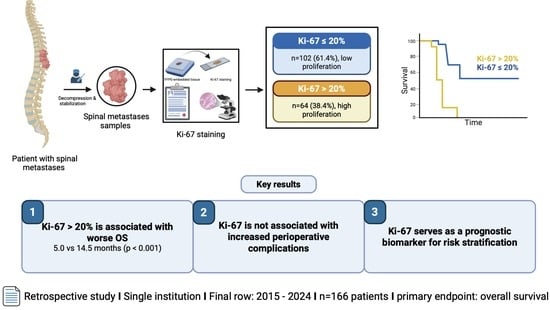
The Prognostic Impact of the Ki-67 Proliferation Index in Patients with Surgically Treated Spinal Metastases
Simple Summary
Abstract
1. Introduction
2. Methods
2.1. Patients and Inclusion Criteria
2.2. Data Recording
2.3. Patient Groups
2.4. Study Design
2.5. Histopathology
2.6. Exclusion Criteria
2.7. Statistical Analysis and Graphical Illustrations
3. Results
3.1. Patient Characteristics
3.2. Patient-Related and Disease-Related Factors Associated with Ki-67 Index
3.3. Prognostic Value of the Ki-67 Index for 1-Year Mortality and Overall Survival
3.4. Multivariate Analysis of Factors Associated with a High Ki-67 Index (>20%)
3.5. Preoperative Risk Score for Elevated Ki-67 Expression and Survival Stratification
4. Discussion
5. Conclusions
6. Limitations
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ASA | American Society of Anesthesiology |
| ASIA | American Spinal Injury Association |
| AUC | Area under the curve |
| CCI | Charlson comorbidity index |
| CUP | Cancer of unknown primary |
| CI | Confidence interval |
| GI | Gastrointestinal |
| IQR | Interquartile range |
| KPS | Karnofsky Performance Scale |
| Meta | Metachronous |
| N | Numbers |
| OR | Odds ratio |
| OS | Overall survival |
| PT | Primary tumor |
| ROC | Receiver operating characteristic |
| SD | Standard deviation |
| SINS | Spinal instability neoplastic score |
| SM | Spinal metastasis |
| SSCs | Spinal surgery-related complications |
| Syn | Synchronous |
| TTP | Time to progression |
References
- Curtin, M.; Piggott, R.P.; Murphy, E.P.; Munigangaiah, S.; Baker, J.F.; McCabe, J.P.; Devitt, A. Spinal Metastatic Disease: A Review of the Role of the Multidisciplinary Team. Orthop. Surg. 2017, 9, 145–151. [Google Scholar] [CrossRef] [PubMed]
- Jemal, A.; Siegel, R.; Ward, E.; Murray, T.; Xu, J.; Thun, M.J. Cancer statistics, 2007. CA Cancer J. Clin. 2007, 57, 43–66. [Google Scholar] [CrossRef] [PubMed]
- Wewel, J.T.; O’Toole, J.E. Epidemiology of spinal cord and column tumors. Neurooncol. Pract. 2020, 7, i5–i9. [Google Scholar] [CrossRef] [PubMed]
- Jaipanya, P.; Chanplakorn, P. Spinal metastasis: Narrative reviews of the current evidence and treatment modalities. J. Int. Med. Res. 2022, 50, 3000605221091665. [Google Scholar] [CrossRef]
- Van den Brande, R.; Cornips, E.M.; Peeters, M.; Ost, P.; Billiet, C.; Van de Kelft, E. Epidemiology of spinal metastases, metastatic epidural spinal cord compression and pathologic vertebral compression fractures in patients with solid tumors: A systematic review. J. Bone Oncol. 2022, 35, 100446. [Google Scholar] [CrossRef]
- Wang, F.; Zhang, H.; Yang, L.; Yang, X.G.; Zhang, H.R.; Li, J.K.; Qiao, R.Q.; Hu, Y.C. Epidemiological Characteristics of 1196 Patients with Spinal Metastases: A Retrospective Study. Orthop. Surg. 2019, 11, 1048–1053. [Google Scholar] [CrossRef]
- Barzilai, O.; Fisher, C.G.; Bilsky, M.H. State of the Art Treatment of Spinal Metastatic Disease. Neurosurgery 2018, 82, 757–769. [Google Scholar] [CrossRef]
- Holmebakk, T.; Solbakken, A.; Mala, T.; Nesbakken, A. Clinical prediction of survival by surgeons for patients with incurable abdominal malignancy. Eur. J. Surg. Oncol. 2011, 37, 571–575. [Google Scholar] [CrossRef]
- Fehlings, M.G.; Nater, A.; Tetreault, L.; Kopjar, B.; Arnold, P.; Dekutoski, M.; Finkelstein, J.; Fisher, C.; France, J.; Gokaslan, Z.; et al. Survival and Clinical Outcomes in Surgically Treated Patients with Metastatic Epidural Spinal Cord Compression: Results of the Prospective Multicenter AOSpine Study. J. Clin. Oncol. 2016, 34, 268–276. [Google Scholar] [CrossRef]
- Patchell, R.A.; Tibbs, P.A.; Regine, W.F.; Payne, R.; Saris, S.; Kryscio, R.J.; Mohiuddin, M.; Young, B. Direct decompressive surgical resection in the treatment of spinal cord compression caused by metastatic cancer: A randomised trial. Lancet 2005, 366, 643–648. [Google Scholar] [CrossRef]
- Fisher, C.G.; DiPaola, C.P.; Ryken, T.C.; Bilsky, M.H.; Shaffrey, C.I.; Berven, S.H.; Harrop, J.S.; Fehlings, M.G.; Boriani, S.; Chou, D.; et al. A novel classification system for spinal instability in neoplastic disease: An evidence-based approach and expert consensus from the Spine Oncology Study Group. Spine 2010, 35, E1221–E1229. [Google Scholar] [CrossRef]
- Tokuhashi, Y.; Matsuzaki, H.; Oda, H.; Oshima, M.; Ryu, J. A revised scoring system for preoperative evaluation of metastatic spine tumor prognosis. Spine 2005, 30, 2186–2191. [Google Scholar] [CrossRef]
- Gerdes, J.; Lemke, H.; Baisch, H.; Wacker, H.H.; Schwab, U.; Stein, H. Cell cycle analysis of a cell proliferation-associated human nuclear antigen defined by the monoclonal antibody Ki-67. J. Immunol. 1984, 133, 1710–1715. [Google Scholar] [CrossRef] [PubMed]
- Duchrow, M.; Schluter, C.; Wohlenberg, C.; Flad, H.D.; Gerdes, J. Molecular characterization of the gene locus of the human cell proliferation-associated nuclear protein defined by monoclonal antibody Ki-67. Cell Prolif. 1996, 29, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Iqbal, A.; Tamgadge, S.; Tamgadge, A.; Pereira, T.; Kumar, S.; Acharya, S.; Jadhav, A. Evaluation of Ki-67 Expression in Oral Submucous Fibrosis and Its Correlation with Clinical and Histopathological Features. J. Microsc. Ultrastruct. 2020, 8, 20–24. [Google Scholar] [CrossRef] [PubMed]
- Luo, Z.W.; Zhu, M.G.; Zhang, Z.Q.; Ye, F.J.; Huang, W.H.; Luo, X.Z. Increased expression of Ki-67 is a poor prognostic marker for colorectal cancer patients: A meta analysis. BMC Cancer 2019, 19, 123. [Google Scholar] [CrossRef]
- Martin, B.; Paesmans, M.; Mascaux, C.; Berghmans, T.; Lothaire, P.; Meert, A.P.; Lafitte, J.J.; Sculier, J.P. Ki-67 expression and patients survival in lung cancer: Systematic review of the literature with meta-analysis. Br. J. Cancer 2004, 91, 2018–2025. [Google Scholar] [CrossRef]
- Tian, Y.; Ma, Z.; Chen, Z.; Li, M.; Wu, Z.; Hong, M.; Wang, H.; Svatek, R.; Rodriguez, R.; Wang, Z. Clinicopathological and Prognostic Value of Ki-67 Expression in Bladder Cancer: A Systematic Review and Meta-Analysis. PLoS ONE 2016, 11, e0158891. [Google Scholar] [CrossRef]
- Inwald, E.C.; Klinkhammer-Schalke, M.; Hofstadter, F.; Zeman, F.; Koller, M.; Gerstenhauer, M.; Ortmann, O. Ki-67 is a prognostic parameter in breast cancer patients: Results of a large population-based cohort of a cancer registry. Breast Cancer Res. Treat. 2013, 139, 539–552. [Google Scholar] [CrossRef]
- Li, L.T.; Jiang, G.; Chen, Q.; Zheng, J.N. Ki67 is a promising molecular target in the diagnosis of cancer (review). Mol. Med. Rep. 2015, 11, 1566–1572. [Google Scholar] [CrossRef]
- Hamed, M.; Potthoff, A.L.; Heimann, M.; Schafer, N.; Borger, V.; Radbruch, A.; Herrlinger, U.; Vatter, H.; Schneider, M. Survival in patients with surgically treated brain metastases: Does infratentorial location matter? Neurosurg. Rev. 2023, 46, 80. [Google Scholar] [CrossRef]
- Schweppe, J.A.; Potthoff, A.L.; Heimann, M.; Ehrentraut, S.F.; Borger, V.; Lehmann, F.; Schaub, C.; Bode, C.; Putensen, C.; Herrlinger, U.; et al. Incurring detriments of unplanned readmission to the intensive care unit following surgery for brain metastasis. Neurosurg. Rev. 2023, 46, 155. [Google Scholar] [CrossRef]
- Sciubba, D.M.; Petteys, R.J.; Dekutoski, M.B.; Fisher, C.G.; Fehlings, M.G.; Ondra, S.L.; Rhines, L.D.; Gokaslan, Z.L. Diagnosis and management of metastatic spine disease. A review. J. Neurosurg. Spine 2010, 13, 94–108. [Google Scholar] [CrossRef] [PubMed]
- Paulino Pereira, N.R.; Janssen, S.J.; van Dijk, E.; Harris, M.B.; Hornicek, F.J.; Ferrone, M.L.; Schwab, J.H. Development of a Prognostic Survival Algorithm for Patients with Metastatic Spine Disease. J. Bone Jt. Surg. Am. 2016, 98, 1767–1776. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, A.K.; Goodwin, C.R.; Heravi, A.; Kim, R.; Abu-Bonsrah, N.; Sankey, E.; Kerekes, D.; De la Garza Ramos, R.; Schwab, J.; Sciubba, D.M. Predicting survival for metastatic spine disease: A comparison of nine scoring systems. Spine J. 2018, 18, 1804–1814. [Google Scholar] [CrossRef] [PubMed]
- Asoglu, H.; Lampmann, T.; Jaber, M.; Khalafov, L.; Dittmer, J.; Ilic, I.; Gielen, G.H.; Toma, M.; Vatter, H.; Bendella, Z.; et al. Bone mineral density as potential individual prognostic biomarker in patients with neurosurgically treated spinal metastasis. J. Cancer Res. Clin. Oncol. 2025, 151, 105. [Google Scholar] [CrossRef]
- Hamed, M.; Brandecker, S.; Rana, S.; Potthoff, A.L.; Eichhorn, L.; Bode, C.; Schmeel, F.C.; Radbruch, A.; Schafer, N.; Herrlinger, U.; et al. Postoperative prolonged mechanical ventilation correlates to poor survival in patients with surgically treated spinal metastasis. Front. Oncol. 2022, 12, 940790. [Google Scholar] [CrossRef]
- Banat, M.; Potthoff, A.L.; Hamed, M.; Borger, V.; Scorzin, J.E.; Lampmann, T.; Asoglu, H.; Khalafov, L.; Schmeel, F.C.; Paech, D.; et al. Synchronous versus metachronous spinal metastasis: A comparative study of survival outcomes following neurosurgical treatment. J. Cancer Res. Clin. Oncol. 2024, 150, 136. [Google Scholar] [CrossRef]
- Siddiqui, S. Assessment of Ki-67 expression in cases of prostatic carcinoma and its correlation with clinical outcomes. Indian J. Pathol. Microbiol. 2024, 67, 362–366. [Google Scholar] [CrossRef]
- Zeng, M.; Zhou, J.; Wen, L.; Zhu, Y.; Luo, Y.; Wang, W. The relationship between the expression of Ki-67 and the prognosis of osteosarcoma. BMC Cancer 2021, 21, 210. [Google Scholar] [CrossRef]
- Ding, X.L.; Su, Y.G.; Yu, L.; Bai, Z.L.; Bai, X.H.; Chen, X.Z.; Yang, X.; Zhao, R.; He, J.X.; Wang, Y.Y. Clinical characteristics and patient outcomes of molecular subtypes of small cell lung cancer (SCLC). World J. Surg. Oncol. 2022, 20, 54. [Google Scholar] [CrossRef] [PubMed]
- Yang, N.; Chen, X.; Wang, H.; Liu, P.; Liu, B.; Xie, B.; Zhou, J. Ki-67 expression stratifies PD-L1-high NSCLC for immune checkpoint inhibitor plus chemotherapy: A real-world biomarker validation. Cancer Immunol. Immunother. CII 2025, 74, 354. [Google Scholar] [CrossRef] [PubMed]
- Kadado, K.J.; Abernathy, O.L.; Salyers, W.J.; Kallail, K.J. Gastrointestinal Stromal Tumor and Ki-67 as a Prognostic Indicator. Cureus 2022, 14, e20868. [Google Scholar] [CrossRef] [PubMed]
- Yang, M.; Zhu, X.; Shen, Y.; He, Q.; Qin, Y.; Shao, Y.; Yuan, L.; Ye, H. [High expression of MYBL2 promotes progression and predicts a poor survival outcome of prostate cancer]. Nan Fang Yi Ke Da Xue Xue Bao = J. South. Med. Univ. 2022, 42, 1109–1118. [Google Scholar] [CrossRef]
- Klayech, Z.; Moussa, A.; Souid, M.; Hadhri, R.; Miled, S.; Gabbouj, S.; Remadi, Y.; Faleh, R.; Bouaouina, N.; Zakhama, A.; et al. Prognostic Significance of Combining Cytokeratin-19, E-Cadherin and Ki-67 Analysis in Triple-Negative Breast Cancer with Basal-Like and Non-Basal-Like Phenotype. Cancer Investig. 2024, 42, 769–781. [Google Scholar] [CrossRef]
- Atrash, S.; Robinson, M.; Taneja, A.; Paul, B.; Cassetta, K.; Ndiaye, A.; Varga, C.; Block, J.; Lipford, E.H.; Smith, E.T.; et al. Bone marrow Ki-67 index is of prognostic value in newly diagnosed multiple myeloma. Eur. J. Haematol. 2023, 111, 373–381. [Google Scholar] [CrossRef]
- Wongmaneerung, P.; Chitapanarux, I.; Traisathit, P.; Prasitwattanaseree, S.; Rottuntikarn, W.; Somwangprasert, A.; Ditsatham, C.; Watcharachan, K.; Klunklin, P.; Onchan, W. The association between Ki-67 expression and survival in breast cancer subtypes: A cross-sectional study of Ki-67 cut-point in northern Thailand. BMC Cancer 2025, 25, 346. [Google Scholar] [CrossRef]
- Pan, X.; Wei, C.; Su, J.; Fang, M.; Lin, Q.; Qin, Y.; Gao, J.; Zhao, J.; Zhao, H.; Liu, F. A comprehensive analysis of the prognostic value, expression characteristics and immune correlation of MKI67 in cancers. Front. Immunol. 2025, 16, 1531708. [Google Scholar] [CrossRef]
- Tsurui, T.; Hosonuma, M.; Sasaki, A.; Maruyama, Y.; Amari, Y.; Funayama, E.; Tajima, K.; Toyoda, H.; Isobe, J.; Yamazaki, Y.; et al. Ki-67 expression in anti-programmed cell death protein-1 antibody-bound CD8+ T cells as a predictor of clinical benefit. Discov. Oncol. 2025, 16, 348. [Google Scholar] [CrossRef]
- Mohammed, A.; Bakry, A.; Gharieb, S.; Hanna, A.; Obaya, A.; Abdelhady, W.; Metwalli, A. Predictive Value of Tumor-Infiltrating Lymphocytes and Ki-67 for Pathological Response to Total Neoadjuvant Therapy in Rectal Cancer. J. Gastrointest. Cancer 2024, 55, 869–876. [Google Scholar] [CrossRef]
- Aghebati, M.; Hossieni, R.; Makeh, A.S.; Shirzadi, A.; Akbari, M.E. Ki-67 and 21-gene recurrence score assay in decision making for adjuvant chemotherapy in breast cancer patients. Discov. Oncol. 2025, 16, 970. [Google Scholar] [CrossRef]
- Atari, M.; Imai, K.; Nanjo, H.; Wakamatsu, Y.; Takashima, S.; Kurihara, N.; Kuriyama, S.; Suzuki, H.; Demura, R.; Harata, Y.; et al. Rapid intraoperative Ki-67 immunohistochemistry for lung cancer using non-contact alternating current electric field mixing. Lung Cancer 2022, 173, 75–82. [Google Scholar] [CrossRef]





| n = 166 | n = 166 | ||
|---|---|---|---|
| Gender, n (%) | Spinal cord compression, n (%) Yes No Involved segments, n (%) ≤2 segments ≥3 segments Extraspinal metastases, n (%) Yes No Postoperative ASIA, n (%) Good (D, E) Poor (A, B, C) Postoperative KPS, n (%) ≥70% <70 Overall survival, months Median (IQR) 1-year mortality, n (%) Yes No | 147 (88.6) 19 (11.4) 86 (51.8) 80 (48.2) 93 (56) 73 (44) 113 (68.1) 53 (31.9) 103 (62) 63 (38) 8.55 (3–17) 86 (51.8) 69 (41.6) | |
| Male | 99 (59.6) | ||
| Female | 67 (40.4) | ||
| Age at SM diagnosis, n (%) | |||
| <70 years | 92 (55.4) | ||
| ≥70 years | 73 (44.6) | ||
| ASA at SM diagnosis, n (%) | |||
| ASA ≤ 2 | 60 (36.1) | ||
| ASA > 2 | 106 (63.9) | ||
| Primarius, n (%) | |||
| Lung cancer | 41 (24.7) | ||
| Breast cancer | 22 (13.3) | ||
| Prostate cancer | 36 (21.7) | ||
| Plasmacytoma | 7 (4.2) | ||
| GI cancer | 10 (6) | ||
| Kidney cancer | 14 (8.4) | ||
| Other pathologies | 27 (16.3) | ||
| CUP | 9 (5.4) | ||
| Ki-67 index, n (%) | |||
| ≤20% | 102 (61.4) | ||
| >20 | 64 (38.6) | ||
| First manifestation as SM, n (%) | |||
| Synchronous | 72 (43.4) | ||
| Metachronous | 85 (51.2) | ||
| SM locations, n (%) | |||
| Craniocervical | 1 (0.6) | ||
| Cervical vertebrae | 14 (8.4) | ||
| Cervicothoracic | 11 (6.6) | ||
| Thoracic vertebrae | 100 (60.2) | ||
| Thoracolumbar | 11 (6.6) | ||
| Lumbar spine | 24 (14.5) | ||
| Lumbosacral | 5 (3.0) |
| Ki-67 ≤ 20%, n = 102 | Ki-67 > 20%, n = 64 | p-Value | |
|---|---|---|---|
| Patient demographics | |||
| Gender, n (%) | 0.481 | ||
| Male | 63 (61.8) | 36 (56.3) | |
| Female | 39 (38.2) | 28 (43.8) | |
| Age at SM diagnosis | 0.768 | ||
| Median (IQR) | 68 (58.75–77.25) | 66.5 (60–74) | |
| Range | 51 | 59 | |
| ASA at SM diagnosis, n (%) | 0.042 | ||
| ASA ≤ 2 | 43 (42.2) | 17 (26.6) | |
| ASA > 2 | 59 (57.8) | 47 (73.4) | |
| CCI, n (%) | 0.445 | ||
| <10 | 83 (81.4) | 55 (85.9) | |
| ≥10 | 19 (18.6) | 9 (14.1) | |
| Tumor characteristics | |||
| Primary, n (%) | 0.074 | ||
| Lung cancer | 19 (18.6) | 22 (34.4) | 0.022 |
| Breast cancer | 17 (16.7) | 5 (7.8) | 0.102 |
| Prostate cancer | 24 (23.5) | 12 (18.8) | 0.467 |
| Plasmacytoma | 5 (4.9) | 2 (3.1) | |
| GI cancer | 4 (3.9) | 6 (9.4) | 0.077 |
| Kidney cancer | 12 (11.8) | 2 (3.1) | 0.051 |
| Other pathologies | 15 (14.7) | 12 (18.8) | |
| CUP | 6 (5.9) | 3 (4.7) | |
| TTP, PT to SM diagnosis, years | 0.012 | ||
| Median (IQR) | 1 (0–8) | 0 (0–1.5) | |
| Range | 30 | 12 | |
| First manifestation as SM, n (%) | 0.048 | ||
| Synchronous | 38 (39.6) | 34 (55.7) | |
| Metachronous | 58 (60.4) | 27 (44.3) | |
| Previous treatment, n (%) | 0.744 | ||
| Yes | 60 (58.8) | 36 (56.3) | |
| No | 42 (41.2) | 28 (43.8) | |
| SM locations, n (%) | 0.887 | ||
| Craniocervical | 0 (0) | 1 (1.6) | |
| Cervical vertebrae | 10 (9.8) | 4 (6.3) | |
| Cervicothoracic | 7 (6.9) | 4 (6.3) | |
| Thoracic vertebrae | 60 (58.8) | 40 (62.5) | |
| Thoracolumbar | 7 (6.9) | 4 (6.3) | |
| Lumbar spine | 15 (14.7) | 9 (14.1) | |
| Lumbosacral | 3 (2.9) | 2 (3.1) | |
| Involved segments, n (%) | 0.491 | ||
| ≤2 segments | 55 (53.9) | 31 (48.4) | |
| ≥3 segments | 47 (46.1) | 33 (51.6) | |
| Spinal cord compression, n (%) | 0.244 | ||
| Yes | 88 (86.3) | 59 (92.2) | |
| No | 14 (13.7) | 5 (7.8) | |
| Spinal instability, n (%) | 0.095 | ||
| Yes | 68 (70.1) | 50 (82) | |
| No | 29 (29.9) | 11 (18) | |
| Systemic disease burden | |||
| Extraspinal metastases, n (%) | 0.119 | ||
| Yes | 62 (60.8) | 31 (48.4) | |
| No | 10 (39.2) | 33 (51.6) | |
| Numbers of metastases sites | 0.348 | ||
| Median (IQR) | 1 (0–2) | 0.5 (0–2) | |
| Range | 6 | 6 | |
| Preoperative functional and neurological status | |||
| Preoperative ASIA, n (%) | 0.106 | ||
| Good (D, E) | 64 (62.7) | 32 (50) | |
| Poor (A, B, C) | 38 (37.3) | 32 (50) | |
| Preoperative KPS, n (%) | 0.104 | ||
| ≥70% | 73 (71.6) | 38 (59.4) | |
| <70% | 29 (28.4) | 26 (40.6) | |
| Surgical treatment and oncological therapy | |||
| Surgery, n (%) | 0.536 | ||
| Only decompression | 30 (29.4) | 16 (25) | |
| Stabilization | 72 (70.6) | 48 (75) | |
| Operative time, minutes | 0.462 | ||
| Median (IQR) | 179 (130.5–282.75) | 195 (152.25–267.75) | |
| Range | 551 | 413 | |
| Intraoperative blood loss, mL | 0.32 | ||
| Median (IQR) | 600 (350–1200) | 600 (400–1000) | |
| Range | 6950 | 2650 | |
| Postoperative outcomes | |||
| Specific SSCs, n (%) | 0.57 | ||
| Yes | 16 (15.7) | 8 (12.5) | |
| No | 86 (84.3) | 56 (87.5) | |
| Postoperative revision, n (%) | 0.169 | ||
| Yes | 91 (89.2) | 61 (95.3) | |
| No | 11 (10.8) | 3 (4.7) | |
| Postoperative ASIA, n (%) | 0.223 | ||
| Good (D, E) | 73 (71.6) | 40 (62.5) | |
| Poor (A, B, C) | 29 (28.4) | 24 (37.5) | |
| Postoperative KPS, n (%) | 0.122 | ||
| ≥70% | 68 (66.7) | 35 (54.7) | |
| <70% | 34 (33.3) | 29 (45.3) | |
| Local tumor recurrence, n (%) | 0.145 | ||
| Yes | 20 (22.5) | 7 (12.7) | |
| No | 69 (77.5) | 48 (87.3) | |
| Readmission in 30 days, n (%) | 0.295 | ||
| Yes | 7 (7.4) | 2 (3.3) | |
| No | 88 (92.6) | 58 (96.7) | |
| Readmission in 3 months, n (%) | 0.206 | ||
| Yes | 12 (13.3) | 4 (6.8) | |
| No | 78 (86.7) | 55 (93.2) | |
| Overall survival, months | 0.001 | ||
| Median | 14.5 | 5 | |
| 1-year mortality, n (%) | 0.001 | ||
| Yes | 41 (41.8) | 45 (78.9) | |
| No | 57 (58.2) | 12 (21.1) |
| Variable | aOR | 95% CI | p-Value |
|---|---|---|---|
| Gender (female/male) | 1.26 | 0.67–2.37 | 0.481 |
| Age (≥70/<70 years) | 0.835 | 0.432–1.613 | 0.591 |
| ASA (>2/≤2) | 2.133 | 1.053–4.32 | 0.035 |
| CCL (≥10/<10) | 0.646 | 0.269–1.555 | 0.33 |
| BMI (normal/obesity) | 0.836 | 0.304–2.301 | 0.729 |
| Lung cancer as primary tumor (yes/no) | 2.288 | 1.117–4.688 | 0.024 |
| Breast cancer as primary tumor (yes/no) | 0.348 | 0.118–1.032 | 0.057 |
| Prostate cancer as primary tumor (yes/no) | 0.592 | 0.262–1.34 | 0.208 |
| GI cancer as primary tumor (yes/no) | 2.072 | 0.560–7.621 | 0.273 |
| Kidney cancer as primary tumor (yes/no) | 0.197 | 0.042–0.937 | 0.041 |
| Previously oncological treatment (yes/no) | 0.9 | 0.478–1.693 | 0.744 |
| First manifestation SM (syn/meta) | 1.922 | 1.003–3.682 | 0.049 |
| Thoracis vertebrae location (yes/no) | 0.667 | 0.128–3.47 | 0.63 |
| Involved segments (≥3/≤2 segments) | 0.908 | 0.444–1.857 | 0.791 |
| Spinal cord compression (yes/no) | 1.287 | 0.408–4.055 | 0.667 |
| Spinal instability (yes/no) | 1.945 | 0.807–4.697 | 0.138 |
| Extraspinal metastases (yes/no) | 0.398 | 0.15–1.051 | 0.063 |
| Surgery (stabilization/only decompression) | 1.438 | 0.651–3.174 | 0.369 |
| Postoperative systematic therapy (yes/no) | 0.881 | 0.147–5.263 | 0.889 |
| Postoperative radiotherapy (yes/no) | 0.506 | 0.087–2.946 | 0.448 |
| Specific SSCs (yes/no) | 0.882 | 0.305–2.548 | 0.817 |
| Postoperative revision (yes/no) | 0.446 | 0.101–1.98 | 0.288 |
| Preoperative ASIA (poor [A, B, C]/good [D, E]) | 1.609 | 0.673–3.846 | 0.285 |
| Preoperative KPS (<70%/≥70%) | 1.056 | 0.445–2.506 | 0.902 |
| Postoperative ASIA (poor [A, B, C]/good [D, E]) | 1.124 | 0.463–2.728 | 0.797 |
| Postoperative KPS (<70%/≥70%) | 1.539 | 0.655–3.617 | 0.323 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Abedellatif, S.-E.; Janjic, M.; Khalafov, L.; Asoglu, H.; Dittmer, J.; Heimann, M.; Jaber, M.; Alenezi, H.; Toma, M.I.; Schneider, M.; et al. The Prognostic Impact of the Ki-67 Proliferation Index in Patients with Surgically Treated Spinal Metastases. Cancers 2026, 18, 1210. https://doi.org/10.3390/cancers18081210
Abedellatif S-E, Janjic M, Khalafov L, Asoglu H, Dittmer J, Heimann M, Jaber M, Alenezi H, Toma MI, Schneider M, et al. The Prognostic Impact of the Ki-67 Proliferation Index in Patients with Surgically Treated Spinal Metastases. Cancers. 2026; 18(8):1210. https://doi.org/10.3390/cancers18081210
Chicago/Turabian StyleAbedellatif, Saif-Eldin, Marija Janjic, Logman Khalafov, Harun Asoglu, Juliane Dittmer, Muriel Heimann, Mohammed Jaber, Haitham Alenezi, Marieta Ioana Toma, Matthias Schneider, and et al. 2026. "The Prognostic Impact of the Ki-67 Proliferation Index in Patients with Surgically Treated Spinal Metastases" Cancers 18, no. 8: 1210. https://doi.org/10.3390/cancers18081210
APA StyleAbedellatif, S.-E., Janjic, M., Khalafov, L., Asoglu, H., Dittmer, J., Heimann, M., Jaber, M., Alenezi, H., Toma, M. I., Schneider, M., Vatter, H., Hamed, M., & Banat, M. (2026). The Prognostic Impact of the Ki-67 Proliferation Index in Patients with Surgically Treated Spinal Metastases. Cancers, 18(8), 1210. https://doi.org/10.3390/cancers18081210