Specific Subtypes of Carcinoma-Associated Fibroblasts Are Correlated with Worse Survival in Resectable Pancreatic Ductal Adenocarcinoma
Simple Summary
Abstract
1. Background
2. Methods
2.1. Patients and Tumor Samples
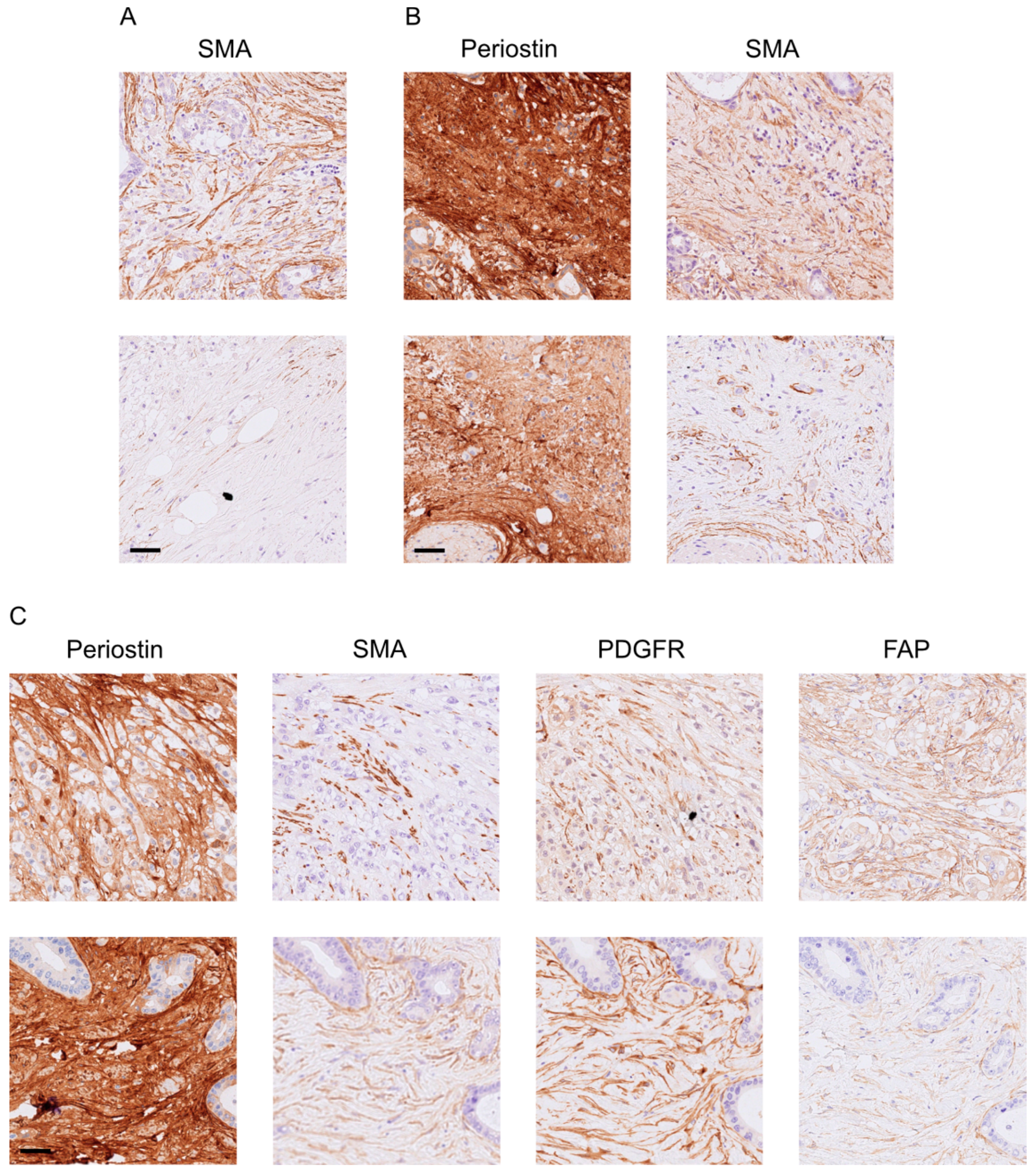
2.2. Immunohistochemistry (IHC) and Analysis
2.3. Statistical Analysis
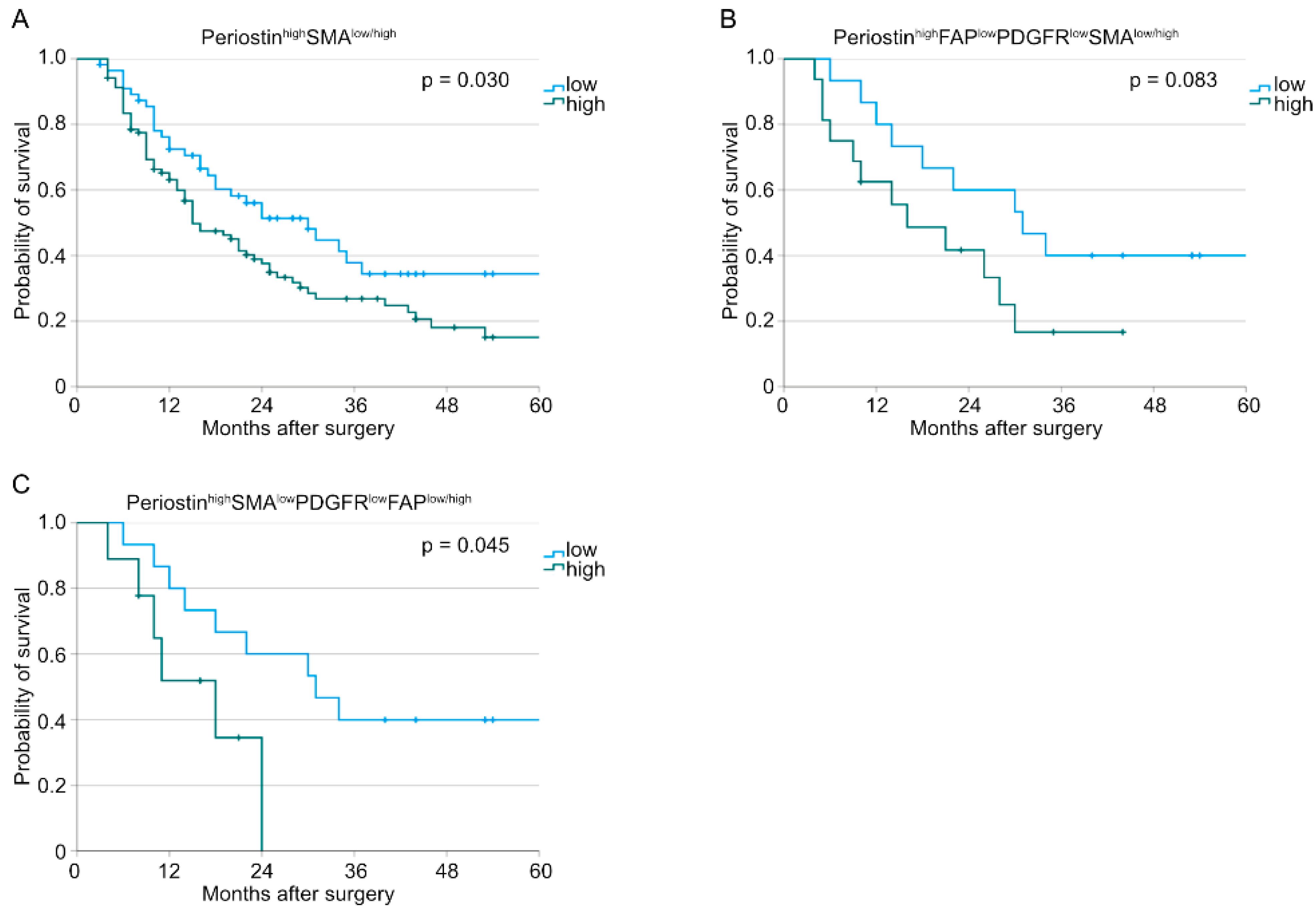
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A. PANCALYZE Study Group
- 1 Department of General, Visceral and Cancer Surgery, University Hospital of Cologne, Faculty of Medicine, University of Cologne, Cologne, Germany
- 3 Department of General and Visceral Surgery, Sana Klinikum Lichtenberg, Berlin, Germany
- 4 Department of General and Visceral Surgery, Klinikum Ernst von Bergmann, Potsdam, Germany
- 5 Department of General and Visceral Surgery, Ostalb-Klinikum Aalen, Aalen, Germany
- 6 Department of General and Visceral Surgery, Carl-von-Basedow Klinikum Saalekreis, Merseburg, Germany
- 7 Department of General and Visceral Surgery, Municipal Hospital Lüneburg, Lüneburg, Germany
- 8 Department of General and Visceral Surgery, Klinikum Osnabrück, Osnabrück, Germany
- 9 Department of General and Visceral Surgery, Evangelisches Krankenhaus Düsseldorf, Düsseldorf, Germany
- 10 Department of General and Visceral Surgery, KMG Klinikum Güstrow, Güstrow, Germany
- 11 Department of General and Visceral Surgery, Klinikum Neumarkt, Neumarkt, Germany
- 12 Department of General and Visceral Surgery, Alfried Krupp Krankenhaus, Essen, Germany
- 13 Department of General and Visceral Surgery, Carl-Thiem-Klinikum Cottbus, Cottbus, Germany
- 14 Department of General and Visceral Surgery, Klinikum Robert Koch Gehrden, Gehrden, Germany
- 15 Department of General and Visceral Surgery, Klinikum Lippe, Detmold, Germany
- 16 Department of General and Visceral Surgery, University Hospital of Jena, Jena, Germany
References
- GBD Pancreatic Cancer Collaborators. The global, regional, and national burden of pancreatic cancer and its attributable risk factors in 195 countries and territories, 1990–2017: A systematic analysis for the Global Burden of Disease Study 2017. Lancet Gastroenterol. Hepatol. 2019, 4, 934–947. [Google Scholar] [CrossRef]
- Ferlay, J.; Partensky, C.; Bray, F. More deaths from pancreatic cancer than breast cancer in the EU by 2017. Acta Oncol. 2016, 55, 1158–1160. [Google Scholar] [CrossRef]
- Hanahan, D.; Coussens, L.M. Accessories to the crime: Functions of cells recruited to the tumor microenvironment. Cancer Cell 2012, 21, 309–322. [Google Scholar] [CrossRef]
- Hanahan, D.; Weinberg, R.A. Hallmarks of cancer: The next generation. Cell 2011, 144, 646–674. [Google Scholar] [CrossRef]
- Huang, X.; Ding, L.; Liu, X.; Tong, R.; Ding, J.; Qian, Z.; Cai, L.; Zhang, P.; Li, D. Regulation of tumor microenvironment for pancreatic cancer therapy. Biomaterials 2021, 270, 120680. [Google Scholar] [CrossRef]
- Awaji, M.; Singh, R.K. Cancer-Associated Fibroblasts’ Functional Heterogeneity in Pancreatic Ductal Adenocarcinoma. Cancers 2019, 11, 290. [Google Scholar] [CrossRef]
- Valls, A.F.; Knipper, K.; Giannakouri, E.; Sarachaga, V.; Hinterkopf, S.; Wuehrl, M.; Shen, Y.; Radhakrishnan, P.; Klose, J.; Ulrich, A.; et al. VEGFR1+ Metastasis–Associated Macrophages Contribute to Metastatic Angiogenesis and Influence Colorectal Cancer Patient Outcome. Clin. Cancer Res. 2019, 25, 5674–5685. [Google Scholar] [CrossRef]
- Manoukian, P.; Bijlsma, M.; van Laarhoven, H. The Cellular Origins of Cancer-Associated Fibroblasts and Their Opposing Contributions to Pancreatic Cancer Growth. Front. Cell Dev. Biol. 2021, 9, 743907. [Google Scholar] [CrossRef]
- Zhang, T.; Ren, Y.; Yang, P.; Wang, J.; Zhou, H. Cancer-associated fibroblasts in pancreatic ductal adenocarcinoma. Cell Death Dis. 2022, 13, 897. [Google Scholar] [CrossRef]
- Gorchs, L.; Kaipe, H. Interactions between Cancer-Associated Fibroblasts and T Cells in the Pancreatic Tumor Microenvironment and the Role of Chemokines. Cancers 2021, 13, 2995. [Google Scholar] [CrossRef]
- Özdemir, B.C.; Pentcheva-Hoang, T.; Carstens, J.L.; Zheng, X.; Wu, C.-C.; Simpson, T.R.; Laklai, H.; Sugimoto, H.; Kahlert, C.; Novitskiy, S.V.; et al. Depletion of carcinoma-associated fibroblasts and fibrosis induces immunosuppression and accelerates pancreas cancer with reduced survival. Cancer Cell 2014, 25, 719–734. [Google Scholar] [CrossRef] [PubMed]
- Biffi, G.; Oni, T.E.; Spielman, B.; Hao, Y.; Elyada, E.; Park, Y.; Preall, J.; Tuveson, D.A. IL1-Induced JAK/STAT Signaling Is Antagonized by TGFβ to Shape CAF Heterogeneity in Pancreatic Ductal Adenocarcinoma. Cancer Discov. 2019, 9, 282–301. [Google Scholar] [CrossRef]
- Kanzaki, R.; Pietras, K. Heterogeneity of cancer-associated fibroblasts: Opportunities for precision medicine. Cancer Sci. 2020, 111, 2708–2717. [Google Scholar] [CrossRef] [PubMed]
- Feig, C.; Gopinathan, A.; Neesse, A.; Chan, D.S.; Cook, N.; Tuveson, D.A. The Pancreas Cancer Microenvironment. Clin. Cancer Res. 2012, 18, 4266–4276. [Google Scholar] [CrossRef]
- Tomasek, J.J.; Gabbiani, G.; Hinz, B.; Chaponnier, C.; Brown, R.A. Myofibroblasts and mechano-regulation of connective tissue remodelling. Nat. Rev. Mol. Cell Biol. 2002, 3, 349–363. [Google Scholar] [CrossRef] [PubMed]
- Shen, Y.; Wang, X.; Lu, J.; Salfenmoser, M.; Wirsik, N.M.; Schleussner, N.; Imle, A.; Valls, A.F.; Radhakrishnan, P.; Liang, J.; et al. Reduction of Liver Metastasis Stiffness Improves Response to Bevacizumab in Metastatic Colorectal Cancer. Cancer Cell 2020, 37, 800–817.e7. [Google Scholar] [CrossRef] [PubMed]
- Fujita, H.; Ohuchida, K.; Mizumoto, K.; Nakata, K.; Yu, J.; Kayashima, T.; Cui, L.; Manabe, T.; Ohtsuka, T.; Tanaka, M. α-Smooth Muscle Actin Expressing Stroma Promotes an Aggressive Tumor Biology in Pancreatic Ductal Adenocarcinoma. Pancreas 2010, 39, 1254–1262. [Google Scholar] [CrossRef] [PubMed]
- Östman, A. PDGF receptors-mediators of autocrine tumor growth and regulators of tumor vasculature and stroma. Cytokine Growth Factor Rev. 2004, 15, 275–286. [Google Scholar] [CrossRef]
- Fjällskog, M.-L.; Hessman, O.; Eriksson, B.; Janson, E.T. Upregulated expression of PDGF receptor beta in endocrine pancreatic tumors and metastases compared to normal endocrine pancreas. Acta Oncol. 2007, 46, 741–746. [Google Scholar] [CrossRef]
- Yuzawa, S.; Kano, M.R.; Einama, T.; Nishihara, H. PDGFRβ expression in tumor stroma of pancreatic adenocarcinoma as a reliable prognostic marker. Med. Oncol. 2012, 29, 2824–2830. [Google Scholar] [CrossRef]
- Ono, J.; Takai, M.; Kamei, A.; Azuma, Y.; Izuhara, K. Pathological Roles and Clinical Usefulness of Periostin in Type 2 Inflammation and Pulmonary Fibrosis. Biomolecules 2021, 11, 1084. [Google Scholar] [CrossRef] [PubMed]
- Baril, P.; Gangeswaran, R.; Mahon, P.C.; Caulee, K.; Kocher, H.M.; Harada, T.; Zhu, M.; Kalthoff, H.; Crnogorac-Jurcevic, T.; Lemoine, N.R. Periostin promotes invasiveness and resistance of pancreatic cancer cells to hypoxia-induced cell death: Role of the beta4 integrin and the PI3k pathway. Oncogene 2007, 26, 2082–2094. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Li, F.; Gao, F.; Xing, L.; Qin, P.; Liang, X.; Zhang, J.; Qiao, X.; Lin, L.; Zhao, Q.; et al. Role of microenvironmental periostin in pancreatic cancer progression. Oncotarget 2016, 8, 89552–89565. [Google Scholar] [CrossRef] [PubMed]
- Jacob, M.; Chang, L.; Pure, E. Fibroblast Activation Protein in Remodeling Tissues. Curr. Mol. Med. 2012, 12, 1220–1243. [Google Scholar] [CrossRef]
- Liu, F.; Qi, L.; Liu, B.; Liu, J.; Zhang, H.; Che, D.; Cao, J.; Shen, J.; Geng, J.; Bi, Y.; et al. Fibroblast Activation Protein Overexpression and Clinical Implications in Solid Tumors: A Meta-Analysis. PLoS ONE 2015, 10, e0116683. [Google Scholar] [CrossRef] [PubMed]
- Shi, M.; Yu, D.-H.; Chen, Y.; Zhao, C.-Y.; Zhang, J.; Liu, Q.-H.; Ni, C.-R.; Zhu, M.-H. Expression of fibroblast activation protein in human pancreatic adenocarcinoma and its clinicopathological significance. World J. Gastroenterol. 2012, 18, 840–846. [Google Scholar] [CrossRef] [PubMed]
- Cohen, S.J.; Alpaugh, R.K.; Palazzo, I.; Meropol, N.J.; Rogatko, A.; Xu, Z.; Hoffman, J.P.; Weiner, L.M.; Cheng, J.D. Fibroblast Activation Protein and Its Relationship to Clinical Outcome in Pancreatic Adenocarcinoma. Pancreas 2008, 37, 154–158. [Google Scholar] [CrossRef]
- Ostermann, E.; Garin-Chesa, P.; Heider, K.H.; Kalat, M.; Lamche, H.; Puri, C.; Kerjaschki, D.; Rettig, W.J.; Adolf, G.R. Effective Immunoconjugate Therapy in Cancer Models Targeting a Serine Protease of Tumor Fibroblasts. Clin. Cancer Res. 2008, 14, 4584–4592. [Google Scholar] [CrossRef]
- Tran, E.; Chinnasamy, D.; Yu, Z.; Morgan, R.A.; Lee, C.-C.R.; Restifo, N.P.; Rosenberg, S.A. Immune targeting of fibroblast activation protein triggers recognition of multipotent bone marrow stromal cells and cachexia. J. Exp. Med. 2013, 210, 1125–1135. [Google Scholar] [CrossRef]
- Deutsche Krebsgesellschaft; DKG; AWMF. S3-Leitlinie Exokrines Pankreaskarzinom, Langversion 2.0; AWMF Registernummer: 032-010OL; AWMF: Düsseldorf, Germany, 2021. [Google Scholar]
- Simon, R. Applications of Tissue Microarray Technology. Methods Mol. Biol. 2010, 664, 1–16. [Google Scholar] [CrossRef]
- Popp, F.C.; Popp, M.C.; Zhao, Y.; Betzler, C.; Kropf, S.; Garlipp, B.; Benckert, C.; Kalinski, T.; Lippert, H.; Bruns, C.J. Protocol of the PANCALYZE trial: A multicenter, prospective study investigating the tumor biomarkers CXCR4, SMAD4, SOX9 and IFIT3 in patients with resected pancreatic adenocarcinoma to predict the pattern of recurrence of the disease. BMC Cancer 2017, 17, 229. [Google Scholar] [CrossRef]
- Bankhead, P.; Loughrey, M.B.; Fernández, J.A.; Dombrowski, Y.; McArt, D.G.; Dunne, P.D.; McQuaid, S.; Gray, R.T.; Murray, L.J.; Coleman, H.G.; et al. QuPath: Open source software for digital pathology image analysis. Sci. Rep. 2017, 7, 16878. [Google Scholar] [CrossRef] [PubMed]
- Conroy, T.; Hammel, P.; Hebbar, M.; Ben Abdelghani, M.; Wei, A.C.; Raoul, J.-L.; Choné, L.; Francois, E.; Artru, P.; Biagi, J.J.; et al. FOLFIRINOX or Gemcitabine as Adjuvant Therapy for Pancreatic Cancer. N. Engl. J. Med. 2018, 379, 2395–2406. [Google Scholar] [CrossRef] [PubMed]
- Conroy, T.; Castan, F.; Lopez, A.; Turpin, A.; Abdelghani, M.B.; Wei, A.C.; Mitry, E.; Biagi, J.J.; Evesque, L.; Artru, P.; et al. Five-Year Outcomes of FOLFIRINOX vs Gemcitabine as Adjuvant Therapy for Pancreatic Cancer: A Randomized Clinical Trial. JAMA Oncol. 2022, 8, 1571–1578. [Google Scholar] [CrossRef] [PubMed]
- Kole, C.; Charalampakis, N.; Tsakatikas, S.; Frountzas, M.; Apostolou, K.; Schizas, D. Immunotherapy in Combination with Well-Established Treatment Strategies in Pancreatic Cancer: Current Insights. Cancer Manag. Res. 2022, 14, 1043–1061. [Google Scholar] [CrossRef] [PubMed]
- Feig, C.; Jones, J.O.; Kraman, M.; Wells, R.J.; Deonarine, A.; Chan, D.S.; Connell, C.M.; Roberts, E.W.; Zhao, Q.; Caballero, O.L.; et al. Targeting CXCL12 from FAP-expressing carcinoma-associated fibroblasts synergizes with anti-PD-L1 immunotherapy in pancreatic cancer. Proc. Natl. Acad. Sci. USA 2013, 110, 20212–20217. [Google Scholar] [CrossRef]
- Ohlund, D.; Handly-Santana, A.; Biffi, G.; Elyada, E.; Almeida, A.S.; Ponz-Sarvise, M.; Corbo, V.; Oni, T.E.; Hearn, S.A.; Lee, E.J.; et al. Distinct populations of inflammatory fibroblasts and myofibroblasts in pancreatic cancer. J. Exp. Med. 2017, 214, 579–596. [Google Scholar] [CrossRef]
- Hu, G.; Huang, L.; Zhong, K.; Meng, L.; Xu, F.; Wang, S.; Zhang, T. PDGFR-β(+) fibroblasts deteriorate survival in human solid tumors: A meta-analysis. Aging 2021, 13, 13693–13707. [Google Scholar] [CrossRef]
- Van den Brand, M.; Hoevenaars, B.M.; Sigmans, J.H.M.; Meijer, J.W.R.; Van Cleef, P.H.J.; Groenen, P.J.T.A.; Hebeda, K.M.; Van Krieken, J.H.J.M. Sequential immunohistochemistry: A promising new tool for the pathology laboratory. Histopathology 2014, 65, 651–657. [Google Scholar] [CrossRef]
- Gorris, M.A.J.; Halilovic, A.; Rabold, K.; van Duffelen, A.; Wickramasinghe, I.N.; Verweij, D.; Wortel, I.M.N.; Textor, J.C.; de Vries, I.J.M.; Figdor, C.G. Eight-Color Multiplex Immunohistochemistry for Simultaneous Detection of Multiple Immune Checkpoint Molecules within the Tumor Microenvironment. J. Immunol. 2018, 200, 347–354. [Google Scholar] [CrossRef]
- Stack, E.C.; Wang, C.; Roman, K.A.; Hoyt, C.C. Multiplexed immunohistochemistry, imaging, and quantitation: A review, with an assessment of Tyramide signal amplification, multispectral imaging and multiplex analysis. Methods 2014, 70, 46–58. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Liu, L.; Yao, H.H.; Zhu, Z.Q.; Ning, Z.L.; Huang, Q. Stromal Myofibroblasts Are Associated with Poor Prognosis in Solid Cancers: A Meta-Analysis of Published Studies. PLoS ONE 2016, 11, e01599472016. [Google Scholar] [CrossRef] [PubMed]
- Sinn, M.; Denkert, C.; Striefler, J.K.; Pelzer, U.; Stieler, J.M.; Bahra, M.; Lohneis, P.; Dörken, B.; Oettle, H.; Riess, H.; et al. α-Smooth muscle actin expression and desmoplastic stromal reaction in pancreatic cancer: Results from the CONKO-001 study. Br. J. Cancer 2014, 111, 1917–1923. [Google Scholar] [CrossRef] [PubMed]
- Tsujino, T.; Seshimo, I.; Yamamoto, H.; Ngan, C.Y.; Ezumi, K.; Takemasa, I.; Ikeda, M.; Sekimoto, M.; Matsuura, N.; Monden, M. Stromal Myofibroblasts Predict Disease Recurrence for Colorectal Cancer. Clin. Cancer Res. 2007, 13, 2082–2090. [Google Scholar] [CrossRef]
- Costa, A.; Kieffer, Y.; Scholer-Dahirel, A.; Pelon, F.; Bourachot, B.; Cardon, M.; Sirven, P.; Magagna, I.; Fuhrmann, L.; Bernard, C.; et al. Fibroblast heterogeneity and immunosuppressive environment in human breast cancer. Cancer Cell 2018, 33, 463–479.e10. [Google Scholar] [CrossRef]
- Helms, E.; Onate, M.K.; Sherman, M.H. Fibroblast Heterogeneity in the Pancreatic Tumor Microenvironment. Cancer Discov. 2020, 10, 648–656. [Google Scholar] [CrossRef]


| Characteristic | Total | SMA-Low | SMA-High | |
|---|---|---|---|---|
| n (%) | n (%) | n (%) | ||
| No. of patients | 321 (100) | 145 (100) | 176 (100) | |
| Sex | 0.270 | |||
| Male | 157 (48.9) | 66 (45.5) | 91 (51.7) | |
| Female | 164 (51.1) | 79 (54.5) | 85 (48.3) | |
| Age | 0.996 | |||
| <65 | 104 (32.4) | 47 (32.4) | 57 (32.4) | |
| ≥65 | 217 (67.6) | 98 (67.6) | 119 (67.6) | |
| Median follow-up period (months) | 18 | 20 | 16 | |
| (range) | (3–98) | (3–72) | (3–98) | |
| Preoperative staging | 0.405 | |||
| Primarily resectable | 277 (86.3) | 126 (86.9) | 151 (85.8) | |
| Borderline resectable | 38 (11.8) | 14 (9.7) | 24 (13.6) | |
| Locally advanced | 1 (0.3) | 0 (0.0) | 1 (0.6) | |
| Unknown | 5 (1.6) | 5 (3.4) | 0 (0.0) | |
| Neoadjuvant therapy | 0.700 | |||
| No | 304 (94.7) | 139 (95.9) | 165 (93.8) | |
| Chemotherapy | 14 (4.4) | 5 (3.4) | 9 (5.1) | |
| Radiochemotherapy | 3 (0.9) | 1 (0.7) | 2 (1.1) | |
| pT | 0.870 | |||
| 1 | 22 (6.9) | 10 (6.9) | 12 (6.8) | |
| 2 | 121 (37.7) | 52 (35.9) | 69 (39.2) | |
| 3 | 171 (53.3) | 79 (54.5) | 92 (52.3) | |
| 4 | 7 (2.2) | 4 (2.8) | 3 (1.7) | |
| pN | 0.324 | |||
| 0 | 93 (29.0) | 46 (31.7) | 47 (26.7) | |
| 1 | 228 (71.0) | 99 (68.3) | 129 (73.3) | |
| R | 0.262 | |||
| 0 | 207 (64.5) | 88 (60.7) | 119 (67.6) | |
| 1 | 113 (35.2) | 56 (38.6) | 57 (32.4) | |
| 2 | 1 (0.3) | 1 (0.7) | 0 (0.0) | |
| Pn | 0.340 | |||
| 0 | 77 (24.0) | 39 (26.9) | 38 (21.6) | |
| 1 | 232 (72.3) | 103 (71.0) | 129 (73.3) | |
| Unknown | 12 (3.7) | 3 (2.1) | 9 (5.1) | |
| L | 0.872 | |||
| 0 | 123 (38.3) | 55 (37.9) | 68 (38.6) | |
| 1 | 195 (60.8) | 89 (61.4) | 106 (60.2) | |
| Unknown | 3 (0.9) | 1 (0.7) | 2 (1.2) | |
| V | 0.877 | |||
| 0 | 221 (68.9) | 99 (68.3) | 122 (69.3) | |
| 1 | 94 (29.3) | 43 (29.6) | 51 (29.0) | |
| Unknown | 6 (1.8) | 3 (2.1) | 3 (1.7) |
| Characteristic | Borders | Hazard Ratio | 95% Confidence Interval | p-Value |
|---|---|---|---|---|
| Preoperative staging | 0.125 | |||
| borderline vs. primarily resectable | 1.497 | 0.973–2.304 | 0.066 | |
| locally advanced vs. primarily resectable | 2.678 | 0.359–19.988 | 0.337 | |
| pT | 0.007 | |||
| 2 vs. 1 | 1.395 | 0.654–2.976 | 0.389 | |
| 3 vs. 1 | 2.316 | 1.091–4.916 | 0.029 | |
| 4 vs. 1 | 1.791 | 0.478–6.717 | 0.387 | |
| pN | 1 vs. 0 | 2.177 | 1.449–3.271 | <0.001 |
| R | ≥1 vs. 0 | 1.228 | 0.900–1.676 | 0.195 |
| Pn | 1 vs. 0 | 0.983 | 0.667–1.449 | 0.931 |
| L | 1 vs. 0 | 0.806 | 0.563–1.154 | 0.238 |
| V | 1 vs. 0 | 1.064 | 0.750–1.509 | 0.730 |
| SMA | high vs. low | 1.389 | 1.019–1.893 | 0.038 |
| FAP | high vs. low | 1.249 | 0.921–1.695 | 0.153 |
| PDGFR | high vs. low | 1.047 | 0.753–1.455 | 0.786 |
| Periostin | high vs. low | 1.078 | 0.790–1.471 | 0.637 |
| Characteristic | PeriostinhighSMAlow | PeriostinhighSMAhigh | PeriostinhighSMAlow | PeriostinhighSMAlow | ||
|---|---|---|---|---|---|---|
| PDGFRlowFAPlow | PDGFRlowFAPhigh | |||||
| n (%) | n (%) | n (%) | n (%) | |||
| No. of patients | 56 (100.0) | 103 (100.0) | 15 (100.0) | 9 (100.0) | ||
| Sex | 0.328 | 0.916 | ||||
| Male | 27 (48.2) | 58 (56.3) | 7 (46.7) | 4 (44.4) | ||
| Female | 29 (51.8) | 45 (43.7) | 8 (53.3) | 5 (55.6) | ||
| Age | 0.411 | 0.562 | ||||
| <65 | 21 (37.5) | 32 (31.1) | 5 (33.3) | 2 (22.2) | ||
| ≥65 | 35 (62.5) | 71 (68.9) | 10 (66.7) | 7 (77.8) | ||
| Median follow-up period (months) | 21 | 14 | 31 | 11 | ||
| (range) | (3–65) | (4–73) | (6–65) | (4–24) | ||
| Preoperative staging | 0.735 | 0.235 | ||||
| Primarily resectable | 48 (85.7) | 89 (86.4) | 12 (80.0) | 9 (100.0) | ||
| Borderline resectable | 6 (10.7) | 13 (12.6) | 2 (13.3) | 0 (0.0) | ||
| Locally advanced | 0 (0.0) | 1 (1.0) | 0 (0.0) | 0 (0.0) | ||
| Unknown | 2 (3.6) | 0 (0.0) | 1 (6.7) | 0 (0.0) | ||
| Neoadjuvant therapy | 0.101 | - | ||||
| No | 56 (100.0) | 95 (92.2) | 15 (100.0) | 9 (100.0) | ||
| Chemotherapy | 0 (0.0) | 6 (5.8) | 0 (0.0) | 0 (0.0) | ||
| Radiochemotherapy | 0 (0.0) | 2 (1.9) | 0 (0.0) | 0 (0.0) | ||
| pT | 0.463 | 0.742 | ||||
| 1 | 7 (12.5) | 6 (5.8) | 1 (6.7) | 1 (11.1) | ||
| 2 | 17 (30.4) | 39 (37.9) | 3 (20.0) | 3 (33.3) | ||
| 3 | 31 (55.4) | 56 (54.4) | 10 (66.7) | 5 (55.6) | ||
| 4 | 1 (1.8) | 2 (1.9) | 1 (6.7) | 0 (0.0) | ||
| pN | 0.749 | 0.572 | ||||
| 0 | 16 (28.6) | 27 (26.2) | 3 (20.0) | 1 (11.1) | ||
| 1 | 40 (71.4) | 76 (73.8) | 12 (80.0) | 8 (88.9) | ||
| R | 0.218 | 0.729 | ||||
| 0 | 32 (57.1) | 69 (67.0) | 8 (53.3) | 5 (55.6) | ||
| 1 | 23 (41.1) | 34 (33.0) | 6 (40.0) | 4 (44.4) | ||
| 2 | 1 (1.8) | 0 (0.0) | 1 (6.7) | 0 (0.0) | ||
| Pn | 0.841 | 0.526 | ||||
| 0 | 12 (21.4) | 23 (22.3) | 3 (20.0) | 3 (33.3) | ||
| 1 | 43 (76.8) | 76 (73.8) | 11 (73.3) | 6 (66.7) | ||
| Unknown | 1 (1.8) | 4 (3.9) | 1 (6.7) | 0 (0.0) | ||
| L | 0.783 | 0.562 | ||||
| 0 | 24 (42.9) | 41 (39.8) | 5 (33.3) | 2 (22.2) | ||
| 1 | 32 (57.1) | 60 (58.3) | 10 (66.7) | 7 (77.8) | ||
| Unknown | 0 (0.0) | 2 (1.9) | 0 (0.0) | 0 (0.0) | ||
| V | 0.487 | 0.285 | ||||
| 0 | 35 (62.5) | 68 (66.0) | 10 (66.7) | 4 (44.4) | ||
| 1 | 21 (37.5) | 32 (31.1) | 5 (33.3) | 5 (55.6) | ||
| Unknown | 0 (0.0) | 3 (2.9) | 0 (0.0) | 0 (0.0) |
| Staining | Treatment-Naïve | Neoadjuvant Treatment | |
|---|---|---|---|
| n (%) | n (%) | ||
| No. of patients | 304 (100.0) | 17 (100.0) | |
| FAP | 0.026 | ||
| Low | 148 (48.7) | 13 (76.5) | |
| High | 156 (51.3) | 4 (23.5) | |
| PDGFR | 0.024 | ||
| Low | 157 (51.6) | 4 (23.5) | |
| High | 147 (48.4) | 13 (76.5) | |
| Periostin | 0.813 | ||
| Low | 152 (50.0) | 9 (52.9) | |
| High | 152 (50.0) | 8 (47.1) | |
| SMA | 0.400 | ||
| Low | 139 (45.7) | 6 (35.3) | |
| High | 165 (54.3) | 11 (64.7) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Knipper, K.; Damanakis, A.I.; Zhao, Y.; Bruns, C.J.; Schmidt, T.; Popp, F.C.; Quaas, A.; Lyu, S.I., on behalf of the PANCALYZE Study Group. Specific Subtypes of Carcinoma-Associated Fibroblasts Are Correlated with Worse Survival in Resectable Pancreatic Ductal Adenocarcinoma. Cancers 2023, 15, 2049. https://doi.org/10.3390/cancers15072049
Knipper K, Damanakis AI, Zhao Y, Bruns CJ, Schmidt T, Popp FC, Quaas A, Lyu SI on behalf of the PANCALYZE Study Group. Specific Subtypes of Carcinoma-Associated Fibroblasts Are Correlated with Worse Survival in Resectable Pancreatic Ductal Adenocarcinoma. Cancers. 2023; 15(7):2049. https://doi.org/10.3390/cancers15072049
Chicago/Turabian StyleKnipper, Karl, Alexander I. Damanakis, Yue Zhao, Christiane J. Bruns, Thomas Schmidt, Felix C. Popp, Alexander Quaas, and Su Ir Lyu on behalf of the PANCALYZE Study Group. 2023. "Specific Subtypes of Carcinoma-Associated Fibroblasts Are Correlated with Worse Survival in Resectable Pancreatic Ductal Adenocarcinoma" Cancers 15, no. 7: 2049. https://doi.org/10.3390/cancers15072049
APA StyleKnipper, K., Damanakis, A. I., Zhao, Y., Bruns, C. J., Schmidt, T., Popp, F. C., Quaas, A., & Lyu, S. I., on behalf of the PANCALYZE Study Group. (2023). Specific Subtypes of Carcinoma-Associated Fibroblasts Are Correlated with Worse Survival in Resectable Pancreatic Ductal Adenocarcinoma. Cancers, 15(7), 2049. https://doi.org/10.3390/cancers15072049








