Feasibility Study of a Network Meta-Analysis and Unanchored Population-Adjusted Indirect Treatment Comparison of Niraparib, Olaparib, and Bevacizumab as Maintenance Therapies in Patients with Newly Diagnosed Advanced Ovarian Cancer
Abstract
Simple Summary
Abstract
1. Introduction
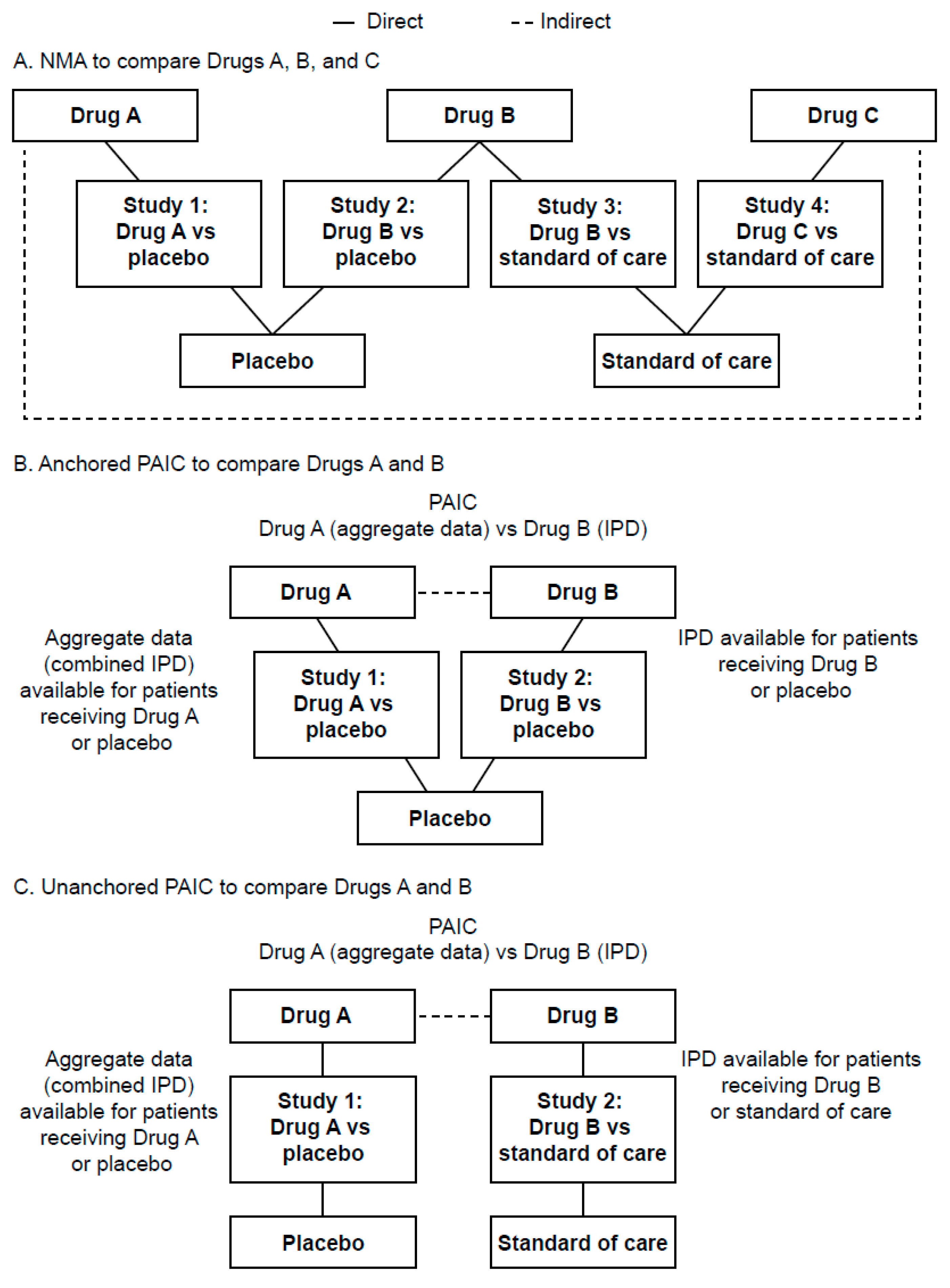
2. Materials and Methods
3. Results
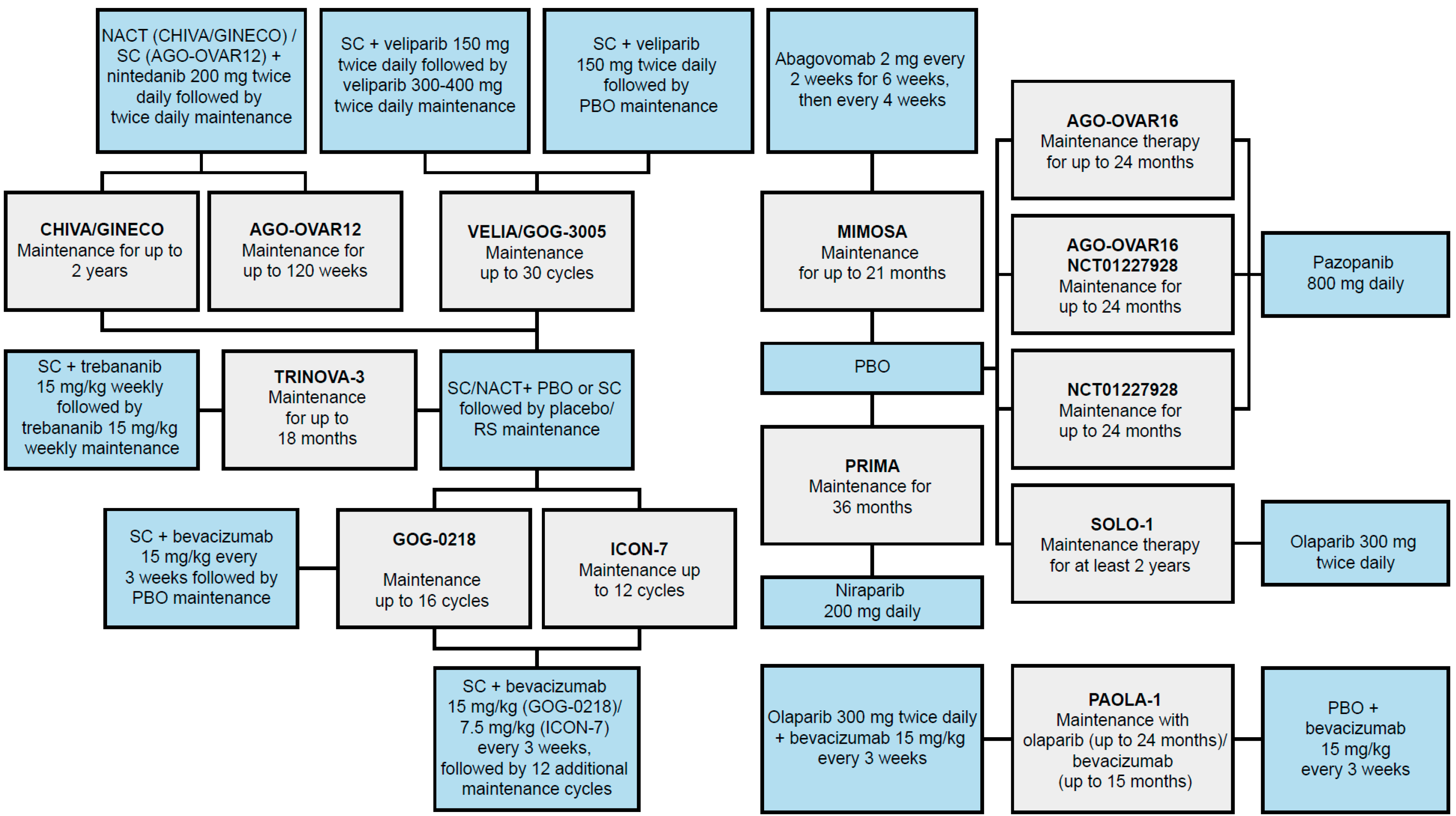
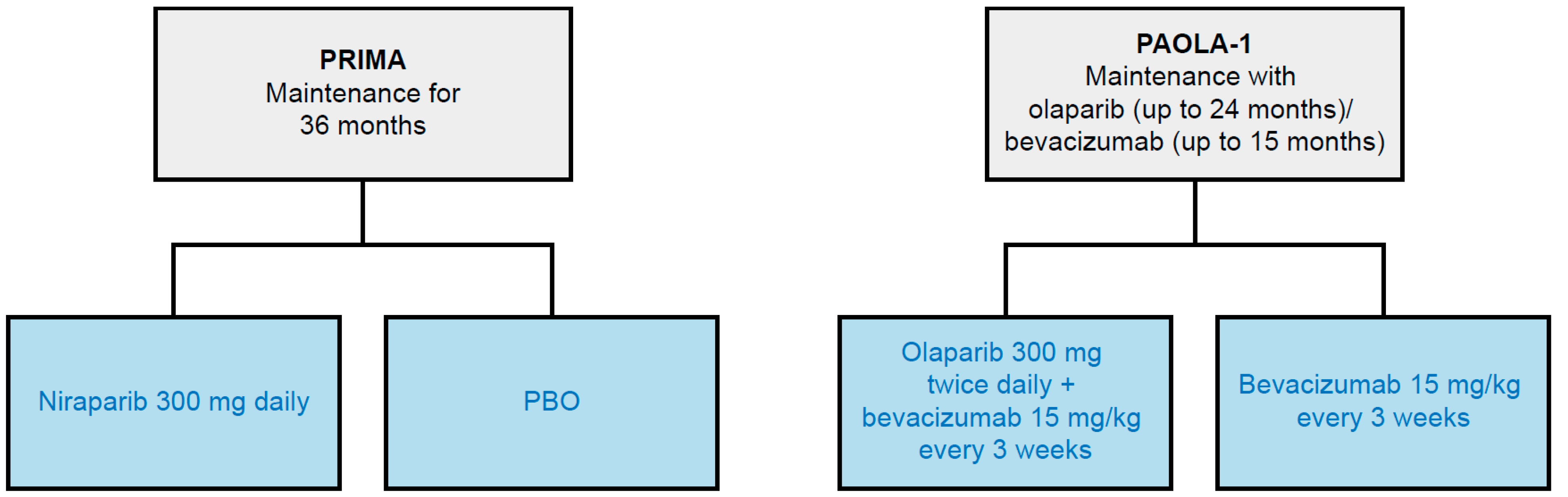
3.1. NMA Feasibility Assessment
3.1.1. Study Design Heterogeneity
3.1.2. Patient Population Heterogeneity
3.1.3. Outcome Heterogeneity
3.2. PAIC Feasibility Assessment
3.2.1. Inclusion/Exclusion Criteria
3.2.2. Bevacizumab Treatment Prior to Study Entry
3.2.3. Receipt of NACT
3.2.4. PFS Method of Assessment and Frequency of Measurement
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Engelhardt, D.; Michor, F. A Quantitative Paradigm for Decision-Making in Precision Oncology. Trends Cancer 2021, 7, 293–300. [Google Scholar] [CrossRef] [PubMed]
- Chan, J.K.; Liang, S.-Y.; Kapp, D.S.; Chan, J.E.; Herzog, T.J.; Coleman, R.L.; Monk, B.J.; Richardson, M.T. Selecting new upfront regimens for advanced ovarian cancer with biomarker guidance. Gynecol. Oncol. 2020, 159, 604–606. [Google Scholar] [CrossRef] [PubMed]
- Lorusso, D.; Ceni, V.; Daniele, G.; Salutari, V.; Pietragalla, A.; Muratore, M.; Nero, C.; Ciccarone, F.; Scambia, G. Newly diagnosed ovarian cancer: Which first-line treatment? Cancer Treat. Rev. 2020, 91, 102111. [Google Scholar] [CrossRef]
- Mirza, M.R.; Coleman, R.L.; González-Martín, A.; Moore, K.N.; Colombo, N.; Ray-Coquard, I.; Pignata, S. The forefront of ovarian cancer therapy: Update on PARP inhibitors. Ann. Oncol. 2020, 31, 1148–1159. [Google Scholar] [CrossRef]
- Dao, F.; Schlappe, B.A.; Tseng, J.; Lester, J.; Nick, A.M.; Lutgendorf, S.K.; McMeekin, S.; Coleman, R.L.; Moore, K.N.; Karlan, B.Y.; et al. Characteristics of 10-year survivors of high-grade serous ovarian carcinoma. Gynecol. Oncol. 2016, 141, 260–263. [Google Scholar] [CrossRef] [PubMed]
- Torre, L.A.; Trabert, B.; DeSantis, C.E.; Miller, K.D.; Samimi, G.; Runowicz, C.D.; Gaudet, M.M.; Jemal, A.; Siegel, R.L. Ovarian cancer statistics, 2018. CA Cancer J. Clin. 2018, 68, 284–296. [Google Scholar] [CrossRef]
- Haddad, F.G.; Karam, E.; Moujaess, E.; Kourie, H.R. Poly-(ADP-ribose) polymerase inhibitors: Paradigm shift in the first-line treatment of newly diagnosed advanced ovarian cancer. Pharmacogenomics 2020, 21, 721–727. [Google Scholar] [CrossRef]
- Lorusso, D.; Mancini, M.; Di Rocco, R.; Fontanelli, R.; Raspagliesi, F. The Role of Secondary Surgery in Recurrent Ovarian Cancer. Int. J. Surg. Oncol. 2012, 2012, 613980. [Google Scholar] [CrossRef]
- Ray-Coquard, I.; Mirza, M.R.; Pignata, S.; Walther, A.; Romero, I.; du Bois, A. Therapeutic options following second-line platinum-based chemotherapy in patients with recurrent ovarian cancer: Comparison of active surveillance and maintenance treatment. Cancer Treat. Rev. 2020, 90, 102107. [Google Scholar] [CrossRef]
- Gupta, S.; Nag, S.; Aggarwal, S.; Rauthan, A.; Warrier, N. Maintenance therapy for recurrent epithelial ovarian cancer: Current therapies and future perspectives—A review. J. Ovarian Res. 2019, 12, 103. [Google Scholar] [CrossRef]
- Ledermann, J.A.; Raja, F.A.; Fotopoulou, C.; Gonzalez-Martin, A.; Colombo, N.; Sessa, C.; ESMO Guidelines Working Group. eUpdate-Ovarian Cancer Treatment Recommendations. 2020. Available online: http://www.esmo.org/Guidelines/Gynaecological-Cancers/Non-Epithelial-Ovarian-Cancer/eUpdate-Treatment-Recommendations (accessed on 1 April 2020).
- National Comprehensive Cancer Network. NCCN Guidelines® for Ovarian Cancer Including Fallopian Tube Cancer and Primary Peritoneal Cancer V.1.2021; © 2020 National Comprehensive Cancer Network, Inc.: Plymouth Meeting, PA, USA, 2021. [Google Scholar]
- Banerjee, S.; Gonzalez-Martin, A.; Harter, P.; Lorusso, D.; Moore, K.N.; Oaknin, A.; Ray-Coquard, I. First-line PARP inhibitors in ovarian cancer: Summary of an ESMO Open—Cancer Horizons round-table discussion. ESMO Open 2020, 5, e001110. [Google Scholar] [CrossRef] [PubMed]
- Tew, W.P.; Lacchetti, C.; Ellis, A.; Maxian, K.; Banerjee, S.; Bookman, M.; Jones, M.B.; Lee, J.-M.; Lheureux, S.; Liu, J.F.; et al. PARP Inhibitors in the Management of Ovarian Cancer: ASCO Guideline. J. Clin. Oncol. 2020, 38, 3468–3493. [Google Scholar] [CrossRef] [PubMed]
- Jiang, X.; Li, W.; Li, X.; Bai, H.; Zhang, Z. Current status and future prospects of PARP inhibitor clinical trials in ovarian cancer. Cancer Manag. Res. 2019, 11, 4371–4390. [Google Scholar] [CrossRef] [PubMed]
- González-Martín, A.; Pothuri, B.; Vergote, I.; DePont Christensen, R.; Graybill, W.; Mirza, M.R.; McCormick, C.; Lorusso, D.; Hoskins, P.; Freyer, G.; et al. Niraparib in Patients with Newly Diagnosed Advanced Ovarian Cancer. N. Engl. J. Med. 2019, 381, 2391–2402. [Google Scholar] [CrossRef] [PubMed]
- GlaxoSmithKline. Zejula 100 mg Hard Capsules, Summary of Product Characteristics. 2021. Available online: https://www.ema.europa.eu/en/documents/product-information/zejula-epar-product-information_en.pdf (accessed on 24 November 2021).
- GlaxoSmithKline. Zejula (Niraparib) Capsules, for Oral Use; Prescribing Information. 2021. Available online: https://www.accessdata.fda.gov/drugsatfda_docs/label/2017/208447lbl.pdf (accessed on 24 November 2021).
- Ray-Coquard, I.; Pautier, P.; Pignata, S.; Pérol, D.; González-Martín, A.; Berger, R.; Fujiwara, K.; Vergote, I.; Colombo, N.; Mäenpää, J.; et al. Olaparib plus Bevacizumab as First-Line Maintenance in Ovarian Cancer. N. Engl. J. Med. 2019, 381, 2416–2428. [Google Scholar] [CrossRef]
- AstraZeneca. Lynparza 100 mg/150 mg Film-Coated Tablets; Summary of Product Characteristics. 2021. Available online: https://www.ema.europa.eu/en/documents/product-information/lynparza-epar-product-information_en.pdf (accessed on 24 November 2021).
- AstraZeneca. Lynparza (Olaparib) Tablets, for Oral Use; Prescribing Information. 2020. Available online: https://nctr-crs.fda.gov/fdalabel/services/spl/set-ids/741ff3e3-dc1a-45a6-84e5-2481b27131aa/spl-doc?hl=olaparib (accessed on 24 November 2021).
- Hoaglin, D.C.; Hawkins, N.; Jansen, J.P.; Scott, D.A.; Itzler, R.; Cappelleri, J.C.; Boersma, C.; Thompson, D.; Larholt, K.M.; Diaz, M.; et al. Conducting Indirect-Treatment-Comparison and Network-Meta-Analysis Studies: Report of the ISPOR Task Force on Indirect Treatment Comparisons Good Research Practices: Part 2. Value Health 2011, 14, 429–437. [Google Scholar] [CrossRef] [PubMed]
- Chaimani, A.; Caldwell, D.M.; Li, T.; Higgins, J.P.; Salanti, G. Chapter 11: Undertaking network meta-analyses. In Cochrane Handbook for Systematic Reviews of Interventions; Higgins, J.P.T., Thomas, J., Chandler, J., Cumpston, M., Li, T., Page, M.J., Welch, V.A., Eds.; Cochrane: London, UK, 2021; Available online: www.training.cochrane.org/handbook (accessed on 24 November 2021).
- Phillippo, D.M.; Ades, T.; Dias, S.; Palmer, S.; Abrams, K.R.; Welton, N. Methods for Population-Adjusted Indirect Comparisons in Health Technology Appraisal. Med. Decis. Mak. 2018, 38, 200–211. [Google Scholar] [CrossRef]
- Oldenhuis, C.N.A.M.; Oosting, S.F.; Gietema, J.A.; de Vries, E.G.E. Prognostic versus predictive value of biomarkers in oncology. Eur. J. Cancer 2008, 44, 946–953. [Google Scholar] [CrossRef] [PubMed]
- Konstantinopoulos, P.A.; Ceccaldi, R.; Shapiro, G.I.; D’Andrea, A.D. Homologous Recombination Deficiency: Exploiting the Fundamental Vulnerability of Ovarian Cancer. Cancer Discov. 2015, 5, 1137–1154. [Google Scholar] [CrossRef]
- Ter Veer, E.; van Oijen, M.G.H.; van Laarhoven, H.W.M. The Use of (Network) Meta-Analysis in Clinical Oncology. Front. Oncol. 2019, 9, 822. [Google Scholar] [CrossRef]
- Clinicaltrials.Gov. Paclitaxel, Polyglutamate Paclitaxel, or Observation in Treating Patients With Stage III or Stage IV Ovarian Epithelial, Peritoneal Cancer, or Fallopian Tube Cancer (NCT00108745); US National Library of Medicine: Bethesda, MD, USA, 2021.
- Markman, M.; Liu, P.; Moon, J.; Monk, B.J.; Copeland, L.; Wilczynski, S.; Alberts, D. Impact on survival of 12 versus 3 monthly cycles of paclitaxel (175 mg/m2) administered to patients with advanced ovarian cancer who attained a complete response to primary platinum-paclitaxel: Follow-up of a Southwest Oncology Group and Gynecologic Oncology Group phase 3 trial. Gynecol. Oncol. 2009, 114, 195–198. [Google Scholar] [CrossRef] [PubMed]
- Markman, M.; Liu, P.; Wilczynski, S.; Monk, B.; Copeland, L.J.; Alvarez, R.D.; Jiang, C.; Alberts, D. Phase III Randomized Trial of 12 Versus 3 Months of Maintenance Paclitaxel in Patients With Advanced Ovarian Cancer After Complete Response to Platinum and Paclitaxel-Based Chemotherapy: A Southwest Oncology Group and Gynecologic Oncology Group Trial. J. Clin. Oncol. 2003, 21, 2460–2465. [Google Scholar] [CrossRef] [PubMed]
- Morgan, M.A.; Darcy, K.M.; Rose, P.G.; DeGeest, K.; Bookman, M.A.; Aikins, J.K.; Sill, M.W.; Mannel, R.S.; Allievi, C.; Egorin, M.J. Paclitaxel poliglumex and carboplatin as first-line therapy in ovarian, peritoneal or fallopian tube cancer: A phase I and feasibility trial of the Gynecologic Oncology Group. Gynecol. Oncol. 2008, 110, 329–335. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Kwon, Y.S.; Nam, J.-H.; Kim, D.-Y.; Suh, D.-S.; Kim, J.-H.; Kim, Y.M.; Kim, Y.T. Hexamethylmelamine as Consolidation Treatment for Patients with Advanced Epithelial Ovarian Cancer in Complete Response after First-Line Chemotherapy. J. Korean Med. Sci. 2009, 24, 679–683. [Google Scholar] [CrossRef][Green Version]
- Guy, H.; Travers, K.; Hawkes, C.; Walder, L.; Malinowska, I.; Gupta, D. 373 Systematic literature review of efficacy and safety of first-line maintenance therapy trials in advanced ovarian cancer. Ovarian Cancer 2020, 30, A73–A74. [Google Scholar]
- Ray-Coquard, I.L.; Pautier, P.; Pignata, S.; Pérol, D.; González-Martín, A.; Sevelda, P.; Fujiwara, K.; Vergote, I.B.; Colombo, N.; Mäenpää, J.; et al. Phase III PAOLA-1/ENGOT-ov25 trial: Olaparib plus bevacizumab (bev) as maintenance therapy in patients (pts) with newly diagnosed, advanced ovarian cancer (OC) treated with platinum-based chemotherapy (PCh) plus bev. Ann. Oncol. 2019, 30 (Suppl. 5), v894–v895. [Google Scholar] [CrossRef]
- Phillippo, D.M.; Ades, A.E.; Dias, S.; Palmer, S.; Abrams, K.R.; Welton, N.J. NICE DSU Technical Support Document 18: Methods for population-adjusted indirect comparisons in submissions to NICE, in Technical Support Documents. Nice Decis. Support Unit 2016, 18, 81. [Google Scholar]
- Eisenkop, S.M.; Spirtos, N.M.; Friedman, R.L.; Lin, W.-C.M.; Pisani, A.L.; Perticucci, S. Relative influences of tumor volume before surgery and the cytoreductive outcome on survival for patients with advanced ovarian cancer: A prospective study. Gynecol. Oncol. 2003, 90, 390–396. [Google Scholar] [CrossRef]
- Ferron, G.; de Rauglaudre, G.; Chevalier, A.; Combe, P.; Joly, F.; Lortholary, A.; Raban, N.; Hamizi, S.; Malaurie, E.; Kaminsky, M.-C.; et al. Impact of adding nintedanib to neoadjuvant chemotherapy (NACT) for advanced epithelial ovarian cancer (EOC) patients: The CHIVA double-blind randomized phase II GINECO study. J. Clin. Oncol. 2019, 37, 5512. [Google Scholar] [CrossRef]
- Hettle, R.; McCrea, C.; Lee, C.K.; Davidson, R. Population-adjusted indirect treatment comparison (PAITC) of maintenance PARP inhibitor (PARPi) with or without bevacizumab versus bevacizumab in women with newly diagnosed ovarian cancer (OC). J. Clin. Oncol. 2020, 38, 6052. [Google Scholar] [CrossRef]
- European Medicines Agency. LYNPARZA® (Olaparib): CHMP Post-Authorization Summary of Positive Opinion 2020. Available online: https://www.ema.europa.eu/en/documents/smop/chmp-post-authorisation-summary-positive-opinion-lynparza-ii-35-ii-36_en.pdf (accessed on 24 November 2021).
- Gonzalez, A.; Mirza, M.R.; Vergote, I.; Li, Y.; Hazard, S.; Clark, R.; Graybill, W.; Pothuri, B.; Monk, B.J. A prospective evaluation of tolerability of niraparib dosing based upon baseline body weight (wt) and platelet (blplt) count: Blinded pooled interim safety data from the PRIMA Study. Ann. Oncol. 2018, 29, viii335–viii336. [Google Scholar] [CrossRef]
- Martín, A.G.; Pothuri, B.; Vergote, I.B.; Christensen, R.D.; Graybill, W.; Mirza, M.R.; McCormick, C.; Lorusso, D.; Hoskins, P.; Freyer, G.; et al. Niraparib therapy in patients with newly diagnosed advanced ovarian cancer (PRIMA/ENGOT-OV26/GOG-3012 study). Ann. Oncol. 2019, 30 (Suppl. 5), v851–v934. [Google Scholar]
- Monk, B.J.; Mirza, M.R.; Vergote, I.; Li, Y.; Malinowska, I.; Gupta, D.; Graybill, W.A.; Pothuri, B.; Gonzalez-Martin, A. A prospective evaluation of tolerability of niraparib dosing based upon baseline body weight and plate-let count: Blinded pooled interim safety data from the ENGOT-OV26/PRIMA study. Gynecol. Oncol. 2019, 154, 3–4. [Google Scholar] [CrossRef]
- González-Martín, A.; Pothuri, B.; Vergote, I.; Christensen, R.D.; Graybill, W.; Mirza, M.R.; McCormick, C.; Lorusso, D.; Hoskins, P.; Freyer, G.; et al. Niraparib therapy in patients with newly diagnosed advanced ovarian cancer after chemo-therapy: PRIMA/ENGOT-OV26/GOG-3012 study. Int. J. Gynecol. Cancer 2019, 29 (Suppl. 4), A9. [Google Scholar]
- Moore, K.; Colombo, N.; Scambia, G.; Kim, B.-G.; Oaknin, A.; Friedlander, M.; Lisyanskaya, A.; Floquet, A.; Leary, A.; Sonke, G.S.; et al. Maintenance Olaparib in Patients with Newly Diagnosed Advanced Ovarian Cancer. N. Engl. J. Med. 2018, 379, 2495–2505. [Google Scholar] [CrossRef] [PubMed]
- Mathews, C.A.; Moore, K.N.; Colombo, N.; Scambia, G.; Kim, B.-G.; Oaknin, A.; Friedlander, M.; Lisyanskaya, A.S.; Floquet, A.; Leary, A.; et al. Maintenance olaparib after platinum-based chemotherapy in patients (pts) with newly diagnosed advanced ovarian cancer (OC) and a BRCA mutation (BRCAm): Efficacy by surgical and tumor status in the Phase III SOLO1 trial. J. Clin. Oncol. 2019, 37, 5541. [Google Scholar] [CrossRef]
- Wu, L.; Zhu, J.; Yin, R.; Wu, X.; Lou, G.; Wang, J.; Gao, Y.; Kong, B.; Lu, X.; Zhou, Q.; et al. Olaparib maintenance therapy in patients (pts) with a BRCA1 and/or BRCA2 mutation (BRCAm) and newly diagnosed advanced ovarian cancer (OC): SOLO1 China cohort. J. Clin. Oncol. 2019, 37, 5554. [Google Scholar] [CrossRef]
- Wu, L.; Zhu, J.; Yin, R.; Wu, X.; Lou, G.; Wang, J.; Gao, Y.; Kong, B.; Lu, X.; Zhou, Q.; et al. Olaparib maintenance therapy in patients (pts) with a BRCA1 and/or BRCA2 mutation (BRCAm) and newly diagnosed advanced ovarian cancer (OC): SOLO1 China cohort. Ann. Oncol. 2019, 30, ix79. [Google Scholar] [CrossRef]
- Friedlander, M.; Rau, J.; Lee, C.K.; Meier, W.; Lesoin, A.; Kim, J.W.; Poveda, A.; Buck, M.; Scambia, G.; Shimada, M.; et al. Maintenance olaparib following platinum-based chemotherapy in newly diagnosed patients (pts) with advanced ovarian cancer (OC) and a BRCA1/2 mutation (BRCAm): Phase III SOLO1 trial, in ESMO ASIA 2018 Congress. Ann. Oncol. 2018, 29, viii727. [Google Scholar] [CrossRef]
- Friedlander, M.; Moore, K.N.; Colombo, N.; Scambia, G.; Kim, B.-G.; Oaknin, A.; Lisyanskaya, A.S.; Floquet, A.; Leary, A.; Sonke, G.S.; et al. Efficacy of maintenance olaparib for newly diagnosed, advanced ovarian cancer patients (pts) by BRCA1 or BRCA2 mutation in the phase III SOLO1 trial. J. Clin. Oncol. 2019, 37, 5551. [Google Scholar] [CrossRef]
- Moore, K.N.; Colombo, N.; Scambia, G.; Kim, B.G.; Oaknin, A.; Friedlander, M.; Lisyanskaya, A.; Floquet, A.; Leary, A.; Sonke, G.S.; et al. Maintenance olaparib following platinum-based chemotherapy in newly diagnosed patients (pts) with advanced ovarian cancer (OC) and a BRCA1/2 mutation (BRCAm): Phase III SOLO1 trial. ESMO Ann. Oncol. 2018, 29, viii727. [Google Scholar] [CrossRef]
- Friedlander, M.; Hettle, R.; Parkhomenko, E. Impact of radiological disease progression on the health status of patients (pts) with BRCA-mutated advanced ovarian cancer (OC) treated with maintenance olaparib (OL) or placebo (PL) after first-line platinum chemotherapy. Ann. Oncol. 2019, 30, ix77–ix78. [Google Scholar] [CrossRef]
- Friedlander, M.; Moore, K.; Colombo, N.; Scambia, G.; Kim, B.-G.; Oaknin, A.; Lisyanskaya, A.; Floquet, A.; Leary, A.; Sonke, G.; et al. Patient-centred outcomes with maintenance olaparib in newly diagnosed patients with advanced ovarian cancer (OC) and a BRCA mutation (BRCAm) in the phase III SOLO1 trial to support the clinical benefit of prolongation of progression-free survival (PFS). Ann. Oncol. 2019, 30, v405–v406. [Google Scholar] [CrossRef]
- Gourley, C.; Brown, J.; Lai, Z.; Lao-Sirieix, P.; Elks, C.; McGarvey, H.; French, T.; Milenkova, T.; Bloomfield, R.; Rowe, P.; et al. Analysis of tumour samples from SOLO1: Frequency of BRCA specific loss of heterozygosity (LOH) and progression-free survival (PFS) according to homologous recombination repair deficiency (HRD)-LOH score. Ann. Oncol. 2019, 30, v407. [Google Scholar] [CrossRef]
- Moore, K.; Colombo, N.; Scambia, G.; Kim, B.-G.; Oaknin, A.; Friedlander, M.; Lisyanskaya, A.; Floquet, A.; Leary, A.; Sonke, G.; et al. Maintenance olaparib after platinum-based chemotherapy in patients with newly diagnosed advanced ovarian cancer and a BRCA mutation: Efficacy by the timing of surgery and residual tumour status following upfront or interval cytoreductive surgery in the Phase III SOLO1 trial. Oral Commun. Gynaecol. Cancer 2019, 29, A14–A15. [Google Scholar] [CrossRef]
- Oaknin, A.; Moore, K.; Colombo, N.; Scambia, G.; Kim, B.-G.; Friedlander, M.; Lisyanskaya, A.; Floquet, A.; Leary, A.; Sonke, G.; et al. Time to second progression (PFS2) and second subsequent therapy (TSST) for patients (pts) with newly diagnosed, advanced ovarian cancer (OC) and a BRCA mutation (BRCAm) treated with maintenance (mt) olaparib (ola): Phase III SOLO1 trial. Ann. Oncol. 2019, 30, v405. [Google Scholar] [CrossRef]
- Colombo, N.; Moore, K.N.; Scambia, G.; Oaknin, A.; Friedlander, M.; Lisyanskaya, A.S.; Floquet, A.; Leary, A.; Sonke, G.S.; Gourley, C.; et al. Adverse events (AEs) with maintenance olaparib in newly diagnosed patients (pts) with advanced ovarian cancer (OC) and a BRCA mutation (BRCAm): Phase III SOLO1 trial. J. Clin. Oncol. 2019, 37, 5539. [Google Scholar] [CrossRef]
- Sabbatini, P.; Harter, P.; Scambia, G.; Sehouli, J.; Meier, W.; Wimberger, P.; Baumann, K.H.; Kurzeder, C.; Schmalfeldt, B.; Cibula, D.; et al. Abagovomab As Maintenance Therapy in Patients With Epithelial Ovarian Cancer: A Phase III Trial of the AGO OVAR, COGI, GINECO, and GEICO—The MIMOSA Study. J. Clin. Oncol. 2013, 31, 1554–1561. [Google Scholar] [CrossRef]
- Buzzonetti, A.; Fossati, M.; Catzola, V.; Scambia, G.; Fattorossi, A.; Battaglia, A. Immunological response induced by abagovomab as a maintenance therapy in patients with epithelial ovarian cancer: Relationship with survival—A substudy of the MIMOSA trial. Cancer Immunol. Immunother. 2014, 63, 1037–1045. [Google Scholar] [CrossRef]
- Vergote, I.; Hanker, L.C.; Floquet, A.; Rau, J.; Kim, J.-W.; Izquierdo, E.O.; Friedlander, M.; Pignata, S.; Fujiwara, K.; Colombo, N.; et al. AGO-OVAR 16: A phase III study to evaluate the efficacy and safety of pazopanib (PZ) monotherapy versus placebo in women who have not progressed after first line chemotherapy for epithelial ovarian, fallopian tube, or primary peritoneal cancer—Overall survival (OS) results. J. Clin. Oncol. 2018, 36, 5518. [Google Scholar] [CrossRef]
- Du Bois, A.; Floquet, A.; Kim, J.-W.; Rau, J.; del Campo, J.M.; Friedlander, M.; Pignata, S.; Fujiwara, K.; Vergote, I.; Colombo, N.; et al. Incorporation of Pazopanib in Maintenance Therapy of Ovarian Cancer. J. Clin. Oncol. 2014, 32, 3374–3382. [Google Scholar] [CrossRef] [PubMed]
- Friedlander, M.; Rau, J.; Lee, C.; Meier, W.; Lesoin, A.; Kim, J.-W.; Poveda, A.; Buck, M.; Scambia, G.; Shimada, M.; et al. Quality of life in patients with advanced epithelial ovarian cancer (EOC) randomized to maintenance pazopanib or placebo after first-line chemotherapy in the AGO-OVAR 16 trial. Measuring what matters—patient-centered end points in trials of maintenance therapy. Ann. Oncol. 2018, 29, 737–743. [Google Scholar] [CrossRef] [PubMed]
- Du Bois, A.; Floquet, A.; Kim, J.W.; Rau, J.; del Campo, J.M.; Friedlander, M.; Pignata, S.; Fujiwara, K.; Vergote, I.; Colombo, N.; et al. Randomized, double-blind, phase III trial of pazopanib versus placebo in women who have not progressed after first-line chemotherapy for advanced epithelial ovarian, fallopian tube, or primary peritoneal cancer (AE-OC): Results of an international Intergroup trial (AGO-OVAR16). J. Clin. Oncol. 2013, 31 (Suppl. 18), LBA5503. [Google Scholar]
- Floquet, A.; Vergote, I.; Colombo, N.; Fiane, B.; Monk, B.J.; Reinthaller, A.; Calvert, P.; Herzog, T.J.; Meier, W.; Kim, J.-W.; et al. Progression-free survival by local investigator versus independent central review: Comparative analysis of the AGO-OVAR16 Trial. Gynecol. Oncol. 2015, 136, 37–42. [Google Scholar] [CrossRef]
- Del Campo, J.; Kurzeder, C.; Berton-Rigaud, D.; Kim, B.; Friedlander, M.; Zamagni, C.; Sakuragi, N.; Canzler, U.; Crescenzo, R.; Bois, A. Progression-free survival by GCIG criteria: Analysis of the secondary endpoints of the AGO-OVAR16 Trial. Int. J. Gynecol. Cancer 2013, 136, 37–42. [Google Scholar]
- Vergote, I.; du Bois, A.; Floquet, A.; Rau, J.; Kim, J.-W.; del Campo, J.; Friedlander, M.; Pignata, S.; Fujiwara, K.; Colombo, N.; et al. Overall survival results of AGO-OVAR16: A phase 3 study of maintenance pazopanib versus placebo in women who have not progressed after first-line chemotherapy for advanced ovarian cancer. Gynecol. Oncol. 2019, 155, 186–191. [Google Scholar] [CrossRef]
- Kim, J.-W.; Mahner, S.; Wu, L.-Y.; Shoji, T.; Kim, B.-G.; Zhu, J.; Takano, T.; Park, S.-Y.; Kong, B.-H.; Wu, Q.; et al. Pazopanib Maintenance Therapy in East Asian Women With Advanced Epithelial Ovarian Cancer: Results From AGO-OVAR16 and an East Asian Study. Int. J. Gynecol. Cancer 2018, 28, 2–10. [Google Scholar] [CrossRef]
- Zang, R.; Wu, L.; Zhu, J.; Kong, B.; Kim, B.-G.; Yao, Y.; Yin, R.; Liu, J.; Wu, Q.; Ngan, H.Y.S.; et al. Pazopanib (Paz) monotherapy in Asian women who have not progressed after first-line chemotherapy for advanced ovarian, Fallopian tube, or primary peritoneal carcinoma. J. Clin. Oncol. 2013, 31, 5512. [Google Scholar] [CrossRef]
- Oza, A.M.; Cook, A.D.; Pfisterer, J.; Embleton, A.; Ledermann, J.A.; Pujade-Lauraine, E.; Kristensen, G.; Carey, M.S.; Beale, P.; Cervantes, A.; et al. Standard chemotherapy with or without bevacizumab for women with newly diagnosed ovarian cancer (ICON7): Overall survival results of a phase 3 randomised trial. Lancet Oncol. 2015, 16, 928–936. [Google Scholar] [CrossRef]
- Perren, T.J.; Swart, A.M.; Pfisterer, J.; Ledermann, J.A.; Pujade-Lauraine, E.; Kristensen, G.; Carey, M.S.; Beale, P.; Cervantes, A.; Kurzeder, C.; et al. A phase 3 trial of bevacizumab in ovarian cancer. N. Engl. J. Med. 2011, 365, 2484–2496. [Google Scholar] [CrossRef] [PubMed]
- Tewari, K.S.; Burger, R.A.; Enserro, D.; Norquist, B.M.; Swisher, E.M.; Brady, M.F.; Bookman, M.A.; Fleming, G.F.; Huang, H.; Homesley, H.D.; et al. Final Overall Survival of a Randomized Trial of Bevacizumab for Primary Treatment of Ovarian Cancer. J. Clin. Oncol. 2019, 37, 2317–2328. [Google Scholar] [CrossRef]
- Burger, R.A.; Brady, M.F.; Bookman, M.A.; Fleming, G.F.; Monk, B.J.; Huang, H.; Mannel, R.S.; Homesley, H.D.; Fowler, J.; Greer, B.E.; et al. Incorporation of Bevacizumab in the Primary Treatment of Ovarian Cancer. N. Engl. J. Med. 2011, 365, 2473–2483. [Google Scholar] [CrossRef] [PubMed]
- Ray-Coquard, I.; Cibula, D.; Mirza, M.R.; Reuss, A.; Ricci, C.; Colombo, N.; Zabernigg, A.; Goffin, F.; Gonzalez-Martin, A.; Ottevanger, P.B.; et al. AGO-OVAR 12: A randomized placebo-controlled GCIG/ENGOT-intergroup phase III trial with chemotherapy +/− nintedanib for advanced ovarian cancer: Overall survival results. Int. J. Gynecol. Cancer. 2017, 27, 3. [Google Scholar]
- Vergote, I.; Scambia, G.; O’Malley, D.M.; van Calster, B.; Park, S.Y.; Del Campo, J.M.; Meier, W.; Bamias, A.; Colombo, N.; Wenham, R.M.; et al. Trebananib or placebo plus carboplatin and paclitaxel as first-line treatment for advanced ovarian cancer (TRINOVA-3/ENGOT-ov2/GOG-3001): A randomised, double-blind, phase 3 trial. Lancet Oncol. 2019, 20, 862–876. [Google Scholar] [CrossRef]
- Coleman, R.L.; Fleming, G.F.; Brady, M.F.; Swisher, E.M.; Steffensen, K.D.; Friedlander, M.; Okamoto, A.; Moore, K.N.; Efrat Ben-Baruch, N.; Werner, T.L.; et al. Veliparib with First-Line Chemotherapy and as Maintenance Therapy in Ovarian Cancer. N. Engl. J. Med. 2019, 381, 2403–2415. [Google Scholar] [CrossRef] [PubMed]
- NCT02470585, Veliparib with Carboplatin and Paclitaxel and as Continuation Maintenance Therapy in Subjects with Newly Diagnosed Stage III or IV, High-Grade Serous, Epithelial Ovarian, Fallopian Tube, or Primary Peritoneal Cancer. 2015. Available online: https://clinicaltrials.gov/show/nct02470585 (accessed on 22 December 2021).
- Burger, R.A.; Brady, M.F.; Rhee, J.; Sovak, M.A.; Kong, G.; Nguyen, H.P.; Bookman, M.A. Independent radiologic review of the Gynecologic Oncology Group Study 0218, a phase III trial of bevacizumab in the primary treatment of advanced epithelial ovarian, primary peritoneal, or fallopian tube cancer. Gynecol. Oncol. 2013, 131, 21–26. [Google Scholar] [CrossRef] [PubMed]
- Amit, O.; Mannino, F.; Stone, A.; Bushnell, W.; Denne, J.; Helterbrand, J.; Burger, H. Blinded independent central review of progression in cancer clinical trials: Results from a meta-analysis. Eur. J. Cancer 2011, 47, 1772–1778. [Google Scholar] [CrossRef] [PubMed]
- Dodd, L.E.; Korn, E.L.; Freidlin, B.; Jaffe, C.C.; Rubinstein, L.V.; Dancey, J.; Mooney, M.M. Blinded Independent Central Review of Progression-Free Survival in Phase III Clinical Trials: Important Design Element or Unnecessary Expense? J. Clin. Oncol. 2008, 26, 3791–3796. [Google Scholar] [CrossRef]
- Harter, P.; Mouret-Reynier, M.A.; Pignata, S.; Cropet, C.; González-Martín, A.; Bogner, G.; Fujiwara, K.; Vergote, I.; Colombo, N.; Nøttrup, T.J.; et al. Efficacy of maintenance olaparib plus bevacizumab by biomarker status in clinical higher- and lower-risk patients with newly diagnosed, advanced ovarian cancer in the PAOLA-1 trial. Int. J. Gynecol. Cancer 2020, 30 (Suppl. 3), A13–A14. [Google Scholar]
- Korach, J.; Graybill, W.; Redondo, A.; O’Malley, D.M.; Han, S.; Cloven, N.G.; Knudsen, A.O.; Lechpammer, S.; Gonzalez-Martin, A.; Monk, B.J. Niraparib in patients with newly diagnosed advanced ovarian brcam cancer: A post hoc analysis of the prima/engot-ov26/gog-3012 trial. Int. J. Gynecol. Cancer 2020, 30, A125–A126. [Google Scholar]
- Melamed, A.; Fink, G.; Wright, A.A.; Keating, N.L.; Gockley, A.A.; del Carmen, M.G.; Schorge, J.O.; Rauh-Hain, J.A. Effect of adoption of neoadjuvant chemotherapy for advanced ovarian cancer on all cause mortality: Quasi-experimental study. BMJ 2018, 360, j5463. [Google Scholar] [CrossRef]
- Tanner, E.; Chi, D.S.; Eisenhauer, E.L.; Diaz-Montes, T.P.; Santillan, A.; Bristow, R.E. Surveillance for the detection of recurrent ovarian cancer: Survival impact or lead-time bias? Gynecol. Oncol. 2010, 117, 336–340. [Google Scholar] [CrossRef] [PubMed]
- Berger, M.L.; Mamdani, M.; Atkins, D.; Johnson, M.L. Good Research Practices for Comparative Effectiveness Research: Defining, Reporting and Interpreting Nonrandomized Studies of Treatment Effects Using Secondary Data Sources: The ISPOR Good Research Practices for Retrospective Database Analysis Task Force Report—Part I. Value Health 2009, 12, 1044–1052. [Google Scholar] [CrossRef]
- Ramsey, S.; Willke, R.; Briggs, A.; Brown, R.; Buxton, M.; Chawla, A.; Cook, J.; Glick, H.; Liljas, B.; Petitti, D.; et al. Good Research Practices for Cost-Effectiveness Analysis Alongside Clinical Trials: The ISPOR RCT-CEA Task Force Report. Value Health 2005, 8, 521–533. [Google Scholar] [CrossRef] [PubMed]
- Vergote, I.; Moore, K.; Hettle, R.; Rhodes, K.; Ouwens, M.; Ray-Coquard, I. Population adjusted indirect comparison of the SOLO1 and PAOLA-1/ENGOT-ov25 studies of olaparib with or without bevacizumab, bev alone and placebo in the maintenance treatment of women with newly diagnosed stage III/IV ovarian cancer with BRCA mutation. Gynecol. Oncol. 2020, 159, 19–20. [Google Scholar] [CrossRef]
- Jansen, J.P.; Fleurence, R.; Devine, B.; Itzler, R.; Barrett, A.; Hawkins, N.; Lee, K.; Boersma, C.; Annemans, L.; Cappelleri, J.C. Interpreting Indirect Treatment Comparisons and Network Meta-Analysis for Health-Care Decision Making: Report of the ISPOR Task Force on Indirect Treatment Comparisons Good Research Practices: Part 1. Value Health 2011, 14, 417–428. [Google Scholar] [CrossRef] [PubMed]
- Balogh, E.P.; Ganz, P.A.; Murphy, S.B.; Nass, S.J.; Ferrell, B.R.; Stovall, E. Patient-Centered Cancer Treatment Planning: Improving the Quality of Oncology Care. Summary of an Institute of Medicine Workshop. Oncologist 2011, 16, 1800–1805. [Google Scholar] [CrossRef] [PubMed]
- Mirza, M.R. When in the treatment continuum to use PARP inhibition in ovarian cancer. Clin. Adv. Hematol. Oncol. 2021, 19, 84–86. [Google Scholar] [PubMed]
| Category | Factor |
|---|---|
| Different quality or methods of randomized trials |
|
| Confounding factors in relation to participant population |
|
| Confounding factors in relation to circumstances |
|
| Different treatment |
|
| Different outcome measures and methods of statistical analysis |
|
| Trial | Study Design Eterogeneity: Lack of Common Comparator within the Network | Patient Population Heterogeneity: Inclusion of Patients with FIGO Stage III Disease with no VRD Following PDS | Outcome Heterogeneity | |
|---|---|---|---|---|
| Interim or Immature OS Data | Differing Measurement of PFS and OS Starting Time Point due to Trial Design | |||
| SOLO-1 [44,45,46,47,48,49,50,51,52,53,54,55,56] | ✓ * | ✓ | ||
| ICON-7 [68,69] | ✓ | ✓ | ||
| MIMOSA [57,58] | ✓ | ✓ | PFS was not assessed | |
| AGO-OVAR16 [59,60,61,62,63,64,65] | ✓ | |||
| NCT01227928 [66,67] | ✓ | |||
| GOG-0218 [70,71] | ✓ | ✓ | ||
| PAOLA-1 [19,34] | ✓ | ✓ | ✓ | |
| CHIVA/GINECO [37] | ✓ | ✓ | ✓ | ✓ |
| TRINOVA-3 [73] | ✓ | ✓ | ✓ | |
| VELIA/GOG-3005 [74,75] | ✓ | ✓ | ✓ | ✓ |
| AGO-OVAR12 [72] | ✓ | ✓ | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lorusso, D.; Guy, H.; Samyshkin, Y.; Hawkes, C.; Estenson, K.; Coleman, R.L. Feasibility Study of a Network Meta-Analysis and Unanchored Population-Adjusted Indirect Treatment Comparison of Niraparib, Olaparib, and Bevacizumab as Maintenance Therapies in Patients with Newly Diagnosed Advanced Ovarian Cancer. Cancers 2022, 14, 1285. https://doi.org/10.3390/cancers14051285
Lorusso D, Guy H, Samyshkin Y, Hawkes C, Estenson K, Coleman RL. Feasibility Study of a Network Meta-Analysis and Unanchored Population-Adjusted Indirect Treatment Comparison of Niraparib, Olaparib, and Bevacizumab as Maintenance Therapies in Patients with Newly Diagnosed Advanced Ovarian Cancer. Cancers. 2022; 14(5):1285. https://doi.org/10.3390/cancers14051285
Chicago/Turabian StyleLorusso, Domenica, Holly Guy, Yevgeniy Samyshkin, Carol Hawkes, Kasey Estenson, and Robert L. Coleman. 2022. "Feasibility Study of a Network Meta-Analysis and Unanchored Population-Adjusted Indirect Treatment Comparison of Niraparib, Olaparib, and Bevacizumab as Maintenance Therapies in Patients with Newly Diagnosed Advanced Ovarian Cancer" Cancers 14, no. 5: 1285. https://doi.org/10.3390/cancers14051285
APA StyleLorusso, D., Guy, H., Samyshkin, Y., Hawkes, C., Estenson, K., & Coleman, R. L. (2022). Feasibility Study of a Network Meta-Analysis and Unanchored Population-Adjusted Indirect Treatment Comparison of Niraparib, Olaparib, and Bevacizumab as Maintenance Therapies in Patients with Newly Diagnosed Advanced Ovarian Cancer. Cancers, 14(5), 1285. https://doi.org/10.3390/cancers14051285






