A Nanoparticle’s Journey to the Tumor: Strategies to Overcome First-Pass Metabolism and Their Limitations
Abstract
:Simple Summary
Abstract
1. Introduction
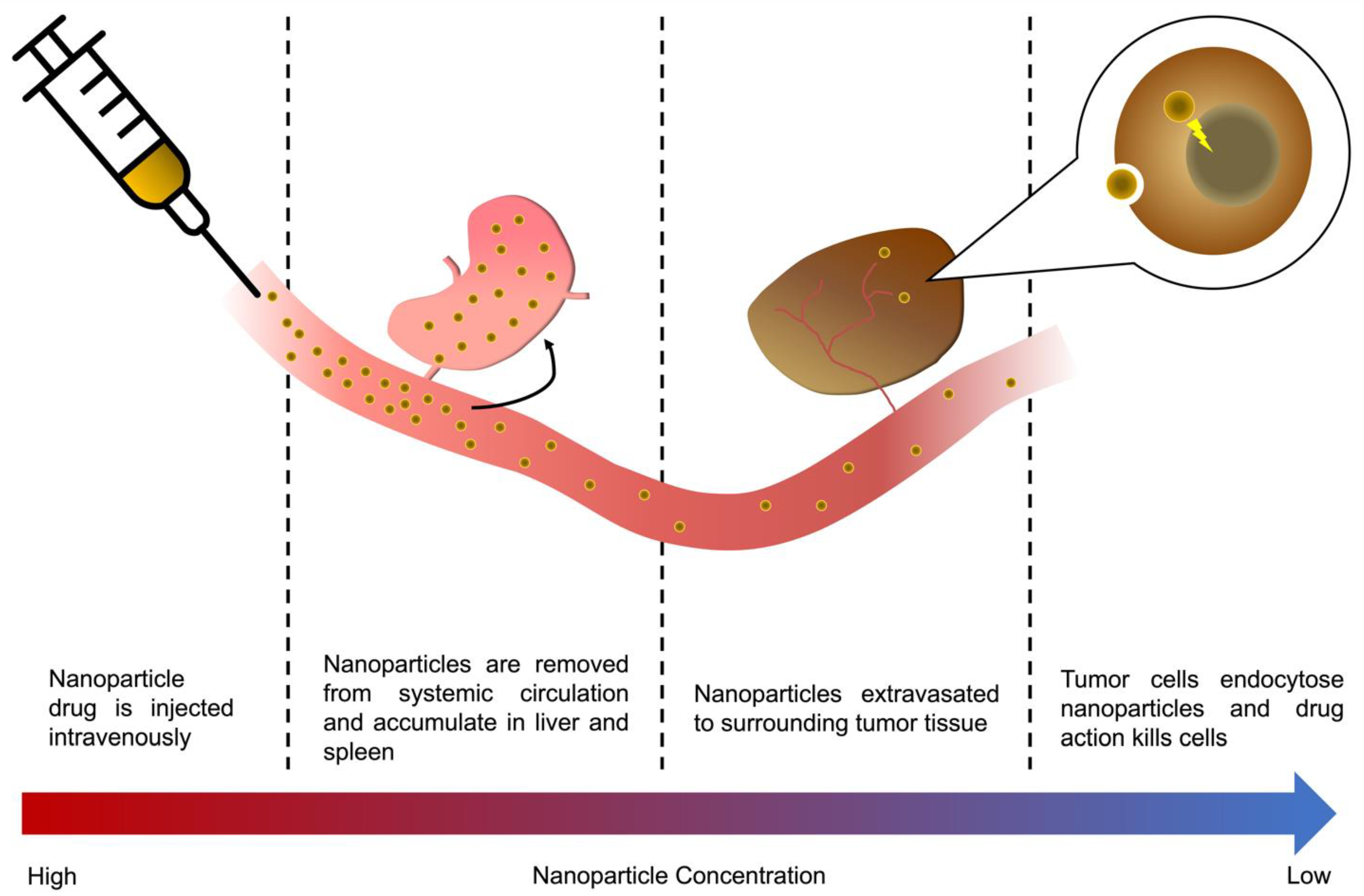
2. A Nanoparticle’s Journey to the Tumor
2.1. Barriers and Challenges of Tumor Nanoparticle Uptake
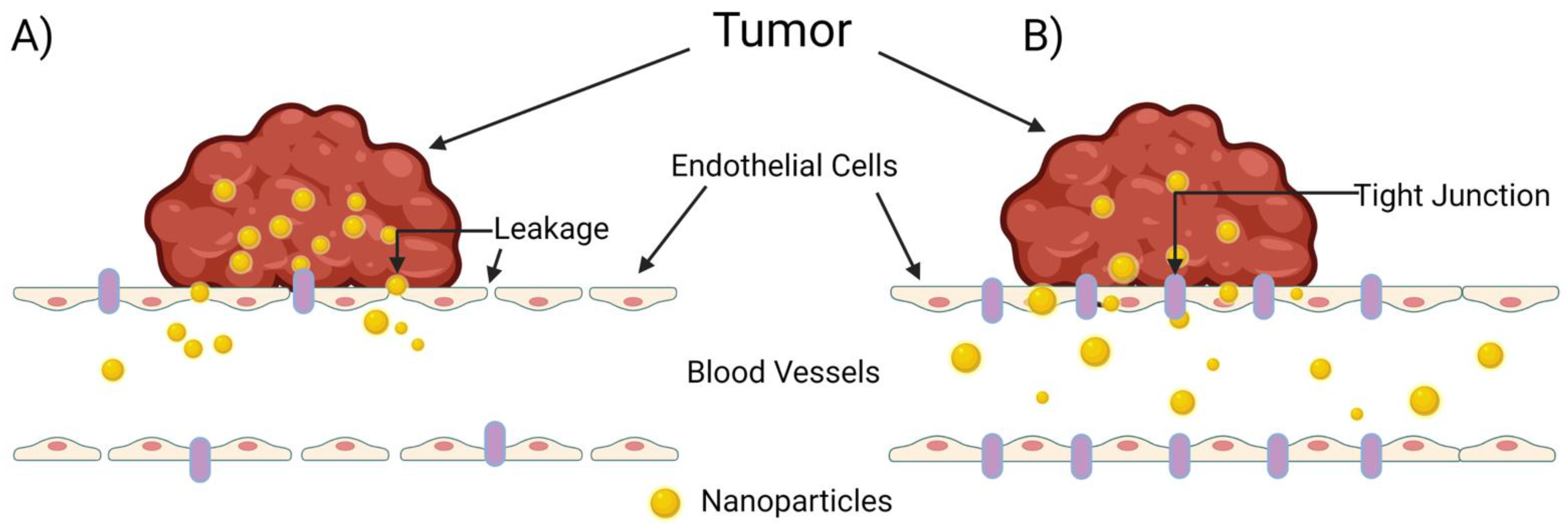
2.2. The Never-Ending Controversy of the EPR Effect
3. Role of First-Pass Organs on Nanoparticle Delivery
3.1. Fates of Different Types of Nanoparticles
3.1.1. Metallic Nanoparticles
3.1.2. Solid Lipid Nanoparticles
3.1.3. Genetically Encoded Micellar Nanoparticles
3.1.4. Other Polymeric Nanoparticles
3.1.5. Next-Generation Nanoparticle Systems
3.2. Role of Size and Surface Chemistry
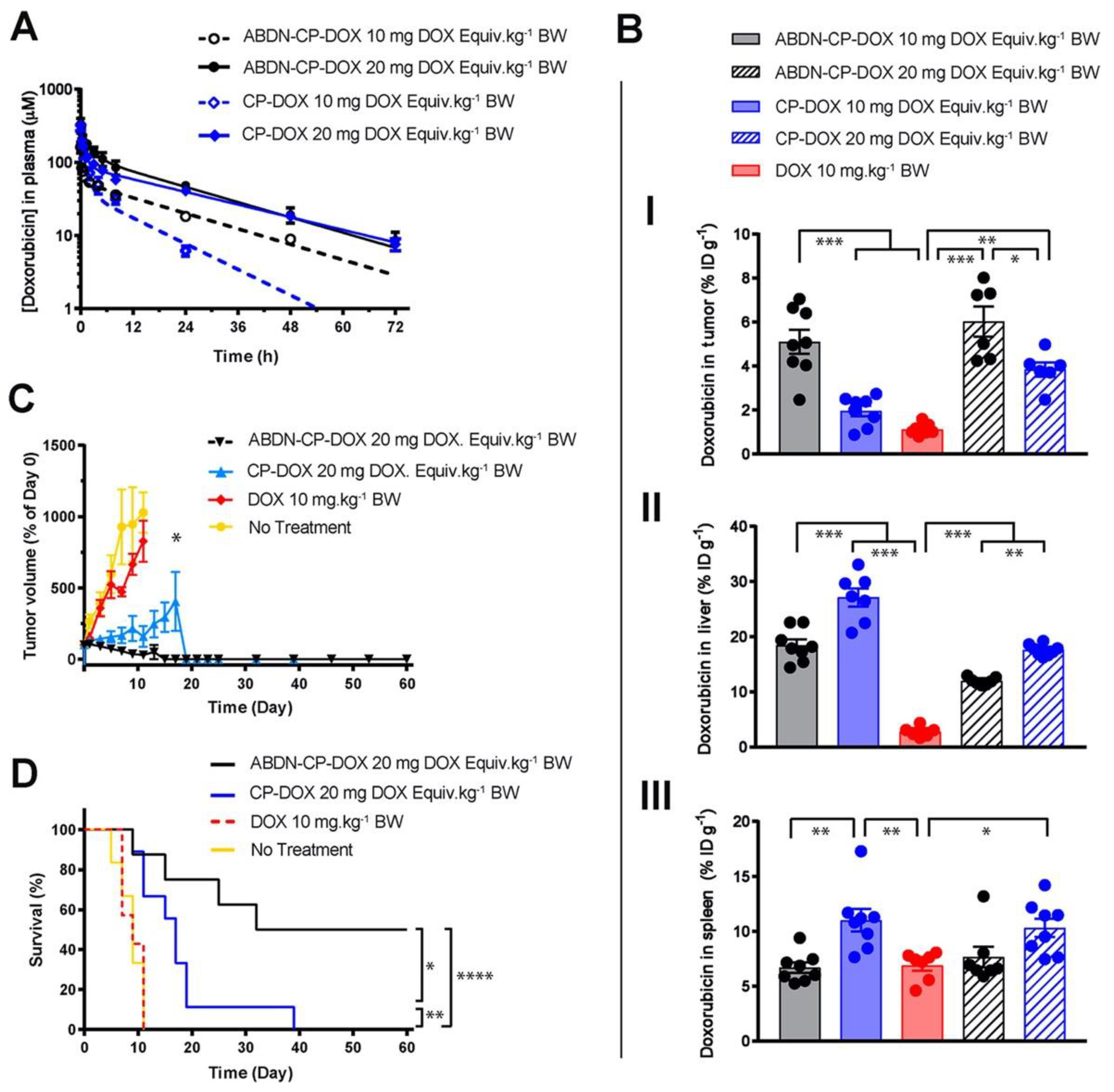
3.3. Role of Dosing
4. Understanding Opsonization of Proteins onto Nanoparticles
5. Strategies to Circumvent First-Pass Metabolism
6. Outlook and Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Salvioni, L.; Rizzuto, M.A.; Bertolini, J.A.; Pandolfi, L.; Colombo, M.; Prosperi, D. Thirty years of cancer nanomedicine: Success, frustration, and hope. Cancers 2019, 11, 1855. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Abbasi Kajani, A.; Haghjooy Javanmard, S.; Asadnia, M.; Razmjou, A. Recent Advances in Nanomaterials Development for Nanomedicine and Cancer. ACS Appl. Biol. Mater. 2021, 4, 5908–5925. [Google Scholar] [CrossRef] [PubMed]
- Wakaskar, R.R. Promising effects of nanomedicine in cancer drug delivery. J. Drug. Target. 2018, 26, 319–324. [Google Scholar] [CrossRef]
- Wakaskar, R.R. Brief overview of nanoparticulate therapy in cancer. J. Drug. Target. 2018, 26, 123–126. [Google Scholar] [CrossRef] [PubMed]
- Milligan, J.J.; Saha, S.; Jenkins, I.C.; Chilkoti, A. Genetically encoded elastin-like polypeptide nanoparticles for drug delivery. Curr. Opin. Biotechnol. 2022, 74, 146–153. [Google Scholar] [CrossRef]
- Varanko, A.; Saha, S.; Chilkoti, A. Recent trends in protein and peptide-based biomaterials for advanced drug delivery. Adv. Drug. Deliv. Rev. 2020, 156, 133–187. [Google Scholar] [CrossRef] [PubMed]
- Barui, S.; Saha, S.; Mondal, G.; Haseena, S.; Chaudhuri, A. Simultaneous delivery of doxorubicin and curcumin encapsulated in liposomes of pegylated RGDK-lipopeptide to tumor vasculature. Biomaterials 2014, 35, 1643–1656. [Google Scholar] [CrossRef]
- Barui, S.; Saha, S.; Yakati, V.; Chaudhuri, A. Systemic Codelivery of a Homoserine Derived Ceramide Analogue and Curcumin to Tumor Vasculature Inhibits Mouse Tumor Growth. Mol. Pharm. 2016, 13, 404–419. [Google Scholar] [CrossRef]
- Mondal, G.; Barui, S.; Saha, S.; Chaudhuri, A. Tumor growth inhibition through targeting liposomally bound curcumin to tumor vasculature. J. Control. Release 2013, 172, 832–840. [Google Scholar] [CrossRef]
- Saha, S.; Venu, Y.; Bhattacharya, D.; Kompella, S.D.; Madhusudana, K.; Chakravarty, S.; Ramakrishna, S.; Chaudhuri, A. Combating Established Mouse Glioblastoma through Nicotinylated-Liposomes-Mediated Targeted Chemotherapy in Combination with Dendritic-Cell-Based Genetic Immunization. Adv. Biosyst. 2017, 1, 1600009. [Google Scholar] [CrossRef]
- Saha, S.; Yakati, V.; Shankar, G.; Jaggarapu, M.M.C.S.; Moku, G.; Madhusudana, K.; Banerjee, R.; Ramkrishna, S.; Srinivas, R.; Chaudhuri, A. Amphetamine decorated cationic lipid nanoparticles cross the blood-brain barrier: Therapeutic promise for combating glioblastoma. J. Mater. Chem. B 2020, 8, 4318–4330. [Google Scholar] [CrossRef] [PubMed]
- Bailly, A.-L.; Correard, F.; Popov, A.; Tselikov, G.; Chaspoul, F.; Appay, R.; Al-Kattan, A.; Kabashin, A.V.; Braguer, D.; Esteve, M.-A. In vivo evaluation of safety, biodistribution and pharmacokinetics of laser-synthesized gold nanoparticles. Sci. Rep. 2019, 9, 12890. [Google Scholar] [CrossRef] [Green Version]
- Walkey, C.D.; Olsen, J.B.; Guo, H.; Emili, A.; Chan, W.C.W. Nanoparticle Size and Surface Chemistry Determine Serum Protein Adsorption and Macrophage Uptake. J. Am. Chem. Soc. 2012, 134, 2139–2147. [Google Scholar] [CrossRef] [PubMed]
- Muntoni, E.; Martina, K.; Marini, E.; Giorgis, M.; Lazzarato, L.; Salaroglio, I.C.; Riganti, C.; Lanotte, M.; Battaglia, L. Methotrexate-Loaded Solid Lipid Nanoparticles: Protein Functionalization to Improve Brain Biodistribution. Pharmaceutics 2019, 11, 65. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chirio, D.; Peira, E.; Dianzani, C.; Muntoni, E.; Gigliotti, C.L.; Ferrara, B.; Sapino, S.; Chindamo, G.; Gallarate, M. Development of Solid Lipid Nanoparticles by Cold Dilution of Microemulsions: Curcumin Loading, Preliminary In Vitro Studies, and Biodistribution. Nanomaterials 2019, 9, 230. [Google Scholar] [CrossRef] [Green Version]
- Banskota, S.; Saha, S.; Bhattacharya, J.; Kirmani, N.; Yousefpour, P.; Dzuricky, M.; Zakharov, N.; Li, X.; Spasojevic, I.; Young, K.; et al. Genetically Encoded Stealth Nanoparticles of a Zwitterionic Polypeptide-Paclitaxel Conjugate Have a Wider Therapeutic Window than Abraxane in Multiple Tumor Models. Nano Lett. 2020, 20, 2396–2409. [Google Scholar] [CrossRef]
- Wang, J.; Saha, S.; Schaal, J.L.; Yousefpour, P.; Li, X.; Chilkoti, A. Heuristics for the Optimal Presentation of Bioactive Peptides on Polypeptide Micelles. Nano Lett. 2019, 19, 7977–7987. [Google Scholar] [CrossRef]
- Saha, S.; Banskota, S.; Roberts, S.; Kirmani, N.; Chilkoti, A. Engineering the Architecture of Elastin-Like Polypeptides: From Unimers to Hierarchical Self-Assembly. Adv. Ther. 2020, 3, 1900164. [Google Scholar] [CrossRef]
- Luginbuhl, K.M.; Mozhdehi, D.; Dzuricky, M.; Yousefpour, P.; Huang, F.C.; Mayne, N.R.; Buehne, K.L.; Chilkoti, A. Recombinant Synthesis of Hybrid Lipid–Peptide Polymer Fusions that Self-Assemble and Encapsulate Hydrophobic Drugs. Angew. Chem. Int. Ed. 2017, 56, 13979–13984. [Google Scholar] [CrossRef]
- Carvalho, M.R.; Reis, R.L.; Oliveira, J.M. Dendrimer nanoparticles for colorectal cancer applications. J. Mater. Chem. B 2020, 8, 1128–1138. [Google Scholar] [CrossRef]
- Salimi, M.; Sarkar, S.; Fathi, S.; Alizadeh, A.M.; Saber, R.; Moradi, F.; Delavari, H. Biodistribution, pharmacokinetics, and toxicity of dendrimer-coated iron oxide nanoparticles in BALB/c mice. Int. J. Nanomed. 2018, 13, 1483–1493. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rafiei, P.; Haddadi, A. Docetaxel-loaded PLGA and PLGA-PEG nanoparticles for intravenous application: Pharmacokinetics and biodistribution profile. Int. J. Nanomed. 2017, 12, 935–947. [Google Scholar] [CrossRef] [Green Version]
- Petersen, G.H.; Alzghari, S.K.; Chee, W.; Sankari, S.S.; La-Beck, N.M. Meta-analysis of clinical and preclinical studies comparing the anticancer efficacy of liposomal versus conventional non-liposomal doxorubicin. J. Control. Release 2016, 232, 255–264. [Google Scholar] [CrossRef]
- de Lázaro, I.; Mooney, D.J. A nanoparticle’s pathway into tumours. Nat. Mater. 2020, 19, 486–487. [Google Scholar] [CrossRef] [PubMed]
- Wilhelm, S.; Tavares, A.J.; Dai, Q.; Ohta, S.; Audet, J.; Dvorak, H.F.; Chan, W.C.W. Analysis of nanoparticle delivery to tumours. Nat. Rev. Mater. 2016, 1, 16014. [Google Scholar]
- Dawidczyk, C.M.; Kim, C.; Park, J.H.; Russell, L.M.; Lee, K.H.; Pomper, M.G.; Searson, P.C. State-of-the-art in design rules for drug delivery platforms: Lessons learned from FDA-approved nanomedicines. J. Control. Release 2014, 187, 133–144. [Google Scholar] [CrossRef] [Green Version]
- Pond, S.M.; Tozer, T.N. First-Pass Elimination Basic Concepts and Clinical Consequences. Clin. Pharm. 1984, 9, 1–25. [Google Scholar] [CrossRef]
- Herman, T.F.; Santos, C. First Pass Effect. xPharm Compr. Pharm. Ref. 2021, 1–2. [Google Scholar] [CrossRef]
- Tam, Y.K. Individual variation in first-pass metabolism. Clin. Pharm. 1993, 25, 300–328. [Google Scholar] [CrossRef]
- Nel, A.E.; Mädler, L.; Velegol, D.; Xia, T.; Hoek, E.M.V.; Somasundaran, P.; Klaessig, F.; Castranova, V.; Thompson, M. Understanding biophysicochemical interactions at the nano–bio interface. Nat. Mater. 2009, 8, 543–557. [Google Scholar] [CrossRef]
- Cedervall, T.; Lynch, I.; Lindman, S.; Berggård, T.; Thulin, E.; Nilsson, H.; Dawson, K.A.; Linse, S. Understanding the nanoparticle–protein corona using methods to quantify exchange rates and affinities of proteins for nanoparticles. Proc. Natl. Acad. Sci. USA 2007, 104, 2050–2055. [Google Scholar] [CrossRef] [Green Version]
- Li, H.; Wang, Y.; Tang, Q.; Yin, D.; Tang, C.; He, E.; Zou, L.; Peng, Q. The protein corona and its effects on nanoparticle-based drug delivery systems. Acta Biomater. 2021, 129, 57–72. [Google Scholar] [CrossRef]
- Wani, T.U.; Raza, S.N.; Khan, N.A. Nanoparticle opsonization: Forces involved and protection by long chain polymers. Polym. Bull. 2019, 77, 3865–3889. [Google Scholar] [CrossRef]
- Hui, Y.; Yi, X.; Hou, F.; Wibowo, D.; Zhang, F.; Zhao, D.; Gao, H.; Zhao, C.-X. Role of Nanoparticle Mechanical Properties in Cancer Drug Delivery. ACS Nano 2019, 13, 7410–7424. [Google Scholar] [CrossRef]
- Sindhwani, S.; Syed, A.M.; Ngai, J.; Kingston, B.R.; Maiorino, L.; Rothschild, J.; MacMillan, P.; Zhang, Y.; Rajesh, N.U.; Hoang, T.; et al. The entry of nanoparticles into solid tumours. Nat. Mater. 2020, 19, 566–575. [Google Scholar] [CrossRef]
- Sanità, G.; Carrese, B.; Lamberti, A. Nanoparticle Surface Functionalization: How to Improve Biocompatibility and Cellular Internalization. Front. Mol. Biosci. 2020, 7, 587012. [Google Scholar] [CrossRef]
- Kalyane, D.; Raval, N.; Maheshwari, R.; Tambe, V.; Kalia, K.; Tekade, R.K. Employment of enhanced permeability and retention effect (EPR): Nanoparticle-based precision tools for targeting of therapeutic and diagnostic agent in cancer. Mater. Sci. Eng. C 2019, 98, 1252–1276. [Google Scholar] [CrossRef]
- Iyer, A.K.; Khaled, G.; Fang, J.; Maeda, H. Exploiting the enhanced permeability and retention effect for tumor targeting. Drug. Discov. Today 2006, 11, 812–818. [Google Scholar] [CrossRef]
- Nakamura, Y.; Mochida, A.; Choyke, P.L.; Kobayashi, H. Nanodrug Delivery: Is the Enhanced Permeability and Retention Effect Sufficient for Curing Cancer? Bioconjug. Chem. 2016, 27, 2225–2238. [Google Scholar] [CrossRef]
- Danhier, F. To exploit the tumor microenvironment: Since the EPR effect fails in the clinic, what is the future of nanomedicine? J. Control. Release 2016, 244, 108–121. [Google Scholar] [CrossRef]
- Shi, Y.; van der Meel, R.; Chen, X.; Lammers, T. The EPR effect and beyond: Strategies to improve tumor targeting and cancer nanomedicine treatment efficacy. Theranostics 2020, 10, 7921–7924. [Google Scholar] [CrossRef]
- Liu, X.; Jiang, J.; Meng, H. Transcytosis—An effective targeting strategy that is complementary to “EPR effect” for pancreatic cancer nano drug delivery. Theranostics 2019, 9, 8018–8025. [Google Scholar] [CrossRef]
- Takeuchi, I.; Nobata, S.; Oiri, N.; Tomoda, K.; Makino, K. Biodistribution and excretion of colloidal gold nanoparticles after intravenous injection: Effects of particle size. Biomed. Mater. Eng. 2017, 28, 315–323. [Google Scholar] [CrossRef]
- Abdel Fattah, W.I.; Ali, G.W. On the anti-cancer activities of silver nanoparticles. J. Appl. Biotechnol. Bioeng. 2018, 5, 43–46. [Google Scholar] [CrossRef] [Green Version]
- Souza, T.A.J.; Franchi, L.P.; Rosa, L.R.; da Veiga, M.A.M.S.; Takahashi, C.S. Cytotoxicity and genotoxicity of silver nanoparticles of different sizes in CHO-K1 and CHO-XRS5 cell lines. Mutat. Res. Genet. Toxicol. Environ. Mutagen. 2016, 795, 70–83. [Google Scholar] [CrossRef]
- Gopisetty, M.K.; Kovács, D.; Igaz, N.; Rónavári, A.; Bélteky, P.; Rázga, Z.; Venglovecz, V.; Csoboz, B.; Boros, I.M.; Kónya, Z.; et al. Endoplasmic reticulum stress: Major player in size-dependent inhibition of P-glycoprotein by silver nanoparticles in multidrug-resistant breast cancer cells. J. Nanobiotechnol. 2019, 17, 9. [Google Scholar] [CrossRef]
- Chugh, H.; Sood, D.; Chandra, I.; Tomar, V.; Dhawan, G.; Chandra, R. Role of gold and silver nanoparticles in cancer nano-medicine. Artif. Cells Nanomed. Biotechnol. 2018, 46, 1210–1220. [Google Scholar] [CrossRef]
- Alphandéry, E. Biodistribution and targeting properties of iron oxide nanoparticles for treatments of cancer and iron anemia disease. Nanotoxicology 2019, 13, 573–596. [Google Scholar] [CrossRef]
- Bai, D.-P.; Zhang, X.-F.; Zhang, G.-L.; Huang, Y.-F.; Gurunathan, S. Zinc oxide nanoparticles induce apoptosis and autophagy in human ovarian cancer cells. Int. J. Nanomed. 2017, 12, 6521–6535. [Google Scholar] [CrossRef] [Green Version]
- Ancona, A.; Dumontel, B.; Garino, N.; Demarco, B.; Chatzitheodoridou, D.; Fazzini, W.; Engelke, H.; Cauda, V. Lipid-Coated Zinc Oxide Nanoparticles as Innovative ROS-Generators for Photodynamic Therapy in Cancer Cells. Nanomater 2018, 8, 143. [Google Scholar] [CrossRef] [Green Version]
- Çeşmeli, S.; Biray Avci, C. Application of titanium dioxide (TiO2) nanoparticles in cancer therapies. J. Drug. Target. 2019, 27, 762–766. [Google Scholar] [CrossRef] [PubMed]
- Kongseng, S.; Yoovathaworn, K.; Wongprasert, K.; Chunhabundit, R.; Sukwong, P.; Pissuwan, D. Cytotoxic and inflammatory responses of TiO2 nanoparticles on human peripheral blood mononuclear cells. J. Appl. Toxicol. 2016, 36, 1364–1373. [Google Scholar] [CrossRef] [PubMed]
- Ziental, D.; Czarczynska-Goslinska, B.; Mlynarczyk, D.T.; Glowacka-Sobotta, A.; Stanisz, B.; Goslinski, T.; Sobotta, L. Titanium Dioxide Nanoparticles: Prospects and Applications in Medicine. Nanomater 2020, 10, 387. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Andrew MacKay, J.; Chen, M.; McDaniel, J.R.; Liu, W.; Simnick, A.J.; Chilkoti, A. Self-assembling chimeric polypeptide–doxorubicin conjugate nanoparticles that abolish tumours after a single injection. Nat. Mater. 2009, 8, 993–999. [Google Scholar] [CrossRef] [Green Version]
- Bhattacharyya, J.; Bellucci, J.J.; Weitzhandler, I.; McDaniel, J.R.; Spasojevic, I.; Li, X.; Lin, C.-C.; Chi, J.-T.A.; Chilkoti, A. A paclitaxel-loaded recombinant polypeptide nanoparticle outperforms Abraxane in multiple murine cancer models. Nat. Commun. 2015, 6, 7939. [Google Scholar] [CrossRef]
- Yousefpour, P.; McDaniel, J.R.; Prasad, V.; Ahn, L.; Li, X.; Subrahmanyan, R.; Weitzhandler, I.; Suter, S.; Chilkoti, A. Genetically Encoding Albumin Binding into Chemotherapeutic-loaded Polypeptide Nanoparticles Enhances Their Antitumor Efficacy. Nano. Lett. 2018, 18, 7784–7793. [Google Scholar] [CrossRef]
- Singh, S.K.; Gordetsky, J.B.; Bae, S.; Acosta, E.P.; Lillard, J.W.; Singh, R. Selective Targeting of the Hedgehog Signaling Pathway by PBM Nanoparticles in Docetaxel-Resistant Prostate Cancer. Cells 2020, 9, 1976. [Google Scholar] [CrossRef]
- Mukherjee, S.; Kotcherlakota, R.; Haque, S.; Das, S.; Nuthi, S.; Bhattacharya, D.; Madhusudana, K.; Chakravarty, S.; Sistla, R.; Patra, C.R. Silver Prussian Blue Analogue Nanoparticles: Rationally Designed Advanced Nanomedicine for Multifunctional Biomedical Applications. ACS Biomater. Sci. Eng. 2020, 6, 690–704. [Google Scholar] [CrossRef]
- Mukherjee, S.; Kotcherlakota, R.; Haque, S.; Bhattacharya, D.; Kumar, J.M.; Chakravarty, S.; Patra, C.R. Improved delivery of doxorubicin using rationally designed PEGylated platinum nanoparticles for the treatment of melanoma. Mater. Sci. Eng. C 2020, 108, 110375. [Google Scholar] [CrossRef]
- Pan, G.; Jia, T.; Huang, Q.; Qiu, Y.; Xu, J.; Yin, P.; Liu, T. Mesoporous silica nanoparticles (MSNs)-based organic/inorganic hybrid nanocarriers loading 5-Fluorouracil for the treatment of colon cancer with improved anticancer efficacy. Colloids Surf. B Biointerfaces 2017, 159, 375–385. [Google Scholar] [CrossRef]
- Abedin, M.R.; Powers, K.; Aiardo, R.; Barua, D.; Barua, S. Antibody–drug nanoparticle induces synergistic treatment efficacies in HER2 positive breast cancer cells. Sci. Rep. 2021, 11, 7347. [Google Scholar] [CrossRef] [PubMed]
- Papi, M.; Caputo, D.; Palmieri, V.; Coppola, R.; Palchetti, S.; Bugli, F.; Martini, C.; Digiacomo, L.; Pozzi, D.; Caracciolo, G. Clinically approved PEGylated nanoparticles are covered by a protein corona that boosts the uptake by cancer cells. Nanoscale 2017, 9, 10327–10334. [Google Scholar] [CrossRef] [PubMed]
- Ouyang, B.; Poon, W.; Zhang, Y.N.; Lin, Z.P.; Kingston, B.R.; Tavares, A.J.; Zhang, Y.; Chen, J.; Valic, M.S.; Syed, A.M.; et al. The dose threshold for nanoparticle tumour delivery. Nat. Mater. 2020, 19, 1362–1371. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, V.H.; Lee, B.J. Protein corona: A new approach for nanomedicine design. Int. J. Nanomed. 2017, 12, 3137–3151. [Google Scholar] [CrossRef] [Green Version]
- Schöttler, S.; Landfester, K.; Mailänder, V. Controlling the Stealth Effect of Nanocarriers through Understanding the Protein Corona. Angew. Chem. Int. Ed. 2016, 55, 8806–8815. [Google Scholar] [CrossRef]
- Papini, E.; Tavano, R.; Mancin, F. Opsonins and Dysopsonins of Nanoparticles: Facts, Concepts, and Methodological Guidelines. Front. Immunol. 2020, 11, 2343. [Google Scholar] [CrossRef]
- Baimanov, D.; Cai, R.; Chen, C. Understanding the Chemical Nature of Nanoparticle–Protein Interactions. Bioconjug. Chem. 2019, 30, 1923–1937. [Google Scholar] [CrossRef]
- Hatziantoniou, S.; Galatou, E.; Bakrania, A.; Zheng, G.; Bhat, M. Nanomedicine in Hepatocellular Carcinoma: A New Frontier in Targeted Cancer Treatment. Pharmaceutics 2021, 14, 41. [Google Scholar]
- Ong, Y.R.; De Rose, R.; Johnston, A.P.R. In Vivo Quantification of Nanoparticle Association with Immune Cell Subsets in Blood. Adv. Health Mater. 2021, 10, 2002160. [Google Scholar] [CrossRef]
- Caruso, F.; Hyeon, T.; Rotello, V.; Walkey, C.D.; Chan, W.C.W. Understanding and controlling the interaction of nanomaterials with proteins in a physiological environment. Chem. Soc. Rev. 2012, 41, 2780–2799. [Google Scholar]
- Deng, Z.J.; Liang, M.; Monteiro, M.; Toth, I.; Minchin, R.F. Nanoparticle-induced unfolding of fibrinogen promotes Mac-1 receptor activation and inflammation. Nat. Nanotechnol. 2010, 6, 39–44. [Google Scholar] [CrossRef] [PubMed]
- Shah, N.B.; Vercellotti, G.M.; White, J.G.; Fegan, A.; Wagner, C.R.; Bischof, J.C. Blood-nanoparticle interactions and in Vivo biodistribution: Impact of surface peg and ligand properties. Mol. Pharm. 2012, 9, 2146–2155. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vogt, C.; Pernemalm, M.; Kohonen, P.; Laurent, S.; Hultenby, K.; Vahter, M.; Lehtiö, J.; Toprak, M.S.; Fadeel, B. Proteomics Analysis Reveals Distinct Corona Composition on Magnetic Nanoparticles with Different Surface Coatings: Implications for Interactions with Primary Human Macrophages. PLoS ONE 2015, 10, e0129008. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mozar, F.S.; Chowdhury, E.H. Impact of PEGylated Nanoparticles on Tumor Targeted Drug Delivery. Curr. Pharm. Des. 2018, 24, 3283–3296. [Google Scholar] [CrossRef] [PubMed]
- Hennig, R.; Pollinger, K.; Veser, A.; Breunig, M.; Goepferich, A. Nanoparticle multivalency counterbalances the ligand affinity loss upon PEGylation. J. Control. Release 2014, 194, 20–27. [Google Scholar] [CrossRef]
- Garay, R.P.; El-Gewely, R.; Armstrong, J.K.; Garratty, G.; Richette, P. Antibodies against polyethylene glycol in healthy subjects and in patients treated with PEG-conjugated agents. Expert. Opin. Drug. Deliv. 2012, 9, 1319–1323. [Google Scholar] [CrossRef]
- Povsic, T.J.; Lawrence, M.G.; Lincoff, A.M.; Mehran, R.; Rusconi, C.P.; Zelenkofske, S.L.; Huang, Z.; Sailstad, J.; Armstrong, P.W.; Steg, P.G.; et al. Pre-existing anti-PEG antibodies are associated with severe immediate allergic reactions to pegnivacogin, a PEGylated aptamer. J. Allergy Clin. Immunol. 2016, 138, 1712–1715. [Google Scholar] [CrossRef] [Green Version]
- Hong, L.; Wang, Z.; Wei, X.; Shi, J.; Li, C. Antibodies against polyethylene glycol in human blood: A literature review. J. Pharmacol. Toxicol. Methods 2020, 102, 106678. [Google Scholar] [CrossRef]
- FDA. Immunogenicity Assessment for Therapeutic Protein Products; U.S. Food and Drug Administration: Silver Spring, MD, USA, 2014. [Google Scholar]
- Ozer, I.; Kelly, G.; Gu, R.; Li, X.; Zakharov, N.; Sirohi, P.; Nair, S.K.; Collier, J.H.; Hershfield, M.S.; Hucknall, A.M.; et al. Polyethylene Glycol-Like Brush Polymer Conjugate of a Protein Drug Does Not Induce an Antipolymer Immune Response and Has Enhanced Pharmacokinetics than Its Polyethylene Glycol Counterpart. Adv. Sci. 2022, 2103672, 2103672. [Google Scholar] [CrossRef]
- Proffitt, R.T.; Williams, L.E.; Presant, C.A.; Tin, G.W.; Uliana, J.A.; Gamble, R.C.; Baldeschwieler, J.D. Liposomal blockade of the reticuloendothelial system: Improved tumor imaging with small unilamellar vesicles. Science 1983, 220, 502–505. [Google Scholar] [CrossRef]
- Liu, T.; Choi, H.; Zhou, R.; Chen, I.W. RES blockade: A strategy for boosting efficiency of nanoparticle drug. Nano Today 2015, 10, 11–21. [Google Scholar] [CrossRef]
- Liu, F.; Han, L.; Huang, X.; Sang, M.; Liu, B.; Li, C.; Ma, C.; Liu, W.; Feng, F.; Qu, W. Reticuloendothelial System Pre-Block Strategy to Improve Tumor Targeting Efficacy for Hyaluronic Acid Related Drug Delivery System. J. Biomed. Nanotechnol. 2018, 14, 1731–1743. [Google Scholar] [CrossRef] [PubMed]
- Diagaradjane, P.; Deorukhkar, A.; Gelovani, J.G.; Maru, D.M.; Krishnan, S. Gadolinium chloride augments tumor-specific imaging of targeted quantum dots in vivo. ACS Nano 2010, 4, 4131–4141. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pelt, J.; Busatto, S.; Ferrari, M.; Thompson, E.A.; Mody, K.; Wolfram, J. Chloroquine and nanoparticle drug delivery: A promising combination. Pharmacol. Ther. 2018, 191, 43–49. [Google Scholar] [CrossRef] [PubMed]
- Opperman, K.S.; Vandyke, K.; Clark, K.C.; Coulter, E.A.; Hewett, D.R.; Mrozik, K.M.; Schwarz, N.; Evdokiou, A.; Croucher, P.I.; Psaltis, P.J.; et al. Clodronate-Liposome Mediated Macrophage Depletion Abrogates Multiple Myeloma Tumor Establishment In Vivo. Neoplasia 2019, 21, 777. [Google Scholar] [CrossRef]
- Sarkar, S.; Khan, M.F.; Kaphalia, B.S.; Ansari, G.A.S. Methyl palmitate inhibits lipopolysaccharide-stimulated phagocytic activity of rat peritoneal macrophages. J. Biochem. Mol. Toxicol. 2006, 20, 302–308. [Google Scholar] [CrossRef]
- Gemsa, D.; Seitz, M.; Kramer, W.; Till, G.; Resch, K. The Effects of Phagocytosis, Dextran Sulfate, and Cell Damage on PGE 1 Sensitivity and PGE 1 Production of Macrophages. J. Immunol. 1978, 120, 1187–1194. [Google Scholar]
- Magaña, I.B.; Yendluri, R.B.; Adhikari, P.; Goodrich, G.P.; Schwartz, J.A.; Sherer, E.A.; O’Neal, D.P. Suppression of the reticuloendothelial system using λ-carrageenan to prolong the circulation of gold nanoparticles. Ther. Deliv. 2015, 6, 777–783. [Google Scholar] [CrossRef]
- Tavares, A.J.; Poon, W.; Zhang, Y.N.; Dai, Q.; Besla, R.; Ding, D.; Ouyang, B.; Li, A.; Chen, J.; Zheng, G.; et al. Effect of removing Kupffer cells on nanoparticle tumor delivery. Proc. Natl. Acad. Sci. USA 2017, 114, E10871–E10880. [Google Scholar] [CrossRef] [Green Version]
- Tang, Y.; Wang, X.; Li, J.; Nie, Y.; Liao, G.; Yu, Y.; Li, C. Overcoming the Reticuloendothelial System Barrier to Drug Delivery with a “don’t-Eat-Us” Strategy. ACS Nano 2019, 13, 13015–13026. [Google Scholar] [CrossRef]
| Phase | Clinical Trial Number | Nanoparticle Type | Active Payload | Conditions |
|---|---|---|---|---|
| Phase 4 | NCT04258631 | Liposome | Bupivacaine | Malignant female reproductive system neoplasms |
| Phase 3 | NCT04033354 | Nab-paclitaxel | Paclitaxel | Squamous non-small cell lung cancer |
| NCT00785291 | Nab-paclitaxel | Paclitaxel | - | |
| NCT00108735 | Paclitaxel-polyglumex | Fallopian tube and ovarian cancer | ||
| NCT03768414 | Nab-paclitaxel | Paclitaxel | Bile duct and gallbladder cancer | |
| NCT02839707 | Pegylated Liposome | Doxorubicin | Fallopian tube and ovarian cancer | |
| NCT02580058 | Pegylated Liposome | Doxorubicin | Ovarian cancer | |
| NCT03197935 | Nab-paclitaxel | Paclitaxel | Triple-negative breast cancer | |
| NCT03941093 | Nab-paclitaxel | Paclitaxel | Non-resectable pancreatic cancer | |
| NCT03088813 | Liposome | Irinotecan | Small cell lung cancer | |
| NCT02101788 | Pegylated Liposome | Doxorubicin | Borderline ovarian serous tumors | |
| NCT03257033 | Nab-paclitaxel | Paclitaxel | Locally advanced pancreatic cancer | |
| NCT04895358 | Nab-paclitaxel | Paclitaxel | Breast neoplasms | |
| Pegylated Liposome | Doxorubicin | |||
| NCT01964430 | Nab-paclitaxel | Paclitaxel | Pancreatic neoplasms |
| Nanoparticle Class | Advantages | Disadvantages |
|---|---|---|
| Gold Nanoparticles (AuNPs) | Strong biocompatibility | Chemical contaminants from synthesis can cause toxicity issues |
| Established delivery platform for a variety of cancer drugs | Less direct anti-cancer effects than other nanoparticle materials | |
| Silver Nanoparticles (AgNPs) | Good biocompatibility | Size-dependent cytotoxicity requires tuning of particle size |
| Direct anti-cancer cell killing capability | Potential off-target effects with little delivery to the tumor | |
| Iron Oxide Nanoparticles (IONPs) | Ability to direct uptake through external magnetic stimulation | Active targeting requires significant research to achieve clinical utility |
| Can be functionalized with ligands to enhance active targeting | ||
| Zinc Oxide Nanoparticles (ZnONPs) | Innate action on molecular pathways inducing ROS, cytokine and chemokine secretion, and cancer cell apoptosis | Off-target effects with poor tumor accumulation must still be addressed in vivo |
| Cytotoxic effects can be tied to external stimulation, such as UV light | ||
| Titanium Dioxide Nanoparticles (TiO2NPs) | Similar direct cytotoxicity mechanisms as ZnONPs, through ROS generation and DNA damage to cancer cells | NPs frequently accumulate in RES organs are cleared through the renal system before significant tumor accumulation |
| Nanoparticle Class | Advantages | Disadvantages |
|---|---|---|
| Solid Lipid Nanoparticles (SLNPs) | Controlled synthesis using oil-in-water microemulsions | Majority of SLNPs accumulate in liver and spleen tissue |
| Optimal for loading of lipophilic agents | ||
| Micellar Chimeric Polypeptide Nanoparticles (CP-NPs) | Significant anti-cancer toxicity with minimal off-target toxicity | Synthesis of CP-NPs requires synthesis in Escherichia coli and purification of endotoxin prior to administration |
| Ease of synthesis with targeting domains and peptides encoded at the gene level into a fusion protein with the CP-NPs | ||
| Dendrimers | Ability to synthesize nanoparticles with targeting ligands for cancer therapies | Accumulation in kidney tissue and likely RES organs reduces anti-cancer efficacy |
| PEG-PLGA Nanoparticles | Significantly less accumulation in liver, kidney, heart, and lung tissue than other nanoparticle systems | Allergic reactions due to anti-PEG antibodies may limit widespread use |
| Simple synthesis and encapsulation of chemotherapeutics |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Milligan, J.J.; Saha, S. A Nanoparticle’s Journey to the Tumor: Strategies to Overcome First-Pass Metabolism and Their Limitations. Cancers 2022, 14, 1741. https://doi.org/10.3390/cancers14071741
Milligan JJ, Saha S. A Nanoparticle’s Journey to the Tumor: Strategies to Overcome First-Pass Metabolism and Their Limitations. Cancers. 2022; 14(7):1741. https://doi.org/10.3390/cancers14071741
Chicago/Turabian StyleMilligan, Joshua J., and Soumen Saha. 2022. "A Nanoparticle’s Journey to the Tumor: Strategies to Overcome First-Pass Metabolism and Their Limitations" Cancers 14, no. 7: 1741. https://doi.org/10.3390/cancers14071741
APA StyleMilligan, J. J., & Saha, S. (2022). A Nanoparticle’s Journey to the Tumor: Strategies to Overcome First-Pass Metabolism and Their Limitations. Cancers, 14(7), 1741. https://doi.org/10.3390/cancers14071741






