Retrospective Long-Term Evaluation of Conventional Transarterial Chemoembolization for Hepatocellular Carcinoma over 20 Years
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Patients
2.2. Imaging Protocol
2.3. TACE Technique
2.4. Statistical Analysis
3. Results
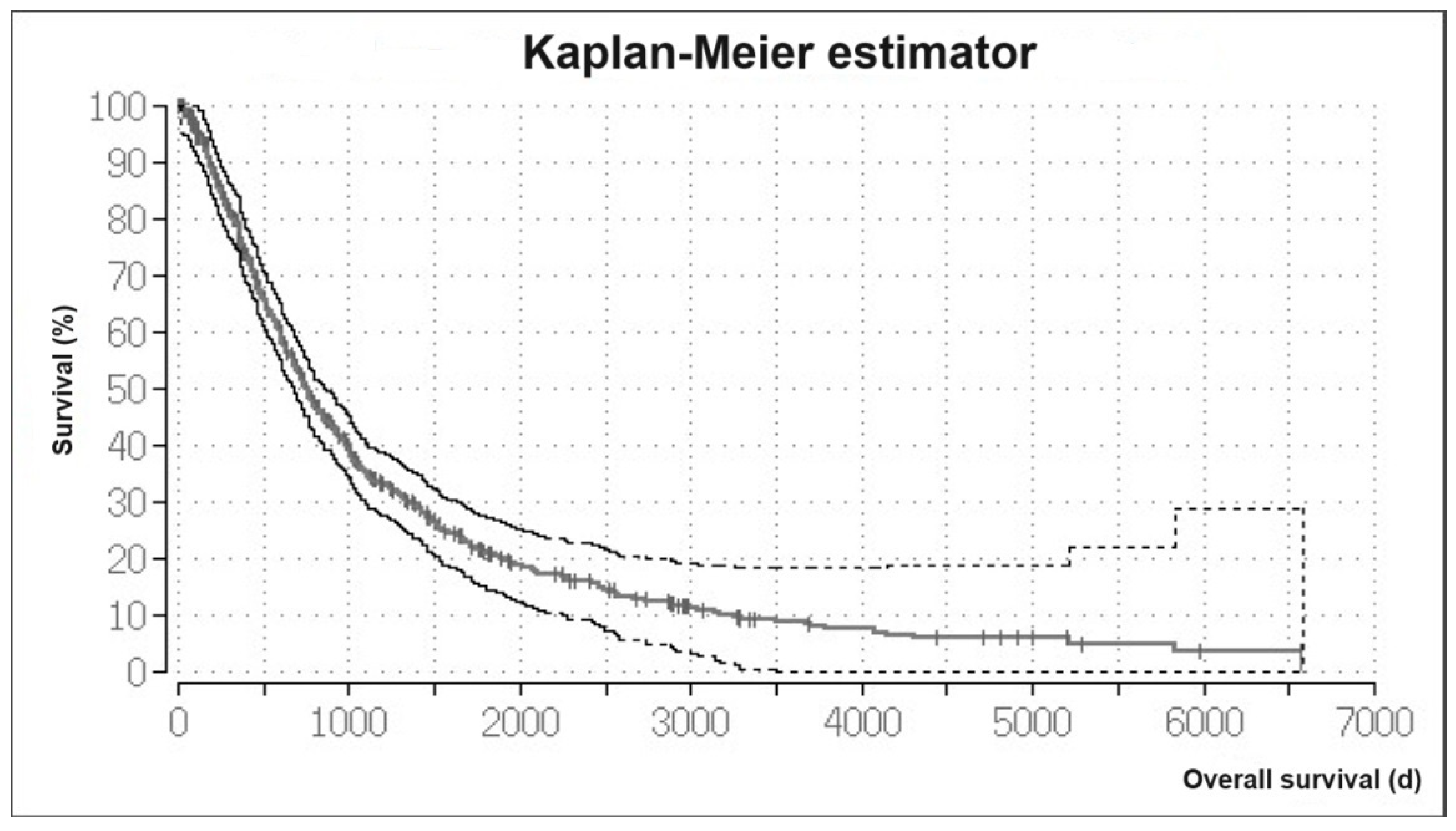
3.1. Overall Survival
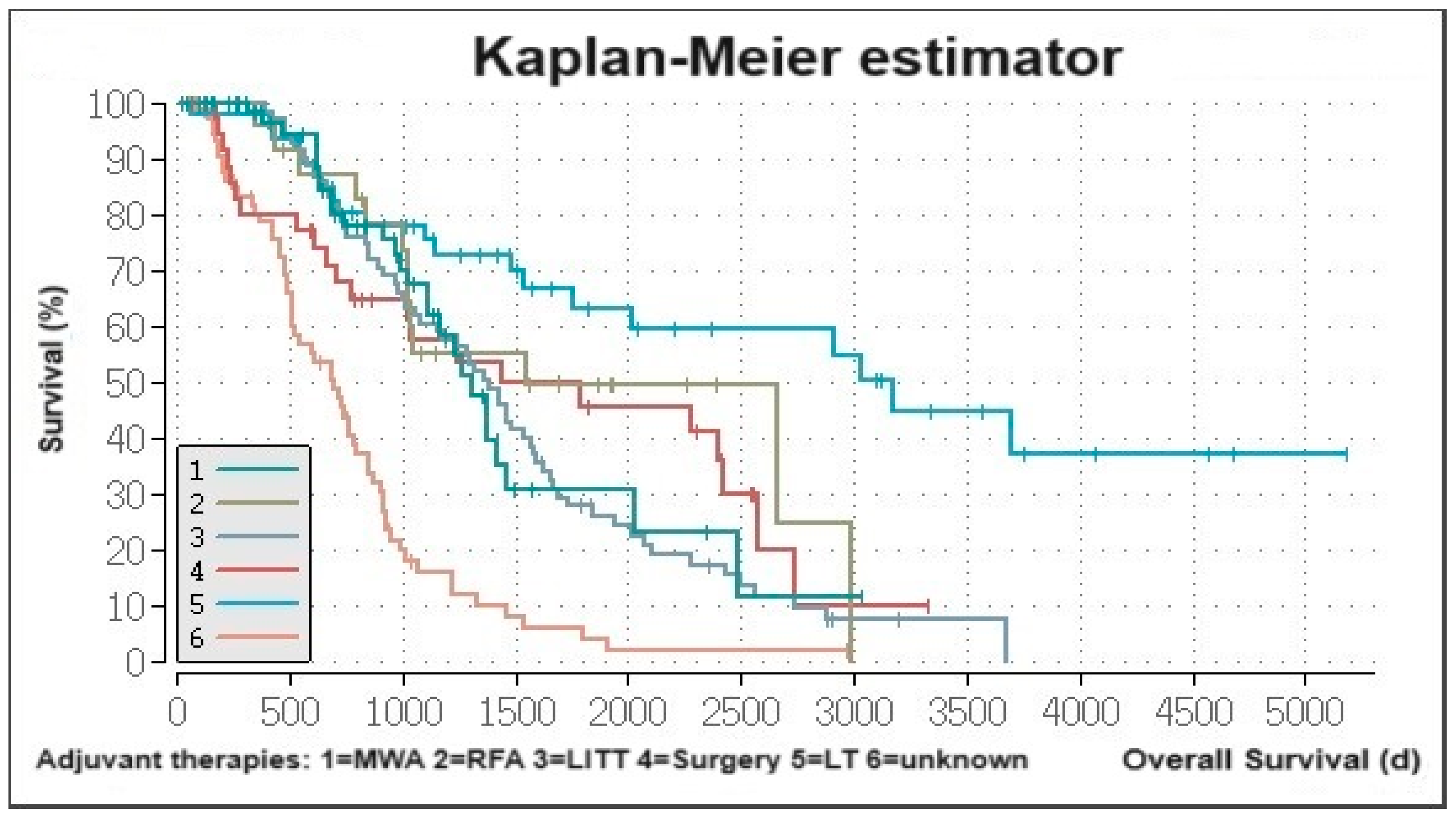
3.2. Determination of Predictors for Survival
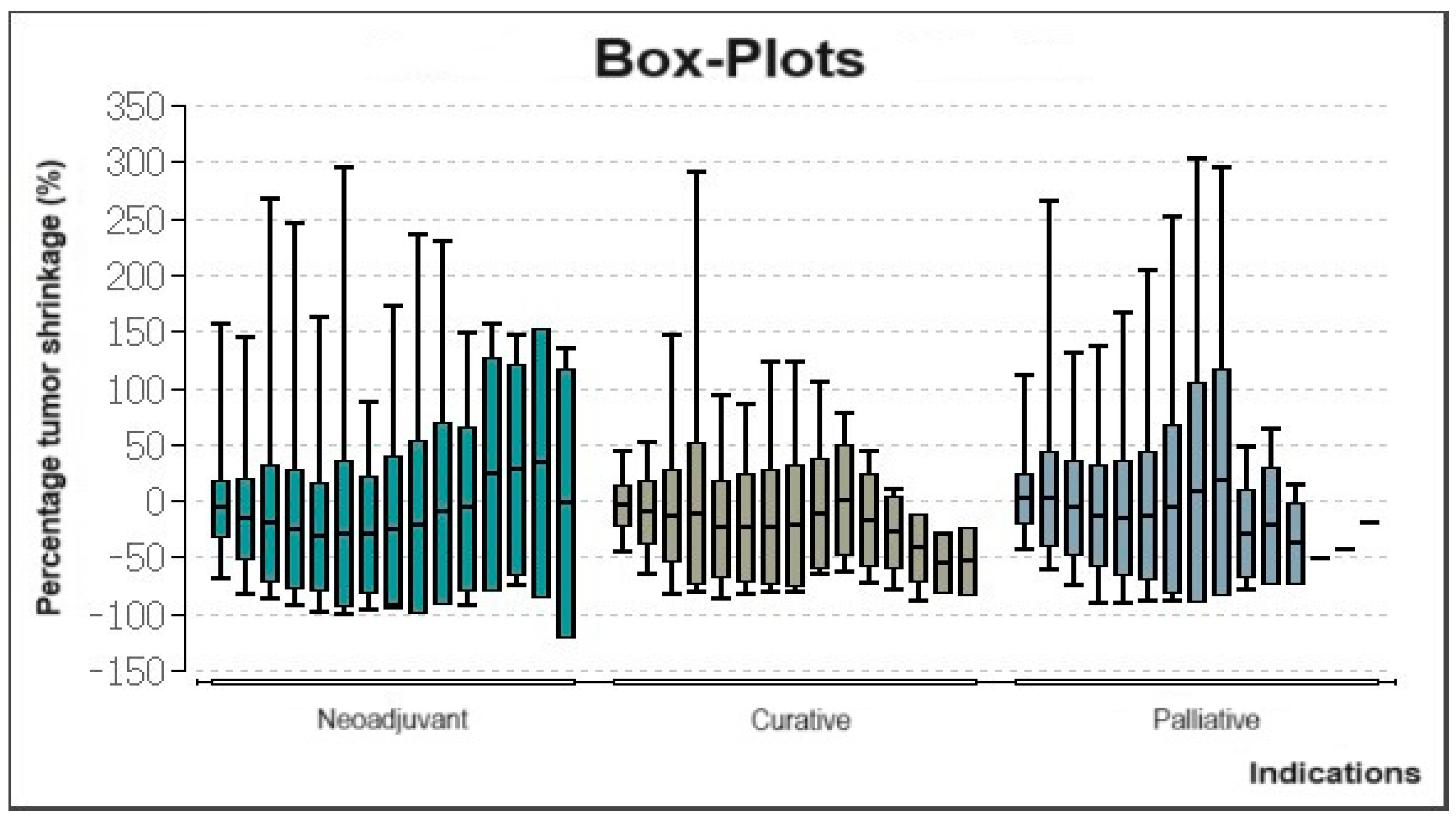
3.3. Evaluation of Tumor Volume Reduction
3.4. Chemotherapeutics
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Balogh, J.; Victor, D., 3rd; Asham, E.H.; Burroughs, S.G.; Boktour, M.; Saharia, A.; Li, X.; Ghobrial, R.M.; Monsour, H.P., Jr. Hepatocellular carcinoma: A review. J. Hepatocell. Carcinoma 2016, 3, 41–53. [Google Scholar] [CrossRef]
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA A Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef]
- Forner, A.; Llovet, J.M.; Bruix, J. Hepatocellular carcinoma. Lancet 2012, 379, 1245–1255. [Google Scholar] [CrossRef]
- Valery, P.C.; Laversanne, M.; Clark, P.J.; Petrick, J.L.; McGlynn, K.A.; Bray, F. Projections of primary liver cancer to 2030 in 30 countries worldwide. Hepatology 2018, 67, 600–611. [Google Scholar] [CrossRef]
- Philips, C.A.; Rajesh, S.; Nair, D.C.; Ahamed, R.; Abduljaleel, J.K.; Augustine, P. Hepatocellular Carcinoma in 2021: An Exhaustive Update. Cureus 2021, 13, e19274. [Google Scholar] [CrossRef]
- Herbst, D.A.; Reddy, K.R. Risk factors for hepatocellular carcinoma. Clin. Liver Dis. 2013, 1, 180–182. [Google Scholar] [CrossRef]
- Ghouri, Y.A.; Mian, I.; Rowe, J.H. Review of hepatocellular carcinoma: Epidemiology, etiology, and carcinogenesis. J. Carcinog. 2017, 16, 1. [Google Scholar] [CrossRef]
- Shimamura, T.; Goto, R.; Watanabe, M.; Kawamura, N.; Takada, Y. Liver Transplantation for Hepatocellular Carcinoma: How Should We Improve the Thresholds? Cancers 2022, 14, 419. [Google Scholar] [CrossRef]
- Lurje, I.; Czigany, Z.; Bednarsch, J.; Roderburg, C.; Isfort, P.; Neumann, U.P.; Lurje, G. Treatment Strategies for Hepatocellular Carcinoma—A Multidisciplinary Approach. Int. J. Mol. Sci. 2019, 20, 1465. [Google Scholar] [CrossRef]
- Reig, M.; Forner, A.; Rimola, J.; Ferrer-Fàbrega, J.; Burrel, M.; Garcia-Criado, Á.; Kelley, R.K.; Galle, P.R.; Mazzaferro, V.; Salem, R.; et al. BCLC strategy for prognosis prediction and treatment recommendation: The 2022 update. J. Hepatol. 2022, 76, 681–693. [Google Scholar] [CrossRef]
- Vogl, T.J.; Zangos, S.; Balzer, J.O.; Nabil, M.; Rao, P.; Eichler, K.; Bechstein, W.O.; Zeuzem, S.; Abdelkader, A. Transarterial chemoembolization (TACE) in hepatocellular carcinoma: Technique, indication and results]. RoFo Fortschritte Geb. Rontgenstrahlen Nukl. 2007, 179, 1113–1126. [Google Scholar] [CrossRef]
- Vogl, T.J.; Gruber-Rouh, T. HCC: Transarterial Therapies-What the Interventional Radiologist Can Offer. Dig. Dis. Sci. 2019, 64, 959–967. [Google Scholar] [CrossRef]
- Zhao, M.; Wang, J.P.; Li, W.; Huang, Z.L.; Zhang, F.J.; Fan, W.J.; Zhang, L.; Li, X.S.; Pan, C.C.; Wu, P.H. Comparison of safety and efficacy for transcatheter arterial chemoembolization alone and plus radiofrequency ablation in the treatment of single branch portal vein tumor thrombus of hepatocellular carcinoma and their prognosis factors. Zhonghua Yi Xue Za Zhi 2011, 91, 1167–1172. [Google Scholar]
- Kim, J.W.; Shin, S.S.; Kim, J.K.; Choi, S.K.; Heo, S.H.; Lim, H.S.; Hur, Y.H.; Cho, C.K.; Jeong, Y.Y.; Kang, H.K. Radiofrequency ablation combined with transcatheter arterial chemoembolization for the treatment of single hepatocellular carcinoma of 2 to 5 cm in diameter: Comparison with surgical resection. Korean J. Radiol. 2013, 14, 626–635. [Google Scholar] [CrossRef]
- Peng, Z.W.; Zhang, Y.J.; Chen, M.S.; Xu, L.; Liang, H.H.; Lin, X.J.; Guo, R.P.; Zhang, Y.Q.; Lau, W.Y. Radiofrequency ablation with or without transcatheter arterial chemoembolization in the treatment of hepatocellular carcinoma: A prospective randomized trial. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2013, 31, 426–432. [Google Scholar] [CrossRef]
- Li, S.; Zhang, L.; Huang, Z.M.; Wu, P.H. Transcatheter arterial chemoembolization combined with CT-guided percutaneous thermal ablation versus hepatectomy in the treatment of hepatocellular carcinoma. Chin. J. Cancer 2015, 34, 254–263. [Google Scholar] [CrossRef]
- Zhang, R.; Shen, L.; Zhao, L.; Guan, Z.; Chen, Q.; Li, W. Combined transarterial chemoembolization and microwave ablation versus transarterial chemoembolization in BCLC stage B hepatocellular carcinoma. Diagn. Interv. Radiol. 2018, 24, 219–224. [Google Scholar] [CrossRef]
- Zhang, Y.J.; Chen, M.S.; Chen, Y.; Lau, W.Y.; Peng, Z. Long-term Outcomes of Transcatheter Arterial Chemoembolization Combined with Radiofrequency Ablation as an Initial Treatment for Early-Stage Hepatocellular Carcinoma. JAMA Netw. Open 2021, 4, e2126992. [Google Scholar] [CrossRef]
- Hendriks, P.; Sudiono, D.R.; Schaapman, J.J.; Coenraad, M.J.; Tushuizen, M.E.; Takkenberg, R.B.; Oosterveer TT, M.; de Geus-Oei, L.F.; van Delden, O.M.; Burgmans, M.C. Thermal ablation combined with transarterial chemoembolization for hepatocellular carcinoma: What is the right treatment sequence? Eur. J. Radiol. 2021, 144, 110006. [Google Scholar] [CrossRef]
- Sun, Y.; Zhang, H.; Long, J.; Zhang, Y.; Zheng, J.; Yuan, C. Percutaneous thermal ablation combined with transcatheter arterial chemoembolization for hepatitis C virus-related hepatocellular carcinoma: Efficacy and survival. Front. Oncol. 2022, 12, 978614. [Google Scholar] [CrossRef]
- Bryce, K.; Tsochatzis, E.A. Downstaging for hepatocellular cancer: Harm or benefit? Transl. Gastroenterol. Hepatol. 2017, 2, 106. [Google Scholar] [CrossRef]
- Renzulli, M.; Peta, G.; Vasuri, F.; Marasco, G.; Caretti, D.; Bartalena, L.; Spinelli, D.; Giampalma, E.; D’Errico, A.; Golfieri, R. Standardization of conventional chemoembolization for hepatocellular carcinoma. Ann. Hepatol. 2021, 22, 100278. [Google Scholar] [CrossRef]
- Ikeda, M.; Arai, Y.; Inaba, Y.; Tanaka, T.; Sugawara, S.; Kodama, Y.; Aramaki, T.; Anai, H.; Morita, S.; Tsukahara, Y.; et al. Conventional or Drug-Eluting Beads? Randomized Controlled Study of Chemoembolization for Hepatocellular Carcinoma: JIVROSG-1302. Liver Cancer 2022, 11, 440–450. [Google Scholar] [CrossRef]
- Lencioni, R. Loco-regional treatment of hepatocellular carcinoma. Hepatology 2010, 52, 762–773. [Google Scholar] [CrossRef]
- Song, J.E.; Kim, D.Y. Conventional vs drug-eluting beads transarterial chemoembolization for hepatocellular carcinoma. World J. Hepatol. 2017, 9, 808–814. [Google Scholar] [CrossRef]
- Seldinger, S.I. Catheter replacement of the needle in percutaneous arteriography; a new technique. Acta Radiol. 1953, 39, 368–376. [Google Scholar] [CrossRef]
- Park, J.W.; Chen, M.; Colombo, M.; Roberts, L.R.; Schwartz, M.; Chen, P.J.; Kudo, M.; Johnson, P.; Wagner, S.; Orsini, L.S.; et al. Global patterns of hepatocellular carcinoma management from diagnosis to death: The BRIDGE Study. Liver Int. Off. J. Int. Assoc. Study Liver 2015, 35, 2155–2166. [Google Scholar] [CrossRef]
- Verslype, C.; Rosmorduc, O.; Rougier, P.; ESMO Guidelines Working Group. Hepatocellular carcinoma: ESMO-ESDO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann. Oncol. Off. J. Eur. Soc. Med. Oncol. 2012, 23 (Suppl. 7), vii41–vii48. [Google Scholar] [CrossRef]
- Kong, J.Y.; Li, S.M.; Fan, H.Y.; Zhang, L.; Zhao, H.J.; Li, S.M. Transarterial chemoembolization extends long-term survival in patients with unresectable hepatocellular carcinoma. Medicine 2018, 97, e11872. [Google Scholar] [CrossRef]
- Akarapatima, K.; Chang, A.; Prateepchaiboon, T.; Pungpipattrakul, N.; Songjamrat, A.; Pakdeejit, S.; Rattanasupar, A. Comparison of Overall Survival between Transarterial Chemoembolization and Best Supportive Care in Intermediate- Stage Hepatocellular Carcinoma. Asian Pac. J. Cancer Prev. 2022, 23, 3173–3178. [Google Scholar] [CrossRef]
- Huo, Y.R.; Eslick, G.D. Transcatheter Arterial Chemoembolization Plus Radiotherapy Compared with Chemoembolization Alone for Hepatocellular Carcinoma: A Systematic Review and Meta-analysis. JAMA Oncol. 2015, 1, 756–765. [Google Scholar] [CrossRef]
- Su, T.S.; Li, L.Q.; Meng, W.W.; Wang, Y.D.; Chen, Y.T.; Li, J.X.; Du, Y.Q.; Qu, S.; Zhao, C.; Huang, D.J.; et al. Long-Term Survival Analysis of Transarterial Chemoembolization Plus Radiotherapy vs. Radiotherapy for Hepatocellular Carcinoma with Macroscopic Vascular Invasion. Front Oncol. 2020, 10, 1205. [Google Scholar] [CrossRef]
- Yang, Y.; Yu, H.; Qi, L.; Liu, C.; Feng, Y.; Qi, J.; Li, J.; Zhu, Q. Combined radiofrequency ablation or microwave ablation with transarterial chemoembolization can increase efficiency in intermediate-stage hepatocellular carcinoma without more complication: A systematic review and meta-analysis. Int. J. Hyperth. 2022, 39, 455–465. [Google Scholar] [CrossRef]
- Parikh, N.D.; Waljee, A.K.; Singal, A.G. Downstaging hepatocellular carcinoma: A systematic review and pooled analysis. Liver Transpl. 2015, 21, 1142–1152. [Google Scholar] [CrossRef]
- Mao, Y.M.; Luo, Z.Y.; Li, B.; Hu, T.Y. Prospective study on the survival of HCC patients treated with transcatheter arterial lipiodol chemoembolization. Asian Pac. J. Cancer Prev. 2012, 13, 1039–1042. [Google Scholar] [CrossRef][Green Version]
- Wu, K.T.; Wang, C.C.; Lu, L.G.; Zhang, W.D.; Zhang, F.J.; Shi, F.; Li, C.X. Hepatocellular carcinoma: Clinical study of long-term survival and choice of treatment modalities. World J. Gastroenterol. 2013, 19, 3649–3657. [Google Scholar] [CrossRef]
- Chang, H.C.; Lin, Y.M.; Yen, A.M.; Chen, S.L.; Wu, W.Y.; Chiu, S.Y.; Fann, J.C.; Lin, Y.S.; Chen, H.H.; Liao, C.S. Predictors of long-term survival in hepatocellular carcinomas: A longitudinal follow-up of 108 patients with small tumors. Anticancer Res. 2013, 33, 5171–5178. [Google Scholar]
- Liu, L.; Zhang, C.; Zhao, Y.; Qi, X.; Chen, H.; Bai, W.; He, C.; Guo, W.; Yin, Z.; Fan, D.; et al. Transarterial chemoembolization for the treatment of advanced hepatocellular carcinoma with portal vein tumor thrombosis: Prognostic factors in a single-center study of 188 patients. BioMed Res. Int. 2014, 2014, 194278. [Google Scholar] [CrossRef]
| Characteristic | |
|---|---|
| Total number of patients | 836 |
| Mean age in years | 74.18 |
| Sex | |
| Male (%) | 653 (78.1%) |
| Female (%) | 183 (21.9%) |
| Child–Pugh class (%) | |
| A | 495 (59.21%) |
| B | 188 (22.49%) |
| C | 122 (14.6%) |
| Unknown | 31 (3.7%) |
| Portal vein thrombosis | |
| Complete | 86 (10.3%) |
| Partial | 57 (6.8%) |
| None | 629 (75.2%) |
| Unknown | 64 (7.7%) |
| Mean spleen size | 12.8 cm |
| Splenomegaly (size ≥ 12.0 cm) | 365 (43.66%) |
| Total number of cTACE | 4084 |
| Mean number of TACE sessions/ patient | 4.89 |
| Patients with ≤3 TACE performed | 460 (55%) |
| Patients with 4–6 TACE performed | 243 (29.1%) |
| Patients with >6 TACE performed | 133 (15.9%) |
| Indications | |
| Neoadjuvant | 394 (47.1%) |
| Curative or Palliative | 431 (51.6%) |
| Unknown | 11 (1.3%) |
| Interventions/treatments after neoadjuvant TACE | |
| Microwave ablation | 112 (28.43%) |
| Radiofrequency ablation | 34 (8.63%) |
| Laser-induced thermotherapy | 78 (19.8%) |
| Selective internal radiation therapy (SIRT) | 2 (0.5%) |
| Surgical resection | 39 (9.9%) |
| Liver transplantation | 49 (12.44%) |
| Unknown | 80 (20.3%) |
| Chemotherapeutic Regime | 1996–2001 | 2002–2006 | 2007–2011 | 2012–2016 |
|---|---|---|---|---|
| Mitomycin C Gemcitabine Cisplatin | 6 (1%) | 44 (3.4%) | 342 (27.3%) | 308 (41.8%) |
| Mitomycin C Cisplatin | 3 (0.5%) | 19 (1.4%) | 245 (19.6%) | 183 (24.9%) |
| Mitomycin C | 506 (81.6%) | 522 (39.8%) | 395 (31.5%) | 203 (27.6%) |
| Mitomycin C Gemcitabine | 67 (10.8%) | 592 (45.1%) | 156 (12.4%) | 7 (1%) |
| Other combinations/ chemotherapeutics | 38 (6.1%) | 135 (10.3%) | 115 (9.2) | 35 (4.7%) |
| ∑ (%) | 620 (100%) | 1312 (100%) | 1253 (100%) | 736 (100%) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vogl, T.J.; Adwan, H.; Wolff, L.; Lahrsow, M.; Gruber-Rouh, T.; Nour-Eldin, N.-E.A.; Trojan, J.; Bechstein, W.-O.; Naguib, N.N.N. Retrospective Long-Term Evaluation of Conventional Transarterial Chemoembolization for Hepatocellular Carcinoma over 20 Years. Cancers 2024, 16, 1498. https://doi.org/10.3390/cancers16081498
Vogl TJ, Adwan H, Wolff L, Lahrsow M, Gruber-Rouh T, Nour-Eldin N-EA, Trojan J, Bechstein W-O, Naguib NNN. Retrospective Long-Term Evaluation of Conventional Transarterial Chemoembolization for Hepatocellular Carcinoma over 20 Years. Cancers. 2024; 16(8):1498. https://doi.org/10.3390/cancers16081498
Chicago/Turabian StyleVogl, Thomas J., Hamzah Adwan, Leonard Wolff, Maximilian Lahrsow, Tatjana Gruber-Rouh, Nour-Eldin Abdelrehim Nour-Eldin, Jörg Trojan, Wolf-Otto Bechstein, and Nagy N. N. Naguib. 2024. "Retrospective Long-Term Evaluation of Conventional Transarterial Chemoembolization for Hepatocellular Carcinoma over 20 Years" Cancers 16, no. 8: 1498. https://doi.org/10.3390/cancers16081498
APA StyleVogl, T. J., Adwan, H., Wolff, L., Lahrsow, M., Gruber-Rouh, T., Nour-Eldin, N.-E. A., Trojan, J., Bechstein, W.-O., & Naguib, N. N. N. (2024). Retrospective Long-Term Evaluation of Conventional Transarterial Chemoembolization for Hepatocellular Carcinoma over 20 Years. Cancers, 16(8), 1498. https://doi.org/10.3390/cancers16081498









