Ibrutinib Associated with Rituximab-Platinum Salt-Based Immunochemotherapy in B-Cell Lymphomas: Results of a Phase 1b-II Study of the LYSA Group
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
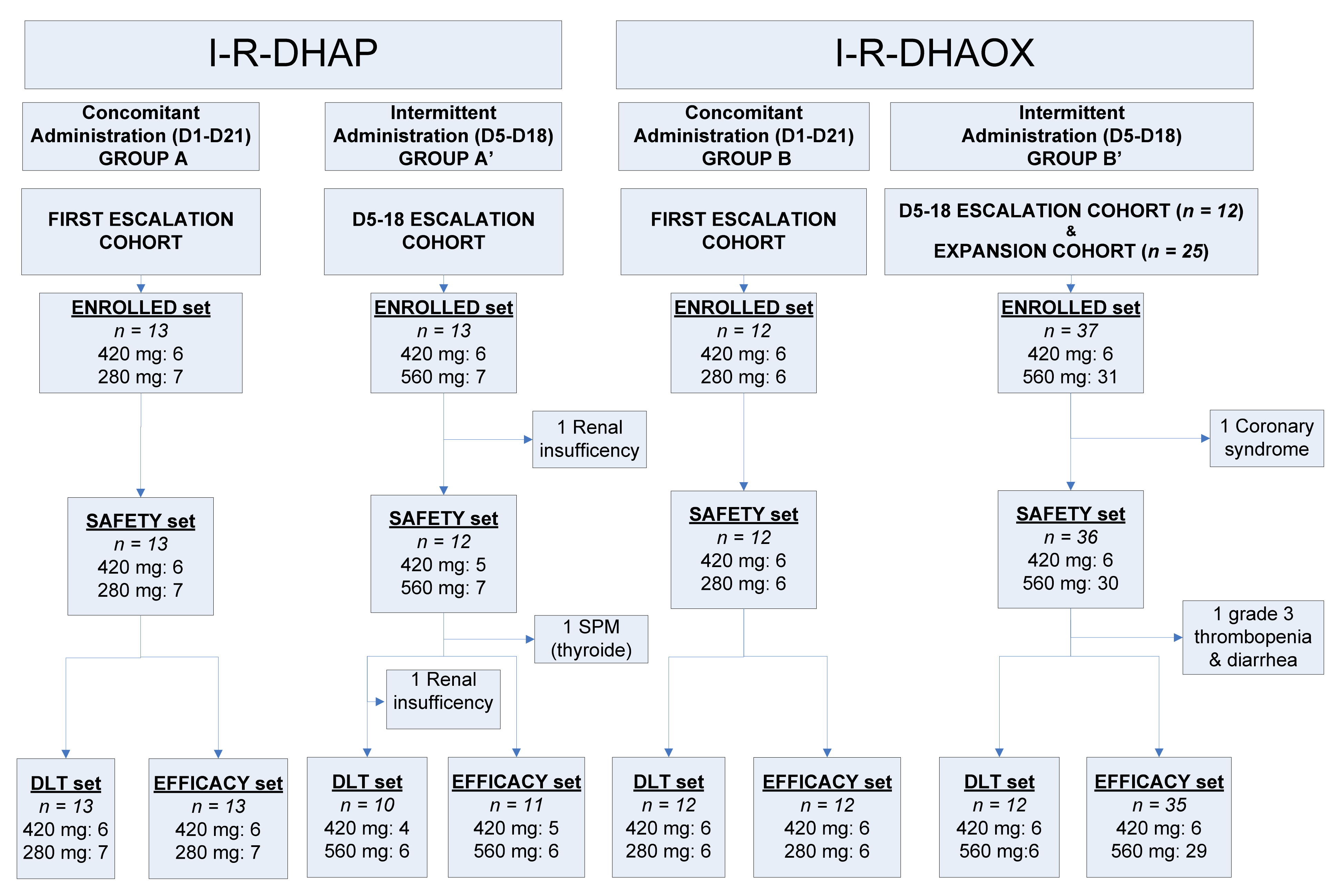
2.1. Study Design and Patients
2.2. Procedures
2.3. Endpoints
2.4. Statistical Analyses
3. Results
3.1. Safety and Tolerability
3.1.1. Ibrutinib in Combination with R-DHAP
3.1.2. Ibrutinib in Combination with R-DHAOx
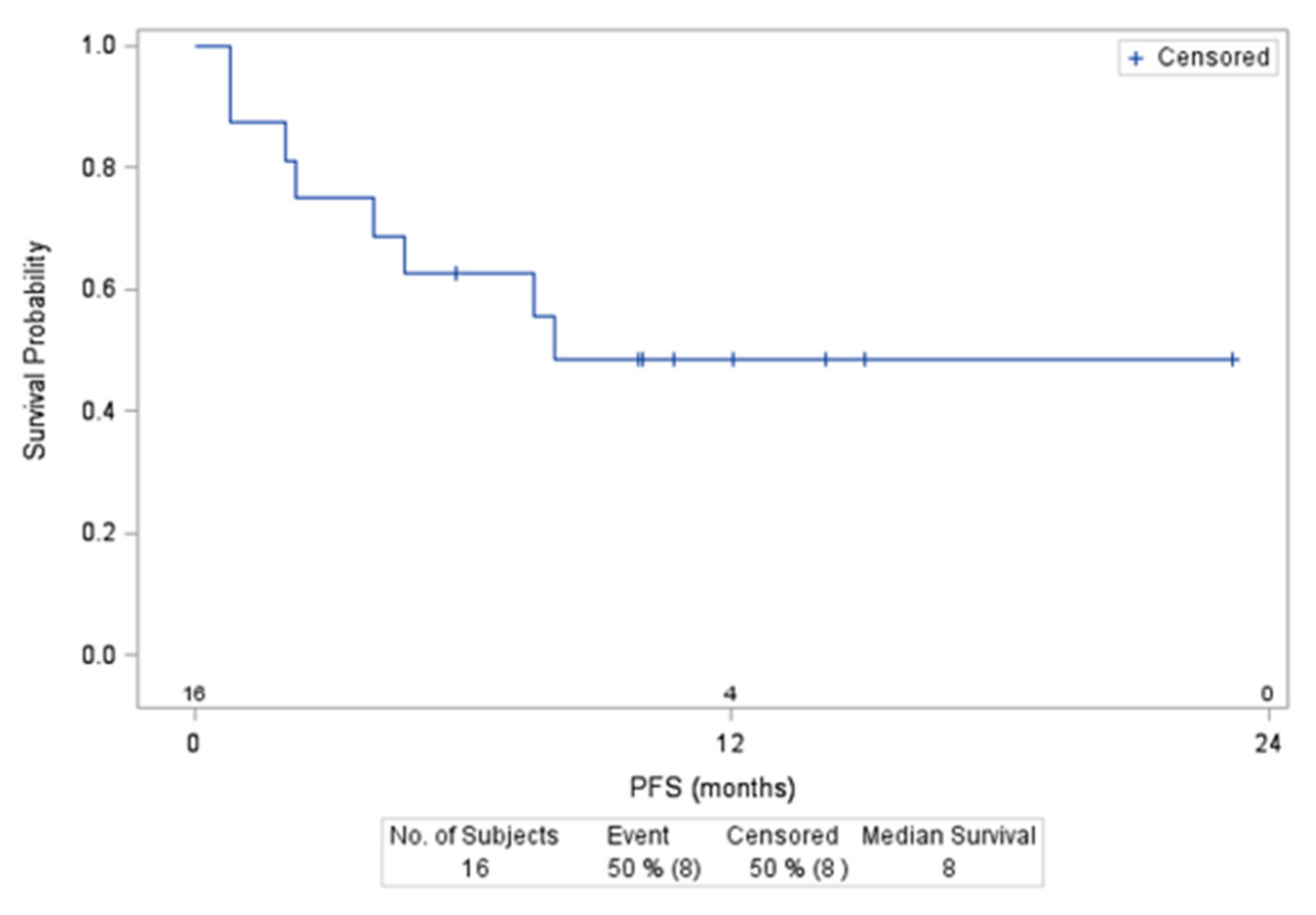
3.2. Patient Outcomes
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Coiffier, B.; Sarkozy, C. Diffuse Large B-Cell Lymphoma: R-CHOP failure—What to Do? Hematology 2016, 2016, 366–378. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bosly, A.; Coiffier, B.; Gisselbrecht, C.; Tilly, H.; Auzanneau, G.; Andrien, F.; Herbrecht, R.; Legros, M.; Devaux, Y.; Jaubert, J. Bone Marrow Transplantation Prolongs Survival after Relapse in Aggres-Sive-Lymphoma Patients Treated with the LNH-84 Regimen. J. Clin. Oncol. 1992, 10, 1615–1623. [Google Scholar] [CrossRef] [PubMed]
- Crump, M.; Neelapu, S.S.; Farooq, U.; Neste, E.V.D.; Kuruvilla, J.; Westin, J.; Link, B.K.; Hay, A.; Cerhan, J.R.; Zhu, L.; et al. Outcomes in Refractory Diffuse Large B-Cell Lymphoma: Results from the International SCHOLAR-1 Study. Blood 2017, 130, 1800–1808. [Google Scholar] [CrossRef] [PubMed]
- Advani, R.H.; Buggy, J.J.; Sharman, J.P.; Smith, S.M.; Boyd, T.E.; Grant, B.; Kolibaba, K.S.; Furman, R.R.; Rodriguez, S.; Chang, B.Y.; et al. Bruton Tyrosine Kinase Inhibitor Ibrutinib (PCI-32765) Has Significant Activity in Patients with Relapsed/Refractory B-Cell Malignancies. J. Clin. Oncol. 2013, 31, 88–94. [Google Scholar] [CrossRef]
- Neelapu, S.S.; Locke, F.L.; Bartlett, N.L.; Lekakis, L.J.; Reagan, P.M.; Miklos, D.B.; Jacobson, C.A.; Braunschweig, I.; Oluwole, O.O.; Siddiqi, T.; et al. Comparison of 2-Year Outcomes with CAR T Cells (ZUMA-1) Vs Salvage Chemotherapy in Refractory Large B-Cell Lymphoma. Blood Adv. 2021, 5, 4149–4155. [Google Scholar] [CrossRef]
- Solimando, A.G.; Ribatti, D.; Vacca, A.; Einsele, H. Targeting B-Cell Non Hodgkin Lymphoma: New and Old Tricks. Leuk. Res. 2016, 42, 93–104. [Google Scholar] [CrossRef]
- Bock, A.M.; Nowakowski, G.S.; Wang, Y. Bispecific Antibodies for Non-Hodgkin Lymphoma Treatment. Curr. Treat. Options Oncol. 2022, 23, 155–170. [Google Scholar] [CrossRef]
- Crump, M.; Kuruvilla, J.; Couban, S.; MacDonald, D.A.; Kukreti, V.; Kouroukis, C.T.; Rubinger, M.; Buckstein, R.; Imrie, K.R.; Federico, M.; et al. Randomized Comparison of Gemcitabine, Dexamethasone, and Cisplatin Versus Dexamethasone, Cytarabine, and Cisplatin Chemotherapy Before Autologous Stem-Cell Transplantation for Relapsed and Refractory Aggressive Lymphomas: NCIC-CTG LY.12. J. Clin. Oncol. 2014, 32, 3490–3496. [Google Scholar] [CrossRef] [Green Version]
- Gisselbrecht, C.; Glass, B.; Mounier, N.; Gill, D.S.; Linch, D.C.; Trneny, M.; Bosly, A.; Ketterer, N.; Shpilberg, O.; Hagberg, H.; et al. Salvage Regimens with Autologous Transplantation for Relapsed Large B-Cell Lym-Phoma in the Rituximab Era. J. Clin. Oncol. 2010, 28, 4184–4190. [Google Scholar] [CrossRef] [Green Version]
- Van Den Neste, E.; Schmitz, N.; Mounier, N.; Gill, D.; Linch, D.; Trneny, M.; Bouadballah, R.; Radford, J.; Bargetzi, M.; Ribrag, V.; et al. Outcomes of Diffuse Large B-Cell Lymphoma Patients Relapsing After Autologous Stem Cell Transplantation: An Analysis of Patients Included in the CORAL Study. Bone Marrow Transplant. 2017, 52, 216–221. [Google Scholar] [CrossRef] [Green Version]
- Wilson, W.H.; Young, R.M.; Schmitz, R.; Yang, Y.; Pittaluga, S.; Wright, G.; Lih, C.-J.; Williams, P.M.; Shaffer, A.L.; Gerecitano, J.; et al. Targeting B Cell Receptor Signaling with Ibrutinib in Diffuse Large B Cell Lymphoma. Nat. Med. 2015, 21, 922–926. [Google Scholar] [CrossRef] [PubMed]
- Younes, A.; Thieblemont, C.; Morschhauser, F.; Flinn, I.; Friedberg, J.W.; Amorim, S.; Hivert, B.; Westin, J.; Vermeulen, J.; Bandyopadhyay, N.; et al. Combination of Ibrutinib with Rituximab, Cyclophosphamide, Doxorubicin, Vincristine, and Prednisone (R-CHOP) for Treatment-Naive Patients with CD20-Positive B-Cell Non-Hodgkin Lymphoma: A Non-Randomised, Phase 1b Study. Lancet Oncol. 2014, 15, 1019–1026. [Google Scholar] [CrossRef]
- Sauter, C.S.; Matasar, M.J.; Schöder, H.; Devlin, S.M.; Drullinsky, P.; Gerecitano, J.; Kumar, A.; Noy, A.; Palomba, M.L.; Portlock, C.S.; et al. A Phase 1 Study of Ibrutinib in Combination With R-ICE in Patients With Relapsed or Primary Refractory DLBCL. Blood 2018, 131, 1805–1808. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zucha, M.A.; Wu, A.T.; Lee, W.H.; Wang, S.-L.; Lin, W.-W.; Yuan, C.-C.; Yeh, C.-T. Bruton’s Tyrosine Kinase (Btk) Inhibitor Ibrutinib Suppresses Stem-Like Traits in Ovarian Cancer. Oncotarget 2015, 6, 13255–13268. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shanafelt, T.D.; Wang, X.V.; Kay, N.E.; Hanson, C.A.; O’Brien, S.; Barrientos, J.; Jelinek, D.F.; Braggio, E.; Leis, J.F.; Zhang, C.C.; et al. Ibrutinib-Rituximab or Chemoimmunotherapy for Chronic Lymphocytic Leukemia. N. Engl. J. Med. 2019, 381, 432–443. [Google Scholar] [CrossRef] [PubMed]
- Dimopoulos, M.A.; Tedeschi, A.; Trotman, J.; Garcia-Sanz, R.; Macdonald, D.; Leblond, V.; Mahe, B.; Herbaux, C.; Tam, C.; Orsucci, L.; et al. Phase 3 Trial of Ibrutinib Plus Rituximab in Waldenström’s Macroglobulinemia. N. Engl. J. Med. 2018, 378, 2399–2410. [Google Scholar] [CrossRef]
- Jain, P.; Zhao, S.; Lee, H.J.; Hill, H.A.; Ok, C.Y.; Kanagal-Shamanna, R.; Hagemeister, F.B.; Fowler, N.; Fayad, L.; Yao, Y.; et al. Ibrutinib with Rituximab in First-Line Treatment of Older Patients with Mantle Cell Lymphoma. J. Clin. Oncol. 2022, 40, 202–212. [Google Scholar] [CrossRef]
- Cheson, B.D.; Pfistner, B.; Juweid, M.E.; Gascoyne, R.D.; Specht, L.; Horning, S.J.; Coiffier, B.; Fisher, R.I.; Hagenbeek, A.; Zucca, E.; et al. Revised Response Criteria for Malignant Lymphoma. J. Clin. Oncol. 2007, 25, 579–586. [Google Scholar] [CrossRef]
- Velasquez, W.S.; Cabanillas, F.; Salvador, P.; McLaughlin, P.; Fridrik, M.; Tucker, S.; Jagannath, S.; Hagemeister, F.B.; Redman, J.R.; Swan, F. Effective Salvage Therapy for Lymphoma with Cisplatin in Combination with High-Dose Ara-C and Dexamethasone (DHAP). Blood 1988, 71, 117–122. [Google Scholar] [CrossRef] [Green Version]
- Machover, D.; Delmas-Marsalet, B.; Misra, S.C.; Gumus, Y.; Goldschmidt, E.; Schilf, A.; Frénoy, N.; Emile, J.-F.; Debuire, B.; Guettier, C.; et al. Dexamethasone, High-Dose Cytarabine, and Oxaliplatin (DHAOx) As Salvage Treatment for Patients with Initially Refractory or Relapsed Non-Hodgkin’s Lymphoma. Ann. Oncol. 2001, 12, 1439–1443. [Google Scholar] [CrossRef]
- Kander, E.M.; Zhao, Q.; Bhat, S.A.; Hirsch, J.; Byrd, J.C.; Ooka, L.; Wiczer, T.; Woyach, J.A.; Awan, F.T.; Rogers, K.A.; et al. Venous and Arterial Thrombosis in Patients with Haematological Malignancy During Treatment with Ibrutinib. Br. J. Haematol. 2019, 187, 399–402. [Google Scholar] [CrossRef] [PubMed]
- Locke, F.L.; Miklos, D.B.; Jacobson, C.; Perales, M.-A.; Kersten, M.J.; Oluwole, O.O.; Ghobadi, A.; Rapoport, A.P.; McGuirk, J.P.; Pagel, J.M.; et al. Primary Analysis of ZUMA-7: A Phase 3 Randomized Trial of Axicabtagene Ciloleucel (Axi-Cel) Versus Standard-of-Care Therapy in Patients with Relapsed/Refractory Large B-Cell Lymphoma. Blood 2021, 138, 2. [Google Scholar] [CrossRef]
- Thieblemont, C.; Briere, J.; Mounier, N.; Voelker, H.-U.; Cuccuini, W.; Hirchaud, E.; Rosenwald, A.; Jack, A.; Sundstrom, C.; Cogliatti, S.; et al. The Germinal Center/Activated B-Cell Subclassification Has a Prognostic Impact for Response to Salvage Therapy in Relapsed/Refractory Diffuse Large B-Cell Lymphoma: A Bio-CORAL Study. J. Clin. Oncol. 2011, 29, 4079–4087. [Google Scholar] [CrossRef] [PubMed]
- Lignon, J.; Sibon, D.; Madelaine, I.; Brice, P.; Franchi, P.; Briere, J.; Mounier, N.; Gisselbrecht, C.; Faure, P.; Thieblemont, C. Rituximab, Dexamethasone, Cytarabine, and Oxaliplatin (R-DHAX) Is an Effective and Safe Salvage Regimen in Relapsed/Refractory B-Cell Non-Hodgkin Lymphoma. Clin. Lymphoma Myeloma Leuk. 2010, 10, 262–269. [Google Scholar] [CrossRef]
- Tixier, F.; Ranchon, F.; Iltis, A.; Vantard, N.; Schwiertz, V.; Bachy, E.; Bouafia-Sauvy, F.; Sarkozy, C.; Tournamille, J.F.; Gyan, E.; et al. Comparative Toxicities of 3 Platinum-Containing Chemotherapy Regimens in Re-lapsed/Refractory Lymphoma Patients. Hematol. Oncol. 2017, 35, 584–590. [Google Scholar] [CrossRef]
- Wright, G.W.; Huang, D.W.; Phelan, J.D.; Coulibaly, Z.A.; Roulland, S.; Young, R.M.; Wang, J.Q.; Schmitz, R.; Morin, R.; Tang, J.; et al. A Probabilistic Classification Tool for Genetic Subtypes of Diffuse Large B Cell Lymphoma with Therapeutic Implications. Cancer Cell 2020, 37, 551–568.e14. [Google Scholar] [CrossRef]
- Younes, A.; Sehn, L.H.; Johnson, P.; Zinzani, P.L.; Hong, X.; Zhu, J.; Patti, C.; Belada, D.; Samoilova, O.; Suh, C.; et al. Randomized Phase III Trial of Ibrutinib and Rituximab Plus Cyclophosphamide, Doxorubicin, Vincristine, and Prednisone in Non–Germinal Center B-Cell Diffuse Large B-Cell Lymphoma. J. Clin. Oncol. 2019, 37, 1285–1295. [Google Scholar] [CrossRef]
- Tam, C.S.; Trotman, J.; Opat, S.; Burger, J.A.; Cull, G.; Gottlieb, D.; Harrup, R.; Johnston, P.B.; Marlton, P.; Munoz, J.; et al. Phase 1 Study of the Selective BTK Inhibitor Zanubrutinib in B-Cell Malignancies and Safety and Efficacy Evaluation in CLL. Blood 2019, 134, 851–859. [Google Scholar] [CrossRef] [Green Version]
- Debureaux, P.-E.; Nailly, D.L.F.D.; Tavernier, E.; Bedoui, M.; Kuhnowski, F.; Tamburini, J.; Fornecker, L.-M.; Camus, V.; Sibon, D.; Moles, M.-P.; et al. Sinusoidal Obstruction Syndrome: A Warning about Autologous Stem Cell Transplantation Preceded by Regimens Containing Oxaliplatin. Bone Marrow Transplant. 2020, 55, 1834–1836. [Google Scholar] [CrossRef]
- Corbacioglu, S.; Jabbour, E.J.; Mohty, M. Risk Factors for Development of and Progression of Hepatic Veno-Occlusive Dis-ease/Sinusoidal Obstruction Syndrome. Biol. Blood Marrow Transplant. 2019, 25, 1271–1280. [Google Scholar] [CrossRef] [Green Version]
- Kuruvilla, J.; Crump, M.; Villa, D.; Aslam, M.; Prica, A.; Scott, D.; Abdel-Samad, N.; Couban, S.; Doucet, S.; Dudebout, J.; et al. Canadian Cancer Trials Group (Cctg) Ly.17: A Randomized Phase Ii Study Evaluating Novel Salvage Therapy Pre-Autologous Stem Cell Transplant (Asct) In Relapsed/Refractory Diffuse Large B Cell Lymphoma (Rr-Dlbcl)—Outcome of Ibrutinib + R-Gdp. Hematol. Oncol. 2017, 35, 88. [Google Scholar] [CrossRef] [Green Version]
- Hutchings, M.; Mous, R.; Clausen, M.R.; Johnson, P.; Linton, K.M.; Chamuleau, M.E.D.; Lewis, D.J.; Balari, A.S.; Cunningham, D.; Oliveri, R.S.; et al. Dose Escalation of Subcutaneous Epcoritamab in Patients with Relapsed or Refractory B-Cell Non-Hodgkin Lymphoma: An Open-Label, Phase 1/2 Study. Lancet 2021, 398, 1157–1169. [Google Scholar] [CrossRef]
- Hutchings, M.; Morschhauser, F.; Iacoboni, G.; Carlo-Stella, C.; Offner, F.C.; Sureda, A.; Salles, G.; Martínez-Lopez, J.; Crump, M.; Thomas, D.N.; et al. Glofitamab, a Novel, Bivalent CD20-Targeting T-Cell–Engaging Bispecific Antibody, Induces Durable Complete Remissions in Relapsed or Refractory B-Cell Lymphoma: A Phase I Trial. J. Clin. Oncol. 2021, 39, 1959–1970. [Google Scholar] [CrossRef] [PubMed]
| Type of Immunochemotherapy | R-DHAP | R-DHAOx | ||
|---|---|---|---|---|
| Administration of Ibrutinib | Concomitant | Intermittent | Concomitant | Intermittent |
| D1–21 | D5–18 | D1–21 | D5–18 | |
| Group | A | A’ | B | B’ |
| Patients enrolled | 13 | 13 | 12 | 37 |
| Patients evaluable for DLT | 13 | 10 | 12 | 12 |
| Patients evaluable for safety | 13 | 12 £ | 12 | 36 ££ |
| Median age * | 63 (40–68) | 60 (49–69) | 58 (42–66) | 62 (25–70) |
| ≤60 years | 5 (38%) | 9 (75%) | 8 (67%) | 16 (44%) |
| >60 years | 8 (62%) | 3 (25%) | 4 (33%) | 20 (56%) |
| Sex | ||||
| Male | 9 (69%) | 8 (67%) | 8 (67%) | 23 (64%) |
| Female | 4 (31%) | 4 (33%) | 4 (33%) | 13 (36%) |
| Stage at inclusion | ||||
| I | 2 (15%) | 4 (33%) | 3 (25%) | 3 (8%) |
| II | 3 (23%) | 1 (8%) | 0 | 3 (8%) |
| III | 2 (15%) | 3 (25%) | 2 (17%) | 6 (17%) |
| IV | 6 (45%) | 4 (33%) | 7 (58%) | 24 (67%) |
| ECOG at inclusion | ||||
| 0 | 8 (63%) | 9 (75%) | 8 (67%) | 25 (69%) |
| 1 | 3 (23%) | 3 (25%) | 3 (25%) | 9 (25%) |
| 2 | 2 (15%) | 0 | 1 (8%) | 2 (6%) |
| LDH > upper normal limit | ||||
| No | 6 (69%) | 7 (64%) | 3 (75%) | 18 (50%) |
| Yes | 7 (31%) | 4 (36%) | 9 (25%) | 18 (50%) |
| Bone marrow | ||||
| Performed | 11 (85%) | 9 (75%) | 11 (92%) | 34 (94%) |
| Involved | 2 (18%) | 0 | 2 (18%) | 6 (79%) |
| Non-involved | 9 (82%) | 9 (100%) | 8 (73%) | 28 (82%) |
| Unspecified | 0 | 0 | 1 (9%) | 0 |
| Histology at inclusion | ||||
| DLBCL | 4 (31%) | 4 (33%) | 7 (58%) | 15 (46%) |
| Other large B-cell lymphoma † | 0 | 2 (16%) | 1 (8%) | 2 (6%) |
| Follicular lymphoma Grade 1, 2, or 3a | 2 (15%) | 4 (33%) | 2 (16%) | 11 (33%) |
| Nodal marginal zone lymphoma | 1 (8%) | 0 | 1 (8%) | 1 (3%) |
| Mantle cell lymphoma | 2 (15%) | 0 | 1 (8%) | 0 |
| Transformed indolent lymphoma ‡ | 4 (32%) | 1 (8%) | 0 | 3 (9%) |
| Missing | 0 | 2 (17%) | 0 | 4 (13%) |
| Disease status | ||||
| Refractory | 3 (23%) | 6 (50%) | 3 (25%) | 13 (36%) |
| Relapsed/progressive | 10 (77%) | 6 (50%) | 9 (75%) | 23 (64%) |
| Number of previous treatment lines | ||||
| 1 | 12 (92%) | 12 (100%) | 10 (83%) | 31 (86%) |
| 2 | 1 (8%) | 0 | 2 (17%) | 5 (14%) |
| Previous treatments | ||||
| Rituximab | 13 (100%) | 11 (92%) | 10 (83%) | 36 (100%) |
| Anthracycline-based chemotherapy | 13 (100%) | 11 (92%) | 12 (100%) | 35 (97%) |
| Radiotherapy | 1 (8%) | 0 | 3 (25%) | 1 (3%) |
| Autologous stem cell transplantation | 2 (15%) | 0 | 0 | 1 (3%) |
| Allogenic stem cell transplantation | 0 | 0 | 1 (8%) | 0 |
| Chemotherapy | R-DHAP | R-DHAOx | ||||||
|---|---|---|---|---|---|---|---|---|
| Enrolled set | 26 | 49 | ||||||
| Safety set | 25 | 48 | ||||||
| Ibrutinib administration | Concomitant | Intermittent | Concomitant | Intermittent | ||||
| Group | A | A’ | B | B’ | ||||
| Enrolled set | 13 | 13 | 12 | 37 | ||||
| Safety set | 13 | 12 | 12 | 36 | ||||
| Grade >2 AEs in >10% of patients and AESIs | Patients (%) | AE | Patients (%) | AE | Patients (%) | AE | Patients (%) | AE |
| 13 (100%) | 127 | 12 (100%) | 98 | 12 (100%) | 81 | 33 (92%) | 171 | |
| Blood and lymphatic disorders | 13 (100%) | 87 | 12 (100%) | 56 | 12 (100)% | 59 | 30 (83%) | 111 |
| Thrombocytopenia | 13 (100%) | 31 | 9 (75%) | 18 | 12 (100%) | 26 | 30 (83%) | 65 |
| Neutropenia | 11 (85%) | 18 | 6 (50%) | 9 | 5 (42%) | 8 | 12 (33%) | 17 |
| Febrile neutropenia | 6 (47%) | 8 | 3 (25%) | 3 | 3 (25%) | 3 | 3 (8%) | 4 |
| Anemia | 8 (62%) | 11 | 3 (25%) | 9 | 2 (17%) | 4 | 8 (22%) | 9 |
| Infections | 4 (31%) | 4 | 3 (25%) | 5 | 4 (33%) | 4 | 8 (22%) | 12 |
| Bacterial sepsis | 2 (15%) | 2 | ||||||
| Bronchitis | 2 (17%) | 3 | ||||||
| Hemorrhagic manifestations | 2 (15%) | 2 | 1 (3%) | 1 | ||||
| General disorders and administration site conditions | 2 (15%) | 3 | 2 (17%) | 2 | 3 (8%) | 3 | ||
| Cardiac disorders | 2 (15%) | 4 | 3 (25%) | 3 | 4 (11%) | 5 | ||
| Atrial fibrillation | 2 (15%) | 2 | 2 (17%) | 2 | 3 (8%) | 3 | ||
| Vascular disorders | 1 (8%) | 1 | 1 (8%) | 1 | ||||
| Metabolism and nutrition disorders | 6 (46%) | 9 | 1 (8%) | 1 | 2 (17%) | 2 | 6 (17%) | 11 |
| Hypocalcemia | 3 (23%) | 3 | ||||||
| Hypokalemia | 2 (15%) | 2 | 1 (8%) | 1 | 2 (6%) | 3 | ||
| Renal and urinary disorders | 7 (54%) | 7 | 8 (67%) | 8 | ||||
| Renal failure | 7 (54%) | 7 | 8 (67%) | 8 | ||||
| Gastrointestinal disorders | 4 (31%) | 5 | 2 (17%) | 3 | 6 (17%) | 7 | ||
| Diarrhea | 4 (11%) | 4 | ||||||
| Hepatobiliary disorders | 2 (17%) | 2 | 5 (14%) | 7 | ||||
| Congenital, familial, and genetic disorders | 2 (17%) | 2 | ||||||
| Aplasia | 2 (17%) | 2 | ||||||
| Nervous system disorders | 3 (25%) | 3 | 8 (22%) | 10 | ||||
| Peripheral neuropathy | 1 (8%) | 1 | 7 (19%) | 7 | ||||
| Skin and subcutaneous disorders | 2 (15%) | 2 | 2 (17%) | 2 | ||||
| Second primary neoplasias | 2 (17%) | 2 | ||||||
| Patients with at least one SAE | Patients (%) | SAE | Patients (%) | SAE | Patients (%) | SAE | Patients (%) | SAE |
| 10 (77) | 27 | 10 (77%) | 34 | 6 (50%) | 17 | 17 (47%) | 30 | |
| Infection | 3 (23%) | 4 | 2 (17%) | 2 | 5 (42%) | 6 | 6 (27%) | 10 |
| Renal failure | 4 (31%) | 4 | 8 (67%) | 8 | ||||
| Atrial fibrillation | 1 (8%) | 1 | 1 (8%) | 1 | 2 (6%) | 2 | ||
| Cutaneous eruption | 1 (8%) | 1 | 2 (17%) | 2 | ||||
| Hemorrhagic complication | 2 (17%) | 2 | 2 (6%) | 2 | ||||
| Veno-occlusive disease | 3 (8%) | 3 | ||||||
| DLT evaluable patients (n) | 13 | 10 | 12 | 12 | ||||
| Patients with DLT | 5 | 3 | 4 | 1 | ||||
| Number of DLTs | 7 | 6 | 6 | 1 | ||||
| Patients who discontinued treatment | 4 | 7 | 5 | 3 | ||||
| Treatment discontinuation due to toxicity | 4 | 2 | 2 | |||||
| Treatment discontinuation due to progression | 3 | 1 | 3 | |||||
| Treatment discontinuation due to consent withdrawal | 1 | 2 | ||||||
| Treatment discontinuation due to concurrent illness | 1 | |||||||
| R-I-DHAP | R-I-DHAOx | ||
|---|---|---|---|
| Group A: Concomitant Administration: 13 Patients | Group B: Concomitant Administration: 12 Patients | ||
| Ibrutinib 420 mg/day: 6 Patients | Ibrutinib 420 mg/day: 6 Patients | ||
| Patient 1 | Grade 4 cutaneous eruption lasting 5 days | Patient 1 | Grade 3 prostate infection lasting 11 days |
| Patient 2 | Grade 4 sepsis lasting 10 days | Grade 3 cutaneous eruption lasting 10 days | |
| Patient 3 | Grade 4 thrombocytopenia lasting 7 days £ | Patient 2 | Grade 3 cutaneous infection lasting 10 days |
| Grade 3 febrile neutropenia lasting 1 day | |||
| Patient 3 | Grade 4 thrombocytopenia lasting 14 days | ||
| Ibrutinib 280 mg/day: 7 patients | Ibrutinib 280 mg/day: 6 patients | ||
| Patient 4 | Grade 4 neutropenia lasting 12 days | Patient 4 | Grade 3 epigastric pain lasting 11 days |
| Patient 5 | Grade 4 gastric hemorrhage lasting 13 days | ||
| Grade 4 thrombocytopenia lasting 13 days | |||
| Grade 3 atrial fibrillation lasting 2 days | |||
| Group A’: Intermittent administration: 10 patients * | Group B’: Intermittent administration: 12 patients | ||
| Ibrutinib 420 mg/day: 4 patients | Ibrutinib 420 mg/day: 6 patients | ||
| No DLT | Patient 1 | Grade 3 hepatitis lasting 141 days | |
| Ibrutinib 560 mg/day: 6 patients | Ibrutinib 560 mg/day: 6 patients | ||
| Patient 1 | Grade 3 acute renal insufficiency lasting 2 days | No DLT | |
| Patient 2 | Grade 3 hyponatremia lasting 1 day | ||
| Grade 3 nausea lasting 19 days | |||
| Grade 3 hypophosphatemia lasting 1 day. | |||
| Patient 3 | Grade 3 acute renal insufficiency lasting 5 days | ||
| Grade 3 vomiting lasting 17 days | |||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bonnet, C.; Dupuis, J.; Tilly, H.; Lamy, T.; Fruchart, C.; le Gouill, S.; Thieblemont, C.; Morschhauser, F.; Casasnovas, O.; Bouabdallah, K.; et al. Ibrutinib Associated with Rituximab-Platinum Salt-Based Immunochemotherapy in B-Cell Lymphomas: Results of a Phase 1b-II Study of the LYSA Group. Cancers 2022, 14, 1761. https://doi.org/10.3390/cancers14071761
Bonnet C, Dupuis J, Tilly H, Lamy T, Fruchart C, le Gouill S, Thieblemont C, Morschhauser F, Casasnovas O, Bouabdallah K, et al. Ibrutinib Associated with Rituximab-Platinum Salt-Based Immunochemotherapy in B-Cell Lymphomas: Results of a Phase 1b-II Study of the LYSA Group. Cancers. 2022; 14(7):1761. https://doi.org/10.3390/cancers14071761
Chicago/Turabian StyleBonnet, Christophe, Jehan Dupuis, Hervé Tilly, Thierry Lamy, Christophe Fruchart, Steven le Gouill, Catherine Thieblemont, Franck Morschhauser, Olivier Casasnovas, Krimo Bouabdallah, and et al. 2022. "Ibrutinib Associated with Rituximab-Platinum Salt-Based Immunochemotherapy in B-Cell Lymphomas: Results of a Phase 1b-II Study of the LYSA Group" Cancers 14, no. 7: 1761. https://doi.org/10.3390/cancers14071761
APA StyleBonnet, C., Dupuis, J., Tilly, H., Lamy, T., Fruchart, C., le Gouill, S., Thieblemont, C., Morschhauser, F., Casasnovas, O., Bouabdallah, K., Ghesquieres, H., Van Den Neste, E., André, M., Cartron, G., & Salles, G. (2022). Ibrutinib Associated with Rituximab-Platinum Salt-Based Immunochemotherapy in B-Cell Lymphomas: Results of a Phase 1b-II Study of the LYSA Group. Cancers, 14(7), 1761. https://doi.org/10.3390/cancers14071761






