In Vitro Diffuse Large B-Cell Lymphoma Cell Line Models as Tools to Investigate Novel Immunotherapeutic Strategies
Simple Summary
Abstract
1. Introduction
2. Established Cell Lines 2-Dimenstional (2D) Models as Tools to Study DLBCL Biology
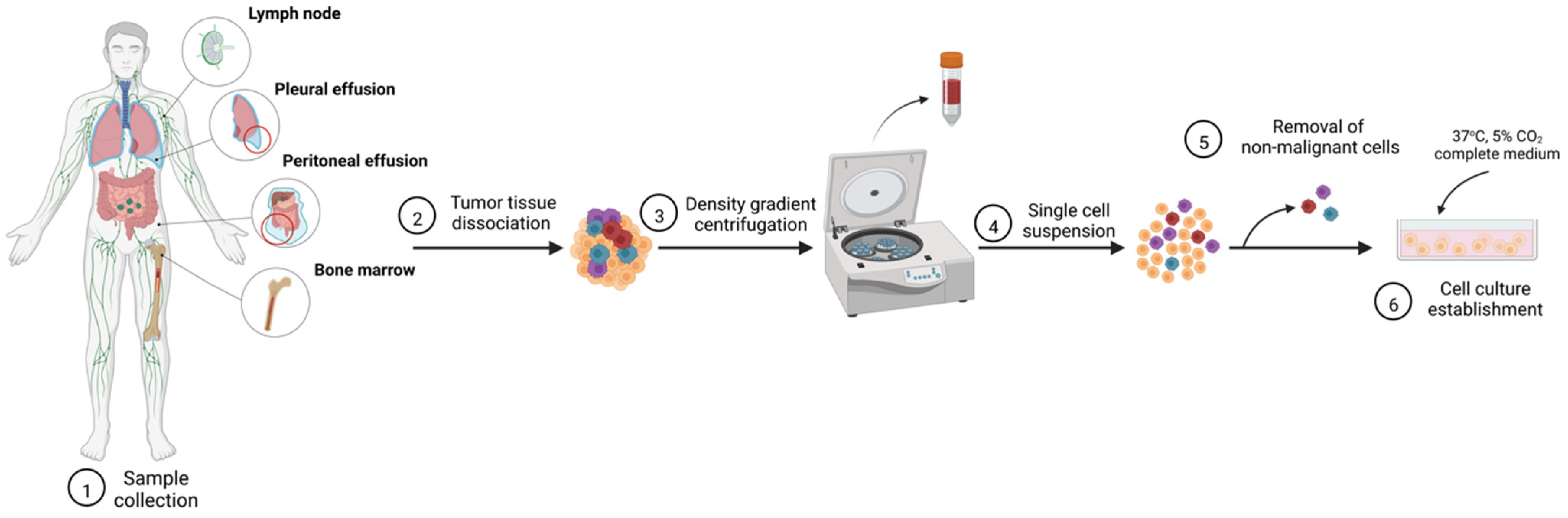
2.1. Cell Line Establishement from Primary Cell Culture
2.2. Challenges in Establishment of Cell Lines Faithfully Recapitulating DLBCL
2.3. DLBCL Cell Line Models Available to Study Biology of the Aggressive B-Cell Lymphoma
2.3.1. Panel of ABC-DLBCL Models
2.3.2. Panel of GCB-DLBCL Models
2.3.3. Cell Line Models with Unique Features Representative of RS-DLBCL and Secondary DLBCL
3. 3-Dimensional (3D) Models
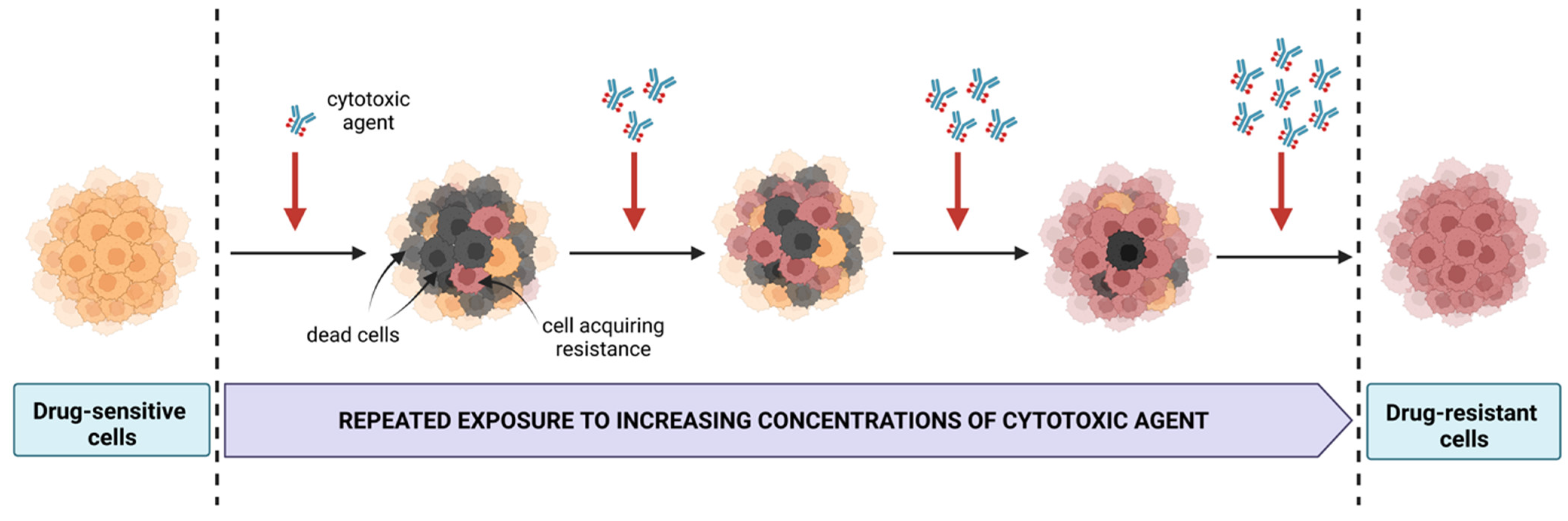
4. Significance of Cell Line Models Resistant to Therapeutic Agents
5. Investigating Immunotherapeutic Strategies
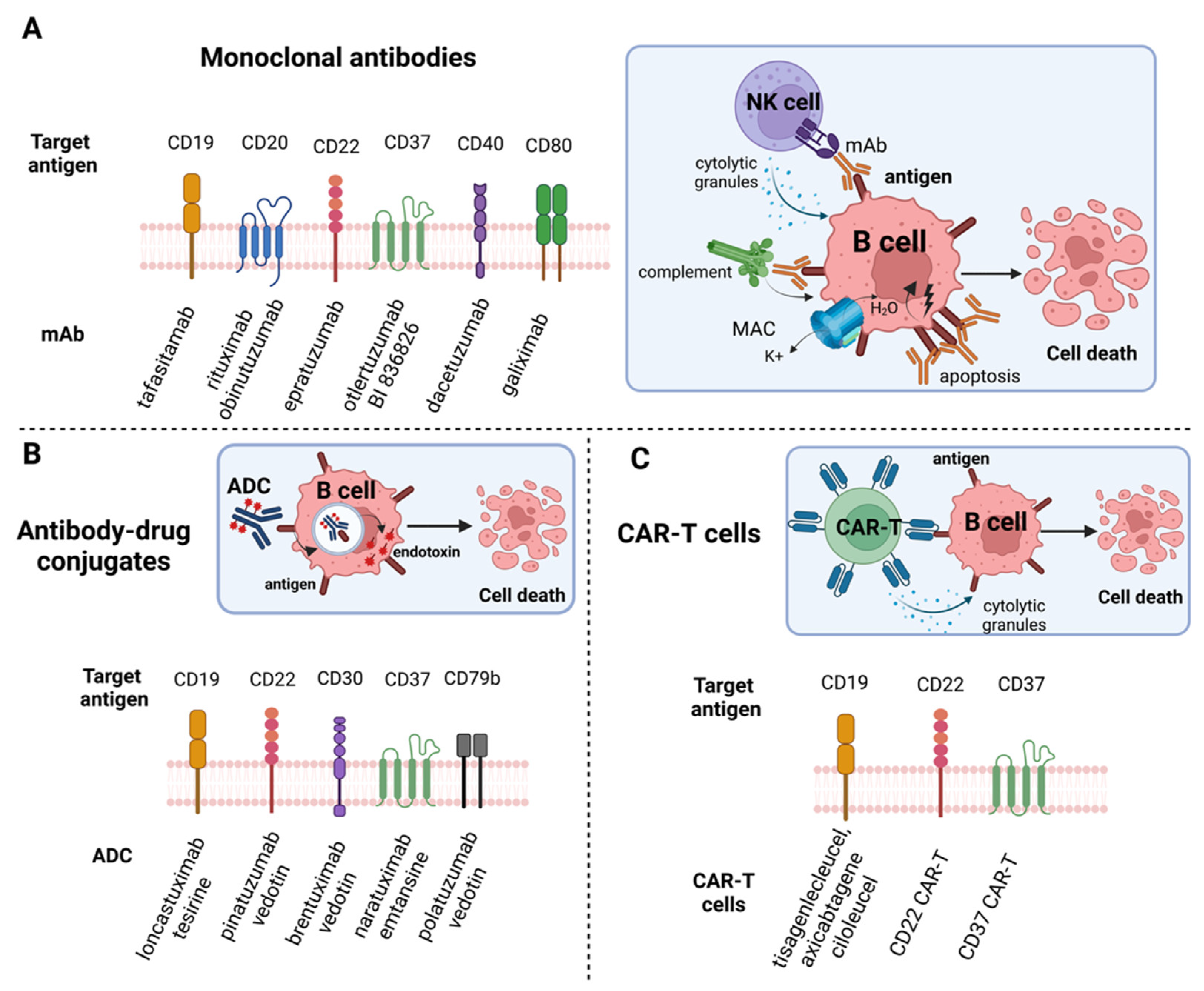
5.1. Exploring Potential of Monoclonal Antibodies (mAbs)
5.2. Antibody-Drug Conjugates (ADCs) and Targeted-Drug Delivery
5.3. CAR-T Cell Therapy
6. Xenograft Mouse Models
7. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Siegel, R.; Naishadham, D.; Jemal, A. Cancer statistics, 2012. CA Cancer J. Clin. 2012, 62, 10–29. [Google Scholar] [CrossRef] [PubMed]
- Rovira, J.; Valera, A.; Colomo, L.; Setoain, X.; Rodríguez, S.; Martínez-Trillos, A.; Giné, E.; Dlouhy, I.; Magnano, L.; Gaya, A.; et al. Prognosis of patients with diffuse large B cell lymphoma not reaching complete response or relapsing after frontline chemotherapy or immunochemotherapy. Ann. Hematol. 2015, 94, 803–812. [Google Scholar] [CrossRef]
- Liu, Y.; Barta, S.K. Diffuse large B-cell lymphoma: 2019 update on diagnosis, risk stratification, and treatment. Am. J. Hematol. 2019, 94, 604–616. [Google Scholar] [CrossRef]
- Zou, L.; Song, G.; Gu, S.; Kong, L.; Sun, S.; Yang, L.; Cho, W.C. Mechanism and Treatment of Rituximab Resistance in Diffuse Large Bcell Lymphoma. Curr. Cancer Drug Targets 2019, 19, 681–687. [Google Scholar] [CrossRef]
- Alizadeh, A.A.; Eisen, M.B.; Davis, R.E.; Ma, C.; Lossos, I.S.; Rosenwald, A.; Boldrick, J.C.; Sabet, H.; Tran, T.; Yu, X.; et al. Distinct types of diffuse large B-cell lymphoma identified by gene expression profiling. Nature 2000, 403, 503–511. [Google Scholar] [CrossRef] [PubMed]
- Susanibar-Adaniya, S.; Barta, S.K. 2021 Update on Diffuse large B cell lymphoma: A review of current data and potential applications on risk stratification and management. Am. J. Hematol. 2021, 96, 617–629. [Google Scholar] [CrossRef] [PubMed]
- Riedell, P.A.; Smith, S.M. Double hit and double expressors in lymphoma: Definition and treatment. Cancer 2018, 124, 4622–4632. [Google Scholar] [CrossRef] [PubMed]
- Rosenthal, A.; Younes, A. High grade B-cell lymphoma with rearrangements of MYC and BCL2 and/or BCL6: Double hit and triple hit lymphomas and double expressing lymphoma. Blood Rev. 2017, 31, 37–42. [Google Scholar] [CrossRef]
- Rossi, D.; Spina, V.; Deambrogi, C.; Rasi, S.; Laurenti, L.; Stamatopoulos, K.; Arcaini, L.; Lucioni, M.; Rocque, G.B.; Xu-Monette, Z.Y.; et al. The genetics of Richter syndrome reveals disease heterogeneity and predicts survival after transformation. Blood 2011, 117, 3391–3401. [Google Scholar] [CrossRef] [PubMed]
- Parikh, S.A.; Shanafelt, T.D. Risk Factors for Richter Syndrome in Chronic Lymphocytic Leukemia. Curr. Hematol. Malig. Rep. 2014, 9, 294–299. [Google Scholar] [CrossRef]
- Bockorny, B.; Codreanu, I.; Dasanu, C.A. Hodgkin lymphoma as Richter transformation in chronic lymphocytic leukaemia: A retrospective analysis of world literature. Br. J. Haematol. 2012, 156, 50–66. [Google Scholar] [CrossRef] [PubMed]
- Yanguas-Casás, N.; Pedrosa, L.; Fernández-Miranda, I.; Sánchez-Beato, M. An Overview on Diffuse Large B-Cell Lymphoma Models: Towards a Functional Genomics Approach. Cancers 2021, 13, 2893. [Google Scholar] [CrossRef] [PubMed]
- Singh, J.; Goswami, A. Applications of cell lines as bioreactors and in vitro models. Int. J. Appl. Biol. Pharm. Technol. 2012, 2, 178–198. [Google Scholar]
- Nowakowski, G.S. Recently approved drugs herald a new era in therapy for diffuse large B-cell lymphoma. Clin. Adv. Hematol. Oncol. 2021, 19, 284–287. [Google Scholar]
- Pulvertaft, R.J.V. Cytology of Burkitt’s Tumour (African Lymphoma). Lancet 1964, 283, 238–240. [Google Scholar] [CrossRef]
- Drexler, H.G.; Matsuo, Y.; MacLeod, R.A.F. Continuous hematopoietic cell lines as model systems for leukemia–lymphoma research. Leuk. Res. 2000, 24, 881–911. [Google Scholar] [CrossRef]
- Matsuo, Y.; Drexler, H.G. Establishment and characterization of human B cell precursor-leukemia cell lines. Leuk. Res. 1998, 22, 567–579. [Google Scholar] [CrossRef]
- Drexler, H.G. Establishment and culture of leukemia-lymphoma cell lines. Methods Mol. Biol. 2011, 731, 181–200. [Google Scholar] [CrossRef]
- Maqsood, M.I.; Matin, M.M.; Bahrami, A.R.; Ghasroldasht, M.M. Immortality of cell lines: Challenges and advantages of establishment. Cell Biol. Int. 2013, 37, 1038–1045. [Google Scholar] [CrossRef]
- Matsuo, Y.; Minowada, J. Human leukemia cell lines--clinical and theoretical significances. Hum. Cell 1988, 1, 263–274. [Google Scholar]
- Drexler, H.G.; Dirks, W.G.; MacLeod, R.A. False human hematopoietic cell lines: Cross-contaminations and misinterpretations. Leukemia 1999, 13, 1601–1607. [Google Scholar] [CrossRef] [PubMed]
- Devin, J.; Kassambara, A.; Bruyer, A.; Moreaux, J.; Bret, C. Phenotypic Characterization of Diffuse Large B-Cell Lymphoma Cells and Prognostic Impact. J. Clin. Med. 2019, 8, 1074. [Google Scholar] [CrossRef] [PubMed]
- Drexler, H.G.; Quentmeier, H. The LL-100 Cell Lines Panel: Tool for Molecular Leukemia-Lymphoma Research. Int. J. Mol. Sci. 2020, 21, 5800. [Google Scholar] [CrossRef] [PubMed]
- Caeser, R.; Di Re, M.; Krupka, J.A.; Gao, J.; Lara-Chica, M.; Dias, J.M.L.; Cooke, S.L.; Fenner, R.; Usheva, Z.; Runge, H.F.P.; et al. Genetic modification of primary human B cells to model high-grade lymphoma. Nat. Commun. 2019, 10, 4543. [Google Scholar] [CrossRef]
- Caeser, R.; Gao, J.; Di Re, M.; Gong, C.; Hodson, D.J. Genetic manipulation and immortalized culture of ex vivo primary human germinal center B cells. Nat. Protoc. 2021, 16, 2499–2519. [Google Scholar] [CrossRef]
- Carbone, A.; Gloghini, A.; Aiello, A.; Testi, A.; Cabras, A. B-cell lymphomas with features intermediate between distinct pathologic entities. From pathogenesis to pathology. Hum. Pathol. 2010, 41, 621–631. [Google Scholar] [CrossRef]
- Matsumoto, Y.; Tsukamoto, T.; Chinen, Y.; Shimura, Y.; Sasaki, N.; Nagoshi, H.; Sato, R.; Adachi, H.; Nakano, M.; Horiike, S.; et al. Detection of novel and recurrent conjoined genes in non-Hodgkin B-cell lymphoma. J. Clin. Exp. Hematop. 2021, 61, 71–77. [Google Scholar] [CrossRef]
- The LL-100 Panel: 100 Cell Lines for Blood Cancer Studies. Available online: https://pubmed.ncbi.nlm.nih.gov/31160637/ (accessed on 26 October 2022).
- Dozzo, M.; Carobolante, F.; Donisi, P.M.; Scattolin, A.; Maino, E.; Sancetta, R.; Viero, P.; Bassan, R. Burkitt lymphoma in adolescents and young adults: Management challenges. Adolesc. Health Med. Ther. 2016, 8, 11–29. [Google Scholar] [CrossRef]
- Epstein, A.L.; Kaplan, H.S. Biology of the human malignant lymphomas. Recent Results Cancer Res. 1978, 64, 190–200. [Google Scholar] [CrossRef]
- Tweeddale, M.; Jamal, N.; Nguyen, A.; Wang, X.H.; Minden, M.D.; Messner, H.A. Production of growth factors by malignant lymphoma cell lines. Blood 1989, 74, 572–578. [Google Scholar] [CrossRef]
- Chang, H.; Messner, H.A.; Wang, X.H.; Yee, C.; Addy, L.; Meharchand, J.; Minden, M.D. A human lymphoma cell line with multiple immunoglobulin rearrangements. J. Clin. Investig. 1992, 89, 1014–1020. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Yee, C.S.; Messner, H.A.; Minden, M.D. Regulation of interleukin-6 expression in the lymphoma cell line OCI-LY3. J. Cell Physiol. 1991, 148, 426–429. [Google Scholar] [CrossRef]
- Nowakowski, G.S.; Czuczman, M.S. ABC, GCB, and Double-Hit Diffuse Large B-Cell Lymphoma: Does Subtype Make a Difference in Therapy Selection? Am. Soc. Clin. Oncol. Educ. Book 2015, 35, e449–e457. [Google Scholar] [CrossRef] [PubMed]
- Kalaitzidis, D.; Davis, R.E.; Rosenwald, A.; Staudt, L.M.; Gilmore, T.D. The human B-cell lymphoma cell line RC-K8 has multiple genetic alterations that dysregulate the Rel/NF-κB signal transduction pathway. Oncogene 2002, 21, 8759–8768. [Google Scholar] [CrossRef] [PubMed]
- Quentmeier, H.; Drexler, H.G.; Hauer, V.; MacLeod, R.A.F.; Pommerenke, C.; Uphoff, C.C.; Zaborski, M.; Berglund, M.; Enblad, G.; Amini, R.-M. Diffuse Large B Cell Lymphoma Cell Line U-2946: Model for MCL1 Inhibitor Testing. PLoS ONE 2016, 11, e0167599. [Google Scholar] [CrossRef]
- Bujisic, B.; De Gassart, A.; Tallant, R.; DeMaria, O.; Zaffalon, L.; Chelbi, S.; Gilliet, M.; Bertoni, F.; Martinon, F. Impairment of both IRE1 expression and XBP1 activation is a hallmark of GCB DLBCL and contributes to tumor growth. Blood 2017, 129, 2420–2428. [Google Scholar] [CrossRef] [PubMed]
- Dyer, M.J.; Fischer, P.; Nacheva, E.; Labastide, W.; Karpas, A. A new human B-cell non-Hodgkin’s lymphoma cell line (Karpas 422) exhibiting both t (14;18) and t(4;11) chromosomal translocations. Blood 1990, 75, 709–714. [Google Scholar] [CrossRef]
- Bakhshi, T.J.; Georgel, P.T. Genetic and epigenetic determinants of diffuse large B-cell lymphoma. Blood Cancer J. 2020, 10, 123. [Google Scholar] [CrossRef]
- Barrans, S.L.; Evans, P.A.S.; O’Connor, S.J.M.; Kendall, S.J.; Owen, R.G.; Haynes, A.P.; Morgan, G.J.; Jack, A.S. The t(14;18) is associated with germinal center-derived diffuse large B-cell lymphoma and is a strong predictor of outcome. Clin. Cancer Res. 2003, 9, 2133–2139. [Google Scholar]
- Fraser, C.R.; Wang, W.; Gomez, M.; Zhang, T.; Mathew, S.; Furman, R.R.; Knowles, D.M.; Orazi, A.; Tam, W. Transformation of Chronic Lymphocytic Leukemia/Small Lymphocytic Lymphoma to Interdigitating Dendritic Cell Sarcoma: Evidence for Transdifferentiation of the Lymphoma Clone. Am. J. Clin. Pathol. 2009, 132, 928–939. [Google Scholar] [CrossRef]
- Shao, H.; Xi, L.; Raffeld, M.; Feldman, A.L.; Ketterling, R.P.; Knudson, R.; Rodriguez-Canales, J.; Hanson, J.; Pittaluga, S.; Jaffe, E.S. Clonally related histiocytic/dendritic cell sarcoma and chronic lymphocytic leukemia/small lymphocytic lymphoma: A study of seven cases. Mod. Pathol. 2011, 24, 1421–1432. [Google Scholar] [CrossRef] [PubMed]
- Petrackova, A.; Turcsanyi, P.; Papajik, T.; Kriegova, E. Revisiting Richter transformation in the era of novel CLL agents. Blood Rev. 2021, 49, 100824. [Google Scholar] [CrossRef] [PubMed]
- Augé, H.; Notarantonio, A.-B.; Morizot, R.; Quinquenel, A.; Fornecker, L.-M.; Hergalant, S.; Feugier, P.; Broséus, J. Microenvironment Remodeling and Subsequent Clinical Implications in Diffuse Large B-Cell Histologic Variant of Richter Syndrome. Front. Immunol. 2020, 11, 594841. [Google Scholar] [CrossRef] [PubMed]
- Iannello, A.; Deaglio, S.; Vaisitti, T. Novel Approaches for the Treatment of Patients with Richter’s Syndrome. Curr. Treat. Options Oncol. 2022, 23, 526–542. [Google Scholar] [CrossRef] [PubMed]
- Matasar, M.J.; Zelenetz, A.D. Overview of lymphoma diagnosis and management. Radiol. Clin. N. Am. 2008, 46, 175–198. [Google Scholar] [CrossRef] [PubMed]
- Cheson, B.D.; Fisher, R.I.; Barrington, S.F.; Cavalli, F.; Schwartz, L.H.; Zucca, E.; Lister, T.A. Recommendations for initial evaluation, staging, and response assessment of Hodgkin and non-Hodgkin lymphoma: The Lugano classification. J. Clin. Oncol. 2014, 32, 3059–3068. [Google Scholar] [CrossRef] [PubMed]
- Borchmann, P.; Behringer, K.; Josting, A.; Rueffer, J.U.; Schnell, R.; Diehl, V.; Engert, A.; Kvasnicka, H.M.; Thiele, J. Secondary malignancies after successful primary treatment of malignant Hodgkin’s lymphoma. Pathologe 2006, 27, 47–52. [Google Scholar] [CrossRef]
- Cappelaere, P. Secondary non-Hodgkin’s lymphomas. Bull. Cancer 1998, 85, 217–231. [Google Scholar]
- Amini, R.-M.; Berglund, M.; Rosenquist, R.; Von Heideman, A.; Lagercrantz, S.; Thunberg, U.; Bergh, J.; Sundström, C.; Glimelius, B.; Enblad, G. A novel B-cell line (U-2932) established from a patient with diffuse large B-cell lymphoma following Hodgkin lymphoma. Leuk. Lymphoma 2002, 43, 2179–2189. [Google Scholar] [CrossRef]
- Quentmeier, H.; Amini, R.M.; Berglund, M.; Dirks, W.; Ehrentraut, S.; Geffers, R.; MacLeod, R.A.F.; Nagel, S.; Romani, J.; Scherr, M.; et al. U-2932: Two clones in one cell line, a tool for the study of clonal evolution. Leukemia 2013, 27, 1155–1164. [Google Scholar] [CrossRef]
- Pinheiro, A.M. Characterization of U2932 Cell Line Subpopulations and Evaluation of Their Sensibility to a Chemotherapeutic Drug. Master’s Thesis, Aalborg University Hospital, Aalborg, Denmark, 2018. Available online: https://projekter.aau.dk/projekter/en/studentthesis/characterization-of-u2932-cell-line-subpopulations-and-evaluation-of-their-sensibility-to-a-chemotherapeutic-drug(f1b1e5d4-71ce-45a6-a961-17777268abba).html (accessed on 30 October 2022).
- Sambade, C.; Berglund, M.; Lagercrantz, S.; Sällström, J.; Reis, R.M.; Enblad, G.; Glimelius, B.; Sundström, C. U-2940, a human B-cell line derived from a diffuse large cell lymphoma sequential to Hodgkin lymphoma. Int. J. Cancer 2006, 118, 555–563. [Google Scholar] [CrossRef] [PubMed]
- Dai, H.; Ehrentraut, S.; Nagel, S.; Eberth, S.; Pommerenke, C.; Dirks, W.G.; Geffers, R.; Kalavalapalli, S.; Kaufmann, M.; Meyer, C.; et al. Genomic Landscape of Primary Mediastinal B-Cell Lymphoma Cell Lines. PLoS ONE 2015, 10, e0139663. [Google Scholar] [CrossRef] [PubMed]
- Drexler, H.G.; Ehrentraut, S.; Nagel, S.; Eberth, S.; MacLeod, R.A.F. Malignant hematopoietic cell lines: In vitro models for the study of primary mediastinal B-cell lymphomas. Leuk. Res. 2015, 39, 18–29. [Google Scholar] [CrossRef]
- Abe, M.; Nozawa, Y.; Wakasa, H.; Ohno, H.; Fukuhara, S. Characterization and comparison of two newly established Epstein-Barr virus-negative lymphoma B-cell lines. Surface markers, growth characteristics, cytogenetics, and transplantability. Cancer 1988, 61, 483–490. [Google Scholar] [CrossRef] [PubMed]
- Berglund, M.; Thunberg, U.; Fridberg, M.; Wingren, A.G.; Gullbo, J.; Leuchowius, K.-J.; Amini, R.-M.; Lagercrantz, S.; Horvat, A.; Enblad, G.; et al. Establishment of a cell line from a chemotherapy resistant diffuse large B-cell lymphoma. Leuk. Lymphoma 2007, 48, 1038–1041. [Google Scholar] [CrossRef]
- Tohda, S.; Sato, T.; Kogoshi, H.; Fu, L.; Sakano, S.; Nara, N. Establishment of a novel B-cell lymphoma cell line with suppressed growth by gamma-secretase inhibitors. Leuk. Res. 2006, 30, 1385–1390. [Google Scholar] [CrossRef] [PubMed]
- Kubonishi, I.; Niiya, K.; Yamashita, M.; Yano, S.; Abe, T.; Ohtsuki, Y.; Miyoshi, I. Characterization of a new human lymphoma cell line (RC-K8) with t(11;14) chromosome abnormality. Cancer 1986, 58, 1453–1460. [Google Scholar] [CrossRef]
- Goy, A.; Ramdas, L.; Remache, Y.K.; Gu, J.; Fayad, L.; Hayes, K.J.; Coombes, K.; Barkoh, B.A.; Katz, R.; Ford, R.; et al. Establishment and characterization by gene expression profiling of a new diffuse large B-cell lymphoma cell line, EJ-1, carrying t(14;18) and t(8;14) translocations. Lab. Investig. 2003, 83, 913–916. [Google Scholar] [CrossRef][Green Version]
- Pham, L.V.; Lu, G.; Tamayo, A.T.; Chen, J.; Challagundla, P.; Jorgensen, J.L.; Medeiros, L.J.; Ford, R.J. Establishment and characterization of a novel MYC/BCL2 «double-hit» diffuse large B cell lymphoma cell line, RC. J. Hematol. Oncol. 2015, 8, 121. [Google Scholar] [CrossRef]
- Boström, H.; Leuchowius, K.-J.; Hallböök, H.; Nordgren, A.; Thörn, I.; Thorselius, M.; Rosenquist, R.; Söderberg, O.; Sundström, C. U-2973, a novel B-cell line established from a patient with a mature B-cell leukemia displaying concurrent t(14;18) and MYC translocation to a non-IG gene partner. Eur. J. Haematol. 2008, 81, 218–225. [Google Scholar] [CrossRef]
- Schmid, T.; Maier, J.; Martin, M.; Tasdogan, A.; Tausch, E.; Barth, T.F.; Stilgenbauer, S.; Bloehdorn, J.; Möller, P.; Mellert, K. U-RT1—A new model for Richter transformation. Neoplasia 2021, 23, 140–148. [Google Scholar] [CrossRef] [PubMed]
- Nichele, I.; Zamò, A.; Bertolaso, A.; Bifari, F.; Tinelli, M.; Franchini, M.; Stradoni, R.; Aprili, F.; Pizzolo, G.; Krampera, M. VR09 Cell Line: An EBV-Positive Lymphoblastoid Cell Line with In Vivo Characteristics of Diffuse Large B Cell Lymphoma of Activated B-Cell Type. PLoS ONE 2012, 7, e52811. [Google Scholar] [CrossRef] [PubMed]
- Zanoni, M.; Cortesi, M.; Zamagni, A.; Arienti, C.; Pignatta, S.; Tesei, A. Modeling neoplastic disease with spheroids and organoids. J. Hematol. Oncol. 2020, 13, 97. [Google Scholar] [CrossRef] [PubMed]
- Ravi, M.; Paramesh, V.; Kaviya, S.R.; Anuradha, E.; Solomon, F.D.P. 3D cell culture systems: Advantages and applications. J. Cell Physiol. 2015, 230, 16–26. [Google Scholar] [CrossRef] [PubMed]
- Langhans, S.A. Three-Dimensional in Vitro Cell Culture Models in Drug Discovery and Drug Repositioning. Front. Pharmacol. 2018, 9, 6. [Google Scholar] [CrossRef]
- Duś-Szachniewicz, K.; Gdesz-Birula, K.; Rymkiewicz, G. Development and Characterization of 3D Hybrid Spheroids for the Investigation of the Crosstalk Between B-Cell Non-Hodgkin Lymphomas and Mesenchymal Stromal Cells. Oncol. Targets Ther. 2022, 15, 683–697. [Google Scholar] [CrossRef]
- Lara, S.; Heilig, J.; Virtanen, A.; Kleinau, S. Exploring complement-dependent cytotoxicity by rituximab isotypes in 2D and 3D-cultured B-cell lymphoma. BMC Cancer 2022, 22, 678. [Google Scholar] [CrossRef]
- Kusowska, A.; Kubacz, M.; Krawczyk, M.; Slusarczyk, A.; Winiarska, M.; Bobrowicz, M. Molecular Aspects of Resistance to Immunotherapies-Advances in Understanding and Management of Diffuse Large B-Cell Lymphoma. Int. J. Mol. Sci. 2022, 23, 1501. [Google Scholar] [CrossRef]
- McDermott, M.; Eustace, A.J.; Busschots, S.; Breen, L.; Crown, J.; Clynes, M.; O’Donovan, N.; Stordal, B.; O’Donovan, N. In vitro Development of Chemotherapy and Targeted Therapy Drug-Resistant Cancer Cell Lines: A Practical Guide with Case Studies. Front. Oncol. 2022, 4, 2014. [Google Scholar] [CrossRef]
- Czuczman, M.S.; Olejniczak, S.; Gowda, A.; Kotowski, A.; Binder, A.; Kaur, H.; Knight, J.; Starostik, P.; Deans, J.; Hernandez-Ilizaliturri, F.J. Acquirement of rituximab resistance in lymphoma cell lines is associated with both global CD20 gene and protein down-regulation regulated at the pretranscriptional and posttranscriptional levels. Clin. Cancer Res. 2008, 14, 1561–1570. [Google Scholar] [CrossRef]
- Olejniczak, S.H.; Hernandez-Ilizaliturri, F.J.; Clements, J.L.; Czuczman, M.S. Acquired resistance to rituximab is associated with chemotherapy resistance resulting from decreased Bax and Bak expression. Clin. Cancer Res. 2008, 14, 1550–1560. [Google Scholar] [CrossRef] [PubMed]
- Arribas, A.J.; Cascione, L.; Aresu, L.; Gaudio, E.; Rinaldi, A.; Tarantelli, C.; Akhmedov, M.; Zucca, E.; Rossi, D.; Stathis, A.; et al. Abstract 2853: Development of novel preclinical models of secondary resistance to the anti-CD37 antibody drug conjugate (ADC) IMGN529/DEBIO1562 in diffuse large B-cell lymphoma (DLBCL). Cancer Res. 2018, 78, 2853. [Google Scholar] [CrossRef]
- Hicks, S.W.; Lai, K.C.; Gavrilescu, L.C.; Yi, Y.; Sikka, S.; Shah, P.; Kelly, M.E.; Lee, J.; Lanieri, L.; Ponte, J.F.; et al. The Antitumor Activity of IMGN529, a CD37-Targeting Antibody-Drug Conjugate, Is Potentiated by Rituximab in Non-Hodgkin Lymphoma Models. Neoplasia 2017, 19, 661–671. [Google Scholar] [CrossRef] [PubMed]
- Miazek-Zapala, N.; Slusarczyk, A.; Kusowska, A.; Zapala, P.; Kubacz, M.; Winiarska, M.; Bobrowicz, M. The «Magic Bullet» Is Here? Cell-Based Immunotherapies for Hematological Malignancies in the Twilight of the Chemotherapy Era. Cells 2021, 10, 1511. [Google Scholar] [CrossRef] [PubMed]
- Gehlert, C.L.; Rahmati, P.; Boje, A.S.; Winterberg, D.; Krohn, S.; Theocharis, T.; Cappuzzello, E.; Lux, A.; Nimmerjahn, F.; Ludwig, R.J.; et al. Dual Fc optimization to increase the cytotoxic activity of a CD19-targeting antibody. Front. Immunol. 2022, 13, 957874. [Google Scholar] [CrossRef]
- Lu, Q.; Huang, H.; Tang, S.; Wang, Y.; Yang, D.-H. Tafasitamab for refractory/relapsed diffuse large B-cell lymphoma. Drugs Today 2021, 57, 571–580. [Google Scholar] [CrossRef]
- Xu-Monette, Z.Y.; Li, L.; Byrd, J.C.; Jabbar, K.J.; Manyam, G.C.; de Winde, C.M.; Brand, M.V.D.; Tzankov, A.; Visco, C.; Wang, J.; et al. Assessment of CD37 B-cell antigen and cell of origin significantly improves risk prediction in diffuse large B-cell lymphoma. Blood 2016, 128, 3083–3100. [Google Scholar] [CrossRef]
- Bobrowicz, M.; Kubacz, M.; Slusarczyk, A.; Winiarska, M. CD37 in B Cell Derived Tumors—More than Just a Docking Point for Monoclonal Antibodies. Int. J. Mol. Sci. 2020, 21, 9531. [Google Scholar] [CrossRef]
- Oostindie, S.C.; Van Der Horst, H.J.; Kil, L.P.; Strumane, K.; Overdijk, M.B.; van den Brink, E.N.; van den Brakel, J.H.N.; Rademaker, H.J.; Van Kessel, B.; van den Noort, J.; et al. DuoHexaBody-CD37®, a novel biparatopic CD37 antibody with enhanced Fc-mediated hexamerization as a potential therapy for B-cell malignancies. Blood Cancer J. 2020, 10, 30. [Google Scholar] [CrossRef]
- Wada, F.; Shimomura, Y.; Yabushita, T.; Yamashita, D.; Ohno, A.; Imoto, H.; Maruoka, H.; Hara, S.; Ishikawa, T. CD38 expression is an important prognostic marker in diffuse large B-cell lymphoma. Hematol. Oncol. 2021, 39, 483–489. [Google Scholar] [CrossRef]
- Vidal-Crespo, A.; Matas-Céspedes, A.; Rodriguez, V.; Rossi, C.; Valero, J.G.; Serrat, N.; Sanjuan-Pla, A.; Menéndez, P.; Roué, G.; López-Guillermo, A.; et al. Daratumumab displays in vitro and in vivo anti-tumor activity in models of B-cell non-Hodgkin lymphoma and improves responses to standard chemo-immunotherapy regimens. Haematologica 2020, 105, 1032–1041. [Google Scholar] [CrossRef]
- Bouwstra, R.; He, Y.; de Boer, J.; Kooistra, H.; Cendrowicz, E.; Fehrmann, R.S.; Ammatuna, E.; zu Eulenburg, C.; Nijland, M.; Huls, G.; et al. CD47 Expression Defines Efficacy of Rituximab with CHOP in Non-Germinal Center B-cell (Non-GCB) Diffuse Large B-cell Lymphoma Patients (DLBCL), but Not in GCB DLBCL. Cancer Immunol. Res. 2019, 7, 1663–1671. [Google Scholar] [CrossRef]
- GPR183 Mediates the Capacity of the Novel CD47-CD19 Bispecific Antibody TG-1801 to Heighten Ublituximab-Umbralisib (U2) Anti-Lymphoma Activity. Available online: https://www.biorxiv.org/content/10.1101/2022.03.31.486558v1 (accessed on 16 November 2022).
- Kline, J.; Bishop, M.R. Update on checkpoint blockade therapy for lymphoma. J. Immunother. Cancer 2015, 3, 33. [Google Scholar] [CrossRef][Green Version]
- Andorsky, D.J.; Yamada, R.E.; Said, J.; Pinkus, G.S.; Betting, D.J.; Timmerman, J.M. Programmed death ligand 1 is expressed by non-hodgkin lymphomas and inhibits the activity of tumor-associated T cells. Clin. Cancer Res. 2011, 17, 4232–4244. [Google Scholar] [CrossRef]
- Wei, T.; Li, M.; Zhu, Z.; Xiong, H.; Shen, H.; Zhang, H.; Du, Q.; Li, Q. Vincristine upregulates PD-L1 and increases the efficacy of PD-L1 blockade therapy in diffuse large B-cell lymphoma. J. Cancer Res. Clin. Oncol. 2021, 147, 691–701. [Google Scholar] [CrossRef]
- Ansell, S.M.; Lesokhin, A.M.; Borrello, I.; Halwani, A.; Scott, E.C.; Gutierrez, M.; Schuster, S.J.; Millenson, M.M.; Cattry, D.; Freeman, G.J.; et al. PD-1 blockade with nivolumab in relapsed or refractory Hodgkin’s lymphoma. N. Engl. J. Med. 2015, 372, 311–319. [Google Scholar] [CrossRef]
- Smith, S.D.; Till, B.G.; Shadman, M.S.; Lynch, R.C.; Cowan, A.J.; Wu, Q.V.; Voutsinas, J.; Rasmussen, H.A.; Blue, K.; Ujjani, C.S.; et al. Pembrolizumab with R-CHOP in previously untreated diffuse large B-cell lymphoma: Potential for biomarker driven therapy. Br. J. Haematol. 2020, 189, 1119–1126. [Google Scholar] [CrossRef]
- Hafeez, U.; Parakh, S.; Gan, H.K.; Scott, A.M. Antibody-Drug Conjugates for Cancer Therapy. Molecules 2020, 25, 4764. [Google Scholar] [CrossRef]
- Tsuchikama, K.; An, Z. Antibody-drug conjugates: Recent advances in conjugation and linker chemistries. Protein Cell 2018, 9, 33–46. [Google Scholar] [CrossRef]
- Dimou, M.; Papageorgiou, S.G.; Stavroyianni, N.; Katodritou, E.; Tsirogianni, M.; Kalpadakis, C.; Banti, A.; Arapaki, M.; Iliakis, T.; Bouzani, M.; et al. Real-life experience with the combination of polatuzumab vedotin, rituximab, and bendamustine in aggressive B-cell lymphomas. Hematol. Oncol. 2021, 39, 336–348. [Google Scholar] [CrossRef]
- Kawasaki, N.; Nishito, Y.; Yoshimura, Y.; Yoshiura, S. The molecular rationale for the combination of polatuzumab vedotin plus rituximab in diffuse large B-cell lymphoma. Br. J. Haematol. 2022, 199, 245–255. [Google Scholar] [CrossRef] [PubMed]
- Tarantelli, C.; Bertoni, F. United we stand: Double targeting of CD79B and CD20 in diffuse large B-cell lymphoma. Br. J. Haematol. 2022, 199, 169–170. [Google Scholar] [CrossRef] [PubMed]
- Pfeifer, M.P.; Zheng, B.; Erdmann, T.; Koeppen, H.; Mccord, R.C.; Grau, M.; Staiger, A.M.; Chai, A.; Sandmann, T.; Madle, H.; et al. Anti-CD22 and anti-CD79B antibody drug conjugates are active in different molecular diffuse large B-cell lymphoma subtypes. Leukemia 2015, 29, 1578–1586. [Google Scholar] [CrossRef] [PubMed]
- Deckert, J.; Park, P.U.; Chicklas, S.; Yi, Y.; Li, M.; Lai, K.C.; Mayo, M.F.; Carrigan, C.N.; Erickson, H.K.; Pinkas, J.; et al. A novel anti-CD37 antibody-drug conjugate with multiple anti-tumor mechanisms for the treatment of B-cell malignancies. Blood 2013, 122, 3500–3510. [Google Scholar] [CrossRef] [PubMed]
- Vaisitti, T.; Vitale, N.; Micillo, M.; Brandimarte, L.; Iannello, A.; Papotti, M.G.; Jaksic, O.; Lopez, G.; Di Napoli, A.; Cutrin, J.C.; et al. Anti-CD37 α-amanitin-conjugated antibodies as potential therapeutic weapons for Richter syndrome. Blood 2022, 140, 1565–1569. [Google Scholar] [CrossRef] [PubMed]
- Zhylko, A.; Winiarska, M.; Graczyk-Jarzynka, A. The Great War of Today: Modifications of CAR-T Cells to Effectively Combat Malignancies. Cancers 2020, 12, 2030. [Google Scholar] [CrossRef]
- Young, P.A.; Yamada, R.E.; Trinh, K.R.; Vasuthasawat, A.; De Oliveira, S.; Yamada, D.H.; Morrison, S.L.; Timmerman, J.M. Activity of Anti-CD19 Chimeric Antigen Receptor T Cells Against B Cell Lymphoma Is Enhanced by Antibody-Targeted Interferon-Alpha. J. Interferon Cytokine Res. 2018, 38, 239–254. [Google Scholar] [CrossRef]
- Liu, M.; Wang, X.; Li, Z.; Zhang, R.; Mu, J.; Jiang, Y.; Deng, Q.; Sun, L. Synergistic effect of ibrutinib and CD19 CAR-T cells on Raji cells in vivo and in vitro. Cancer Sci. 2020, 111, 4051–4060. [Google Scholar] [CrossRef]
- Köksal, H.; Dillard, P.; Josefsson, S.E.; Maggadottir, S.M.; Pollmann, S.; Fåne, A.; Blaker, Y.N.; Beiske, K.; Huse, K.; Kolstad, A.; et al. Preclinical development of CD37CAR T-cell therapy for treatment of B-cell lymphoma. Blood Adv. 2019, 3, 1230–1243. [Google Scholar] [CrossRef]
- Maloney, D.G. Anti-CD19 CAR T cell therapy for lymphoma-off to the races! Nat. Rev. Clin. Oncol. 2019, 16, 279–280. [Google Scholar] [CrossRef]
- Kohnken, R.; Porcu, P.; Mishra, A. Overview of the Use of Murine Models in Leukemia and Lymphoma Research. Front. Oncol. 2017, 7, 22. [Google Scholar] [CrossRef] [PubMed]
- Köberle, M.; Müller, K.; Kamprad, M.; Horn, F.; Scholz, M. Monitoring Disease Progression and Therapeutic Response in a Disseminated Tumor Model for Non-Hodgkin Lymphoma by Bioluminescence Imaging. Mol. Imaging 2015, 14, 400–413. [Google Scholar] [CrossRef] [PubMed]
- Klerk, C.P.; Overmeer, R.M.; Niers, T.M.; Versteeg, H.H.; Richel, D.J.; Buckle, T.; Van Noorden, C.J.; van Tellingen, O. Validity of bioluminescence measurements for noninvasive in vivo imaging of tumor load in small animals. Biotechniques 2007, 43, 7–13. [Google Scholar] [CrossRef] [PubMed]
- Flümann, R.; Nieper, P.; Reinhardt, H.C.; Knittel, G. New murine models of aggressive lymphoma. Leuk. Lymphoma 2020, 61, 788–798. [Google Scholar] [CrossRef] [PubMed]
- Horton, H.M.; Bernett, M.J.; Pong, E.; Peipp, M.; Karki, S.; Chu, S.Y.; Richards, J.O.; Vostiar, I.; Joyce, P.F.; Repp, R.; et al. Potent In vitro and In vivo Activity of an Fc-Engineered Anti-CD19 Monoclonal Antibody against Lymphoma and Leukemia. Cancer Res. 2008, 68, 8049–8057. [Google Scholar] [CrossRef]
- Pereira, D.S.; Guevara, C.I.; Jin, L.; Mbong, N.; Verlinsky, A.; Hsu, S.J.; Aviña, H.; Karki, S.; Abad, J.D.; Yang, P.; et al. AGS67E, an Anti-CD37 Monomethyl Auristatin E Antibody-Drug Conjugate as a Potential Therapeutic for B/T-Cell Malignancies and AML: A New Role for CD37 in AML. Mol. Cancer Ther. 2015, 14, 1650–1660. [Google Scholar] [CrossRef]
- Barth, M.J.; Hernandez-Ilizaliturri, F.J.; Mavis, C.; Tsai, P.-C.; Gibbs, J.F.; Deeb, G.; Czuczman, M.S. Ofatumumab demonstrates activity against rituximab-sensitive and -resistant cell lines, lymphoma xenografts and primary tumour cells from patients with B-cell lymphoma. Br. J. Haematol. 2012, 156, 490–498. [Google Scholar] [CrossRef]
- Ahmadbeigi, N.; Alatab, S.; Vasei, M.; Ranjbar, A.; Aghayan, S.; Khorsand, A.; Moradzadeh, K.; Darvishyan, Z.; Jamali, M.; Muhammadnejad, S. Characterization of a xenograft model for anti-CD19 CAR T cell studies. Clin. Transl. Oncol. 2021, 23, 2181–2190. [Google Scholar] [CrossRef]
- Webster, B.; Xiong, Y.; Hu, P.; Wu, D.; Alabanza, L.; Orentas, R.J.; Dropulic, B.; Schneider, D. Self-driving armored CAR-T cells overcome a suppressive milieu and eradicate CD19+ Raji lymphoma in preclinical models. Mol. Ther. 2021, 29, 2691–2706. [Google Scholar] [CrossRef]
- Ruella, M.; Kenderian, S.S.; Shestova, O.; Fraietta, J.A.; Qayyum, S.; Zhang, Q.; Maus, M.V.; Liu, X.; Nunez-Cruz, S.; Klichinsky, M.; et al. The Addition of the BTK Inhibitor Ibrutinib to Anti-CD19 Chimeric Antigen Receptor T Cells (CART19) Improves Responses against Mantle Cell Lymphoma. Clin. Cancer Res. 2016, 22, 2684–2696. [Google Scholar] [CrossRef]
- Ormhøj, M.; Scarfò, I.; Cabral, M.L.; Bailey, S.R.; Lorrey, S.J.; Bouffard, A.A.; Castano, A.P.; Larson, R.C.; Riley, L.S.; Schmidts, A.; et al. Chimeric Antigen Receptor T Cells Targeting CD79b Show Efficacy in Lymphoma with or without Cotargeting CD19. Clin. Cancer Res. 2019, 25, 7046–7057. [Google Scholar] [CrossRef] [PubMed]
- Jin, C.-H.; Xia, J.; Rafiq, S.; Huang, X.; Hu, Z.; Zhou, X.; Brentjens, R.J.; Yang, Y.-G. Modeling anti-CD19 CAR T cell therapy in humanized mice with human immunity and autologous leukemia. EBioMedicine 2019, 39, 173–181. [Google Scholar] [CrossRef] [PubMed]
| Cell Line | Citation | Age, Sex | Cell Source and Clinical Stage | EBV-Status | Immunophenotype | Characteristic Mutations, Aberrations or Translocations | Culture Requirements |
|---|---|---|---|---|---|---|---|
| ABC-DLBCL cell line models | |||||||
| HBL-1 | Abe et al. [56] | 65 y.o. male | Pleural effusion | Negative | (IgM, K), Bl’, BA-I+, and HLA-DR’. | t(14;16) | RPMI-1640 No glutamine Doubling time: 48 h |
| HBL-2 | Abe et al. [56] | 84 y.o. male | Cervical lymph node biopsy | Negative | Monoclonal surface Igs (IgM, D, A), Fcy-receptors, C3 receptors, Bl’, and HLA-DR’. Weak positivity for IgD | t(11; 14)(q13,32) | RPMI-1640 Glutamine Doubling time: 48 h |
| ULA | Berglund et al. [57] | 57 y.o. male | Ascites IV stage | Negative | CD10+, CD19+, CD20+, CD22+, CD27+, CD38+, CD40+, CD79b+, IgM+, IgD+, lambda light chain+, FMC7+, BCL-2+, BCL-6+, MHC class I and II+ | p53-gene loss (14;18)(q32;q21) | Opti-MEM (45%) IMDM (45%) Glutamine Could not survive in RPMI-1640 Doubling time: N/A |
| TDM8 | Tohda et al. [58] | 62 y.o male | Bone marrow | Negative | CD5+, CD19+, CD20+, HLA-DR+, s-IgM, s-kappa | 48, XY, add(1)(p3?), add(1)(q42), add(6)(p2?), del(6)(q?), +9, i(9)(p10) × 2, 15p, +18, −19, +mar karyotype | α-MEM No glutamine Doubling time: 30 h |
| OCI-Ly18 | Chang et al. [32] | 56 y.o male | Pleural effusion, High grade DLBCL | Negative | CD19+, CD20+, CD21+, CD23+, CD34+ | Translocations involving bands
| IMDM No glutamine Doubling time: 36 h |
| RC-K8 | Kubonishi et al. [59] | 55 y.o male | Peritoneal effusion | Negative | Complement receptors+, Ia+, B1+, and Leu 12 antigens+ | 14q+ chromosome, EBNA- t(11;14)(q23;q32) | RPMI-1640 No glutamine Doubling time: 48–60 h |
| U-2946 | Quentmeier et al. [36] | 52 y.o male | Pleural effusion, IV stage DLBCL | Negative | CD20+, CD79a+, CD10+, BCL-6+, MYC+, p53+ Partial expression of MUM1 and FOXP1 | t(8;14) | RPMI-1640 Glutamine Doubling time: 48 h |
| GCB-DLBCL cell line models | |||||||
| Karpas422 | Dyer et al. [38] | 72 y.o female | Pleural effusion | Negative | CD19+, CD37+, IgM+, and IgG+, 30% of cells IgD+ Weak CD10+ Stable expression of CAMPATH-1 (CDw52) as in normal lymphocytes | t(14;18) t(4;11) | RPMI-1640 No glutamine Doubling time: 60–90 h |
| MYC, BCL-2/BCL-6 rearrangement models | |||||||
| EJ-1 | Goy et al. [60] | 43 y.o female | Ascites, IV stage DLBCL | Negative | CD10+, CD19+, CD20+, CD22+, CD45+, CD79b, sIgM, and light chain lambda+ | t(14;8) t(8;14) del(7)(q31q32) | RPMI-1640 Glutamine Doubling time: 24 h |
| RC | Pham et al. [61] | Unknown | Pleural effusion, High-grade DLBCL | Negative | CD10+, CD19+, CD20+ (a small subset), CD22+, CD23+, CD38+, CD43+, CD44 (only partially), CD45+, CD79b+ | t(2;8)(p12;q24.2) t(14;18)(q32;q21.3) | RPMI-1640 No glutamine Optimally could be split 1:2 every 3–4 days. |
| U-2973 | Boström et al. [62] | 42 y.o male | Peripheral blood mononucleated cells at diagnosis | Negative | CD19+, CD20+, CD22+, CD10+, CD38+, cytoplasmic CD79a, and dim kappa surface Ig. FMC7+ (only partially), CD52+ CMYC+, and BCL2+ | t(14;18)(q32;q) | RPMI-1640 No glutamine Doubling time: 34 h |
| Models or Richter’s transformation | |||||||
| U-RT1 | Schmid et al. [63] | 60 y.o male | Lymph node biopsy | Positive | CD20+, CD23+, BCL-2+, PAX-5+ CDKN2A- a chromosomal gain of the NOTCH1 gene locus | CLL cells: 13q14.3 17p13.1 (loss of a single copy of TP53) deletions as well as a mutation in the other TP53 copy (c.342-343del2bpins1bp) No TP53 loss in a lymph node biopsy material; however, a subset of cells still carried 13q14.3 deletion. | IMDM No glutamine Doubling time: approximately 36 h |
| VR09 | Nichele et al. [64] | 75 y.o male | Bone marrow sample | Positive | CD19+, CD20+, CD22+, CD23+, CD43+, CD45+, CD38+, CD138+, IgD+, IgM+, IgG+, kappa chain+, ZAP-70+ BCL-2+, MNDA+, and MUM1+ | Chromosome 12 trisomy | RPMI-1640 No glutamine 10% DMSO Doubling time: 84 h |
| Models of secondary lymphomas | |||||||
| U-2932 | Amini et al. [50] | 29 y.o female | Pleural effusion Nodular sclerosis HL type 1 with progression to ABC-DLBCL | Negative | The Hodgkin and Reed–Sternberg (HRS)-cells of the HL: CD30+, CD45+, CD15+, LMP-1+, p53+, Rb+, BCL-2+, BCL-6+ DLBCL cells: CD20+, CD30+, CD45+, CD15+, LMP-1+, p53þ, Rb+, BCL-2þ, BCL-6þ U-2932 line: LMP-1+, p53+, Rb+, BCL-2+, and BCL-6+ | R1: upregulation of BCL2 and BCL6 R2: upregulation of BCL2 and MYC | RPMI-1640 Glutamine Doubling time: N/A |
| Immunotherapy | Cell Lines | Reference |
|---|---|---|
| Monoclonal Antibodies (mAbs) | ||
| Tafasitamab (anti-CD19 mAb) | B-ALL cell lines: SEM, Jurkat, CEM, MOLT-16, Nalm-6 cells | [78] |
| DuoHexaBody-CD37 | DLBCL cell lines: OCI-Ly7, OCI-Ly19, RC-K8, Ri-1, SU-DHL-4, SU-DHL-8, WSU-DLCL-2, U-2932 | [81] |
| Daratumumab (anti-CD38 mAb) | DLBCL cell lines: Toledo, WSU-DLC2, SU-DHL-4, SU-DHL-6 MCL cell lines: Jeko, REC-1, Mino, UPN1 FL cell lines: SC-1, WSU-FSCCL BL cell lines: Daudi | [83] |
| Anti-CD47 in combination with RTX | DLBCL cell lines: OCI-Ly3, U-2932, SU-DHL-2, SU-DHL-4, SU-DHL-6, SU-DHL-10 | [84] |
| Anti-PD-L1 in combination with vincristine | DLBCL cell lines: OCI-Ly-3, TDM8, SU-DHL-4 | [88] |
| Antibody-drug conjugates (ADCs) | ||
| Polatuzumab vedotin (anti-CD79b ADC) | DLBCL cell lines: Pola-sensitive: DB, STR-428, SU-DHL10, SU-DHL-4, NU-DUL-1, U-2932 Pola-resistant: SU-DHL-8, HT, SU-DHL-2, RC-K8 | [94] |
| Pinatuzumab vedotin (anti-CD22 ADC) versus Polatuzumab vedotin (anti-CD79b ADC) | DLBCL cell lines: U-2932, RIVA, TDM8, OCI-Ly10, OCI-Ly3, HBL1, BJAB, Pfeiffer, Farage, SU-DHL-6, SU-DHL-10 | [96] |
| Naratuximab emtansine (Anti-CD37 ADC) | DLBCL cell lines: Farage, RL BL cell lines: Ramos, Raji, Daudi | [74] |
| Naratuximab emtansine (Anti-CD37 ADC) with RTX | DLBCL cell lines: U-2932, SU-DHL-4, DOHH-2, OCI-Ly18, OCI-Ly7, Farage | [75] |
| Anti-CD37 alfa-amanitin conjugated ADCs | DLBCL cell line: U-RT-1 | [98] |
| CAR-T cell therapy | ||
| Anti-CD19 CAR-T cells with anti-CD20-IFN fusion protein | DLBCL cell lines: OCI-Ly2 and OCI-Ly19 BL cell lines: Raji, Daudi Anaplastic large cell lymphoma cell line: DEL MCL cell lines: Granta-519, Jeko-1 | [100] |
| Anti-CD37 CAR-T cells | DLBCL cell lines: SU-DHL-6, SU-DHL-4, Oci-Ly3, Oci-Ly7, Oci-Ly10, K422 MCL cell lines: Granta-519, Jeko-1, MINO, Maver-1, FL cell lines: SC-1 BL cell lines: Daudi, Raji, Ramos, BL-41 | [102] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kubacz, M.; Kusowska, A.; Winiarska, M.; Bobrowicz, M. In Vitro Diffuse Large B-Cell Lymphoma Cell Line Models as Tools to Investigate Novel Immunotherapeutic Strategies. Cancers 2023, 15, 235. https://doi.org/10.3390/cancers15010235
Kubacz M, Kusowska A, Winiarska M, Bobrowicz M. In Vitro Diffuse Large B-Cell Lymphoma Cell Line Models as Tools to Investigate Novel Immunotherapeutic Strategies. Cancers. 2023; 15(1):235. https://doi.org/10.3390/cancers15010235
Chicago/Turabian StyleKubacz, Matylda, Aleksandra Kusowska, Magdalena Winiarska, and Małgorzata Bobrowicz. 2023. "In Vitro Diffuse Large B-Cell Lymphoma Cell Line Models as Tools to Investigate Novel Immunotherapeutic Strategies" Cancers 15, no. 1: 235. https://doi.org/10.3390/cancers15010235
APA StyleKubacz, M., Kusowska, A., Winiarska, M., & Bobrowicz, M. (2023). In Vitro Diffuse Large B-Cell Lymphoma Cell Line Models as Tools to Investigate Novel Immunotherapeutic Strategies. Cancers, 15(1), 235. https://doi.org/10.3390/cancers15010235





