Netrin Family Genes as Prognostic Markers and Therapeutic Targets for Clear Cell Renal Cell Carcinoma: Netrin-4 Acts through the Wnt/β-Catenin Signaling Pathway
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Data Collection
2.2. mRNA Expression Analysis
2.3. Diagnostic and Prognostic Value Analysis
2.4. Tissue Specimens and Tumor Cells
2.5. Western Blotting
2.6. Cell Transfection
2.7. CCK-8 Proliferation Assay
2.8. Colony Growth Assay
2.9. Transwell Experiment
2.10. The Scratch Wound Assay
2.11. Immunohistochemistry
2.12. Immunofluorescence
2.13. Flow Cytometric Analysis
2.14. In Vivo Experiments
2.15. Statistical Analysis
3. Results
3.1. Expression of the Netrin Family in Normal Human Tissues
3.2. Expression of the Netrin Family in ccRCC
3.3. Correlation of Netrin Family Members with Survival Prognosis in Patients with ccRCC
3.4. Independent Diagnostic Value of Netrin Family Members in the Survival of Patients with ccRCC
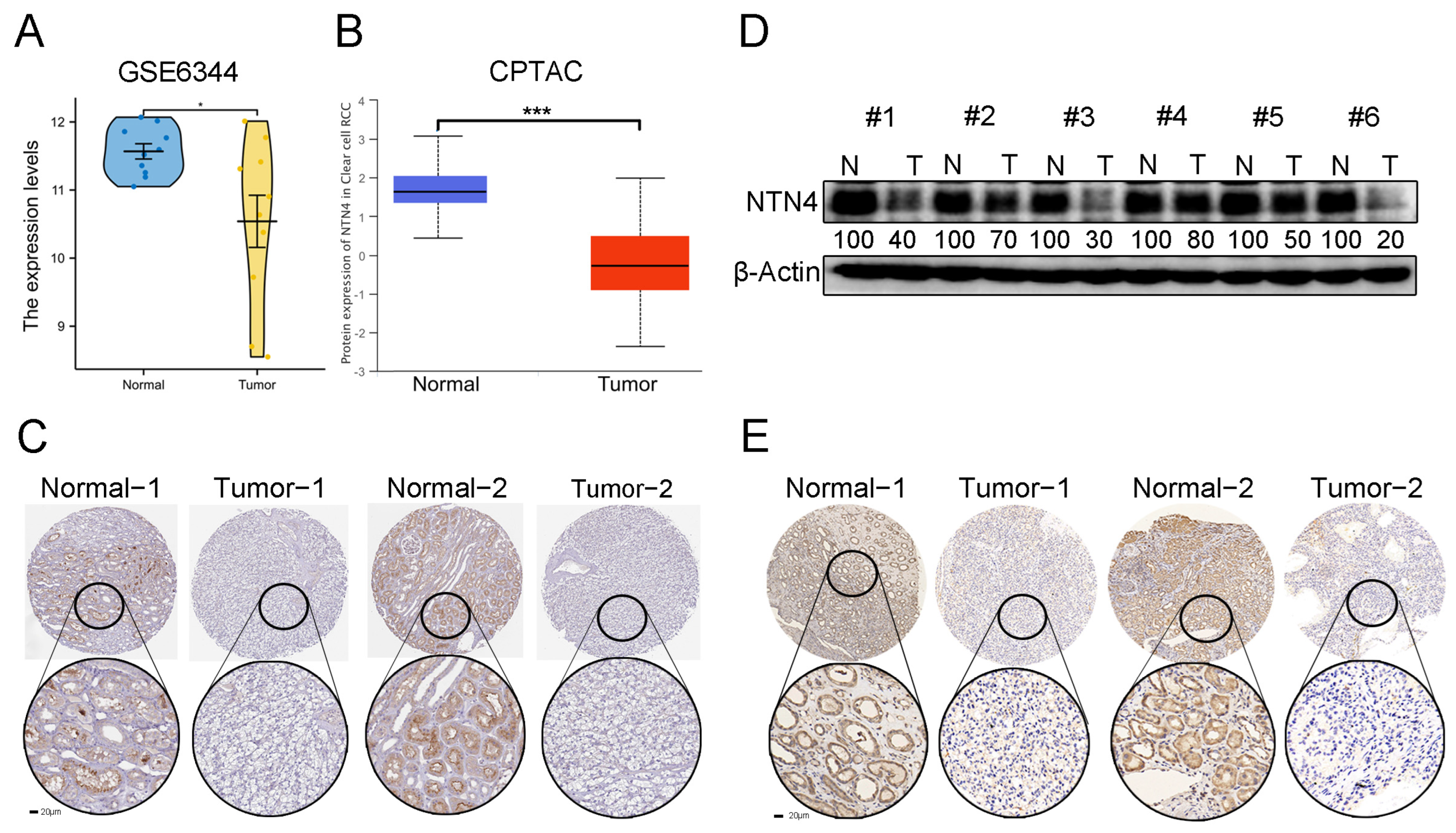
3.5. Expression of Netrins Family Members in Patients with ccRCC
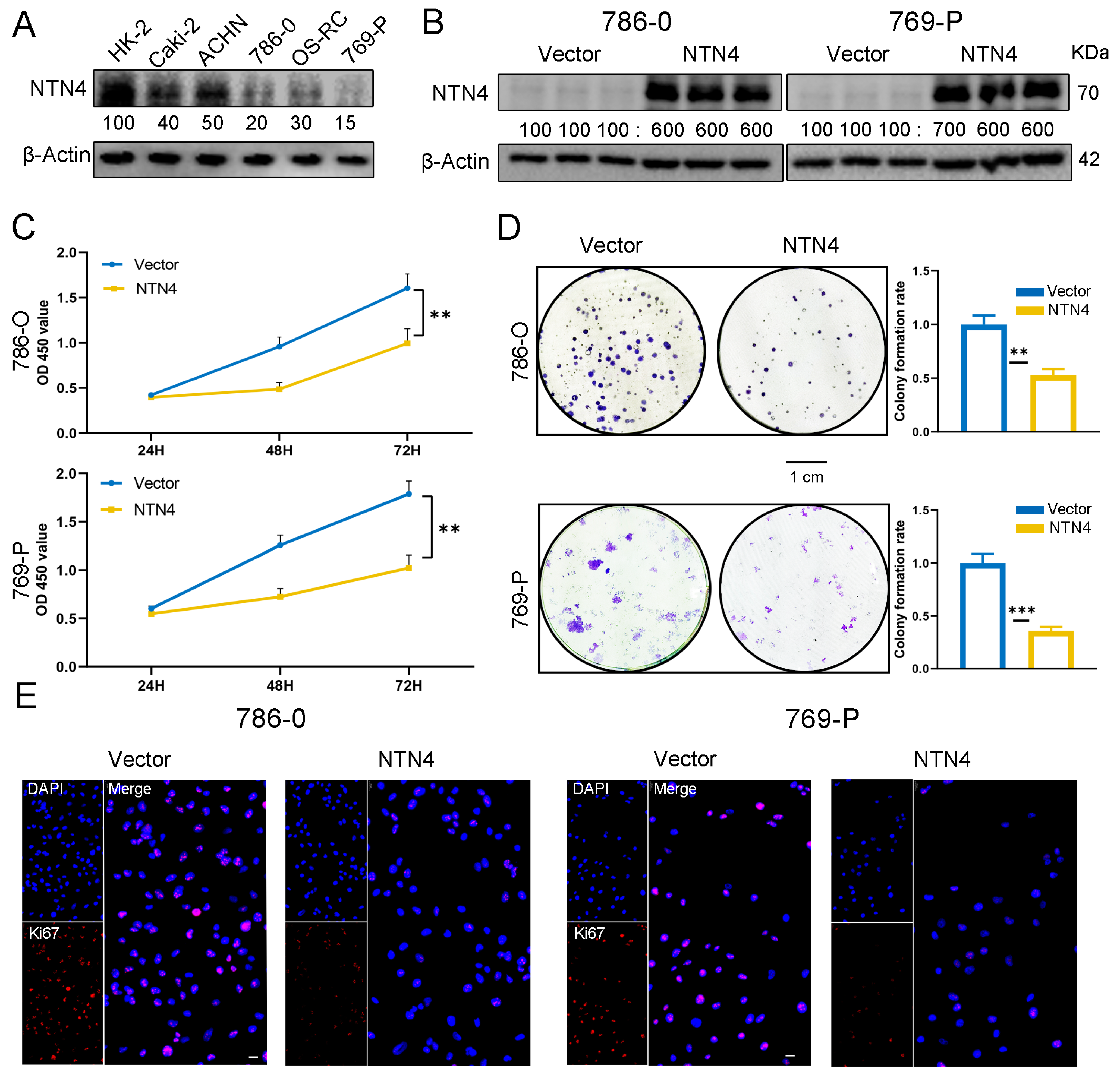
3.6. Overexpression of NTN4 Inhibits the Growth of ccRCC Cells In Vitro
3.7. We Evaluated the Effect of NTN4 Overexpression on ccRCC Cell Migration In Vitro
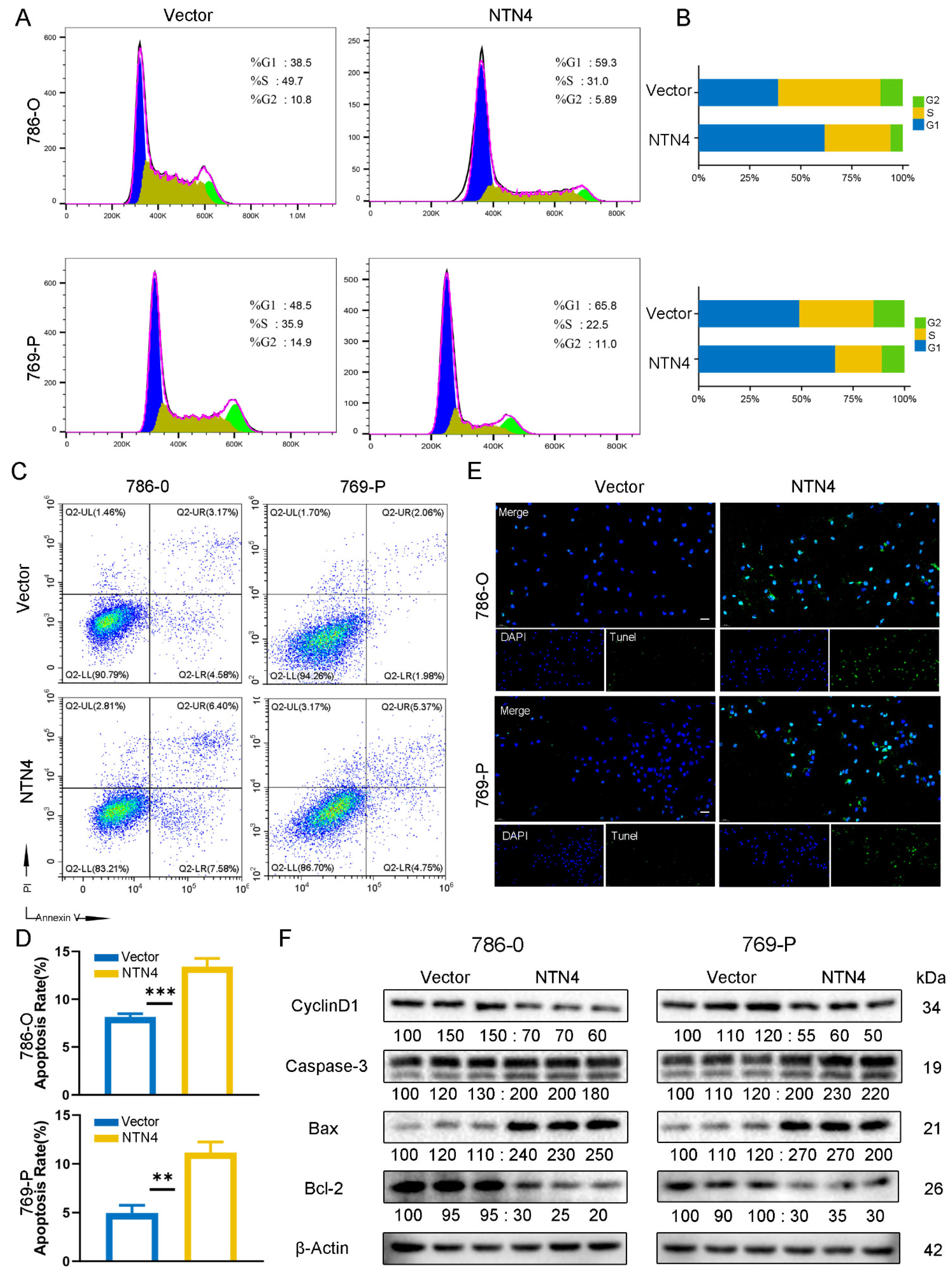
3.8. Regulation of the Cell Cycle and Apoptosis by NTN4
3.9. NTN4-Mediated Regulation of the WNT/β-Catenin Signaling Pathway in ccRCC
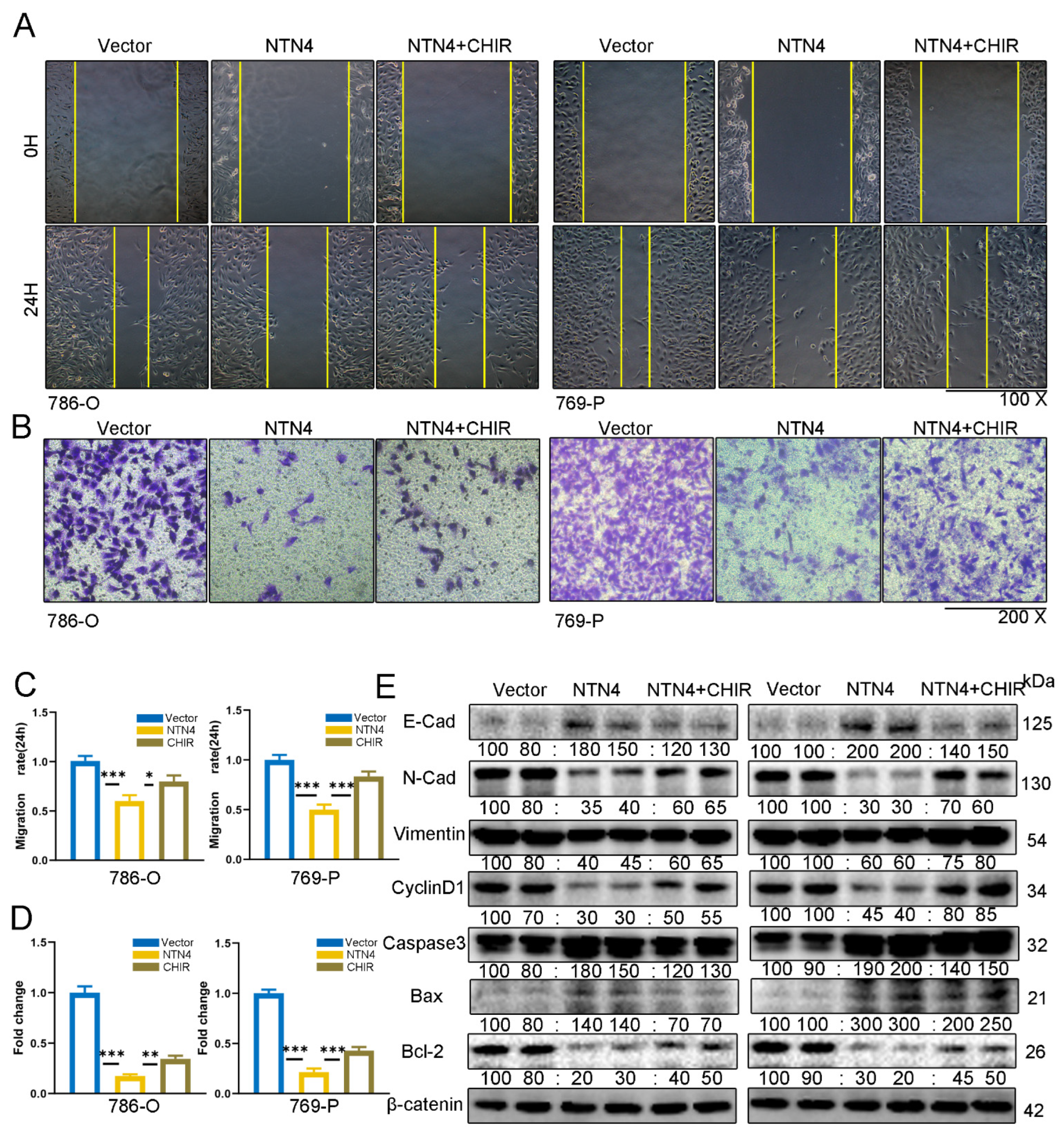
3.10. Enhancement of β-Catenin Signaling Can Counteract the Impact of NTN4
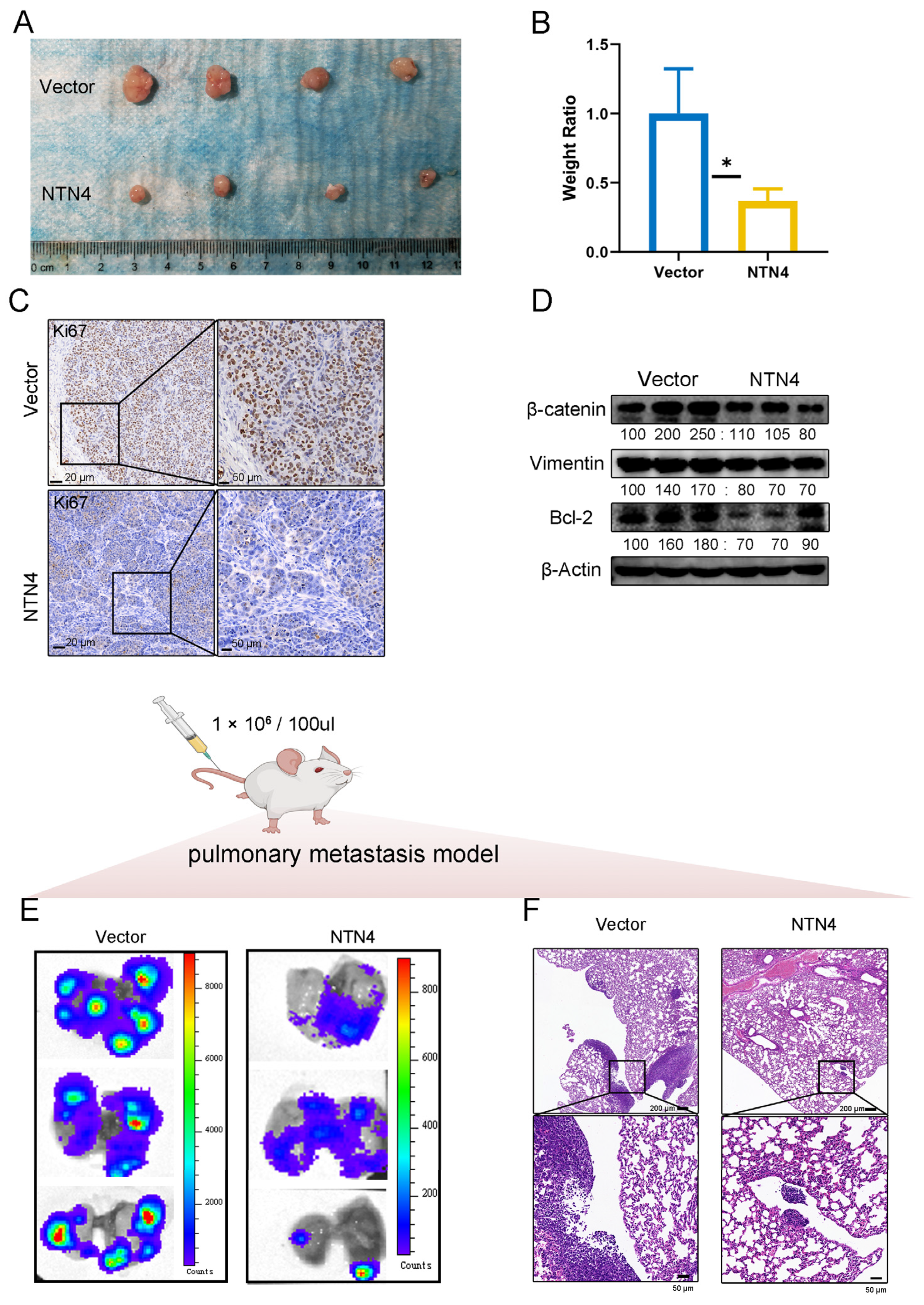
3.11. Subcutaneous Proliferation Model and Lung Metastasis Model in Nude Mice
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ljungberg, B.; Albiges, L.; Abu-Ghanem, Y.; Bedke, J.; Capitanio, U.; Dabestani, S.; Fernandez-Pello, S.; Giles, R.H.; Hofmann, F.; Hora, M.; et al. European Association of Urology Guidelines on Renal Cell Carcinoma: The 2022 Update. Eur. Urol. 2022, 82, 399–410. [Google Scholar] [CrossRef] [PubMed]
- Benson, A.; Venook, A.P.; Al-Hawary, M.M.; Arain, M.A.; Chen, Y.J.; Ciombor, K.K.; Cohen, S.; Cooper, H.S.; Deming, D.; Farkas, L.; et al. Colon Cancer, Version 2.2021. J. Natl. Compr. Cancer Netw. 2021, 19, 329–359. [Google Scholar] [CrossRef]
- Porta, C.; Cosmai, L.; Leibovich, B.C.; Powles, T.; Gallieni, M.; Bex, A. The adjuvant treatment of kidney cancer: A multidisciplinary outlook. Nat. Rev. Nephrol. 2019, 15, 423–433. [Google Scholar] [CrossRef] [PubMed]
- Serafini, T.; Kennedy, T.E.; Galko, M.J.; Mirzayan, C.; Jessell, T.M.; Tessierlavigne, M. The Netrins Define a Family of Axon Outgrowth-Promoting Proteins Homologous to C. elegans UNC-6. Cell 1994, 78, 409–424. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Copeland, N.G.; Gilbert, D.J.; Jenkins, N.A.; Tessier-Lavigne, M. Netrin-3, a mouse homolog of human NTN2L, is highly expressed in sensory ganglia and shows differential binding to netrin receptors. J. Neurosci. 1999, 19, 4938–4947. [Google Scholar] [CrossRef]
- Nakashiba, T.; Ikeda, T.; Nishimura, S.; Tashiro, K.; Honjo, T.; Culotti, J.G.; Itohara, S. Netrin-G1: A novel glycosyl phosphatidylinositol-linked mammalian netrin that is functionally divergent from classical netrins. J. Neurosci. 2000, 20, 6540–6550. [Google Scholar] [CrossRef]
- Nakashiba, T.; Nishimura, S.; Ikeda, T.; Itohara, S. Complementary expression and neurite outgrowth activity of netrin-G subfamily members. Mech. Dev. 2002, 111, 47–60. [Google Scholar] [CrossRef]
- Yin, Y.; Sanes, J.R.; Miner, J.H. Identification and expression of mouse netrin-4. Mech. Dev. 2000, 96, 115–119. [Google Scholar] [CrossRef]
- Jasmin, M.; Ahn, E.H.; Voutilainen, M.H.; Fombonne, J.; Guix, C.; Viljakainen, T.; Kang, S.S.; Yu, L.Y.; Saarma, M.; Mehlen, P.; et al. Netrin-1 and its receptor DCC modulate survival and death of dopamine neurons and Parkinson’s disease features. Embo J. 2021, 40, e105537. [Google Scholar] [CrossRef]
- Castets, M.; Broutier, L.; Molin, Y.; Brevet, M.; Chazot, G.; Gadot, N.; Paquet, A.; Mazelin, L.; Jarrosson-Wuilleme, L.; Scoazec, J.Y.; et al. DCC constrains tumour progression via its dependence receptor activity. Nature 2012, 482, 534–537. [Google Scholar] [CrossRef]
- Untiveros, G.; Raskind, A.; Linares, L.; Dotti, A.; Strizzi, L. Netrin-1 Stimulates Migration of Neogenin Expressing Aggressive Melanoma Cells. Int. J. Mol. Sci. 2022, 23, 12751. [Google Scholar] [CrossRef]
- Wischhusen, J.; Wilson, K.E.; Delcros, J.G.; Pena, R.M.; Gibert, B.; Jiang, S.; Ngo, J.; Goldschneider, D.; Mehlen, P.; Willmann, J.K.; et al. Ultrasound molecular imaging as a non-invasive companion diagnostic for netrin-1 interference therapy in breast cancer. Theranostics 2018, 8, 5126–5142. [Google Scholar] [CrossRef]
- Han, P.; Liu, J.M.; Lei, Y.; Lin, Z.Y.; Tian, D.; Yan, W. Netrin-1 promotes the collective cell migration of liver cancer cells in a 3D cell culture model. J. Physiol. Biochem. 2019, 75, 489–498. [Google Scholar] [CrossRef]
- Fang, S.Y.; Luo, Y.Y.; Zhang, Y.; Wang, H.M.; Liu, Q.F.; Li, X.Y.; Yu, T.H. NTNG1 Modulates Cisplatin Resistance in Epithelial Ovarian Cancer Cells via the GAS6/AXL/Akt Pathway. Front. Cell Dev. Biol. 2021, 9, 652325. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.Z.; Ylivinkka, I.; Chen, P.; Li, L.; Hautaniemi, S.; Nyman, T.A.; Keski-Oja, J.; Hyytiainen, M. Netrin-4 Promotes Glioblastoma Cell Proliferation through Integrin beta(4) Signaling. Neoplasia 2012, 14, 219–227. [Google Scholar] [CrossRef] [PubMed]
- Eveno, C.; Broqueres-You, D.; Feron, J.G.; Rampanou, A.; Tijeras-Raballand, A.; Ropert, S.; Leconte, L.; Levy, B.I.; Pocard, M. Netrin-4 Delays Colorectal Cancer Carcinomatosis by Inhibiting Tumor Angiogenesis. Am. J. Pathol. 2011, 178, 1861–1869. [Google Scholar] [CrossRef] [PubMed]
- Reuten, R.; Zendehroud, S.; Nicolau, M.; Fleischhauer, L.; Laitala, A.; Kiderlen, S.; Nikodemus, D.; Wullkopf, L.; Nielsen, S.R.; McNeilly, S.; et al. Basement membrane stiffness determines metastases formation. Nat. Mater. 2021, 20, 892–903. [Google Scholar] [CrossRef]
- Gumz, M.L.; Zou, H.; Kreinest, P.A.; Childs, A.C.; Belmonte, L.S.; LeGrand, S.N.; Wu, K.J.; Luxon, B.A.; Sinha, M.; Parker, A.S.; et al. Secreted frizzled-related protein 1 loss contributes to tumor phenotype of clear cell renal cell carcinoma. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2007, 13, 4740–4749. [Google Scholar] [CrossRef]
- Ritchie, M.E.; Phipson, B.; Wu, D.; Hu, Y.; Law, C.W.; Shi, W.; Smyth, G.K. Limma powers differential expression analyses for RNA-sequencing and microarray studies. Nucleic Acids Res. 2015, 43, e47. [Google Scholar] [CrossRef]
- Robin, X.; Turck, N.; Hainard, A.; Tiberti, N.; Lisacek, F.; Sanchez, J.C.; Müller, M. pROC: An open-source package for R and S+ to analyze and compare ROC curves. BMC Bioinform. 2011, 12, 77. [Google Scholar] [CrossRef]
- Rajasekharan, S.; Kennedy, T.E. The netrin protein family. Genome Biol. 2009, 10, 239. [Google Scholar] [CrossRef] [PubMed]
- Sung, P.J.; Rama, N.; Imbach, J.; Fiore, S.; Ducarouge, B.; Neves, D.; Chen, H.W.; Bernard, D.; Yang, P.C.; Bernet, A.; et al. Cancer-Associated Fibroblasts Produce Netrin-1 to Control Cancer Cell Plasticity. Cancer Res. 2019, 79, 3651–3661. [Google Scholar] [CrossRef] [PubMed]
- Ko, S.Y.; Blatch, G.L.; Dass, C.R. Netrin-1 as a potential target for metastatic cancer: Focus on colorectal cancer. Cancer Metastasis Rev. 2014, 33, 101–113. [Google Scholar] [CrossRef] [PubMed]
- Paradisi, A.; Maisse, C.; Coissieux, M.M.; Gadot, N.; Lepinasse, F.; Delloye-Bourgeois, C.; Delcros, J.G.; Svrcek, M.; Neufert, C.; Flejou, J.F.; et al. Netrin-1 up-regulation in inflammatory bowel diseases is required for colorectal cancer progression. Proc. Natl. Acad. Sci. USA 2009, 106, 17146–17151. [Google Scholar] [CrossRef]
- Eveno, C.; Contreres, J.O.; Hainaud, P.; Nemeth, J.; Dupuy, E.; Pocard, M. Netrin-4 overexpression suppresses primary and metastatic colorectal tumor progression. Oncol. Rep. 2013, 29, 73–78. [Google Scholar] [CrossRef]
- Beesley, J.; Sivakumaran, H.; Marjaneh, M.M.; Shi, W.; Hillman, K.M.; Kaufmann, S.; Hussein, N.; Kar, S.; Lima, L.G.; Ham, S.; et al. eQTL Colocalization Analyses Identify NTN4 as a Candidate Breast Cancer Risk Gene. Am. J. Hum. Genet. 2020, 107, 778–787. [Google Scholar] [CrossRef]
- Xu, X.P.; Yan, Q.Y.; Wang, Y.A.; Dong, X.J. NTN4 is associated with breast cancer metastasis via regulation of EMT-related biomarkers. Oncol. Rep. 2017, 37, 449–457. [Google Scholar] [CrossRef]
- Fagerberg, L.; Hallstrom, B.M.; Oksvold, P.; Kampf, C.; Djureinovic, D.; Odeberg, J.; Habuka, M.; Tahmasebpoor, S.; Danielsson, A.; Edlund, K.; et al. Analysis of the Human Tissue-specific Expression by Genome-wide Integration of Transcriptomics and Antibody-based Proteomics. Mol. Cell. Proteom. 2014, 13, 397–406. [Google Scholar] [CrossRef] [PubMed]
- Lonsdale, J.; Thomas, J.; Salvatore, M.; Phillips, R.; Lo, E.; Shad, S.; Hasz, R.; Walters, G.; Garcia, F.; Young, N.; et al. The Genotype-Tissue Expression (GTEx) project. Nat. Genet. 2013, 45, 580–585. [Google Scholar] [CrossRef]
- Uhlen, M.; Fagerberg, L.; Hallstrom, B.M.; Lindskog, C.; Oksvold, P.; Mardinoglu, A.; Sivertsson, A.; Kampf, C.; Sjostedt, E.; Asplund, A.; et al. Tissue-based map of the human proteome. Science 2015, 347, 1260419. [Google Scholar] [CrossRef]
- Broekman, M.L.; Maas, S.L.N.; Abels, E.R.; Mempel, T.R.; Krichevsky, A.M.; Breakefield, X.O. Multidimensional communication in the microenvirons of glioblastoma. Nat. Rev. Neurol. 2018, 14, 482–495. [Google Scholar] [CrossRef] [PubMed]
- Xu, M.S.; Zhang, T.; Xia, R.L.; Wei, Y.Q.; Wei, X.W. Targeting the tumor stroma for cancer therapy. Mol. Cancer 2022, 21, 208. [Google Scholar] [CrossRef] [PubMed]
- Tian, Q.; Stepaniants, S.B.; Mao, M.; Weng, L.; Feetham, M.C.; Doyle, M.J.; Yi, E.C.; Dai, H.Y.; Thorsson, V.; Eng, J.; et al. Integrated genomic and proteomic analyses of gene expression in mammalian cells. Mol. Cell. Proteom. 2004, 3, 960–969. [Google Scholar] [CrossRef] [PubMed]
- Vogel, C.; Abreu, R.D.; Ko, D.J.; Le, S.Y.; Shapiro, B.A.; Burns, S.C.; Sandhu, D.; Boutz, D.R.; Marcotte, E.M.; Penalva, L.O. Sequence signatures and mRNA concentration can explain two-thirds of protein abundance variation in a human cell line. Mol. Syst. Biol. 2010, 6, 400. [Google Scholar] [CrossRef] [PubMed]
- Uhlen, M.; Zhang, C.; Lee, S.; Sjostedt, E.; Fagerberg, L.; Bidkhori, G.; Benfeitas, R.; Arif, M.; Liu, Z.T.; Edfors, F.; et al. A pathology atlas of the human cancer transcriptome. Science 2017, 357, 2507. [Google Scholar] [CrossRef]
- Yi, L.L.; Lei, Y.Q.; Yuan, F.J.; Tian, C.H.; Chai, J.; Gu, M.L. NTN4 as a prognostic marker and a hallmark for immune infiltration in breast cancer. Sci. Rep. 2022, 12, 10567. [Google Scholar] [CrossRef]
- Ma, X.G.; Xiang, F.; Pei, Z.; Miao, J.F.; Wu, P.; Song, X.F.; Li, Y.H.; Zhang, Y.M. Circ-Smad5 retards the G1/S transition of cell cycle via inhibiting the activity of wnt/lef/cyclind1 signaling in JB6 cells. Genes Dis. 2021, 8, 364–372. [Google Scholar] [CrossRef]
- Koohpeyma, H.; Goudarzi, I.; Salmani, M.E.; Lashkarbolouki, T.; Shabani, M. Folic Acid Protects Rat Cerebellum Against Oxidative Damage Caused by Homocysteine: The Expression of Bcl-2, Bax, and Caspase-3 Apoptotic Genes. Neurotox. Res. 2020, 37, 564–577. [Google Scholar] [CrossRef]
- Pastushenko, I.; Blanpain, C. EMT Transition States during Tumor Progression and Metastasis. Trends Cell Biol. 2019, 29, 212–226. [Google Scholar] [CrossRef]
- Gong, G.; Ganesan, K.; Xiong, Q.; Zheng, Y. Anti-Invasive and Anti-Migratory Effects of Ononin on Human Osteosarcoma Cells by Limiting the MMP2/9 and EGFR-Erk1/2 Pathway. Cancers 2023, 15, 758. [Google Scholar] [CrossRef]
- Davis, F.M.; Stewart, T.A.; Thompson, E.W.; Monteith, G.R. Targeting EMT in cancer: Opportunities for pharmacological intervention. Trends Pharmacol. Sci. 2014, 35, 479–488. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Zhang, S.J.; Li, X.Y.; Zhang, F.; Zhao, L. HOXB5 promotes the progression of breast cancer through wnt/ beta-catenin pathway. Pathol. Res. Pract. 2021, 224, 153117. [Google Scholar] [CrossRef] [PubMed]
- Song, L.L.; Li, Y.M.; He, B.M.; Gong, Y. Development of Small Molecules Targeting the Wnt Signaling Pathway in Cancer Stem Cells for the Treatment of Colorectal Cancer. Clin. Color. Cancer 2015, 14, 133–145. [Google Scholar] [CrossRef] [PubMed]
- Cai, J.S.; Huang, Z.Y.; Zhou, J.; Wu, W.F.; Ye, Y.L. TLN2 functions as a tumor suppressor in clear cell renal cell carcinoma via inactivation of the Wnt/beta-catenin signaling pathway. Transl. Androl. Urol. 2022, 11, 39–52. [Google Scholar] [CrossRef]
- Zhu, L.Z.; Tian, Q.; Gao, H.; Wu, K.J.; Wang, B.; Ge, G.Q.; Jiang, S.Y.; Wang, K.; Zhou, C.; He, J.J.; et al. PROX1 promotes breast cancer invasion and metastasis through WNT/beta-catenin pathway via interacting with hnRNPK. Int. J. Biol. Sci. 2022, 18, 2032–2046. [Google Scholar] [CrossRef]
- Gao, S.L.; Gao, L.; Wang, S.M.; Shi, X.K.; Yue, C.; Wei, S.Z.; Zuo, L.; Zhang, L.F.; Qin, X.H. ATF3 Suppresses Growth and Metastasis of Clear Cell Renal Cell Carcinoma by Deactivating EGFR/AKT/GSK3 beta/beta-Catenin Signaling Pathway. Front. Cell Dev. Biol. 2021, 9, 618987. [Google Scholar] [CrossRef]
- Fendler, A.; Bauer, D.; Busch, J.; Jung, K.; Wulf-Goldenberg, A.; Kunz, S.; Song, K.; Myszczyszyn, A.; Elezkurtaj, S.; Erguen, B.; et al. Inhibiting WNT and NOTCH in renal cancer stem cells and the implications for human patients. Nat. Commun. 2020, 11, 929. [Google Scholar] [CrossRef]





| Antibody | Specificity | WB | IF | IHC | Company |
|---|---|---|---|---|---|
| Bax | Rabbit | 1:800 | − | − | Abcam |
| Bcl-2 | Rabbit | 1:800 | − | − | Abcam |
| Cleaved caspase-3 | Rabbit | 1:800 | − | − | Abcam |
| Cyclin D1 | Rabbit | 1:800 | − | − | Proteintech |
| E-cadherin | Rabbit | 1:800 | 1:100 | − | Abcam |
| GSK3β | Rabbit | 1:800 | − | − | Proteintech |
| Ki67 | Rabbit | 1:800 | 1:100 | 1:200 | Proteintech |
| Lamin B | Rabbit | 1:800 | − | − | Proteintech |
| MMP-9 | Rabbit | 1:800 | − | − | Proteintech |
| N-cadherin | Rabbit | 1:800 | 1:100 | − | Abcam |
| NTN4 | Rabbit | 1:500 | − | 1:200 | ABclonal |
| p-GSK3β | Rabbit | 1:800 | − | − | Proteintech |
| Vimentin | Rabbit | 1:800 | 1:100 | − | Abcam |
| β-actin | Rabbit | − | − | − | Proteintech |
| β-catenin | Rabbit | 1:800 | 1:100 | − | Proteintech |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ke, S.; Guo, J.; Wang, Q.; Shao, H.; He, M.; Li, T.; Qiu, T.; Guo, J. Netrin Family Genes as Prognostic Markers and Therapeutic Targets for Clear Cell Renal Cell Carcinoma: Netrin-4 Acts through the Wnt/β-Catenin Signaling Pathway. Cancers 2023, 15, 2816. https://doi.org/10.3390/cancers15102816
Ke S, Guo J, Wang Q, Shao H, He M, Li T, Qiu T, Guo J. Netrin Family Genes as Prognostic Markers and Therapeutic Targets for Clear Cell Renal Cell Carcinoma: Netrin-4 Acts through the Wnt/β-Catenin Signaling Pathway. Cancers. 2023; 15(10):2816. https://doi.org/10.3390/cancers15102816
Chicago/Turabian StyleKe, Shuai, Jiayu Guo, Qinghua Wang, Haoren Shao, Mu He, Tao Li, Tao Qiu, and Jia Guo. 2023. "Netrin Family Genes as Prognostic Markers and Therapeutic Targets for Clear Cell Renal Cell Carcinoma: Netrin-4 Acts through the Wnt/β-Catenin Signaling Pathway" Cancers 15, no. 10: 2816. https://doi.org/10.3390/cancers15102816
APA StyleKe, S., Guo, J., Wang, Q., Shao, H., He, M., Li, T., Qiu, T., & Guo, J. (2023). Netrin Family Genes as Prognostic Markers and Therapeutic Targets for Clear Cell Renal Cell Carcinoma: Netrin-4 Acts through the Wnt/β-Catenin Signaling Pathway. Cancers, 15(10), 2816. https://doi.org/10.3390/cancers15102816








