Epigenetics of Thymic Epithelial Tumors
Abstract
Simple Summary
Abstract
1. Introduction
2. Thymic Epithelial Tumors
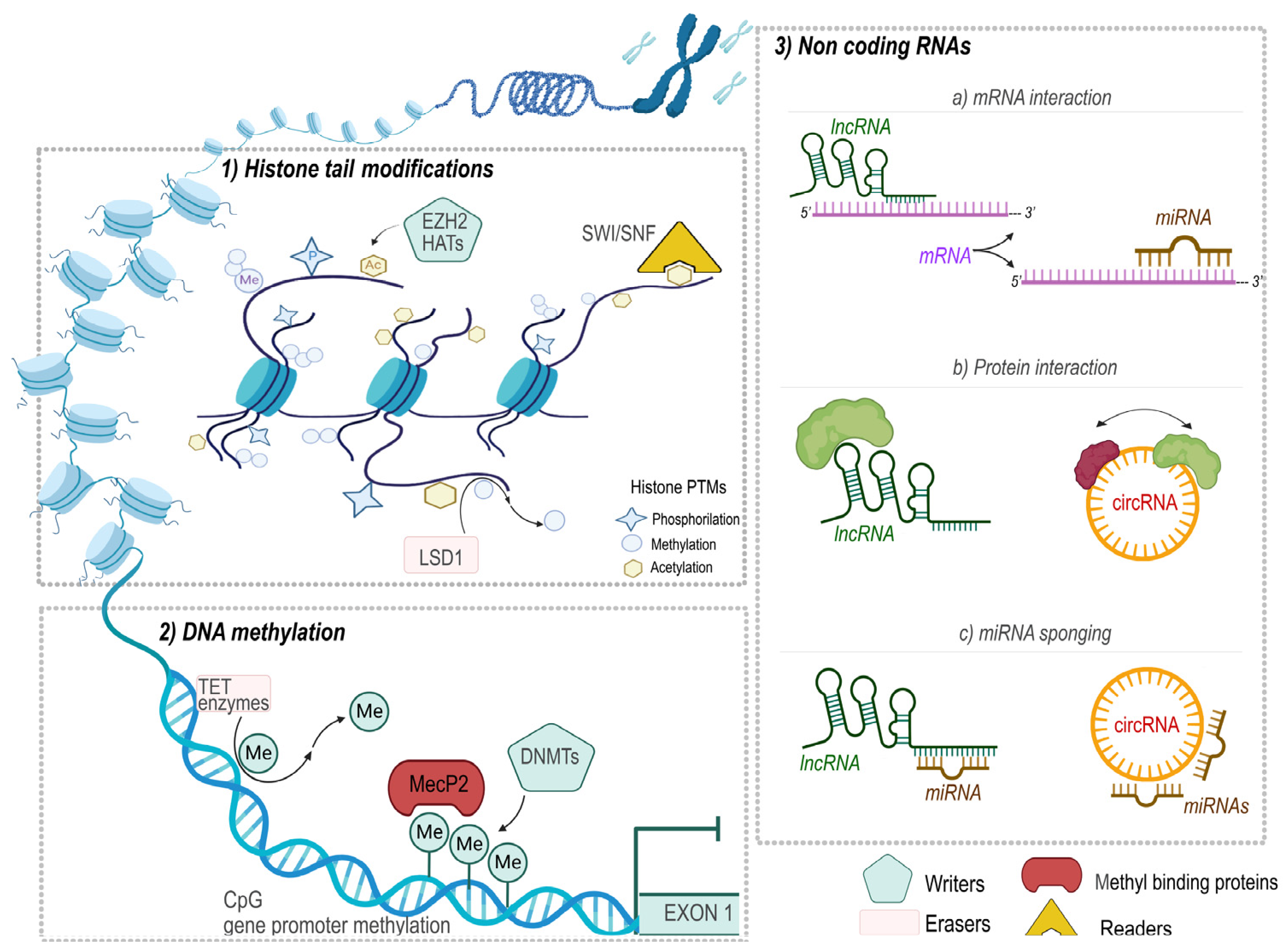
3. Epigenetic Mechanisms
3.1. DNA Methylation
3.2. Histone Post-Translational Modifications
3.3. Non-Protein Coding RNAs
4. DNA Methylation in Thymomas and Thymic Carcinomas
4.1. Thymomas
4.2. Thymic Carcinomas
5. Non-Coding RNAs in Thymomas
5.1. Thymomas
5.2. Thymic Carcinomas
6. Histone Tail Modifications in TETs
7. Epigenetic Deregulation of Neuroendocrine Thymic Epithelial Tumors
8. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Thapa, P.; Farber, D.L. The Role of the Thymus in the Immune Response. Thorac. Surg. Clin. 2019, 29, 123–131. [Google Scholar] [CrossRef] [PubMed]
- Kitada, M.; Sato, K.; Matsuda, Y.; Hayashi, S.; Tokusashi, Y.; Miyokawa, N.; Sasajima, T. Ectopic Thymoma Presenting as a Giant Intrathoracic Tumor: A Case Report. World J. Surg. Oncol. 2011, 9, 66. [Google Scholar] [CrossRef] [PubMed]
- Marx, A.; Chan, J.K.C.; Chalabreysse, L.; Dacic, S.; Detterbeck, F.; French, C.A.; Hornick, J.L.; Inagaki, H.; Jain, D.; Lazar, A.J.; et al. The 2021 WHO Classification of Tumors of the Thymus and Mediastinum: What Is New in Thymic Epithelial, Germ Cell, and Mesenchymal Tumors? J. Thorac. Oncol. 2022, 17, 200–213. [Google Scholar] [CrossRef] [PubMed]
- Gao, N.; Li, Y.; Li, J.; Gao, Z.; Yang, Z.; Li, Y.; Liu, H.; Fan, T. Long Non-Coding RNAs: The Regulatory Mechanisms, Research Strategies, and Future Directions in Cancers. Front. Oncol. 2020, 10, 598817. [Google Scholar] [CrossRef] [PubMed]
- Nishiyama, A.; Nakanishi, M. Navigating the DNA Methylation Landscape of Cancer. Trends Genet. 2021, 37, 1012–1027. [Google Scholar] [CrossRef] [PubMed]
- Bure, I.V.; Nemtsova, M.V.; Kuznetsova, E.B. Histone Modifications and Non-Coding RNAs: Mutual Epigenetic Regulation and Role in Pathogenesis. Int. J. Mol. Sci. 2022, 23, 5801. [Google Scholar] [CrossRef]
- Jankowska, A.M.; Millward, C.L.; Caldwell, C.W. The Potential of DNA Modifications as Biomarkers and Therapeutic Targets in Oncology. Expert Rev. Mol. Diagn. 2015, 15, 1325–1337. [Google Scholar] [CrossRef]
- Dumitrescu, R.G. Early Epigenetic Markers for Precision Medicine. Methods Mol. Biol. 2018, 1856, 3–17. [Google Scholar] [CrossRef]
- Pan, Y.; Liu, G.; Zhou, F.; Su, B.; Li, Y. DNA Methylation Profiles in Cancer Diagnosis and Therapeutics. Clin. Exp. Med. 2018, 18, 1–14. [Google Scholar] [CrossRef]
- Constâncio, V.; Nunes, S.P.; Henrique, R.; Jerónimo, C. DNA Methylation-Based Testing in Liquid Biopsies as Detection and Prognostic Biomarkers for the Four Major Cancer Types. Cells 2020, 9, 624. [Google Scholar] [CrossRef]
- Ahmed, Y.W.; Alemu, B.A.; Bekele, S.A.; Gizaw, S.T.; Zerihun, M.F.; Wabalo, E.K.; Teklemariam, M.D.; Mihrete, T.K.; Hanurry, E.Y.; Amogne, T.G.; et al. Epigenetic Tumor Heterogeneity in the Era of Single-Cell Profiling with Nanopore Sequencing. Clin. Epigenetics 2022, 14, 107. [Google Scholar] [CrossRef] [PubMed]
- Gatta, G.; Capocaccia, R.; Botta, L.; Mallone, S.; De Angelis, R.; Ardanaz, E.; Comber, H.; Dimitrova, N.; Leinonen, M.K.; Siesling, S.; et al. Burden and Centralised Treatment in Europe of Rare Tumours: Results of RARECAREnet—A Population-Based Study. Lancet Oncol. 2017, 18, 1022–1039. [Google Scholar] [CrossRef] [PubMed]
- Minervini, F.; Kocher, G.J. When to Suspect a Thymoma: Clinical Point of View. J. Thorac. Dis. 2020, 12, 7613–7618. [Google Scholar] [CrossRef] [PubMed]
- Ea, E.; Rm, P. Malignant Thymoma in the United States: Demographic Patterns in Incidence and Associations with Subsequent Malignancies. Int. J. Cancer 2003, 105, 546–551. [Google Scholar] [CrossRef]
- Engels, E.A. Epidemiology of Thymoma and Associated Malignancies. J. Thorac. Oncol. 2010, 5, S260–S265. [Google Scholar] [CrossRef]
- Hsu, C.-H.; Chan, J.K.; Yin, C.-H.; Lee, C.-C.; Chern, C.-U.; Liao, C.-I. Trends in the Incidence of Thymoma, Thymic Carcinoma, and Thymic Neuroendocrine Tumor in the United States. PLoS ONE 2019, 14, e0227197. [Google Scholar] [CrossRef]
- Fang, W.; Fu, J.; Shen, Y.; Wei, Y.; Tan, L.; Zhang, P.; Han, Y.; Chen, C.; Zhang, R.; Li, Y.; et al. Management of Thymic Tumors–Consensus Based on the Chinese Alliance for Research in Thymomas Multi-institutional Retrospective Studies. Zhongguo Fei Ai Za Zhi 2016, 19, 414–417. [Google Scholar] [CrossRef]
- Jeong, D.Y.; Lee, K.S.; Chung, M.J.; Zo, J.I.; Shim, Y.M.; Moon, J.W. JOURNAL CLUB: Doubling Time of Thymic Epithelial Tumors Correlates with World Health Organization Histopathologic Classification. AJR Am. J. Roentgenol. 2017, 209, W202–W210. [Google Scholar] [CrossRef]
- Valavanis, C.; Stanc, G.M.; Baltayiannis, N. Classification, Histopathology and Molecular Pathology of Thymic Epithelial Tumors: A Review. J. BUON 2021, 26, 1198–1207. [Google Scholar]
- Marx, A.; Ströbel, P.; Badve, S.S.; Chalabreysse, L.; Chan, J.K.C.; Chen, G.; de Leval, L.; Detterbeck, F.; Girard, N.; Huang, J.; et al. ITMIG Consensus Statement on the Use of the WHO Histological Classification of Thymoma and Thymic Carcinoma: Refined Definitions, Histological Criteria, and Reporting. J. Thorac. Oncol. 2014, 9, 596–611. [Google Scholar] [CrossRef]
- Zhou, Q.; Huang, X.; Xue, C.; Zhou, J. Correlation of Clinical and Computed Tomography Features of Thymic Epithelial Tumours with World Health Organization Classification and Masaoka-Koga Staging. Eur. J. Cardiothorac. Surg. 2022, 61, 742–748. [Google Scholar] [CrossRef] [PubMed]
- Detterbeck, F.C.; Stratton, K.; Giroux, D.; Asamura, H.; Crowley, J.; Falkson, C.; Filosso, P.L.; Frazier, A.A.; Giaccone, G.; Huang, J.; et al. The IASLC/ITMIG Thymic Epithelial Tumors Staging Project: Proposal for an Evidence-Based Stage Classification System for the Forthcoming (8th) Edition of the TNM Classification of Malignant Tumors. J. Thorac. Oncol. 2014, 9, S65–S72. [Google Scholar] [CrossRef] [PubMed]
- Markowiak, T.; Hofmann, H.-S.; Ried, M. Classification and Staging of Thymoma. J. Thorac. Dis. 2020, 12, 7607–7612. [Google Scholar] [CrossRef]
- Kang, G.; Yoon, N.; Han, J.; Kim, Y.E.; Kim, T.S.; Kim, K. Metaplastic Thymoma: Report of 4 Cases. Korean J. Pathol. 2012, 46, 92–95. [Google Scholar] [CrossRef]
- Tajima, S.; Koda, K. A Case Report of Sclerosing Thymoma of the Anterior Mediastinum: An Exceedingly Rare Morphology. Int. J. Clin. Exp. Pathol. 2015, 8, 4233–4237. [Google Scholar] [PubMed]
- Qu, L.; Xiong, Y.; Yao, Q.; Zhang, B.; Li, T. Micronodular Thymoma with Lymphoid Stroma: Two Cases, One in a Multilocular Thymic Cyst, and Literature Review. Thorac. Cancer 2017, 8, 734–740. [Google Scholar] [CrossRef]
- Yang, L.; Zeng, Q.; Du, X.; Li, W.; Liu, L.; Wang, X.; Zhou, Y.; Ying, J.; Feng, Q. Sclerosing Thymoma: A Case Report and Literature Review. Transl. Cancer Res. 2020, 9, 3034–3039. [Google Scholar] [CrossRef] [PubMed]
- Scorsetti, M.; Leo, F.; Trama, A.; D’Angelillo, R.; Serpico, D.; Macerelli, M.; Zucali, P.; Gatta, G.; Garassino, M.C. Thymoma and Thymic Carcinomas. Crit. Rev. Oncol. Hematol. 2016, 99, 332–350. [Google Scholar] [CrossRef]
- Li, J.; Liu, Y.; Zhang, X.; Zheng, X.; Qi, G. Prognostic Factors for Overall Survival after Surgical Resection in Patients with Thymic Epithelial Tumors: A Systematic Review and Meta-Analysis. Medicine 2022, 101, e30867. [Google Scholar] [CrossRef]
- Alkaaki, A.; Abo Al-Saud, A.; Di Lena, É.; Ramirez-GarciaLuna, J.L.; Najmeh, S.; Spicer, J.; Ferri, L.; Mulder, D.; Sirois, C.; Cools-Lartigue, J. Factors Predicting Recurrence in Thymic Epithelial Neoplasms. Eur. J. Cardiothorac. Surg. 2022, 62, ezac274. [Google Scholar] [CrossRef]
- Karlin, K.; Michaels, P.D. Thymic Carcinoma: Review and Update. J. Cancer Metastasis Treat. 2022, 8, 15. [Google Scholar] [CrossRef]
- Filosso, P.L.; Ruffini, E.; Solidoro, P.; Roffinella, M.; Lausi, P.O.; Lyberis, P.; Oliaro, A.; Guerrera, F. Neuroendocrine Tumors of the Thymus. J. Thorac. Dis. 2017, 9, S1484–S1490. [Google Scholar] [CrossRef] [PubMed]
- Dinter, H.; Bohnenberger, H.; Beck, J.; Bornemann-Kolatzki, K.; Schütz, E.; Küffer, S.; Klein, L.; Franks, T.J.; Roden, A.; Emmert, A.; et al. Molecular Classification of Neuroendocrine Tumors of the Thymus. J. Thorac. Oncol. 2019, 14, 1472–1483. [Google Scholar] [CrossRef] [PubMed]
- Vernino, S.; Lennon, V.A. Autoantibody Profiles and Neurological Correlations of Thymoma. Clin. Cancer Res. 2004, 10, 7270–7275. [Google Scholar] [CrossRef] [PubMed]
- Bernard, C.; Frih, H.; Pasquet, F.; Kerever, S.; Jamilloux, Y.; Tronc, F.; Guibert, B.; Isaac, S.; Devouassoux, M.; Chalabreysse, L.; et al. Thymoma Associated with Autoimmune Diseases: 85 Cases and Literature Review. Autoimmun. Rev. 2016, 15, 82–92. [Google Scholar] [CrossRef] [PubMed]
- Zekeridou, A.; McKeon, A.; Lennon, V.A. Frequency of Synaptic Autoantibody Accompaniments and Neurological Manifestations of Thymoma. JAMA Neurol. 2016, 73, 853–859. [Google Scholar] [CrossRef]
- Padda, S.K.; Yao, X.; Antonicelli, A.; Riess, J.W.; Shang, Y.; Shrager, J.B.; Korst, R.; Detterbeck, F.; Huang, J.; Burt, B.M.; et al. Paraneoplastic Syndromes and Thymic Malignancies: An Examination of the International Thymic Malignancy Interest Group Retrospective Database. J. Thorac. Oncol. 2018, 13, 436–446. [Google Scholar] [CrossRef]
- Conforti, F.; Pala, L.; Giaccone, G.; De Pas, T. Thymic Epithelial Tumors: From Biology to Treatment. Cancer Treat. Rev. 2020, 86, 102014. [Google Scholar] [CrossRef]
- Takaba, H.; Takayanagi, H. The Mechanisms of T Cell Selection in the Thymus. Trends Immunol. 2017, 38, 805–816. [Google Scholar] [CrossRef]
- Marx, A.; Hohenberger, P.; Hoffmann, H.; Pfannschmidt, J.; Schnabel, P.; Hofmann, H.-S.; Wiebe, K.; Schalke, B.; Nix, W.; Gold, R.; et al. The Autoimmune Regulator AIRE in Thymoma Biology: Autoimmunity and Beyond. J. Thorac. Oncol. 2010, 5, S266–S272. [Google Scholar] [CrossRef]
- Okumura, M.; Fujii, Y.; Shiono, H.; Inoue, M.; Minami, M.; Utsumi, T.; Kadota, Y.; Sawa, Y. Immunological Function of Thymoma and Pathogenesis of Paraneoplastic Myasthenia Gravis. Gen. Thorac. Cardiovasc. Surg. 2008, 56, 143–150. [Google Scholar] [CrossRef] [PubMed]
- Fujii, Y. The Thymus, Thymoma and Myasthenia Gravis. Surg. Today 2013, 43, 461–466. [Google Scholar] [CrossRef] [PubMed]
- Radovich, M.; Pickering, C.R.; Felau, I.; Ha, G.; Zhang, H.; Jo, H.; Hoadley, K.A.; Anur, P.; Zhang, J.; McLellan, M.; et al. The Integrated Genomic Landscape of Thymic Epithelial Tumors. Cancer Cell 2018, 33, 244–258.e10. [Google Scholar] [CrossRef] [PubMed]
- Saito, M.; Fujiwara, Y.; Asao, T.; Honda, T.; Shimada, Y.; Kanai, Y.; Tsuta, K.; Kono, K.; Watanabe, S.; Ohe, Y.; et al. The Genomic and Epigenomic Landscape in Thymic Carcinoma. Carcinogenesis 2017, 38, 1084–1091. [Google Scholar] [CrossRef]
- Uccella, S.; La Rosa, S.; Metovic, J.; Marchiori, D.; Scoazec, J.-Y.; Volante, M.; Mete, O.; Papotti, M. Genomics of High-Grade Neuroendocrine Neoplasms: Well-Differentiated Neuroendocrine Tumor with High-Grade Features (G3 NET) and Neuroendocrine Carcinomas (NEC) of Various Anatomic Sites. Endocr. Pathol. 2021, 32, 192–210. [Google Scholar] [CrossRef]
- Volante, M.; Mete, O.; Pelosi, G.; Roden, A.C.; Speel, E.J.M.; Uccella, S. Molecular Pathology of Well-Differentiated Pulmonary and Thymic Neuroendocrine Tumors: What Do Pathologists Need to Know? Endocr. Pathol. 2021, 32, 154–168. [Google Scholar] [CrossRef]
- Gentili, F.; Monteleone, I.; Mazzei, F.G.; Luzzi, L.; Del Roscio, D.; Guerrini, S.; Volterrani, L.; Mazzei, M.A. Advancement in Diagnostic Imaging of Thymic Tumors. Cancers 2021, 13, 3599. [Google Scholar] [CrossRef]
- Strange, C.D.; Ahuja, J.; Shroff, G.S.; Truong, M.T.; Marom, E.M. Imaging Evaluation of Thymoma and Thymic Carcinoma. Front. Oncol. 2021, 11, 810419. [Google Scholar] [CrossRef]
- Agrafiotis, A.C.; Siozopoulou, V.; Hendriks, J.M.H.; Pauwels, P.; Koljenovic, S.; Van Schil, P.E. Prognostic Factors and Genetic Markers in Thymic Epithelial Tumors: A Narrative Review. Thorac. Cancer 2022, 13, 3242–3249. [Google Scholar] [CrossRef]
- Conforti, F.; Marino, M.; Vitolo, V.; Spaggiari, L.; Mantegazza, R.; Zucali, P.; Ruffini, E.; di Tommaso, L.; Pelosi, G.; Barberis, M.; et al. Clinical Management of Patients with Thymic Epithelial Tumors: The Recommendations Endorsed by the Italian Association of Medical Oncology (AIOM). ESMO Open 2021, 6, 100188. [Google Scholar] [CrossRef]
- Zucali, P.A.; De Vincenzo, F.; Perrino, M.; Digiacomo, N.; Cordua, N.; D’Antonio, F.; Borea, F.; Santoro, A. Systemic Treatments for Thymic Tumors: A Narrative Review. Mediastinum 2021, 5, 24. [Google Scholar] [CrossRef] [PubMed]
- Rajan, A.; Heery, C.R.; Thomas, A.; Mammen, A.L.; Perry, S.; O’Sullivan Coyne, G.; Guha, U.; Berman, A.; Szabo, E.; Madan, R.A.; et al. Efficacy and Tolerability of Anti-Programmed Death-Ligand 1 (PD-L1) Antibody (Avelumab) Treatment in Advanced Thymoma. J. Immunother. Cancer 2019, 7, 269. [Google Scholar] [CrossRef] [PubMed]
- Falkson, C.B.; Vella, E.T.; Ellis, P.M.; Maziak, D.E.; Ung, Y.C.; Yu, E. Surgical, Radiation, and Systemic Treatments of Patients with Thymic Epithelial Tumors: A Systematic Review. J. Thorac. Oncol. 2022; in press. [Google Scholar] [CrossRef]
- Falkson, C.B.; Vella, E.T.; Ellis, P.M.; Maziak, D.E.; Ung, Y.C.; Yu, E. Surgical, Radiation, and Systemic Treatments of Patients with Thymic Epithelial Tumours: A Clinical Practice Guideline. J. Thorac. Oncol. 2022; in press. [Google Scholar] [CrossRef]
- Baxter, E.; Windloch, K.; Gannon, F.; Lee, J.S. Epigenetic Regulation in Cancer Progression. Cell Biosci. 2014, 4, 45. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Qi, X.; Liu, L.; Hu, X.; Liu, J.; Yang, J.; Yang, J.; Lu, L.; Zhang, Z.; Ma, S.; et al. Emerging Epigenetic Regulation of Circular RNAs in Human Cancer. Mol. Ther. Nucleic Acids 2019, 16, 589–596. [Google Scholar] [CrossRef] [PubMed]
- Nowak, E.; Bednarek, I. Aspects of the Epigenetic Regulation of EMT Related to Cancer Metastasis. Cells 2021, 10, 3435. [Google Scholar] [CrossRef] [PubMed]
- Rotondo, J.C.; Mazziotta, C.; Lanzillotti, C.; Tognon, M.; Martini, F. Epigenetic Dysregulations in Merkel Cell Polyomavirus-Driven Merkel Cell Carcinoma. Int. J. Mol. Sci. 2021, 22, 11464. [Google Scholar] [CrossRef]
- Hyun, K.; Jeon, J.; Park, K.; Kim, J. Writing, Erasing and Reading Histone Lysine Methylations. Exp. Mol. Med. 2017, 49, e324. [Google Scholar] [CrossRef]
- Biswas, S.; Rao, C.M. Epigenetic Tools (The Writers, The Readers and The Erasers) and Their Implications in Cancer Therapy. Eur. J. Pharmacol. 2018, 837, 8–24. [Google Scholar] [CrossRef]
- Lu, Y.; Chan, Y.-T.; Tan, H.-Y.; Li, S.; Wang, N.; Feng, Y. Epigenetic Regulation in Human Cancer: The Potential Role of Epi-Drug in Cancer Therapy. Mol. Cancer 2020, 19, 79. [Google Scholar] [CrossRef]
- Lyko, F. The DNA Methyltransferase Family: A Versatile Toolkit for Epigenetic Regulation. Nat. Rev. Genet. 2018, 19, 81–92. [Google Scholar] [CrossRef]
- Barau, J.; Teissandier, A.; Zamudio, N.; Roy, S.; Nalesso, V.; Hérault, Y.; Guillou, F.; Bourc’his, D. The DNA Methyltransferase DNMT3C Protects Male Germ Cells from Transposon Activity. Science 2016, 354, 909–912. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.; Xie, T.; Wang, Z.; Wang, X.; Zeng, S.; Kang, Y.; Hou, T. DNA Methyltransferases: Emerging Targets for the Discovery of Inhibitors as Potent Anticancer Drugs. Drug Discov. Today 2019, 24, 2323–2331. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Zhu, D.; Wu, J.; Ma, Y.; Cai, C.; Chen, Y.; Qin, M.; Dai, H. New Substrates and Determinants for TRNA Recognition of RNA Methyltransferase DNMT2/TRDMT1. RNA Biol. 2021, 18, 2531–2545. [Google Scholar] [CrossRef] [PubMed]
- Kaiser, S.; Jurkowski, T.P.; Kellner, S.; Schneider, D.; Jeltsch, A.; Helm, M. The RNA Methyltransferase Dnmt2 Methylates DNA in the Structural Context of a TRNA. RNA Biol. 2017, 14, 1241–1251. [Google Scholar] [CrossRef]
- Rasmussen, K.D.; Helin, K. Role of TET Enzymes in DNA Methylation, Development, and Cancer. Genes Dev. 2016, 30, 733–750. [Google Scholar] [CrossRef]
- Jimenez-Useche, I.; Yuan, C. The Effect of DNA CpG Methylation on the Dynamic Conformation of a Nucleosome. Biophys. J. 2012, 103, 2502–2512. [Google Scholar] [CrossRef]
- Blackledge, N.P.; Klose, R. CpG Island Chromatin: A Platform for Gene Regulation. Epigenetics 2011, 6, 147–152. [Google Scholar] [CrossRef]
- Rao, S.; Chiu, T.-P.; Kribelbauer, J.F.; Mann, R.S.; Bussemaker, H.J.; Rohs, R. Systematic Prediction of DNA Shape Changes Due to CpG Methylation Explains Epigenetic Effects on Protein-DNA Binding. Epigenetics Chromatin 2018, 11, 6. [Google Scholar] [CrossRef]
- Buitrago, D.; Labrador, M.; Arcon, J.P.; Lema, R.; Flores, O.; Esteve-Codina, A.; Blanc, J.; Villegas, N.; Bellido, D.; Gut, M.; et al. Impact of DNA Methylation on 3D Genome Structure. Nat. Commun. 2021, 12, 3243. [Google Scholar] [CrossRef]
- Greenberg, M.V.C.; Bourc’his, D. The Diverse Roles of DNA Methylation in Mammalian Development and Disease. Nat. Rev. Mol. Cell Biol. 2019, 20, 590–607. [Google Scholar] [CrossRef]
- Rodriguez-Casanova, A.; Costa-Fraga, N.; Castro-Carballeira, C.; González-Conde, M.; Abuin, C.; Bao-Caamano, A.; García-Caballero, T.; Brozos-Vazquez, E.; Rodriguez-López, C.; Cebey, V.; et al. A Genome-Wide Cell-Free DNA Methylation Analysis Identifies an Episignature Associated with Metastatic Luminal B Breast Cancer. Front. Cell Dev. Biol. 2022, 10, 1016955. [Google Scholar] [CrossRef] [PubMed]
- Stuckel, A.J.; Zeng, S.; Lyu, Z.; Zhang, W.; Zhang, X.; Dougherty, U.; Mustafi, R.; Khare, T.; Zhang, Q.; Joshi, T.; et al. Sprouty4 Is Epigenetically Upregulated in Human Colorectal Cancer. Epigenetics, 2022; ahead of print. [Google Scholar] [CrossRef]
- Dong, Z.; Zhou, H. Pan-Cancer Landscape of Aberrant DNA Methylation across Childhood Cancers: Molecular Characteristics and Clinical Relevance. Exp. Hematol. Oncol. 2022, 11, 89. [Google Scholar] [CrossRef] [PubMed]
- Boot, J.; Rosser, G.; Kancheva, D.; Vinel, C.; Lim, Y.M.; Pomella, N.; Zhang, X.; Guglielmi, L.; Sheer, D.; Barnes, M.; et al. Global Hypo-Methylation in a Proportion of Glioblastoma Enriched for an Astrocytic Signature Is Associated with Increased Invasion and Altered Immune Landscape. eLife 2022, 11, e77335. [Google Scholar] [CrossRef] [PubMed]
- Weisenberger, D.J.; Lakshminarasimhan, R.; Liang, G. The Role of DNA Methylation and DNA Methyltransferases in Cancer. Adv. Exp. Med. Biol. 2022, 1389, 317–348. [Google Scholar] [CrossRef]
- Chen, S.; Petricca, J.; Ye, W.; Guan, J.; Zeng, Y.; Cheng, N.; Gong, L.; Shen, S.Y.; Hua, J.T.; Crumbaker, M.; et al. The Cell-Free DNA Methylome Captures Distinctions between Localized and Metastatic Prostate Tumors. Nat. Commun. 2022, 13, 6467. [Google Scholar] [CrossRef]
- Kurumizaka, H.; Kujirai, T.; Takizawa, Y. Contributions of Histone Variants in Nucleosome Structure and Function. J. Mol. Biol. 2021, 433, 166678. [Google Scholar] [CrossRef]
- Hergeth, S.P.; Schneider, R. The H1 Linker Histones: Multifunctional Proteins beyond the Nucleosomal Core Particle. EMBO Rep. 2015, 16, 1439–1453. [Google Scholar] [CrossRef]
- Kouzarides, T. Chromatin Modifications and Their Function. Cell 2007, 128, 693–705. [Google Scholar] [CrossRef]
- Yang, Y.; Zhang, M.; Wang, Y. The Roles of Histone Modifications in Tumorigenesis and Associated Inhibitors in Cancer Therapy. J. Natl. Cancer Cent. 2022, 2, 277–290. [Google Scholar] [CrossRef]
- Chen, Z.; Tyler, J.K. The Chromatin Landscape Channels DNA Double-Strand Breaks to Distinct Repair Pathways. Front. Cell Dev. Biol. 2022, 10, 909696. [Google Scholar] [CrossRef]
- Allshire, R.C.; Madhani, H.D. Ten Principles of Heterochromatin Formation and Function. Nat. Rev. Mol. Cell Biol. 2018, 19, 229–244. [Google Scholar] [CrossRef] [PubMed]
- Morgan, M.A.J.; Shilatifard, A. Reevaluating the Roles of Histone-Modifying Enzymes and Their Associated Chromatin Modifications in Transcriptional Regulation. Nat. Genet. 2020, 52, 1271–1281. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Gonzalez, E.A.; Rameshwar, P.; Etchegaray, J.-P. Non-Coding RNAs as Mediators of Epigenetic Changes in Malignancies. Cancers 2020, 12, 3657. [Google Scholar] [CrossRef] [PubMed]
- Statello, L.; Guo, C.-J.; Chen, L.-L.; Huarte, M. Gene Regulation by Long Non-Coding RNAs and Its Biological Functions. Nat. Rev. Mol. Cell Biol. 2021, 22, 96–118. [Google Scholar] [CrossRef]
- Panda, A.C. Circular RNAs Act as MiRNA Sponges. Adv. Exp. Med. Biol. 2018, 1087, 67–79. [Google Scholar] [CrossRef] [PubMed]
- Xiong, Q.; Zhang, Y.; Li, J.; Zhu, Q. Small Non-Coding RNAs in Human Cancer. Genes 2022, 13, 2072. [Google Scholar] [CrossRef] [PubMed]
- Cheloufi, S.; Dos Santos, C.O.; Chong, M.M.W.; Hannon, G.J. A Dicer-Independent MiRNA Biogenesis Pathway That Requires Ago Catalysis. Nature 2010, 465, 584–589. [Google Scholar] [CrossRef] [PubMed]
- Müller, M.; Fazi, F.; Ciaudo, C. Argonaute Proteins: From Structure to Function in Development and Pathological Cell Fate Determination. Front. Cell Dev. Biol. 2020, 7, 360. [Google Scholar] [CrossRef] [PubMed]
- Yao, Q.; Chen, Y.; Zhou, X. The Roles of MicroRNAs in Epigenetic Regulation. Curr. Opin. Chem. Biol. 2019, 51, 11–17. [Google Scholar] [CrossRef]
- Heydarnezhad Asl, M.; Pasban Khelejani, F.; Bahojb Mahdavi, S.Z.; Emrahi, L.; Jebelli, A.; Mokhtarzadeh, A. The Various Regulatory Functions of Long Noncoding RNAs in Apoptosis, Cell Cycle, and Cellular Senescence. J. Cell. Biochem. 2022, 123, 995–1024. [Google Scholar] [CrossRef]
- Hirabayashi, H.; Fujii, Y.; Sakaguchi, M.; Tanaka, H.; Yoon, H.E.; Komoto, Y.; Inoue, M.; Miyoshi, S.; Matsuda, H. P16INK4, PRB, P53 and Cyclin D1 Expression and Hypermethylation of CDKN2 Gene in Thymoma and Thymic Carcinoma. Int. J. Cancer 1997, 73, 639–644. [Google Scholar] [CrossRef]
- Suzuki, M.; Chen, H.; Shigematsu, H.; Ando, S.; Iida, T.; Nakajima, T.; Fujisawa, T.; Kimura, H. Aberrant Methylation: Common in Thymic Carcinomas, Rare in Thymomas. Oncol. Rep. 2005, 14, 1621–1624. [Google Scholar] [CrossRef] [PubMed]
- Gu, Z.; Mitsui, H.; Inomata, K.; Honda, M.; Endo, C.; Sakurada, A.; Sato, M.; Okada, Y.; Kondo, T.; Horii, A. The Methylation Status of FBXW7 Beta-Form Correlates with Histological Subtype in Human Thymoma. Biochem. Biophys. Res. Commun. 2008, 377, 685–688. [Google Scholar] [CrossRef]
- Hirose, Y.; Kondo, K.; Takizawa, H.; Nagao, T.; Nakagawa, Y.; Fujino, H.; Toba, H.; Kenzaki, K.; Sakiyama, S.; Tangoku, A. Aberrant Methylation of Tumour-Related Genes in Thymic Epithelial Tumours. Lung Cancer 2009, 64, 155–159. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Yin, B.; Wei, Q.; Li, D.; Hu, J.; Yu, F.; Lu, Q. Aberrant DNA Methylation in Thymic Epithelial Tumors. Cancer Investig. 2009, 27, 582–591. [Google Scholar] [CrossRef]
- Mokhtar, M.; Kondo, K.; Namura, T.; Ali, A.H.K.; Fujita, Y.; Takai, C.; Takizawa, H.; Nakagawa, Y.; Toba, H.; Kajiura, K.; et al. Methylation and Expression Profiles of MGMT Gene in Thymic Epithelial Tumors. Lung Cancer 2014, 83, 279–287. [Google Scholar] [CrossRef]
- Lopomo, A.; Ricciardi, R.; Maestri, M.; De Rosa, A.; Melfi, F.; Lucchi, M.; Mussi, A.; Coppedè, F.; Migliore, L. Gene-Specific Methylation Analysis in Thymomas of Patients with Myasthenia Gravis. Int. J. Mol. Sci. 2016, 17, 2121. [Google Scholar] [CrossRef]
- Kajiura, K.; Takizawa, H.; Morimoto, Y.; Masuda, K.; Tsuboi, M.; Kishibuchi, R.; Wusiman, N.; Sawada, T.; Kawakita, N.; Toba, H.; et al. Frequent Silencing of RASSF1A by DNA Methylation in Thymic Neuroendocrine Tumours. Lung Cancer 2017, 111, 116–123. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Yuan, Y.; Xiao, H.; Dai, J.; Ye, Y.; Zhang, Q.; Zhang, Z.; Jiang, Y.; Luo, J.; Hu, J.; et al. Discovery and Validation of DNA Methylation Markers for Overall Survival Prognosis in Patients with Thymic Epithelial Tumors. Clin. Epigenetics 2019, 11, 38. [Google Scholar] [CrossRef]
- Bi, Y.; Meng, Y.; Niu, Y.; Li, S.; Liu, H.; He, J.; Zhang, Y.; Liang, N.; Liu, L.; Mao, X.; et al. Genome-Wide DNA Methylation Profile of Thymomas and Potential Epigenetic Regulation of Thymoma Subtypes. Oncol. Rep. 2019, 41, 2762–2774. [Google Scholar] [CrossRef]
- Coppedè, F.; Ricciardi, R.; Lopomo, A.; Stoccoro, A.; De Rosa, A.; Guida, M.; Petrucci, L.; Maestri, M.; Lucchi, M.; Migliore, L. Investigation of MLH1, MGMT, CDKN2A, and RASSF1A Gene Methylation in Thymomas From Patients With Myasthenia Gravis. Front. Mol. Neurosci. 2020, 13, 567676. [Google Scholar] [CrossRef]
- Coppedè, F.; Stoccoro, A.; Nicolì, V.; Gallo, R.; De Rosa, A.; Guida, M.; Maestri, M.; Lucchi, M.; Ricciardi, R.; Migliore, L. Investigation of GHSR Methylation Levels in Thymomas from Patients with Myasthenia Gravis. Gene 2020, 752, 144774. [Google Scholar] [CrossRef]
- Kishibuchi, R.; Kondo, K.; Soejima, S.; Tsuboi, M.; Kajiura, K.; Kawakami, Y.; Kawakita, N.; Sawada, T.; Toba, H.; Yoshida, M.; et al. DNA Methylation of GHSR, GNG4, HOXD9 and SALL3 Is a Common Epigenetic Alteration in Thymic Carcinoma. Int. J. Oncol. 2020, 56, 315–326. [Google Scholar] [CrossRef] [PubMed]
- Yang, Z.; Liu, S.; Wang, Y.; Chen, Y.; Zhang, P.; Liu, Y.; Zhang, H.; Zhang, P.; Tao, Z.; Xiong, K. High Expression of KITLG Is a New Hallmark Activating the MAPK Pathway in Type A and AB Thymoma. Thorac. Cancer 2020, 11, 1944–1954. [Google Scholar] [CrossRef] [PubMed]
- Yan, M.; Wu, J.; Xue, M.; Mo, J.; Zheng, L.; Zhang, J.; Gao, Z.; Bao, Y. The Studies of Prognostic Factors and the Genetic Polymorphism of Methylenetetrahydrofolate Reductase C667T in Thymic Epithelial Tumors. Front. Oncol. 2022, 12, 847957. [Google Scholar] [CrossRef] [PubMed]
- Soejima, S.; Kondo, K.; Tsuboi, M.; Muguruma, K.; Tegshee, B.; Kawakami, Y.; Kajiura, K.; Kawakita, N.; Toba, H.; Yoshida, M.; et al. GAD1 Expression and Its Methylation as Indicators of Malignant Behavior in Thymic Epithelial Tumors. Oncol. Lett. 2021, 21, 483. [Google Scholar] [CrossRef]
- Liu, D.; Zhang, P.; Zhao, J.; Yang, L.; Wang, W. Identification of Molecular Characteristics and New Prognostic Targets for Thymoma by Multiomics Analysis. BioMed Res. Int. 2021, 2021, e5587441. [Google Scholar] [CrossRef] [PubMed]
- Guan, W.; Li, S.; Zhang, Z.; Xiao, H.; He, J.; Li, J.; He, X.; Luo, J.; Liu, Y.; Lei, L.; et al. Promotor Methylation Status of MAPK4 Is a Novel Epigenetic Biomarker for Prognosis of Recurrence in Patients with Thymic Epithelial Tumors. Thorac. Cancer 2022, 13, 2844–2853. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Li, Q.; Sun, Q.; Wang, B.; Cui, Y.; Lou, C.; Yao, Y.; Zhang, Y. Epigenetic Modifications Inhibit the Expression of MARVELD1 and in Turn Tumorigenesis by Regulating the Wnt/β-Catenin Pathway in Pan-Cancer. J. Cancer 2022, 13, 225–242. [Google Scholar] [CrossRef] [PubMed]
- Waters, C.E.; Saldivar, J.C.; Hosseini, S.A.; Huebner, K. The FHIT Gene Product: Tumor Suppressor and Genome ‘Caretaker’. Cell. Mol. Life Sci. 2014, 71, 4577–4587. [Google Scholar] [CrossRef] [PubMed]
- Qiang, L.; Shah, P.; Barcellos-Hoff, M.H.; He, Y.Y. TGF-β Signaling Links E-Cadherin Loss to Suppression of Nucleotide Excision Repair. Oncogene 2016, 35, 3293–3302. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Liu, Y.; Sun, H.; Ge, A.; Li, D.; Fu, J.; Li, Y.; Pang, D.; Zhao, Y. DNA Methylation in RARβ Gene as a Mediator of the Association Between Healthy Lifestyle and Breast Cancer: A Case–Control Study. Cancer Manag. Res. 2020, 12, 4677–4684. [Google Scholar] [CrossRef] [PubMed]
- Ortiz-Barahona, V.; Joshi, R.S.; Esteller, M. Use of DNA Methylation Profiling in Translational Oncology. Semin. Cancer Biol. 2022, 83, 523–535. [Google Scholar] [CrossRef] [PubMed]
- Botezatu, A.; Socolov, D.; Iancu, I.V.; Huica, I.; Plesa, A.; Ungureanu, C.; Anton, G. Methylenetetrahydrofolate Reductase (MTHFR) Polymorphisms and Promoter Methylation in Cervical Oncogenic Lesions and Cancer. J. Cell. Mol. Med. 2013, 17, 543–549. [Google Scholar] [CrossRef] [PubMed]
- Ferlazzo, N.; Currò, M.; Zinellu, A.; Caccamo, D.; Isola, G.; Ventura, V.; Carru, C.; Matarese, G.; Ientile, R. Influence of MTHFR Genetic Background on P16 and MGMT Methylation in Oral Squamous Cell Cancer. Int. J. Mol. Sci. 2017, 18, 724. [Google Scholar] [CrossRef]
- Coppedè, F.; Stoccoro, A.; Tannorella, P.; Gallo, R.; Nicolì, V.; Migliore, L. Association of Polymorphisms in Genes Involved in One-Carbon Metabolism with MTHFR Methylation Levels. Int. J. Mol. Sci. 2019, 20, 3754. [Google Scholar] [CrossRef]
- Xiong, M.; Pan, B.; Wang, X.; Nie, J.; Pan, Y.; Sun, H.; Xu, T.; Cho, W.C.S.; Wang, S.; He, B. Susceptibility of Genetic Variations in Methylation Pathway to Gastric Cancer. Pharmgenomics Pers. Med. 2022, 15, 441–448. [Google Scholar] [CrossRef]
- Lin, T.-C.; Hsiao, M. Ghrelin and Cancer Progression. Biochim. Biophys. Acta (BBA) Rev. Cancer 2017, 1868, 51–57. [Google Scholar] [CrossRef]
- Tegshee, B.; Kondo, K.; Soejima, S.; Muguruma, K.; Tsuboi, M.; Kajiura, K.; Kawakami, Y.; Kawakita, N.; Toba, H.; Yoshida, M.; et al. GHSR Methylation-Dependent Expression of a Variant Ligand and Receptor of the Ghrelin System Induces Thymoma Tumorigenesis. Oncol. Lett. 2021, 22, 793. [Google Scholar] [CrossRef]
- Kont, V.; Murumägi, A.; Tykocinski, L.-O.; Kinkel, S.A.; Webster, K.E.; Kisand, K.; Tserel, L.; Pihlap, M.; Ströbel, P.; Scott, H.S.; et al. DNA Methylation Signatures of the AIRE Promoter in Thymic Epithelial Cells, Thymomas and Normal Tissues. Mol. Immunol. 2011, 49, 518–526. [Google Scholar] [CrossRef]
- Herzig, Y.; Nevo, S.; Bornstein, C.; Brezis, M.R.; Ben-Hur, S.; Shkedy, A.; Eisenberg-Bord, M.; Levi, B.; Delacher, M.; Goldfarb, Y.; et al. Transcriptional Programs That Control Expression of the Autoimmune Regulator Gene Aire. Nat. Immunol. 2017, 18, 161–172. [Google Scholar] [CrossRef] [PubMed]
- Merino, V.F.; Nguyen, N.; Jin, K.; Sadik, H.; Cho, S.; Korangath, P.; Han, L.; Foster, Y.M.N.; Zhou, X.C.; Zhang, Z.; et al. Combined Treatment with Epigenetic, Differentiating, and Chemotherapeutic Agents Cooperatively Targets Tumor-Initiating Cells in Triple-Negative Breast Cancer. Cancer Res. 2016, 76, 2013–2024. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, A.B.; Zidi, S.; Sghaier, I.; Ghazouani, E.; Mezlini, A.; Almawi, W.; Loueslati, B.Y. Common Variants in IL-1RN, IL-1β and TNF-α and the Risk of Ovarian Cancer: A Case Control Study. Cent. Eur. J. Immunol. 2017, 42, 150–155. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Thomas, A.; Lau, C.; Rajan, A.; Zhu, Y.; Killian, J.K.; Petrini, I.; Pham, T.; Morrow, B.; Zhong, X.; et al. Mutations of Epigenetic Regulatory Genes Are Common in Thymic Carcinomas. Sci. Rep. 2014, 4, 7336. [Google Scholar] [CrossRef]
- Anastasiadou, E.; Jacob, L.S.; Slack, F.J. Non-Coding RNA Networks in Cancer. Nat. Rev. Cancer 2018, 18, 5–18. [Google Scholar] [CrossRef]
- Yan, H.; Bu, P. Non-Coding RNA in Cancer. Essays Biochem. 2021, 65, 625–639. [Google Scholar] [CrossRef]
- Xu, M.; Gan, T.; Ning, H.; Wang, L. MicroRNA Functions in Thymic Biology: Thymic Development and Involution. Front. Immunol. 2018, 9, 2063. [Google Scholar] [CrossRef]
- Huang, B.; Belharazem, D.; Li, L.; Kneitz, S.; Schnabel, P.A.; Rieker, R.J.; Körner, D.; Nix, W.; Schalke, B.; Müller-Hermelink, H.K.; et al. Anti-Apoptotic Signature in Thymic Squamous Cell Carcinomas—Functional Relevance of Anti-Apoptotic BIRC3 Expression in the Thymic Carcinoma Cell Line 1889c. Front. Oncol. 2013, 3, 316. [Google Scholar] [CrossRef]
- Ganci, F.; Vico, C.; Korita, E.; Sacconi, A.; Gallo, E.; Mori, F.; Cambria, A.; Russo, E.; Anile, M.; Vitolo, D.; et al. MicroRNA Expression Profiling of Thymic Epithelial Tumors. Lung Cancer 2014, 85, 197–204. [Google Scholar] [CrossRef]
- Bellissimo, T.; Russo, E.; Ganci, F.; Vico, C.; Sacconi, A.; Longo, F.; Vitolo, D.; Anile, M.; Disio, D.; Marino, M.; et al. Circulating MiR-21-5p and MiR-148a-3p as Emerging Non-Invasive Biomarkers in Thymic Epithelial Tumors. Cancer Biol. Ther. 2016, 17, 79–82. [Google Scholar] [CrossRef][Green Version]
- Wang, Z.; Chen, Y.; Xu, S.; Yang, Y.; Wei, D.; Wang, W.; Huang, X. Aberrant Decrease of MicroRNA19b Regulates TSLP Expression and Contributes to Th17 Cells Development in Myasthenia Gravis Related Thymomas. J. Neuroimmunol. 2015, 288, 34–39. [Google Scholar] [CrossRef] [PubMed]
- Radovich, M.; Solzak, J.P.; Hancock, B.A.; Conces, M.L.; Atale, R.; Porter, R.F.; Zhu, J.; Glasscock, J.; Kesler, K.A.; Badve, S.S.; et al. A Large MicroRNA Cluster on Chromosome 19 Is a Transcriptional Hallmark of WHO Type A and AB Thymomas. Br. J. Cancer 2016, 114, 477–484. [Google Scholar] [CrossRef]
- Xin, Y.; Cai, H.; Lu, T.; Zhang, Y.; Yang, Y.; Cui, Y. MiR-20b Inhibits T Cell Proliferation and Activation via NFAT Signaling Pathway in Thymoma-Associated Myasthenia Gravis. Biomed. Res. Int. 2016, 2016, 9595718. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Qiu, D.; Chen, Z.; Du, W.; Liu, J.; Mo, X. Altered Expression of MiR-125a-5p in Thymoma-Associated Myasthenia Gravis and Its down-Regulation of Foxp3 Expression in Jurkat Cells. Immunol. Lett. 2016, 172, 47–55. [Google Scholar] [CrossRef] [PubMed]
- Bellissimo, T.; Ganci, F.; Gallo, E.; Sacconi, A.; Tito, C.; De Angelis, L.; Pulito, C.; Masciarelli, S.; Diso, D.; Anile, M.; et al. Thymic Epithelial Tumors Phenotype Relies on MiR-145-5p Epigenetic Regulation. Mol. Cancer 2017, 16, 88. [Google Scholar] [CrossRef]
- Enkner, F.; Pichlhöfer, B.; Zaharie, A.T.; Krunic, M.; Holper, T.M.; Janik, S.; Moser, B.; Schlangen, K.; Neudert, B.; Walter, K.; et al. Molecular Profiling of Thymoma and Thymic Carcinoma: Genetic Differences and Potential Novel Therapeutic Targets. Pathol. Oncol. Res. 2017, 23, 551–564. [Google Scholar] [CrossRef]
- Wei, J.; Liu, Z.; Wu, K.; Yang, D.; He, Y.; Chen, G.G.; Zhang, J.; Lin, J. Identification of Prognostic and Subtype-Specific Potential MiRNAs in Thymoma. Epigenomics 2017, 9, 647–657. [Google Scholar] [CrossRef]
- Han, Z.; Zhang, J.; Zhang, K.; Zhao, Y. Curcumin Inhibits Cell Viability, Migration, and Invasion of Thymic Carcinoma Cells via Downregulation of MicroRNA-27a. Phytother. Res. 2020, 34, 1629–1637. [Google Scholar] [CrossRef]
- Wang, B.; Xiao, H.; Yang, X.; Zeng, Y.; Zhang, Z.; Yang, R.; Chen, H.; Chen, C.; Chen, J. A Novel Immune-Related MicroRNA Signature for Prognosis of Thymoma. Aging (Albany NY) 2022, 14, 4739–4754. [Google Scholar] [CrossRef]
- Su, Y.; Chen, Y.; Tian, Z.; Lu, C.; Chen, L.; Ma, X. LncRNAs Classifier to Accurately Predict the Recurrence of Thymic Epithelial Tumors. Thorac. Cancer 2020, 11, 1773–1783. [Google Scholar] [CrossRef]
- Ji, G.; Ren, R.; Fang, X. Identification and Characterization of Non-Coding RNAs in Thymoma. Med. Sci. Monit. 2021, 27, e929727. [Google Scholar] [CrossRef] [PubMed]
- Ke, J.; Du, X.; Cui, J.; Yu, L.; Li, H. LncRNA and MRNA Expression Associated with Myasthenia Gravis in Patients with Thymoma. Thorac. Cancer 2022, 13, 15–23. [Google Scholar] [CrossRef] [PubMed]
- Wu, Q.; Luo, X.; Li, H.; Zhang, L.; Su, F.; Hou, S.; Yin, J.; Zhang, W.; Zou, L. Identification of Differentially Expressed Circular RNAs Associated with Thymoma. Thorac. Cancer 2021, 12, 1312–1319. [Google Scholar] [CrossRef]
- Tito, C.; Ganci, F.; Sacconi, A.; Masciarelli, S.; Fontemaggi, G.; Pulito, C.; Gallo, E.; Laquintana, V.; Iaiza, A.; De Angelis, L.; et al. LINC00174 Is a Novel Prognostic Factor in Thymic Epithelial Tumors Involved in Cell Migration and Lipid Metabolism. Cell Death Dis. 2020, 11, 959. [Google Scholar] [CrossRef] [PubMed]
- Tan, S.; Chen, J. Si-MALAT1 Attenuates Thymic Cancer Cell Proliferation and Promotes Apoptosis via the MiR-145-5p/HMGA2 Pathway. Oncol. Lett. 2021, 22, 585. [Google Scholar] [CrossRef]
- Iaiza, A.; Tito, C.; Ianniello, Z.; Ganci, F.; Laquintana, V.; Gallo, E.; Sacconi, A.; Masciarelli, S.; De Angelis, L.; Aversa, S.; et al. METTL3-Dependent MALAT1 Delocalization Drives c-Myc Induction in Thymic Epithelial Tumors. Clin. Epigenetics 2021, 13, 173. [Google Scholar] [CrossRef]
- Zhuang, J.; Guan, M.; Liu, M.; Liu, Y.; Yang, S.; Hu, Z.; Lai, F.; He, F. Immune-Related Molecular Profiling of Thymoma With Myasthenia Gravis. Front. Genet. 2021, 12, 756493. [Google Scholar] [CrossRef]
- Wang, F.; Zhang, H.; Qiu, G.; Li, Z.; Wang, Y. The LINC00452/MiR-204/CHST4 Axis Regulating Thymic Tregs Might Be Involved in the Progression of Thymoma-Associated Myasthenia Gravis. Front. Neurol. 2022, 13, 828970. [Google Scholar] [CrossRef]
- Wang, J.; Huang, H.; Zhang, X.; Ma, H. LOXL1-AS1 Promotes Thymoma and Thymic Carcinoma Progression by Regulating MiR-525-5p-HSPA9. Oncol. Rep. 2021, 45, 117. [Google Scholar] [CrossRef]
- Noguer-Dance, M.; Abu-Amero, S.; Al-Khtib, M.; Lefèvre, A.; Coullin, P.; Moore, G.E.; Cavaillé, J. The Primate-Specific MicroRNA Gene Cluster (C19MC) Is Imprinted in the Placenta. Hum. Mol. Genet. 2010, 19, 3566–3582. [Google Scholar] [CrossRef]
- Ucar, O.; Tykocinski, L.-O.; Dooley, J.; Liston, A.; Kyewski, B. An Evolutionarily Conserved Mutual Interdependence between Aire and MicroRNAs in Promiscuous Gene Expression. Eur. J. Immunol. 2013, 43, 1769–1778. [Google Scholar] [CrossRef] [PubMed]
- Nadal, E.; Zhong, J.; Lin, J.; Reddy, R.M.; Ramnath, N.; Orringer, M.B.; Chang, A.C.; Beer, D.G.; Chen, G. A MicroRNA Cluster at 14q32 Drives Aggressive Lung Adenocarcinoma. Clin. Cancer Res. 2014, 20, 3107–3117. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Nayak, S.; Pathak, P.; Purkait, S.; Malgulawar, P.B.; Sharma, M.C.; Suri, V.; Mukhopadhyay, A.; Suri, A.; Sarkar, C. Identification of MiR-379/MiR-656 (C14MC) Cluster Downregulation and Associated Epigenetic and Transcription Regulatory Mechanism in Oligodendrogliomas. J. Neurooncol. 2018, 139, 23–31. [Google Scholar] [CrossRef] [PubMed]
- Kabekkodu, S.P.; Shukla, V.; Varghese, V.K.; Adiga, D.; Vethil Jishnu, P.; Chakrabarty, S.; Satyamoorthy, K. Cluster MiRNAs and Cancer: Diagnostic, Prognostic and Therapeutic Opportunities. Wiley Interdiscip. Rev. RNA 2020, 11, e1563. [Google Scholar] [CrossRef] [PubMed]
- Kandettu, A.; Adiga, D.; Devi, V.; Suresh, P.S.; Chakrabarty, S.; Radhakrishnan, R.; Kabekkodu, S.P. Deregulated MiRNA Clusters in Ovarian Cancer: Imperative Implications in Personalized Medicine. Genes Dis. 2022, 9, 1443–1465. [Google Scholar] [CrossRef]
- Saghazadeh, A.; Rezaei, N. MicroRNA Expression Profiles of Peripheral Blood and Mononuclear Cells in Myasthenia Gravis: A Systematic Review. Int. Immunopharmacol. 2022, 112, 109205. [Google Scholar] [CrossRef]
- Ströbel, P.; Rosenwald, A.; Beyersdorf, N.; Kerkau, T.; Elert, O.; Murumägi, A.; Sillanpää, N.; Peterson, P.; Hummel, V.; Rieckmann, P.; et al. Selective Loss of Regulatory T Cells in Thymomas. Ann. Neurol. 2004, 56, 901–904. [Google Scholar] [CrossRef]
- Wang, X.; Liu, C.; Zhang, S.; Yan, H.; Zhang, L.; Jiang, A.; Liu, Y.; Feng, Y.; Li, D.; Guo, Y.; et al. N6-Methyladenosine Modification of MALAT1 Promotes Metastasis via Reshaping Nuclear Speckles. Dev. Cell 2021, 56, 702–715.e8. [Google Scholar] [CrossRef]
- Hu, C.; Zhang, K.; Jiang, F.; Wang, H.; Shao, Q. Epigenetic Modifications in Thymic Epithelial Cells: An Evolutionary Perspective for Thymus Atrophy. Clin. Epigenetics 2021, 13, 210. [Google Scholar] [CrossRef]
- Martinez-Ruíz, G.U.; Morales-Sánchez, A.; Bhandoola, A. Transcriptional and Epigenetic Regulation in Thymic Epithelial Cells. Immunol. Rev. 2022, 305, 43–58. [Google Scholar] [CrossRef]
- Org, T.; Rebane, A.; Kisand, K.; Laan, M.; Haljasorg, U.; Andreson, R.; Peterson, P. AIRE Activated Tissue Specific Genes Have Histone Modifications Associated with Inactive Chromatin. Hum. Mol. Genet. 2009, 18, 4699–4710. [Google Scholar] [CrossRef] [PubMed]
- Holstein, E.; Dittmann, A.; Kääriäinen, A.; Pesola, V.; Koivunen, J.; Pihlajaniemi, T.; Naba, A.; Izzi, V. The Burden of Post-Translational Modification (PTM)-Disrupting Mutations in the Tumor Matrisome. Cancers 2021, 13, 1081. [Google Scholar] [CrossRef] [PubMed]
- Qiu, L.; Meng, Y.; Wang, L.; Gunewardena, S.; Liu, S.; Han, J.; Krieg, A.J. Histone Lysine Demethylase 4B Regulates General and Unique Gene Expression Signatures in Hypoxic Cancer Cells. MedComm (2020) 2021, 2, 414–429. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Zhang, X.; Huang, A. Aggresome-Autophagy Associated Gene HDAC6 Is a Potential Biomarker in Pan-Cancer, Especially in Colon Adenocarcinoma. Front. Oncol. 2021, 11, 718589. [Google Scholar] [CrossRef] [PubMed]
- Geng, F.; Yang, W.; Song, D.; Hou, H.; Han, B.; Chen, Y.; Zhao, H. MDIG, a 2-oxoglutarate-dependent Oxygenase, Acts as an Oncogene and Predicts the Prognosis of Multiple Types of Cancer. Int. J. Oncol. 2022, 61, 82. [Google Scholar] [CrossRef]
- Kim, H.-R.; Seo, C.-W.; Kim, J. Zinc Finger E-Box Binding Homeobox 1 as Prognostic Biomarker and Its Correlation with Infiltrating Immune Cells and Telomerase in Lung Cancer. Biomed. Sci. Lett. 2022, 28, 9–24. [Google Scholar] [CrossRef]
- Yang, J.; Zhang, Y.; Song, H. A Disparate Role of RP11-424C20.2/UHRF1 Axis through Control of Tumor Immune Escape in Liver Hepatocellular Carcinoma and Thymoma. Aging 2019, 11, 6422–6439. [Google Scholar] [CrossRef]
- Ashraf, W.; Ibrahim, A.; Alhosin, M.; Zaayter, L.; Ouararhni, K.; Papin, C.; Ahmad, T.; Hamiche, A.; Mély, Y.; Bronner, C.; et al. The Epigenetic Integrator UHRF1: On the Road to Become a Universal Biomarker for Cancer. Oncotarget 2017, 8, 51946–51962. [Google Scholar] [CrossRef]
- American Association of Neurological Surgeons (AANS); American Society of Neuroradiology (ASNR); Cardiovascular and Interventional Radiology Society of Europe (CIRSE); Canadian Interventional Radiology Association (CIRA); Congress of Neurological Surgeons (CNS); European Society of Minimally Invasive Neurological Therapy (ESMINT); European Society of Neuroradiology (ESNR); European Stroke Organization (ESO); Society for Cardiovascular Angiography and Interventions (SCAI); Society of Interventional Radiology (SIR); et al. Multisociety Consensus Quality Improvement Revised Consensus Statement for Endovascular Therapy of Acute Ischemic Stroke. Int. J. Stroke 2018, 13, 612–632. [Google Scholar] [CrossRef]
- Giaccone, G.; Rajan, A.; Berman, A.; Kelly, R.J.; Szabo, E.; Lopez-Chavez, A.; Trepel, J.; Lee, M.-J.; Cao, L.; Espinoza-Delgado, I.; et al. Phase II Study of Belinostat in Patients with Recurrent or Refractory Advanced Thymic Epithelial Tumors. J. Clin. Oncol. 2011, 29, 2052–2059. [Google Scholar] [CrossRef]
- Thomas, A.; Rajan, A.; Szabo, E.; Tomita, Y.; Carter, C.A.; Scepura, B.; Lopez-Chavez, A.; Lee, M.-J.; Redon, C.E.; Frosch, A.; et al. A Phase I/II Trial of Belinostat in Combination with Cisplatin, Doxorubicin, and Cyclophosphamide in Thymic Epithelial Tumors: A Clinical and Translational Study. Clin. Cancer Res. 2014, 20, 5392–5402. [Google Scholar] [CrossRef] [PubMed]
- Arjonen, A.; Mäkelä, R.; Härmä, V.; Rintanen, N.; Kuopio, T.; Kononen, J.; Rantala, J.K. Image-Based Ex Vivo Drug Screen to Assess Targeted Therapies in Recurrent Thymoma. Lung Cancer 2020, 145, 27–32. [Google Scholar] [CrossRef] [PubMed]
- Bohnenberger, H.; Dinter, H.; König, A.; Ströbel, P. Neuroendocrine Tumors of the Thymus and Mediastinum. J. Thorac. Dis. 2017, 9, S1448–S1457. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.-Y.; Rumilla, K.M.; Jin, L.; Nakamura, N.; Stilling, G.A.; Ruebel, K.H.; Hobday, T.J.; Erlichman, C.; Erickson, L.A.; Lloyd, R.V. Association of DNA Methylation and Epigenetic Inactivation of RASSF1A and Beta-Catenin with Metastasis in Small Bowel Carcinoid Tumors. Endocrine 2006, 30, 299–306. [Google Scholar] [CrossRef] [PubMed]
- Karpathakis, A.; Dibra, H.; Thirlwell, C. Neuroendocrine Tumours: Cracking the Epigenetic Code. Endocr. Relat. Cancer 2013, 20, R65–R82. [Google Scholar] [CrossRef]
- Mapelli, P.; Aboagye, E.O.; Stebbing, J.; Sharma, R. Epigenetic Changes in Gastroenteropancreatic Neuroendocrine Tumours. Oncogene 2015, 34, 4439–4447. [Google Scholar] [CrossRef]
- Tirosh, A.; Killian, J.K.; Petersen, D.; Zhu, Y.J.; Walker, R.L.; Blau, J.E.; Nilubol, N.; Patel, D.; Agarwal, S.K.; Weinstein, L.S.; et al. Distinct DNA Methylation Signatures in Neuroendocrine Tumors Specific for Primary Site and Inherited Predisposition. J. Clin. Endocrinol. Metab. 2020, 105, 3285–3294. [Google Scholar] [CrossRef]
- Wong, J.J.M.; Ginter, P.S.; Tyryshkin, K.; Yang, X.; Nanayakkara, J.; Zhou, Z.; Tuschl, T.; Chen, Y.-T.; Renwick, N. Classifying Lung Neuroendocrine Neoplasms through MicroRNA Sequence Data Mining. Cancers 2020, 12, 2653. [Google Scholar] [CrossRef]
- Colao, A.; de Nigris, F.; Modica, R.; Napoli, C. Clinical Epigenetics of Neuroendocrine Tumors: The Road Ahead. Front. Endocrinol. 2020, 11, 604341. [Google Scholar] [CrossRef]
- Korotaeva, A.; Mansorunov, D.; Apanovich, N.; Kuzevanova, A.; Karpukhin, A. MiRNA Expression in Neuroendocrine Neoplasms of Frequent Localizations. Noncoding RNA 2021, 7, 38. [Google Scholar] [CrossRef]
- Soto, J.A.; Rodríguez-Antolín, C.; Vera, O.; Pernía, O.; Esteban-Rodríguez, I.; Dolores Diestro, M.; Benitez, J.; Sánchez-Cabo, F.; Alvarez, R.; De Castro, J.; et al. Transcriptional Epigenetic Regulation of Fkbp1/Pax9 Genes Is Associated with Impaired Sensitivity to Platinum Treatment in Ovarian Cancer. Clin. Epigenetics 2021, 13, 167. [Google Scholar] [CrossRef] [PubMed]
- Mohanad, M.; Yousef, H.F.; Bahnassy, A.A. Epigenetic Inactivation of DNA Repair Genes as Promising Prognostic and Predictive Biomarkers in Urothelial Bladder Carcinoma Patients. Mol. Genet. Genom. 2022, 297, 1671–1687. [Google Scholar] [CrossRef] [PubMed]
- Saleh, R.; Toor, S.M.; Sasidharan Nair, V.; Elkord, E. Role of Epigenetic Modifications in Inhibitory Immune Checkpoints in Cancer Development and Progression. Front. Immunol. 2020, 11, 1469. [Google Scholar] [CrossRef] [PubMed]
| Investigated Genes | Cohort | Tissues | Methods | Findings | Ref. |
|---|---|---|---|---|---|
| Cyclin-dependent kinase inhibitor 2A (CDKN2A) | 36 TMs 3 TCs | Tumor tissue samples | MSP | Hypermethylation of CDKN2 was observed in 4 TMs and 1 TCs. | [94] |
| Hyperpigmentation, progressive, 1 (HPP1), secreted protein acidic and cysteine rich (SPARC), Reprimo (RPRM), retinoic acid receptor (RAR6), ras association domain family member 1 (RASSF1A), cadherin 13 (CDH13), pl6 (CDKN2A), secretoglobin family 3A member 1 (SCGB3A1), cadherin 1 (CDH1), gremlin 1 (GRM1), RAD | 6 TMs 5TCs | Tumor and adjacent NT samples | MSP | The overall methylation pattern was higher in TC. | [95] |
| F-box and WD repeat domain containing 7 (FBXW7) | 13 TMs | Tumor tissue samples | Bisulphite sequencing | FBXW7 b-form promoter was hypermethylated in types B1 TMs or higher histotypes. | [96] |
| Death-associated protein kinase 1 (DAPK), p16 (CDKN2A), O-6-methylguanine-DNA methyltransferase (MGMT), hyperpigmentation, progressive, 1 (HPP1) | 26 TMs 6 TCs | Tumor tissue samples | MSP | The investigated genes were more methylated in TCs than in TMs (60% vs. 20%). MGMT methylation was detected in 23% of TMs and in 83% of TCs. | [97] |
| Global methylation (5 mC content) 9 candidate TSGs | 65 TETs | 40 PEFF samples and 25 fresh frozen tissues | ELISA (for global methylation) Nested MSP (for gene-specific methylation) | Global DNA methylation levels were reduced, and DNA methyltransferase expression was increased in advanced-stage TETs. E-cadherin (E-cad), retinoic acid receptor beta (RARβ), human mutL homolog 1 (hMLH1), RASSF1A, adenomatous polyposis coli promoter 1A (APC1A), and fragile histidine triad diadenosine triphosphatase (FHIT) genes were hypermethylated in B2/B3/C TMs relative to A/AB/B1 TMs. | [98] |
| MGMT | 66 TETs | Tumor tissue samples | MSP | MGMT methylation was more frequent in TCs than in TMs, also correlated with loss of protein expression. | [99] |
| Methylenetetrahydrofolate reductase (MTHFR), DNA methyltransferases (DNMT1, DNMT3A, DNMT3B) | 69 TAMGs | Tumor tissue and 44 paired NT samples. Blood samples | MS-HRM | MTHFR and DNMT3A promoters were more methylated in tumor tissue with respect to blood. MTHFR promoter was more methylated in tumor tissue respect to healthy adjacent thymic epithelial cells. DNMT1 and DNMT3B promoters were hypomethylated in all tissues. | [100] |
| RASSF1A | 8 TMs 6 TCs 3 NETs | Tumor tissue and adjacent NT samples | Bisulphite pyrosequencing | RASSF1A was hypermethylated in NETs, but not in TCS or TMs. | [101] |
| GWAS | 124 TETs | TCGA dataset | Illumina Infinium HumanMethylation450K | 5155 CpG sites hypomethylated and 6967 CpG sites hypermethylated in TMs and TCs. High methylation in kinase suppressor of Ras 1 (KSR1), E74-like ETS transcription factor 3 (ELF3), interleukin 1 receptor antagonist (IL1RN) correlates with a good prognosis; low methylation in recombination activating 1 (RAG1) correlates with longer OS. | [102] |
| GWAS | 6 TETs | Tumor tissue and adjacent NT samples | Infinium MethylationEPIC BeadChip microarray (850 K) | 119 hypermethylated and 18,999 hypomethylated DMCs in tumor vs. normal tissue. 7 hypermethylated and 29 hypomethylated genes. Fasciculation and elongation protein zeta 2 (FEZ2), protein tyrosine phosphatase receptor type E (PTPRE), ATPase sarcoplasmic/endoplasmic reticulum Ca2+ transporting 2 (ATP2A2), Cbl proto-oncogene B (CBlB), chromosome 5 open reading frame 46 (C5orf45), carboxypeptidase E (CPE), follistatin-like 1 (FSTl1), zinc finger protein 396 (ZNF396), fraser extracellular matrix complex subunit 1 (FRAS1), neuron navigator 2 (NAV2) and lebercilin LCA5 (lCA5) may be potential biomarkers for the diagnosis of type A and type B thymomas. | [103] |
| MLH1, MGMT, CDKN2A, RASSF1A | 69 TAMGs | Tumor tissue and 44 paired NT samples. Blood samples | MS-HRM | MLH1, MGMT, CDKN2A, and RASSF1A genes promoter methylation were not increased in TAMGs with respect to healthy tissues. | [104] |
| Growth hormone secretagogue receptor (GHSR) | 65 TAMG | Tumor tissue and 43 paired NT samples. Blood samples | MS-HRM | GHSR hypermethylation was observed in 18 TMs compared to the healthy thymic tissues and demethylated in the remaining 47 TMs. | [105] |
| GWAS | 46 TETs | Tumor tissue and 20 paired adjacent NT samples. | Illumina HumanMethylation450 K BeadChip, pyrosequencing | 92 CGI were hypermethylated in the TCs with respect to B3 TMs. 5 CpG sites in the GHSR gene were more methylated in TMs and TCs than in the thymus. G protein subunit gamma 4 (GNG4), spalt-like transcription factor 3 (SALL3), and homeobox D9 (HOXD9) DNA methylation rates were significantly higher in TC than in the thymus. | [106] |
| KIT proto-oncogene ligand (KITLG) | 121 TMs + 37 TMs * | TCGA/GEO datasets | Methylation data from TCGA/GEO datasets | KITLG overexpression is associated with 28 hypermethylated and 163 hypomethylated regions. | [107] |
| MTHFR | 123 TETs | TCGA dataset | Methylation data from TCGA dataset | Negative correlation between the transcriptional and methylation levels of MTHFR. | [108] |
| Glutamate decarboxylase 1 (GAD1) | 73 TMs 17 TCs | 47 tumor tissues and 21 paired adjacent NT samples | Pyrosequencing | GAD1 hypermethylation is higher in TCs than TMs. | [109] |
| GWAS | TCGA cohort | TCGA dataset | Methylation data from TCGA dataset | 943 DMCs between TMs and controls; 14 genes, including lipase E (LIPE) resulted epigenetically regulated. | [110] |
| Mitogen-activated protein kinase 4 (MAPK4) | 124 TETs from TCGA 95 TET * | Tumor tissues | TCGA dataset/pyrosequencing | MAPK4 methylation is a prognostic factor for RFS. | [111] |
| MARVEL domain containing 1 (MARVELD1) | 10,534 patients representing 33 cancer types | TCGA dataset | Methylation data from TCGA dataset | Hypermethylation in the MARVELD1 promoter locus synergistically affected its expression in pan-cancer. | [112] |
| Investigated ncRNA | Cohort | Tissues | Methods | Findings | Ref. |
|---|---|---|---|---|---|
| MiRNome | 51 TMs 8 TCs 7 NT + 36 TMs * 11 TCs | Tumor and NT sample | Data from the TCGA dataset | Hsa-mir-142-5p, hsa-mir-363-3p, and hsa-mir-16-2-3p were dysregulated in TC and linked to antiapoptotic pathways. | [131] |
| MiRNome | 54 TETs, 12 NT | FFPE tissues | Microarray analysis | 87 miRNAs differently expressed between TETs and normal samples. Hsa-mir-7-5p, hsa-mir-489, hsa-mir-1208, hsa-mir-1323, hsa-mir-519e-5p, hsa-mir-516b-5p, hsa-mir-921, hsa-mir-509-5p, hsa-mir-138-2-3p, and hsa-mir-342-5p were dysregulated in TCs vs. B2/B3 TMs. Hsa-mir-650, hsa-mir-181c-5p, hsa-mir-363-3p, hsa-mir-181a-3p were dysregulated in TCs vs. A/AB/B1 TMs. | [132] |
| MiRNome | 5 TETs | Blood samples | RT-qPCR | Hsa-mir-21-5p and 148a-3p were found significantly upregulated in plasma of the TET patients. | [133] |
| hsa-mir-19b | 52 TAMG 12 TMs 11 NT | Tumor tissues and healthy thymus | RT-qPCR | Hsa-mir-19b-5p was increased in TMs from TAMG patients. | [134] |
| C19MC C14MC | 13 TET 3 NT | Tumor tissue and NT sample | RNA-seq | C19MC is a genomic hallmark of type A and AB TMs. | [135] |
| hsa-mir-20b | 30 TAMG | Tumor tissue samples and serum | RT-qPCR | Hsa-mir-20b expression level is downregulated in both TMs and serum from patients with TAMG. | [136] |
| hsa-mir-125a-5p | 13 TAMG | Tumor tissues, adjacent NT and normal thymus samples | Microarray analysis | 137 miRNAs in NT adjacent to the TM from TAMG patients that were significantly dysregulated compared with normal thymus in controls. Hsa-mir-125a-5p expression negatively correlates with foxp3 expression in NT with respect to the thymoma from TAMG. | [137] |
| hsa-mir-145-5p | 14 TMs 5 TCs | FFPE tissues | Microarray analysis | Hsa-mir-145-5p contributes to the tumor phenotype. | [138] |
| C19MC | 37 TMs 35 TCs 6 NT | FFPE tissues | RNA-seq | C14MC and C19MC miRNA clusters are highly expressed in type A thymomas but silenced in TCs. Hsa-mir-21, hsa-mir-9-3 and hsa-mir-375 and hsa-mir-34b, hsa-mir-34c, hsa-mir-130a, and hsa-mir-195 were respectively upregulated and downregulated in TCs. | [139] |
| MiRNome | 123 TETs (9% TCs) | Tumor tissues sample | Data from the TCGA dataset | 9 miRNAs upregulated and 72 miRNAs downregulated in TCs. Let-7a-1, let-7a-2, let-7a-3, hsa-mir-140, hsa-mir-324, hsa-mir-639, and hsa-mir-3613 dysregulation was associated with DFS or OS in TCs. | [140] |
| hsa-mir-27-a | 15 TETs | Tumor and normal tissues sample | RT-qPCR | Hsa-mir-27-a is upregulated in TC tissues. Curcumin treatment decrease hsa-mir-27-a levels in TC1889 cells. | [141] |
| MiRNome | 119 TETs + 99 TETs * | FFPE tissues | RNA seq from TCGA dataset | Hsa-mir-130b-5p, hsa-mir-1307-3p, and hsa-mir-425-5p, were among prognostic factors for RLFS and OS in TCs. | [142] |
| Investigated lncRNA | Cohort | Tissues | Methods | Findings | Ref. |
|---|---|---|---|---|---|
| Whole LncRNA analysis | 114 TMs from TCGA | Tumor tissue samples | RNA-seq | HSD52, LINC0098, ADAMTS9-AS1, and LNC01697 were proposed as a viable prognostic factor for low- and high-risk recurrence outcomes. | [143] |
| Whole LncRNA analysis | 25 TMs 25 NT | Tumor tissue samples | RNA-seq | AFAP1-AS1, LINC00324, ADAMTS9-AS1, VLDR-AS1, LINC00968, and NEAT1 are deregulated in TMs compared to NT. | [144] |
| Whole LncRNA analysis | 5 TMs 3 TAMG | Tumor tissue samples | Microarray analysis | 4360 lncRNAs and 2545 mRNAs were differentially expressed between TAMG and non-TAMG thymomas. | [145] |
| Whole LncRNA analysis | 22 TETs | Tumor tissue samples and NT tissues | RNA-seq | The circulating RNA hsa_circ_0001173, hsa_circ_000729.1, hsa_circ_0003550, and hsa_circ_0001947 are upregulated in TMs and associated with PFS. | [146] |
| LINC00174 | 119 TETs + 6 TETs * 3 NT | Tumor and NT samples | Microarray analysis/ Data from the TCGA dataset | LINC00174 is upregulated in TETs and sponges has-mir-145-5p. | [147] |
| MALAT1 | IU-TAB-1 cell line | Cell models | RT-qPCR | MALAT1 silencing attenuated cell proliferation and apoptosis via the miR-145-5p/HMGA2 pathway in TC cell models. | [148] |
| MALAT1 | TC1889 cell line | Cell models | RT-qPCR | c-MYC protein is enabled by MALAT1, which is delocalized by METTL3, acting as a tumor promoter in TCs. | [149] |
| Whole LncRNA analysis | 29 TAMG 58 TETs | Tumor tissues | TCGA dataset | TAMG showed specific up and downregulation of 56 and 84 lncRNAs, respectively. The lncRNA AP000787.1, AC004943.1, WT1-AS, FOXG1-AS1 are shown to be regulated by methylation of their promoter region. | [150] |
| RNA expression analysis | 27 TAMG 67 TMs | Tumor tissues | TCGA dataset | The LINC00425/hsa-mir-204/CHST4 axis is potentially involved in the progression of TAMG. | [151] |
| LOXL1-AS1 | 42 TMs 28 TCs | Tumor samples and Thy0517/Ty-82 cell models | RT-qPCR/Data from the TCGA dataset | LOXL1-AS1 sponges hsa-mir--525-5p-HSPA9 target. Downregulation of hsa-mir-525-5p and high levels of LOXL1-AS1 and HSPA9 were associated with poor prognosis in TCs. | [152] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nicolì, V.; Coppedè, F. Epigenetics of Thymic Epithelial Tumors. Cancers 2023, 15, 360. https://doi.org/10.3390/cancers15020360
Nicolì V, Coppedè F. Epigenetics of Thymic Epithelial Tumors. Cancers. 2023; 15(2):360. https://doi.org/10.3390/cancers15020360
Chicago/Turabian StyleNicolì, Vanessa, and Fabio Coppedè. 2023. "Epigenetics of Thymic Epithelial Tumors" Cancers 15, no. 2: 360. https://doi.org/10.3390/cancers15020360
APA StyleNicolì, V., & Coppedè, F. (2023). Epigenetics of Thymic Epithelial Tumors. Cancers, 15(2), 360. https://doi.org/10.3390/cancers15020360






