Efficacy of Organ Preservation Strategy by Adjuvant Chemoradiotherapy after Non-Curative Endoscopic Resection for Superficial SCC: A Multicenter Western Study
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Population
2.2. Endoscopic Resection
2.3. Histological Definitions
2.4. Objectives and Outcomes
2.5. Follow-Up after Endoscopic Resection and Adjuvant Therapy
2.6. Statistical Analysis
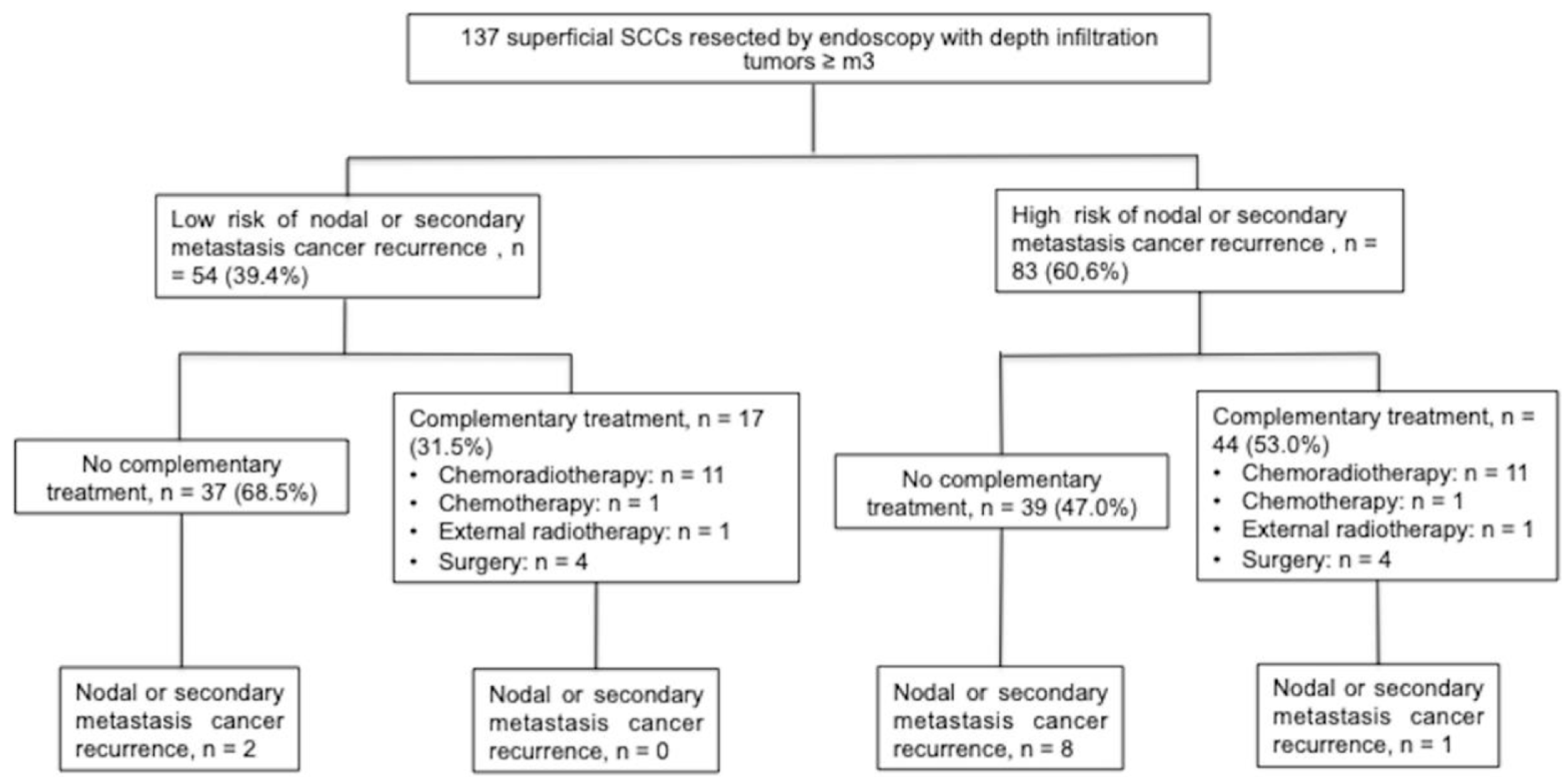
3. Results
3.1. Study Population and Tumor Characteristics
3.2. Main Outcome
3.3. Secondary Outcomes
3.3.1. Risk Factors for Nodal or Metastatic Cancer Recurrence
3.3.2. Adjuvant Treatment and Risk of Nodal or Metastatic Recurrence
3.3.3. Complications after Adjuvant Treatment
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, L.A.; Jemal, A. Global Cancer Statistics 2018: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J. Clin. 2018, 68, 394–424. [Google Scholar] [CrossRef]
- Lorenzo, D.; Barret, M.; Leblanc, S.; Terris, B.; Beuvon, F.; Coriat, R.; Chaussade, S.; Prat, F. Outcomes of Endoscopic Submucosal Dissection for Early Oesophageal Squamous Cell Neoplasia at a Western Centre. United Eur. Gastroenterol. J. 2019, 7, 1084–1092. [Google Scholar] [CrossRef]
- Berger, A.; Rahmi, G.; Perrod, G.; Pioche, M.; Canard, J.-M.; Cesbron-Métivier, E.; Boursier, J.; Samaha, E.; Vienne, A.; Lépilliez, V.; et al. Long-Term Follow-up after Endoscopic Resection for Superficial Esophageal Squamous Cell Carcinoma: A Multicenter Western Study. Endoscopy 2018, 51, 298–306. [Google Scholar] [CrossRef]
- Pimentel-Nunes, P.; Dinis-Ribeiro, M.; Ponchon, T.; Repici, A.; Vieth, M.; De Ceglie, A.; Amato, A.; Berr, F.; Bhandari, P.; Bialek, A.; et al. Endoscopic Submucosal Dissection: European Society of Gastrointestinal Endoscopy (ESGE) Guideline. Endoscopy 2015, 47, 829–854. [Google Scholar] [CrossRef]
- Bollschweiler, E.; Baldus, S.E.; Schröder, W.; Prenzel, K.; Gutschow, C.; Schneider, P.M.; Hölscher, A.H. High Rate of Lymph-Node Metastasis in Submucosal Esophageal Squamous-Cell Carcinomas and Adenocarcinomas. Endoscopy 2006, 38, 149–156. [Google Scholar] [CrossRef]
- Eguchi, T.; Nakanishi, Y.; Shimoda, T.; Iwasaki, M.; Igaki, H.; Tachimori, Y.; Kato, H.; Yamaguchi, H.; Saito, D.; Umemura, S. Histopathological Criteria for Additional Treatment after Endoscopic Mucosal Resection for Esophageal Cancer: Analysis of 464 Surgically Resected Cases. Mod. Pathol. 2006, 19, 475–480. [Google Scholar] [CrossRef]
- Tajima, Y.; Nakanishi, Y.; Tachimori, Y.; Kato, H.; Watanabe, H.; Yamaguchi, H.; Yoshimura, K.; Kusano, M.; Shimoda, T. Significance of Involvement by Squamous Cell Carcinoma of the Ducts of Esophageal Submucosal Glands. Analysis of 201 Surgically Resected Superficial Squamous Cell Carcinomas. Cancer 2000, 89, 248–254. [Google Scholar] [CrossRef] [PubMed]
- Kadota, T.; Yano, T.; Fujita, T.; Daiko, H.; Fujii, S. Submucosal Invasive Depth Predicts Lymph Node Metastasis and Poor Prognosis in Submucosal Invasive Esophageal Squamous Cell Carcinoma. Am. J. Clin. Pathol. 2017, 148, 416–426. [Google Scholar] [CrossRef] [PubMed]
- Dubecz, A.; Kern, M.; Solymosi, N.; Schweigert, M.; Stein, H.J. Predictors of Lymph Node Metastasis in Surgically Resected T1 Esophageal Cancer. Ann. Thorac. Surg. 2015, 99, 1879–1885; discussion 1886. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, K.; Hashimoto, S.; Mizuno, K.-I.; Kobayashi, T.; Tominaga, K.; Sato, H.; Kohisa, J.; Ikarashi, S.; Hayashi, K.; Takeuchi, M.; et al. Management Decision Based on Lymphovascular Involvement Leads to Favorable Outcomes after Endoscopic Treatment of Esophageal Squamous Cell Carcinoma. Endoscopy 2017, 50, 662–670. [Google Scholar] [CrossRef]
- Fuchs, H.F.; Harnsberger, C.R.; Broderick, R.C.; Chang, D.C.; Sandler, B.J.; Jacobsen, G.R.; Bouvet, M.; Horgan, S. Mortality after Esophagectomy Is Heavily Impacted by Center Volume: Retrospective Analysis of the Nationwide Inpatient Sample. Surg. Endosc. 2017, 31, 2491–2497. [Google Scholar] [CrossRef] [PubMed]
- Kato, K.; Muro, K.; Minashi, K.; Ohtsu, A.; Ishikura, S.; Boku, N.; Takiuchi, H.; Komatsu, Y.; Miyata, Y.; Fukuda, H.; et al. Phase II Study of Chemoradiotherapy with 5-Fluorouracil and Cisplatin for Stage II-III Esophageal Squamous Cell Carcinoma: JCOG Trial (JCOG 9906). Int. J. Radiat. Oncol. Biol. Phys. 2011, 81, 684–690. [Google Scholar] [CrossRef] [PubMed]
- Ikeda, A.; Hoshi, N.; Yoshizaki, T.; Fujishima, Y.; Ishida, T.; Morita, Y.; Ejima, Y.; Toyonaga, T.; Kakechi, Y.; Yokosaki, H.; et al. Endoscopic Submucosal Dissection (ESD) with Additional Therapy for Superficial Esophageal Cancer with Submucosal Invasion. Intern. Med. Tokyo Jpn. 2015, 54, 2803–2813. [Google Scholar] [CrossRef] [PubMed]
- Yoshii, T.; Ohkawa, S.; Tamai, S.; Kameda, Y. Clinical Outcome of Endoscopic Mucosal Resection for Esophageal Squamous Cell Cancer Invading Muscularis Mucosa and Submucosal Layer: EMR for ESCC with Deeper Layer. Dis. Esophagus 2013, 26, 496–502. [Google Scholar] [CrossRef] [PubMed]
- Hisano, O.; Nonoshita, T.; Hirata, H.; Sasaki, T.; Watanabe, H.; Wakiyama, H.; Ono, M.; Ohga, S.; Honda, H. Additional Radiotherapy Following Endoscopic Submucosal Dissection for T1a-MM/T1b-SM Esophageal Squamous Cell Carcinoma Improves Locoregional Control. Radiat. Oncol. Lond. Engl. 2018, 13, 14. [Google Scholar] [CrossRef]
- Naito, S.; Yoshio, T.; Ishiyama, A.; Tsuchida, T.; Tokura, J.; Namikawa, K.; Tokai, Y.; Yoshimizu, S.; Horiuchi, Y.; Hirasawa, T.; et al. Long-Term Outcomes of Esophageal Squamous Cell Carcinoma with Invasion Depth of Pathological T1a-Muscularis Mucosae and T1b-Submucosa by Endoscopic Resection Followed by Appropriate Additional Treatment. Dig. Endosc. Off. J. Jpn. Gastroenterol. Endosc. Soc. 2022, 34, 793–804. [Google Scholar] [CrossRef]
- Shimizu, Y.; Kato, M.; Yamamoto, J.; Nakagawa, S.; Tsukagoshi, H.; Fujita, M.; Hosokawa, M.; Asaka, M. EMR Combined with Chemoradiotherapy: A Novel Treatment for Superficial Esophageal Squamous-Cell Carcinoma. Gastrointest. Endosc. 2004, 59, 199–204. [Google Scholar] [CrossRef]
- Kawaguchi, G.; Sasamoto, R.; Abe, E.; Ohta, A.; Sato, H.; Tanaka, K.; Maruyama, K.; Kaizu, M.; Ayukawa, F.; Yamana, N.; et al. The Effectiveness of Endoscopic Submucosal Dissection Followed by Chemoradiotherapy for Superficial Esophageal Cancer. Radiat. Oncol. Lond. Engl. 2015, 10, 31. [Google Scholar] [CrossRef]
- Minashi, K.; Nihei, K.; Mizusawa, J.; Takizawa, K.; Yano, T.; Ezoe, Y.; Tsuchida, T.; Ono, H.; Iizuka, T.; Hanaoka, N.; et al. Efficacy of Endoscopic Resection and Selective Chemoradiotherapy for Stage I Esophageal Squamous Cell Carcinoma. Gastroenterology 2019, 157, P382–P390. [Google Scholar] [CrossRef]
- Cancer de l’œsophage|SNFGE.Org-Société Savante Médicale Française d’hepato-Gastroenterologie Et D’oncologie Digestive. Available online: https://www.snfge.org/content/1-cancer-de-loesophage#ancre3205 (accessed on 4 July 2019).
- Conroy, T.; Piessen, G.; Tougeron, D.; Créhange, G.; Lepillez, V.; Artru, P.; Drouillard, A. Cancer de l’œsophage. Thésaurus Natl. Cancérologie Dig. 2016. Available online: http://www.tncd.org (accessed on 12 February 2022).
- Dermine, S.; Lévi-Strauss, T.; Ali, E.A.; Belle, A.; Leblanc, S.; Bibault, J.-E.; Barré, A.; Palmieri, L.-J.; Brezault, C.; Dhooge, M.; et al. Organ Preservation after Endoscopic Resection of Early Esophageal Cancer with a High Risk of Lymph Node Involvement. Cancers 2020, 12, 3598. [Google Scholar] [CrossRef]
- Motoori, M.; Yano, M.; Ishihara, R.; Yamamoto, S.; Kawaguchi, Y.; Tanaka, K.; Kishi, K.; Miyashiro, I.; Fujiwara, Y.; Shingai, T.; et al. Comparison between Radical Esophagectomy and Definitive Chemoradiotherapy in Patients with Clinical T1bN0M0 Esophageal Cancer. Ann. Surg. Oncol. 2012, 19, 2135–2141. [Google Scholar] [CrossRef]
- Yoshimizu, S.; Yoshio, T.; Ishiyama, A.; Tsuchida, T.; Horiuchi, Y.; Omae, M.; Hirasawa, T.; Asari, T.; Chin, K.; Fujisaki, J. Long-Term Outcomes of Combined Endoscopic Resection and Chemoradiotherapy for Esophageal Squamous Cell Carcinoma with Submucosal Invasion. Dig. Liver Dis. Off. J. Ital. Soc. Gastroenterol. Ital. Assoc. Study Liver 2018, 50, 833–838. [Google Scholar] [CrossRef]
- Pech, O.; Gossner, L.; May, A.; Vieth, M.; Stolte, M.; Ell, C. Endoscopic Resection of Superficial Esophageal Squamous-Cell Carcinomas: Western Experience. Am. J. Gastroenterol. 2004, 99, 1226–1232. [Google Scholar] [CrossRef] [PubMed]
- Katada, C.; Muto, M.; Manabe, T.; Ohtsu, A.; Yoshida, S. Local Recurrence of Squamous-Cell Carcinoma of the Esophagus after EMR. Gastrointest. Endosc. 2005, 61, 219–225. [Google Scholar] [CrossRef]
- Yamashina, T.; Ishihara, R.; Nagai, K.; Matsuura, N.; Matsui, F.; Ito, T.; Fujii, M.; Yamamoto, S.; Hanaoka, N.; Takeuchi, Y.; et al. Long-Term Outcome and Metastatic Risk After Endoscopic Resection of Superficial Esophageal Squamous Cell Carcinoma. Am. J. Gastroenterol. 2013, 108, 544–551. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Lu, H.; Wu, L.; Yuan, B.; Liu, J.; Shi, H.; Wang, F. Long-Term Outcomes of Endoscopic Multiband Mucosectomy for Early Esophageal Squamous Cell Neoplasia: A Retrospective, Single-Center Study. Gastrointest. Endosc. 2016, 84, 893–899. [Google Scholar] [CrossRef]
- Jiang, D.; Li, X.; Wang, H.; Xu, C.; Li, X.; Sujie, A.; Zeng, H.; Hou, Y.; Zhong, Y. A Retrospective Study of Endoscopic Resection for 368 Patients with Early Esophageal Squamous Cell Carcinoma or Precancerous Lesions. Surg. Endosc. 2017, 31, 2122–2130. [Google Scholar] [CrossRef]
- Tsujii, Y.; Nishida, T.; Nishiyama, O.; Yamamoto, K.; Kawai, N.; Yamaguchi, S.; Yamada, T.; Yoshio, T.; Kitamura, S.; Nakamura, T.; et al. Clinical Outcomes of Endoscopic Submucosal Dissection for Superficial Esophageal Neoplasms: A Multicenter Retrospective Cohort Study. Endoscopy 2015, 47, 775–783. [Google Scholar] [CrossRef]
- Joo, D.C.; Kim, G.H.; Park, D.Y.; Jhi, J.H.; Song, G.A. Long-Term Outcome after Endoscopic Submucosal Dissection in Patients with Superficial Esophageal Squamous Cell Carcinoma: A Single-Center Study. Gut Liver 2014, 8, 612–618. [Google Scholar] [CrossRef]
- Probst, A.; Aust, D.; Märkl, B.; Anthuber, M.; Messmann, H. Early Esophageal Cancer in Europe: Endoscopic Treatment by Endoscopic Submucosal Dissection. Endoscopy 2014, 47, 113–121. [Google Scholar] [CrossRef]
- Park, H.C.; Kim, D.H.; Gong, E.J.; Na, H.K.; Ahn, J.Y.; Lee, J.H.; Jung, K.W.; Choi, K.D.; Song, H.J.; Lee, G.H.; et al. Ten-Year Experience of Esophageal Endoscopic Submucosal Dissection of Superficial Esophageal Neoplasms in a Single Center. Korean J. Intern. Med. 2016, 31, 1064–1072. [Google Scholar] [CrossRef] [PubMed]



| Patients | Overall (n = 137) |
|---|---|
| Male n (%) | 103 (75.2) |
| Median age (years, (range)) | 63 (35–90) |
| ASA score ≥ 3 n (%) | 71 (51.8) |
| Tumor | |
| Median size (mm, (range)) | 25 (3.0–90.0) |
| Circumference of the esophageal lumen (%) (<1/3; 1/3–3/4; >3/4) | 38.7; 39.4; 21.9 |
| Resections n (%) | |
| Method of endoscopic resection | |
| - EMR | 66 (48.2) |
| - ESD | 71 (51.8) |
| Histology n (%) | |
| SCC | 137 (100) |
| Lateral margin-free cancer (%) (R0; R1; Rx) a | 51.1; 12.4; 36.5 |
| Cancer infiltration depth | |
| - m3 | 56 (40.9) |
| - sm1 | 35 (25.5) |
| - sm2–3 | 46 (33.6) |
| Differentiation n (%) | |
| - G1 b | 95 (69.3) |
| - G2 b and G3 b | 42 (30.7) |
| Lymphovascular invasion n (%) | 19 (13.9) |
| Patient Characteristics | Complementary Treatment | |
|---|---|---|
| Yes (n = 61) | No (n = 76) | |
| Male, n (%) | 47 (77.0) | 56 (73.7) |
| Age, mean (range), years | 64.7 (44–90) | 62.8 (35–83) |
| ASA score ≥3, n (%) | 32 (52.5) | 39 (51.3) |
| Tumor size, median (range), mm | 25 (5–81) | 20 (3–90) |
| EMR resection, n (%) | 30 (49.2) | 41(53.9) |
| Tumor infiltration depth m3–sm, n (%) | 34 (55.7) | 57 (75.0) |
| Differentiation G2 and G3 †, n (%) | 22 (36.1) | 20 (26.3) |
| Lymphovascular invasion, n (%) | 11 (18.0) | 8 (10.5) |
| Risk Factors to Nodal or Distal Metastasis | Nodal or Secondary Metastasis Cancer Recurrence | Univariate Analysis | Multivariate Analysis | |
|---|---|---|---|---|
| Yes (n = 11) | No (n = 126) | p-Value 1 | Hazard Ratio (95% CI) | |
| Male n (%) | 8 (72.7) | 95 (75.5) | 0.691 | |
| Mean age, (years, (range)) | 70.2 (56–83) | 63.3 (35–90) | 0.007 | 1.075 (1.007–1.149) |
| History of head-and-neck cancer n (%) | 4 (36.4) | 51 (40.5) | 1.000 | |
| Median tumor size (mm, (range)) | 20.0 (7.0–71.5) | 25.0 (3.0–90.0) | 0.827 | |
| Tumor circumference of the esophageal lumen > 1/3 n (%) | 4 (36.4) | 80 (63.5) | 0.096 | |
| EMR resection n (%) | 7 (63.6) | 64 (50.8) | 0.667 | |
| Piecemeal resection n (%) | 6 (54.5) | 44 (34.9) | 0.295 | |
| R1 or Rx a lateral margins, n (%) | 6 (54.5) | 61 (48.4) | 0.901 | |
| Rx a depth margins, n (%) | 3 (27.3) | 24 (19.0) | 0.570 | |
| Tumor infiltration depth > Sm1 n (%) | 6 (54.5) | 40 (31.7) | 0.145 | 4.129 (1.067–15.977) |
| Differentiation G2 and G3 †, n (%) | 6 (54.5) | 36 (28.6) | 0.067 | |
| Lymphovascular invasion, n (%) | 1 (9.1) | 18 (15.7) | 0.677 | |
| No adjuvant treatment by chemoradiotherapy and/or surgery n (%) | 1 (9.1) | 60 (47.6) | 0.011 | 11.322 (1.281–100.033) |
| Median follow-up (months, (range)) | 34.0 (9–112) | 21.5 (1–135) | 0.547 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Berger, A.; Perrod, G.; Pioche, M.; Barret, M.; Cesbron-Métivier, E.; Lépilliez, V.; Hupé, M.; Perez-Cuadrado-Robles, E.; Cholet, F.; Daubigny, A.; et al. Efficacy of Organ Preservation Strategy by Adjuvant Chemoradiotherapy after Non-Curative Endoscopic Resection for Superficial SCC: A Multicenter Western Study. Cancers 2023, 15, 590. https://doi.org/10.3390/cancers15030590
Berger A, Perrod G, Pioche M, Barret M, Cesbron-Métivier E, Lépilliez V, Hupé M, Perez-Cuadrado-Robles E, Cholet F, Daubigny A, et al. Efficacy of Organ Preservation Strategy by Adjuvant Chemoradiotherapy after Non-Curative Endoscopic Resection for Superficial SCC: A Multicenter Western Study. Cancers. 2023; 15(3):590. https://doi.org/10.3390/cancers15030590
Chicago/Turabian StyleBerger, Arthur, Guillaume Perrod, Mathieu Pioche, Maximilien Barret, Elodie Cesbron-Métivier, Vincent Lépilliez, Marianne Hupé, Enrique Perez-Cuadrado-Robles, Franck Cholet, Augustin Daubigny, and et al. 2023. "Efficacy of Organ Preservation Strategy by Adjuvant Chemoradiotherapy after Non-Curative Endoscopic Resection for Superficial SCC: A Multicenter Western Study" Cancers 15, no. 3: 590. https://doi.org/10.3390/cancers15030590
APA StyleBerger, A., Perrod, G., Pioche, M., Barret, M., Cesbron-Métivier, E., Lépilliez, V., Hupé, M., Perez-Cuadrado-Robles, E., Cholet, F., Daubigny, A., Texier, C., Ali, E. A., Chabrun, E., Jacques, J., Wallenhorst, T., Chevaux, J. B., Schaefer, M., Cellier, C., & Rahmi, G. (2023). Efficacy of Organ Preservation Strategy by Adjuvant Chemoradiotherapy after Non-Curative Endoscopic Resection for Superficial SCC: A Multicenter Western Study. Cancers, 15(3), 590. https://doi.org/10.3390/cancers15030590





