Rare Hematologic Malignancies and Pre-Leukemic Entities in Children and Adolescents Young Adults
Abstract
Simple Summary
Abstract
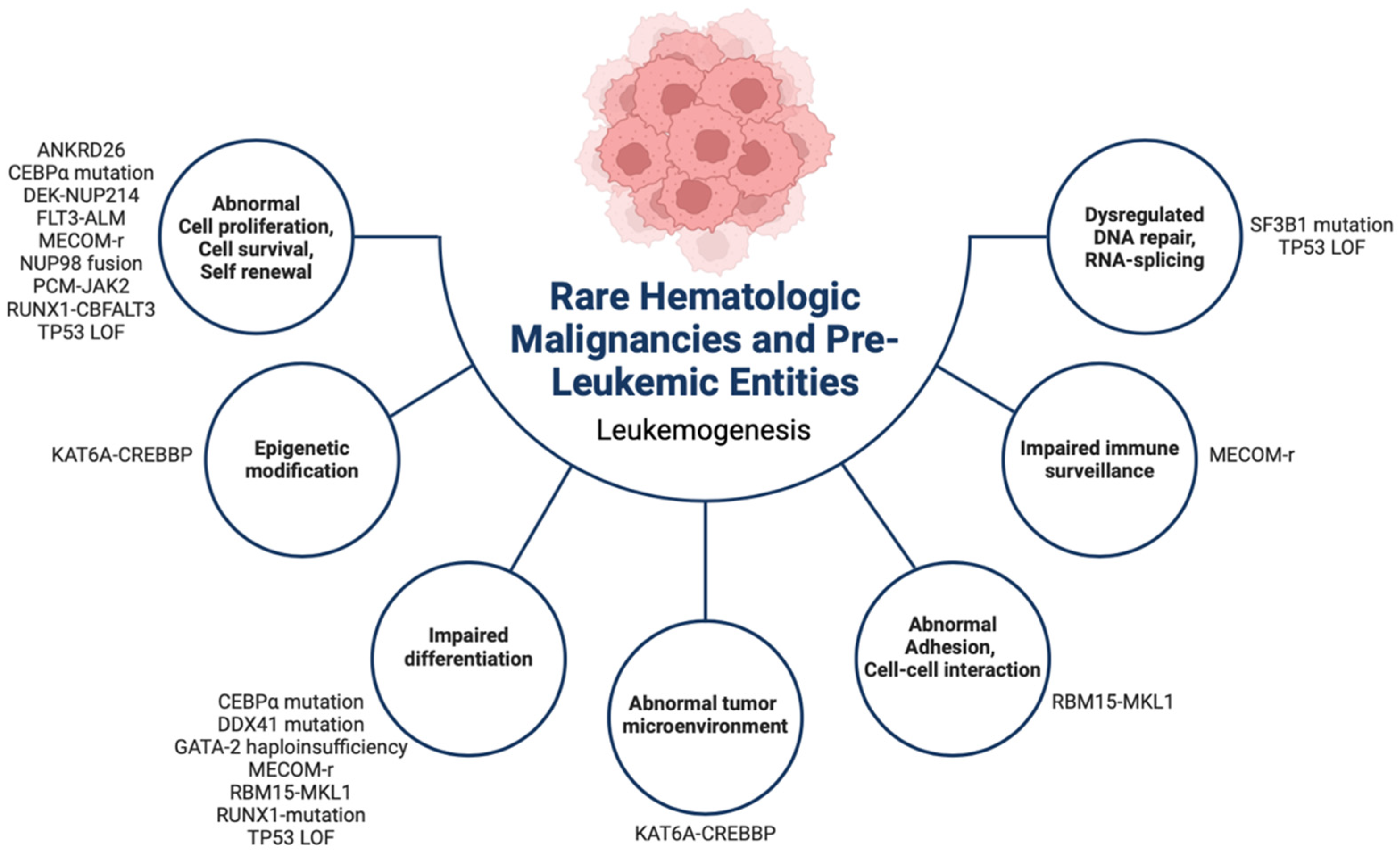
1. Introduction
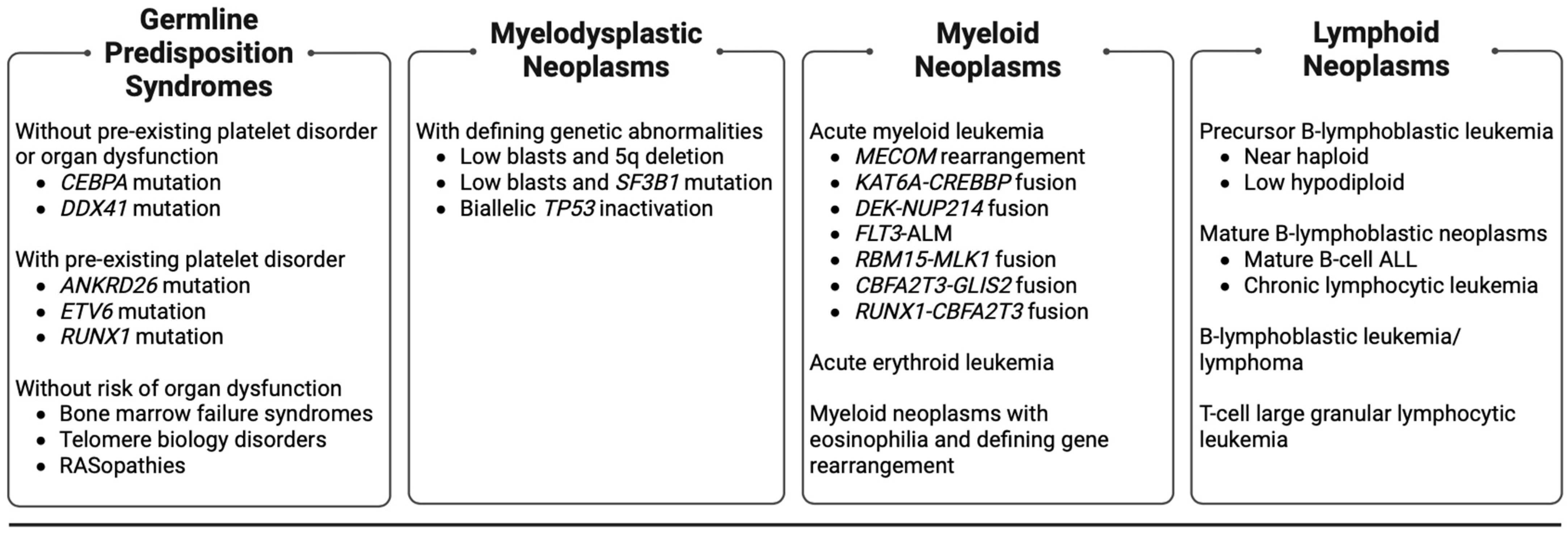
2. Myelodysplastic Neoplasms, Rare Entities
2.1. Myelodysplastic Neoplasms
2.2. MDS with Low Blasts and 5q Deletion
2.2.1. Pathologic and Cytogenetic Features
2.2.2. Treatment
2.3. MDS with Low Blasts and SF3B1 Mutation
2.3.1. Pathologic and Cytogenetic Features
2.3.2. Treatment
2.4. MDS with Biallelic TP53 Inactivation
2.4.1. Pathologic and Cytogenetic Features
2.4.2. Treatment
3. Myeloid Neoplasms, Rare Entities
3.1. AML
3.2. AML with MECOM Rearrangement
3.2.1. Pathologic and Cytogenetic Features
3.2.2. Treatment
3.2.3. MECOM-Associated Syndrome
3.3. AML with KAT6A-CREBBP Fusion
3.3.1. Pathologic and Cytogenetic Features
3.3.2. Treatment
3.4. AML with DEK-NUP214 Fusion
3.4.1. Pathologic and Cytogenetic Features
3.4.2. Treatment
3.5. AML with FLT3-ALM
3.5.1. Pathologic and Cytogenetic Features
3.5.2. Treatment
3.6. AML with Megakaryoblastic Differentiation, RBM15-MKL1 Fusion
3.6.1. Pathologic and Cytogenetic Features
3.6.2. Treatment
3.7. AML with Megakaryoblastic Differentiation, CBFA2T3-GLIS2 Fusion
3.7.1. Pathologic and Cytogenetic Features
3.7.2. Treatment
3.8. AML with RUNX1-CBFA2T3 Fusion
3.8.1. Pathologic and Cytogenetic Features
3.8.2. Treatment
3.9. Acute Erythroid Leukemia
3.9.1. Pathologic and Cytogenetic Features
3.9.2. Treatment
3.10. Myeloid Neoplasms with Eosinophilia and Defining Gene Rearrangement
3.10.1. Pathologic and Cytogenetic Features
3.10.2. Treatment
4. Syndromes Predisposing to Myelodysplastic and Myeloid Neoplasms, Rare Entities
4.1. Syndromes Predisposing to Myelodysplastic and Myeloid Neoplasms
4.2. Myeloid Neoplasms with Germline Predisposition without a Pre-Existing Platelet Disorder or Risk of Organ Dysfunction with CEBPA and DDX41 Mutations
4.3. Myeloid Neoplasms with Germline Predisposition and Pre-Existing Platelet Disorders with RUNX1, ANKRD26, and ETV6 Mutations
4.4. Myeloid Neoplasms with Germline Predisposition and Risk of Organ Dysfunctions
5. B-Cell Lymphoid Proliferations, Rare Entities
5.1. B-Cell Lymphoid Neoplasms
5.2. Near Haploid ALL (24 to 30 Chromosomes)
5.2.1. Pathologic and Cytogenetic Features
5.2.2. Treatment
5.3. Low Hypodiploid ALL (31 to 39 Chromosomes)
5.3.1. Pathologic and Cytogenetic Features
5.3.2. Treatment
5.4. Mature B-Cell ALL
5.4.1. Pathologic and Cytogenetic Features
5.4.2. Treatment
5.5. B-Lymphoblastic Leukemia/Lymphoma
5.5.1. Pathologic and Cytogenetic Features
5.5.2. Treatment
5.6. Chronic Lymphocytic Leukemia
5.6.1. Pathologic and Cytogenetic Features
5.6.2. Treatment
6. T-Cell Lymphoid Proliferations, Rare Entities
6.1. T-Cell Large Granular Lymphocytic Leukemia
6.1.1. Pathologic and Cytogenetic Features
6.1.2. Treatment
7. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Orkin, S.H.; Nathan, D.G.; Ginsburg, D.; Look, A.T.; Fisher, D.E.; Lux, S. Nathan and Oski’s Hematology and Oncology of Infancy and Childhood; Elsevier Health Sciences: Philedelphia, PA, USA, 2014. [Google Scholar]
- Khoury, J.D.; Solary, E.; Abla, O.; Akkari, Y.; Alaggio, R.; Apperley, J.F.; Bejar, R.; Berti, E.; Busque, L.; Chan, J.K.C.; et al. The 5th edition of the World Health Organization Classification of Haematolymphoid Tumours: Myeloid and Histiocytic/Dendritic Neoplasms. Leukemia 2022, 36, 1703–1719. [Google Scholar] [CrossRef]
- Malcovati, L.; Porta, M.G.; Pascutto, C.; Invernizzi, R.; Boni, M.; Travaglino, E.; Passamonti, F.; Arcaini, L.; Maffioli, M.; Bernasconi, P.; et al. Prognostic factors and life expectancy in myelodysplastic syndromes classified according to WHO criteria: A basis for clinical decision making. J. Clin. Oncol. 2005, 23, 7594–7603. [Google Scholar] [CrossRef]
- Chisholm, K.M.; Bohling, S.D. Childhood Myelodysplastic Syndrome. Clin. Lab. Med. 2023, 43, 639–655. [Google Scholar] [CrossRef]
- Pastor, V.; Hirabayashi, S.; Karow, A.; Wehrle, J.; Kozyra, E.J.; Nienhold, R.; Ruzaike, G.; Lebrecht, D.; Yoshimi, A.; Niewisch, M.; et al. Mutational landscape in children with myelodysplastic syndromes is distinct from adults: Specific somatic drivers and novel germline variants. Leukemia 2017, 31, 759–762. [Google Scholar] [CrossRef]
- Marques, F.K.; Sabino, A.P. Myelodysplastic neoplasms: An overview on diagnosis, risk-stratification, molecular pathogenesis, and treatment. Biomed. Pharmacother. 2022, 156, 113905. [Google Scholar] [CrossRef]
- Kardos, G.; Baumann, I.; Passmore, S.J.; Locatelli, F.; Hasle, H.; Schultz, K.R.; Starý, J.; Schmitt-Graeff, A.; Fischer, A.; Harbott, J.; et al. Refractory anemia in childhood: A retrospective analysis of 67 patients with particular reference to monosomy 7. Blood 2003, 102, 1997–2003. [Google Scholar] [CrossRef]
- Aalbers, A.M.; van den Heuvel-Eibrink, M.M.; de Haas, V.; Te Marvelde, J.G.; de Jong, A.X.; van der Burg, M.; Dworzak, M.; Hasle, H.; Locatelli, F.; De Moerloose, B.; et al. Applicability of a reproducible flow cytometry scoring system in the diagnosis of refractory cytopenia of childhood. Leukemia 2013, 27, 1923–1925. [Google Scholar] [CrossRef]
- Aalbers, A.M.; van den Heuvel-Eibrink, M.M.; Baumann, I.; Dworzak, M.; Hasle, H.; Locatelli, F.; De Moerloose, B.; Schmugge, M.; Mejstrikova, E.; Nováková, M.; et al. Bone marrow immunophenotyping by flow cytometry in refractory cytopenia of childhood. Haematologica 2015, 100, 315–323. [Google Scholar] [CrossRef]
- Chisholm, K.M.; Xu, M.; Davis, B.; Ogi, A.; Pacheco, M.C.; Geddis, A.E.; Tsuchiya, K.D.; Rutledge, J.C. Evaluation of the Utility of Bone Marrow Morphology and Ancillary Studies in Pediatric Patients Under Surveillance for Myelodysplastic Syndrome. Am. J. Clin. Pathol. 2018, 149, 499–513. [Google Scholar] [CrossRef]
- Haferlach, T.; Nagata, Y.; Grossmann, V.; Okuno, Y.; Bacher, U.; Nagae, G.; Schnittger, S.; Sanada, M.; Kon, A.; Alpermann, T.; et al. Landscape of genetic lesions in 944 patients with myelodysplastic syndromes. Leukemia 2014, 28, 241–247. [Google Scholar] [CrossRef]
- Greenberg, P.L.; Tuechler, H.; Schanz, J.; Sanz, G.; Garcia-Manero, G.; Solé, F.; Bennett, J.M.; Bowen, D.; Fenaux, P.; Dreyfus, F.; et al. Revised international prognostic scoring system for myelodysplastic syndromes. Blood 2012, 120, 2454–2465. [Google Scholar] [CrossRef] [PubMed]
- Hasle, H.; Baumann, I.; Bergsträsser, E.; Fenu, S.; Fischer, A.; Kardos, G.; Kerndrup, G.; Locatelli, F.; Rogge, T.; Schultz, K.R.; et al. The International Prognostic Scoring System (IPSS) for childhood myelodysplastic syndrome (MDS) and juvenile myelomonocytic leukemia (JMML). Leukemia 2004, 18, 2008–2014. [Google Scholar] [CrossRef]
- Schwartz, J.R.; Ma, J.; Lamprecht, T.; Walsh, M.; Wang, S.; Bryant, V.; Song, G.; Wu, G.; Easton, J.; Kesserwan, C.; et al. The genomic landscape of pediatric myelodysplastic syndromes. Nat. Commun. 2017, 8, 1557. [Google Scholar] [CrossRef]
- Veltroni, M.; Sainati, L.; Zecca, M.; Fenu, S.; Tridello, G.; Testi, A.M.; Merlone, A.D.; Buldini, B.; Leszl, A.; Lo Nigro, L.; et al. Advanced pediatric myelodysplastic syndromes: Can immunophenotypic characterization of blast cells be a diagnostic and prognostic tool? Pediatr. Blood Cancer 2009, 52, 357–363. [Google Scholar] [CrossRef]
- Locatelli, F.; Strahm, B. How I treat myelodysplastic syndromes of childhood. Blood 2018, 131, 1406–1414. [Google Scholar] [CrossRef]
- Boultwood, J.; Lewis, S.; Wainscoat, J. The 5q-syndrome. Blood 1994, 84, 3253–3260. [Google Scholar] [CrossRef]
- Antillón, F.; Raimondi, S.C.; Fairman, J.; Liang, H.; Nagarajan, L.; Head, D.; Ribeiro, R.C. 5q- in a child with refractory anemia with excess blasts: Similarities to 5q- syndrome in adults. Cancer Genet. Cytogenet. 1998, 105, 119–122. [Google Scholar] [CrossRef]
- Chantrain, C.; Vermylen, C.; Michaux, L.; Brichard, B.; Cornu, G. Clonal monosomy 7 and 5q--in a child with myelodysplastic syndrome. Pediatr. Hematol. Oncol. 2000, 17, 505–509. [Google Scholar] [CrossRef]
- Uyttebroeck, A.; Brock, P.; De Groote, B.; Renard, M.; Dal Cin, P.; Van den Berghe, H.; Casteels-Van Daele, M. 5q- syndrome in a child. Cancer Genet. Cytogenet. 1995, 80, 121–123. [Google Scholar] [CrossRef] [PubMed]
- Sugita, K.; Kaneko, T.; Sekine, Y.; Taguchi, N.; Miyauchi, J. Mast cell leukemia evolved from RAEB-T (5q-syndrome) in a 12 year-old girl. Rinsho Ketsueki 1996, 37, 430–436. [Google Scholar] [PubMed]
- Pitman, S.D.; Victorio, A.; Rowsell, E.; Morris, J.; Wang, J. 5q- syndrome in a child with slowly progressive pancytopenia: A case report and review of the literature. J. Pediatr. Hematol. Oncol. 2006, 28, 115–119. [Google Scholar] [CrossRef]
- Elkattawy, S.; Ayad, S.; El-Feki, I.; Guo, X.; Appiah-Kubi, E.; Talpur, A.; Kessler, W. 5q Deletion Myelodysplastic Syndrome in a Young Male Patient. Cureus 2021, 13, e17466. [Google Scholar] [CrossRef]
- Adams, R.H.; Lemons, R.S.; Thangavelu, M.; Le Beau, M.M.; Christensen, R.D. Interstitial deletion of chromosome 5, del(5q), in a newborn with Down syndrome and an unusual hematologic disorder. Am. J. Hematol. 1989, 31, 273–279. [Google Scholar] [CrossRef]
- Shikano, T.; Ishikawa, Y.; Anakura, M. Myelodysplastic syndrome with partial deletion of the long arm of chromosome 5: First report of a case in a child. Acta Paediatr. Jpn. 1992, 34, 539–542. [Google Scholar] [CrossRef]
- Van den Berghe, H.; Cassiman, J.J.; David, G.; Fryns, J.P.; Michaux, J.L.; Sokal, G. Distinct haematological disorder with deletion of long arm of no. 5 chromosome. Nature 1974, 251, 437–438. [Google Scholar] [CrossRef]
- Sokal, G.; Michaux, J.L.; Van Den Berghe, H.; Cordier, A.; Rodhain, J.; Ferrant, A.; Moriau, M.; De Bruyere, M.; Sonnet, J. A new hematologic syndrome with a distinct karyotype: The 5 q--chromosome. Blood 1975, 46, 519–533. [Google Scholar] [CrossRef]
- Adema, V.; Bejar, R. What lies beyond del(5q) in myelodysplastic syndrome? Haematologica 2013, 98, 1819–1821. [Google Scholar] [CrossRef] [PubMed]
- Gaballa, M.R.; Besa, E.C. Myelodysplastic syndromes with 5q deletion: Pathophysiology and role of lenalidomide. Ann. Hematol. 2014, 93, 723–733. [Google Scholar] [CrossRef] [PubMed]
- Hosono, N.; Makishima, H.; Mahfouz, R.; Przychodzen, B.; Yoshida, K.; Jerez, A.; LaFramboise, T.; Polprasert, C.; Clemente, M.J.; Shiraishi, Y.; et al. Recurrent genetic defects on chromosome 5q in myeloid neoplasms. Oncotarget 2017, 8, 6483–6495. [Google Scholar] [CrossRef] [PubMed]
- Mahmood, T.; Robinson, W.A.; Hamstra, R.D.; Wallner, S.F. Macrocytic anemia, thrombocytosis and nonlobulated megakaryocytes: The 5q-syndrome, a distinct entity. Am. J. Med. 1979, 66, 946–950. [Google Scholar] [CrossRef] [PubMed]
- Van den Berghe, H.; Vermaelen, K.; Mecucci, C.; Barbieri, D.; Tricot, G. The 5q-anomaly. Cancer Genet. Cytogenet. 1985, 17, 189–255. [Google Scholar] [CrossRef]
- List, A.; Dewald, G.; Bennett, J.; Giagounidis, A.; Raza, A.; Feldman, E.; Powell, B.; Greenberg, P.; Thomas, D.; Stone, R.; et al. Lenalidomide in the myelodysplastic syndrome with chromosome 5q deletion. N. Engl. J. Med. 2006, 355, 1456–1465. [Google Scholar] [CrossRef] [PubMed]
- Fenaux, P.; Giagounidis, A.; Selleslag, D.; Beyne-Rauzy, O.; Mufti, G.; Mittelman, M.; Muus, P.; Te Boekhorst, P.; Sanz, G.; Del Cañizo, C.; et al. A randomized phase 3 study of lenalidomide versus placebo in RBC transfusion-dependent patients with Low-/Intermediate-1-risk myelodysplastic syndromes with del5q. Blood 2011, 118, 3765–3776. [Google Scholar] [CrossRef]
- Raza, A.; Reeves, J.A.; Feldman, E.J.; Dewald, G.W.; Bennett, J.M.; Deeg, H.J.; Dreisbach, L.; Schiffer, C.A.; Stone, R.M.; Greenberg, P.L.; et al. Phase 2 study of lenalidomide in transfusion-dependent, low-risk, and intermediate-1 risk myelodysplastic syndromes with karyotypes other than deletion 5q. Blood 2008, 111, 86–93. [Google Scholar] [CrossRef]
- Adès, L.; Boehrer, S.; Prebet, T.; Beyne-Rauzy, O.; Legros, L.; Ravoet, C.; Dreyfus, F.; Stamatoullas, A.; Chaury, M.P.; Delaunay, J.; et al. Efficacy and safety of lenalidomide in intermediate-2 or high-risk myelodysplastic syndromes with 5q deletion: Results of a phase 2 study. Blood 2009, 113, 3947–3952. [Google Scholar] [CrossRef] [PubMed]
- Möllgård, L.; Saft, L.; Treppendahl, M.B.; Dybedal, I.; Nørgaard, J.M.; Astermark, J.; Ejerblad, E.; Garelius, H.; Dufva, I.H.; Jansson, M.; et al. Clinical effect of increasing doses of lenalidomide in high-risk myelodysplastic syndrome and acute myeloid leukemia with chromosome 5 abnormalities. Haematologica 2011, 96, 963–971. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Platzbecker, U.; Braulke, F.; Kündgen, A.; Götze, K.; Bug, G.; Schönefeldt, C.; Shirneshan, K.; Röllig, C.; Bornhäuser, M.; Naumann, R.; et al. Sequential combination of azacitidine and lenalidomide in del(5q) higher-risk myelodysplastic syndromes or acute myeloid leukemia: A phase I study. Leukemia 2013, 27, 1403–1407. [Google Scholar] [CrossRef]
- Scherman, E.; Malak, S.; Perot, C.; Gorin, N.C.; Rubio, M.T.; Isnard, F. Interest of the association azacitidine-lenalidomide as frontline therapy in high-risk myelodysplasia or acute myeloid leukemia with complex karyotype. Leukemia 2012, 26, 822–824. [Google Scholar] [CrossRef][Green Version]
- Sekeres, M.A.; Tiu, R.V.; Komrokji, R.; Lancet, J.; Advani, A.S.; Afable, M.; Englehaupt, R.; Juersivich, J.; Cuthbertson, D.; Paleveda, J.; et al. Phase 2 study of the lenalidomide and azacitidine combination in patients with higher-risk myelodysplastic syndromes. Blood 2012, 120, 4945–4951. [Google Scholar] [CrossRef][Green Version]
- Kantarjian, H.; O’Brien, S.; Ravandi, F.; Borthakur, G.; Faderl, S.; Bueso-Ramos, C.; Abruzzo, L.; Pierce, S.; Shan, J.; Issa, J.P.; et al. The heterogeneous prognosis of patients with myelodysplastic syndrome and chromosome 5 abnormalities: How does it relate to the original lenalidomide experience in MDS? Cancer 2009, 115, 5202–5209. [Google Scholar] [CrossRef]
- Jädersten, M.; Saft, L.; Pellagatti, A.; Göhring, G.; Wainscoat, J.S.; Boultwood, J.; Porwit, A.; Schlegelberger, B.; Hellström-Lindberg, E. Clonal heterogeneity in the 5q- syndrome: p53 expressing progenitors prevail during lenalidomide treatment and expand at disease progression. Haematologica 2009, 94, 1762–1766. [Google Scholar] [CrossRef]
- Hirabayashi, S.; Flotho, C.; Moetter, J.; Heuser, M.; Hasle, H.; Gruhn, B.; Klingebiel, T.; Thol, F.; Schlegelberger, B.; Baumann, I.; et al. Spliceosomal gene aberrations are rare, coexist with oncogenic mutations, and are unlikely to exert a driver effect in childhood MDS and JMML. Blood 2012, 119, e96–e99. [Google Scholar] [CrossRef][Green Version]
- Boles, B.; Shiel, M.; Gardner, J.A.; Conant, J.L. Pediatric Myelodysplastic Syndrome with SF3B1 Mutation. J. Assoc. Genet. Technol. 2023, 49, 69–72. [Google Scholar]
- Hosono, N. Genetic abnormalities and pathophysiology of MDS. Int. J. Clin. Oncol. 2019, 24, 885–892. [Google Scholar] [CrossRef]
- Malcovati, L.; Stevenson, K.; Papaemmanuil, E.; Neuberg, D.; Bejar, R.; Boultwood, J.; Bowen, D.T.; Campbell, P.J.; Ebert, B.L.; Fenaux, P.; et al. SF3B1-mutant MDS as a distinct disease subtype: A proposal from the International Working Group for the Prognosis of MDS. Blood 2020, 136, 157–170. [Google Scholar] [CrossRef]
- Yoshida, K.; Sanada, M.; Shiraishi, Y.; Nowak, D.; Nagata, Y.; Yamamoto, R.; Sato, Y.; Sato-Otsubo, A.; Kon, A.; Nagasaki, M.; et al. Frequent pathway mutations of splicing machinery in myelodysplasia. Nature 2011, 478, 64–69. [Google Scholar] [CrossRef]
- Papaemmanuil, E.; Cazzola, M.; Boultwood, J.; Malcovati, L.; Vyas, P.; Bowen, D.; Pellagatti, A.; Wainscoat, J.S.; Hellstrom-Lindberg, E.; Gambacorti-Passerini, C.; et al. Somatic SF3B1 mutation in myelodysplasia with ring sideroblasts. N. Engl. J. Med. 2011, 365, 1384–1395. [Google Scholar] [CrossRef] [PubMed]
- Malcovati, L.; Papaemmanuil, E.; Bowen, D.T.; Boultwood, J.; Della Porta, M.G.; Pascutto, C.; Travaglino, E.; Groves, M.J.; Godfrey, A.L.; Ambaglio, I.; et al. Clinical significance of SF3B1 mutations in myelodysplastic syndromes and myelodysplastic/myeloproliferative neoplasms. Blood 2011, 118, 6239–6246. [Google Scholar] [CrossRef] [PubMed]
- Visconte, V.; Makishima, H.; Maciejewski, J.P.; Tiu, R.V. Emerging roles of the spliceosomal machinery in myelodysplastic syndromes and other hematological disorders. Leukemia 2012, 26, 2447–2454. [Google Scholar] [CrossRef] [PubMed]
- Chan, O.; Ali, N.A.; Sallman, D.; Padron, E.; Lancet, J.; Komrokji, R. Therapeutic Outcomes and Prognostic Impact of Gene Mutations Including TP53 and SF3B1 in Patients with Del(5q) Myelodysplastic Syndromes (MDS). Clin. Lymphoma Myeloma Leuk. 2022, 22, e467–e476. [Google Scholar] [CrossRef] [PubMed]
- Choi, H.W.; Kim, H.R.; Baek, H.J.; Kook, H.; Cho, D.; Shin, J.H.; Suh, S.P.; Ryang, D.W.; Shin, M.G. Alteration of the SETBP1 gene and splicing pathway genes SF3B1, U2AF1, and SRSF2 in childhood acute myeloid leukemia. Ann. Lab. Med. 2015, 35, 118–122. [Google Scholar] [CrossRef] [PubMed][Green Version]
- van der Werf, I.; Wojtuszkiewicz, A.; Yao, H.; Sciarrillo, R.; Meggendorfer, M.; Hutter, S.; Walter, W.; Janssen, J.; Kern, W.; Haferlach, C.; et al. SF3B1 as therapeutic target in FLT3/ITD positive acute myeloid leukemia. Leukemia 2021, 35, 2698–2702. [Google Scholar] [CrossRef] [PubMed]
- Haase, D.; Stevenson, K.E.; Neuberg, D.; Maciejewski, J.P.; Nazha, A.; Sekeres, M.A.; Ebert, B.L.; Garcia-Manero, G.; Haferlach, C.; Haferlach, T.; et al. TP53 mutation status divides myelodysplastic syndromes with complex karyotypes into distinct prognostic subgroups. Leukemia 2019, 33, 1747–1758. [Google Scholar] [CrossRef] [PubMed]
- Bernard, E.; Nannya, Y.; Hasserjian, R.P.; Devlin, S.M.; Tuechler, H.; Medina-Martinez, J.S.; Yoshizato, T.; Shiozawa, Y.; Saiki, R.; Malcovati, L.; et al. Implications of TP53 allelic state for genome stability, clinical presentation and outcomes in myelodysplastic syndromes. Nat. Med. 2020, 26, 1549–1556. [Google Scholar] [CrossRef] [PubMed]
- Saito, S.; Matsuda, K.; Taira, C.; Sano, K.; Tanaka-Yanagisawa, M.; Yanagisawa, R.; Nakazawa, Y.; Sakashita, K.; Shiohara, M.; Koike, K. Genetic analysis of TP53 in childhood myelodysplastic syndrome and juvenile myelomonocytic leukemia. Leuk. Res. 2011, 35, 1578–1584. [Google Scholar] [CrossRef]
- Jekic, B.; Novakovic, I.; Lukovic, L.; Kuzmanovic, M.; Popovic, B.; Milasin, J.; Bunjevacki, G.; Damnjanovic, T.; Cvjeticanin, S.; Bunjevacki, V. Lack of TP53 and FMS gene mutations in children with myelodysplastic syndrome. Cancer Genet. Cytogenet. 2006, 166, 163–165. [Google Scholar] [CrossRef]
- Praulich, I.; Tauscher, M.; Göhring, G.; Glaser, S.; Hofmann, W.; Feurstein, S.; Flotho, C.; Lichter, P.; Niemeyer, C.M.; Schlegelberger, B.; et al. Clonal heterogeneity in childhood myelodysplastic syndromes--challenge for the detection of chromosomal imbalances by array-CGH. Genes Chromosomes Cancer 2010, 49, 885–900. [Google Scholar] [CrossRef]
- Newman, A.M.; Zaka, M.; Zhou, P.; Blain, A.E.; Erhorn, A.; Barnard, A.; Crossland, R.E.; Wilkinson, S.; Enshaei, A.; De Zordi, J.; et al. Genomic abnormalities of TP53 define distinct risk groups of paediatric B-cell non-Hodgkin lymphoma. Leukemia 2022, 36, 781–789. [Google Scholar] [CrossRef]
- Pugh, T.J.; Yu, W.; Yang, J.; Field, A.L.; Ambrogio, L.; Carter, S.L.; Cibulskis, K.; Giannikopoulos, P.; Kiezun, A.; Kim, J.; et al. Exome sequencing of pleuropulmonary blastoma reveals frequent biallelic loss of TP53 and two hits in DICER1 resulting in retention of 5p-derived miRNA hairpin loop sequences. Oncogene 2014, 33, 5295–5302. [Google Scholar] [CrossRef]
- Elgarten, C.W.; Aplenc, R. Pediatric acute myeloid leukemia: Updates on biology, risk stratification, and therapy. Curr. Opin. Pediatr. 2020, 32, 57–66. [Google Scholar] [CrossRef]
- Pommert, L.; Tarlock, K. The evolution of targeted therapy in pediatric AML: Gemtuzumab ozogamicin, FLT3/IDH/BCL2 inhibitors, and other therapies. Hematol. Am. Soc. Hematol. Educ. Program 2022, 2022, 603–610. [Google Scholar] [CrossRef]
- Dwivedi, A.; Fu, L.; Chien, C.D.; Pouzolles, M.; Shah, N.N.; Taylor, N. Engineering Off-the-Shelf Gamma Delta CAR T Cells for the Treatment of Acute Myeloid Leukemia. Blood 2023, 142, 4827. [Google Scholar] [CrossRef]
- Shah, N.; Azzi, J.; Cooper, B.; Deol, A.; DiiPersio, J.; Koura, D.; McClune, B.; Muffly, L.; Umair Mushtaq, M.; Narayan, R.; et al. Phase 1/2 Study of Donor-Derived Anti-CD33 Chimeric Antigen Receptor Expressing T Cells (VCAR33) in Patients with Relapsed or Refractory Acute Myeloid Leukemia after Allogeneic Hematopoietic Cell Transplantation. In Proceedings of the ASH Annual Meeting & Exposition, San Diego, CA, USA, 11 December 2023. [Google Scholar]
- Calviño, C.; Ceballos, C.; Alfonso, A.; Jauregui, P.; Calleja-Cervantes, M.E.; San Martin-Uriz, P.; Rodriguez-Marquez, P.; Martin-Mallo, A.; Iglesias, E.; Abizanda, G.; et al. Optimization of universal allogeneic CAR-T cells combining CRISPR and transposon-based technologies for treatment of acute myeloid leukemia. Front. Immunol. 2023, 14, 1270843. [Google Scholar] [CrossRef]
- Bhagwat, A.S.; Torres, L.; Shestova, O.; Shestov, M.; Frey, N.V.; Hexner, E.O.; Luger, S.M.; Loren, A.W.; Martin, M.E.; McCurdy, S.R.; et al. Cytokine Release Syndrome Results in Reduced AML Killing By CD123 CAR T Cells. Blood 2023, 142, 217. [Google Scholar] [CrossRef]
- Rohrbacher, L.; Nixdorf, D.; Stadler, H.; Brauchle, B.; Märkl, F.; Philipp, N.; Hänel, G.; Marcinek, A.; Kazerani, M.; Emhardt, A.J.; et al. Two Players, One Goal: BiTE ® Vs CART Targeting FLT3 in AML. Blood 2023, 142, 3444. [Google Scholar] [CrossRef]
- Le, Q.; Hadland, B.; Smith, J.L.; Leonti, A.; Huang, B.J.; Ries, R.; Hylkema, T.A.; Castro, S.; Tang, T.T.; McKay, C.N.; et al. CBFA2T3-GLIS2 model of pediatric acute megakaryoblastic leukemia identifies FOLR1 as a CAR T cell target. J. Clin. Investig. 2022, 132, e157101. [Google Scholar] [CrossRef] [PubMed]
- Kataoka, K.; Kurokawa, M. Ecotropic viral integration site 1, stem cell self-renewal and leukemogenesis. Cancer Sci. 2012, 103, 1371–1377. [Google Scholar] [CrossRef] [PubMed]
- Tang, Z.; Tang, G.; Hu, S.; Patel, K.P.; Yin, C.C.; Wang, W.; Lin, P.; Toruner, G.A.; Ok, C.Y.; Gu, J.; et al. Deciphering the complexities of MECOM rearrangement-driven chromosomal aberrations. Cancer Genet. 2019, 233–234, 21–31. [Google Scholar] [CrossRef] [PubMed]
- Lugthart, S.; Gröschel, S.; Beverloo, H.B.; Kayser, S.; Valk, P.J.; van Zelderen-Bhola, S.L.; Jan Ossenkoppele, G.; Vellenga, E.; van den Berg-de Ruiter, E.; Schanz, U.; et al. Clinical, molecular, and prognostic significance of WHO type inv(3)(q21q26.2)/t(3;3)(q21;q26.2) and various other 3q abnormalities in acute myeloid leukemia. J. Clin. Oncol. 2010, 28, 3890–3898. [Google Scholar] [CrossRef]
- Lugthart, S.; van Drunen, E.; van Norden, Y.; van Hoven, A.; Erpelinck, C.A.; Valk, P.J.; Beverloo, H.B.; Löwenberg, B.; Delwel, R. High EVI1 levels predict adverse outcome in acute myeloid leukemia: Prevalence of EVI1 overexpression and chromosome 3q26 abnormalities underestimated. Blood 2008, 111, 4329–4337. [Google Scholar] [CrossRef]
- Gröschel, S.; Lugthart, S.; Schlenk, R.F.; Valk, P.J.; Eiwen, K.; Goudswaard, C.; van Putten, W.J.; Kayser, S.; Verdonck, L.F.; Lübbert, M.; et al. High EVI1 expression predicts outcome in younger adult patients with acute myeloid leukemia and is associated with distinct cytogenetic abnormalities. J. Clin. Oncol. 2010, 28, 2101–2107. [Google Scholar] [CrossRef] [PubMed]
- Baldazzi, C.; Luatti, S.; Zuffa, E.; Papayannidis, C.; Ottaviani, E.; Marzocchi, G.; Ameli, G.; Bardi, M.A.; Bonaldi, L.; Paolini, R.; et al. Complex chromosomal rearrangements leading to MECOM overexpression are recurrent in myeloid malignancies with various 3q abnormalities. Genes Chromosomes Cancer 2016, 55, 375–388. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.; Konoplev, S.N.; Wang, X.; Cui, W.; Chen, S.S.; Medeiros, L.J.; Lin, P. De novo acute myeloid leukemia with inv(3)(q21q26.2) or t(3;3)(q21;q26.2): A clinicopathologic and cytogenetic study of an entity recently added to the WHO classification. Mod. Pathol. 2011, 24, 384–389. [Google Scholar] [CrossRef] [PubMed]
- Grimwade, D.; Hills, R.K.; Moorman, A.V.; Walker, H.; Chatters, S.; Goldstone, A.H.; Wheatley, K.; Harrison, C.J.; Burnett, A.K. Refinement of cytogenetic classification in acute myeloid leukemia: Determination of prognostic significance of rare recurring chromosomal abnormalities among 5876 younger adult patients treated in the United Kingdom Medical Research Council trials. Blood 2010, 116, 354–365. [Google Scholar] [CrossRef]
- De Braekeleer, M.; Le Bris, M.J.; De Braekeleer, E.; Basinko, A.; Morel, F.; Douet-Guilbert, N. 3q26/EVI1 rearrangements in myeloid hemopathies: A cytogenetic review. Future Oncol. 2015, 11, 1675–1686. [Google Scholar] [CrossRef] [PubMed]
- Haas, K.; Kundi, M.; Sperr, W.R.; Esterbauer, H.; Ludwig, W.D.; Ratei, R.; Koller, E.; Gruener, H.; Sauerland, C.; Fonatsch, C.; et al. Expression and prognostic significance of different mRNA 5’-end variants of the oncogene EVI1 in 266 patients with de novo AML: EVI1 and MDS1/EVI1 overexpression both predict short remission duration. Genes Chromosomes Cancer 2008, 47, 288–298. [Google Scholar] [CrossRef]
- Balgobind, B.V.; Lugthart, S.; Hollink, I.H.; Arentsen-Peters, S.T.; van Wering, E.R.; de Graaf, S.S.; Reinhardt, D.; Creutzig, U.; Kaspers, G.J.; de Bont, E.S.; et al. EVI1 overexpression in distinct subtypes of pediatric acute myeloid leukemia. Leukemia 2010, 24, 942–949. [Google Scholar] [CrossRef]
- Ho, P.A.; Alonzo, T.A.; Gerbing, R.B.; Pollard, J.A.; Hirsch, B.; Raimondi, S.C.; Cooper, T.; Gamis, A.S.; Meshinchi, S. High EVI1 expression is associated with MLL rearrangements and predicts decreased survival in paediatric acute myeloid leukaemia: A report from the children’s oncology group. Br. J. Haematol. 2013, 162, 670–677. [Google Scholar] [CrossRef]
- Duan, J.X.; Liu, F.; Chang, L.; Che, G.L.; Yang, Q.X.; Teng, J.; Jian, H.; Liu, X.J.; Lai, S.Y. A primary pediatric acute myelomonocytic leukemia with t(3;21)(q26;q22): A case report. Medicine 2023, 102, e35721. [Google Scholar] [CrossRef]
- Johansson, B.; Fioretos, T.; Garwicz, S.; Heim, S.; Mitelman, F. t(3;21)(q26;q22) with AML1 rearrangement in a de novo childhood acute monoblastic leukaemia. Br. J. Haematol. 1996, 92, 429–431. [Google Scholar] [CrossRef]
- Li, S.; Yin, C.C.; Medeiros, L.J.; Bueso-Ramos, C.; Lu, G.; Lin, P. Myelodysplastic syndrome/acute myeloid leukemia with t(3;21)(q26.2;q22) is commonly a therapy-related disease associated with poor outcome. Am. J. Clin. Pathol. 2012, 138, 146–152. [Google Scholar] [CrossRef]
- Chisholm, K.M.; Smith, J.; Heerema-McKenney, A.E.; Choi, J.K.; Ries, R.E.; Hirsch, B.A.; Raimondi, S.C.; Wang, Y.C.; Dang, A.; Alonzo, T.A.; et al. Pathologic, cytogenetic, and molecular features of acute myeloid leukemia with megakaryocytic differentiation: A report from the Children’s Oncology Group. Pediatr. Blood Cancer 2023, 70, e30251. [Google Scholar] [CrossRef]
- Connelly, J.A.; Mody, R.J.; Wu, Y.M.; Robinson, D.R.; Lonigro, R.J.; Vats, P.; Rabban, E.; Anderson, B.; Walkovich, K. Identification of novel MECOM gene fusion and personalized therapeutic targets through integrative clinical sequencing in secondary acute myeloid leukemia in a patient with severe congenital neutropenia: A case report and literature review. Cold Spring Harb. Mol. Case Stud. 2018, 4, a002204. [Google Scholar] [CrossRef]
- Elsherif, M.; Hammad, M.; Hafez, H.; Yassin, D.; Ashraf, M.; Yasser, N.; Lehmann, L.; Elhaddad, A. MECOM gene overexpression in pediatric patients with acute myeloid leukemia. Acta Oncol. 2022, 61, 516–522. [Google Scholar] [CrossRef] [PubMed]
- Jo, A.; Mitani, S.; Shiba, N.; Hayashi, Y.; Hara, Y.; Takahashi, H.; Tsukimoto, I.; Tawa, A.; Horibe, K.; Tomizawa, D.; et al. High expression of EVI1 and MEL1 is a compelling poor prognostic marker of pediatric AML. Leukemia 2015, 29, 1076–1083. [Google Scholar] [CrossRef] [PubMed]
- Ammeti, D.; Marzollo, A.; Gabelli, M.; Zanchetta, M.E.; Tretti-Parenzan, C.; Bottega, R.; Capaci, V.; Biffi, A.; Savoia, A.; Bresolin, S.; et al. A novel mutation in MECOM affects MPL regulation in vitro and results in thrombocytopenia and bone marrow failure. Br. J. Haematol. 2023, 203, 852–859. [Google Scholar] [CrossRef]
- Germeshausen, M.; Ancliff, P.; Estrada, J.; Metzler, M.; Ponstingl, E.; Rütschle, H.; Schwabe, D.; Scott, R.H.; Unal, S.; Wawer, A.; et al. MECOM-associated syndrome: A heterogeneous inherited bone marrow failure syndrome with amegakaryocytic thrombocytopenia. Blood Adv. 2018, 2, 586–596. [Google Scholar] [CrossRef] [PubMed]
- Irie, M.; Niihori, T.; Nakano, T.; Suzuki, T.; Katayama, S.; Moriya, K.; Niizuma, H.; Suzuki, N.; Saito-Nanjo, Y.; Onuma, M.; et al. Reduced-intensity conditioning is effective for allogeneic hematopoietic stem cell transplantation in infants with MECOM-associated syndrome. Int. J. Hematol. 2023, 117, 598–606. [Google Scholar] [CrossRef]
- Ripperger, T.; Hofmann, W.; Koch, J.C.; Shirneshan, K.; Haase, D.; Wulf, G.; Issing, P.R.; Karnebogen, M.; Schmidt, G.; Auber, B.; et al. MDS1 and EVI1 complex locus (MECOM): A novel candidate gene for hereditary hematological malignancies. Haematologica 2018, 103, e55–e58. [Google Scholar] [CrossRef]
- Dreveny, I.; Deeves, S.E.; Fulton, J.; Yue, B.; Messmer, M.; Bhattacharya, A.; Collins, H.M.; Heery, D.M. The double PHD finger domain of MOZ/MYST3 induces α-helical structure of the histone H3 tail to facilitate acetylation and methylation sampling and modification. Nucleic Acids Res. 2014, 42, 822–835. [Google Scholar] [CrossRef]
- Perez-Campo, F.M.; Costa, G.; Lie-a-Ling, M.; Kouskoff, V.; Lacaud, G. The MYSTerious MOZ, a histone acetyltransferase with a key role in haematopoiesis. Immunology 2013, 139, 161–165. [Google Scholar] [CrossRef]
- Borrow, J.; Stanton, V.P., Jr.; Andresen, J.M.; Becher, R.; Behm, F.G.; Chaganti, R.S.; Civin, C.I.; Disteche, C.; Dubé, I.; Frischauf, A.M.; et al. The translocation t(8;16)(p11;p13) of acute myeloid leukaemia fuses a putative acetyltransferase to the CREB-binding protein. Nat. Genet. 1996, 14, 33–41. [Google Scholar] [CrossRef] [PubMed]
- Troke, P.J.; Kindle, K.B.; Collins, H.M.; Heery, D.M. MOZ fusion proteins in acute myeloid leukaemia. Biochem. Soc. Symp. 2006, 73, 23–39. [Google Scholar] [CrossRef]
- Lamble, A.J.; Gerbing, R.B.; Smith, J.L.; Ries, R.E.; Kolb, E.A.; Alonzo, T.A.; Meshinchi, S. Crebbp Alterations Are Associated with a Poor Prognosis in De Novo AML. Blood 2021, 138, 3451. [Google Scholar] [CrossRef]
- Coenen, E.A.; Zwaan, C.M.; Reinhardt, D.; Harrison, C.J.; Haas, O.A.; de Haas, V.; Mihál, V.; De Moerloose, B.; Jeison, M.; Rubnitz, J.E.; et al. Pediatric acute myeloid leukemia with t(8;16)(p11;p13), a distinct clinical and biological entity: A collaborative study by the International-Berlin-Frankfurt-Münster AML-study group. Blood 2013, 122, 2704–2713. [Google Scholar] [CrossRef] [PubMed]
- Baell, J.B.; Leaver, D.J.; Hermans, S.J.; Kelly, G.L.; Brennan, M.S.; Downer, N.L.; Nguyen, N.; Wichmann, J.; McRae, H.M.; Yang, Y.; et al. Inhibitors of histone acetyltransferases KAT6A/B induce senescence and arrest tumour growth. Nature 2018, 560, 253–257. [Google Scholar] [CrossRef] [PubMed]
- Rowley, J.D.; Potter, D. Chromosomal banding patterns in acute nonlymphocytic leukemia. Blood 1976, 47, 705–721. [Google Scholar] [CrossRef] [PubMed]
- Ageberg, M.; Drott, K.; Olofsson, T.; Gullberg, U.; Lindmark, A. Identification of a novel and myeloid specific role of the leukemia-associated fusion protein DEK-NUP214 leading to increased protein synthesis. Genes Chromosomes Cancer 2008, 47, 276–287. [Google Scholar] [CrossRef]
- Gupta, M.; Ashok Kumar, J.; Sitaram, U.; Neeraj, S.; Nancy, A.; Balasubramanian, P.; Abraham, A.; Mathews, V.; Viswabandya, A.; George, B.; et al. The t(6;9)(p22;q34) in myeloid neoplasms: A retrospective study of 16 cases. Cancer Genet. Cytogenet. 2010, 203, 297–302. [Google Scholar] [CrossRef]
- Visconte, V.; Shetty, S.; Przychodzen, B.; Hirsch, C.; Bodo, J.; Maciejewski, J.P.; Hsi, E.D.; Rogers, H.J. Clinicopathologic and molecular characterization of myeloid neoplasms with isolated t(6;9)(p23;q34). Int. J. Lab. Hematol. 2017, 39, 409–417. [Google Scholar] [CrossRef]
- Cuneo, A.; Kerim, S.; Vandenberghe, E.; Van Orshoven, A.; Rodhain, J.; Bosly, A.; Zachee, P.; Louwagie, A.; Michaux, J.L.; Dal Cin, P.; et al. Translocation t(6;9) occurring in acute myelofibrosis, myelodysplastic syndrome, and acute nonlymphocytic leukemia suggests multipotent stem cell involvement. Cancer Genet. Cytogenet. 1989, 42, 209–219. [Google Scholar] [CrossRef] [PubMed]
- Soekarman, D.; von Lindern, M.; Daenen, S.; de Jong, B.; Fonatsch, C.; Heinze, B.; Bartram, C.; Hagemeijer, A.; Grosveld, G. The translocation (6;9) (p23;q34) shows consistent rearrangement of two genes and defines a myeloproliferative disorder with specific clinical features. Blood 1992, 79, 2990–2997. [Google Scholar] [CrossRef] [PubMed]
- Fang, H.; Yabe, M.; Zhang, X.; Kim, Y.; Wu, X.; Wei, P.; Chi, S.; Zheng, L.; Garcia-Manero, G.; Shao, L.; et al. Myelodysplastic syndrome with t(6;9)(p22;q34.1)/DEK-NUP214 better classified as acute myeloid leukemia? A multicenter study of 107 cases. Mod. Pathol. 2021, 34, 1143–1152. [Google Scholar] [CrossRef]
- Sandahl, J.D.; Coenen, E.A.; Forestier, E.; Harbott, J.; Johansson, B.; Kerndrup, G.; Adachi, S.; Auvrignon, A.; Beverloo, H.B.; Cayuela, J.M.; et al. t(6;9)(p22;q34)/DEK-NUP214-rearranged pediatric myeloid leukemia: An international study of 62 patients. Haematologica 2014, 99, 865–872. [Google Scholar] [CrossRef] [PubMed]
- Tarlock, K.; Alonzo, T.A.; Moraleda, P.P.; Gerbing, R.B.; Raimondi, S.C.; Hirsch, B.A.; Ravindranath, Y.; Lange, B.; Woods, W.G.; Gamis, A.S.; et al. Acute myeloid leukaemia (AML) with t(6;9)(p23;q34) is associated with poor outcome in childhood AML regardless of FLT3-ITD status: A report from the Children’s Oncology Group. Br. J. Haematol. 2014, 166, 254–259. [Google Scholar] [CrossRef] [PubMed]
- Meshinchi, S.; Woods, W.G.; Stirewalt, D.L.; Sweetser, D.A.; Buckley, J.D.; Tjoa, T.K.; Bernstein, I.D.; Radich, J.P. Prevalence and prognostic significance of Flt3 internal tandem duplication in pediatric acute myeloid leukemia. Blood 2001, 97, 89–94. [Google Scholar] [CrossRef]
- Thiede, C.; Steudel, C.; Mohr, B.; Schaich, M.; Schäkel, U.; Platzbecker, U.; Wermke, M.; Bornhäuser, M.; Ritter, M.; Neubauer, A.; et al. Analysis of FLT3-activating mutations in 979 patients with acute myelogenous leukemia: Association with FAB subtypes and identification of subgroups with poor prognosis. Blood 2002, 99, 4326–4335. [Google Scholar] [CrossRef]
- Bolouri, H.; Farrar, J.E.; Triche, T., Jr.; Ries, R.E.; Lim, E.L.; Alonzo, T.A.; Ma, Y.; Moore, R.; Mungall, A.J.; Marra, M.A.; et al. The molecular landscape of pediatric acute myeloid leukemia reveals recurrent structural alterations and age-specific mutational interactions. Nat. Med. 2018, 24, 103–112. [Google Scholar] [CrossRef]
- Shih, L.Y.; Kuo, M.C.; Liang, D.C.; Huang, C.F.; Lin, T.L.; Wu, J.H.; Wang, P.N.; Dunn, P.; Lai, C.L. Internal tandem duplication and Asp835 mutations of the FMS-like tyrosine kinase 3 (FLT3) gene in acute promyelocytic leukemia. Cancer 2003, 98, 1206–1216. [Google Scholar] [CrossRef]
- Au, W.Y.; Fung, A.; Chim, C.S.; Lie, A.K.; Liang, R.; Ma, E.S.; Chan, C.H.; Wong, K.F.; Kwong, Y.L. FLT-3 aberrations in acute promyelocytic leukaemia: Clinicopathological associations and prognostic impact. Br. J. Haematol. 2004, 125, 463–469. [Google Scholar] [CrossRef]
- Gale, R.E.; Hills, R.; Pizzey, A.R.; Kottaridis, P.D.; Swirsky, D.; Gilkes, A.F.; Nugent, E.; Mills, K.I.; Wheatley, K.; Solomon, E.; et al. Relationship between FLT3 mutation status, biologic characteristics, and response to targeted therapy in acute promyelocytic leukemia. Blood 2005, 106, 3768–3776. [Google Scholar] [CrossRef]
- Kutny, M.A.; Moser, B.K.; Laumann, K.; Feusner, J.H.; Gamis, A.; Gregory, J.; Larson, R.A.; Powell, B.L.; Stock, W.; Willman, C.L.; et al. FLT3 mutation status is a predictor of early death in pediatric acute promyelocytic leukemia: A report from the Children’s Oncology Group. Pediatr. Blood Cancer 2012, 59, 662–667. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, Y.; Kiyoi, H.; Nakano, Y.; Suzuki, R.; Kodera, Y.; Miyawaki, S.; Asou, N.; Kuriyama, K.; Yagasaki, F.; Shimazaki, C.; et al. Activating mutation of D835 within the activation loop of FLT3 in human hematologic malignancies. Blood 2001, 97, 2434–2439. [Google Scholar] [CrossRef] [PubMed]
- Mizuki, M.; Fenski, R.; Halfter, H.; Matsumura, I.; Schmidt, R.; Müller, C.; Grüning, W.; Kratz-Albers, K.; Serve, S.; Steur, C.; et al. Flt3 mutations from patients with acute myeloid leukemia induce transformation of 32D cells mediated by the Ras and STAT5 pathways. Blood 2000, 96, 3907–3914. [Google Scholar] [CrossRef]
- Meshinchi, S.; Alonzo, T.A.; Stirewalt, D.L.; Zwaan, M.; Zimmerman, M.; Reinhardt, D.; Kaspers, G.J.; Heerema, N.A.; Gerbing, R.; Lange, B.J.; et al. Clinical implications of FLT3 mutations in pediatric AML. Blood 2006, 108, 3654–3661. [Google Scholar] [CrossRef] [PubMed]
- Pollard, J.A.; Alonzo, T.A.; Gerbing, R.; Brown, P.; Fox, E.; Choi, J.; Fisher, B.; Hirsch, B.; Kahwash, S.; Getz, K.; et al. Sorafenib in Combination With Standard Chemotherapy for Children With High Allelic Ratio FLT3/ITD+ Acute Myeloid Leukemia: A Report From the Children’s Oncology Group Protocol AAML1031. J. Clin. Oncol. 2022, 40, 2023–2035. [Google Scholar] [CrossRef]
- Tarlock, K.; Chang, B.; Cooper, T.; Gross, T.; Gupta, S.; Neudorf, S.; Adlard, K.; Ho, P.A.; McGoldrick, S.; Watt, T.; et al. Sorafenib treatment following hematopoietic stem cell transplant in pediatric FLT3/ITD acute myeloid leukemia. Pediatr. Blood Cancer 2015, 62, 1048–1054. [Google Scholar] [CrossRef]
- Stone, R.M.; Mandrekar, S.J.; Sanford, B.L.; Laumann, K.; Geyer, S.; Bloomfield, C.D.; Thiede, C.; Prior, T.W.; Döhner, K.; Marcucci, G.; et al. Midostaurin plus Chemotherapy for Acute Myeloid Leukemia with a FLT3 Mutation. N. Engl. J. Med. 2017, 377, 454–464. [Google Scholar] [CrossRef] [PubMed]
- Perl, A.E.; Martinelli, G.; Cortes, J.E.; Neubauer, A.; Berman, E.; Paolini, S.; Montesinos, P.; Baer, M.R.; Larson, R.A.; Ustun, C.; et al. Gilteritinib or Chemotherapy for Relapsed or Refractory FLT3-Mutated AML. N. Engl. J. Med. 2019, 381, 1728–1740. [Google Scholar] [CrossRef]
- Voso, M.T.; Larson, R.A.; Jones, D.; Marcucci, G.; Prior, T.; Krauter, J.; Heuser, M.; Lavorgna, S.; Nomdedeu, J.; Geyer, S.M.; et al. Midostaurin in patients with acute myeloid leukemia and FLT3-TKD mutations: A subanalysis from the RATIFY trial. Blood Adv. 2020, 4, 4945–4954. [Google Scholar] [CrossRef]
- Gruber, T.A.; Downing, J.R. The biology of pediatric acute megakaryoblastic leukemia. Blood 2015, 126, 943–949. [Google Scholar] [CrossRef]
- Inaba, H.; Zhou, Y.; Abla, O.; Adachi, S.; Auvrignon, A.; Beverloo, H.B.; de Bont, E.; Chang, T.-T.; Creutzig, U.; Dworzak, M.; et al. Heterogeneous cytogenetic subgroups and outcomes in childhood acute megakaryoblastic leukemia: A retrospective international study. Blood 2015, 126, 1575–1584. [Google Scholar] [CrossRef]
- de Rooij, J.D.E.; Masetti, R.; van den Heuvel-Eibrink, M.M.; Cayuela, J.-M.; Trka, J.; Reinhardt, D.; Rasche, M.; Sonneveld, E.; Alonzo, T.A.; Fornerod, M.; et al. Recurrent abnormalities can be used for risk group stratification in pediatric AMKL: A retrospective intergroup study. Blood 2016, 127, 3424–3430. [Google Scholar] [CrossRef]
- Hara, Y.; Shiba, N.; Ohki, K.; Tabuchi, K.; Yamato, G.; Park, M.-j.; Tomizawa, D.; Kinoshita, A.; Shimada, A.; Arakawa, H.; et al. Prognostic impact of specific molecular profiles in pediatric acute megakaryoblastic leukemia in non-Down syndrome. Genes Chromosomes Cancer 2017, 56, 394–404. [Google Scholar] [CrossRef]
- Langenberg-Ververgaert, K.; Renzi, S.; Fuligni, F.; Davidson, S.; Abdelhaleem, M.; Lo, W.; Malkin, D.; Shlien, A.; Shago, M.; Villani, A.; et al. TERT promotor variant associated with poor clinical outcome in a patient with novel RBM15-MKL1 fusion-positive pediatric acute megakaryoblastic leukemia. Pediatr. Blood Cancer 2021, 68, e28542. [Google Scholar] [CrossRef] [PubMed]
- Messiaen, J.; Uyttebroeck, A.; Michaux, L.; Vandenberghe, P.; Boeckx, N.; Jacobs, S.A. t(1;7;22)(p13;q21;q13) is a novel 3-way variant of t(1;22)(p13;q13) neonatal acute megakaryoblastic leukemia: A case report. Mol. Clin. Oncol. 2023, 18, 18. [Google Scholar] [CrossRef]
- Torres, L.; Lisboa, S.; Vieira, J.; Cerveira, N.; Santos, J.; Pinheiro, M.; Correia, C.; Bizarro, S.; Almeida, M.; Teixeira, M.R. Acute megakaryoblastic leukemia with a four-way variant translocation originating the RBM15-MKL1 fusion gene. Pediatr. Blood Cancer 2011, 56, 846–849. [Google Scholar] [CrossRef]
- de Rooij, J.D.; Branstetter, C.; Ma, J.; Li, Y.; Walsh, M.P.; Cheng, J.; Obulkasim, A.; Dang, J.; Easton, J.; Verboon, L.J.; et al. Pediatric non-Down syndrome acute megakaryoblastic leukemia is characterized by distinct genomic subsets with varying outcomes. Nat. Genet. 2017, 49, 451–456. [Google Scholar] [CrossRef] [PubMed]
- Bernstein, J.; Dastugue, N.; Haas, O.A.; Harbott, J.; Heerema, N.A.; Huret, J.L.; Landman-Parker, J.; LeBeau, M.M.; Leonard, C.; Mann, G.; et al. Nineteen cases of the t(1;22)(p13;q13) acute megakaryblastic leukaemia of infants/children and a review of 39 cases: Report from a t(1;22) study group. Leukemia 2000, 14, 216–218. [Google Scholar] [CrossRef]
- Schweitzer, J.; Zimmermann, M.; Rasche, M.; von Neuhoff, C.; Creutzig, U.; Dworzak, M.; Reinhardt, D.; Klusmann, J.H. Improved outcome of pediatric patients with acute megakaryoblastic leukemia in the AML-BFM 04 trial. Ann. Hematol. 2015, 94, 1327–1336. [Google Scholar] [CrossRef] [PubMed]
- Gruber, T.A.; Larson Gedman, A.; Zhang, J.; Koss, C.S.; Marada, S.; Ta, H.Q.; Chen, S.C.; Su, X.; Ogden, S.K.; Dang, J.; et al. An Inv(16)(p13.3q24.3)-encoded CBFA2T3-GLIS2 fusion protein defines an aggressive subtype of pediatric acute megakaryoblastic leukemia. Cancer Cell 2012, 22, 683–697. [Google Scholar] [CrossRef]
- Masetti, R.; Pigazzi, M.; Togni, M.; Astolfi, A.; Indio, V.; Manara, E.; Casadio, R.; Pession, A.; Basso, G.; Locatelli, F. CBFA2T3-GLIS2 fusion transcript is a novel common feature in pediatric, cytogenetically normal AML, not restricted to FAB M7 subtype. Blood 2013, 121, 3469–3472. [Google Scholar] [CrossRef]
- Masetti, R.; Bertuccio, S.N.; Pession, A.; Locatelli, F. CBFA2T3-GLIS2-positive acute myeloid leukaemia. A peculiar paediatric entity. Br. J. Haematol. 2019, 184, 337–347. [Google Scholar] [CrossRef]
- Eidenschink Brodersen, L.; Alonzo, T.A.; Menssen, A.J.; Gerbing, R.B.; Pardo, L.; Voigt, A.P.; Kahwash, S.B.; Hirsch, B.; Raimondi, S.; Gamis, A.S.; et al. A recurrent immunophenotype at diagnosis independently identifies high-risk pediatric acute myeloid leukemia: A report from Children’s Oncology Group. Leukemia 2016, 30, 2077–2080. [Google Scholar] [CrossRef]
- Smith, J.L.; Ries, R.E.; Hylkema, T.; Alonzo, T.A.; Gerbing, R.B.; Santaguida, M.T.; Eidenschink Brodersen, L.; Pardo, L.; Cummings, C.L.; Loeb, K.R.; et al. Comprehensive Transcriptome Profiling of Cryptic CBFA2T3-GLIS2 Fusion-Positive AML Defines Novel Therapeutic Options: A COG and TARGET Pediatric AML Study. Clin. Cancer Res. 2020, 26, 726–737. [Google Scholar] [CrossRef]
- Noort, S.; Zimmermann, M.; Reinhardt, D.; Cuccuini, W.; Pigazzi, M.; Smith, J.; Ries, R.E.; Alonzo, T.A.; Hirsch, B.; Tomizawa, D.; et al. Prognostic impact of t(16;21)(p11;q22) and t(16;21)(q24;q22) in pediatric AML: A retrospective study by the I-BFM Study Group. Blood 2018, 132, 1584–1592. [Google Scholar] [CrossRef] [PubMed]
- Shiba, N.; Yoshida, K.; Hara, Y.; Yamato, G.; Shiraishi, Y.; Matsuo, H.; Okuno, Y.; Chiba, K.; Tanaka, H.; Kaburagi, T.; et al. Transcriptome analysis offers a comprehensive illustration of the genetic background of pediatric acute myeloid leukemia. Blood Adv. 2019, 3, 3157–3169. [Google Scholar] [CrossRef]
- Park, I.J.; Park, J.E.; Kim, H.J.; Jung, H.J.; Lee, W.G.; Cho, S.R. Acute myeloid leukemia with t(16;21)(q24;q22) and eosinophilia: Case report and review of the literature. Cancer Genet. Cytogenet. 2010, 196, 105–108. [Google Scholar] [CrossRef]
- Kawashima, N.; Shimada, A.; Taketani, T.; Hayashi, Y.; Yoshida, N.; Matsumoto, K.; Takahashi, Y.; Kojima, S.; Kato, K. Childhood acute myeloid leukemia with bone marrow eosinophilia caused by t(16;21)(q24;q22). Int. J. Hematol. 2012, 95, 577–580. [Google Scholar] [CrossRef] [PubMed]
- Kondoh, K.; Nakata, Y.; Furuta, T.; Hosoda, F.; Gamou, T.; Kurosawa, Y.; Kinoshita, A.; Ohki, M.; Tomita, Y.; Mori, T. A pediatric case of secondary leukemia associated with t(16;21)(q24;q22) exhibiting the chimeric AML1-MTG16 gene. Leuk. Lymphoma 2002, 43, 415–420. [Google Scholar] [CrossRef] [PubMed]
- Jeandidier, E.; Dastugue, N.; Mugneret, F.; Lafage-Pochitaloff, M.; Mozziconacci, M.J.; Herens, C.; Michaux, L.; Verellen-Dumoulin, C.; Talmant, P.; Cornillet-Lefebvre, P.; et al. Abnormalities of the long arm of chromosome 21 in 107 patients with hematopoietic disorders: A collaborative retrospective study of the Groupe Français de Cytogénétique Hématologique. Cancer Genet. Cytogenet. 2006, 166, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Raimondi, S.C.; Kalwinsky, D.K.; Hayashi, Y.; Behm, F.G.; Mirro, J., Jr.; Williams, D.L. Cytogenetics of childhood acute nonlymphocytic leukemia. Cancer Genet. Cytogenet. 1989, 40, 13–27. [Google Scholar] [CrossRef] [PubMed]
- Salomon-Nguyen, F.; Busson-Le Coniat, M.; Lafage Pochitaloff, M.; Mozziconacci, J.; Berger, R.; Bernard, O.A. AML1-MTG16 fusion gene in therapy-related acute leukemia with t(16;21)(q24;q22): Two new cases. Leukemia 2000, 14, 1704–1705. [Google Scholar] [CrossRef]
- Zatkova, A.; Fonatsch, C.; Sperr, W.R.; Valent, P. A patient with de novo AML M1 and t(16;21) with karyotype evolution. Leuk. Res. 2007, 31, 1319–1321. [Google Scholar] [CrossRef] [PubMed]
- Bennett, J.M.; Begg, C.B. Eastern Cooperative Oncology Group study of the cytochemistry of adult acute myeloid leukemia by correlation of subtypes with response and survival. Cancer Res. 1981, 41, 4833–4837. [Google Scholar] [PubMed]
- Barnard, D.R.; Alonzo, T.A.; Gerbing, R.B.; Lange, B.; Woods, W.G. Comparison of childhood myelodysplastic syndrome, AML FAB M6 or M7, CCG 2891: Report from the Children’s Oncology Group. Pediatr Blood Cancer 2007, 49, 17–22. [Google Scholar] [CrossRef] [PubMed]
- Chisholm, K.M.; Heerema-McKenney, A.E.; Choi, J.K.; Smith, J.; Ries, R.E.; Hirsch, B.A.; Raimondi, S.C.; Alonzo, T.A.; Wang, Y.C.; Aplenc, R.; et al. Acute erythroid leukemia is enriched in NUP98 fusions: A report from the Children’s Oncology Group. Blood Adv. 2020, 4, 6000–6008. [Google Scholar] [CrossRef] [PubMed]
- Reichard, K.K.; Tefferi, A.; Abdelmagid, M.; Orazi, A.; Alexandres, C.; Haack, J.; Greipp, P.T. Pure (acute) erythroid leukemia: Morphology, immunophenotype, cytogenetics, mutations, treatment details, and survival data among 41 Mayo Clinic cases. Blood Cancer J. 2022, 12, 147. [Google Scholar] [CrossRef]
- Iacobucci, I.; Wen, J.; Meggendorfer, M.; Choi, J.K.; Shi, L.; Pounds, S.B.; Carmichael, C.L.; Masih, K.E.; Morris, S.M.; Lindsley, R.C.; et al. Genomic subtyping and therapeutic targeting of acute erythroleukemia. Nat. Genet. 2019, 51, 694–704. [Google Scholar] [CrossRef]
- Ping, N.; Sun, A.; Song, Y.; Wang, Q.; Yin, J.; Cheng, W.; Xu, Y.; Wen, L.; Yao, H.; Ma, L.; et al. Exome sequencing identifies highly recurrent somatic GATA2 and CEBPA mutations in acute erythroid leukemia. Leukemia 2017, 31, 195–202. [Google Scholar] [CrossRef]
- Saft, L.; Kvasnicka, H.M.; Boudova, L.; Gianelli, U.; Lazzi, S.; Rozman, M. Myeloid/lymphoid neoplasms with eosinophilia and tyrosine kinase fusion genes: A workshop report with focus on novel entities and a literature review including paediatric cases. Histopathology 2023, 83, 829–849. [Google Scholar] [CrossRef]
- Clark, A.; Thomas, S.; Hamblin, A.; Talley, P.; Kulasekararaj, A.; Grinfeld, J.; Speight, B.; Snape, K.; McVeigh, T.P.; Snowden, J.A. Management of patients with germline predisposition to haematological malignancies considered for allogeneic blood and marrow transplantation: Best practice consensus guidelines from the UK Cancer Genetics Group (UKCGG), CanGene-CanVar, NHS England Genomic Laboratory Hub (GLH) Haematological Malignancies Working Group and the British Society of Blood and Marrow Transplantation and cellular therapy (BSBMTCT). Br. J. Haematol. 2023, 201, 35–44. [Google Scholar] [CrossRef] [PubMed]
- Gilad, O.; Dgany, O.; Noy-Lotan, S.; Krasnov, T.; Yacobovich, J.; Rabinowicz, R.; Goldberg, T.; Kuperman, A.A.; Abu-Quider, A.; Miskin, H.; et al. Syndromes predisposing to leukemia are a major cause of inherited cytopenias in children. Haematologica 2022, 107, 2081–2095. [Google Scholar] [CrossRef]
- Mendoza, H.; Podoltsev, N.A.; Siddon, A.J. Laboratory evaluation and prognostication among adults and children with CEBPA-mutant acute myeloid leukemia. Int. J. Lab. Hematol. 2021, 43 (Suppl. S1), 86–95. [Google Scholar] [CrossRef]
- Owen, C.; Barnett, M.; Fitzgibbon, J. Familial myelodysplasia and acute myeloid leukaemia—A review. Br. J. Haematol. 2008, 140, 123–132. [Google Scholar] [CrossRef]
- Tawana, K.; Rio-Machin, A.; Preudhomme, C.; Fitzgibbon, J. Familial CEBPA-mutated acute myeloid leukemia. Semin. Hematol. 2017, 54, 87–93. [Google Scholar] [CrossRef]
- Cheah, J.J.C.; Hahn, C.N.; Hiwase, D.K.; Scott, H.S.; Brown, A.L. Myeloid neoplasms with germline DDX41 mutation. Int. J. Hematol. 2017, 106, 163–174. [Google Scholar] [CrossRef]
- Tawana, K.; Wang, J.; Renneville, A.; Bödör, C.; Hills, R.; Loveday, C.; Savic, A.; Van Delft, F.W.; Treleaven, J.; Georgiades, P.; et al. Disease evolution and outcomes in familial AML with germline CEBPA mutations. Blood 2015, 126, 1214–1223. [Google Scholar] [CrossRef] [PubMed]
- Brown, A.L.; Hahn, C.N.; Scott, H.S. Secondary leukemia in patients with germline transcription factor mutations (RUNX1, GATA2, CEBPA). Blood 2020, 136, 24–35. [Google Scholar] [CrossRef] [PubMed]
- Martin, E.S.; Ferrer, A.; Mangaonkar, A.A.; Khan, S.P.; Kohorst, M.A.; Joshi, A.Y.; Hogan, W.J.; Olteanu, H.; Moyer, A.M.; Al-Kali, A.; et al. Spectrum of hematological malignancies, clonal evolution and outcomes in 144 Mayo Clinic patients with germline predisposition syndromes. Am. J. Hematol. 2021, 96, 1450–1460. [Google Scholar] [CrossRef] [PubMed]
- Song, W.J.; Sullivan, M.G.; Legare, R.D.; Hutchings, S.; Tan, X.; Kufrin, D.; Ratajczak, J.; Resende, I.C.; Haworth, C.; Hock, R.; et al. Haploinsufficiency of CBFA2 causes familial thrombocytopenia with propensity to develop acute myelogenous leukaemia. Nat. Genet. 1999, 23, 166–175. [Google Scholar] [CrossRef]
- Brown, A.L.; Arts, P.; Carmichael, C.L.; Babic, M.; Dobbins, J.; Chong, C.E.; Schreiber, A.W.; Feng, J.; Phillips, K.; Wang, P.P.S.; et al. RUNX1-mutated families show phenotype heterogeneity and a somatic mutation profile unique to germline predisposed AML. Blood Adv. 2020, 4, 1131–1144. [Google Scholar] [CrossRef]
- Noris, P.; Perrotta, S.; Seri, M.; Pecci, A.; Gnan, C.; Loffredo, G.; Pujol-Moix, N.; Zecca, M.; Scognamiglio, F.; De Rocco, D.; et al. Mutations in ANKRD26 are responsible for a frequent form of inherited thrombocytopenia: Analysis of 78 patients from 21 families. Blood 2011, 117, 6673–6680. [Google Scholar] [CrossRef]
- Zhang, M.Y.; Churpek, J.E.; Keel, S.B.; Walsh, T.; Lee, M.K.; Loeb, K.R.; Gulsuner, S.; Pritchard, C.C.; Sanchez-Bonilla, M.; Delrow, J.J.; et al. Germline ETV6 mutations in familial thrombocytopenia and hematologic malignancy. Nat. Genet. 2015, 47, 180–185. [Google Scholar] [CrossRef]
- Hahn, C.N.; Chong, C.E.; Carmichael, C.L.; Wilkins, E.J.; Brautigan, P.J.; Li, X.C.; Babic, M.; Lin, M.; Carmagnac, A.; Lee, Y.K.; et al. Heritable GATA2 mutations associated with familial myelodysplastic syndrome and acute myeloid leukemia. Nat. Genet. 2011, 43, 1012–1017. [Google Scholar] [CrossRef]
- Brown, P.; Inaba, H.; Annesley, C.; Beck, J.; Colace, S.; Dallas, M.; DeSantes, K.; Kelly, K.; Kitko, C.; Lacayo, N.; et al. Pediatric Acute Lymphoblastic Leukemia, Version 2.2020, NCCN Clinical Practice Guidelines in Oncology. J. Natl. Compr. Cancer Netw. 2020, 18, 81–112. [Google Scholar] [CrossRef]
- Akkari, Y.M.N.; Bruyere, H.; Hagelstrom, R.T.; Kanagal-Shamanna, R.; Liu, J.; Luo, M.; Mikhail, F.M.; Pitel, B.A.; Raca, G.; Shago, M.; et al. Evidence-based review of genomic aberrations in B-lymphoblastic leukemia/lymphoma: Report from the cancer genomics consortium working group for lymphoblastic leukemia. Cancer Genet. 2020, 243, 52–72. [Google Scholar] [CrossRef] [PubMed]
- Alaggio, R.; Amador, C.; Anagnostopoulos, I.; Attygalle, A.D.; Araujo, I.B.O.; Berti, E.; Bhagat, G.; Borges, A.M.; Boyer, D.; Calaminici, M.; et al. The 5th edition of the World Health Organization Classification of Haematolymphoid Tumours: Lymphoid Neoplasms. Leukemia 2022, 36, 1720–1748. [Google Scholar] [CrossRef]
- Rheingold, S.R.; Ji, L.; Gore, L.; Xu, X.; Bhojwani, D.; Shah, N.N.; Raetz, E.A.; Loh, M.L.; O’Brien, M.M. Impact of Treatment with Inotuzumab Ozogamicin before or after Chimeric Antigen Receptor T-Cell Therapy in Children with Relapsed/Refractory Acute Lymphoblastic Leukemia. Blood 2023, 142, 2876. [Google Scholar] [CrossRef]
- Hogan, L.E.; Brown, P.A.; Ji, L.; Xu, X.; Devidas, M.; Bhatla, T.; Borowitz, M.J.; Raetz, E.A.; Carroll, A.; Heerema, N.A.; et al. Children’s Oncology Group AALL1331: Phase III Trial of Blinatumomab in Children, Adolescents, and Young Adults With Low-Risk B-Cell ALL in First Relapse. J. Clin. Oncol. 2023, 41, 4118–4129. [Google Scholar] [CrossRef] [PubMed]
- Winestone, L.E.; Bhojwani, D.; Ghorashian, S.; Muffly, L.; Leahy, A.B.; Chao, K.; Steineck, A.; Rössig, C.; Lamble, A.; Maude, S.L.; et al. INSPIRED Symposium Part 4A: Access to CAR T Cell Therapy in Unique Populations with B Cell Acute Lymphoblastic Leukemia. Transplant. Cell Ther. 2024, 30, 56–70. [Google Scholar] [CrossRef] [PubMed]
- Lamble, A.J.; Moskop, A.; Pulsipher, M.A.; Maude, S.L.; Summers, C.; Annesley, C.; Baruchel, A.; Gore, L.; Amrolia, P.; Shah, N. INSPIRED Symposium Part 2: Prevention and Management of Relapse Following Chimeric Antigen Receptor T Cell Therapy for B Cell Acute Lymphoblastic Leukemia. Transplant. Cell Ther. 2023, 29, 674–684. [Google Scholar] [CrossRef] [PubMed]
- Brady, S.W.; Roberts, K.G.; Gu, Z.; Shi, L.; Pounds, S.; Pei, D.; Cheng, C.; Dai, Y.; Devidas, M.; Qu, C.; et al. The genomic landscape of pediatric acute lymphoblastic leukemia. Nat. Genet. 2022, 54, 1376–1389. [Google Scholar] [CrossRef] [PubMed]
- Safavi, S.; Paulsson, K. Near-haploid and low-hypodiploid acute lymphoblastic leukemia: Two distinct subtypes with consistently poor prognosis. Blood 2017, 129, 420–423. [Google Scholar] [CrossRef] [PubMed]
- Holmfeldt, L.; Wei, L.; Diaz-Flores, E.; Walsh, M.; Zhang, J.; Ding, L.; Payne-Turner, D.; Churchman, M.; Andersson, A.; Chen, S.C.; et al. The genomic landscape of hypodiploid acute lymphoblastic leukemia. Nat. Genet. 2013, 45, 242–252. [Google Scholar] [CrossRef] [PubMed]
- Harrison, C.J.; Moorman, A.V.; Broadfield, Z.J.; Cheung, K.L.; Harris, R.L.; Reza Jalali, G.; Robinson, H.M.; Barber, K.E.; Richards, S.M.; Mitchell, C.D.; et al. Three distinct subgroups of hypodiploidy in acute lymphoblastic leukaemia. Br. J. Haematol. 2004, 125, 552–559. [Google Scholar] [CrossRef]
- Nachman, J.B.; Heerema, N.A.; Sather, H.; Camitta, B.; Forestier, E.; Harrison, C.J.; Dastugue, N.; Schrappe, M.; Pui, C.H.; Basso, G.; et al. Outcome of treatment in children with hypodiploid acute lymphoblastic leukemia. Blood 2007, 110, 1112–1115. [Google Scholar] [CrossRef]
- Arpas, T.; Jelinkova, H.; Hrabovsky, S.; Orsulova, M.; Vrzalova, Z.; Navrkalova, V.; Brhelova, E.; Bryjova, L.; Bulikova, A.; Ondrouskova, E.; et al. Very rare near-haploid acute lymphoblastic leukemia resistant to immunotherapy and CAR-T therapy in 19-year-old male patient. Clin. Case Rep. 2022, 10, e05545. [Google Scholar] [CrossRef]
- Mühlbacher, V.; Zenger, M.; Schnittger, S.; Weissmann, S.; Kunze, F.; Kohlmann, A.; Bellos, F.; Kern, W.; Haferlach, T.; Haferlach, C. Acute lymphoblastic leukemia with low hypodiploid/near triploid karyotype is a specific clinical entity and exhibits a very high TP53 mutation frequency of 93%. Genes Chromosomes Cancer 2014, 53, 524–536. [Google Scholar] [CrossRef]
- Safavi, S.; Forestier, E.; Golovleva, I.; Barbany, G.; Nord, K.H.; Moorman, A.V.; Harrison, C.J.; Johansson, B.; Paulsson, K. Loss of chromosomes is the primary event in near-haploid and low-hypodiploid acute lymphoblastic leukemia. Leukemia 2013, 27, 248–250. [Google Scholar] [CrossRef][Green Version]
- Qu, C.; Song, Y.; Yin, J.; Ma, Y.; Kang, L.; Li, Z.; Dai, H.; Zhu, X.; Yu, L.; Wu, D.; et al. Decitabine may improve CAR-T efficacy in refractory/relapsed acute leukemia patients carrying TP53 alterations. Bone Marrow Transplant. 2021, 56, 1710–1713. [Google Scholar] [CrossRef]
- Cui, Y.; Zhou, M.; Zou, P.; Liao, X.; Xiao, J. Mature B cell acute lymphoblastic leukaemia with KMT2A-MLLT3 transcripts in children: Three case reports and literature reviews. Orphanet J. Rare Dis. 2021, 16, 331. [Google Scholar] [CrossRef] [PubMed]
- Worch, J.; Rohde, M.; Burkhardt, B. Mature B-cell lymphoma and leukemia in children and adolescents-review of standard chemotherapy regimen and perspectives. Pediatr. Hematol. Oncol. 2013, 30, 465–483. [Google Scholar] [CrossRef] [PubMed]
- Sajaroff, E.O.; Mansini, A.; Rubio, P.; Alonso, C.N.; Gallego, M.S.; Coccé, M.C.; Eandi-Eberle, S.; Bernasconi, A.R.; Ampatzidou, M.; Paterakis, G.; et al. B-cell acute lymphoblastic leukemia with mature phenotype and MLL rearrangement: Report of five new cases and review of the literature. Leuk. Lymphoma 2016, 57, 2289–2297. [Google Scholar] [CrossRef] [PubMed]
- Kenkel, T.J.; Sridhar, N.; Hammons, L.R.; Hintzke, M.; Shah, N.N. Bone Marrow Aplasia after CAR-T-Cell Therapy for Relapsed/Refractory Burkitt’s Lymphoma. Med. Sci. 2023, 11, 67. [Google Scholar] [CrossRef] [PubMed]
- Jeha, S.; Pei, D.; Raimondi, S.C.; Onciu, M.; Campana, D.; Cheng, C.; Sandlund, J.T.; Ribeiro, R.C.; Rubnitz, J.E.; Howard, S.C.; et al. Increased risk for CNS relapse in pre-B cell leukemia with the t(1;19)/TCF3-PBX1. Leukemia 2009, 23, 1406–1409. [Google Scholar] [CrossRef] [PubMed]
- Zenz, T.; Mertens, D.; Küppers, R.; Döhner, H.; Stilgenbauer, S. From pathogenesis to treatment of chronic lymphocytic leukaemia. Nat. Rev. Cancer 2010, 10, 37–50. [Google Scholar] [CrossRef] [PubMed]
- Demir, H.A.; Bayhan, T.; Üner, A.; Kurtulan, O.; Karakuş, E.; Emir, S.; Özyörük, D.; Ceylaner, S. Chronic lymphocytic leukemia in a child: A challenging diagnosis in pediatric oncology practice. Pediatr. Blood Cancer 2014, 61, 933–935. [Google Scholar] [CrossRef] [PubMed]
- Hallek, M.; Fischer, K.; Fingerle-Rowson, G.; Fink, A.M.; Busch, R.; Mayer, J.; Hensel, M.; Hopfinger, G.; Hess, G.; von Grünhagen, U.; et al. Addition of rituximab to fludarabine and cyclophosphamide in patients with chronic lymphocytic leukaemia: A randomised, open-label, phase 3 trial. Lancet 2010, 376, 1164–1174. [Google Scholar] [CrossRef]
- Siddiqi, T.; Maloney, D.G.; Kenderian, S.S.; Brander, D.M.; Dorritie, K.; Soumerai, J.; Riedell, P.A.; Shah, N.N.; Nath, R.; Fakhri, B.; et al. Lisocabtagene Maraleucel (liso-cel) in R/R CLL/SLL: 24-Month Median Follow-up of TRANSCEND CLL 004. Blood 2023, 142, 330. [Google Scholar] [CrossRef]
- Siddiqi, T.; Maloney, D.G.; Kenderian, S.S.; Brander, D.M.; Dorritie, K.; Soumerai, J.; Riedell, P.A.; Shah, N.N.; Nath, R.; Fakhri, B.; et al. Lisocabtagene maraleucel in chronic lymphocytic leukaemia and small lymphocytic lymphoma (TRANSCEND CLL 004): A multicentre, open-label, single-arm, phase 1-2 study. Lancet 2023, 402, 641–654. [Google Scholar] [CrossRef]
- Fries, C.; Evans, A.G.; Cheon, H.; Korones, D.N.; Loughran, T.P., Jr.; Andolina, J.R. Allogeneic Bone Marrow Transplant as a Cure for Refractory T-Cell Large Granular Lymphocytic Leukemia in an Adolescent. J. Pediatr. Hematol. Oncol. 2022, 44, e960–e963. [Google Scholar] [CrossRef]
- Mittal, N.; Loughran, T., Jr.; Rivers, A. T-cell large granular lymphocytic leukemia successfully treated with oral cyclosporine in a child: Case report and review of literature. Pediatr. Blood Cancer 2020, 67, e28487. [Google Scholar] [CrossRef]
- Kondoh, K.; Morimoto, M.; Keino, D.; Oyama, R.; Nagae, C.; Ashikaga, T.; Arai, K.; Nakazawa, A.; Kinoshita, A. T-cell large granular lymphocyte leukemia in a child with anemia and Crohn’s disease. Pediatr. Int. 2013, 55, 111–114. [Google Scholar] [CrossRef]
- Blanchong, C.A.; Olshefski, R.; Kahwash, S. Large granular lymphocyte leukemia: Case report of chronic neutropenia and rheumatoid arthritis-like symptoms in a child. Pediatr. Dev. Pathol. 2001, 4, 94–99. [Google Scholar] [CrossRef] [PubMed]
- Kitchen, B.J.; Boxer, L.A. Large granular lymphocyte leukemia (LGL) in a child with hyper IgM syndrome and autoimmune hemolytic anemia. Pediatr. Blood Cancer 2008, 50, 142–145. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.; Loughran, T.P., Jr. Large granular lymphocytic leukemia: Molecular pathogenesis, clinical manifestations, and treatment. Hematol. Am. Soc. Hematol. Educ. Program 2012, 2012, 652–659. [Google Scholar] [CrossRef]
- Park, S.H.; Lee, Y.J.; Kim, Y.; Kim, H.K.; Lim, J.H.; Jo, J.C. T-large granular lymphocytic leukemia. Blood Res. 2023, 58, S52–S57. [Google Scholar] [CrossRef] [PubMed]
- Alfano, G.; Fontana, F.; Colaci, E.; Mori, G.; Cerami, C.; Messerotti, A.; Potenza, L.; Luppi, M.; Cappelli, G. T-cell large granular lymphocyte leukemia in solid organ transplant recipients: Case series and review of the literature. Int. J. Hematol. 2019, 110, 313–321. [Google Scholar] [CrossRef]
- Rahul, E.; Ningombam, A.; Acharya, S.; Tanwar, P.; Ranjan, A.; Chopra, A. Large granular lymphocytic leukemia: A brief review. Am. J. Blood Res. 2022, 12, 17–32. [Google Scholar] [PubMed]
- Cheon, H.; Dziewulska, K.H.; Moosic, K.B.; Olson, K.C.; Gru, A.A.; Feith, D.J.; Loughran, T.P., Jr. Advances in the Diagnosis and Treatment of Large Granular Lymphocytic Leukemia. Curr. Hematol. Malig. Rep. 2020, 15, 103–112. [Google Scholar] [CrossRef] [PubMed]
- Lamy, T.; Loughran, T.P., Jr. How I treat LGL leukemia. Blood 2011, 117, 2764–2774. [Google Scholar] [CrossRef] [PubMed]
- Loughran, T.P., Jr.; Zickl, L.; Olson, T.L.; Wang, V.; Zhang, D.; Rajala, H.L.; Hasanali, Z.; Bennett, J.M.; Lazarus, H.M.; Litzow, M.R.; et al. Immunosuppressive therapy of LGL leukemia: Prospective multicenter phase II study by the Eastern Cooperative Oncology Group (E5998). Leukemia 2015, 29, 886–894. [Google Scholar] [CrossRef] [PubMed]
- Magnano, L.; Rivero, A.; Matutes, E. Large Granular Lymphocytic Leukemia: Current State of Diagnosis, Pathogenesis and Treatment. Curr. Oncol. Rep. 2022, 24, 633–644. [Google Scholar] [CrossRef]
| MDS Subtypes | Blasts | Cytogenetics | Mutations |
|---|---|---|---|
| MDS with defining genetic abnormalities | |||
| MDS with low blasts and 5q deletion | <5% BM and <2% PB | 5q deletion with or without 1 other abnormality other than 7q deletion or monosomy 7 | |
| MDS with low blasts and SF3B1 mutation | <5% BM and <2% PB | Absence of 5q deletion, monosomy 7, or abnormal karyotype | SF3B1 |
| MDS with biallelic TP53 inactivation 1 | <20% BM and PB | Often complex | TP53 1 |
| MDS morphologically defined | |||
| MDS with low blasts | <5% BM and <2% PB | ||
| MDS with increased blasts 2 | 5–19% BM or 2–20% PB | ||
| MDS, hypoplastic 3 |
| Risk Category | Cytogenetic Aberration |
|---|---|
| Very good | –Y, del(11q) |
| Good | Normal, del(5q), del(12p), del(20q), double including del(5q) |
| Intermediate | del(7q), +8, +19, isochromosome(17q), Any other single or double independent clones |
| Poor | –7, inv(3)/t(3q)/del(3q), double including –7/del(7q), Complex karyotype: 3 aberrations |
| Very poor | Complex karyotype: >3 aberrations |
| Variable | Score | ||||||
|---|---|---|---|---|---|---|---|
| 0 | 0.5 | 1 | 1.5 | 2 | 3 | 4 | |
| Cytogenetics | Very good | - | Good | - | Intermediate | Poor | Very poor |
| BM blast (%) | ≤2 | - | >2–<5 | - | 5–10 | >10 | - |
| Hemoglobin (g/dL) | ≥10 | - | 8–<10 | <8 | - | - | - |
| Platelets (×109/L) | ≥100 | 50–<100 | <50 | - | - | - | - |
| ANC (×109/L) | ≥0.8 | <0.8 | - | - | - | - | - |
| IPSS-R Risk Category | Cumulative Score |
|---|---|
| Very low | ≤1.5 |
| Low | >1.5–≤3.0 |
| Intermediate | >3.0–≤4.5 |
| High | >4.5–≤6.0 |
| Very high | >6.0 |
| FAB Subtype | Description |
|---|---|
| M0 | AML without differentiation |
| M1 | AML with minimal differentiation |
| M2 | AML with differentiation |
| M3 | APL, hypergranular |
| M3v | APL, microgranular |
| M4 | AMML |
| M4Eo | AMML with eosinophilia |
| M5 | Acute monocytic leukemia |
| M6 | Acute erythroblastic leukemia |
| M7 | Acute megakaryoblastic leukemia |
| AML with defining genetic abnormalities 1 |
| APL with PML-RARA fusion |
| AML with RUNX1-RUNX1T1 fusion |
| AML with CBFB-MYH11 fusion |
| AML with DEK-NUP214 fusion |
| AML with RBM15-MRTFA fusion |
| AML with BCR-ABL1 fusion |
| AML with KMT2A rearrangement |
| AML with MECOM rearrangement |
| AML with NUP98 rearrangement |
| AML with NPM1 mutation |
| AML with CEBPA mutation |
| AML, myelodysplasia-related |
| AML with other defined genetic alterations |
| AML defined by differentiation 2 |
| AML with minimal differentiation |
| AML without maturation |
| AML with maturation |
| Acute basophilic leukemia |
| Acute myelomonocytic leukemia |
| Acute monocytic leukemia |
| Acute erythroid leukemia |
| Acute megakaryoblastic leukemia |
| Target | Genomic Alteration 1 | Agent | Clinical Trial 2 |
|---|---|---|---|
| FLT3 | FLT3-ITD | Gilteritinib Quizartinib | NCT04293562 3 NCT04240002 NCT03793478 |
| FLT3-TKD | Gilteritinib | NCT04293562 3 | |
| BCL2 | - | Venetoclax | NCT03194932 NCT03826992 NCT03236857 NCT04898894 NCT05317403 |
| IDH | IDH2 | Enasidenib | NCT04203316 |
| Menin | KMT2Ar, NPM1, NUP98 | SNDX-5613 | NCT04065399 NCT05326516 |
| E-selectin | E-selectin ligand expression | Uproleselan | NCT05146739 |
| XPO1 | - | Selinexor | NCT04898894 |
| CD33 | - | Gemtuzumab ozogamicin (ADC) Anti-CD33 CAR-T | NCT02724163 NCT03971799 |
| CD123 | - | Anti-CD123 CAR-T | NCT04318678 NCT04678336 |
| Without pre-existing platelet disorder or organ dysfunction |
|
| With pre-existing platelet disorder |
|
| With risk of organ dysfunction |
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Brown, A.; Batra, S. Rare Hematologic Malignancies and Pre-Leukemic Entities in Children and Adolescents Young Adults. Cancers 2024, 16, 997. https://doi.org/10.3390/cancers16050997
Brown A, Batra S. Rare Hematologic Malignancies and Pre-Leukemic Entities in Children and Adolescents Young Adults. Cancers. 2024; 16(5):997. https://doi.org/10.3390/cancers16050997
Chicago/Turabian StyleBrown, Amber, and Sandeep Batra. 2024. "Rare Hematologic Malignancies and Pre-Leukemic Entities in Children and Adolescents Young Adults" Cancers 16, no. 5: 997. https://doi.org/10.3390/cancers16050997
APA StyleBrown, A., & Batra, S. (2024). Rare Hematologic Malignancies and Pre-Leukemic Entities in Children and Adolescents Young Adults. Cancers, 16(5), 997. https://doi.org/10.3390/cancers16050997







