Feasibility of Longitudinal ctDNA Assessment in Patients with Uterine and Extra-Uterine Leiomyosarcoma
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Patient Population
2.2. Specimen Collection and Processing
2.3. Personalized, Tumor-Informed ctDNA Detection
2.4. Genomic Analysis to Assess TMB and Identify Driver Mutations
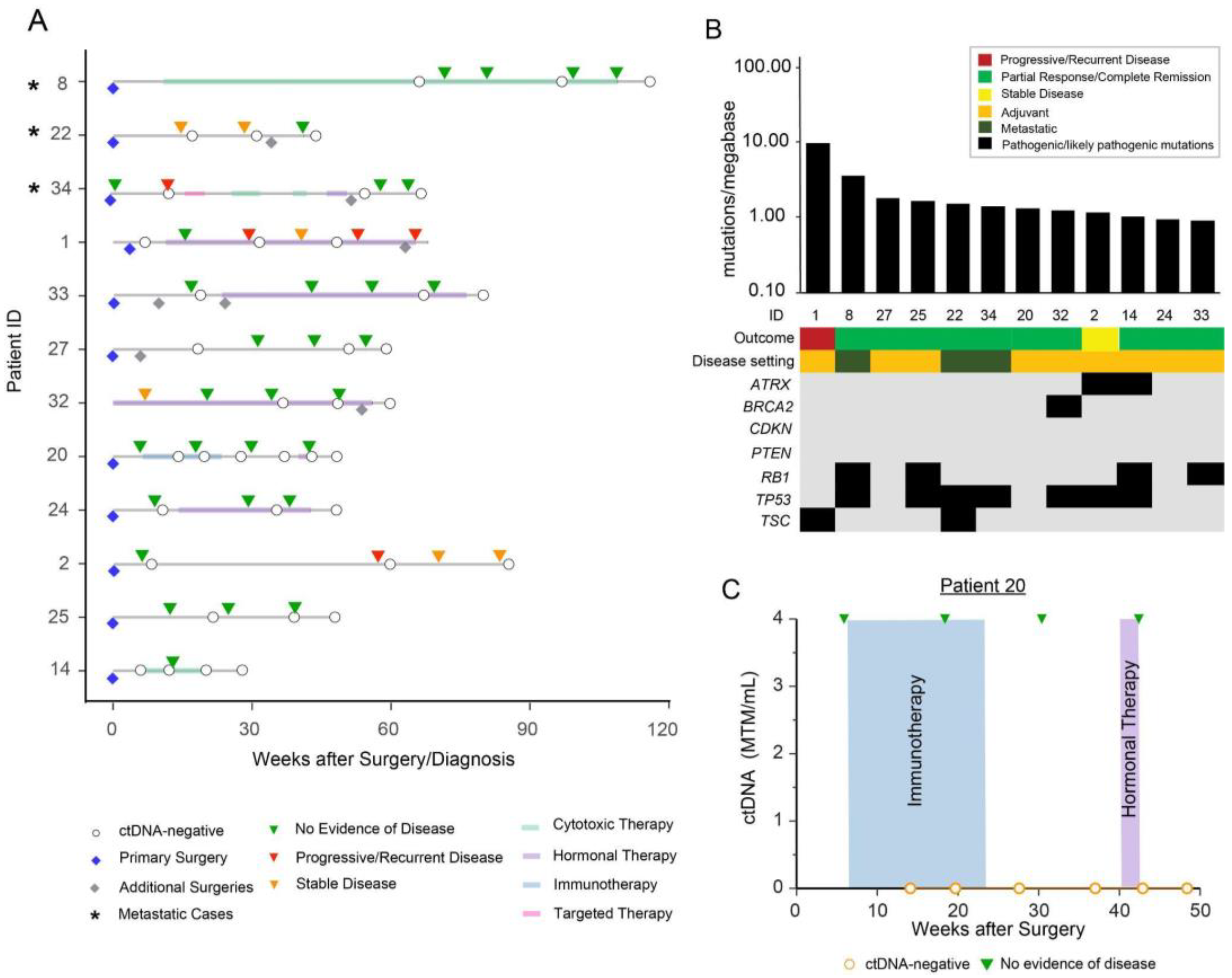
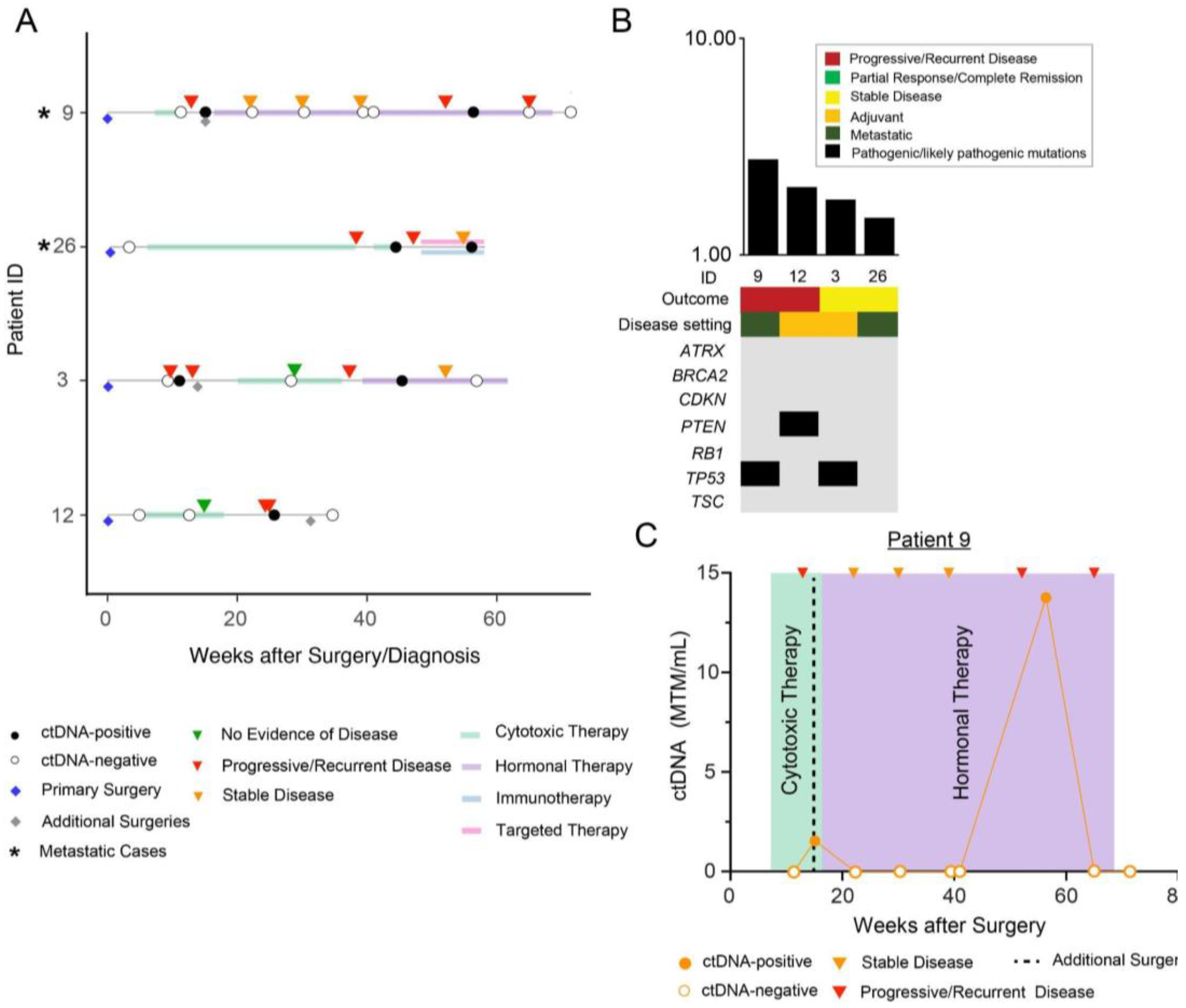
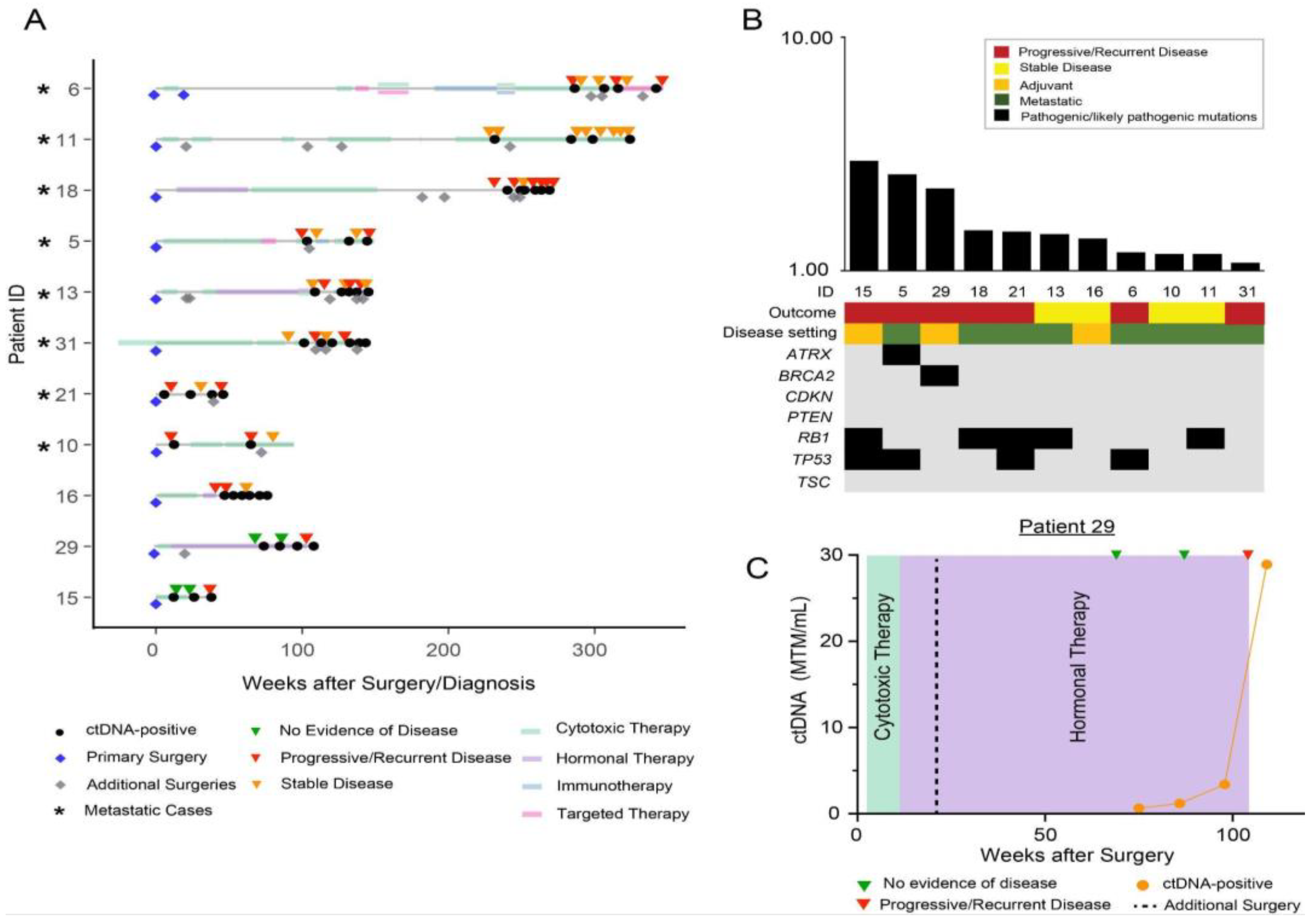
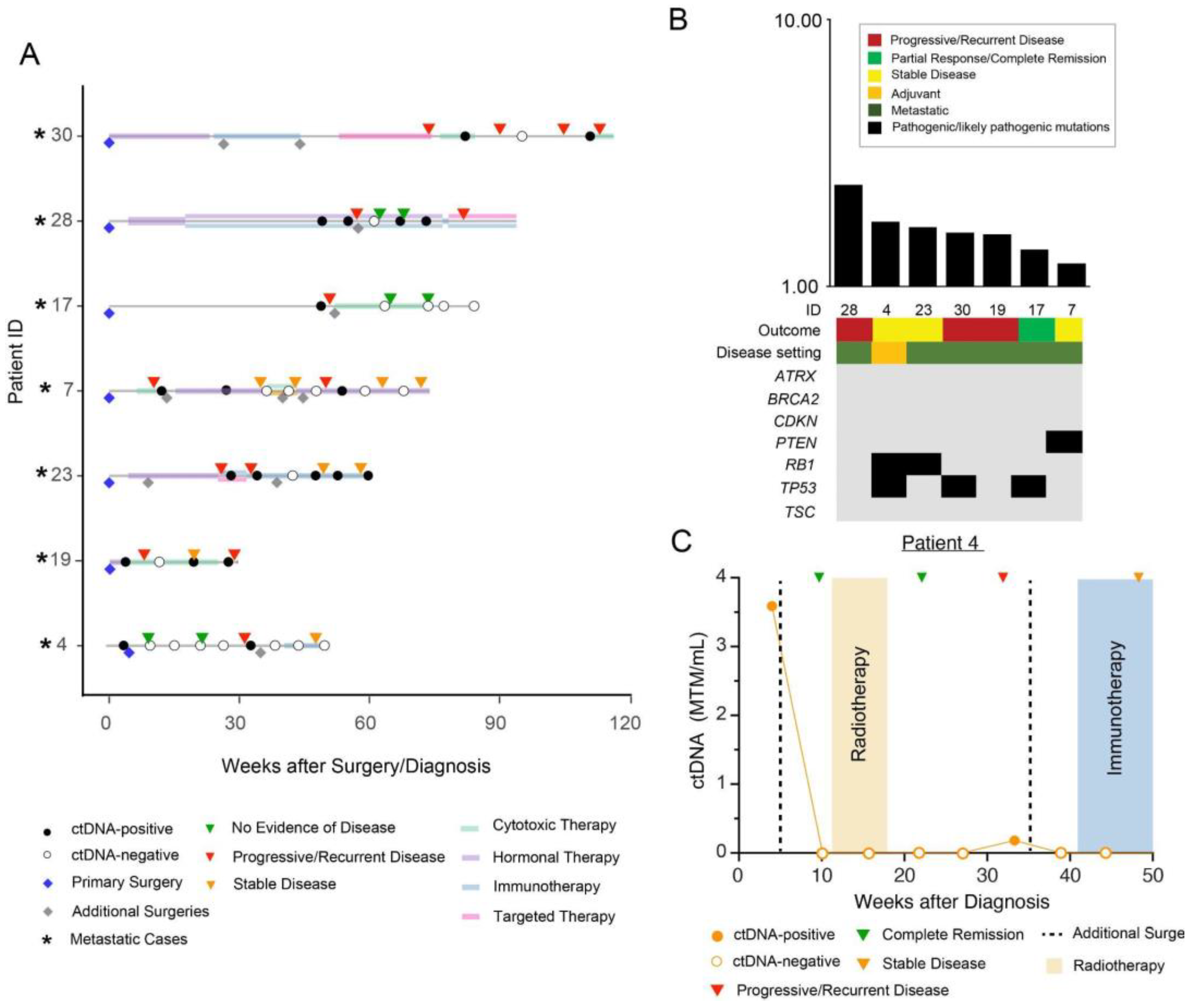
3. Results
Patient Cohort
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Siegel, R.L.; Miller, K.D.; Fuchs, H.E.; Jemal, A. Cancer statistics, 2022. CA Cancer J. Clin. 2022, 72, 7–33. [Google Scholar] [CrossRef] [PubMed]
- von Konow, A.; Ghanei, I.; Styring, E.; Hainaut, L. Late Local Recurrence and Metastasis in Soft Tissue Sarcoma of the Extremities and Trunk Wall: Better Outcome After Treatment of Late Events Compared with Early. Ann. Surg. Oncol. 2021, 28, 7891–7902. [Google Scholar] [CrossRef] [PubMed]
- Nacev, B.A.; Sanchez-Vega, F.; Smith, S.A.; Antonescu, C.R.; Rosenbaum, E.; Shi, H.; Tang, C.; Socci, N.D.; Rana, S.; Gularte-Mérida, R.; et al. Clinical sequencing of soft tissue and bone sarcomas delineates diverse genomic landscapes and potential therapeutic targets. Nat. Commun. 2022, 13, 3405. [Google Scholar] [CrossRef]
- Ducimetière, F.; Lurkin, A.; Ranchère-Vince, D.; Decouvelaere, A.-V.; Peoc’H, M.; Istier, L.; Chalabreysse, P.; Muller, C.; Alberti, L.; Bringuier, P.-P.; et al. Incidence of Sarcoma Histotypes and Molecular Subtypes in a Prospective Epidemiological Study with Central Pathology Review and Molecular Testing. PLoS ONE 2011, 6, e20294. [Google Scholar] [CrossRef] [PubMed]
- Toro, J.R.; Travis, L.B.; Wu, H.J.; Zhu, K.; Fletcher, C.D.; Devesa, S.S. Incidence patterns of soft tissue sarcomas, regardless of primary site, in the surveillance, epidemiology and end results program, 1978–2001: An analysis of 26,758 cases. Int. J. Cancer 2006, 119, 2922–2930. [Google Scholar] [CrossRef] [PubMed]
- Mangla, A.; Yadav, U. Leiomyosarcoma; StatPearls: Treasure Island, FL, USA, 2022. [Google Scholar]
- Venkatraman, L.; Goepel, J.R.; Steele, K.; Dobbs, S.P.; Lyness, R.W.; McCluggage, W.G. Soft tissue, pelvic, and urinary bladder leiomyosarcoma as second neoplasm following hereditary retinoblastoma. J. Clin. Pathol. 2003, 56, 233–236. [Google Scholar] [CrossRef]
- Ognjanovic, S.; Olivier, M.; Bergemann, T.L.; Hainaut, P. Sarcomas in TP53 germline mutation carriers: A review of the IARC TP53 database. Cancer 2012, 118, 1387–1396. [Google Scholar] [CrossRef]
- Bjerkehagen, B.; Smeland, S.; Walberg, L.; Skjeldal, S.; Hall, K.S.; Nesland, J.M.; Småstuen, M.C.; Fosså, S.D.; Saeter, G. Radiation-induced sarcoma: 25-year experience from The Norwegian Radium Hospital. Acta Oncol. 2008, 47, 1475–1482. [Google Scholar] [CrossRef]
- Robinson, E.; Neugut, A.I.; Wylie, P. Clinical Aspects of Postirradiation Sarcomas1. JNCI: J. Natl. Cancer Inst. 1988, 80, 233–240. [Google Scholar] [CrossRef]
- Hemming, M.L.; Klega, K.; Rhoades, J.; Ha, G.; Acker, K.E.; Andersen, J.L.; Thai, E.; Nag, A.; Thorner, A.R.; Raut, C.P.; et al. Detection of Circulating Tumor DNA in Patients With Leiomyosarcoma With Progressive Disease. JCO Precis. Oncol. 2019, 2019, 1–11. [Google Scholar] [CrossRef]
- Blaye, C.; Kind, M.; Stoeckle, E.; Brouste, V.; Kantor, G.; Le Loarer, F.; Italiano, A.; Toulmonde, M. Local and Metastatic Relapse Features in Patients After a Primary Soft Tissue Sarcoma: Advocating for a Better-Tailored Follow-Up. Front. Oncol. 2019, 9, 559. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Li, S.; Zhang, Z.; Jia, J.; Shan, B. Prevalence and occult rates of uterine leiomyosarcoma. Medicine 2020, 99, e21766. [Google Scholar] [CrossRef] [PubMed]
- Cuppens, T.; Moisse, M.; Depreeuw, J.; Annibali, D.; Colas, E.; Gil-Moreno, A.; Huvila, J.; Carpén, O.; Zikán, M.; Matias-Guiu, X.; et al. Integrated genome analysis of uterine leiomyosarcoma to identify novel driver genes and targetable pathways. Int. J. Cancer 2017, 142, 1230–1243. [Google Scholar] [CrossRef] [PubMed]
- NCCN. Uterine Neoplasms, Version 1. 2022. Available online: https://www.nccn.org/professionals/physician_gls/pdf/uterine.pdf (accessed on 3 December 2022).
- Madanat-Harjuoja, L.M.; Klega, K.; Lu, Y.; Shulman, D.S.; Thorner, A.R.; Nag, A.; Tap, W.D.; Reinke, D.K.; Diller, L.; Ballman, K.V.; et al. Circulating Tumor DNA Is Associated with Response and Survival in Patients with Advanced Leiomyosarcoma. Clin. Cancer Res. 2022, 28, 2579–2586. [Google Scholar] [CrossRef] [PubMed]
- Abbosh, C.; Birkbak, N.J.; Wilson, G.A.; Jamal-Hanjani, M.; Constantin, T.; Salari, R.; Le Quesne, J.; Moore, D.A.; Veeriah, S.; Rosenthal, R.; et al. Phylogenetic ctDNA analysis depicts early-stage lung cancer evolution. Nature 2017, 545, 446–451. [Google Scholar] [CrossRef] [PubMed]
- Christensen, E.; Birkenkamp-Demtröder, K.; Sethi, H.; Shchegrova, S.; Salari, R.; Nordentoft, I.K.; Wu, H.-T.; Knudsen, M.; Lamy, P.; Lindskrog, S.V.; et al. Early Detection of Metastatic Relapse and Monitoring of Therapeutic Efficacy by Ultra-Deep Sequencing of Plasma Cell-Free DNA in Patients With Urothelial Bladder Carcinoma. J. Clin. Oncol. 2019, 37, 1547–1557. [Google Scholar] [CrossRef]
- Coombes, R.C.; Page, K.; Salari, R.; Hastings, R.K.; Armstrong, A.C.; Ahmed, S.; Ali, S.; Cleator, S.J.; Kenny, L.M.; Stebbing, J.; et al. Personalized Detection of Circulating Tumor DNA Antedates Breast Cancer Metastatic Recurrence. Clin. Cancer Res. 2019, 25, 4255–4263. [Google Scholar] [CrossRef]
- Reinert, T.; Henriksen, T.V.; Christensen, E.; Sharma, S.; Salari, R.; Sethi, H.; Knudsen, M.; Nordentoft, I.K.; Wu, H.-T.; Tin, A.S.; et al. Analysis of Plasma Cell-Free DNA by Ultradeep Sequencing in Patients With Stages I to III Colorectal Cancer. JAMA Oncol. 2019, 5, 1124–1131. [Google Scholar] [CrossRef]
- Szabados, B.; Kockx, M.; Assaf, Z.J.; van Dam, P.-J.; Rodriguez-Vida, A.; Duran, I.; Crabb, S.J.; Van Der Heijden, M.S.; Pous, A.F.; Gravis, G.; et al. Final Results of Neoadjuvant Atezolizumab in Cisplatin-ineligible Patients with Muscle-invasive Urothelial Cancer of the Bladder. Eur. Urol. 2022, 82, 212–222. [Google Scholar] [CrossRef]
- Kasi, P.M.; Sawyer, S.; Guilford, J.; Munro, M.; Ellers, S.; Wulff, J.; Hook, N.; Krinshpun, S.; Malashevich, A.K.; Malhotra, M.; et al. BESPOKE study protocol: A multicentre, prospective observational study to evaluate the impact of circulating tumour DNA guided therapy on patients with colorectal cancer. BMJ Open 2021, 11, e047831. [Google Scholar] [CrossRef]
- Henriksen, T.V.; Tarazona, N.; Frydendahl, A.; Reinert, T.; Gimeno-Valiente, F.; Carbonell-Asins, J.A.; Sharma, S.; Renner, D.; Hafez, D.; Roda, D.; et al. Circulating Tumor DNA in Stage III Colorectal Cancer, beyond Minimal Residual Disease Detection, toward Assessment of Adjuvant Therapy Efficacy and Clinical Behavior of Recurrences. Clin. Cancer Res. 2022, 28, 507–517. [Google Scholar] [CrossRef]
- Kasi, P.M.; Fehringer, G.; Taniguchi, H.; Starling, N.; Nakamura, Y.; Kotani, D.; Powles, T.; Li, B.T.; Pusztai, L.; Aushev, V.N.; et al. Impact of Circulating Tumor DNA–Based Detection of Molecular Residual Disease on the Conduct and Design of Clinical Trials for Solid Tumors. JCO Precis. Oncol. 2022, 6, e2100181. [Google Scholar] [CrossRef] [PubMed]
- Dall, G.V.; Hamilton, A.; Ratnayake, G.; Scott, C.; Barker, H. Interrogating the Genomic Landscape of Uterine Leiomyosarcoma: A Potential for Patient Benefit. Cancers 2022, 14, 1561. [Google Scholar] [CrossRef] [PubMed]
- Gajic, Z.Z.; Deshpande, A.; Legut, M.; Imieliński, M.; Sanjana, N.E. Recurrent somatic mutations as predictors of immunotherapy response. Nat. Commun. 2022, 13, 3938. [Google Scholar] [CrossRef] [PubMed]
- Powles, T.; Assaf, Z.J.; Davarpanah, N.; Banchereau, R.; Szabados, B.E.; Yuen, K.C.; Grivas, P.; Hussain, M.; Oudard, S.; Gschwend, J.E.; et al. ctDNA guiding adjuvant immunotherapy in urothelial carcinoma. Nature 2021, 595, 432–437. [Google Scholar] [CrossRef]
- Chiang, S.; Vasudevaraja, V.; Serrano, J.; Stewart, C.J.R.; Oliva, E.; Momeni-Boroujeni, A.; Jungbluth, A.A.; Paula, A.D.C.; da Silva, E.M.; Weigelt, B.; et al. TSC2-mutant uterine sarcomas with JAZF1-SUZ12 fusions demonstrate hybrid features of endometrial stromal sarcoma and PEComa and are responsive to mTOR inhibition. Mod. Pathol. 2021, 35, 117–127. [Google Scholar] [CrossRef]
- Bui, N.Q.; Przybyl, J.; Trabucco, S.E.; Frampton, G.; Hastie, T.; Van De Rijn, M.; Ganjoo, K.N. A clinico-genomic analysis of soft tissue sarcoma patients reveals CDKN2A deletion as a biomarker for poor prognosis. Clin. Sarcoma Res. 2019, 9, 12. [Google Scholar] [CrossRef]
- Chudasama, P.; Mughal, S.S.; Sanders, M.A.; Hübschmann, D.; Chung, I.; Deeg, K.I.; Wong, S.-H.; Rabe, S.; Hlevnjak, M.; Zapatka, M.; et al. Integrative genomic and transcriptomic analysis of leiomyosarcoma. Nat. Commun. 2018, 9, 144. [Google Scholar] [CrossRef]
- Mäkinen, N.; Aavikko, M.; Heikkinen, T.; Taipale, M.; Taipale, J.; Koivisto-Korander, R.; Bützow, R.; Vahteristo, P. Exome Sequencing of Uterine Leiomyosarcomas Identifies Frequent Mutations in TP53, ATRX, and MED12. PLOS Genet. 2016, 12, e1005850. [Google Scholar] [CrossRef]
- Darmusey, L.; Pérot, G.; Thébault, N.; Le Guellec, S.; Desplat, N.; Gaston, L.; Delespaul, L.; Lesluyes, T.; Darbo, E.; Gomez-Brouchet, A.; et al. ATRX Alteration Contributes to Tumor Growth and Immune Escape in Pleomorphic Sarcomas. Cancers 2021, 13, 2151. [Google Scholar] [CrossRef]
- Seligson, N.D.; Tang, J.; Jin, D.X.; Bennett, M.P.; Elvin, J.A.; Graim, K.; Hays, J.L.; Millis, S.Z.; Miles, W.O.; Chen, J.L. Drivers of genomic loss of heterozygosity in leiomyosarcoma are distinct from carcinomas. npj Precis. Oncol. 2022, 6, 29. [Google Scholar] [CrossRef] [PubMed]
- Seligson, N.D.; Kautto, E.A.; Passen, E.N.; Stets, C.; Toland, A.E.; Millis, S.Z.; Meyer, C.F.; Hays, J.L.; Chen, J.L. BRCA1/2 Functional Loss Defines a Targetable Subset in Leiomyosarcoma. Oncologist 2018, 24, 973–979. [Google Scholar] [CrossRef] [PubMed]
- Astolfi, A.; Nannini, M.; Indio, V.; Schipani, A.; Rizzo, A.; Perrone, A.M.; De Iaco, P.; Pirini, M.G.; De Leo, A.; Urbini, M.; et al. Genomic Database Analysis of Uterine Leiomyosarcoma Mutational Profile. Cancers 2020, 12, 2126. [Google Scholar] [CrossRef] [PubMed]
- Kantidakis, G.; Litière, S.; Neven, A.; Vinches, M.; Judson, I.; Schöffski, P.; Wardelmann, E.; Stacchiotti, S.; D’Ambrosio, L.; Marréaud, S.; et al. Efficacy thresholds for clinical trials with advanced or metastatic leiomyosarcoma patients: A European Organisation for Research and Treatment of Cancer Soft Tissue and Bone Sarcoma Group meta-analysis based on a literature review for soft-tissue sarcomas. Eur. J. Cancer 2021, 154, 253–268. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhou, M.; Bui, N.; Rathore, R.; Sudhaman, S.; George, G.V.; Malashevich, A.K.; Malhotra, M.; Liu, M.C.; Aleshin, A.; Ganjoo, K.N. Feasibility of Longitudinal ctDNA Assessment in Patients with Uterine and Extra-Uterine Leiomyosarcoma. Cancers 2023, 15, 157. https://doi.org/10.3390/cancers15010157
Zhou M, Bui N, Rathore R, Sudhaman S, George GV, Malashevich AK, Malhotra M, Liu MC, Aleshin A, Ganjoo KN. Feasibility of Longitudinal ctDNA Assessment in Patients with Uterine and Extra-Uterine Leiomyosarcoma. Cancers. 2023; 15(1):157. https://doi.org/10.3390/cancers15010157
Chicago/Turabian StyleZhou, Maggie, Nam Bui, Richa Rathore, Sumedha Sudhaman, Giby V. George, Allyson K. Malashevich, Meenakshi Malhotra, Minetta C. Liu, Alexey Aleshin, and Kristen N. Ganjoo. 2023. "Feasibility of Longitudinal ctDNA Assessment in Patients with Uterine and Extra-Uterine Leiomyosarcoma" Cancers 15, no. 1: 157. https://doi.org/10.3390/cancers15010157
APA StyleZhou, M., Bui, N., Rathore, R., Sudhaman, S., George, G. V., Malashevich, A. K., Malhotra, M., Liu, M. C., Aleshin, A., & Ganjoo, K. N. (2023). Feasibility of Longitudinal ctDNA Assessment in Patients with Uterine and Extra-Uterine Leiomyosarcoma. Cancers, 15(1), 157. https://doi.org/10.3390/cancers15010157




