Diagnostic and Therapeutic Pathway of Advanced Ovarian Cancer with Peritoneal Metastases
Abstract
Simple Summary
Abstract
1. Introduction
2. Sources and Methodology
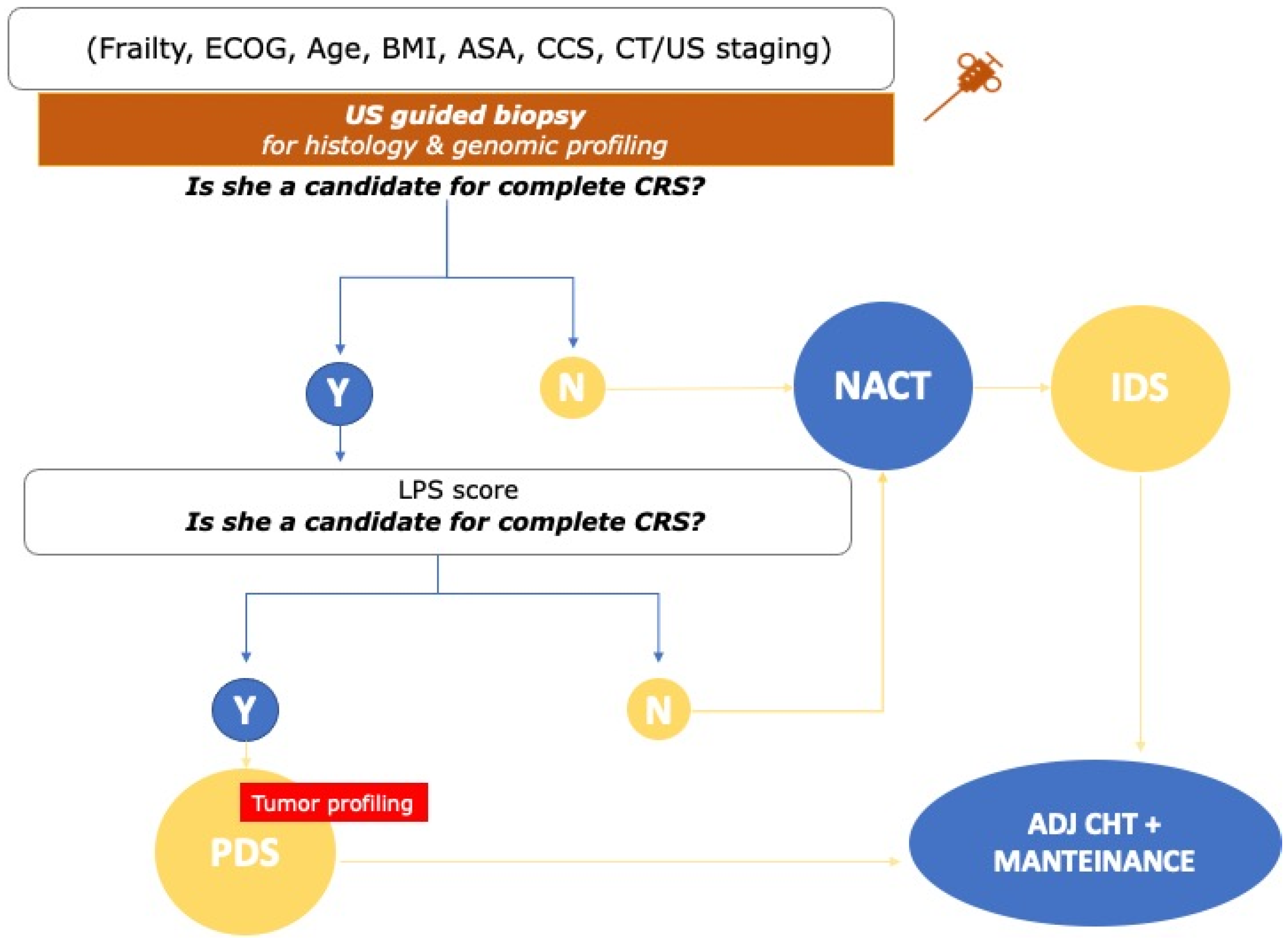
3. Diagnostic Pathway
3.1. Radiologic Assessment and Role of Tumor Markers
3.2. Selection of the Appropriate Surgical Candidate
3.3. The Role of Laparoscopy in Treatment Selection
4. Treatment Pathway
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Filippova, O.T.; Tin, A.L.; Alonso, J.; Vickers, A.J.; Tew, W.P.; Gardner, G.J.; Sonoda, Y.; Roche, K.L.; Zivanovic, O.; Chi, D.S.; et al. Frailty based on the memorial Sloan Kettering Frailty Index is associated with surgical decision making, clinical trial participation, and overall survival among older women with ovarian cancer. Gynecol. Oncol. 2021, 161, 687–692. [Google Scholar] [CrossRef]
- Sioulas, V.D.; Schiavone, M.B.; Kadouri, D.; Zivanovic, O.; Roche, K.L.; O’Cearbhaill, R.; Abu-Rustum, N.R.; Levine, D.A.; Sonoda, Y.; Gardner, G.J.; et al. Optimal primary management of bulky stage IIIC ovarian, fallopian tube and peritoneal carcinoma: Are the only options complete gross resection at primary debulking surgery or neoadjuvant chemotherapy? Gynecol. Oncol. 2017, 145, 15–20. [Google Scholar] [CrossRef]
- Tseng, J.H.; Cowan, R.A.; Zhou, Q.; Iasonos, A.; Byrne, M.; Polcino, T.; Polen-De, C.; Gardner, G.J.; Sonoda, Y.; Zivanovic, O.; et al. Continuous improvement in primary Debulking surgery for advanced ovarian cancer: Do increased complete gross resection rates independently lead to increased progression-free and overall survival? Gynecol. Oncol. 2018, 151, 24–31. [Google Scholar] [CrossRef]
- Onda, T.; Satoh, T.; Ogawa, G.; Saito, T.; Kasamatsu, T.; Nakanishi, T.; Mizutani, T.; Takehara, K.; Okamoto, A.; Ushijima, K.; et al. Comparison of survival between primary debulking surgery and neoadjuvant chemotherapy for stage III/IV ovarian, tubal and peritoneal cancers in phase III randomised trial. Eur. J. Cancer 2020, 130, 114–125. [Google Scholar] [CrossRef] [PubMed]
- Mueller, J.; Zhou, Q.C.; Iasonos, A.; O’Cearbhaill, R.E.; Alvi, F.A.; El Haraki, A.; Eriksson, A.G.Z.; Gardner, G.J.; Sonoda, Y.; Levine, D.A.; et al. Neoadjuvant chemotherapy and primary debulking surgery utilization for advanced-stage ovarian cancer at a comprehensive cancer center. Gynecol. Oncol. 2016, 140, 436–442. [Google Scholar] [CrossRef] [PubMed]
- May, T.; Comeau, R.; Sun, P.; Kotsopoulos, J.; Narod, S.A.; Rosen, B.; Ghatage, P. A Comparison of Survival Outcomes in Advanced Serous Ovarian Cancer Patients Treated With Primary Debulking Surgery Versus Neoadjuvant Chemotherapy. Int. J. Gynecol. Cancer 2017, 27, 668–674. [Google Scholar] [CrossRef] [PubMed]
- Kehoe, S.; Hook, J.; Nankivell, M.; Jayson, G.C.; Kitchener, H.; Lopes, T.; Luesley, D.; Perren, T.; Bannoo, S.; Mascarenhas, M.; et al. Primary chemotherapy versus primary surgery for newly diagnosed advanced ovarian cancer (CHORUS): An open-label, randomised, controlled, non-inferiority trial. Lancet 2015, 386, 249–257. [Google Scholar] [CrossRef] [PubMed]
- Vergote, I.; Tropé, C.G.; Amant, F.; Kristensen, G.B.; Ehlen, T.; Johnson, N.; Verheijen, R.H.; van der Burg, M.E.; Lacave, A.J.; Panici, P.B.; et al. European Organization for Research and Treatment of Cancer-Gynaecological Cancer Group; NCIC Clinical Trials Group. Neoadjuvant chemotherapy or primary surgery in stage IIIC or IV ovarian cancer. N. Engl. J. Med. 2010, 363, 943–953. [Google Scholar] [CrossRef]
- Fagotti, A.; Ferrandina, M.G.; Vizzielli, G.; Pasciuto, T.; Fanfani, F.; Gallotta, V.; Margariti, P.A.; Chiantera, V.; Costantini, B.; Alletti, S.; et al. Randomized trial of primary debulking surgery versus neoadjuvant chemotherapy for advanced epithelial ovarian cancer (SCORPION-NCT01461850). Int. J. Gynecol. Cancer 2020, 30, 1657–1664. [Google Scholar] [CrossRef]
- Suidan, R.S.; Ramirez, P.T.; Sarasohn, D.; Teitcher, J.B.; Mironov, S.; Iyer, R.B.; Zhou, Q.; Iasonos, A.; Paul, H.; Hosaka, M.; et al. A multicenter prospective trial evaluating the ability of preoperative computed tomography scan and serum CA-125 to predict suboptimal cytoreduction at primary debulking surgery for advanced ovarian, fallopian tube, and peritoneal cancer. Gynecol. Oncol. 2014, 134, 455–461. [Google Scholar] [CrossRef]
- Bristow, R.E.; Duska, L.R.; Lambrou, N.C.; Fishman, E.K.; O’Neill, M.J.; Trimble, E.L.; Montz, F.J. A model for predicting surgical outcome in patients with advanced ovarian carcinoma using computed tomography. Cancer 2000, 89, 1532–1540. [Google Scholar] [CrossRef]
- Axtell, A.E.; Lee, M.H.; Bristow, R.E.; Dowdy, S.C.; Cliby, W.A.; Raman, S.; Weaver, J.P.; Gabbay, M.; Ngo, M.; Lentz, S.; et al. Multi-Institutional Reciprocal Validation Study of Computed Tomography Predictors of Suboptimal Primary Cytoreduction in Patients With Advanced Ovarian Cancer. J. Clin. Oncol. 2007, 25, 384–389. [Google Scholar] [CrossRef] [PubMed]
- Chi, D.S.; Venkatraman, E.S.; Masson, V.; Hoskins, W.J. The ability of preoperative serum CA-125 to predict optimal primary tumor cytoreduction in stage III epithelial ovarian carcinoma. Gynecol. Oncol. 2000, 77, 227–231. [Google Scholar] [CrossRef]
- Gemer, O.; Lurian, M.; Gdalevich, M.; Kapustian, V.; Piura, E.; Schneider, D.; Lavie, O.; Levy, T.; Fishman, A.; Dgani, R.; et al. A multicenter study of CA 125 level as a predictor of non-optimal primary cytoreduction of advanced epithelial ovarian cancer. Eur. J. Surg. Oncol. (EJSO) 2005, 31, 1006–1010. [Google Scholar] [CrossRef]
- Chi, D.S.; Zivanovic, O.; Palayekar, M.J.; Eisenhauer, E.L.; Abu-Rustum, N.R.; Sonoda, Y.; Levine, D.A.; Leitao, M.M.; Brown, C.L.; Barakat, R.R. A contemporary analysis of the ability of preoperative serum CA-125 to predict primary cytoreductive outcome in patients with advanced ovarian, tubal and peritoneal carcinoma. Gynecol. Oncol. 2009, 112, 6–10. [Google Scholar] [CrossRef]
- Kang, S.; Kim, T.-J.; Nam, B.-H.; Seo, S.-S.; Kim, B.-G.; Bae, D.-S.; Park, S.-Y. Preoperative serum CA-125 levels and risk of suboptimal cytoreduction in ovarian cancer: A meta-analysis. J. Surg. Oncol. 2010, 101, 13–17. [Google Scholar] [CrossRef] [PubMed]
- Tozzi, R.; Traill, Z.; Valenti, G.; Ferrari, F.; Gubbala, K.; Campanile, R.G. A prospective study on the diagnostic pathway of patients with stage IIIC-IV ovarian cancer: Exploratory laparoscopy (EXL) + CT scan VS. CT scan. Gynecol. Oncol. 2021, 161, 188–193. [Google Scholar] [CrossRef] [PubMed]
- Fagotti, A.; Ferrandina, M.G.; Fanfani, F.; Ercoli, A.; Lorusso, D.; Rossi, M.; Scambia, G. A Laparoscopy-Based Score to Predict Surgical Outcome in Patients with Advanced Ovarian Carcinoma: A Pilot Study. Ann. Surg. Oncol. 2006, 13, 1156–1161. [Google Scholar] [CrossRef]
- Llueca, A.; Climent, M.T.; Escrig, J.; Carrasco, P.; Serra, A. MUAPOS working group (Multidisciplinary Unit of Abdominal Pelvic Oncology Surgery). Validation of three predictive models for suboptimal cytoreductive surgery in advanced ovarian cancer. Sci. Rep. 2021, 11, 8111. [Google Scholar] [CrossRef]
- Shahrokni, A.; Tin, A.; Alexander, K.; Sarraf, S.; Afonso, A.; Filippova, O.; Harris, J.; Downey, R.J.; Vickers, A.J.; Korc-Grodzicki, B. Development and Evaluation of a New Frailty Index for Older Surgical Patients With Cancer. JAMA Netw. Open 2019, 2, e193545. [Google Scholar] [CrossRef]
- Rocha, V.; Marmelo, F.; Leite-Moreira, A.; Gonçalves, D. Clinical Utility of Frailty Scales for the Prediction of Postoperative Complications: Systematic Review and Meta-Analysis. Rev. Port. Cir. Cardiotorac. Vasc. 2017, 24, 132. [Google Scholar] [PubMed]
- Fried, L.P.; Tangen, C.M.; Walston, J.; Newman, A.B.; Hirsch, C.; Gottdiener, J.; Seeman, T.; Tracy, R.; Kop, W.J.; Burke, G.; et al. Cardiovascular Health Study Collaborative Research Group. Frailty in older adults: Evidence for a phenotype. J. Gerontol. A Biol. Sci. Med. Sci. 2001, 56, M146–M156. [Google Scholar] [CrossRef] [PubMed]
- Mitnitski, A.B.; Mogilner, A.J.; Rockwood, K. Accumulation of deficits as aproxy measure of aging. Sci. World J. 2001, 1, 323–336. [Google Scholar] [CrossRef] [PubMed]
- Uppal, S.; Igwe, E.; Rice, L.W.; Spencer, R.J.; Rose, S.L. Frailty index predicts severe complications in gynecologic oncology patients. Gynecol. Oncol. 2015, 137, 98–101. [Google Scholar] [CrossRef]
- Kumar, A.; Langstraat, C.L.; DeJong, S.R.; McGree, M.E.; Bakkum-Gamez, J.N.; Weaver, A.L.; LeBrasseur, N.K.; Cliby, W.A. Functional not chronologic age: Frailty index predicts outcomes in advanced ovarian cancer. Gynecol. Oncol. 2017, 147, 104–109. [Google Scholar] [CrossRef]
- Inci, M.G.; Anders, L.; Woopen, H.; Richter, R.; Guzel, D.; Armbrust, R.; Sehouli, J. Frailty Index for prediction of surgical outcome in ovarian cancer: Results of a prospective study. Gynecol. Oncol. 2021, 161, 396–401. [Google Scholar] [CrossRef]
- Aletti, G.D.; Dowdy, S.C.; Podratz, K.C.; Cliby, W.A. Relationship among surgical com- plexity, short-term morbidity, and overall survival in primary surgery for advanced ovarian cancer. Am. J. Obstet. Gynecol. 2007, 197, 676.e1–676.e7. [Google Scholar] [CrossRef]
- Chambers, L.M.; Chalif, J.; Yao, M.; Chichura, A.; Morton, M.; Gruner, M.; Costales, A.B.; Horowitz, M.; Chau, D.B.; Vargas, R.; et al. Modified frailty index predicts postoperative complications in women with gynecologic cancer undergoing cytoreductive surgery and hyperthermic intraperitoneal chemotherapy. Gynecol. Oncol. 2021, 162, 368–374. [Google Scholar] [CrossRef]
- Narasimhulu, D.M.; Thannickal, A.; Kumar, A.; Weaver, A.L.; McGree, M.E.; Langstraat, C.L.; Cliby, W.A. Appropriate triage allows aggressive primary debulking surgery with rates of morbidity and mortality comparable to interval surgery after chemotherapy. Gynecol. Oncol. 2021, 160, 681–687. [Google Scholar] [CrossRef]
- Colombo, N.; Sessa, C.; du Bois, A.; Ledermann, J.; McCluggage, W.G.; McNeish, I.; Morice, P.; Pignata, S.; Ray-Coquard, I.; Vergote, I.; et al. ESMO-ESGO consensus conference recommendations on ovarian cancer: Pathology and molecular biology, early and advanced stages, borderline tumours and recurrent disease. Int. J. Gynecol. Cancer 2019, 29, ijgc-2019-000308. [Google Scholar] [CrossRef]
- Armstrong, D.K.; Alvarez, R.D.; Bakkum-Gamez, J.N.; Barroilhet, L.; Behbakht, K.; Berchuck, A.; Chen, L.M.; Cristea, M.; DeRosa, M.; Eisenhauer, E.L.; et al. Ovarian Cancer, Version 2.2020, NCCN Clinical Practice Guidelines in Oncology. J. Natl. Compr. Canc. Netw. 2021, 19, 191–226. [Google Scholar] [CrossRef]
- Petrillo, M.; Vizzielli, G.; Fanfani, F.; Gallotta, V.; Cosentino, F.; Chiantera, V.; Legge, F.; Carbone, V.; Scambia, G.; Fagotti, A. Definition of a dynamic laparoscopic model for the prediction of incomplete cytoreduction in advanced epithelial ovarian cancer: Proof of a concept. Gynecol. Oncol. 2015, 139, 5–9. [Google Scholar] [CrossRef]
- Hansen, J.M.; Sood, A.K.; Coleman, R.L.; Westin, S.N.; Soliman, P.T.; Ramirez, P.T.; Fellman, B.M.; Schmeler, K.M.; Fleming, N.D. Concordance of a laparoscopic scoring algorithm with primary surgery findings in advanced stage ovarian cancer. Gynecol. Oncol. 2018, 151, 428–432. [Google Scholar] [CrossRef] [PubMed]
- Fagotti, A.; Fanfani, F.; Vizzielli, G.; Gallotta, V.; Ercoli, A.; Paglia, A.; Costantini, B.; Vigliotta, M.; Scambia, G.; Ferrandina, G. Should laparoscopy be included in the work-up of advanced ovarian cancer patients attempting interval debulking surgery? Gynecol. Oncol. 2010, 116, 72–77. [Google Scholar] [CrossRef] [PubMed]
- Llueca, A.; Serra, A.; Delgado, K.; Maiocchi, K.; Jativa, R.; Gomez, L.; Escrig, J. A radiologic-laparoscopic model to predict suboptimal (or complete and optimal) debulking surgery in advanced ovarian cancer: A pilot study. Int. J. Women’s Health 2019, 11, 333–342. [Google Scholar] [CrossRef]
- Wright, A.A.; Bohlke, K.; Armstrong, D.K.; Bookman, M.A.; Cliby, W.A.; Coleman, R.L.; Dizon, D.S.; Kash, J.J.; Meyer, L.A.; Moore, K.N.; et al. Neoadjuvant chemotherapy for newly diagnosed, advanced ovarian cancer: Society of Gynecologic Oncology and American Society of Clinical Oncology Clinical Practice Guideline. Gynecol. Oncol. 2016, 143, 3–15. [Google Scholar] [CrossRef] [PubMed]
- Mascilini, F.; Quagliozzi, L.; Moro, F.; Moruzzi, M.C.; De Blasis, I.; Paris, V.; Scambia, G.; Fagotti, A.; Testa, A.C. Role of transvaginal ultrasound-guided biopsy in gynecology. Int. J. Gynecol. Cancer 2020, 30, 128–132. [Google Scholar] [CrossRef]
- Grabowski, J.P.; Harter, P.; Heitz, F.; Pujade-Lauraine, E.; Reuss, A.; Kristensen, G.; Ray-Coquard, I.; Heitz, J.; Traut, A.; Pfisterer, J.; et al. Operability and chemotherapy responsiveness in advanced low-grade serous ovarian cancer. An analysis of the AGO Study Group metadatabase. Gynecol. Oncol. 2016, 140, 457–462. [Google Scholar] [CrossRef] [PubMed]
- Sugiyama, T.; Kamura, T.; Kigawa, J.; Terakawa, N.; Kikuchi, Y.; Kita, T.; Suzuki, M.; Sato, I.; Taguchi, K. Clinical characteristics of clear cell carcinoma of the ovary: A distinct histologic type with poor prognosis and resistance to platinum-based chemotherapy. Cancer 2000, 88, 2584–2589. [Google Scholar] [CrossRef]
- Morice, P.; Leary, A.; Gouy, S. Mucinous Ovarian Carcinoma. N. Engl. J. Med. 2019, 381, e3. [Google Scholar]
- Johnson, R.L.; Laios, A.; Jackson, D.; Nugent, D.; Orsi, N.M.; Theophilou, G.; Thangavelu, A.; de Jong, D. The Uncertain Benefit of Adjuvant Chemotherapy in Advanced Low-Grade Serous Ovarian Cancer and the Pivotal Role of Surgical Cytoreduction. J. Clin. Med. 2021, 10, 5927. [Google Scholar] [CrossRef]
- Pisano, C.; Greggi, S.; Tambaro, R.; Losito, S.; Iodice, F.; Di Maio, M.; Ferrari, E.; Falanga, M.; Formato, R.; Iaffaioli, V.R.; et al. Activity of chemotherapy in mucinous epithelial ovarian cancer: A retrospective study. Anticancer Res. 2005, 25, 3501–3505. [Google Scholar]
- Prat, J. FIGO Committee on Gynecologic Oncology. Staging classification for cancer of the ovary, fallopian tube, and peritoneum. Int. J. Gynaecol. Obstet. 2014, 124, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Vergote, I.; Coens, C.; Nankivell, M.; Kristensen, G.B.; Parmar, M.K.B.; Ehlen, T.; Jayson, G.C.; Johnson, N.; Swart, A.M.; Verheijen, R.; et al. Neoadjuvant chemotherapy versus debulking surgery in advanced tubo-ovarian cancers: Pooled analysis of individual patient data from the EORTC 55971 and CHORUS trials. Lancet Oncol. 2018, 19, 1680–1687. [Google Scholar] [CrossRef]
- Reuss, A.; du Bois, A.; Harter, P.; Fotopoulou, C.; Sehouli, J.; Aletti, G.; Guyon, F.; Greggi, S.; Mosgaard, B.J.; Reinthaller, A.; et al. TRUST: Trial of Radical Upfront Surgical Therapy in advanced ovarian cancer (ENGOT ov33/AGO-OVAR OP7). Int. J. Gynecol. Cancer 2019, 29, 1327–1331. [Google Scholar] [CrossRef]
- Bristow, R.E.; Tomacruz, R.S.; Armstrong, D.K.; Trimble, E.L.; Montz, F.J. Survival effect of maximal cytoreductive surgery for advanced ovarian carcinoma during the platinum era: A meta-analysis. J. Clin. Oncol. 2002, 20, 1248–1259. [Google Scholar] [CrossRef] [PubMed]
- Chang, S.-J.; Hodeib, M.; Chang, J.; Bristow, R.E. Survival impact of complete cytoreduction to no gross residual disease for advanced-stage ovarian cancer: A meta-analysis. Gynecol. Oncol. 2013, 130, 493–498. [Google Scholar] [CrossRef]
- Ghirardi, V.; Moruzzi, M.; Bizzarri, N.; Vargiu, V.; D’Indinosante, M.; Garganese, G.; Pasciuto, T.; Loverro, M.; Scambia, G.; Fagotti, A. Minimal residual disease at primary debulking surgery versus complete tumor resection at interval debulking surgery in advanced epithelial ovarian cancer: A survival analysis. Gynecol. Oncol. 2020, 157, 209–213. [Google Scholar] [CrossRef] [PubMed]
- van Driel, W.J.; Koole, S.N.; Sikorska, K.; van Leeuwen, J.H.; Schreuder, H.W.R.; Hermans, R.H.M.; de Hingh, I.H.J.T.; van der Velden, J.; Arts, H.J.; Massuger, L.F.A.G.; et al. Hyperthermic Intraperitoneal Chemotherapy in Ovarian Cancer. N. Engl. J. Med. 2018, 378, 230–240. [Google Scholar] [CrossRef]
- Ghirardi, V.; Ronsini, C.; Trozzi, R.; Di Ilio, C.; Di Giorgio, A.; Cianci, S.; Draisci, G.; Scambia, G.; Fagotti, A. Hyperthermic intraperitoneal chemotherapy in interval debulking surgery for advanced epithelial ovarian cancer: A single-center, real-life experience. Cancer 2020, 126, 5256–5262. [Google Scholar] [CrossRef]
- Koole, S.; van Stein, R.; Sikorska, K.; Barton, D.; Perrin, L.; Brennan, D.; Zivanovic, O.; Mosgaard, B.J.; Fagotti, A.; Colombo, P.E.; et al. Primary cytoreductive surgery with or without hyperthermic intraperitoneal chemotherapy (HIPEC) for FIGO stage III epithelial ovarian cancer: OVHIPEC-2, a phase III randomized clinical trial. Int. J. Gynecol. Cancer 2020, 30, 888–892. [Google Scholar] [CrossRef]
- Vergote, I.; Harter, P.; Chiva, L. Hyperthermic intraperitoneal chemotherapy does not improve survival in advanced ovarian cancer. Cancer 2019, 125 (Suppl. S24), 4594–4597. [Google Scholar] [CrossRef]
- van Stein, R.M.; Aalbers, A.G.J.; Sonke, G.S.; van Driel, W.J. Hyperthermic Intraperitoneal Chemotherapy for Ovarian and Colorectal Cancer: A Review. JAMA Oncol. 2021, 7, 1231–1238. [Google Scholar] [CrossRef] [PubMed]
- Lim, M.C.; Chang, S.J.; Park, B.; Yoo, H.J.; Yoo, C.W.; Nam, B.H.; Park, S.Y. HIPEC for Ovarian Cancer Collaborators. Survival after Hyperthermic Intraperitoneal Chemotherapy and Primary or Interval Cytoreductive Surgery in Ovarian Cancer: A Randomized Clinical Trial. JAMA Surg. 2022, 157, 374–383. [Google Scholar] [CrossRef]
- Zivanovic, O.; Chi, D.S.; Zhou, Q.; Iasonos, A.; Konner, J.A.; Makker, V.; Grisham, R.N.; Brown, A.K.; Nerenstone, S.; Diaz, J.P.; et al. Secondary Cytoreduction and Carboplatin Hyperthermic Intraperitoneal Chemotherapy for Platinum-Sensitive Recurrent Ovarian Cancer: An MSK Team Ovary Phase II Study. J. Clin. Oncol. 2021, 39, 2594–2604. [Google Scholar] [CrossRef]
- Kim, S.I.; Kim, J.H.; Lee, S.; Cho, H.; van Driel, W.J.; Sonke, G.S.; Bristow, R.E.; Park, S.-Y.; Fotopoulou, C.; Lim, M.C. Hyperthermic intraperitoneal chemotherapy for epithelial ovarian cancer: A meta-analysis. Gynecol. Oncol. 2022, 167, 547–556. [Google Scholar] [CrossRef] [PubMed]
- Koole, S.N.; Schouten, P.C.; Hauke, J.; Kluin, R.J.C.; Nederlof, P.; Richters, L.K.; Krebsbach, G.; Sikorska, K.; Alkemade, M.; Opdam, M.; et al. Effect of HIPEC according to HRD/BRCAwt genomic profile in stage III ovarian cancer: Results from the phase III OVHIPEC trial. Int. J. Cancer 2022, 151, 1394–1404. [Google Scholar] [CrossRef]
- McGuire, W.P.; Hoskins, W.J.; Brady, M.F.; Kucera, P.R.; Partridge, E.E.; Look, K.Y.; Clarke-Pearson, D.L.; Davidson, M. Cyclophosphamide and cisplatin compared with paclitaxel and cisplatin in patients with stage III and stage IV ovarian cancer. N. Engl. J. Med. 1996, 334, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Ozols, R.F.; Bundy, B.N.; Greer, B.E.; Fowler, J.M.; Clarke-Pearson, D.; Burger, R.A.; Mannel, R.S.; DeGeest, K.; Hartenbach, E.M.; Baergen, R.; et al. Gynecologic Oncology Group. Phase III Trial of Carboplatin and Paclitaxel Compared With Cisplatin and Paclitaxel in Patients With Optimally Resected Stage III Ovarian Cancer: A Gynecologic Oncology Group Study. J. Clin. Oncol. 2003, 21, 3194–3200. [Google Scholar] [CrossRef]
- Clamp, A.R.; James, E.C.; McNeish, I.A.; Dean, A.; Kim, J.-W.; O’Donnell, D.M.; Hook, J.; Coyle, C.; Blagden, S.; Brenton, J.D.; et al. Weekly dose-dense chemotherapy in first-line epithelial ovarian, fallopian tube, or primary peritoneal carcinoma treatment (ICON8): Primary progression free survival analysis results from a GCIG phase 3 randomised controlled trial. Lancet 2019, 394, 2084–2095. [Google Scholar] [CrossRef]
- Oza, A.M.; Cook, A.D.; Pfisterer, J.; Embleton, A.; Ledermann, J.A.; Pujade-Lauraine, E.; Kristensen, G.; Carey, M.S.; Beale, P.; Cervantes, A.; et al. ICON7 trial investigators. Standard chemotherapy with or without bevacizumab for women with newly diagnosed ovarian cancer (ICON7): Overall survival results of a phase 3 randomised trial. Lancet Oncol. 2015, 16, 928–936. [Google Scholar] [CrossRef]
- Tewari, K.S.; Burger, R.A.; Enserro, D.; Norquist, B.M.; Swisher, E.M.; Brady, M.F.; Bookman, M.A.; Fleming, G.F.; Huang, H.; Homesley, H.D.; et al. Final Overall Survival of a Randomized Trial of Bevacizumab for Primary Treatment of Ovarian Cancer. J. Clin. Oncol. 2019, 37, 2317–2328. [Google Scholar] [CrossRef]
- Moore, K.; Colombo, N.; Scambia, G.; Kim, B.G.; Oaknin, A.; Friedlander, M.; Lisyanskaya, A.; Floquet, A.; Leary, A.; Sonke, G.S.; et al. Maintenance Olaparib in Patients with Newly Diagnosed Advanced Ovarian Cancer. N. Engl. J. Med. 2018, 379, 2495–2505. [Google Scholar] [CrossRef] [PubMed]
- Coleman, R.L.; Fleming, G.F.; Brady, M.F.; Swisher, E.M.; Steffensen, K.D.; Friedlander, M.; Okamoto, A.; Moore, K.N.; Efrat Ben-Baruch, N.; Werner, T.L.; et al. Veliparib with First-Line Chemotherapy and as Maintenance Therapy in Ovarian Cancer. N. Engl. J. Med. 2019, 381, 2403–2415. [Google Scholar] [CrossRef] [PubMed]
- González-Martín, A.; Pothuri, B.; Vergote, I.; DePont Christensen, R.; Graybill, W.; Mirza, M.R.; McCormick, C.; Lorusso, D.; Hoskins, P.; Freyer, G.; et al. PRIMA/ENGOT-OV26/GOG-3012 Investigators. Niraparib in Patients with Newly Diagnosed Advanced Ovarian Cancer. N. Engl. J. Med. 2019, 381, 2391–2402. [Google Scholar] [CrossRef]
- Sehouli, J.; Richter, R.; Braicu, E.I.; Bühling, K.J.; Bahra, M.; Neuhaus, P.; Lichtenegger, W.; Fotopoulou, C. Role of secondary cytoreductive surgery in ovarian cancer relapse: Who will benefit? A systematic analysis of 240 consecutive patients. J. Surg. Oncol. 2010, 102, 656–662. [Google Scholar] [CrossRef] [PubMed]
- Harter, P.; Du Bois, A.; Hahmann, M.; Hasenburg, A.; Burges, A.; Loibl, S.; Gropp, M.; Huober, J.; Fink, D.; Schröder, W.; et al. Surgery in Recurrent Ovarian Cancer: The Arbeitsgemeinschaft Gynaekologische Onkologie (AGO) DESKTOP OVAR Trial. Ann. Surg. Oncol. 2006, 13, 1702–1710. [Google Scholar] [CrossRef] [PubMed]
- Harter, P.; Sehouli, J.; Reuss, A.; Hasenburg, A.; Scambia, G.; Cibula, D.; Mahner, S.; Vergote, I.; Reinthaller, A.; Burges, A.; et al. Prospective Validation Study of a Predictive Score for Operability of Recurrent Ovarian Cancer: The Multicenter Intergroup Study DESKTOP II. A Project of the AGO Kommission OVAR, AGO Study Group, NOGGO, AGO-Austria, and MITO. Int. J. Gynecol. Cancer 2011, 21, 289–295. [Google Scholar] [CrossRef]
- Harter, P.; Sehouli, J.; Vergote, I.; Ferron, G.; Reuss, A.; Meier, W.; Greggi, S.; Mosgaard, B.J.; Selle, F.; Guyon, F.; et al. Randomized Trial of Cytoreductive Surgery for Relapsed Ovarian Cancer. N. Engl. J. Med. 2021, 385, 2123–2131. [Google Scholar] [CrossRef] [PubMed]
- Shi, T.; Zhu, J.; Feng, Y.; Tu, D.; Zhang, Y.; Zhang, P.; Jia, H.; Huang, X.; Cai, Y.; Yin, S.; et al. Secondary cytoreduction followed by chemotherapy versus chemotherapy alone in platinum-sensitive relapsed ovarian cancer (SOC-1): A multicentre, open-label, randomised, phase 3 trial. Lancet Oncol. 2021, 22, 439–449. [Google Scholar] [CrossRef]
- Coleman, R.L.; Spirtos, N.M.; Enserro, D.; Herzog, T.J.; Sabbatini, P.; Armstrong, D.K.; Kim, J.W.; Park, S.Y.; Kim, B.G.; Nam, J.H.; et al. Secondary Surgical Cytoreduction for Recurrent Ovarian Cancer. N. Engl. J. Med. 2019, 381, 1929–1939. [Google Scholar] [CrossRef]
- Fotopoulou, C.; Richter, R.; Braicu, I.E.; Schmidt, S.C.; Neuhaus, P.; Lichtenegger, W.; Sehouli, J. Clinical outcome of tertiary surgical cytoreduction in patients with recurrent epithelial ovarian cancer. Ann. Surg. Oncol. 2011, 18, 49–57. [Google Scholar] [CrossRef] [PubMed]
- Fotopoulou, C.; Richter, R.; Braicu, E.I.; Schmidt, S.C.; Lichtenegger, W.; Sehouli, J. Can complete tumor resection be predicted in advanced primary epithelial ovarian cancer? A systematic evaluation of 360 consecutive patients. Eur. J. Surg. Oncol. 2010, 36, 1202–1210. [Google Scholar] [CrossRef] [PubMed]
- Buechel, M.; Herzog, T.; Westin, S.; Coleman, R.; Monk, B.; Moore, K. Treatment of patients with recurrent epithelial ovarian cancer for whom platinum is still an option. Ann. Oncol. 2019, 30, 721–732. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ghirardi, V.; Fagotti, A.; Ansaloni, L.; Valle, M.; Roviello, F.; Sorrentino, L.; Accarpio, F.; Baiocchi, G.; Piccini, L.; De Simone, M.; et al. Diagnostic and Therapeutic Pathway of Advanced Ovarian Cancer with Peritoneal Metastases. Cancers 2023, 15, 407. https://doi.org/10.3390/cancers15020407
Ghirardi V, Fagotti A, Ansaloni L, Valle M, Roviello F, Sorrentino L, Accarpio F, Baiocchi G, Piccini L, De Simone M, et al. Diagnostic and Therapeutic Pathway of Advanced Ovarian Cancer with Peritoneal Metastases. Cancers. 2023; 15(2):407. https://doi.org/10.3390/cancers15020407
Chicago/Turabian StyleGhirardi, Valentina, Anna Fagotti, Luca Ansaloni, Mario Valle, Franco Roviello, Lorena Sorrentino, Fabio Accarpio, Gianluca Baiocchi, Lorenzo Piccini, Michele De Simone, and et al. 2023. "Diagnostic and Therapeutic Pathway of Advanced Ovarian Cancer with Peritoneal Metastases" Cancers 15, no. 2: 407. https://doi.org/10.3390/cancers15020407
APA StyleGhirardi, V., Fagotti, A., Ansaloni, L., Valle, M., Roviello, F., Sorrentino, L., Accarpio, F., Baiocchi, G., Piccini, L., De Simone, M., Coccolini, F., Visaloco, M., Bacchetti, S., Scambia, G., & Marrelli, D. (2023). Diagnostic and Therapeutic Pathway of Advanced Ovarian Cancer with Peritoneal Metastases. Cancers, 15(2), 407. https://doi.org/10.3390/cancers15020407





