P-21 Activated Kinases in Liver Disorders
Abstract
Simple Summary
Abstract
1. Introduction
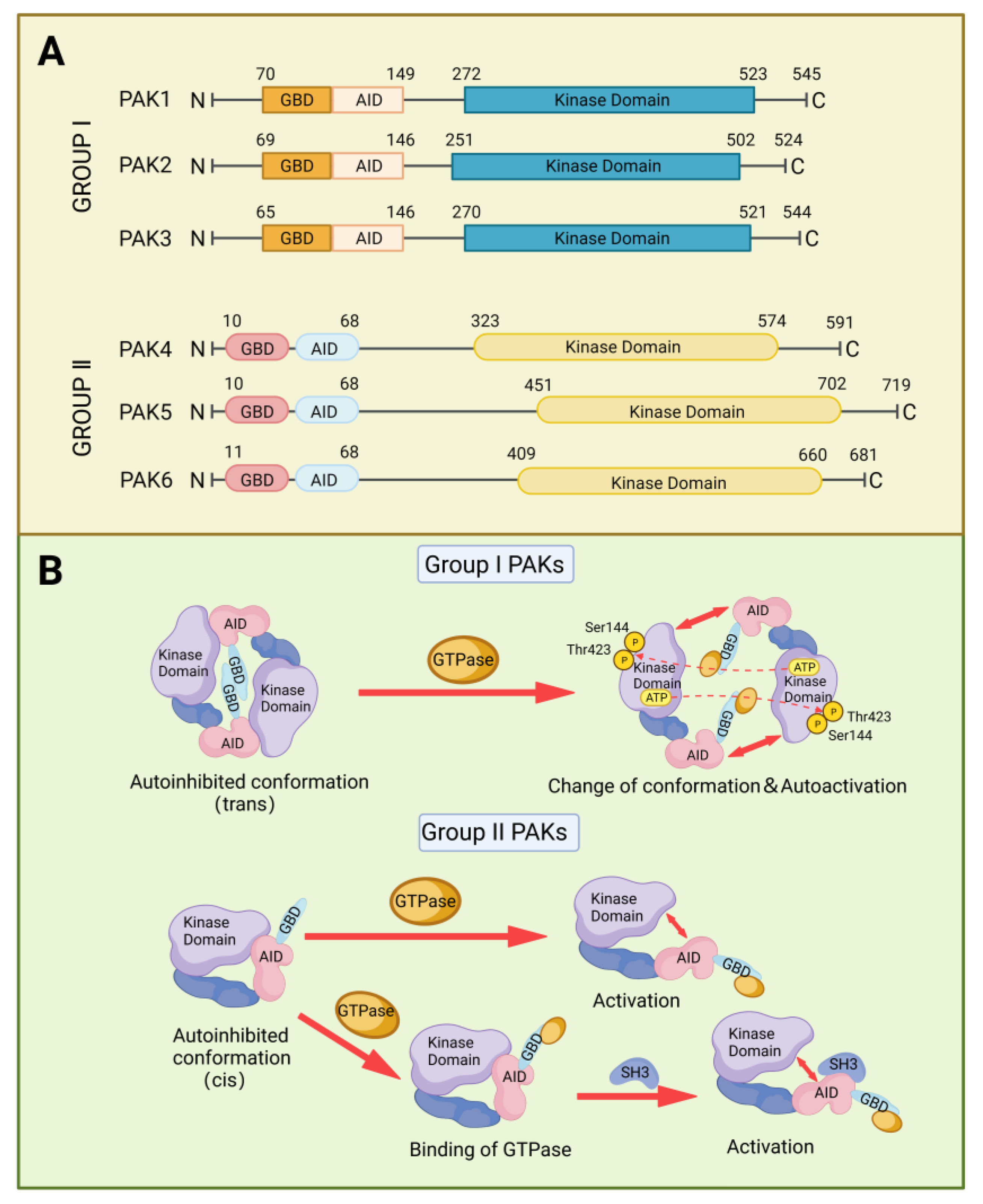
2. Structure and Activation of PAKs Family
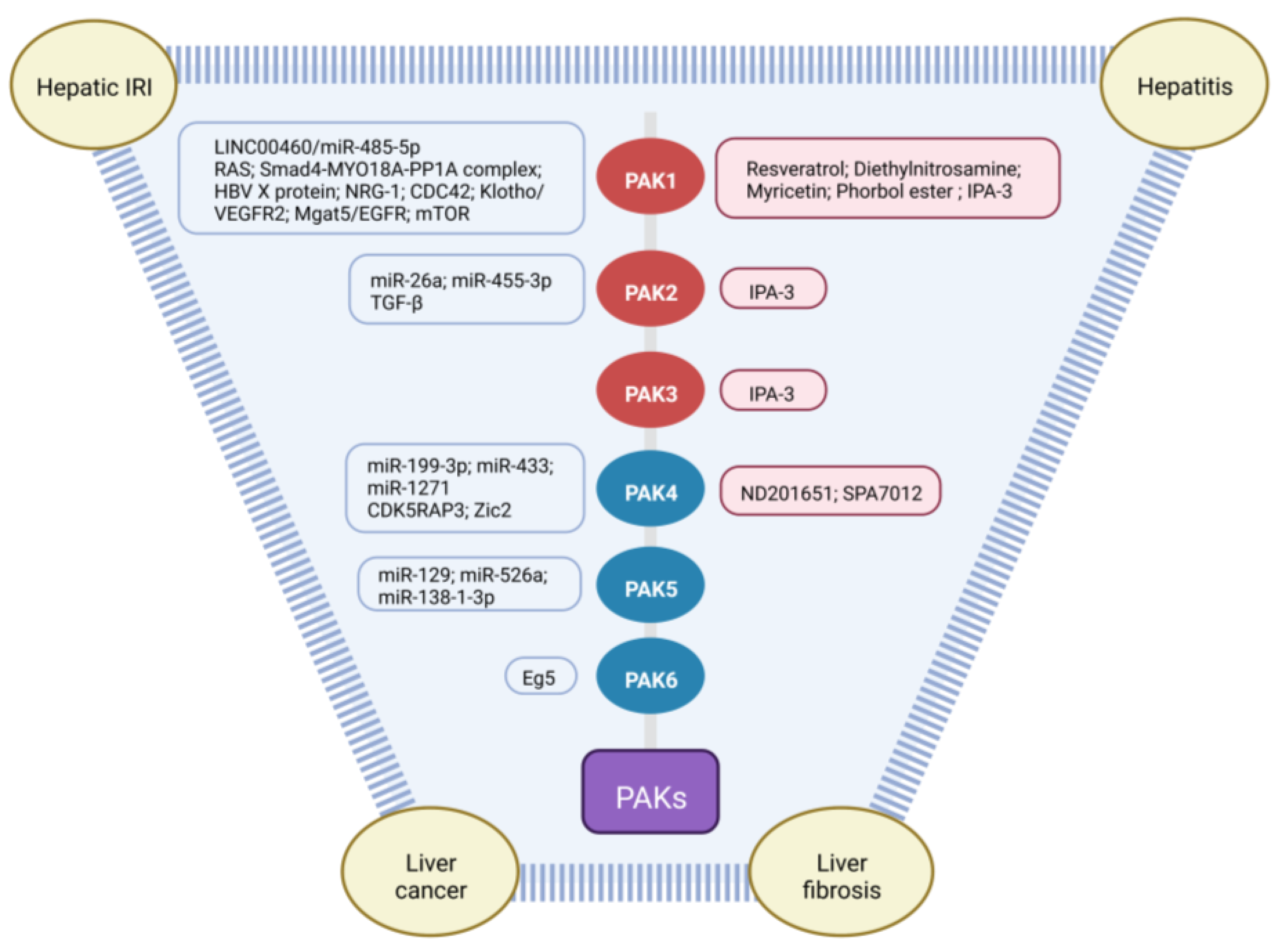
3. PAKs in Liver Cancers
3.1. Roles of Group I PAKs in Liver Cancer
3.2. Roles of Group II PAKs in Liver Cancer
4. PAKs in Hepatic Ischemia/Reperfusion Injury
5. PAKs in Hepatitis
6. PAKs in Liver Fibrosis
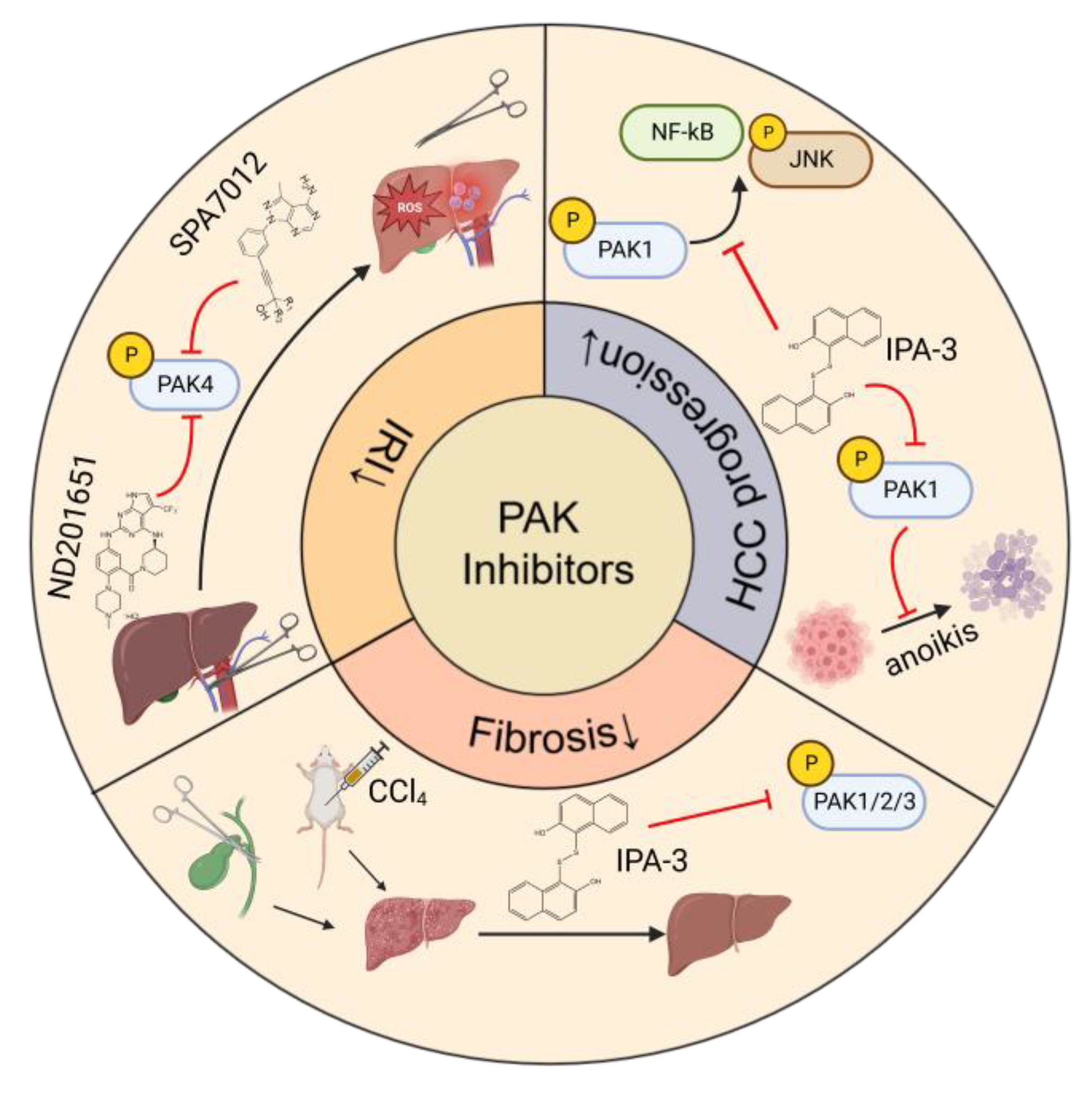
7. Therapeutic Effects of PAK Inhibitors in Liver Disorders
8. Discussion
8.1. Potential Role of PAKs in Glucolipid Metabolism of Liver
8.2. Potential Role of PAKs in Immunotherapy for Liver Disorders
9. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Rane, C.K.; Minden, A. P21 activated kinase signaling in cancer. Semin. Cancer Biol. 2019, 54, 40–49. [Google Scholar] [CrossRef] [PubMed]
- Fruman, D.A.; Chiu, H.; Hopkins, B.D.; Bagrodia, S.; Cantley, L.C.; Abraham, R.T. The PI3K Pathway in Human Disease. Cell 2017, 170, 605–635. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Sun, M.; Wang, M.; Feng, M.; Yang, F.; Li, L.; Zhao, J.; Chang, C.; Dong, H.; Xie, T.; et al. Dysregulation of Wnt/β-catenin signaling by protein kinases in hepatocellular carcinoma and its therapeutic application. Cancer Sci. 2021, 112, 1695–1706. [Google Scholar] [CrossRef]
- Zhou, Q.; Gu, T.; Zhang, Y.; Li, H.; Zhuansun, X.; Xu, S.; Kuang, Y. Human Umbilical Cord Mesenchymal Stem Cells Ameliorate Hepatic Stellate Cell Activation and Liver Fibrosis by Upregulating MicroRNA-455-3p through Suppression of p21-Activated Kinase-2. BioMed Res. Int. 2021, 2021, 6685605. [Google Scholar] [CrossRef] [PubMed]
- Ruan, P.; Dai, X.; Sun, J.; He, C.; Huang, C.; Zhou, R.; Chemin, I. Integration of hepatitis B virus DNA into p21-activated kinase 3 (PAK3) gene in HepG2.2.15 cells. Virus Genes 2020, 56, 168–173. [Google Scholar] [CrossRef] [PubMed]
- Mao, Y.; Han, C.Y.; Hao, L.; Lee, Y.; Son, J.B.; Choi, H.; Lee, M.R.; Yang, J.D.; Hong, S.K.; Suh, K.S.; et al. p21-activated kinase 4 inhibition protects against liver ischemia/reperfusion injury: Role of nuclear factor erythroid 2-related factor 2 phosphorylation. Hepatology 2022, 76, 345–356. [Google Scholar] [CrossRef] [PubMed]
- Tse, E.Y.; Ching, Y.P. The role of p21-activated kinases in hepatocellular carcinoma metastasis. J. Mol. Signal. 2014, 9, 7. [Google Scholar] [CrossRef]
- Rane, C.K.; Minden, A. P21 activated kinases: Structure, regulation, and functions. Small GTPases 2014, 5, e28003. [Google Scholar] [CrossRef]
- Liu, H.; Liu, K.; Dong, Z. The Role of p21-Activated Kinases in Cancer and Beyond: Where Are We Heading? Front. Cell Dev. Biol. 2021, 9, 41381. [Google Scholar] [CrossRef]
- Baskaran, Y.; Ng, Y.; Selamat, W.; Ling, F.T.P.; Manser, E. Group I and II mammalian PAKs have different modes of activation by Cdc42. EMBO Rep. 2012, 13, 653–659. [Google Scholar] [CrossRef]
- Ha, B.H.; Davis, M.J.; Chen, C.; Lou, H.J.; Gao, J.; Zhang, R.; Krauthammer, M.; Halaban, R.; Schlessinger, J.; Turk, B.E.; et al. Type II p21-activated kinases (PAKs) are regulated by an autoinhibitory pseudosubstrate. Proc. Natl. Acad. Sci. USA 2012, 109, 16107–16112. [Google Scholar] [CrossRef] [PubMed]
- Chan, C.H.; Chiou, L.W.; Lee, T.Y.; Liu, Y.R.; Hsieh, T.H.; Yang, C.Y.; Jeng, Y.M. PAK and PI3K pathway activation confers resistance to KRAS(G12C) inhibitor sotorasib. Br. J. Cancer 2023, 128, 148–159. [Google Scholar] [CrossRef] [PubMed]
- Higuchi, M.; Onishi, K.; Kikuchi, C.; Gotoh, Y. Scaffolding function of PAK in the PDK1–Akt pathway. Nature 2008, 10, 1356–1364. [Google Scholar] [CrossRef] [PubMed]
- King, H.; Thillai, K.; Whale, A.; Arumugam, P.; Eldaly, H.; Kocher, H.M.; Wells, C.M. PAK4 interacts with p85 alpha: Implications for pancreatic cancer cell migration. Sci. Rep. 2017, 7, 2575. [Google Scholar] [CrossRef]
- Park, S.Y.; An, J.M.; Seo, J.T.; Seo, S.R. Y-27632 Induces Neurite Outgrowth by Activating the NOX1-Mediated AKT and PAK1 Phosphorylation Cascades in PC12 Cells. Int. J. Mol. Sci. 2020, 21, 7679. [Google Scholar] [CrossRef]
- Fu, X.; Feng, J.; Zeng, D.; Ding, Y.; Yu, C.; Yang, B. PAK4 confers cisplatin resistance in gastric cancer cells via PI3K/Akt- and MEK/ERK-dependent pathways. Biosci. Rep. 2014, 34, e00094. [Google Scholar] [CrossRef]
- Huang, N.; Li, S.; Xie, Y.; Han, Q.; Xu, X.M.; Sheng, Z.H. Reprogramming an energetic AKT-PAK5 axis boosts axon energy supply and facilitates neuron survival and regeneration after injury and ischemia. Curr. Biol. 2021, 31, 3098–3114. [Google Scholar] [CrossRef]
- Beeser, A.; Jaffer, Z.M.; Hofmann, C.; Chernoff, J. Role of Group A p21-activated Kinases in Activation of Extracellular-regulated Kinase by Growth Factors. J. Biol. Chem. 2005, 280, 36609–36615. [Google Scholar] [CrossRef]
- Frost, J.A.; Swantek, J.L.; Stippec, S.; Yin, M.J.; Gaynor, R.; Cobb, M.H. Stimulation of NFkappa B activity by multiple signaling pathways requires PAK1. J. Biol. Chem. 2000, 275, 19693–19699. [Google Scholar] [CrossRef]
- Zhu, M.; Xu, Y.; Zhang, W.; Gu, T.; Wang, D. Inhibition of PAK1 alleviates cerulein-induced acute pancreatitis via p38 and NF-κB pathways. Biosci. Rep. 2019, 39, BSR20182221. [Google Scholar] [CrossRef]
- Wong, L.L.; Lam, I.P.; Wong, T.Y.; Lai, W.L.; Liu, H.F.; Yeung, L.L.; Ching, Y.P. IPA-3 inhibits the growth of liver cancer cells by suppressing PAK1 and NF-κB activation. PLoS ONE 2013, 8, e68843. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Ren, G.; Li, K.; Liu, Z.; Wang, Y.; Chen, T.; Mu, W.; Yang, X.; Li, X.; Shi, A.; et al. The Smad4-MYO18A-PP1A complex regulates β-catenin phosphorylation and pemigatinib resistance by inhibiting PAK1 in cholangiocarcinoma. Cell Death Differ. 2022, 29, 818–831. [Google Scholar] [CrossRef]
- Yi, Y.; Li, P.; Huang, Y.; Chen, D.; Fan, S.; Wang, J.; Yang, M.; Zeng, S.; Deng, J.; Lv, X.; et al. P21-activated kinase 2-mediated β-catenin signaling promotes cancer stemness and osimertinib resistance in EGFR-mutant non-small-cell lung cancer. Oncogene 2022, 41, 4318–4329. [Google Scholar] [CrossRef] [PubMed]
- Li, T.T.; Mou, J.; Pan, Y.J.; Huo, F.C.; Du, W.Q.; Liang, J.; Wang, Y.; Zhang, L.S.; Pei, D.S. MicroRNA-138-1-3p sensitizes sorafenib to hepatocellular carcinoma by targeting PAK5 mediated β-catenin/ABCB1 signaling pathway. J. Biomed. Sci. 2021, 28, 56. [Google Scholar] [CrossRef]
- Li, Y.; Shao, Y.; Tong, Y.; Shen, T.; Zhang, J.; Li, Y.; Gu, H.; Li, F. Nucleo-cytoplasmic shuttling of PAK4 modulates β-catenin intracellular translocation and signaling. Biochim. Biophys. Acta (BBA)—Mol. Cell Res. 2012, 1823, 465–475. [Google Scholar] [CrossRef] [PubMed]
- Vershinin, Z.; Feldman, M.; Chen, A.; Levy, D. PAK4 Methylation by SETD6 Promotes the Activation of the Wnt/β-Catenin Pathway. J. Biol. Chem. 2016, 291, 6786–6795. [Google Scholar] [CrossRef] [PubMed]
- Bastea, L.I.; Döppler, H.; Pearce, S.E.; Durand, N.; Spratley, S.J.; Storz, P. Protein kinase D-mediated phosphorylation at Ser99 regulates localization of p21-activated kinase 4. Biochem. J. 2013, 455, 251–260. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Hirose, H.; Du, G.; Chong, K.; Kiyohara, E.; Witz, I.P.; Hoon, D.S.B. P-REX1 amplification promotes progression of cutaneous melanoma via the PAK1/P38/MMP-2 pathway. Cancer Lett. 2017, 407, 6–75. [Google Scholar] [CrossRef]
- Zhang, K.; Wang, Y.; Fan, T.; Zeng, C.; Sun, Z.S. The p21-activated kinases in neural cytoskeletal remodeling and related neurological disorders. Protein Cell 2022, 13, 6–25. [Google Scholar] [CrossRef]
- Wang, Y.; Wang, S.; Lei, M.; Boyett, M.; Tsui, H.; Liu, W.; Wang, X. The p21-activated kinase 1 (Pak1) signalling pathway in cardiac disease: From mechanistic study to therapeutic exploration. Br. J. Pharmacol. 2017, 175, 1362–1374. [Google Scholar] [CrossRef]
- Ramos-Alvarez, I.; Jensen, R.T. P21-activated kinase 4 in pancreatic acinar cells is activated by numerous gastrointestinal hormones/neurotransmitters and growth factors by novel signaling, and its activation stimulates secretory/growth cascades. Am. J. Physiol. Gastrointest. Liver Physiol. 2018, 315, G302–G317. [Google Scholar] [CrossRef] [PubMed]
- Dammann, K.; Khare, V.; Coleman, C.; Berdel, H.; Gasche, C. p-21 Activated Kinase as a Molecular Target for Chemoprevention in Diabetes. Geriatrics 2018, 3, 73. [Google Scholar] [CrossRef] [PubMed]
- El-Serag, H.B.; Rudolph, K.L. Hepatocellular carcinoma: Epidemiology and molecular carcinogenesis. Gastroenterology 2007, 132, 2557–2576. [Google Scholar] [CrossRef]
- McGlynn, K.A.; London, W.T. The global epidemiology of hepatocellular carcinoma: Present and future. Clin. Liver Dis. 2011, 15, 223–243. [Google Scholar] [CrossRef]
- Parekh, P.; Rao, K.V. Overexpression of cyclin D1 is associated with elevated levels of MAP kinases, Akt and Pak1 during diethylnitrosamine-induced progressive liver carcinogenesis. Cell Biol. Int. 2007, 31, 35–43. [Google Scholar] [CrossRef]
- Razavi, H.; Waked, I.; Sarrazin, C.; Myers, R.P.; Idilman, R.; Calinas, F.; Vogel, W.; Mendes Correa, M.C.; Hézode, C.; Lázaro, P.; et al. The present and future disease burden of hepatitis C virus (HCV) infection with today’s treatment paradigm. J. Viral Hepat. 2014, 21 (Suppl. 1), 4–59. [Google Scholar] [CrossRef]
- Best, M.; Gale, M.E.; Wells, C.M. PAK-dependent regulation of actin dynamics in breast cancer cells. Int. J. Biochem. Cell Biol. 2022, 146, 06207. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Zhang, C.; Zheng, Q.; Ma, Z.; Qi, M.; Di, G.; Ling, S.; Xu, H.; Qi, B.; Yao, C.; et al. RhoJ facilitates angiogenesis in glioblastoma via JNK/VEGFR2 mediated activation of PAK and ERK signaling pathways. Int. J. Biol. Sci. 2022, 18, 942–955. [Google Scholar] [CrossRef] [PubMed]
- Lei, K.; Luo, M.; Tu, Z.; Lv, S.; Liu, J.; Gong, C.; Ye, M.; Wu, M.; Sheng, Y.; Long, X.; et al. Comprehensive analysis of the prognostic implications and functional exploration of PAK gene family in human cancer. Cancer Cell Int. 2022, 22, 275. [Google Scholar] [CrossRef]
- Yu, X.; Huang, C.; Liu, J.; Shi, X.; Li, X. The significance of PAK4 in signaling and clinicopathology: A review. Open Life Sci. 2022, 17, 586–598. [Google Scholar] [CrossRef]
- Kumar, R.; Li, D.Q. PAKs in Human Cancer Progression: From Inception to Cancer Therapeutic to Future Oncobiology. Adv. Cancer Res. 2016, 130, 37–209. [Google Scholar] [CrossRef]
- Cao, F.; Yin, L.X. PAK1 promotes proliferation, migration and invasion of hepatocellular carcinoma by facilitating EMT via directly up-regulating Snail. Genomics 2020, 112, 694–702. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.-L.; Liu, G.-C.; Peng, L.; Zhang, C.; Jia, Y.-M.; Yang, W.-H.; Mao, L. Effect of PAK1 gene silencing on proliferation and apoptosis in hepatocellular carcinoma cell lines MHCC97-H and HepG2 and cells in xenograft tumor. Gene Ther. 2018, 25, 284–296. [Google Scholar] [CrossRef]
- Ching, Y.P.; Leong, V.Y.; Lee, M.F.; Xu, H.T.; Jin, D.Y.; Ng, I.O. P21-activated protein kinase is overexpressed in hepatocellular carcinoma and enhances cancer metastasis involving c-Jun NH2-terminal kinase activation and paxillin phosphorylation. Cancer Res. 2007, 67, 3601–3608. [Google Scholar] [CrossRef] [PubMed]
- Gujdár, A.; Sipeki, S.; Bander, E.; Buday, L.; Faragó, A. Phorbol ester-induced migration of HepG2 cells is accompanied by intensive stress fibre formation, enhanced integrin expression and transient down-regulation of p21-activated kinase 1. Cell. Signal. 2003, 15, 307–318. [Google Scholar] [CrossRef]
- Zhang, J.G.; Zhou, H.M.; Zhang, X.; Mu, W.; Hu, J.N.; Liu, G.L.; Li, Q. Hypoxic induction of vasculogenic mimicry in hepatocellular carcinoma: Role of HIF-1 α, RhoA/ROCK and Rac1/PAK signaling. BMC Cancer 2020, 20, 32. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Liu, H.; Liu, J.; Zhu, Y.; Xu, L.; He, H.; Zhang, H.; Wang, S.; Wu, Q.; Liu, W.; et al. Klotho endows hepatoma cells with resistance to anoikis via VEGFR2/PAK1 activation in hepatocellular carcinoma. PLoS ONE 2013, 8, e58413. [Google Scholar] [CrossRef]
- Liu, J.; Liu, H.; Zhang, W.; Wu, Q.; Liu, W.; Liu, Y.; Pan, D.; Xu, J.; Gu, J. N-acetylglucosaminyltransferase V confers hepatoma cells with resistance to anoikis through EGFR/PAK1 activation. Glycobiology 2013, 23, 1097–1109. [Google Scholar] [CrossRef]
- Parekh, P.; Motiwale, L.; Naik, N.; Rao, K.V. Downregulation of cyclin D1 is associated with decreased levels of p38 MAP kinases, Akt/PKB and Pak1 during chemopreventive effects of resveratrol in liver cancer cells. Exp. Toxicol. Pathol. 2011, 63, 67–173. [Google Scholar] [CrossRef]
- Iyer, S.C.; Gopal, A.; Halagowder, D. Myricetin induces apoptosis by inhibiting P21 activated kinase 1 (PAK1) signaling cascade in hepatocellular carcinoma. Mol. Cell. Biochem. 2015, 407, 223–237. [Google Scholar] [CrossRef]
- Janardhan, H.P.; Meng, X.; Dresser, K.; Hutchinson, L.; Trivedi, C.M. KRAS or BRAF mutations cause hepatic vascular cavernomas treatable with MAP2K-MAPK1 inhibition. J. Exp. Med. 2020, 217, e20192205. [Google Scholar] [CrossRef]
- Chen, L.; Bi, S.; Hou, J.; Zhao, Z.; Wang, C.; Xie, S. Targeting p21-activated kinase 1 inhibits growth and metastasis via Raf1/MEK1/ERK signaling in esophageal squamous cell carcinoma cells. Cell Commun. Signal. 2019, 17, 31. [Google Scholar] [CrossRef] [PubMed]
- Cheng, C.; Kong, X.; Wang, H.; Gan, H.; Hao, Y.; Zou, W.; Wu, J.; Chi, Y.; Yang, J.; Hong, Y.; et al. Trihydrophobin 1 Interacts with PAK1 and Regulates ERK/MAPK Activation and Cell Migration. J. Biol. Chem. 2009, 284, 8786–8796. [Google Scholar] [CrossRef] [PubMed]
- Tu, J.; Zhao, Z.; Xu, M.; Chen, M.; Weng, Q.; Ji, J. LINC00460 promotes hepatocellular carcinoma development through sponging miR-485-5p to up-regulate PAK1. Biomed. Pharmacother. 2019, 118, 109213. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Fan, R.G.; Qin, C.L.; Jia, J.; Wu, X.D.; Zha, W.Z. LncRNA-H19 activates CDC42/PAK1 pathway to promote cell proliferation, migration and invasion by targeting miR-15b in hepatocellular carcinoma. Genomics 2019, 111, 1862–1872. [Google Scholar] [CrossRef]
- Xu, J.; Liu, H.; Chen, L.; Wang, S.; Zhou, L.; Yun, X.; Sun, L.; Wen, Y.; Gu, J. Hepatitis B Virus X Protein Confers Resistance of Hepatoma Cells to Anoikis by Up-regulating and Activating p21-Activated Kinase 1. Gastroenterology 2012, 143, 199–212.e4. [Google Scholar] [CrossRef]
- Zhang, X.; Zhang, X.; Wang, T.; Wang, L.; Tan, Z.; Wei, W.; Yan, B.; Zhao, J.; Wu, K.; Yang, A.; et al. MicroRNA-26a is a key regulon that inhibits progression and metastasis of c-Myc/EZH2 double high advanced hepatocellular carcinoma. Cancer Lett. 2018, 426, 8–108. [Google Scholar] [CrossRef]
- Sato, M.; Matsuda, Y.; Wakai, T.; Kubota, M.; Osawa, M.; Fujimaki, S.; Sanpei, A.; Takamura, M.; Yamagiwa, S.; Aoyagi, Y. p21-activated kinase-2 is a critical mediator of transforming growth factor-β-induced hepatoma cell migration. J. Gastroenterol. Hepatol. 2013, 28, 1047–1055. [Google Scholar] [CrossRef]
- Koth, A.P.; Oliveira, B.R.; Parfitt, G.M.; Buonocore Jde, Q.; Barros, D.M. Participation of group I p21-activated kinases in neuroplasticity. J. Physiol. 2014, 108, 70–277. [Google Scholar] [CrossRef]
- Gao, Z.; Zhong, M.; Ye, Z.; Wu, Z.; Xiong, Y.; Ma, J.; Chen, H.; Zhu, Y.; Yang, Y.; Zhao, Y.; et al. PAK3 promotes the metastasis of hepatocellular carcinoma by regulating EMT process. J. Cancer 2022, 13, 153–161. [Google Scholar] [CrossRef]
- Li, Q.; Zhang, X.; Wei, N.; Liu, S.; Ling, Y.; Wang, H. p21-activated kinase 4 as a switch between caspase-8 apoptosis and NF-κB survival signals in response to TNF-α in hepatocarcinoma cells. Biochem. Biophys. Res. Commun. 2018, 503, 3003–3010. [Google Scholar] [CrossRef] [PubMed]
- Callegari, E.; D’Abundo, L.; Guerriero, P.; Simioni, C.; Elamin, B.K.; Russo, M.; Cani, A.; Bassi, C.; Zagatti, B.; Giacomelli, L.; et al. miR-199a-3p Modulates MTOR and PAK4 Pathways and Inhibits Tumor Growth in a Hepatocellular Carcinoma Transgenic Mouse Model. Mol. Ther. Nucleic Acids 2018, 11, 85–493. [Google Scholar] [CrossRef] [PubMed]
- Hou, J.; Lin, L.; Zhou, W.; Wang, Z.; Ding, G.; Dong, Q.; Qin, L.; Wu, X.; Zheng, Y.; Yang, Y.; et al. Identification of miRNomes in human liver and hepatocellular carcinoma reveals miR-199a/b-3p as therapeutic target for hepatocellular carcinoma. Cancer Cell 2011, 19, 232–243. [Google Scholar] [CrossRef] [PubMed]
- Mak, G.W.; Chan, M.M.; Leong, V.Y.; Lee, J.M.; Yau, T.O.; Ng, I.O.; Ching, Y.P. Overexpression of a novel activator of PAK4, the CDK5 kinase-associated protein CDK5RAP3, promotes hepatocellular carcinoma metastasis. Cancer Res. 2011, 71, 2949–2958. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.T.; Lai, W.L.; Liu, H.F.; Wong, L.L.; Ng, I.O.; Ching, Y.P. PAK4 Phosphorylates p53 at Serine 215 to Promote Liver Cancer Metastasis. Cancer Res. 2016, 76, 5732–5742. [Google Scholar] [CrossRef] [PubMed]
- Lu, S.X.; Zhang, C.Z.; Luo, R.Z.; Wang, C.H.; Liu, L.L.; Fu, J.; Zhang, L.; Wang, H.; Xie, D.; Yun, J.P. Zic2 promotes tumor growth and metastasis via PAK4 in hepatocellular carcinoma. Cancer Lett. 2017, 402, 1–80. [Google Scholar] [CrossRef] [PubMed]
- Xue, J.; Chen, L.Z.; Li, Z.Z.; Hu, Y.Y.; Yan, S.P.; Liu, L.Y. MicroRNA-433 inhibits cell proliferation in hepatocellular carcinoma by targeting p21 activated kinase (PAK4). Mol. Cell. Biochem. 2015, 399, 7–86. [Google Scholar] [CrossRef]
- Shao, S.; Hu, Q.; Wu, W.; Wang, M.; Huang, J.; Zhao, X.; Tang, G.; Liang, T. Tumor-triggered personalized microRNA cocktail therapy for hepatocellular carcinoma. Biomater. Sci. 2020, 8, 6579–6591. [Google Scholar] [CrossRef]
- Zhang, D.G.; Gong, C.C.; Wu, X.J.; Ren, X.; Sedaka, R.S.; Chen, W.C.; Huo, F.C.; Chen, C.; Du, W.Q.; Pei, D.S. P21-activated kinase 5 potentiates the chemoresistant phenotype of liver cancer. Signal Transduct. Target. Ther. 2021, 6, 47. [Google Scholar] [CrossRef]
- Zhai, J.; Qu, S.; Li, X.; Zhong, J.; Chen, X.; Qu, Z.; Wu, D. miR-129 suppresses tumor cell growth and invasion by targeting PAK5 in hepatocellular carcinoma. Biochem. Biophys. Res. Commun. 2015, 464, 161–167. [Google Scholar] [CrossRef]
- Zhan, L.; Pan, Y.; Chen, L.; Chen, Z.; Zhang, H.; Sun, C. MicroRNA-526a targets p21-activated kinase 7 to inhibit tumorigenesis in hepatocellular carcinoma. Mol. Med. Rep. 2017, 16, 837–844. [Google Scholar] [CrossRef] [PubMed]
- Fang, Z.P.; Jiang, B.G.; Gu, X.F.; Zhao, B.; Ge, R.L.; Zhang, F.B. P21-activated kinase 5 plays essential roles in the proliferation and tumorigenicity of human hepatocellular carcinoma. Acta Pharmacol. Sin. 2014, 35, 82–88. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.G.; Zhang, J.; Mao, L.L.; Wu, J.X.; Cao, W.J.; Zheng, J.N.; Pei, D.S. p21-Activated kinase 5 affects cisplatin-induced apoptosis and proliferation in hepatocellular carcinoma cells. Tumour. Biol. 2015, 36, 3685–3691. [Google Scholar] [CrossRef] [PubMed]
- Gu, Y.F.; Kong, L.T. Inhibiting p21-activated kinase (PAK7) enhances radiosensitivity in hepatocellular carcinoma. Hum. Exp. Toxicol. 2021, 40, 2202–2214. [Google Scholar] [CrossRef]
- Chen, H.; Miao, J.; Li, H.; Wang, C.; Li, J.; Zhu, Y.; Wang, J.; Wu, X.; Qiao, H. Expression and prognostic significance of p21-activated kinase 6 in hepatocellular carcinoma. J. Surg. Res. 2014, 189, 81–88. [Google Scholar] [CrossRef]
- Zheng, J.; Zhang, C.; Li, Y.; Jiang, Y.; Xing, B.; Du, X. p21-activated kinase 6 controls mitosis and hepatocellular carcinoma progression by regulating Eg5. Biochim. Biophys. Acta (BBA)—Mol. Cell Res. 2020, 1868, 118888. [Google Scholar] [CrossRef]
- Liu, W.; Liu, Y.; Liu, H.; Zhang, W.; Fu, Q.; Xu, J.; Gu, J. Tumor Suppressive Function of p21-activated Kinase 6 in Hepatocellular Carcinoma. J. Biol. Chem. 2015, 290, 28489–28501. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Zhang, J.; Gong, N. Role of the PI3K/Akt signaling pathway in liver ischemia reperfusion injury: A narrative review. Ann. Palliat. Med. 2022, 11, 806–817. [Google Scholar] [CrossRef]
- Dery, K.J.; Kupiec-Weglinski, J.W. New insights into ischemia-reperfusion injury signaling pathways in organ transplantation. Curr. Opin. Organ Transplant. 2022, 27, 424–433. [Google Scholar] [CrossRef]
- Gao, F.; Qiu, X.; Wang, K.; Shao, C.; Jin, W.; Zhang, Z.; Xu, X. Targeting the Hepatic Microenvironment to Improve Ischemia/Reperfusion Injury: New Insights into the Immune and Metabolic Compartments. Aging Dis. 2022, 13, 1196–1214. [Google Scholar] [CrossRef]
- Ma, Y.; Nikfarjam, M.; He, H. The trilogy of P21 activated kinase, autophagy and immune evasion in pancreatic ductal adenocarcinoma. Cancer Lett. 2022, 24, 215868. [Google Scholar] [CrossRef] [PubMed]
- Wang, K.; Zhan, Y.; Huynh, N.; Dumesny, C.; Wang, X.; Asadi, K.; Herrmann, D.; Timpson, P.; Yang, Y.; Walsh, K.; et al. Inhibition of PAK1 suppresses pancreatic cancer by stimulation of anti-tumour immunity through down-regulation of PD-L1. Cancer Lett. 2020, 472, 8–18. [Google Scholar] [CrossRef] [PubMed]
- Micó-Carnero, M.; Casillas-Ramírez, A.; Sánchez-González, A.; Rojano-Alfonso, C.; Peralta, C. The Role of Neuregulin-1 in Steatotic and Non-Steatotic Liver Transplantation from Brain-Dead Donors. Biomedicines 2022, 10, 978. [Google Scholar] [CrossRef]
- Castaneda, D.; Gonzalez, A.J.; Alomari, M.; Tandon, K.; Zervos, X.B. From hepatitis A to E: A critical review of viral hepatitis. World J. Gastroenterol. 2021, 27, 1691–1715. [Google Scholar] [CrossRef]
- Hosseini, N.; Shor, J.; Szabo, G. Alcoholic Hepatitis: A Review. Alcohol Alcohol. 2019, 54, 408–416. [Google Scholar] [CrossRef]
- Sucher, E.; Sucher, R.; Gradistanac, T.; Brandacher, G.; Schneeberger, S.; Berg, T. Autoimmune Hepatitis—Immunologically Triggered Liver Pathogenesis—Diagnostic and Therapeutic Strategies. J. Immunol. Res. 2019, 2019, 9437043. [Google Scholar] [CrossRef]
- Targher, G.; Tilg, H.; Byrne, C.D. Non-alcoholic fatty liver disease: A multisystem disease requiring a multidisciplinary and holistic approach. Lancet Gastroenterol. Hepatol. 2021, 6, 578–588. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Siddiqui, A. Hepatitis B Virus X Protein Stimulates the Mitochondrial Translocation of Raf-1 via Oxidative Stress. J. Virol. 2007, 81, 6757–6760. [Google Scholar] [CrossRef]
- Yu, F.X.; Zhang, X.L.; Wang, Y.P.; Ma, N.; Du, H.; Ma, J.M.; Liu, D.W. Gene polymorphisms of interleukin-28, p21-activated protein kinases 4, and response to interferon-α based therapy in Chinese patients with chronic hepatitis B. Chin. Med. J. (Engl.) 2013, 126, 1726–1731. [Google Scholar]
- Ma, N.; Zhang, X.; Yu, F.; Gao, P.; Fan, Q.; Liu, L.; Liu, D. Role of IFN-ks, IFN-ks related genes and the DEPDC5 gene in Hepatitis B virus-related liver disease. J. Viral Hepat. 2014, 21, e29–e38. [Google Scholar] [CrossRef]
- Ishida, H.; Li, K.; Yi, M.; Lemon, S.M. p21-activated kinase 1 is activated through the mammalian target of rapamycin/p70 S6 kinase pathway and regulates the replication of hepatitis C virus in human hepatoma cells. J. Biol. Chem. 2007, 282, 11836–11848. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, H.; Sankaran, S.; Dandekar, S. Hepatitis C virus core protein induces expression of genes regulating immune evasion and anti-apoptosis in hepatocytes. Virology 2006, 354, 58–68. [Google Scholar] [CrossRef] [PubMed]
- Fan, M.; Luo, Y.; Zhang, B.; Wang, J.; Chen, T.; Liu, B.; Sun, Y.; Nan, Y.; Hiscox, J.A.; Zhao, Q.; et al. Cell Division Control Protein 42 Interacts With Hepatitis E Virus Capsid Protein and Participates in Hepatitis E Virus Infection. Front. Microbiol. 2021, 12, 75083. [Google Scholar] [CrossRef] [PubMed]
- Zhang, B.; Fan, M.; Fan, J.; Luo, Y.; Wang, J.; Wang, Y.; Liu, B.; Sun, Y.; Zhao, Q.; Hiscox, J.A.; et al. Avian Hepatitis E Virus ORF2 Protein Interacts with Rap1b to Induce Cytoskeleton Rearrangement That Facilitates Virus Internalization. Microbiol. Spectr. 2022, 10, e0226521. [Google Scholar] [CrossRef]
- Chang, H.; He, K.Y.; Li, C.; Ni, Y.Y.; Li, M.N.; Chen, L.; Hou, M.; Zhou, Z.; Xu, Z.P.; Ji, M.J. P21 activated kinase-1 (PAK1) in macrophages is required for promotion of Th17 cell response during helminth infection. J. Cell. Mol. Med. 2020, 24, 14325–14338. [Google Scholar] [CrossRef]
- Lin, R.; Zhang, J.; Zhou, L.; Wang, B. Altered function of monocytes/macrophages in patients with autoimmune hepatitis. Mol. Med. Rep. 2016, 13, 3874–3880. [Google Scholar] [CrossRef]
- Afifiyan, N.; Tillman, B.; French, B.A.; Sweeny, O.; Masouminia, M.; Samadzadeh, S.; French, S.W. The role of Tec kinase signaling pathways in the development of Mallory Denk Bodies in balloon cells in alcoholic hepatitis. Exp. Mol. Pathol. 2017, 103, 191–199. [Google Scholar] [CrossRef]
- Kisseleva, T.; Brenner, D. Molecular and cellular mechanisms of liver fibrosis and its regression. Nat. Rev. Gastroenterol. Hepatol. 2020, 18, 151–166. [Google Scholar] [CrossRef]
- Roehlen, N.; Crouchet, E.; Baumert, T.F. Liver Fibrosis: Mechanistic Concepts and Therapeutic Perspectives. Cells 2020, 9, 875. [Google Scholar] [CrossRef]
- Martin, K.; Pritchett, J.; Llewellyn, J.; Mullan, A.F.; Athwal, V.S.; Dobie, R.; Harvey, E.; Zeef, L.; Farrow, S.; Streuli, C.; et al. PAK proteins and YAP-1 signalling downstream of integrin beta-1 in myofibroblasts promote liver fibrosis. Nat. Commun. 2016, 7, 2502. [Google Scholar] [CrossRef]
- Romermann, D.; Ansari, N.; Schultz-Moreira, A.R.; Michael, A.; Marhenke, S.; Hardtke-Wolenski, M.; Longerich, T.; Manns, M.P.; Wedemeyer, H.; Vogel, A.; et al. Absence of Atg7 in the liver disturbed hepatic regeneration after liver injury. Liver Int. 2020, 40, 1225–1238. [Google Scholar] [CrossRef] [PubMed]
- Odenwald, M.A.; Paul, S. Viral hepatitis: Past, present, and future. World J. Gastroenterol. 2022, 28, 1405–1429. [Google Scholar] [CrossRef] [PubMed]
- Fanning, G.C.; Zoulim, F.; Hou, J.; Bertoletti, A. Therapeutic strategies for hepatitis B virus infection: Towards a cure. Nat. Rev. Drug Discov. 2019, 18, 827–844. [Google Scholar] [CrossRef]
- Powell, E.E.; Wong, V.W.; Rinella, M. Non-alcoholic fatty liver disease. Lancet 2021, 397, 2212–2224. [Google Scholar] [CrossRef] [PubMed]
- Ganne-Carrié, N.; Nahon, P. Hepatocellular carcinoma in the setting of alcohol-related liver disease. J. Hepatol. 2019, 70, 284–293. [Google Scholar] [CrossRef] [PubMed]
- Fan, Y.; Xue, H.; Zheng, H. Systemic Therapy for Hepatocellular Carcinoma: Current Updates and Outlook. J. Hepatocell. Carcinoma 2022, 9, 233–263. [Google Scholar] [CrossRef] [PubMed]
- Kudo, M.; Finn, R.S.; Qin, S.; Han, K.H.; Ikeda, K.; Piscaglia, F.; Baron, A.; Park, J.W.; Han, G.; Jassem, J.; et al. Lenvatinib versus sorafenib in first-line treatment of patients with unresectable hepatocellular carcinoma: A randomised phase 3 non-inferiority trial. Lancet 2018, 391, 1163–1173. [Google Scholar] [CrossRef]
- Chakraborty, E.; Sarkar, D. Emerging Therapies for Hepatocellular Carcinoma (HCC). Cancers 2022, 14, 2798. [Google Scholar] [CrossRef]
- Yao, D.; Li, C.; Rajoka, M.S.R.; He, Z.; Huang, J.; Wang, J.; Zhang, J. P21-Activated Kinase 1: Emerging biological functions and potential therapeutic targets in Cancer. Theranostics 2020, 10, 9741–9766. [Google Scholar] [CrossRef]
- Rudolph, J.; Crawford, J.J.; Hoeflich, K.P.; Chernoff, J. p21-activated kinase inhibitors. Enzymes 2013, 34, 57–180. [Google Scholar]
- Amirthalingam, M.; Palanisamy, S.; Tawata, S. p21-Activated kinase 1 (PAK1) in aging and longevity: An overview. Ageing Res. Rev. 2021, 71, 101443. [Google Scholar] [CrossRef]
- Mao, Y.; Lee, E.; Yang, X.; Bae, E.J.; Jeon, R.; Park, B.H. Targeting p21-activated kinase 4 (PAK4) with pyrazolo[3,4-d]pyrimidine derivative SPA7012 attenuates hepatic ischaemia-reperfusion injury in mice. J. Enzyme Inhib. Med. Chem. 2022, 37, 2133–2146. [Google Scholar] [CrossRef] [PubMed]
- Deacon, S.W.; Beeser, A.; Fukui, J.A.; Rennefahrt, U.E.; Myers, C.; Chernoff, J.; Peterson, J.R. An isoform-selective, small-molecule inhibitor targets the autoregulatory mechanism of p21-activated kinase. Chem. Biol. 2008, 15, 322–331. [Google Scholar] [CrossRef]
- Abu Aboud, O.; Chen, C.H.; Senapedis, W.; Baloglu, E.; Argueta, C.; Weiss, R.H. Dual and Specific Inhibition of NAMPT and PAK4 By KPT-9274 Decreases Kidney Cancer Growth. Mol. Cancer Ther. 2016, 15, 2119–2129. [Google Scholar] [CrossRef] [PubMed]
- Li, N.; Lopez, M.A.; Linares, M.; Kumar, S.; Oliva, S.; Martinez-Lopez, J.; Xu, L.; Xu, Y.; Perini, T.; Senapedis, W.; et al. Dual PAK4-NAMPT Inhibition Impacts Growth and Survival, and Increases Sensitivity to DNA-Damaging Agents in Waldenström Macroglobulinemia. Clin. Cancer Res. 2019, 25, 369–377. [Google Scholar] [CrossRef] [PubMed]
- Cordover, E.; Wei, J.; Patel, C.; Shan, N.L.; Gionco, J.; Sargsyan, D.; Wu, R.; Cai, L.; Kong, A.-N.; Jacinto, E.; et al. KPT-9274, an Inhibitor of PAK4 and NAMPT, Leads to Downregulation of mTORC2 in Triple Negative Breast Cancer Cells. Chem. Res. Toxicol. 2020, 33, 482–491. [Google Scholar] [CrossRef]
- Wang, J.; Zhu, Y.; Chen, J.; Yang, Y.; Zhu, L.; Zhao, J.; Yang, Y.; Cai, X.; Hu, C.; Rosell, R.; et al. Identification of a novel PAK1 inhibitor to treat pancreatic cancer. Acta Pharm. Sin. B 2020, 10, 603–614. [Google Scholar] [CrossRef]
- He, H.; Dumesny, C.; Ang, C.-S.; Dong, L.; Ma, Y.; Zeng, J.; Nikfarjam, M. A novel PAK4 inhibitor suppresses pancreatic cancer growth and enhances the inhibitory effect of gemcitabine. Transl. Oncol. 2022, 16, 101329. [Google Scholar] [CrossRef]
- Jones, J.G. Hepatic glucose and lipid metabolism. Diabetologia 2016, 59, 1098–1103. [Google Scholar] [CrossRef]
- Asrani, S.K.; Devarbhavi, H.; Eaton, J.; Kamath, P.S. Burden of liver diseases in the world. J. Hepatol. 2019, 70, 151–171. [Google Scholar] [CrossRef]
- Chiang, Y.-T.A.; Ip, W.; Shao, W.; Song, Z.E.; Chernoff, J.; Jin, T. Activation of cAMP Signaling Attenuates Impaired Hepatic Glucose Disposal in Aged Male p21-Activated Protein Kinase-1 Knockout Mice. Endocrinology 2014, 155, 2122–2132. [Google Scholar] [CrossRef] [PubMed]
- Du, D.; Liu, C.; Qin, M.; Zhang, X.; Xi, T.; Yuan, S.; Hao, H.; Xiong, J. Metabolic dysregulation and emerging therapeutical targets for hepatocellular carcinoma. Acta Pharm. Sin. B 2021, 12, 558–580. [Google Scholar] [CrossRef] [PubMed]
- Ren, Y.; Shen, H.M. Critical role of AMPK in redox regulation under glucose starvation. Redox Biol. 2019, 25, 01154. [Google Scholar] [CrossRef] [PubMed]
- Mao, Z.; Zhang, W. Role of mTOR in Glucose and Lipid Metabolism. Int. J. Mol. Sci. 2018, 19, 2043. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Chen, J.; Huang, J.; Li, Z.; Gong, Y.; Zou, B.; Liu, X.; Ding, L.; Li, P.; Zhu, Z.; et al. HIF-2α upregulation mediated by hypoxia promotes NAFLD-HCC progression by activating lipid synthesis via the PI3K-AKT-mTOR pathway. Aging (Albany NY) 2019, 11, 10839–10860. [Google Scholar] [CrossRef]
- Morrice, N.A.; Fecondo, J.; Wettenhall, R.E. Differential effects of fatty acid and phospholipid activators on the catalytic activities of a structurally novel protein kinase from rat liver. FEBS Lett. 1994, 351, 171–175. [Google Scholar] [CrossRef]
- Yu, W.; Liu, J.; Morrice, N.A.; Wettenhall, R.E. Isolation and characterization of a structural homologue of human PRK2 from rat liver. Distinguishing substrate and lipid activator specificities. J. Biol. Chem. 1997, 272, 10030–10034. [Google Scholar] [CrossRef]
- Joshi, A.; Azuma, R.; Akumuo, R.; Goetzl, L.; Pinney, S.E. Gestational diabetes and maternal obesity are associated with sex-specific changes in miRNA and target gene expression in the fetus. Int. J. Obes. (Lond.) 2020, 44, 1497–1507. [Google Scholar] [CrossRef]
- Zhang, X.; Zhang, X.; Li, Y.; Shao, Y.; Xiao, J.; Zhu, G.; Li, F. PAK4 regulates G6PD activity by p53 degradation involving colon cancer cell growth. Cell Death Dis. 2017, 8, e2820. [Google Scholar] [CrossRef]
- Zhou, J.; Zhou, F.; Wang, W.; Zhang, X.-J.; Ji, Y.-X.; Zhang, P.; She, Z.G.; Zhu, L.; Cai, J.; Li, H. Epidemiological Features of NAFLD From 1999 to 2018 in China. Hepatology 2020, 71, 1851–1864. [Google Scholar] [CrossRef]
- Younossi, Z.M.; Koenig, A.B.; Abdelatif, D.; Fazel, Y.; Henry, L.; Wymer, M. Global epidemiology of nonalcoholic fatty liver disease-Meta-analytic assessment of prevalence, incidence, and outcomes. Hepatology 2016, 64, 73–84. [Google Scholar] [CrossRef] [PubMed]
- Dewidar, B.; Kahl, S.; Pafili, K.; Roden, M. Metabolic liver disease in diabetes—From mechanisms to clinical trials. Metabolism 2020, 111, 154299. [Google Scholar] [CrossRef] [PubMed]
- Tang, S.P.; Mao, X.L.; Chen, Y.H.; Yan, L.L.; Ye, L.P.; Li, S.W. Reactive Oxygen Species Induce Fatty Liver and Ischemia-Reperfusion Injury by Promoting Inflammation and Cell Death. Front. Immunol. 2022, 13, 870239. [Google Scholar] [CrossRef] [PubMed]
- Guo, J.; Akahoshi, T.; Mizuta, Y.; Murata, M.; Narahara, S.; Kawano, T.; Nagao, Y.; Zhang, S.; Tomikawa, M.; Kawanaka, H.; et al. Histidine-Rich Glycoprotein Alleviates Liver Ischemia/Reperfusion Injury in Mice With Nonalcoholic Steatohepatitis. Liver Transpl. 2021, 27, 840–853. [Google Scholar] [CrossRef] [PubMed]
- Yang, F.; Zhang, Y.; Ren, H.; Wang, J.; Shang, L.; Liu, Y.; Zhu, W.; Shi, X. Ischemia reperfusion injury promotes recurrence of hepatocellular carcinoma in fatty liver via ALOX12-12HETE-GPR31 signaling axis. J. Exp. Clin. Cancer Res. 2019, 38, 1–14. [Google Scholar] [CrossRef]
- Czigany, Z.; Lurje, I.; Schmelzle, M.; Schöning, W.; Öllinger, R.; Raschzok, N.; Sauer, I.M.; Tacke, F.; Strnad, P.; Trautwein, C.; et al. Ischemia-Reperfusion Injury in Marginal Liver Grafts and the Role of Hypothermic Machine Perfusion: Molecular Mechanisms and Clinical Implications. J. Clin. Med. 2020, 9, 846. [Google Scholar] [CrossRef]
- Ringelhan, M.; Pfister, D.; O’Connor, T.; Pikarsky, E.; Heikenwalder, M. The immunology of hepatocellular carcinoma. Nat. Immunol. 2018, 19, 222–232. [Google Scholar] [CrossRef]
- Huynh, N.; Wang, K.; Yim, M.; Dumesny, C.J.; Sandrin, M.S.; Baldwin, G.S.; Nikfarjam, M.; He, H. Depletion of p21-activated kinase 1 up-regulates the immune system of APC(∆14/+) mice and inhibits intestinal tumorigenesis. BMC Cancer 2017, 17, 431. [Google Scholar] [CrossRef]
- Abril-Rodriguez, G.; Torrejon, D.Y.; Liu, W.; Zaretsky, J.M.; Nowicki, T.S.; Tsoi, J.; Puig-Saus, C.; Baselga-Carretero, I.; Medina, E.; Quist, M.J.; et al. PAK4 inhibition improves PD-1 blockade immunotherapy. Nat. Cancer 2020, 1, 46–58. [Google Scholar] [CrossRef]

| Liver Disorders | Group I PAKs | Group II PAKs |
|---|---|---|
| Liver cancer | PAK1: β-catenin → Proliferation/invasion↑ and apoptosis↓ MAPK/ERK, PI3K/AKT → Apoptosis↓ Snail → Migration and invasion↑ Snail, β-catenin, p53/p21 → Xenograft tumor growth↑ Paxillin, JNK → Actin stress fibers and metastasis↑ Anchorage-independent growth and anoikis resistance ↑ HIF-1α, Vimentin → Vasculogenic mimicry↑ PAK2: Migration↑ PAK3: TGF-β/Smad → EMT↑ | PAK4: Caspase-8, NF-κB → Apoptosis↓ RAF/MEK/ERK → Tumor growth and metastasis↑ P53 → Migration and invasion↑ PAK5: Proliferation and invasion↑ Apoptosis induced by cisplatin↓ β-catenin/ABCB1 → Resistance to adriamycin, 5-fluorouracil and sorafenib↑ Radiotherapy resistance↑ PAK6: Proliferation↑ Eg5 → mitosis↑ HCC metastasis↑ |
| Liver IRI | PAK1: VEGFα, IGF1 → IRI↓ | PAK4: Nrf2 → Inflammation and necrosis of hepatocytes↑ |
| Hepatitis | PAK1: RAF-1 → HBV X protein → Apoptosis↓ HCV replication↓ NMIIA/Cofilin → HEV infection↑ NF-κB/IRF1/IL-6 → The differentiation of CD4+ T cells to Th17 cells and inflammation in Schistosoma japonicum infection↑ Autoimmune hepatitis progression↑ PAK2: Apoptosis and inflammatory reaction in cells infected with HCV↓ PAK3: HBV replication↑ | PAK6: Tumorigenesis in alcoholic hepatitis↑ |
| Liver fibrosis | PAK1: Integrin β-1 → Myofibroblasts activation and ECM production↑ PAK2: Myofibroblast activation and liver fibrosis↑ PAK3: Integrin β-1 → Myofibroblasts activation and ECM production↑ | PAK4: Perisinusoidal/pericellular fibrosis↑ |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Qiu, X.; Xu, H.; Wang, K.; Gao, F.; Xu, X.; He, H. P-21 Activated Kinases in Liver Disorders. Cancers 2023, 15, 551. https://doi.org/10.3390/cancers15020551
Qiu X, Xu H, Wang K, Gao F, Xu X, He H. P-21 Activated Kinases in Liver Disorders. Cancers. 2023; 15(2):551. https://doi.org/10.3390/cancers15020551
Chicago/Turabian StyleQiu, Xun, Hanzhi Xu, Kai Wang, Fengqiang Gao, Xiao Xu, and Hong He. 2023. "P-21 Activated Kinases in Liver Disorders" Cancers 15, no. 2: 551. https://doi.org/10.3390/cancers15020551
APA StyleQiu, X., Xu, H., Wang, K., Gao, F., Xu, X., & He, H. (2023). P-21 Activated Kinases in Liver Disorders. Cancers, 15(2), 551. https://doi.org/10.3390/cancers15020551





