Nurse-Led Consultation and Symptom Burden in Patients with Head and Neck Cancer: A Comparative Analysis of Routine Clinical Data
Abstract
Simple Summary
Abstract
1. Introduction
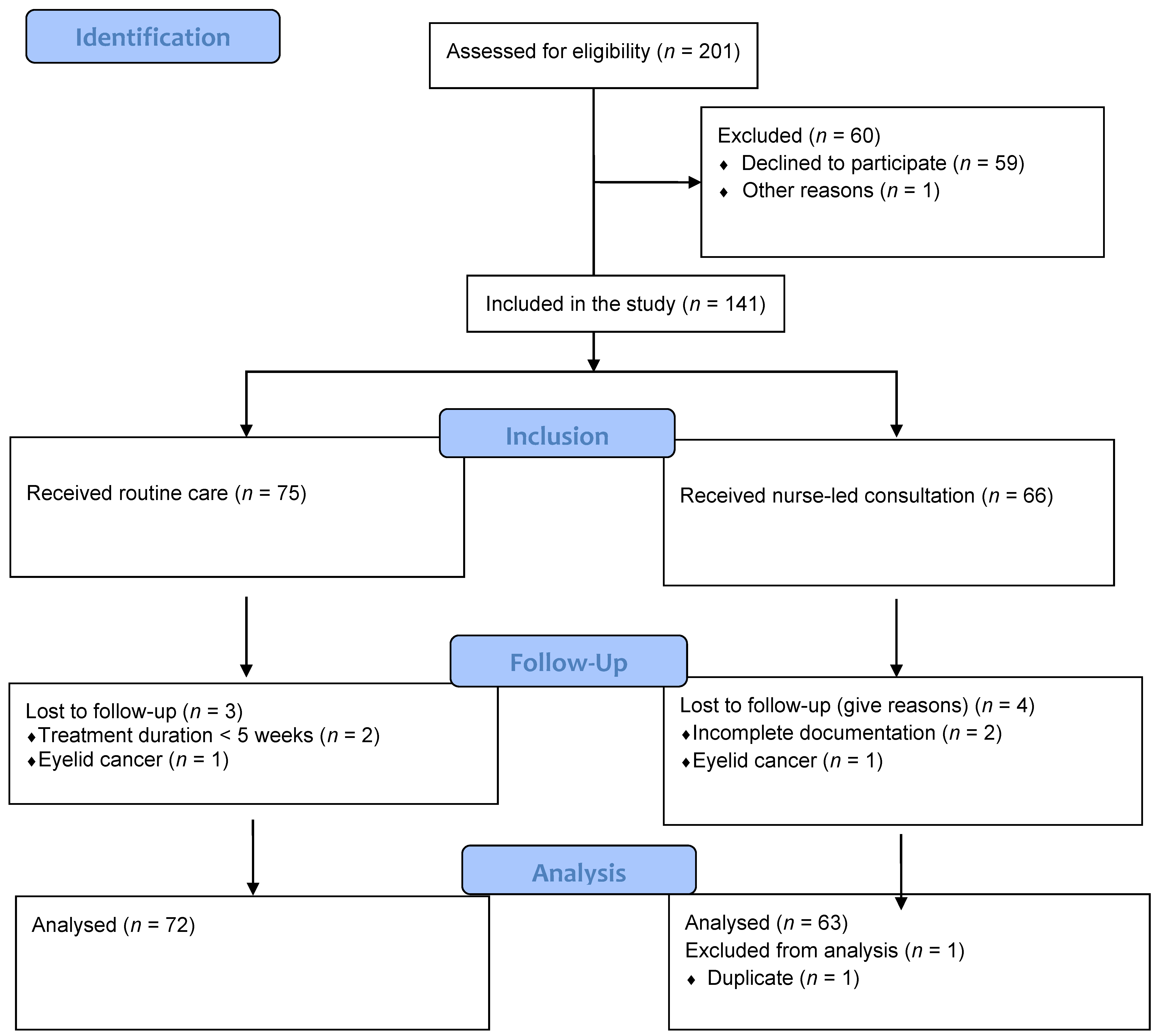
2. Materials and Methods
2.1. Study Design and Setting
2.2. Study Population
2.3. Data Collection
2.4. Routine Care
2.5. Nurse-Led Consultation
2.6. Data Collection
2.7. Statistical Analysis
2.8. Ethics
3. Results
3.1. Participant Demographic and Clinical Characteristics
3.2. Nurse-Led Consultations
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, L.A.; Jemal, A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2018, 68, 394–424. [Google Scholar] [CrossRef] [PubMed]
- Lo Nigro, C.; Denaro, N.; Merlotti, A.; Merlano, M. Head and neck cancer: Improving outcomes with a multidisciplinary approach. Cancer Manag. Res. 2017, 9, 363–371. [Google Scholar] [CrossRef] [PubMed]
- Yete, S.; D’Souza, W.; Saranath, D. High-Risk Human Papillomavirus in Oral Cancer: Clinical Implications. Oncology 2018, 94, 133–141. [Google Scholar] [CrossRef] [PubMed]
- Argiris, A.; Karamouzis, M.V.; Raben, D.; Ferris, R.L. Head and neck cancer. Lancet 2008, 371, 1695–1709. [Google Scholar] [CrossRef]
- Ang, K.K.; Harris, J.; Wheeler, R.; Weber, R.; Rosenthal, D.I.; Nguyen-Tân, P.F.; Westra, W.H.; Chung, C.H.; Jordan, R.C.; Lu, C.; et al. Human Papillomavirus and Survival of Patients with Oropharyngeal Cancer. N. Engl. J. Med. 2010, 363, 24–35. [Google Scholar] [CrossRef]
- Mendenhall, W.M.; Dagan, R.; Bryant, C.M.; Fernandes, R.P. Radiation Oncology for Head and Neck Cancer: Current Standards and Future Changes. Oral Maxillofac. Surg. Clin. N. Am. 2019, 31, 31–38. [Google Scholar] [CrossRef]
- Naghavi, A.O.; Echevarria, M.I.; Strom, T.J.; Abuodeh, Y.A.; Venkat, P.S.; Ahmed, K.A.; Demetriou, S.; Frakes, J.M.; Kim, Y.; Kish, J.A.; et al. Patient choice for high-volume center radiation impacts head and neck cancer outcome. Cancer Med. 2018, 7, 4964–4979. [Google Scholar] [CrossRef]
- Meccariello, G.; Maniaci, A.; Bianchi, G.; Cammaroto, G.; Iannella, G.; Catalano, A.; Sgarzani, R.; De Vito, A.; Capaccio, P.; Pelucchi, S.; et al. Neck dissection and trans oral robotic surgery for oropharyngeal squamous cell carcinoma. Auris Nasus Larynx 2022, 49, 117–125. [Google Scholar] [CrossRef] [PubMed]
- Uptodate. Available online: https://www.uptodate.com/contents/overview-of-treatment-for-head-and-neck-cancer?sectionName=TNM%20STAGING%20SYSTEM&search=Head%20and%20Neck%20Cancer%20Symptom%20Management&topicRef=89977&anchor=H256481212&source=see_link#H256481212 (accessed on 17 November 2021).
- Humphris, G. Psychological management for head and neck cancer patients: United Kingdom National Multidisciplinary Guidelines. J. Laryngol. Otol. 2016, 130, S45–S48. [Google Scholar] [CrossRef][Green Version]
- Wang, X.S.; Shi, Q.; Williams, L.A.; Mao, L.; Cleeland, C.S.; Komaki, R.R.; Mobley, G.M.; Liao, Z. Inflammatory cytokines are associated with the development of symptom burden in patients with NSCLC undergoing concurrent chemoradiation therapy. Brain Behav. Immun. 2010, 24, 968–974. [Google Scholar] [CrossRef]
- Dempsey, L.; Orr, S.; Lane, S.; Scott, A. The clinical nurse specialist’s role in head and neck cancer care: United Kingdom National Multidisciplinary Guidelines. J. Laryngol. Otol. 2016, 130, S212–S215. [Google Scholar] [CrossRef] [PubMed]
- Barata, A.; Wood, W.A.; Choi, S.W.; Jim, H.S. Unmet Needs for Psychosocial Care in Hematologic Malignancies and Hematopoietic Cell Transplant. Curr. Hematol. Malig. Rep. 2016, 11, 280–287. [Google Scholar] [CrossRef] [PubMed]
- Hanna, E.Y.; Mendoza, T.R.; Rosenthal, D.I.; Gunn, G.B.; Sehra, P.; Yucel, E.; Cleeland, C.S. The symptom burden of treatment-naive patients with head and neck cancer. Cancer 2015, 121, 766–773. [Google Scholar] [CrossRef] [PubMed]
- Wulff-Burchfield, E.; Dietrich, M.S.; Ridner, S.; Murphy, B.A. Late systemic symptoms in head and neck cancer survivors. Support. Care Cancer. 2019, 27, 2893–2902. [Google Scholar] [CrossRef]
- Allen-Ayodabo, C.O.; Eskander, A.; Davis, L.E.; Zhao, H.; Mahar, A.L.; Karam, I.; Singh, S.; Gupta, V.; Bubis, L.D.; Moody, L.; et al. Symptom burden among head and neck cancer patients in the first year after diagnosis: Association with primary treatment modality. Oral Oncol. 2019, 99, 104434. [Google Scholar] [CrossRef] [PubMed]
- Wheless, S.A.; McKinney, K.A.; Zanation, A.M. A prospective study of the clinical impact of a multidisciplinary head and neck tumor board. Otolaryngol. Head Neck Surg. 2010, 143, 650–654. [Google Scholar] [CrossRef]
- Eilers, J.; Harris, D.; Henry, K.; Johnson, L.A. Evidence-based interventions for cancer treatment-related mucositis: Putting evidence into practice. Clin. J. Oncol. Nurs. 2014, 18, 80–96. [Google Scholar] [CrossRef]
- Elad, S.; Cheng, K.K.F.; Lalla, R.V.; Yarom, N.; Hong, C.; Logan, R.M.; Bowen, J.; Gibson, R.; Saunders, D.P.; Zadik, Y.; et al. MASCC/ISOO clinical practice guidelines for the management of mucositis secondary to cancer therapy. Cancer 2020, 126, 4423–4431. [Google Scholar] [CrossRef]
- Gosselin, T.; Ginex, P.K.; Backler, C.; Bruce, S.D.; Hutton, A.; Marquez, C.M.; McGee, L.A.; Shaftic, A.M.; Suarez, L.V.; Moriarty, K.A.; et al. ONS Guidelines™ for Cancer Treatment-Related Radiodermatitis. Oncol. Nurs. Forum. 2020, 47, 654–670. [Google Scholar] [CrossRef]
- Plemons, J.M.; Al-Hashimi, I.; Marek, C.L. Managing xerostomia and salivary gland hypofunction: Executive summary of a report from the American Dental Association Council on Scientific Affairs. J. Am. Dent. Assoc. 2014, 145, 867–873. [Google Scholar] [CrossRef]
- Buglione, M.; Cavagnini, R.; Di Rosario, F.; Maddalo, M.; Vassalli, L.; Grisanti, S.; Salgarello, S.; Orlandi, E.; Bossi, P.; Majorana, A.; et al. Oral toxicity management in head and neck cancer patients treated with chemotherapy and radiation: Xerostomia and trismus (Part 2). Literature review and consensus statement. Crit. Rev. Oncol. Hematol. 2016, 102, 47–54. [Google Scholar] [CrossRef] [PubMed]
- Braat, C.; Verduijn, G.M.; van der Stege, H.A.; Offerman, M.P.J.; Peeters, M.A.C.; van Staa, A.; Oldenmenger, W.H. Evaluation of a Nurse-led Aftercare Intervention for Patients With Head and Neck Cancer Treated With Radiotherapy and Cisplatin or Cetuximab. Cancer Nurs. 2022, 45, E436–E446. [Google Scholar] [CrossRef] [PubMed]
- Jones, G.; Rutkowski, N.; Trudel, G.; St-Gelais, C.; Ladouceur, M.; Brunet, J.; Lebel, S. Translating guidelines to practice: A training session about cancer-related fatigue. Curr. Oncol. 2020, 27, e163–e170. [Google Scholar] [CrossRef] [PubMed]
- Howell, D.; Mayo, S.; Currie, S.; Jones, G.; Boyle, M.; Hack, T.; Green, E.; Hoffman, L.; Collacutt, V.; McLeod, D.; et al. Psychosocial health care needs assessment of adult cancer patients: A consensus-based guideline. Support. Care Cancer 2012, 20, 3343–3354. [Google Scholar] [CrossRef] [PubMed]
- Pearson, E.J.; Morris, M.E.; McKinstry, C.E. Cancer-related fatigue: Appraising evidence-based guidelines for screening, assessment and management. Support. Care Cancer 2016, 4, 3935–3942. [Google Scholar] [CrossRef] [PubMed]
- de Leeuw, J.; Prins, J.B.; Uitterhoeve, R.; Merkx, M.A.; Marres, H.A.; van Achterberg, T. Nurse-patient communication in follow-up consultations after head and neck cancer treatment. Cancer Nurs. 2014, 37, E1–E9. [Google Scholar] [CrossRef]
- Li, G.; Zhang, S.-X.; Xu, B. Effects of nurse-led telephone follow-up for discharged patients treated with chemotherapy. Asia Pac. J. Oncol. Nurs. 2014, 1, 46–49. [Google Scholar] [CrossRef]
- Gandhi, A.K.; Roy, S.; Thakar, A.; Sharma, A.; Mohanti, B.K. Symptom Burden and Quality of Life in Advanced Head and Neck Cancer Patients: AIIMS Study of 100 Patients. Indian J. Palliat. Care 2014, 20, 189–193. [Google Scholar] [CrossRef]
- Hauth, F.; Bizu, V.; App, R.; Lautenbacher, H.; Tenev, A.; Bitzer, M.; Malek, N.P.; Zips, D.; Gani, C. Electronic Patient-Reported Outcome Measures in Radiation Oncology: Initial Experience After Workflow Implementation. JMIR Mhealth Uhealth 2019, 7, e12345. [Google Scholar] [CrossRef]
- de Leeuw, J.; Prins, J.B.; Teerenstra, S.; Merkx, M.A.; Marres, H.A.; van Achterberg, T. Nurse-led follow-up care for head and neck cancer patients: A quasi-experimental prospective trial. Support. Care Cancer 2013, 21, 537–547. [Google Scholar] [CrossRef]
- Rosenthal, D.I.; Mendoza, T.R.; Chambers, M.S.; Asper, J.A.; Gning, I.; Kies, M.S.; Weber, R.S.; Lewin, J.S.; Garden, A.S.; Ang, K.K.; et al. Measuring head and neck cancer symptom burden: The development and validation of the M. D. Anderson symptom inventory, head and neck module. Head Neck 2007, 29, 923–931. [Google Scholar] [CrossRef] [PubMed]
- Wells, M.; Donnan, P.T.; Sharp, L.; Ackland, C.; Fletcher, J.; Dewar, J.A. A study to evaluate nurse-led on-treatment review for patients undergoing radiotherapy for head and neck cancer. J. Clin. Nurs. 2008, 17, 1428–1439. [Google Scholar] [CrossRef] [PubMed]
- Support, M.C. Holistic Needs Assessments. Available online: https://www.macmillan.org.uk/healthcare-professionals/innovation-in-cancer-care/holistic-needs-assessment (accessed on 6 December 2021).
- Support, M.C. Health and Wellbeing Clinics and Events. Available online: https://www.macmillan.org.uk/documents/aboutus/health_professionals/recoverypackage/healthandwellbeingclinics.pdf (accessed on 6 December 2021).
- Pisani, P.; Airoldi, M.; Allais, A.; Valletti, P.A.; Battista, M.; Benazzo, M.; Briatore, R.; Cacciola, S.; Cocuzza, S.; Colombo, A.; et al. Metastatic disease in head & neck oncology. Acta Otorhinolaryngol. Ital. 2020, 40 (Suppl. 1), S1–S86. [Google Scholar]
- Bussu, F.; Sali, M.; Gallus, R.; Petrone, G.; Autorino, R.; Santangelo, R.; Pandolfini, M.; Miccichè, F.; Delogu, G.; Almadori, G.; et al. HPV and EBV Infections in Neck Metastases from Occult Primary Squamous Cell Carcinoma: Another Virus-Related Neoplastic Disease in the Head and Neck Region. Ann. Surg. Oncol. 2015, 22 (Suppl. 3), S979–S984. [Google Scholar] [CrossRef]
- Wade, D.M.; Mouncey, P.R.; Richards-Belle, A.; Wulff, J.; Harrison, D.A.; Sadique, M.Z.; Grieve, R.D.; Emerson, L.M.; Mason, A.J.; Aaronovitch, D.; et al. Effect of a Nurse-Led Preventive Psychological Intervention on Symptoms of Posttraumatic Stress Disorder Among Critically Ill Patients: A Randomized Clinical Trial. JAMA 2019, 321, 665–675. [Google Scholar] [CrossRef] [PubMed]
- Clarke, G.M.; Conti, S.; Wolters, A.T.; Steventon, A. Evaluating the impact of healthcare interventions using routine data. BMJ 2019, 365, l2239. [Google Scholar] [CrossRef] [PubMed]
- Amit, M.; Hutcheson, K.; Zaveri, J.; Lewin, J.; Kupferman, M.E.; Hessel, A.C.; Goepfert, R.P.; Gunn, G.B.; Garden, A.S.; Ferraratto, R.; et al. Patient-reported outcomes of symptom burden in patients receiving surgical or nonsurgical treatment for low-intermediate risk oropharyngeal squamous cell carcinoma: A comparative analysis of a prospective registry. Oral Oncol. 2019, 91, 13–20. [Google Scholar] [CrossRef] [PubMed]
- Badr, H.; Herbert, K.; Chhabria, K.; Sandulache, V.C.; Chiao, E.Y.; Wagner, T. Self-management intervention for head and neck cancer couples: Results of a randomized pilot trial. Cancer 2019, 125, 1176–1184. [Google Scholar] [CrossRef]
- Bossi, P.; Di Pede, P.; Guglielmo, M.; Granata, R.; Alfieri, S.; Iacovelli, N.A.; Orlandi, E.; Guzzo, M.; Bianchi, R.; Ferella, L.; et al. Prevalence of Fatigue in Head and Neck Cancer Survivors. Ann. Otol. Rhinol. Laryngol. 2019, 128, 413–419. [Google Scholar] [CrossRef]


| Characteristics | Routine Care | NLC | p-Value |
|---|---|---|---|
| Age (mean, SD) | 63.7 (11.0) | 63.5 (12.0) | 0.62 |
| Gender N (%) Male Female | 55 (76.4) 17 (23.6) | 47 (75.8) 15 (24.2) | 0.93 |
| Marital status N (%) | 0.39 | ||
| Married | 39 (54.2) | 29 (46.7) | |
| Not married | 33 (45.8) | 33 (53.2) | |
| Alcohol N (%) consumption | 0.50 | ||
| Yes | 33 (45.8) | 32 (51.6) | |
| No | 39 (54.2) | 30 (48.4) | |
| Smoking N (%) | |||
| Yes | 26 (36.1) | 14 (22.6) | 0.08 |
| No | 46 (63.9) | 48 (77.4) | |
| Weight before radiotherapy (kg), mean (SD) | 72.6 (13.9) | 71.7 (14.9) | 0.29 |
| Pre-existing conditions N (%) | <0.0001 | ||
| Yes | 64 (88.9) | 39 (62.9) | |
| No | 8 (11.1) | 23 (37.1) | |
| Pre-existing oncological conditions N (%) | <0.000 | ||
| Yes | 21 (29.2) | 20 (32.3) | |
| No | 51 (70.8) | 42 (67.7) | |
| Cancer recurrence N (%) | 0.04 | ||
| Yes | 7 (9.7) | 14 (22.6) | |
| No | 65 (90.3) | 48 (77.4) | |
| Cancer location N (%) | <0.000 | ||
| Oropharynx | 27 (37.5) | 33 (53.2) | |
| Oral cavity | 15 (20.8) | 1 (1.6) | |
| Nasopharynx | 2 (2.8) | 4 (6.4) | |
| Larynx | 11 (15.3) | 3 (4.8) | |
| Hypopharynx | 11 (15.3) | 3 (4.8) | |
| Salivary glands | 4 (5.6) | 8 (12.9) | |
| Nasal cavity | 0 (0.0) | 7 (11.3) | |
| Thyroid | 2 (2.8) | 1 (1.6) | |
| Stage N (%) | 0.14 | ||
| I | 3 (4.2) | 4 (6.4) | |
| II | 9 (12.5) | 9 (14.5) | |
| III | 17 (23.6) | 13 (20.9 | |
| IV | 42 (58.3) | 29 (46.7) | |
| Unknown | 1 (1.4) | 7 (11.3) | |
| Treatment type N (%) | 0.38 | ||
| Radiotherapy only | 6 (8.3) | 8 (12.9) | |
| Radiotherapy and other | 66 (91.6) | 54 (87.1) | |
| Radiotherapy sessions (mean, SD) | 32.2 (1.4) | 32.5 (1.3) | 0.29 |
| Radiation dose (Gy, mean SD) | 63.6 (6.8) | 64.4 (6.7) | 0.68 |
| Radiotherapy interruption | 0.68 | ||
| Yes | 12 (16.7) | 12 (19.3) | |
| No | 60 (83.3) | 50 (80.6) | |
| Tube placement during radiotherapy | 0.02 | ||
| Yes | 29 (59.7) | 14 (77.4) | |
| No | 43 (40.3) | 48 (22.6) | |
| Hospitalization during radiotherapy | 0.37 | ||
| Yes | 12 (16.7) | 7 (11.3) | |
| No | 60 (83.3) | 55 (88.7) | |
| Total | 72 (100.0) | 62 (100.0) |
| Consultations | T0 N = 63 | T1 N = 63 | T2 N = 41 * | |
|---|---|---|---|---|
| Variables | Interventions | n (%) | n (%) | n (%) |
| Nutrition | Screening | 63 (100) | 62 (98.4) | 40 (97.6) |
| Education | 37 (58.7) | 11 (17.5) | 12 (29.3) | |
| Coordination | 19 (30.2) | 13 (20.6) | 9 (22.0) | |
| Smoking | Screening | 61 (96.8) | 59 (93.7) | 34 (83.0) |
| Education | 10 (15.9) | 6 (9.5) | 0 | |
| Coordination | 4 (6.4) | 1 (1.6) | 0 | |
| Pain | Screening | 44 (69.8) | 41 (65.1) | 26 (63.4) |
| Education | 3 (4.8) | 16 (25.4) | 11 (26.8) | |
| Coordination | 4 (6.3) | 0 | 1 (2.4) | |
| Oral cavity status | Screening | 60 (95.2) | 59 (93.7) | NA |
| Education | 60 (95.2) | 46 (73.0) | NA | |
| Coordination | 0 | 0 | NA | |
| Swallowing/chewing capacity | Screening | 57 (90.5) | 51(81.0) | NA |
| Education | 0 | 0 | NA | |
| Coordination | 6 (9.5) | 3 (4.8) | NA |
| Variables | T0 N = 62 | T1 N = 62 | T2 N = 41 | ||||
|---|---|---|---|---|---|---|---|
| n (%) | MED (IQR) | n (%) | MED (IQR) | n (%) | MED (IQR) | ||
| Nutrition | |||||||
| Weight | Screening | 62 (100) | 62 (98.4) | 40 (97.6) | |||
| Kg | 70 (19.9) | 67.2 (17.4) | 67.4 (14.8) | ||||
| Kondrup a | Screening | 57 (90.5) | 2.0 (1.0) | NA | NA | NA | NA |
| Kondrup ≥ 3 | 13 (20.6) | 3.0 (1.0) | |||||
| Smoking | Screening | 61 (96.8) | 59 (93.7) | 34 (83.0) | |||
| Status | Non-smoker | 46 (73.0) | 48 (76.2) | 32 (78.1) | |||
| Smoker | 15 (23.8) | 11 (17.5) | 2 (4.9) | ||||
| Attitude b | Ambivalent | 5 (33.0) | 3 (27.0) | 1 (50.0) | |||
| Motivated | 7 (47.0) | 4 (36.0) | 1 (50.0) | ||||
| Consumption | Screening | 11(73.0) | 10 (91.0) | 2 (100) | |||
| n/day c | 20 (21) | 10.0 (10) | 25 (10) | ||||
| Pain | Screening | 44 (69.8) | 41 (65.1) | 26 (63.4) | |||
| Score EVA d | 0 (3.0) | 2.0 (4.0) | 3.0 (5.0) | ||||
| Oral status | Screening | 60 (95.2) | 59 (93.7) | NA | NA | ||
| Score OAG e | 1.5 (3.0) | 3.0 (3.0) | |||||
| Swallowing/chewing capacity (dysphagia) | Screening | 57 (90.5) | 51 (81.0) | NA | NA | ||
| Absence | 45 (71.4) | 38 (60.3) | |||||
| Presence | 5 (7.9) | 9 (14.3) | |||||
| Dysphagia associated with other symptoms f | 7 (11.1) | 4 (6.4) | |||||
| MDASI-HN Dimensions | T1 (3rd–4th Week) | T2 (5th–6th Week) | ||||||
|---|---|---|---|---|---|---|---|---|
| Mean (RC) | Mean (NLC) | Mean Difference (95% CI) | p-Value | Mean (RC) | Mean (NLC) | Mean Difference (95% CI) | p-Value | |
| Core symptoms | ||||||||
| Pain | 3.4 | 3.0 | 0.40 (−1.35–0.53) | 0.39 | 4.5 | 4.0 | −0.49 (−1.45–0.46) | 0.31 |
| Fatigue | 3.8 | 3.8 | −0.01 (−0.84–0.82) | 0.98 | 4.9 | 4.6 | −0.30 (−1.13–0.52) | 0.47 |
| Nausea | 1.4 | 1.5 | 0.11 (0.66–0.88) | 0.77 | 2.0 | 2.0 | −0.02 (−0.94–0.89) | 0.96 |
| Disturbed sleep | 2.3 | 2.7 | 0.37 (−0.55–1.29) | 0.43 | 2.5 | 2.8 | 0.24 (−0.63–1.12) | 0.58 |
| Being distressed (worried) | 2.7 | 2.9 | 0.18 (−0.72–1.09) | 0.69 | 2.8 | 2.9 | 0.13 (−0.78–1.04) | 0.77 |
| Shortness of breath | 1.9 | 1.9 | 0.06 (−0.75–0.88) | 0.87 | 2.0 | 1.9 | −0.09 (−0.90–0.70) | 0.81 |
| Difficulty remembering | 2.7 | 2.9 | 0.18 (−0.72–1.09) | 0.69 | 2.8 | 2.9 | 0.13 (−0.78–1.04) | 0.77 |
| Lack of appetite | 3.1 | 2.7 | −0.36 (−1.37–0.64) | 0.47 | 4.3 | 3.7 | −0.60 (−1.64–0.43) | 0.25 |
| Drowsiness | 2.3 | 2.2 | −0.07 (−0.84–0.70) | 0.85 | 3.2 | 2.9 | −0.24 (−1.17–0.68) | 0.60 |
| Dry mouth | 4.7 | 4.3 | 0.23 (−1.27–0.81) | 0.45 | 5.1 | 4.9 | −0.23 (−1.27–0.81) | 0.66 |
| Sadness | 2.5 | 2.9 | 0.41 (−0.58–1.39) | 0.41 | 2.7 | 2.9 | 0.18 (−0.84–1.20) | 0.72 |
| Vomiting | 0.6 | 0.7 | 0.02 (−0.55–0.61) | 0.92 | 1.5 | 1.0 | −0.43 (−1.25–0.38) | 0.29 |
| Numbness/tingling | 1.1 | 1.5 | 0.42 (−0.24–1.10) | 0.21 | 1.7 | 1.8 | 0.12 (−0.75–1.00) | 0.77 |
| Head and neck symptoms | ||||||||
| Mucus | 3.5 | 3.7 | 0.21 (−0.78–1.21) | 0.67 | 4.4 | 4.6 | 0.27 (−0.68–1.22) | 0.57 |
| Difficulty with swallowing/chewing | 4.4 | 3.8 | −0.57 (−1.61–0.46) | 0.28 | 5.3 | 4.6 | −0.63 (−1.69–0.42) | 0.24 |
| Coughing/choking | 1.7 | 1.6 | 0.32 (−1.30–0.65) | 0.86 | 1.6 | 1.7 | −0.32 (−1.30–0.65) | 0.51 |
| Difficulty with voice/speech | 3.1 | 3.2 | 0.07 (−0.86–1.02) | 0.87 | 3.9 | 3.9 | −0.02 (−1.05–1.01) | 0.96 |
| Burning/rash | 2.5 | 2.3 | 0.52 (−1.56–0.50 | 0.31 | 4.4 | 3.9 | −0.52 (−1.56–0.50) | 0.31 |
| Constipation | 1.8 | 2.2 | 0.37 (−0.58–1.33) | 0.63 | 2.7 | 2.4 | −0.24 (−1.25–0.77) | 0.63 |
| Problem with tasting food | 4.0 | 3.7 | −0.37 (−1.55–0.79) | 0.53 | 5.8 | 5.1 | −0.64 (−1.83–0.54) | 0.28 |
| Mouth/throat sores | 3.4 | 3.5 | 0.11 (−0.97–1.20) | 0.83 | 4.4 | 4.3 | −0.03 (−1.13–1.06) | 0.94 |
| Problem with teeth or gums | 2.3 | 3.0 | 0.69 (−0.27–1.67) | 0.16 | 2.8 | 3.0 | 0.20 (−0.80–1.21) | 0.69 |
| Interference symptoms | ||||||||
| General activity | 3.4 | 3.4 | 0.07 (−0.88–1.03) | 0.87 | 4.0 | 4.1 | 0.09 (−0.88–1.07) | 0.84 |
| Mood | 2.9 | 3.3 | 0.36 (−0.61–1.33) | 0.46 | 3.5 | 3.3 | 0.36 (−0.61–1.33) | 0.46 |
| Work | 3.8 | 3.4 | −0.46 (−1.58–0.64) | 0.41 | 3.7 | 4.0 | 0.35 (−0.72–1.42) | 0.52 |
| Relationships with others | 2.4 | 2.3 | −0.12 (−0.98–0.73) | 0.77 | 2.8 | 2.7 | −0.11 (−0.98–0.75) | 0.79 |
| Walking | 2.2 | 2.1 | −0.17 (−1.09–0.75) | 0.71 | 2.7 | 1.7 | −0.96 (−1.85–0.08) | 0.03 |
| Joy of living | 2.6 | 3.5 | 0.93 (−0.08–1.95) | 0.07 | 2.6 | 3.3 | 0.66 (−0.35–1.67) | 0.20 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Luta, X.; Colomer-Lahiguera, S.; Martins Cardoso, R.J.; Hof, F.; Savoie, M.; Schuler, C.; Wicht, J.; Fucina, N.; Debarge, P.; Ninane, F.; et al. Nurse-Led Consultation and Symptom Burden in Patients with Head and Neck Cancer: A Comparative Analysis of Routine Clinical Data. Cancers 2022, 14, 1227. https://doi.org/10.3390/cancers14051227
Luta X, Colomer-Lahiguera S, Martins Cardoso RJ, Hof F, Savoie M, Schuler C, Wicht J, Fucina N, Debarge P, Ninane F, et al. Nurse-Led Consultation and Symptom Burden in Patients with Head and Neck Cancer: A Comparative Analysis of Routine Clinical Data. Cancers. 2022; 14(5):1227. https://doi.org/10.3390/cancers14051227
Chicago/Turabian StyleLuta, Xhyljeta, Sara Colomer-Lahiguera, Rodrigo Jose Martins Cardoso, Frank Hof, Manon Savoie, Cosette Schuler, Justine Wicht, Nadia Fucina, Patricia Debarge, Françoise Ninane, and et al. 2022. "Nurse-Led Consultation and Symptom Burden in Patients with Head and Neck Cancer: A Comparative Analysis of Routine Clinical Data" Cancers 14, no. 5: 1227. https://doi.org/10.3390/cancers14051227
APA StyleLuta, X., Colomer-Lahiguera, S., Martins Cardoso, R. J., Hof, F., Savoie, M., Schuler, C., Wicht, J., Fucina, N., Debarge, P., Ninane, F., Bourhis, J., & Eicher, M. (2022). Nurse-Led Consultation and Symptom Burden in Patients with Head and Neck Cancer: A Comparative Analysis of Routine Clinical Data. Cancers, 14(5), 1227. https://doi.org/10.3390/cancers14051227







