.S.; Brawer, M.K.; Bowdern, G.T. Expression of metalloproteinase genes in human prostate cancer. J. Cancer Res. Clin. Oncol. 1991, 117, 144–150. [Google Scholar] [CrossRef]
- Yoshimoto, M.; Itoh, F.; Yamamoto, H.; Hinoda, Y.; Imai, K.; Yachi, A. Expression of MMP-7 (PUMP-1) mRNA in human colorectal cancers. Int. J. Cancer 1993, 54, 614–618. [Google Scholar] [CrossRef]
- Adachi, Y.; Itoh, F.; Yamamoto, H.; Matsuno, K.; Arimura, Y.; Kusano, M.; Endoh, T.; Hinoda, Y.; Oohara, M.; Hosokawa, M.; et al. Matrix metalloproteinase matrilysin (MMP-7) participates in the progression of human gastric and esophageal cancers. Int. J. Oncol. 1998, 13, 1031–1035. [Google Scholar]
- Chuang, H.C.; Su, C.Y.; Huang, H.Y.; Huang, C.C.; Chien, C.Y.; Du, Y.Y.; Chuang, J.H. Active matrix metalloproteinase-7 is associated with invasion in buccal squamous cell carcinoma. Mod. Pathol. 2008, 21, 1444–1450. [Google Scholar] [CrossRef]
- Kimura, R.; Kasamatsu, A.; Koyama, T.; Fukumoto, C.; Kouzu, Y.; Higo, M.; Endo-Sakamoto, Y.; Ogawara, K.; Shiiba, M.; Tanzawa, H.; et al. Glutamate acid decarboxylase 1 promotes metastasis of human oral cancer by beta-catenin translocation and MMP7 activation. BMC Cancer 2013, 13, 555. [Google Scholar] [CrossRef]
- Dey, N.; Young, B.; Abramovitz, M.; Bouzyk, M.; Barwick, B.; De, P.; Leyland-Jones, B. Differential activation of Wnt-β-catenin pathway in triple negative breast cancer increases MMP7 in a PTEN dependent manner. PLoS One 2013, 8, e77425. [Google Scholar]
- Stransky, N.; Egloff, A.M.; Tward, A.D.; Kostic, A.D.; Cibulskis, K.; Sivachenko, A.; Kryukov, G.V.; Lawrence, M.S.; Sougnez, C.; McKenna, A.; et al. The mutational landscape of head and neck squamous cell carcinoma. Science 2011, 6046, 1157–1160. [Google Scholar]
- Poetsch, M.; Lorenz, G.; Kleist, B. Detection of new PTEN/MMAC1 mutations in head and neck squamous cell carcinomas with loss of chromosome 10. Cancer Genet. Cytogenet. 2002, 1, 20–24. [Google Scholar] [CrossRef]
- Shao, X.; Tandon, R.; Samara, G.; Kanki, H.; Yano, H.; Close, L.G.; Parsons, R.; Sato, T. Mutational analysis of the PTEN gene in head and neck squamous cell carcinoma. Int. J. Cancer 1998, 5, 684–688. [Google Scholar]
- Sirum, K.; Brinckerhoff, C.E. Cloning of the genes for human stromelysin and stromelysin 2: Differential expression in rheumatoid synovial fibroblasts. Biochemistry 1989, 28, 8691–8698. [Google Scholar] [CrossRef]
- Saarialho-Kere, U.K.; Pentland, A.P.; Birkedal-Hansen, H.; Parks, W.C.; Welgus, H.G. Distinct populations of basal keratinocytes express stromelysin-1 and stromelysin-2 in chronic wounds. J. Clin. Investig. 1994, 94, 79–88. [Google Scholar] [CrossRef]
- Windsor, L.J.; Grenett, H.; Birkedal-Hansen, B.; Boddern, M.K.; Engler, J.A.; Birkedal-Hansen, H. Cell type-specific regulation of SL-1 and SL-2 genes. Induction of the SL-2 gene but not the SL-1 gene by human keratinocytes in response to cytokines and phorbolesters. J. Biol. Chem. 1993, 268, 17341–17347. [Google Scholar]
- Madlener, M.; Werner, S. cDNA cloning and expression of the gene encoding murine stromelysin-2 (MMP10). Gene 1997, 202, 75–81. [Google Scholar] [CrossRef]
- Frederick, L.A.; Matthews, J.A.; Jamieson, L.; Justilien, V.; Thompson, E.A.; Radisky, D.C.; Fields, A.P. Matrix metalloproteinase-10 is a critical effector of protein kinase Ciota-Par6alpha-mediated lung cancer. Oncogene 2008, 27, 4841–4853. [Google Scholar] [CrossRef]
- Gill, J.H.; Kirwan, I.G.; Seargent, J.M.; Martin, S.W.; Tijani, S.; Anikin, V.A.; Mearns, A.J.; Bibby, M.C.; Anthone, A.; Loadman, P.M. MMP-10 is overexpressed, proteolytically active, and a potential target for therapeutic intervention in human lung carcinomas. Neoplasia 2004, 6, 777–785. [Google Scholar] [CrossRef]
- Tsang, R.K.; Tang, W.W.; Gao, W.; Ho, W.K.; Chan, J.Y.; Wei, W.I.; Wong, T.S. Curcumin inhibits tongue carcinoma cells migration and invasion through downregulation of matrix metalloproteinase 10. Cancer Investig. 2012, 30, 503–512. [Google Scholar] [CrossRef]
- Epstein, J.; Sanderson, I.R.; Macdonald, T.T. Curcumin as a therapeutic agent: The evidence from in vitro, animal and human studies. Br. J. Nutr. 2010, 103, 1545–1557. [Google Scholar] [CrossRef]
- LoTempio, M.M.; Veena, M.S.; Steele, H.L.; Ramamurthy, B.; Ramalingam, T.S.; Cohen, A.N.; Chakrabarti, R.; Srivatsan, E.S.; Wang, M.B. Curcumin suppresses growth of head and neck squamous cell carcinoma. Clin. Cancer Res. 2005, 11, 6994–7002. [Google Scholar] [CrossRef]
- Chandler, S.; Cossins, J.; Lury, J.; Wells, G. Macrophage metalloelastase degrades matrix and myelin proteins and processes a tumor necrosis factor-alpha fusion protein. Biochem. Biophys. Res. Commun. 1996, 228, 421–429. [Google Scholar] [CrossRef]
- Shipley, J.M.; Wesselschmidt, R.; Kobayashi, D.; Ley, T.; Shapiro, S. Metalloelastase is required for macrophage mediated proteolysis and matrix invasion in mice. Proc. Natl. Acad. Sci. USA 1996, 93, 3942–3946. [Google Scholar] [CrossRef]
- Gronski, T.J., Jr.; Martin, R.L.; Kobayashi, K.D.; Walsh, B.C.; Holman, M.C.; Huber, M.; van Wart, H.E.; Shapiro, S.D. Hydrolysis of a broad spectrum of extracellular matrix proteins by human macrophage elastase. J. Biol. Chem. 1997, 272, 12189–12194. [Google Scholar] [CrossRef]
- Dong, Z.; Kumar, R.; Yang, X.; Fidler, I.J. Macrophage-derived metalloelastase is responsible for the generation of angiostatin in Lewis lung carcinoma. Cell 1997, 88, 801–810. [Google Scholar] [CrossRef]
- Cornelius, L.A.; Nehring, L.C.; Harding, E.; Bolanowski, M.; Welgus, H.G.; Kobayashi, D.K.; Pierce, R.A.; Shapiro, S.D. Matrix metalloproteinase generate angiostatin: Effects on neovascularization. J. Immunol. 1998, 161, 6845–6852. [Google Scholar]
- Kerkelä, E.; Ala-Aho, R.; Jeskanen, L.; Rechardt, O.; Grénman, R.; Shapiro, S.D.; Kähäri, V.M.; Saarialho-Kere, U. Expression of human macrophage metalloelastase (MMP-12) by tumor cells in skin cancer. J. Investig. Dermatol. 2000, 114, 1113–1119. [Google Scholar] [CrossRef]
- Impola, U.; Uitto, V.J.; Hietanen, J.; Hakkinen, L.; Zhang, L.; Larjava, H.; Isaka, K.; Saarialho-Kere, U. Differential expression of matrilysin-1 (MMP-7), 92 kD gelatinase (MMP-9), and metalloelastase (MMP-12) in oral verrucous and squamous cell cancer. J. Pathol. 2004, 202, 14–22. [Google Scholar] [CrossRef]
- Choi, P.; Chen, C. Genetic expression profiles and biologic pathway alterations in head and neck squamous cell carcinoma. Cancer 2005, 104, 1113–1128. [Google Scholar] [CrossRef]
- Thurlow, J.K.; Pena Murillo, C.L.; Hunter, K.D.; Buffa, F.M.; Patiar, S.; Betts, G.; West, C.M.; Harris, A.L.; Parkinson, E.K.; Harrison, P.R.; et al. Spectral clustering of microarray data elucidates the roles of microenvironment remodeling and immune responses in survival of head and neck squamous cell carcinoma. J. Clin. Oncol. 2010, 28, 2881–2888. [Google Scholar] [CrossRef]
- Kerkelä, E.; Ala-aho, R.; Klemi, P.; Grénman, S.; Kähäri, V.M.; Saarialho-Kere, U. Metalloelastase (MMP-12) expression by tumor cells in squamous cell carcinoma of the vulva correlates with invasiveness, while that by macrophages predicts better outcome. J. Pathol. 2002, 198, 258–269. [Google Scholar] [CrossRef]
- Chin, D.; Blyle, G.M.; Williams, R.M.; Ferguson, K.; Pandeya, N.; Pedley, J.; Campbell, C.M.; Theile, D.R.; Parsons, P.G.; Coman, W.B. Novel markers for poor prognosis in head and neck cancer. Int. J. Cancer 2004, 113, 789–797. [Google Scholar]
- Kim, J.M.; Kim, H.J.; Koo, B.S.; Rha, K.S.; Yoon, Y.H. Expression of matrix metalloproteinase-12 is correlated with extracapsular spread of tumor from nodes with metastasis in head and neck squamous cell carcinoma. Eur. Arch. Otorhinolaryngol. 2013, 270, 1137–1142. [Google Scholar] [CrossRef]
- Folkman, J. Tumor Angiogenesis in Cancer. In Medicine, 5th ed.; B.C. Decker, Inc.: Hamilton, ON, Canada, 2000; pp. 132–152. [Google Scholar]
- Littlepage, L.E.; Sternlicht, M.D.; Rougier, N.; Phillips, J.; Gallo, E.; Yu, Y.; Willams, K.; Brenot, A.; Gordon, J.L.; Werb, Z. Matrix metalloproteinases contribute distinct roles in neuroendocrine prostate carcinogenesis, metastasis, and angiogenesis progression. Cancer Res. 2010, 70, 2224–2234. [Google Scholar] [CrossRef]
- Bergers, G.; Brekken, R.; McMahon, G.; Vu, T.H.; Ithoh, T.; Tamaki, K.; Tanzawa, K.; Thorpe, P.; Itohara, S.; Werb, Z.; et al. Matrix metalloproteinase-9 triggers the angiogenic switch during carcinogenesis. Nat. Cell Biol. 2000, 2, 737–774. [Google Scholar] [CrossRef]
- Lee, S.; Jilani, S.M.; Nikolova, G.V.; Carpizao, D.; Iruela-Arispe, M.L. Processing of VEGF-A by matrix metalloproteinases regulates bioavailability and vascular patterning in tumors. J. Cell Biol. 2005, 169, 681–691. [Google Scholar] [CrossRef]
- Ribatti, D. Endogenous inhibitors of angiogenesis: A historical review. Leuk. Res. 2009, 33, 638–644. [Google Scholar] [CrossRef]
- Freije, J.M.; Díez-Itza, I.; Balbín, M.; Sánchez, L.M.; Blasco, R.; Tolivia, J.; López-Otín, C. Molecular cloning and expression of collagenase-3, a novel human matrix metalloproteinase produced by breast carcinomas. J. Biol. Chem. 1994, 269, 16766–16773. [Google Scholar]
- Kosaki, N.; Takaishi, H.; Kamekura, S.; Kimura, T.; Okada, Y.; Minqi, L.; Amizuka, N.; Chung, U.I.; Nakamura, K.; Kawaguchi, H.; et al. Impaired bone fracture healing in matrix metalloproteinase-13 deficient mice. Biochem. Biophys. Res. Commun. 2007, 354, 846–851. [Google Scholar] [CrossRef]
- Zijlstra, A.; Aimes, R.T.; Zhu, D.; Regazzoni, K.; Kupriyanova, T.; Seandel, M.; Deryugina, E.I.; Quigley, J.P. Collagenolysis-dependent angiogenesis mediated by matrix metalloproteinase-13 (collagenase-3). J. Biol. Chem. 2004, 279, 27633–27645. [Google Scholar] [CrossRef]
- Nielsen, B.S.; Rank, F.; López, J.M.; Balbin, M.; Vizoso, F.; Lund, L.R.; Danø, K.; López-Otín, C. Collagenase-3 expression in breast myofibroblasts as a molecular marker of transition of ductal carcinoma in situ lesions to invasive ductal carcinomas. Cancer Res. 2001, 61, 7091–7100. [Google Scholar]
- Lederle, W.; Hartenstein, B.; Meides, A.; Kunzelmann, H.; Werb, Z.; Angel, P.; Mueller, M.M. MMP13 as a stromal mediator in controlling persistent angiogenesis in skin carcinoma. Carcinogenesis 2010, 31, 1175–1184. [Google Scholar] [CrossRef]
- Kudo, Y.; Iizuka, S.; Yoshida, M.; Tsunematsu, T.; Kondo, T.; Subarnbhesaj, A.; Deraz, E.M.; Siriwardena, S.B.S.M.; Tahara, H.; Ishimaru, N.; et al. Matrix metalloproteinase-13 (MMP-13) directly and indirectly promotes tumor angiogensis. J. Biol. Chem. 2012, 287, 38716–38728. [Google Scholar] [CrossRef]
- Luukkaa, M.; Vihinen, P.; Kronqvist, P.; Vahlberg, T.; Pyrhonen, S.; Kähäri, V.M.; Grénman, R. Association between high collagenase-3 expression levels and poor prognosis in patients with head and neck cancer. Head Neck 2006, 28, 225–234. [Google Scholar] [CrossRef]
- Murphy, D.A.; Courtneidge, S.A. The “ins” and “outs” of podosomes and invadopodia; characteristics, formation and function. Nat. Rev. Mol. Cell Biol. 2011, 12, 413–426. [Google Scholar] [CrossRef]
- Artym, V.V.; Matsumoto, K.; Mueller, S.C.; Yamada, K.M. Dynamic membrane remodeling at invadopodia differentiates invadopodia from podosomes. Eur. J. Cell Biol. 2011, 90, 172–180. [Google Scholar]
- Yu, X.; Machesky, L.M. Cells assemble invadopodia-like structures and invade into matrigel in a matrix metalloprotease dependent manner in the circular invasion assay. PLoS One 2012, 7, e30605. [Google Scholar]
- Eckert, M.A.; Yang, J. Targeting invadopodia to block breast cancer metastasis. Oncotarget 2011, 2, 562–568. [Google Scholar]
- Díaz, B.; Yuen, A.; Iizuka, S.; Higashiyama, S.; Courtneidge, S.A. Notch increases the shedding of HB-EGF by ADAM12 to potentiate invadopodia formation in hypoxia. J. Cell Biol. 2013, 201, 279–292. [Google Scholar] [CrossRef]
- Stylli, S.S.; Kaye, A.H.; Lock, P. Invadopodia; at the cutting edge of tumor invasion. J. Clin. Neurosci. 2008, 15, 725–737. [Google Scholar] [CrossRef]
- Linder, S. The matrix corroded: Podosomes and invadopodia in extracellular matrix degradation. Trends Cell Biol. 2007, 17, 107–117. [Google Scholar]
- Clark, E.S.; Weaver, A.M. A new role for cortactin in invadopodia: Regulation of protease secretion. Eur. J. Cell Biol. 2008, 87, 581–590. [Google Scholar] [CrossRef]
- Clark, E.S.; Whigham, A.S.; Yarbrough, W.G.; Weaver, A.M. Cortactin is an essential regulator of matrix metalloproteinase secretion and extracellular matrix degradation in invadopodia. Cancer Res. 2007, 67, 4227–4235. [Google Scholar] [CrossRef]
- Buschman, M.D.; Bromann, P.A.; Cejudo-Martin, P.; Wen, F.; Pass, I.; Courtneidge, S.A. The novel adaptor protein Tks4 (SH3PXD2B) is required for functional podosome formation. Mol. Biol. Cell 2009, 20, 1302–1311. [Google Scholar] [CrossRef]
- Seals, D.F.; Azucena, E.F., Jr.; Pass, I.; Tesfay, L.; Gordon, R.; Woodrow, M.; Resau, J.H.; Courtneidge, S.A. The adaptor protein Tks5/Fish is required for podosome formation and function, and for the protease driven invasion of cancer cells. Cancer Cell 2005, 7, 155–165. [Google Scholar] [CrossRef]
- Blouw, B.; Seals, D.F.; Pass, I.; Díaz, B.; Courtneidge, S.A. A role for the podosome/invadopodia scaffold protein Tks5 in tumor growth in vivo. Eur. J. Cell Biol. 2008, 87, 555–567. [Google Scholar]
- Hotary, K.; Allen, E.; Punturieri, A.; Yana, I.; Weiss, S.J. Regulation of cell invasion and morphogenesis in a three-dimensional type 1 collagen matrix by membrane-type matrix metalloproteinases 1, 2, and 3. J. Cell Biol. 2000, 149, 1309–1323. [Google Scholar] [CrossRef]
- Hotary, K.B.; Allen, E.D.; Brooks, P.C.; Datta, N.S.; Long, M.W.; Weiss, S.J. Membrane type 1 matrix metalloproteinase usurps tumor growth control imposed by the three-dimensional extracellular matrix. Cell 2003, 114, 33–45. [Google Scholar] [CrossRef]
- Clark, E.S.; Brown, B.; Whigham, A.S.; Kochaishvili, A.; Yarbrough, W.G.; Weaver, A.M. Aggressiveness of HNSCC tumors depends on expression levels of cortactin, a gene in the 11q13 amplicon. Oncogene 2009, 28, 431–444. [Google Scholar] [CrossRef]
- Yoshizaki, T.; Sato, H.; Maruyama, Y.; Murono, S.; Furukawa, M.; Park, C.S.; Seiki, M. Increased expression of membrane type 1-matrix metalloproteinase in head and neck carcinoma. Cancer 1997, 79, 139–144. [Google Scholar] [CrossRef]
- Imanishi, Y.; Fujii, M.; Tokumaru, Y.; Tomita, T.; Kanke, M.; Kanzaki, J.; Kameyama, K.; Otani, Y.; Sato, H. Clinical significance of expression of membrane type 1 matrix metalloproteinase and matrix metalloproteinase-2 in human head and neck squamous cell carcinoma. Hum. Pathol. 2000, 31, 895–904. [Google Scholar] [CrossRef]
- Zhang, W.; Matrisian, L.M.; Holmbeck, K.; Vick, C.C.; Rosenthal, E.L. Fibroblast-derived MT1-MMP promotes tumor progression in vitro and in vivo. BMC Cancer 2006, 6, 52. [Google Scholar] [CrossRef] [Green Version]
- Basile, J.R.; Holmbeck, K.; Bugge, T.H.; Gutkind, S. MT1-MMP controls tumor-induced angiogenesis through the release of semaphoring 4D. J. Biol. Chem. 2007, 282, 6899–6905. [Google Scholar]
- Cossins, J.; Dudgeon, T.J.; Catlin, G.; Gearing, A.J.; Clements, J.M. Identification of MMP-18, a putative novel human matrix metalloproteinase. Biochem. Biophys. Res. Commun. 1996, 12, 494–498. [Google Scholar]
- Pendás, A.M.; Knäuper, V.; Puente, X.S.; Llano, E.; Mattei, M.G.; Apte, S.; Murphy, G.; López-Otín, C. Identification and characterization of a novel human marix metalloproteinase with unique structural characteristics, chromosomal location, and tissue distribution. J. Biol. Chem. 1997, 14, 4281–4286. [Google Scholar]
- Stracke, J.O.; Fosang, A.J.; Last, K.; Mercuri, F.A.; Pendás, A.M.; Llano, E.; Perris, R.; di Cesare, P.E.; Murphy, G.; Knáuper, V. Matrix metalloproteinases 19 and 20 cleave aggrecan and cartilage oligomeric matrix protein (COMP). FEBS Lett. 2000, 28, 52–56. [Google Scholar]
- Stracke, J.O.; Hutton, M.; Stewart, M.; Pendás, A.M.; Smith, B.; Lopez-Otin, C.; Murph, G.; Knáuper, V. Biochemical characterization of the catalytic domain of human matrix metalloproteinase 19. Evidence for a role as a potent basement membrane degrading enzyme. J. Biol. Chem. 2000, 19, 14809–14816. [Google Scholar]
- Djonov, V.; Högger, K.; Sedlacek, R.; Laissue, J.; Draeger, A. MMP-19: Cellular localization of a novel metalloproteinase within normal breast tissue and mammary gland tumors. J. Pathol. 2001, 195, 147–155. [Google Scholar] [CrossRef]
- Velinov, N.; Aebersold, D.; Haeni, N.; Hlushchuk, R.; Mishev, G.; Weinstein, F.; Sedlacek, R.; Djonov, V. Matrix metalloproteinase-19 is a predictive marker for tumor invasiveness in patients with oropharyngeal squamous cell carcinoma. Int. J. Biol. Markers 2007, 22, 265–273. [Google Scholar]
- Müller, M.; Beck, I.M.; Gadesmann, J.; Karschuk, N.; Paschen, A.; Proksch, E.; Djonov, V.; Sedlacek, R. MMP19 is upregulated during melanoma progression and increases invasion of melanoma cells. Mod. Pathol. 2010, 23, 511–521. [Google Scholar] [CrossRef]
- Zhao, H.; Yang, Z.; Wang, X.; Wang, M.; Wang, Y.; Mei, Q.; Wang, Z. Triptolide inhibits ovarian cancer cell invasion by repression of matrix metalloproteinase 7 and 19 and upregulation of E-cadherin. Exp. Mol. Med. 2012, 30, 633–641. [Google Scholar]
- Wang, L.; Yuan, J.; Tu, Y.; Mao, X.; He, S.; Fu, G.; Zong, J.; Zhang, Y. Co-expression of MMP-14 and MMP-19 predicts poor survival in human glioma. Clin. Transl. Oncol. 2013, 15, 139–145. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Iizuka, S.; Ishimaru, N.; Kudo, Y. Matrix Metalloproteinases: The Gene Expression Signatures of Head and Neck Cancer Progression. Cancers 2014, 6, 396-415. https://doi.org/10.3390/cancers6010396
Iizuka S, Ishimaru N, Kudo Y. Matrix Metalloproteinases: The Gene Expression Signatures of Head and Neck Cancer Progression. Cancers. 2014; 6(1):396-415. https://doi.org/10.3390/cancers6010396
Chicago/Turabian StyleIizuka, Shinji, Naozumi Ishimaru, and Yasusei Kudo. 2014. "Matrix Metalloproteinases: The Gene Expression Signatures of Head and Neck Cancer Progression" Cancers 6, no. 1: 396-415. https://doi.org/10.3390/cancers6010396
APA StyleIizuka, S., Ishimaru, N., & Kudo, Y. (2014). Matrix Metalloproteinases: The Gene Expression Signatures of Head and Neck Cancer Progression. Cancers, 6(1), 396-415. https://doi.org/10.3390/cancers6010396





