Racial Differences in the Detection Rate of Bladder Cancer Using Blue Light Cystoscopy: Insights from a Multicenter Registry
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Population
2.2. Procedure
2.3. Study Parameters and Outcome Measures
2.4. Statistical Analysis
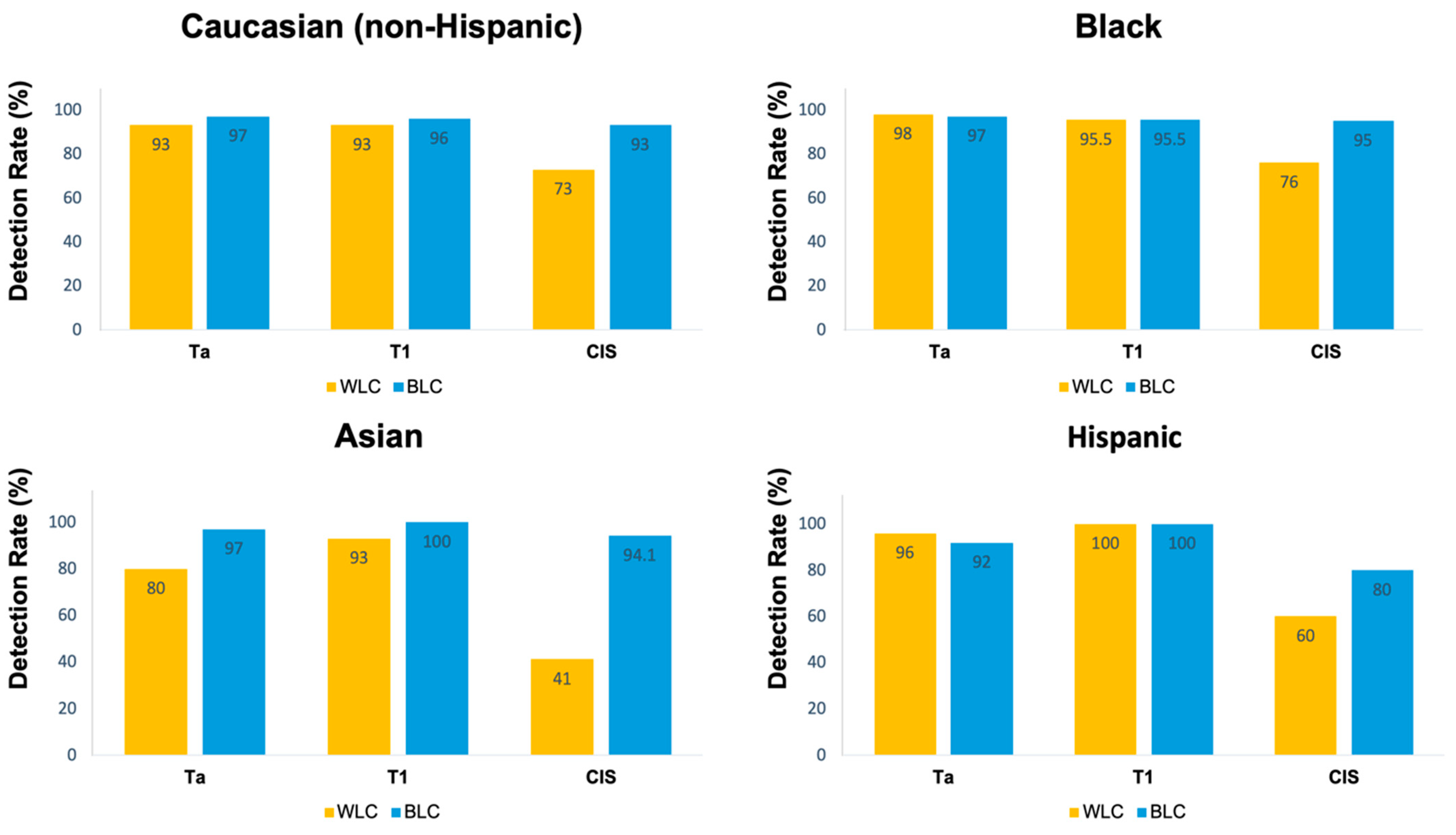
3. Results
3.1. Baseline Clinical Features
3.2. Sensitivity
3.3. PPV and NPV
3.4. CIS Lesions
3.5. Adverse Events
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Siegel, R.L.; Miller, K.D.; Wagle, N.S.; Jemal, A. Cancer statistics, 2023. CA Cancer J. Clin. 2023, 73, 17–48. [Google Scholar] [CrossRef] [PubMed]
- Sylvester, R.J.; Van Der Meijden, A.P.; Oosterlinck, W.; Witjes, J.A.; Bouffioux, C.; Denis, L.; Newling, D.W.; Kurth, K. Predicting recurrence and progression in individual patients with stage Ta T1 bladder cancer using EORTC risk tables: A combined analysis of 2596 patients from seven EORTC trials. Eur. Urol. 2006, 49, 466–477; discussion 475–477. [Google Scholar] [CrossRef] [PubMed]
- Chang, S.S.; Boorjian, S.A.; Chou, R.; Clark, P.E.; Daneshmand, S.; Konety, B.R.; Pruthi, R.; Quale, D.Z.; Ritch, C.R.; Seigne, J.D.; et al. Diagnosis and Treatment of Non-Muscle Invasive Bladder Cancer: AUA/SUO Guideline. J. Urol. 2016, 196, 1021–1029. [Google Scholar] [CrossRef] [PubMed]
- Fradet, Y.; Grossman, H.B.; Gomella, L.; Lerner, S.; Cookson, M.; Albala, D.; Droller, M.J.; PC B302/01 Study Group. A comparison of hexaminolevulinate fluorescence cystoscopy and white light cystoscopy for the detection of carcinoma in situ in patients with bladder cancer: A phase III, multicenter study. J. Urol. 2007, 178, 68–73; discussion 73. [Google Scholar] [CrossRef] [PubMed]
- Daneshmand, S.; Bazargani, S.T.; Bivalacqua, T.J.; Holzbeierlein, J.M.; Willard, B.; Taylor, J.M.; Liao, J.C.; Pohar, K.; Tierney, J.; Konety, B. Blue light cystoscopy for the diagnosis of bladder cancer: Results from the US prospective multicenter registry. Urol. Oncol. 2018, 36, 361.e1–361.e6. [Google Scholar] [CrossRef]
- Palou, J.; Hernández, C.; Solsona, E.; Abascal, R.; Burgués, J.P.; Rioja, C.; Cabrera, J.A.; Gutiérrez, C.; Rodríguez, O.; Iborra, I.; et al. Effectiveness of hexaminolevulinate fluorescence cystoscopy for the diagnosis of non-muscle-invasive bladder cancer in daily clinical practice: A Spanish multicentre observational study. BJU Int. 2015, 116, 37–43. [Google Scholar] [CrossRef] [PubMed]
- Kamat, A.M.; Cookson, M.; Witjes, J.A.; Stenzl, A.; Grossman, H.B. The Impact of Blue Light Cystoscopy with Hexaminolevulinate (HAL) on Progression of Bladder Cancer—A New Analysis. Bladder Cancer Amst. Neth. 2016, 2, 273–278. [Google Scholar] [CrossRef] [PubMed]
- Jocham, D.; Witjes, F.; Wagner, S.; Zeylemaker, B.; van Moorselaar, J.E.R.O.E.N.; Grimm, M.O.; Muschter, R.; Popken, G.; König, F.R.A.N.K.; Knüchel, R.U.T.H.; et al. Improved detection and treatment of bladder cancer using hexaminolevulinate imaging: A prospective, phase III multicenter study. J. Urol. 2005, 174, 862–866; discussion 866. [Google Scholar] [CrossRef] [PubMed]
- Fletcher, S.A.; Bivalacqua, T.J.; Brawley, O.W.; Kates, M. Race, ethnicity, and gender reporting in North American clinical trials for BCG-unresponsive non-muscle invasive bladder cancer. Urol. Oncol. 2022, 40, 195.e13–195.e18. [Google Scholar] [CrossRef] [PubMed]
- Yuan, H.; Qiu, J.; Liu, L.; Zheng, S.; Yang, L.; Liu, Z.; Pu, C.; Li, J.; Wei, Q.; Han, P. Therapeutic outcome of fluorescence cystoscopy guided transurethral resection in patients with non-muscle invasive bladder cancer: A meta-analysis of randomized controlled trials. PLoS ONE 2013, 8, e74142. [Google Scholar] [CrossRef]
- Daneshmand, S.; Schuckman, A.K.; Bochner, B.H.; Cookson, M.S.; Downs, T.M.; Gomella, L.G.; Grossman, H.B.; Kamat, A.M.; Konety, B.R.; Lee, C.T.; et al. Hexaminolevulinate blue-light cystoscopy in non-muscle-invasive bladder cancer: Review of the clinical evidence and consensus statement on appropriate use in the USA. Nat. Rev. Urol. 2014, 11, 589–596. [Google Scholar] [CrossRef] [PubMed]
- Burger, M.; Grossman, H.B.; Droller, M.; Schmidbauer, J.; Hermann, G.; Drăgoescu, O.; Ray, E.; Fradet, Y.; Karl, A.; Burgués, J.P.; et al. Photodynamic diagnosis of non-muscle-invasive bladder cancer with hexaminolevulinate cystoscopy: A meta-analysis of detection and recurrence based on raw data. Eur. Urol. 2013, 64, 846–854. [Google Scholar] [CrossRef] [PubMed]
- Hermann, G.G.; Mogensen, K.; Carlsson, S.; Marcussen, N.; Duun, S. Fluorescence-guided transurethral resection of bladder tumours reduces bladder tumour recurrence due to less residual tumour tissue in Ta/T1 patients: A randomized two-centre study. BJU Int. 2011, 108, E297–E303. [Google Scholar] [CrossRef] [PubMed]
- Stenzl, A.; Burger, M.; Fradet, Y.; Mynderse, L.A.; Soloway, M.S.; Witjes, J.A.; Kriegmair, M.; Karl, A.; Shen, Y.; Grossman, H.B. Hexaminolevulinate guided fluorescence cystoscopy reduces recurrence in patients with nonmuscle invasive bladder cancer. J. Urol. 2010, 184, 1907–1913. [Google Scholar] [CrossRef] [PubMed]
- Grossman, H.B.; Gomella, L.; Fradet, Y.; Morales, A.; Presti, J.; Ritenour, C.; Nseyo, U.; Droller, M.J.; PC B302/01 Study Group. A phase III, multicenter comparison of hexaminolevulinate fluorescence cystoscopy and white light cystoscopy for the detection of superficial papillary lesions in patients with bladder cancer. J. Urol. 2007, 178, 62–67. [Google Scholar] [CrossRef] [PubMed]
- Babjuk, M.; Burger, M.; Capoun, O.; Cohen, D.; Compérat, E.M.; Escrig, J.L.D.; Gontero, P.; Liedberg, F.; Masson-Lecomte, A.; Mostafid, A.H.; et al. European Association of Urology Guidelines on Non-muscle-invasive Bladder Cancer (Ta, T1, and Carcinoma In Situ). Eur. Urol. 2022, 81, 75–94. [Google Scholar] [CrossRef] [PubMed]
- Mandalapu, R.S.; Remzi, M.; de Reijke, T.M.; Margulis, V.; Palou, J.; Kapoor, A.; Yossepowitch, O.; Coleman, J.; Traxer, O.; Anderson, J.K.; et al. Update of the ICUD-SIU consultation on upper tract urothelial carcinoma 2016: Treatment of localized high-risk disease. World J. Urol. 2017, 35, 327–335. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Chang, Q.; Li, Y. Racial differences in Urinary Bladder Cancer in the United States. Sci. Rep. 2018, 8, 12521. [Google Scholar] [CrossRef]
- Sung, J.M.; Martin, J.W.; Jefferson, F.A.; Sidhom, D.A.; Piranviseh, K.; Huang, M.; Nguyen, N.; Chang, J.; Ziogas, A.; Anton-Culver, H.; et al. Racial and Socioeconomic Disparities in Bladder Cancer Survival: Analysis of the California Cancer Registry. Clin. Genitourin. Cancer 2019, 17, e995–e1002. [Google Scholar] [CrossRef] [PubMed]
- Ghoreifi, A.; Mitra, A.P.; Cai, J.; Miranda, G.; Daneshmand, S.; Djaladat, H. Perioperative complications and oncological outcomes following radical cystectomy among different racial groups: A long-term, single-center study. Can. Urol. Assoc. J. J. Assoc. Urol. Can. 2020, 14, E493–E498. [Google Scholar] [CrossRef]
- Washington, S.L.; Gregorich, S.E.; Meng, M.V.; Suskind, A.M.; Porten, S.P. Race modifies survival benefit of guideline-based treatment: Implications for reducing disparities in muscle invasive bladder cancer. Cancer Med. 2020, 9, 8310–8317. [Google Scholar] [CrossRef] [PubMed]
- Tang, D.H.; Chang, S.S. Management of carcinoma in situ of the bladder: Best practice and recent developments. Ther. Adv. Urol. 2015, 7, 351–364. [Google Scholar] [CrossRef] [PubMed]
- Freudenburg, E.; Bagheri, I.; Srinivas, S.; Martinez, A.; Putluri, N.; Klaassen, Z.; Kamat, A.M.; Konety, B.R.; Kim, W.Y.; Dyrskjøt, L.; et al. Race reporting and disparities regarding clinical trials in bladder cancer: A systematic review. Cancer Causes Control 2022, 33, 1071–1081. [Google Scholar] [CrossRef] [PubMed]
| Caucasian (Non-Hispanic) | African American | Asian | Hispanic | |
|---|---|---|---|---|
| Age (mean ± SD), year | 70.2 ± 9.4 | 67.5 ± 8.6 | 73.8 ± 6.3 | 72.2 ± 9.5 |
| Gender (Male%) | 83 | 80 | 78 | 84.5 |
| Smoker (%) | 67 | 70 | 57 | 64 |
| Primary occurrence (%) | 34 | 40 | 50 | 53 |
| Pathological stage (%) | ||||
| Ta | 60 | 61 | 50 | 62 |
| T1 | 20 | 20 | 23.5 | 26 |
| CIS * | 20 | 19 | 26.5 | 12 |
| Pathological grade (%) | ||||
| Benign | 24.5 | 28 | 19 | 41 |
| PUNLMP | 0.5 | 4 | 0 | 1 |
| Low grade | 22 | 20 | 6 | 18 |
| High grade | 53 | 48 | 80 | 40 |
| Race (Number of Lesions) | Sensitivity | DR of BLC | Increase in DR by BLC | PPV for BLC | NPV for BLC | |||
|---|---|---|---|---|---|---|---|---|
| WLC Only | BLC Only | Either WLC or BLC | HG | CIS | ||||
| Caucasian (non-Hispanic) (2011) | 87 | 96 | 99 | 94 | 93 | 11 | 82 | 69 |
| African American (178) | 93 | 97 | 99 | 97 | 95 | 6 | 77 | 60 |
| Asian (99) | 78 | 96 | 97 | 94 | 94 | 18 | 94 | 60 |
| Hispanic (91) | 95 | 95 | 97 | 97 | 80 | 2 | 88 | 86 |
| Total (2379) | 89 | 96 | 99 | 94 | 93 | 10 | 82 | 70 |
| Race | Odds Ratio | 95% CI | p-Value (Score Test) # |
|---|---|---|---|
| Caucasian (non-Hispanic) | 3.0 | 2.2–4.1 | <0.001 |
| African American | 2.7 | 0.7–10.1 | 0.13 |
| Asian | 16.0 | 2.1–120.6 | <0.001 |
| Hispanic | 1.0 | 0.06–16.0 | 1.0 |
| Race | Sensitivity | Specificity | PPV | NPV |
|---|---|---|---|---|
| Caucasian (non-Hispanic) | 93 | 21 | 53 | 76 |
| African American | 95 | 11 | 39 | 80 |
| Asian | 94 | 40 | 73 | 80 |
| Hispanic | 80 | 70 | 36 | 94 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ladi-Seyedian, S.-S.; Ghoreifi, A.; Konety, B.; Pohar, K.; Holzbeierlein, J.M.; Taylor, J.; Kates, M.; Willard, B.; Taylor, J.M.; Liao, J.C.; et al. Racial Differences in the Detection Rate of Bladder Cancer Using Blue Light Cystoscopy: Insights from a Multicenter Registry. Cancers 2024, 16, 1268. https://doi.org/10.3390/cancers16071268
Ladi-Seyedian S-S, Ghoreifi A, Konety B, Pohar K, Holzbeierlein JM, Taylor J, Kates M, Willard B, Taylor JM, Liao JC, et al. Racial Differences in the Detection Rate of Bladder Cancer Using Blue Light Cystoscopy: Insights from a Multicenter Registry. Cancers. 2024; 16(7):1268. https://doi.org/10.3390/cancers16071268
Chicago/Turabian StyleLadi-Seyedian, Seyedeh-Sanam, Alireza Ghoreifi, Badrinath Konety, Kamal Pohar, Jeffrey M. Holzbeierlein, John Taylor, Max Kates, Brian Willard, Jennifer M. Taylor, Joseph C. Liao, and et al. 2024. "Racial Differences in the Detection Rate of Bladder Cancer Using Blue Light Cystoscopy: Insights from a Multicenter Registry" Cancers 16, no. 7: 1268. https://doi.org/10.3390/cancers16071268
APA StyleLadi-Seyedian, S.-S., Ghoreifi, A., Konety, B., Pohar, K., Holzbeierlein, J. M., Taylor, J., Kates, M., Willard, B., Taylor, J. M., Liao, J. C., Kaimakliotis, H. Z., Porten, S. P., Steinberg, G. D., Tyson, M. D., Lotan, Y., Daneshmand, S., & Blue Light Cystoscopy with Cysview Registry Group. (2024). Racial Differences in the Detection Rate of Bladder Cancer Using Blue Light Cystoscopy: Insights from a Multicenter Registry. Cancers, 16(7), 1268. https://doi.org/10.3390/cancers16071268







