Plasma Micronutrient Profile of Prostate Cancer Cases Is Altered Relative to Healthy Controls—Results of a Pilot Study in South Australia
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Population
2.2. Laboratory Assays
2.3. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Chu, K.C.; Tarone, R.E.; Freeman, H.P. Trends in prostate cancer mortality among black men and white men in the United States. Cancer 2003, 97, 1507–1516. [Google Scholar] [CrossRef]
- Chen, C.B.; Eurich, D.T.; Majumdar, S.R.; Johnson, J.A. Risk of prostate cancer across different racial/ethnic groups in men with diabetes: A retrospective cohort study. Diabet. Med. 2018, 35, 107–111. [Google Scholar] [CrossRef]
- Rebbeck, T.R. Prostate Cancer Disparities by Race and Ethnicity: From Nucleotide to Neighborhood. Cold Spring Harb. Perspect. Med. 2018, 8, a030387. [Google Scholar] [CrossRef]
- Ames, B.N. DNA damage from micronutrient deficiencies is likely to be a major cause of cancer. Mutat. Res. 2001, 475, 7–20. [Google Scholar] [CrossRef]
- Hernando-Herraez, I.; Heyn, H.; Fernandez-Callejo, M.; Vidal, E.; Fernandez-Bellon, H.; Prado-Martinez, J.; Sharp, A.J.; Esteller, M.; Marques-Bonet, T. The interplay between DNA methylation and sequence divergence in recent human evolution. Nucleic Acids Res. 2015, 43, 8204–8214. [Google Scholar] [CrossRef]
- Sanchez, H.; Hossain, M.B.; Lera, L.; Hirsch, S.; Albala, C.; Uauy, R.; Broberg, K.; Ronco, A.M. High levels of circulating folate concentrations are associated with DNA methylation of tumor suppressor and repair genes p16, MLH1, and MGMT in elderly Chileans. Clin. Epigenet. 2017, 9, 74. [Google Scholar] [CrossRef]
- Selhub, J.; Jacques, P.F.; Wilson, P.W.; Rush, D.; Rosenberg, I.H. Vitamin status and intake as primary determinants of homocysteinemia in an elderly population. JAMA 1993, 270, 2693–2698. [Google Scholar] [CrossRef]
- Halliwell, B. Free radicals and antioxidants: A personal view. Nutr. Rev. 1994, 52 Pt 1, 253–265. [Google Scholar] [CrossRef]
- Yang, C.S.; Luo, P.; Zeng, Z.; Wang, H.; Malafa, M.; Suh, N. Vitamin E and cancer prevention: Studies with different forms of tocopherols and tocotrienols. Mol. Carcinog. 2020, 59, 365–389. [Google Scholar] [CrossRef]
- Grenier, E.; Maupas, F.S.; Beaulieu, J.F.; Seidman, E.; Delvin, E.; Sane, A.; Tremblay, E.; Garofalo, C.; Levy, E. Effect of retinoic acid on cell proliferation and differentiation as well as on lipid synthesis, lipoprotein secretion, and apolipoprotein biogenesis. Am. J. Physiol. Gastrointest. Liver Physiol. 2007, 293, G1178–G1189. [Google Scholar] [CrossRef]
- Mishra, S.; Kelly, K.K.; Rumian, N.L.; Siegenthaler, J.A. Retinoic Acid Is Required for Neural Stem and Progenitor Cell Proliferation in the Adult Hippocampus. Stem Cell Rep. 2018, 10, 1705–1720. [Google Scholar] [CrossRef]
- Krinsky, N.I.; Johnson, E.J. Carotenoid actions and their relation to health and disease. Mol. Asp. Med. 2005, 26, 459–516. [Google Scholar] [CrossRef]
- Mohsenzadegan, M.; Seif, F.; Farajollahi, M.M.; Khoshmirsafa, M. Anti-Oxidants as Chemopreventive Agents in Prostate Cancer: A Gap Between Preclinical and Clinical Studies. Recent Pat. Anticancer Drug Discov. 2018, 13, 224–239. [Google Scholar] [CrossRef]
- Ripple, M.O.; Henry, W.F.; Rago, R.P.; Wilding, G. Prooxidant-antioxidant shift induced by androgen treatment of human prostate carcinoma cells. J. Natl. Cancer Inst. 1997, 89, 40–48. [Google Scholar] [CrossRef]
- Iynem, A.H.; Alademir, A.Z.; Obek, C.; Kural, A.R.; Konukoğlu, D.; Akçay, T. The effect of prostate cancer and antiandrogenic therapy on lipid peroxidation and antioxidant systems. Int. Urol. Nephrol. 2004, 36, 57–62. [Google Scholar]
- Chan, J.M.; Gann, P.H.; Giovannucci, E.L. Role of diet in prostate cancer development and progression. J. Clin. Oncol. 2005, 23, 8152–8160. [Google Scholar] [CrossRef]
- Oczkowski, M.; Dziendzikowska, K.; Pasternak-Winiarska, A.; Włodarek, D.; Gromadzka-Ostrowska, J. Dietary Factors and Prostate Cancer Development, Progression, and Reduction. Nutrients 2021, 13, 496. [Google Scholar] [CrossRef]
- Chang, S.; Erdman, J.W., Jr.; Clinton, S.K.; Vadiveloo, M.; Strom, S.S.; Yamamura, Y.; Duphorne, C.M.; Spitz, M.R.; Amos, C.I.; Contois, J.H.; et al. Relationship between plasma carotenoids and prostate cancer. Nutr. Cancer 2005, 53, 127–134. [Google Scholar] [CrossRef]
- Antwi, S.O.; Steck, S.E.; Zhang, H.; Stumm, L.; Zhang, J.; Hurley, T.G.; Hebert, J.R. Plasma carotenoids and tocopherols in relation to prostate-specific antigen (PSA) levels among men with biochemical recurrence of prostate cancer. Cancer Epidemiol. 2015, 39, 752–762. [Google Scholar] [CrossRef]
- Nordström, T.; Van Blarigan, E.L.; Ngo, V.; Roy, R.; Weinberg, V.; Song, X.; Simko, J.; Carroll, P.R.; Chan, J.M.; Paris, P.L. Associations between circulating carotenoids, genomic instability and the risk of high-grade prostate cancer. Prostate 2016, 76, 339–348. [Google Scholar] [CrossRef]
- Peters, U.; Leitzmann, M.F.; Chatterjee, N.; Wang, Y.; Albanes, D.; Gelmann, E.P.; Friesen, M.D.; Riboli, E.; Hayes, R.B. Serum lycopene, other carotenoids, and prostate cancer risk: A nested case-control study in the prostate, lung, colorectal, and ovarian cancer screening trial. Cancer Epidemiol. Biomark. Prev. 2007, 16, 962–968. [Google Scholar] [CrossRef]
- van Breemen, R.B.; Sharifi, R.; Viana, M.; Pajkovic, N.; Zhu, D.; Yuan, L.; Yang, Y.; Bowen, P.E.; Stacewicz-Sapuntzakis, M. Antioxidant effects of lycopene in African American men with prostate cancer or benign prostate hyperplasia: A randomized, controlled trial. Cancer Prev. Res. 2011, 4, 711–718. [Google Scholar] [CrossRef]
- Srivastava, A.R.; Dalela, D. Prostate cancer: Altering the natural history by dietary changes. Natl. Med. J. India 2004, 17, 248–253. [Google Scholar]
- Allen, N.E.; Sauvaget, C.; Roddam, A.W.; Appleby, P.; Nagano, J.; Suzuki, G.; Key, T.J.; Koyama, K. A prospective study of diet and prostate cancer in Japanese men. Cancer Causes Control 2004, 15, 911–920. [Google Scholar] [CrossRef]
- Bostwick, D.G.; Burke, H.B.; Djakiew, D.; Euling, S.; Ho, S.M.; Landolph, J.; Morrison, H.; Sonawane, B.; Shifflett, T.; Waters, D.J.; et al. Human prostate cancer risk factors. Cancer 2004, 101 (Suppl. 10), 2371–2490. [Google Scholar] [CrossRef]
- Capiod, T.; Barry Delongchamps, N.; Pigat, N.; Souberbielle, J.C.; Goffin, V. Do dietary calcium and vitamin D matter in men with prostate cancer? Nat. Rev. Urol. 2018, 15, 453–461. [Google Scholar] [CrossRef]
- Hayashi, T.; Fujita, K.; Nojima, S.; Hayashi, Y.; Nakano, K.; Ishizuya, Y.; Wang, C.; Yamamoto, Y.; Kinouchi, T.; Matsuzaki, K.; et al. High-Fat Diet-Induced Inflammation Accelerates Prostate Cancer Growth via IL6 Signaling. Clin. Cancer Res. 2018, 24, 4309–4318. [Google Scholar] [CrossRef]
- Di Maso, M.; Augustin, L.S.A.; Toffolutti, F.; Stocco, C.; Dal Maso, L.; Jenkins, D.J.A.; Fleshner, N.E.; Serraino, D.; Polesel, J. Adherence to Mediterranean Diet, Physical Activity and Survival after Prostate Cancer Diagnosis. Nutrients 2021, 13, 243. [Google Scholar] [CrossRef]
- Wilson, R.L.; Taaffe, D.R.; Newton, R.U.; Hart, N.H.; Lyons-Wall, P.; Galvão, D.A. Using Exercise and Nutrition to Alter Fat and Lean Mass in Men with Prostate Cancer Receiving Androgen Deprivation Therapy: A Narrative Review. Nutrients 2021, 13, 1664. [Google Scholar] [CrossRef]
- Stanisławska, I.J.; Figat, R.; Kiss, A.K.; Bobrowska-Korczak, B. Essential Elements and Isoflavonoids in the Prevention of Prostate Cancer. Nutrients 2022, 14, 1225. [Google Scholar] [CrossRef]
- Feng, P.; Li, T.L.; Guan, Z.X.; Franklin, R.B.; Costello, L.C. Effect of zinc on prostatic tumorigenicity in nude mice. Ann. N. Y. Acad. Sci. 2003, 1010, 316–320. [Google Scholar] [CrossRef] [PubMed]
- Platz, E.A.; Helzlsouer, K.J.; Hoffman, S.C.; Morris, J.S.; Baskett, C.K.; Comstock, G.W. Prediagnostic toenail cadmium and zinc and subsequent prostate cancer risk. Prostate 2002, 52, 288–296. [Google Scholar] [CrossRef] [PubMed]
- Masih, K.; Vadehra, P.L.; Prabhakar, B.R. Serum copper levels in prostatic disorders. Indian J. Pathol. MicroBiol. 1985, 28, 67–70. [Google Scholar] [PubMed]
- Nyman, D.W.; Suzanne Stratton, M.; Kopplin, M.J.; Dalkin, B.L.; Nagle, R.B.; Jay Gandolfi, A. Selenium and selenomethionine levels in prostate cancer patients. Cancer Detect. Prev. 2004, 28, 8–16. [Google Scholar] [CrossRef]
- Sun, Y.; Selvaraj, S.; Varma, A.; Derry, S.; Sahmoun, A.E.; Singh, B.B. Increase in serum Ca2+/Mg2+ ratio promotes proliferation of prostate cancer cells by activating TRPM7 channels. J. Biol. Chem. 2013, 288, 255–263. [Google Scholar] [CrossRef]
- Li, H.; Stampfer, M.J.; Giovannucci, E.L.; Morris, J.S.; Willett, W.C.; Gaziano, J.M.; Ma, J. A prospective study of plasma selenium levels and prostate cancer risk. J. Natl. Cancer Inst. 2004, 96, 696–703. [Google Scholar] [CrossRef]
- Pourmand, G.; Salem, S.; Moradi, K.; Nikoobakht, M.R.; Tajik, P.; Mehrsai, A. Serum selenium level and prostate cancer: A case-control study. Nutr. Cancer 2008, 60, 171–176. [Google Scholar] [CrossRef]
- Cui, Z.; Liu, D.; Liu, C.; Liu, G. Serum selenium levels and prostate cancer risk: A MOOSE-compliant meta-analysis. Medicine 2017, 96, e5944. [Google Scholar] [CrossRef]
- Tapiero, H.; Townsend, D.M.; Tew, K.D. The antioxidant role of selenium and seleno-compounds. BioMed. Pharmacother. 2003, 57, 134–144. [Google Scholar] [CrossRef]
- Donadio, J.L.S.; Rogero, M.M.; Guerra-Shinohara, E.M.; Barbosa, F., Jr.; Desmarchelier, C.; Borel, P.; Sneddon, A.A.; Hesketh, J.E.; Cozzolino, S.M.F. Genetic variants in selenoprotein genes modulate biomarkers of selenium status in response to Brazil nut supplementation (the SU.BRA.NUT study). Clin. Nutr. 2019, 38, 539–548. [Google Scholar] [CrossRef]
- Tappel, A. Heme of consuMed. red meat can act as a catalyst of oxidative damage and could initiate colon, breast and prostate cancers, heart disease and other diseases. Med. Hypotheses 2007, 68, 562–564. [Google Scholar] [CrossRef] [PubMed]
- Prá, D.; Bortoluzzi, A.; Müller, L.L.; Hermes, L.; Horta, J.A.; Maluf, S.W.; Henriques, J.A.; Fenech, M.; Franke, S.I. Iron intake, red cell indicators of iron status, and DNA damage in young subjects. Nutrition 2011, 27, 293–297. [Google Scholar] [CrossRef]
- Ho, E. Zinc deficiency, DNA damage and cancer risk. J. Nutr. BioChem. 2004, 15, 572–578. [Google Scholar] [CrossRef] [PubMed]
- Sharif, R.; Thomas, P.; Zalewski, P.; Fenech, M. The role of zinc in genomic stability. Mutat. Res. 2012, 733, 111–121. [Google Scholar] [CrossRef]
- Franklin, R.B.; Milon, B.; Feng, P.; Costello, L.C. Zinc and zinc transporters in normal prostate and the pathogenesis of prostate cancer. Front. BioSci. 2005, 10, 2230–2239. [Google Scholar] [CrossRef] [PubMed]
- Gleason, D.F. Classification of prostatic carcinomas. Cancer Chemother Rep. 1966, 50, 125–128. [Google Scholar] [PubMed]
- Dhillon, V.S.; Zabaras, D.; Almond, T.; Cavuoto, P.; James-Martin, G.; Fenech, M. Whey protein isolate improves vitamin B(12) and folate status in elderly Australians with subclinical deficiency of vitamin B(12). Mol. Nutr. Food Res. 2017, 61. [Google Scholar] [CrossRef]
- Fenech, M.; Baghurst, P.; Luderer, W.; Turner, J.; Record, S.; Ceppi, M.; Bonassi, S. Low intake of calcium, folate, nicotinic acid, vitamin E, retinol, beta-carotene and high intake of pantothenic acid, biotin and riboflavin are significantly associated with increased genome instability--results from a dietary intake and micronucleus index survey in South Australia. Carcinogenesis 2005, 26, 991–999. [Google Scholar]
- Fenech, M. Folate (vitamin B9) and vitamin B12 and their function in the maintenance of nuclear and mitochondrial genome integrity. Mutat. Res. 2012, 733, 21–33. [Google Scholar] [CrossRef]
- Trujillo, E.; Davis, C.; Milner, J. Nutrigenomics, proteomics, metabolomics, and the practice of dietetics. J. Am. Diet. Assoc. 2006, 106, 403–413. [Google Scholar] [CrossRef]
- Ulrich, C.M.; Potter, J.D. Folate supplementation: Too much of a good thing? Cancer Epidemiol. Biomark. Prev. 2006, 15, 189–193. [Google Scholar] [CrossRef]
- Rees, J.R.; Morris, C.B.; Peacock, J.L.; Ueland, P.M.; Barry, E.L.; McKeown-Eyssen, G.E.; Figueiredo, J.C.; Snover, D.C.; Baron, J.A. Unmetabolized Folic Acid, Tetrahydrofolate, and Colorectal Adenoma Risk. Cancer Prev. Res. 2017, 10, 451–458. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Stabler, S.P. Clinical practice. Vitamin B12 deficiency. N. Engl. J. Med. 2013, 368, 149–160. [Google Scholar] [CrossRef] [PubMed]
- Green, R.; Allen, L.H.; Bjørke-Monsen, A.L.; Brito, A.; Guéant, J.L.; Miller, J.W.; Molloy, A.M.; Nexo, E.; Stabler, S.; Toh, B.H.; et al. Vitamin B(12) deficiency. Nat. Rev. Dis. Prim. 2017, 3, 17040. [Google Scholar] [CrossRef] [PubMed]
- Smith, A.D.; Warren, M.J.; Refsum, H. Vitamin B(12). Adv. Food Nutr. Res. 2018, 83, 215–279. [Google Scholar]
- Collin, S.M.; Metcalfe, C.; Refsum, H.; Lewis, S.J.; Zuccolo, L.; Smith, G.D.; Chen, L.; Harris, R.; Davis, M.; Marsden, G.; et al. Circulating folate, vitamin B12, homocysteine, vitamin B12 transport proteins, and risk of prostate cancer: A case-control study, systematic review, and meta-analysis. Cancer Epidemiol. Biomark. Prev. 2010, 19, 1632–1642. [Google Scholar] [CrossRef]
- Awwad, H.M.; Ohlmann, C.H.; Stoeckle, M.; Geisel, J.; Obeid, R. Serum concentrations of folate vitamers in patients with a newly diagnosed prostate cancer or hyperplasia. Clin. BioChem. 2018, 56, 41–46. [Google Scholar] [CrossRef]
- Obeid, R.; Kirsch, S.H.; Dilmann, S.; Klein, C.; Eckert, R.; Geisel, J.; Herrmann, W. Folic acid causes higher prevalence of detectable unmetabolized folic acid in serum than B-complex: A randomized trial. Eur. J. Nutr. 2016, 55, 1021–1028. [Google Scholar] [CrossRef]
- Price, A.J.; Travis, R.C.; Appleby, P.N.; Albanes, D.; Barricarte Gurrea, A.; Bjørge, T.; Bueno-de-Mesquita, H.B.; Chen, C.; Donovan, J.; Gislefoss, R.; et al. Circulating Folate and Vitamin B(12) and Risk of Prostate Cancer: A Collaborative Analysis of Individual Participant Data from Six Cohorts Including 6875 Cases and 8104 Controls. Eur. Urol. 2016, 70, 941–951. [Google Scholar] [CrossRef]
- Weinstein, S.J.; Hartman, T.J.; Stolzenberg-Solomon, R.; Pietinen, P.; Barrett, M.J.; Taylor, P.R.; Virtamo, J.; Albanes, D. Null association between prostate cancer and serum folate, vitamin B(6), vitamin B(12), and homocysteine. Cancer Epidemiol. Biomark. Prev. 2003, 12 Pt 1, 1271–1272. [Google Scholar]
- Ebbing, M.; Bønaa, K.H.; Nygård, O.; Arnesen, E.; Ueland, P.M.; Nordrehaug, J.E.; Rasmussen, K.; Njølstad, I.; Refsum, H.; Nilsen, D.W.; et al. Cancer incidence and mortality after treatment with folic acid and vitamin B12. JAMA 2009, 302, 2119–2126. [Google Scholar] [CrossRef] [PubMed]
- Milani, A.; Basirnejad, M.; Shahbazi, S.; Bolhassani, A. Carotenoids: Biochemistry, pharmacology and treatment. Br. J. Pharmacol. 2017, 174, 1290–1324. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, T.; Shnimizu, M.; Moriwaki, H. Cancer chemoprevention by carotenoids. Molecules 2012, 17, 3202–3242. [Google Scholar] [CrossRef] [PubMed]
- Clinton, S.K.; Emenhiser, C.; Schwartz, S.J.; Bostwick, D.G.; Williams, A.W.; Moore, B.J.; Erdman, J.W., Jr. cis-trans lycopene isomers, carotenoids, and retinol in the human prostate. Cancer Epidemiol. Biomark. Prev. 1996, 5, 823–833. [Google Scholar]
- Zu, K.; Mucci, L.; Rosner, B.A.; Clinton, S.K.; Loda, M.; Stampfer, M.J.; Giovannucci, E. Dietary lycopene, angiogenesis, and prostate cancer: A prospective study in the prostate-specific antigen era. J. Natl. Cancer Inst. 2014, 106, djt430. [Google Scholar] [CrossRef] [PubMed]
- Rowles, J.L., 3rd; Ranard, K.M.; Smith, J.W.; An, R.; Erdman, J.W., Jr. Increased dietary and circulating lycopene are associated with reduced prostate cancer risk: A systematic review and meta-analysis. Prostate Cancer Prostatic Dis. 2017, 20, 361–377. [Google Scholar] [CrossRef]
- López-Guarnido, O.; Urquiza-Salvat, N.; Saiz, M.; Lozano-Paniagua, D.; Rodrigo, L.; Pascual-Geler, M.; Lorente, J.A.; Alvarez-Cubero, M.J.; Rivas, A. Bioactive compounds of the Mediterranean diet and prostate cancer. Aging Male 2018, 21, 251–260. [Google Scholar] [CrossRef]
- Key, T.J.; Appleby, P.N.; Travis, R.C.; Albanes, D.; Alberg, A.J.; Barricarte, A.; Black, A.; Boeing, H.; Bueno-de-Mesquita, H.B.; Chan, J.M.; et al. Carotenoids, retinol, tocopherols, and prostate cancer risk: Pooled analysis of 15 studies. Am. J. Clin. Nutr. 2015, 102, 1142–1157. [Google Scholar] [CrossRef]
- Carazo, A.; Macáková, K.; Matoušová, K.; Krčmová, L.K.; Protti, M.; Mladěnka, P. Vitamin A Update: Forms, Sources, Kinetics, Detection, Function, Deficiency, Therapeutic Use and Toxicity. Nutrients 2021, 13, 1703. [Google Scholar] [CrossRef]
- Knape, M.J.; Ballez, M.; Burghardt, N.C.; Zimmermann, B.; Bertinetti, D.; Kornev, A.P.; Herberg, F.W. Divalent metal ions control activity and inhibition of protein kinases. Metallomics 2017, 9, 1576–1584. [Google Scholar] [CrossRef]
- Pasha, Q.; Malik, S.A.; Shah, M.H. Statistical analysis of trace metals in the plasma of cancer patients versus controls. J. Hazard Mater. 2008, 153, 1215–1221. [Google Scholar] [CrossRef] [PubMed]
- Jablonska, E.; Vinceti, M. Selenium and Human Health: Witnessing a Copernican Revolution? J. Environ. Sci. Health C Environ. Carcinog Ecotoxicol. Rev. 2015, 33, 328–368. [Google Scholar] [CrossRef] [PubMed]
- Vinceti, M.; Filippini, T.; Cilloni, S.; Crespi, C.M. The Epidemiology of Selenium and Human Cancer. Adv. Cancer Res. 2017, 136, 1–48. [Google Scholar] [PubMed]
- Allen, N.E.; Travis, R.C.; Appleby, P.N.; Albanes, D.; Barnett, M.J.; Black, A.; Bueno-de-Mesquita, H.B.; Deschasaux, M.; Galan, P.; Goodman, G.E.; et al. Selenium and Prostate Cancer: Analysis of Individual Participant Data From Fifteen Prospective Studies. J. Natl. Cancer Inst. 2016, 108, djw153. [Google Scholar] [CrossRef]
- Yarmolinsky, J.; Bonilla, C.; Haycock, P.C.; Langdon, R.J.Q.; Lotta, L.A.; Langenberg, C.; Relton, C.L.; Lewis, S.J.; Evans, D.M.; Davey Smith, G.; et al. Circulating Selenium and Prostate Cancer Risk: A Mendelian Randomization Analysis. J. Natl. Cancer Inst. 2018, 110, 1035–1038. [Google Scholar] [CrossRef] [PubMed]
- Brigelius-Flohé, R.; Kelly, F.J.; Salonen, J.T.; Neuzil, J.; Zingg, J.M.; Azzi, A. The European perspective on vitamin E: Current knowledge and future research. Am. J. Clin. Nutr. 2002, 76, 703–716. [Google Scholar] [CrossRef]
- Mascitelli, L.; Goldstein, M.R. Metabolic syndrome and liver cancer: Is excess iron the link? Hepatology 2011, 54, 1487. [Google Scholar] [CrossRef]
- Karimi, G.; Shahar, S.; Homayouni, N.; Rajikan, R.; Abu Bakar, N.F.; Othman, M.S. Association between trace element and heavy metal levels in hair and nail with prostate cancer. Asian Pac. J. Cancer Prev. 2012, 13, 4249–4253. [Google Scholar] [CrossRef]
- Nandeesha, H.; Eldhose, A.; Dorairajan, L.N.; Anandhi, B. Hypoadiponectinemia, elevated iron and high-sensitivity C-reactive protein levels and their relation with prostate size in benign prostatic hyperplasia. Andrologia 2017, 49, e12715. [Google Scholar] [CrossRef]
- Ozmen, H.; Erulas, F.A.; Karatas, F.; Cukurovali, A.; Yalcin, O. Comparison of the concentration of trace metals (Ni, Zn, Co, Cu and Se), Fe, vitamins A, C and E, and lipid peroxidation in patients with prostate cancer. Clin. Chem. Lab. Med. 2006, 44, 175–179. [Google Scholar] [CrossRef]
- World Cancer Research Fund. Diet, Nutrition, Physical Activity, and Prostate Cancer; WCRF International: London, UK, 2014. [Google Scholar]
- Aune, D.; Navarro Rosenblatt, D.A.; Chan, D.S.; Vieira, A.R.; Vieira, R.; Greenwood, D.C.; Vatten, L.J.; Norat, T. Dairy products, calcium, and prostate cancer risk: A systematic review and meta-analysis of cohort studies. Am. J. Clin. Nutr. 2015, 101, 87–117. [Google Scholar] [CrossRef] [PubMed]
- Thorning, T.K.; Raben, A.; Tholstrup, T.; Soedamah-Muthu, S.S.; Givens, I.; Astrup, A. Milk and dairy products: Good or bad for human health? An assessment of the totality of scientific evidence. Food Nutr. Res. 2016, 60, 32527. [Google Scholar] [CrossRef] [PubMed]
- Giovannucci, E.; Pollak, M.; Liu, Y.; Platz, E.A.; Majeed, N.; Rimm, E.B.; Willett, W.C. Nutritional predictors of insulin-like growth factor I and their relationships to cancer in men. Cancer Epidemiol. Biomark. Prev. 2003, 12, 84–89. [Google Scholar]
- Skinner, H.G.; Schwartz, G.G. A prospective study of total and ionized serum calcium and fatal prostate cancer. Cancer Epidemiol. Biomark. Prev. 2009, 18, 575–578. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Brändstedt, J.; Almquist, M.; Ulmert, D.; Manjer, J.; Malm, J. Vitamin D, PTH, and calcium and tumor aggressiveness in prostate cancer: A prospective nested case-control study. Cancer Causes Control 2016, 27, 69–80. [Google Scholar] [CrossRef] [PubMed]
- Bonjour, J.P.; Chevalley, T.; Fardellone, P. Calcium intake and vitamin D metabolism and action, in healthy conditions and in prostate cancer. Br. J. Nutr. 2007, 97, 611–616. [Google Scholar] [CrossRef]
- Kim, B.E.; Nevitt, T.; Thiele, D.J. Mechanisms for copper acquisition, distribution and regulation. Nat. Chem. Biol. 2008, 4, 176–185. [Google Scholar] [CrossRef]
- An, Y.; Li, S.; Huang, X.; Chen, X.; Shan, H.; Zhang, M. The Role of Copper Homeostasis in Brain Disease. Int. J. Mol. Sci. 2022, 23, 13850. [Google Scholar] [CrossRef]
- Linder, M.C. The relationship of copper to DNA damage and damage prevention in humans. Mutat. Res. 2012, 733, 83–91. [Google Scholar] [CrossRef]
- Nayak, S.B.; Bhat, V.R.; Upadhyay, D.; Udupa, S.L. Copper and ceruloplasmin status in serum of prostate and colon cancer patients. Indian J. Physiol. Pharmacol 2003, 47, 108–110. [Google Scholar]
- Leone, N.; Courbon, D.; Ducimetiere, P.; Zureik, M. Zinc, copper, and magnesium and risks for all-cause, cancer, and cardiovascular mortality. Epidemiology 2006, 17, 308–314. [Google Scholar] [CrossRef] [PubMed]
- Gupte, A.; Mumper, R.J. Elevated copper and oxidative stress in cancer cells as a target for cancer treatment. Cancer Treat. Rev. 2009, 35, 32–46. [Google Scholar] [CrossRef] [PubMed]
- Kaba, M.; Pirincci, N.; Yuksel, M.B.; Gecit, I.; Gunes, M.; Ozveren, H.; Eren, H.; Demir, H. Serum levels of trace elements in patients with prostate cancer. Asian Pac. J. Cancer Prev. 2014, 15, 2625–2629. [Google Scholar] [CrossRef] [PubMed]
- Turski, M.L.; Brady, D.C.; Kim, H.J.; Kim, B.E.; Nose, Y.; Counter, C.M.; Winge, D.R.; Thiele, D.J. A novel role for copper in Ras/mitogen-activated protein kinase signaling. Mol. Cell Biol. 2012, 32, 1284–1295. [Google Scholar] [CrossRef] [PubMed]
- Wach, S.; Weigelt, K.; Michalke, B.; Lieb, V.; Stoehr, R.; Keck, B.; Hartmann, A.; Wullich, B.; Taubert, H.; Chaudhri, A. Diagnostic potential of major and trace elements in the serum of bladder cancer patients. J. Trace Elem Med. Biol. 2018, 46, 150–155. [Google Scholar] [CrossRef]
- Szabo, C.; Coletta, C.; Chao, C.; Módis, K.; Szczesny, B.; Papapetropoulos, A.; Hellmich, M.R. Tumor-derived hydrogen sulfide, produced by cystathionine-β-synthase, stimulates bioenergetics, cell proliferation, and angiogenesis in colon cancer. Proc. Natl. Acad. Sci. USA 2013, 110, 12474–12479. [Google Scholar] [CrossRef]
- Hoffman, R.M. The wayward methyl group and the cascade to cancer. Cell Cycle 2017, 16, 825–829. [Google Scholar] [CrossRef]
- Hoffman, R.M. Methionine Dependency Determination of Human Patient Tumors in Gelfoam(®) Histoculture. Methods Mol. Biol. 2018, 1760, 125–131. [Google Scholar]
| Characteristics | Cases | Controls | p Value |
|---|---|---|---|
| Age (years; Mean ± SD) | 71.24 ± 7.18 | 69.07 ± 7.99 | 0.88 |
| Total plasma PSA (ng/mL; mean ± SD) | 9.5 ± 8.5 | 2.4 ± 2.45 | 0.0001 * |
| Gleason Score | 6–9 | - | - |
| Smoking Status | |||
| Current Smokers | 9 | 3 | 0.0001 * |
| Ex-Smokers | 60 | 39 | |
| Non-Smokers | 25 | 54 | |
| Undeclared | 24 | 36 |
| Micronutrients/ Trace Elements | Controls | Prostate Cancer | p Value |
|---|---|---|---|
| Homocysteine (μmol/L) | 10.27 ± 0.28 (5.5–23.4) | 12.21 ± 1.009 (4.6–94.0) | 0.25 |
| B12 (pmol/L) | 284.5 ± 11.4 (121–862) | 276.8 ± 15.97 (44–1450) | 0.24 |
| Folate (nmol/L) | 19.26 ± 0.78 (5.4–37.7) | 18.22 ± 0.96 (2.9–45) | 0.2 |
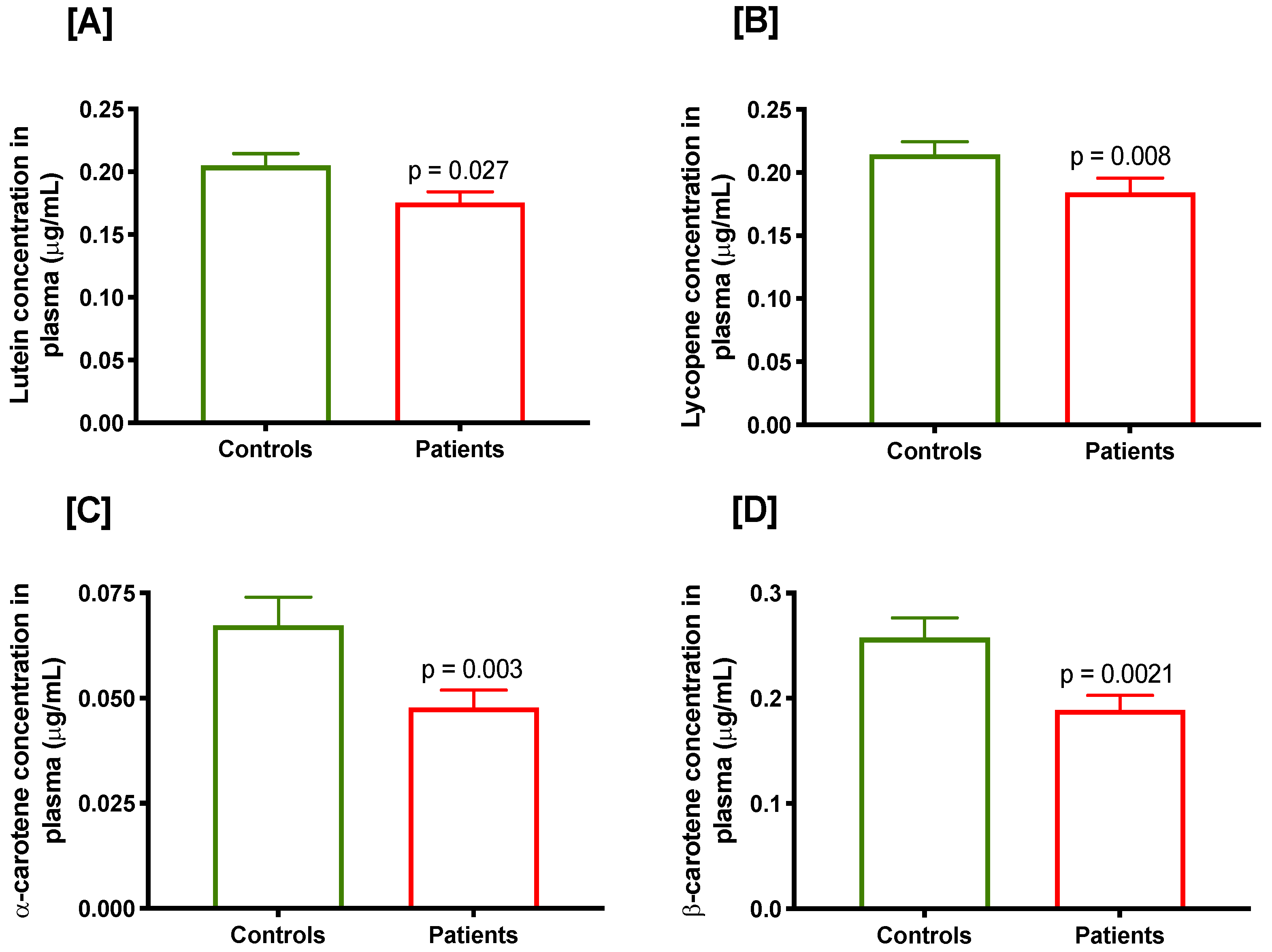
| Lutein (μg/mL) | 0.205 ± 0.009 (0.06–0.64) | 0.176 ± 0.009 (0.029–0.553) | 0.027 * |
| Retinol (μg/mL) | 0.678 ± 0.015 (0.38–1.32) | 0.654 ± 0.015 (0.414–1.086) | 0.29 |
| α-Tocopherol (μg/mL) | 13.17 ± 0.36 (6.35–29.4) | 13.31 ± 0.42 (7.07–37.48) | 0.63 |
| Lycopene (μg/mL) | 0.215 ± 0.009 (0.03–0.58) | 0.184 ± 0.011 (0/013–0.655) | 0.008 * |
| α-Carotene (μg/mL) | 0.067 ± 0.006 (0.01–0.61) | 0.048 ± 0.004 (0.002–0.273) | 0.003 * |
| β-Carotene (μg/mL) | 0.258 ± 0.019 (0.03–1.36) | 0.189 ± 0.014 (0/017–0.704) | 0.002 * |
| Trace Elements | Controls | Prostate Cancer | p Value |
|---|---|---|---|
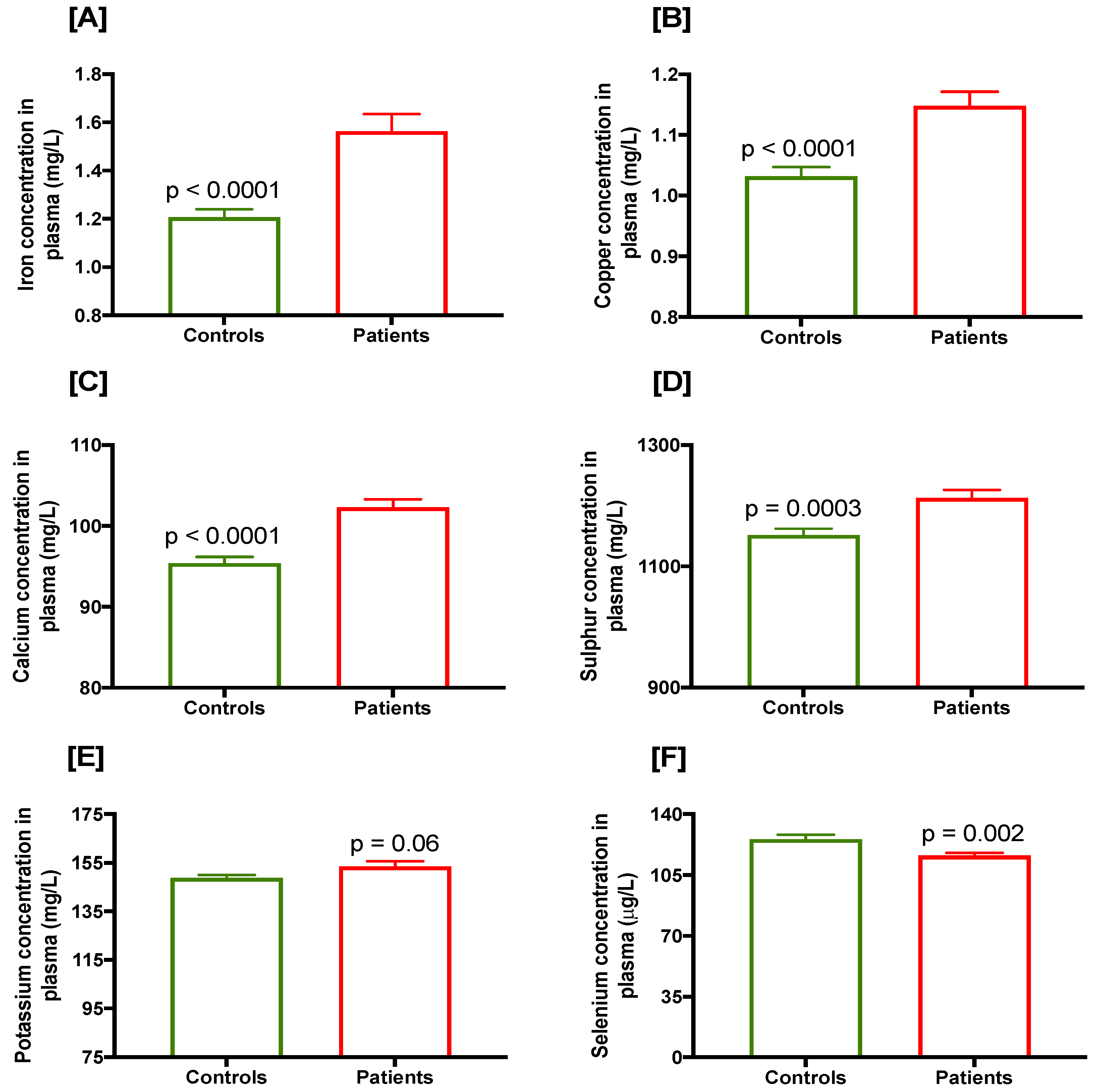
| Selenium (μg/L) | 125.6 ± 2.56 (79.17–238.1) | 116.1 ± 1.59 (71.83–157.6) | 0.002 * |
| Zinc (μg/mL) | 0.78 ± 0.01 (0.52–1.04) | 0.77 ± 0.1 (0.52–1.24) | 0.23 |
| Iron (mg/L) | 1.207 ± 0.03 (0.5–2.3) | 1.564 ± 0.07 (0.52–4.7) | <0.0001 * |
| Copper (mg/L) | 1.032 ± 0.015 (0.6–1.5) | 1.148 ± 0.023 (0.58–2.0) | <0.0001 * |
| Calcium (mg/L) | 95.4 ± 0.79 (79–119) | 102.3 ± 1.006 (77–128) | <0.0001 * |
| Magnesium (mg/L) | 21.35 ± 0.208 (17–28) | 21.78 ± 0.261 (15–27) | 0.19 |
| Sodium (mg/L) | 3338 ± 22.89 (2835–4095) | 3395 ± 27.41 (2600–4000) | 0.19 |
| Potassium (mg/L) | 148.9 ± 1.18 (114–182) | 153.5 ± 2.22 (100–222) | 0.06 |
| Phosphorus (mg/L) | 124.6 ± 1.81 (87–176) | 128.2 ± 2.15 (81–189) | 0.19 |
| Sulphur (mg/L) | 1152 ± 10.19 (960–1403) | 1213 ± 13.18 (870–1560) | 0.0003 * |
| Micronutrient/ Trace Element | Odds Ratio (95% CI) | p Value | |
|---|---|---|---|
| Micronutrients which Tend to be Higher in Controls | Lutein | ||
| ≥Median (0.18 μg/mL) | 1 (Ref) | ||
| <Median (0.18 μg/mL) | 1.35 (0.79–2.31) | 0.28 | |
| Lycopene # | |||
| ≥Median (0.205 μg/mL) | 1 (Ref) | ||
| <Median (0.205 μg/mL) | 2.24 (1.29–3.88) | 0.006 * | |
| α-Carotene # | |||
| ≥Median (0.05 μg/mL) | 1 (Ref) | ||
| <Median (0.05 μg/mL) | 2.13 (1.23–3.69) | 0.009 * | |
| β-Carotene # | 1 (Ref) | ||
| ≥Median (0.21 μg/mL) | 1 (Ref) | ||
| <Median (0.21 μg/mL) | 1.97 (1.41–3.40) | 0.01 * | |
| Selenium | |||
| ≥Median (118.5 μmol/L) | 1 (Ref) | ||
| <Median (118.5 μmol/L) | 1.36 (0.79–2.33) | 0.28 | |
| Micronutrients which Tend to be Lower in Controls | Iron # | ||
| <Median (1.2 mg/L) | 1 (Ref) | ||
| ≥Median (1.2 mg/L) | 2.31 (1.29–4.16) | 0.006 * | |
| Copper | |||
| <Median (1 mg/L) | 1 (Ref) | ||
| ≥Median (1 mg/L) | 0.94 (0.5–1.77) | 0.87 | |
| Calcium # | |||
| <Median (94 mg/L) | 1 (Ref) | ||
| ≥Median (94 mg/L) | 4.35 (2.24–8.44) | <0.0001 * | |
| Sulphur # | |||
| <Median (1150 mg/L) | 1 (Ref) | ||
| ≥Median (1150 mg/L) | 2.39 (1.33–4.27) | 0.004 * | |
| Selenium/Lycopene # | |||
| >120 Selenium/>0.25 Lycopene | 1 (Ref) | ||
| ≤120 Selenium/≤0.25 Lycopene | 2.97 (1.19–7.41) | 0.02 * | |
| Micronutrient Risk Index | ||||||
|---|---|---|---|---|---|---|
| ≥1 | ≥2 | ≥3 | ≥4 | ≥5 | 6 | |
| p Value | 0.13 | 0.005 | 0.0003 | < 0.0001 | < 0.0001 | 0.001 |
| Odds Ratio (95% CI) | 3.23 (0.64–16.41) | 4.03 (1.42–11.4) | 3.37 (1.72–6.58) | 3.14 (1.75–5.62) | 3.89 (2.09–7.24) | 4.88 (1.76–13.54) |
| RR (95% CI) | 2.07 (0.62–6.94) | 2.37 (1.09–5.22) | 1.98 (1.28–3.05) | 1.79 (1.3–2.46) | 1.83 (1.41–2.38) | 1.75 (1.36–2.24) |
| Sensitivity | 0.98 | 0.95 | 0.84 | 0.69 | 0.51 | 0.21 |
| Specificity | 0.06 | 0.17 | 0.38 | 0.58 | 0.78 | 0.95 |
| PPV | 0.52 | 0.54 | 0.59 | 0.63 | 0.71 | 0.81 |
| NPV | 0.75 | 0.77 | 0.7 | 0.65 | 0.61 | 0.54 |
| Likelihood Ratio | 1.04 | 1.15 | 1.38 | 1.66 | 2.4 | 4.08 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dhillon, V.S.; Deo, P.; Fenech, M. Plasma Micronutrient Profile of Prostate Cancer Cases Is Altered Relative to Healthy Controls—Results of a Pilot Study in South Australia. Cancers 2023, 15, 77. https://doi.org/10.3390/cancers15010077
Dhillon VS, Deo P, Fenech M. Plasma Micronutrient Profile of Prostate Cancer Cases Is Altered Relative to Healthy Controls—Results of a Pilot Study in South Australia. Cancers. 2023; 15(1):77. https://doi.org/10.3390/cancers15010077
Chicago/Turabian StyleDhillon, Varinderpal S., Permal Deo, and Michael Fenech. 2023. "Plasma Micronutrient Profile of Prostate Cancer Cases Is Altered Relative to Healthy Controls—Results of a Pilot Study in South Australia" Cancers 15, no. 1: 77. https://doi.org/10.3390/cancers15010077
APA StyleDhillon, V. S., Deo, P., & Fenech, M. (2023). Plasma Micronutrient Profile of Prostate Cancer Cases Is Altered Relative to Healthy Controls—Results of a Pilot Study in South Australia. Cancers, 15(1), 77. https://doi.org/10.3390/cancers15010077






