Investigation of siRNA Nanoparticle Formation Using Mono-Cationic Detergents and Its Use in Gene Silencing in Human HeLa Cells
Abstract
:1. Introduction
2. Results and Discussion
2.1. Preparation of siRNA/MCD Complex
| Group | Name | Basic structure | R (L-chain) |
|---|---|---|---|
| Type-C | C-1 |  | -C14H25 |
| C-2 | -C16H33 | ||
| Type-D | D-1 | 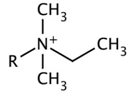 | -C12H25 |
| D-2 | -C16H33 | ||
| Type-E | E-1 | 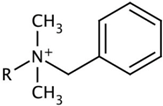 | -C12H25 |
| E-2 | -C14H29 | ||
| E-3 | -C16H33 |

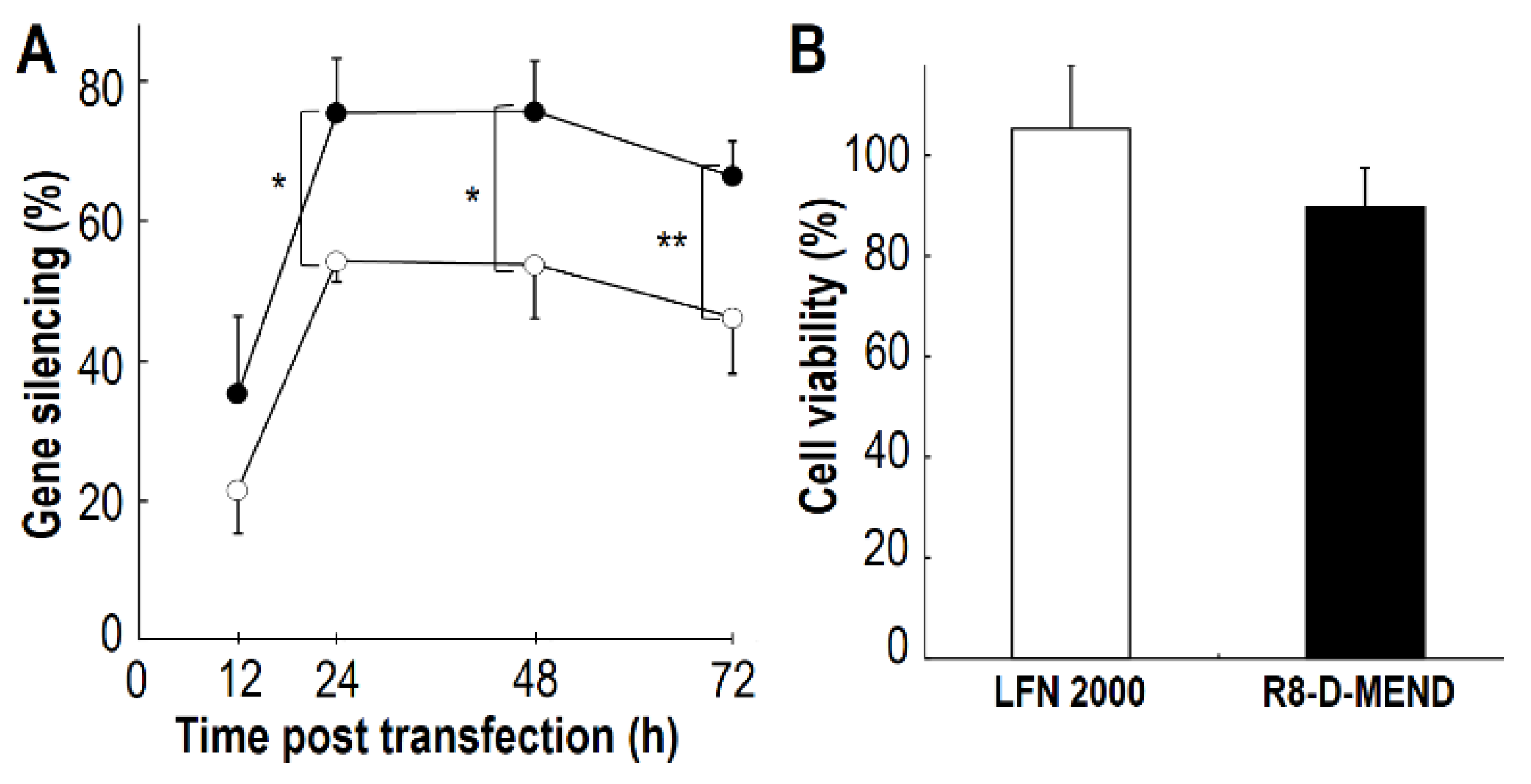
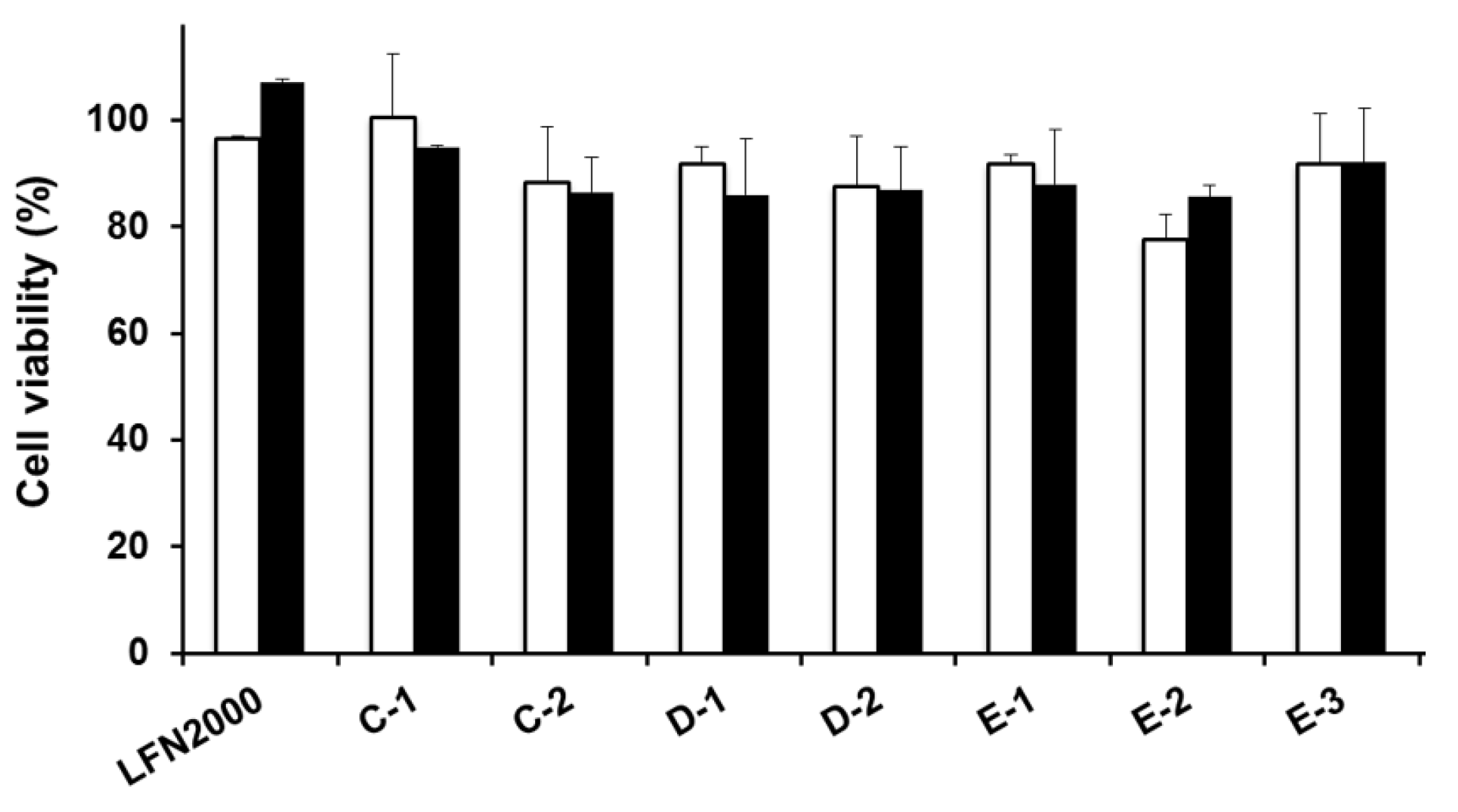
2.2. Comparison of Gene Silencing Efficiencies and Cell Viabilities Between siRNA/MCD Nanoparticles and LFN 2000
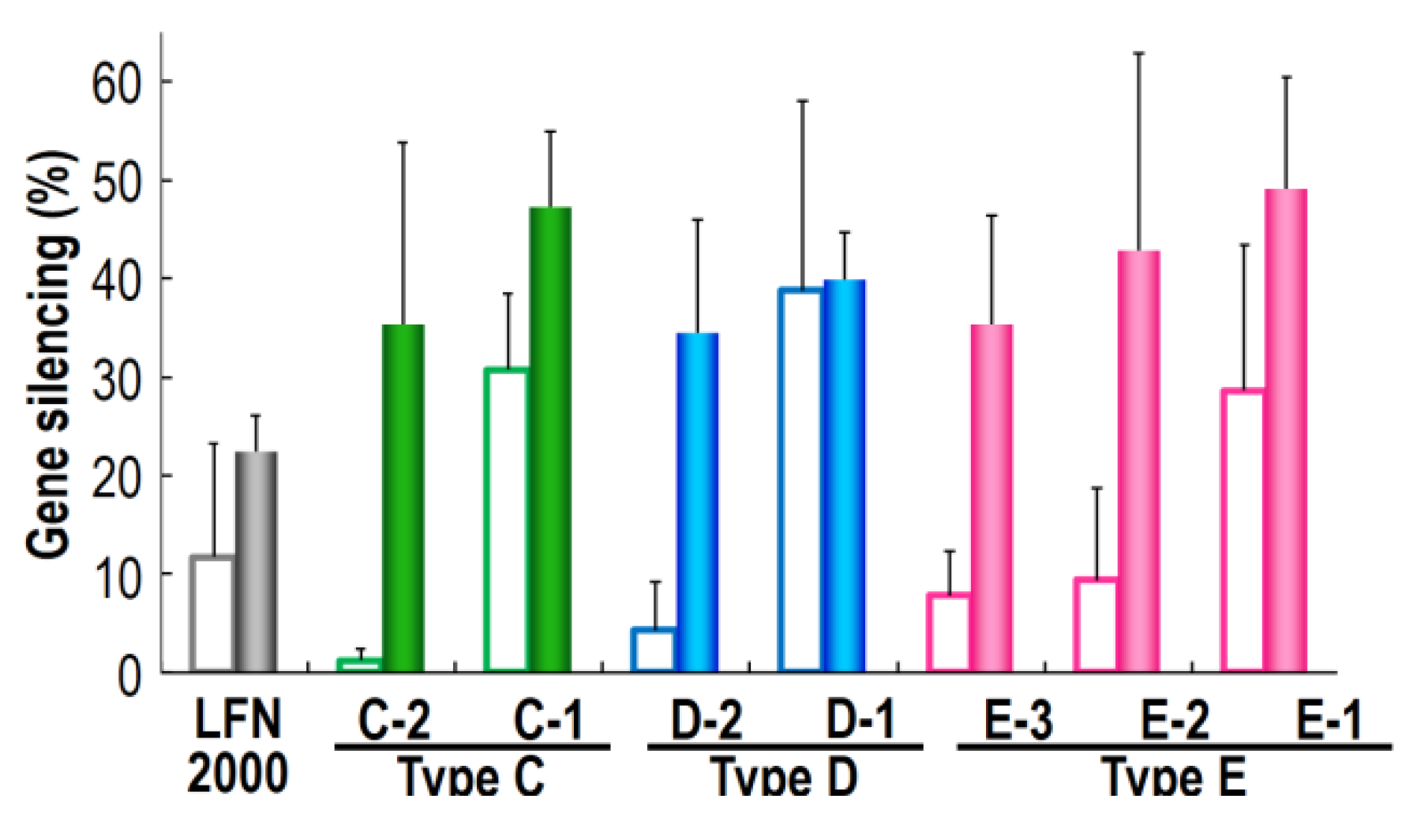
2.3. Investigation of siRNA Nanoparticle Formed with Various Type of MCD on Gene Silencing Efficiencies
| MCD (N/P ratio) | siRNA/MCD nanoparticle | R8-D-MEND | ||||
|---|---|---|---|---|---|---|
| Size (nm) | Zeta potential (mV) | PDI | Size (nm) | Zeta potential (mV) | PDI | |
| C-1 (1.0) | 130 ± 65 | −15.5 ± 19.3 | 0.48 ± 0.08 | 111 ± 9 | 32.4 ± 3.6 | 0.22 ± 0.04 |
| C-2 (5.0) | 131 ± 60 | 13.1 ± 5.6 | 0.35 ± 0.11 | 110 ± 3 | 43.3 ± 3.1 | 0.24 ± 0.06 |
| D-1 (5.0) | 51 ± 18 | −13.1 ± 7.7 | 0.32 ± 0.15 | 92 ± 13 | 30.3 ± 13.4 | 0.20 ± 0.03 |
| D-2 (5.0) | 85 ± 27 | 10.2 ± 3.9 | 0.25 ± 0.02 | 123 ± 10 | 45.0 ± 3.6 | 0.18 ± 0.03 |
| E-1 (2.5) | 85 ± 26 | −34.6 ± 11.4 | 0.28 ± 0.08 | 105 ± 9 | 30.9 ± 6.4 | 0.20 ± 0.05 |
| E-2 (7.5) | 60 ± 8 | 4.9 ± 3.4 | 0.24 ± 0.03 | 109 ± 11 | 43.7 ± 0.8 | 0.20 ± 0.03 |
| E-3 (5.0) | 67 ± 7 | 32.6 ± 2.5 | 0.33 ± 0.06 | 113 ± 7 | 51.0 ± 2.5 | 0.26 ± 0.02 |
3. Experimental
3.1. Materials
3.2. Preparation of siRNA/MCD Nanoparticle
3.3. Measurement of Size and Zeta-Potential of Carriers
3.4. Construction of R8-D-MEND Containing siRNA/MCD Nanoparticle
3.5. Evaluation of Gene Silencing Efficiency and Cell Viability
3.6. Statistical Analysis
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Nakamura, Y.; Kogure, K.; Futaki, S.; Harashima, H. Octaarginine-modified multifunctional envelope-type nano device for siRNA. J. Control. Release 2007, 119, 360–367. [Google Scholar] [CrossRef]
- Takemoto, H.; Ishii, A.; Miyata, K.; Nakanishi, M.; Oba, M.; Ishii, T.; Yamasaki, Y.; Nishiyama, N.; Kataoka, K. Polyion complex stability and gene silencing efficiency with a siRNA-grafted polymer delivery system. Biomaterials 2010, 31, 8097–8105. [Google Scholar] [CrossRef]
- Zintchenko, A.; Philipp, A.; Dehshahri, A.; Wagner, E. Simple modifications of branched PEI lead to highly efficient siRNA carriers with low toxicity. Bioconjug. Chem. 2008, 19, 1448–1455. [Google Scholar] [CrossRef]
- Sato, Y.; Hatakeyama, H.; Sakurai, Y.; Hyodo, M.; Akita, H.; Harashima, H. A pH-sensitive cationic lipid facilitates the delivery of liposomal siRNA and gene silencing activity in vitro and in vivo. J. Control. Release 2012, 163, 267–276. [Google Scholar] [CrossRef]
- Semple, S.C.; Akinc, A.; Chen, J.; Sandhu, A.P.; Mui, B.L.; Cho, C.K.; Sah, D.W.; Stebbing, D.; Crosley, E.J.; Yaworski, E.; et al. Rational design of cationic lipids for siRNA delivery. Nat. Biotechnol. 2010, 28, 172–176. [Google Scholar] [CrossRef]
- Akinc, A.; Zumbuehl, A.; Goldberg, M.; Leshchiner, E.S.; Busini, V.; Hossain, N.; Bacallado, S.A.; Nguyen, D.N.; Fuller, J.; Alvarez, R.; et al. A combinatorial library of lipid-like materials for delivery of RNAi therapeutics. Nat. Biotechnol. 2008, 26, 561–569. [Google Scholar] [CrossRef]
- Mao, S.; Neu, M.; Germershaus, O.; Merkel, O.; Sitterberg, J.; Bakowsky, U.; Kissel, T. Influence of polyethylene glycol chain length on the physicochemical and biological properties of poly(ethylene imine)-graft-poly(ethylene glycol) block copolymer/SiRNA polyplexes. Bioconjug. Chem. 2006, 17, 1209–1218. [Google Scholar] [CrossRef]
- Godbey, W.T.; Mikos, A.G. Recent progress in gene delivery using non-viral transfer complexes. J. Control. Release 2001, 72, 115–125. [Google Scholar] [CrossRef]
- Yang, Y.; Xu, Z.; Chen, S.; Gao, Y.; Gu, W.; Chen, L.; Pei, Y.; Li, Y. Histidylated cationic polyorganophosphazene/DNA self-assembled nanoparticles for gene delivery. Int. J. Pharm. 2008, 353, 277–282. [Google Scholar] [CrossRef]
- Osada, K.; Oshima, H.; Kobayashi, D.; Doi, M.; Enoki, M.; Yamasaki, Y.; Kataoka, K. Quantized folding of plasmid DNA condensed with block catiomer into characteristic rod structures promoting transgene efficacy. J. Am. Chem. Soc. 2010, 132, 12343–12348. [Google Scholar] [CrossRef]
- Dias, R.S.; Pais, A.A.C.C.; Miguel, M.G.; Lindman, B. Modeling of DNA compaction by polycations. J. Chem. Phys. 2003, 119, 8150–8157. [Google Scholar] [CrossRef]
- Liu, Y.; Wenning, L.; Lynch, M.; Reineke, T.M. New poly(D-glucaramidoamine)s induce DNA nanoparticle formation and efficient gene delivery into mammalian cells. J. Am. Chem. Soc. 2004, 126, 7422–7423. [Google Scholar] [CrossRef]
- Brumbach, J.H.; Lin, C.; Yockman, J.; Kim, W.J.; Blevins, K.S.; Engbersen, J.F.; Feijen, J.; Kim, S.W. Mixtures of poly(triethylenetetramine/cystamine bisacrylamide) and poly(triethylenetetramine/cystamine bisacrylamide)-g-poly(ethylene glycol) for improved gene delivery. Bioconjug. Chem. 2010, 21, 1753–1761. [Google Scholar] [CrossRef]
- Vijayanathan, V.; Thomas, T.; Thomas, T.J. DNA nanoparticles and development of DNA delivery vehicles for gene therapy. Biochemistry 2002, 41, 14085–14094. [Google Scholar] [CrossRef]
- Hama, S.; Akita, H.; Iida, S.; Mizuguchi, H.; Harashima, H. Quantitative and mechanism-based investigation of post-nuclear delivery events between adenovirus and lipoplex. Nucleic Acids Res. 2007, 35, 1533–1543. [Google Scholar] [CrossRef]
- Hama, S.; Akita, H.; Ito, R.; Mizuguchi, H.; Hayakawa, T.; Harashima, H. Quantitative comparison of intracellular trafficking and nuclear transcription between adenoviral and lipoplex systems. Mol. Ther. 2006, 13, 786–794. [Google Scholar] [CrossRef]
- Yamada, Y.; Nomura, T.; Harashima, H.; Yamashita, A.; Katoono, R.; Yui, N. Intranuclear DNA release is a determinant of transfection activity for a non-viral vector: Biocleavable polyrotaxane as a supramolecularly dissociative condenser for efficient intranuclear DNA release. Biol. Pharm. Bull. 2010, 33, 1218–1222. [Google Scholar] [CrossRef]
- Yamada, Y.; Nomura, T.; Harashima, H.; Yamashita, A.; Yui, N. Post-nuclear gene delivery events for transgene expression by biocleavable polyrotaxanes. Biomaterials 2012, 33, 3952–3958. [Google Scholar] [CrossRef]
- Yamada, Y.; Hashida, M.; Nomura, T.; Harashima, H.; Yamasaki, Y.; Kataoka, K.; Yamashita, A.; Katoono, R.; Yui, N. Different mechanisms for nanoparticle formation between pDNA and siRNA using polyrotaxane as the polycation. Chemphyschem 2012, 13, 1161–1165. [Google Scholar] [CrossRef]
- Suzuki, R.; Yamada, Y.; Harashima, H. Development of small, homogeneous pDNA particles condensed with mono-cationic detergents and encapsulated in a multifunctional envelope-type nano device. Biol. Pharm. Bull. 2008, 31, 1237–1243. [Google Scholar] [CrossRef]
- Kogure, K.; Moriguchi, R.; Sasaki, K.; Ueno, M.; Futaki, S.; Harashima, H. Development of a non-viral multifunctional envelope-type nano device by a novel lipid film hydration method. J. Control. Release 2004, 98, 317–323. [Google Scholar] [CrossRef]
- Khalil, I.A.; Kogure, K.; Futaki, S.; Harashima, H. High density of octaarginine stimulates macropinocytosis leading to efficient intracellular trafficking for gene expression. J. Biol. Chem. 2006, 281, 3544–3551. [Google Scholar]
- Kogure, K.; Akita, H.; Yamada, Y.; Harashima, H. Multifunctional envelope-type nano device (MEND) as a non-viral gene delivery system. Adv. Drug Deliv. Rev. 2008, 60, 559–571. [Google Scholar] [CrossRef]
- Khalil, I.A.; Kogure, K.; Yamada, M.; Harashima, H. Octaarginine-modified multifunctional envelope-type nanoparticles for gene delivery. J. Gene Med. 2009, 11, 1171–1171. [Google Scholar]
- Khalil, I.A.; Kogure, K.; Futaki, S.; Hama, S.; Akita, H.; Ueno, M.; Kishida, H.; Kudoh, M.; Mishina, Y.; Kataoka, K.; et al. Octaarginine-modified multifunctional envelope-type nanoparticles for gene delivery. Gene Ther. 2007, 14, 682–689. [Google Scholar] [CrossRef]
- Akita, H.; Kudo, A.; Minoura, A.; Yamaguti, M.; Khalil, I.A.; Moriguchi, R.; Masuda, T.; Danev, R.; Nagayama, K.; Kogure, K.; et al. Multi-layered nanoparticles for penetrating the endosome and nuclear membrane via a step-wise membrane fusion process. Biomaterials 2009, 30, 2940–2949. [Google Scholar] [CrossRef]
- El-Sayed, A.; Khalil, I.A.; Kogure, K.; Futaki, S.; Harashima, H. Octaarginine- and octalysine-modified nanoparticles have different modes of endosomal escape. J. Biol. Chem. 2008, 283, 23450–23461. [Google Scholar] [CrossRef]
Supplementary Materials
| Nanoparticles prepared using C-1 | Nanoparticles prepared using C-2 | ||||||
|---|---|---|---|---|---|---|---|
| N/P ratio | Size (nm) | Zeta potential (mV) | PDI | N/P ratio | Size (nm) | Zeta potential (mV) | PDI |
| 0.25 | 94 | −37.9 | 0.72 | 0.25 | 162 | −42.9 | 0.63 |
| 0.5 | 124 | −29.8 | 0.55 | 0.5 | 2520 | −19.9 | 0.37 |
| 1 | 66 | −28.8 | 0.33 | 2.5 | 616 | 1.6 | 0.6 |
| 1.5 | 90 | −37.5 | 0.36 | 3.75 | 584 | 10.7 | 0.57 |
| 2 | 90 | −28.6 | 0.26 | 5 | 151 | 18.7 | 0.43 |
| 2.5 | 1480 | −28.1 | 0.58 | 6.25 | 108 | 19.9 | 0.27 |
| 5 | 1650 | −9.3 | 0.39 | 7.5 | 58 | 27.7 | 0.37 |
| 10 | 1360 | 0.8 | 0.56 | 8.75 | 84 | 29.3 | 0.42 |
| 10 | 57 | 31.7 | 0.36 | ||||
| Nanoparticles prepared using D-1 | Nanoparticles prepared using D-2 | ||||||
| N/P ratio | Size (nm) | Zeta potential (mV) | PDI | N/P ratio | Size (nm) | Zeta potential (mV) | PDI |
| 0.25 | 244 | −5.6 | 0.67 | 0.25 | 156 | −47.8 | 0.44 |
| 0.5 | 302 | −19.9 | 0.35 | 0.5 | 2560 | −18.6 | 0.7 |
| 2.5 | 162 | −18.5 | 0.34 | 2.5 | 888 | 4.5 | 0.62 |
| 3.75 | 113 | −34.4 | 0.34 | 3.75 | 69 | 18.3 | 0.24 |
| 5 | 123 | −37.0 | 0.22 | 5 | 95 | 18.7 | 0.32 |
| 6.25 | 69 | −35.0 | 0.3 | 6.25 | 82 | 21 | 0.34 |
| 7.5 | 55 | −31.9 | 0.21 | 7.5 | 65 | 30.4 | 0.36 |
| 8.75 | 99 | −33.5 | 0.24 | 8.75 | 50 | 30 | 0.34 |
| 10 | 102 | −35.0 | 0.25 | 10 | 53 | 31.3 | 0.36 |
| Nanoparticles prepared using E-1 | Nanoparticles prepared using E-2 | ||||||
| N/P ratio | Size (nm) | Zeta potential (mV) | PDI | N/P ratio | Size (nm) | Zeta potential (mV) | PDI |
| 0.25 | 713 | −19.9 | 0.85 | 0.25 | 133 | −50.2 | 0.41 |
| 0.5 | 198 | −20.0 | 0.49 | 0.5 | 129 | −29.6 | 0.24 |
| 1.25 | 324 | −20.7 | 0.66 | 2.5 | 3140 | −9.3 | 0.455 |
| 2.5 | 74 | −34.2 | 0.28 | 3.75 | 918 | 0.3 | 0.45 |
| 3.75 | 159 | −29.1 | 0.18 | 5 | 221 | 8.2 | 0.386 |
| 5 | 165 | −28.5 | 0.14 | 6.25 | 132 | 12.9 | 0.22 |
| 7.5 | 2030 | −20.5 | 0.7 | 7.5 | 53 | 16.5 | 0.26 |
| 10 | 2160 | −18.8 | 0.6 | 8.75 | 54 | 16.9 | 0.28 |
| 10 | 63 | 20.5 | 0.313 | ||||
| Nanoparticles prepared using E-3 | |||||||
| N/P ratio | Size (nm) | Zeta potential (mV) | PDI | ||||
| 2.5 | 54 | 25.2 | 0.27 | ||||
| 3.75 | 63 | 40.1 | 0.25 | ||||
| 5 | 53 | 39.7 | 0.36 | ||||
| 7.5 | 45 | 44.4 | 0.32 | ||||
| 10 | 57 | 42.8 | 0.42 | ||||
| Size (nm) | Zata potential (mV) | |
|---|---|---|
| SUV | 74 ± 10 | −66.8 ± 8.9 |
| R8-SUV | 86 ± 14 | 29.9 ± 11.3 |
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Yamada, Y.; Suzuki, R.; Harashima, H. Investigation of siRNA Nanoparticle Formation Using Mono-Cationic Detergents and Its Use in Gene Silencing in Human HeLa Cells. Cancers 2013, 5, 1413-1425. https://doi.org/10.3390/cancers5041413
Yamada Y, Suzuki R, Harashima H. Investigation of siRNA Nanoparticle Formation Using Mono-Cationic Detergents and Its Use in Gene Silencing in Human HeLa Cells. Cancers. 2013; 5(4):1413-1425. https://doi.org/10.3390/cancers5041413
Chicago/Turabian StyleYamada, Yuma, Ryosuke Suzuki, and Hideyoshi Harashima. 2013. "Investigation of siRNA Nanoparticle Formation Using Mono-Cationic Detergents and Its Use in Gene Silencing in Human HeLa Cells" Cancers 5, no. 4: 1413-1425. https://doi.org/10.3390/cancers5041413
APA StyleYamada, Y., Suzuki, R., & Harashima, H. (2013). Investigation of siRNA Nanoparticle Formation Using Mono-Cationic Detergents and Its Use in Gene Silencing in Human HeLa Cells. Cancers, 5(4), 1413-1425. https://doi.org/10.3390/cancers5041413




