Cancer Worry Distribution and Willingness to Undergo Colonoscopy at Three Levels of Hypothetical Cancer Risk—A Population-Based Survey in Sweden
Abstract
Simple Summary
Abstract
1. Introduction
- (1)
- How is cancer worry distributed in the general population in Sweden?
- (2)
- Are cancer worry and the intention to undergo a colonoscopy screening associated?
2. Materials and Methods
2.1. Data Collection
2.2. Measurements
2.3. Statistical Methods
3. Results
3.1. Distribution of Cancer Worry and Possible Determinants
3.1.1. CWS Scores in Respondents without a Personal Cancer History
3.1.2. CWS Score in the Full Population-Based Sample
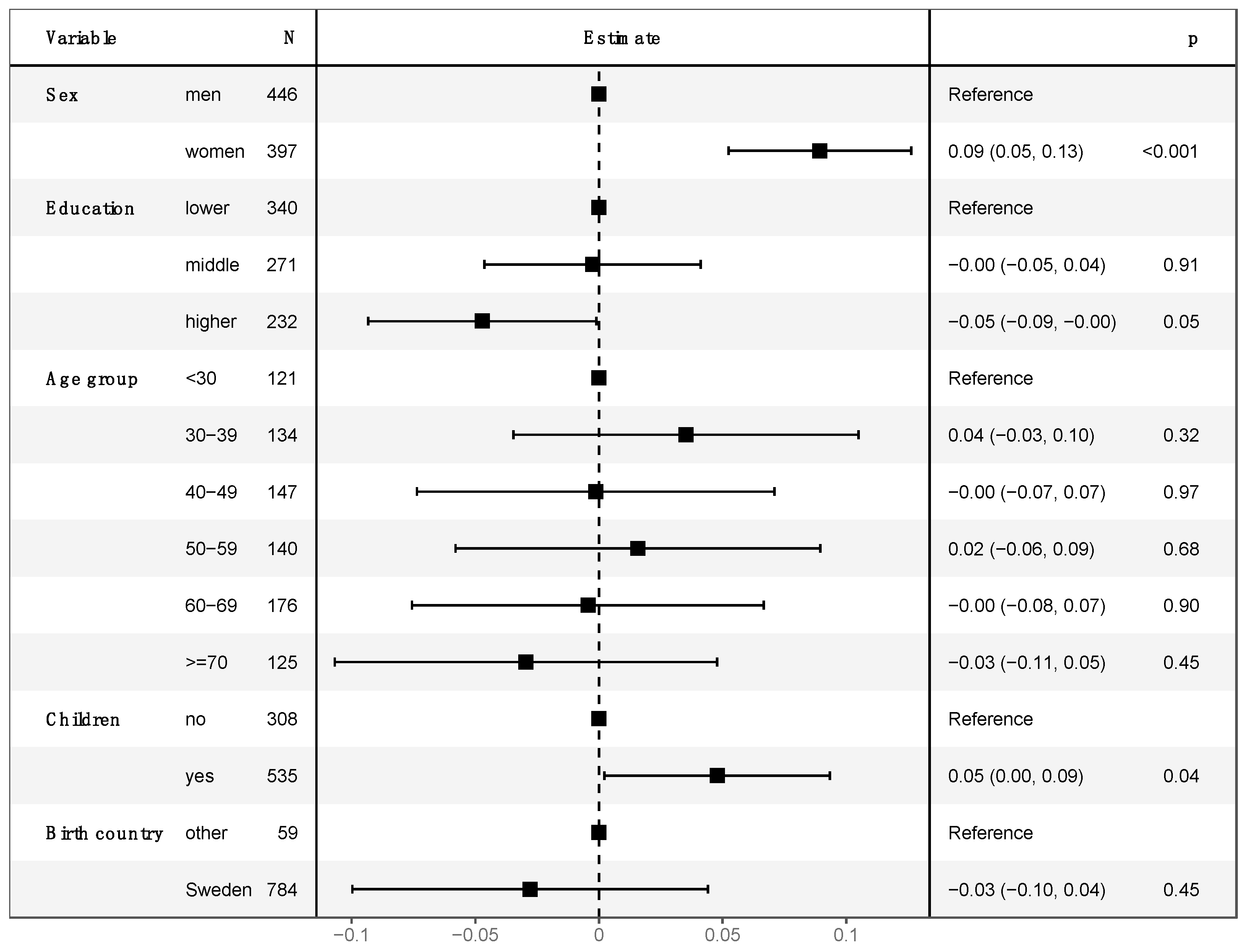
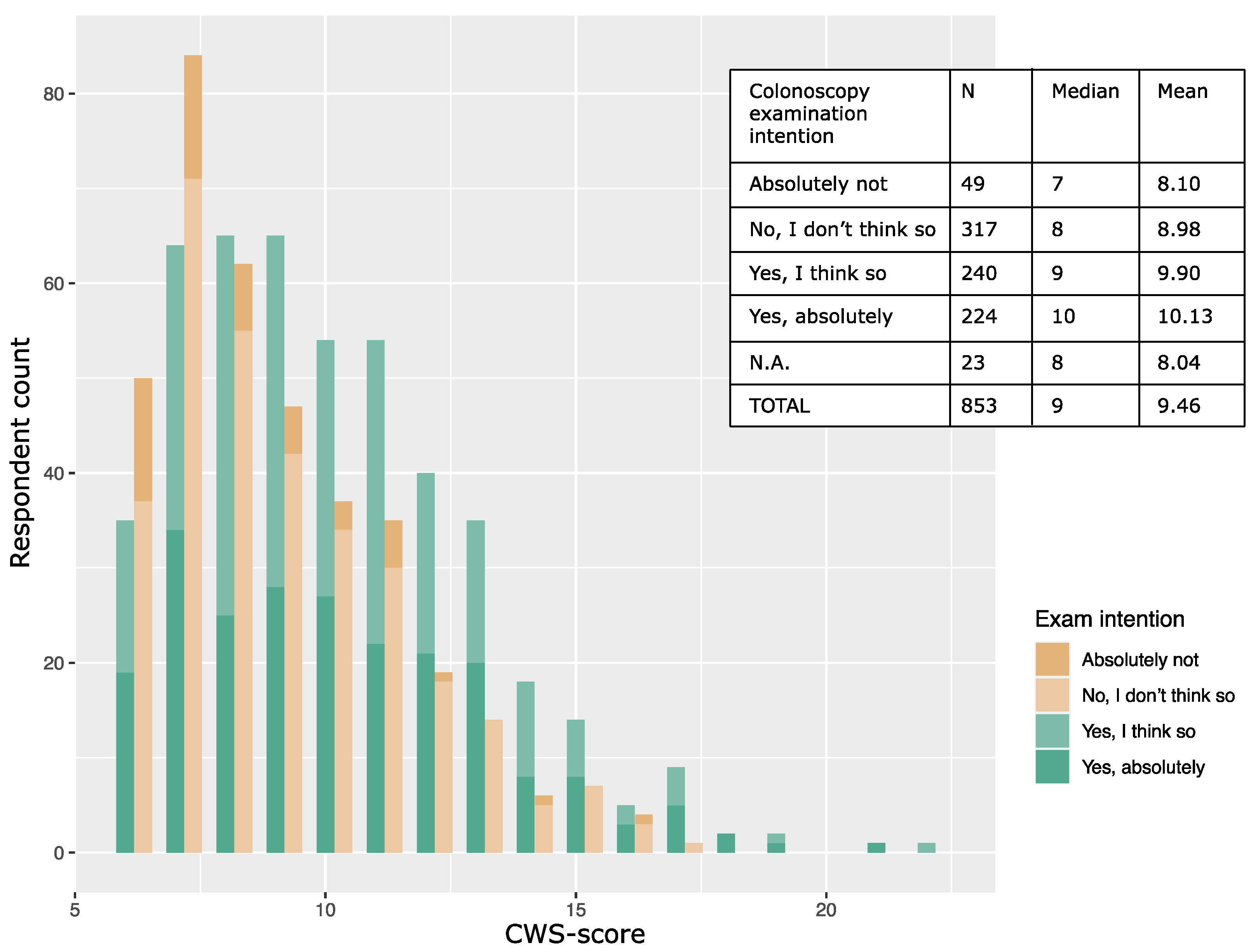
3.2. Cancer Worry and Intention to Participate in Colorectal Cancer Screening
4. Discussion
4.1. Cancer Worry in the General Population and in Those without a Cancer History
4.2. Gender Differences in Cancer Worry
4.3. What Constitutes “Clinically Relevant” Cancer Worry?
4.4. Cancer Worry and Intention to Undergo Colonoscopy
4.5. Study Limitations
4.6. Clinical Implications
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Van de Wal, M.; van de Poll-Franse, L.; Prins, J.; Gielissen, M. Does fear of cancer recurrence differ between cancer types? A study from the population-based PROFILES registry. Psychooncology 2016, 25, 772–778. [Google Scholar] [CrossRef] [PubMed]
- Simard, S.; Thewes, B.; Humphris, G.; Dixon, M.; Hayden, C.; Mireskandari, S.; Ozakinci, G. Fear of cancer recurrence in adult cancer survivors: A systematic review of quantitative studies. J. Cancer Surviv. 2013, 7, 300–322. [Google Scholar] [CrossRef] [PubMed]
- Loescher, L.J. Cancer worry in women with hereditary risk factors for breast cancer. Oncol. Nurs. Forum 2003, 30, 767–772. [Google Scholar] [CrossRef][Green Version]
- Custers, J.A.E.; Kwakkenbos, L.; van de Wal, M.; Prins, J.B.; Thewes, B. Re-validation and screening capacity of the 6-item version of the Cancer Worry Scale. Psychooncology 2018, 27, 2609–2615. [Google Scholar] [CrossRef] [PubMed]
- Jensen, J.D.; Bernat, J.K.; Davis, L.A.; Yale, R. Dispositional cancer worry: Convergent, divergent, and predictive validity of existing scales. J. Psychosoc. Oncol. 2010, 28, 470–489. [Google Scholar] [CrossRef] [PubMed]
- Koitsalu, M.; Eklund, M.; Adolfsson, J.; Sprangers, M.A.G.; Gronberg, H.; Brandberg, Y. Predictors of participation in risk-based prostate cancer screening. PLoS ONE 2018, 13, e0200409. [Google Scholar] [CrossRef] [PubMed]
- Hay, J.L.; Buckley, T.R.; Ostroff, J.S. The role of cancer worry in cancer screening: A theoretical and empirical review of the literature. Psycho-Oncology 2005, 14, 517–534. [Google Scholar] [CrossRef] [PubMed]
- Vrinten, C.; Waller, J.; von Wagner, C.; Wardle, J. Cancer fear: Facilitator and deterrent to participation in colorectal cancer screening. Cancer Epidemiol. Biomark. Prev. 2015, 24, 400–405. [Google Scholar] [CrossRef]
- Custers, J.A.; van den Berg, S.W.; van Laarhoven, H.W.; Bleiker, E.M.; Gielissen, M.F.; Prins, J.B. The Cancer Worry Scale: Detecting fear of recurrence in breast cancer survivors. Cancer Nurs. 2014, 37, E44–E50. [Google Scholar] [CrossRef]
- Yang, Y.; Li, W.; Wen, Y.; Wang, H.; Sun, H.; Liang, W.; Zhang, B.; Humphris, G. Fear of cancer recurrence in adolescent and young adult cancer survivors: A systematic review of the literature. Psychooncology 2019, 28, 675–686. [Google Scholar] [CrossRef]
- Tilburt, J.C.; James, K.M.; Sinicrope, P.S.; Eton, D.T.; Costello, B.A.; Carey, J.; Lane, M.A.; Ehlers, S.L.; Erwin, P.J.; Nowakowski, K.E.; et al. Factors influencing cancer risk perception in high risk populations: A systematic review. Hered. Cancer Clin. Pract. 2011, 9, 2. [Google Scholar] [CrossRef]
- Wardle, J.; Sutton, S.; Williamson, S.; Taylor, T.; McCaffery, K.; Cuzick, J.; Hart, A.; Atkin, W. Psychosocial influences on older adults’ interest in participating in bowel cancer screening. Prev. Med. 2000, 31, 323–334. [Google Scholar] [CrossRef] [PubMed]
- Lerman, C.; Daly, M.; Masny, A.; Balshem, A. Attitudes about genetic testing for breast-ovarian cancer susceptibility. J. Clin. Oncol. 1994, 12, 843–850. [Google Scholar] [CrossRef] [PubMed]
- Douma, K.F.; Aaronson, N.K.; Vasen, H.F.; Gerritsma, M.A.; Gundy, C.M.; Janssen, E.P.; Vriends, A.H.; Cats, A.; Verhoef, S.; Bleiker, E.M. Psychological distress and use of psychosocial support in familial adenomatous polyposis. Psychooncology 2010, 19, 289–298. [Google Scholar] [CrossRef]
- Lerman, C.; Trock, B.; Rimer, B.K.; Jepson, C.; Brody, D.; Boyce, A. Psychological side effects of breast cancer screening. Health Psychol. 1991, 10, 259–267. [Google Scholar] [CrossRef]
- Konings, I.C.; Harinck, F.; Kuenen, M.A.; Sidharta, G.N.; Kieffer, J.M.; Aalfs, C.M.; Poley, J.W.; Smets, E.M.; Wagner, A.; van Rens, A.; et al. Factors associated with cancer worries in individuals participating in annual pancreatic cancer surveillance. Fam. Cancer 2017, 16, 143–151. [Google Scholar] [CrossRef]
- Vrinten, C.; van Jaarsveld, C.H.; Waller, J.; von Wagner, C.; Wardle, J. The structure and demographic correlates of cancer fear. BMC Cancer 2014, 14, 597. [Google Scholar] [CrossRef]
- Bresner, L.; Banach, R.; Rodin, G.; Thabane, L.; Ezzat, S.; Sawka, A.M. Cancer-related worry in Canadian thyroid cancer survivors. J. Clin. Endocrinol. Metab. 2015, 100, 977–985. [Google Scholar] [CrossRef]
- Martinsson, J.; Andreasson, M.; Johansson, J.; Holgersson, E. Technical Report Citizen Panel 31—2018; University of Gothenburg, LORE: Gothenburg, Sweden, 2018. [Google Scholar]
- Andersson, A.; Hawranek, C.; Öfverholm, A.; Ehrencrona, H.; Grill, K.; Hajdarevic, S.; Melin, B.; Tham, E.; Hellquist, B.N.; Rosén, A. Public support for healthcare-mediated disclosure of hereditary cancer risk information: Results from a population-based survey in Sweden. Hered. Cancer Clin. Pract. 2020, 18, 18. [Google Scholar] [CrossRef]
- Custers, J.A.E.; Gielissen, M.F.M.; Janssen, S.H.V.; de Wilt, J.H.W.; Prins, J.B. Fear of cancer recurrence in colorectal cancer survivors. Supportive Care Cancer 2016, 24, 555–562. [Google Scholar] [CrossRef] [PubMed]
- R Core Team. R: A Language and Environment for Statistical Computing; R Project for Statistical Computing: Vienna, Austria, 2018; Available online: https://www.R-project.org/ (accessed on 12 January 2022).
- Timur Taşhan, S.; Uçar, T.; Aksoy Derya, Y.; Nacar, G.; Erci, B. Validity and Reliability of the Turkish Version of the Modified Breast Cancer Worry Scale. Iran. J. Public Health 2018, 47, 1681–1687. [Google Scholar]
- Smits, S.; Boivin, J.; Menon, U.; Brain, K. Influences on anticipated time to ovarian cancer symptom presentation in women at increased risk compared to population risk of ovarian cancer. BMC Cancer 2017, 17, 814. [Google Scholar] [CrossRef] [PubMed]
- Hidalgo, J.L.; Sotos, J.R.; Herraez, M.J.; Rosa, M.C.; Lopez, J.L.; Ortiz, M.P. Factors Associated With Cancer Worry Among People Aged 50 or Older, Spain, 2012–2014. Prev. Chronic Dis. 2015, 12, E226. [Google Scholar] [CrossRef] [PubMed]
- Saya, S.; McIntosh, J.G.; Winship, I.M.; Clendenning, M.; Milton, S.; Oberoi, J.; Dowty, J.G.; Buchanan, D.D.; Jenkins, M.A.; Emery, J.D. A Genomic Test for Colorectal Cancer Risk: Is This Acceptable and Feasible in Primary Care? Public Health Genom. 2020, 23, 110–121. [Google Scholar] [CrossRef]
- Qaderi, S.M.; Swartjes, H.; Vromen, H.; Bremers, A.J.A.; Custers, J.A.E.; de Wilt, J.H.W. Acceptability, quality of life and cost overview of a remote follow-up plan for patients with colorectal cancer. Eur. J. Surg. Oncol. 2021, 47, 1637–1644. [Google Scholar] [CrossRef] [PubMed]
- Custers, J.A.; Kwakkenbos, L.; van der Graaf, W.T.; Prins, J.B.; Gielissen, M.F.; Thewes, B. Not as Stable as We Think: A Descriptive Study of 12 Monthly Assessments of Fear of Cancer Recurrence Among Curatively-Treated Breast Cancer Survivors 0–5 Years After Surgery. Front. Psychol. 2020, 11, 580979. [Google Scholar] [CrossRef]
- Van de Beek, I.; Smets, E.M.A.; Legdeur, M.A.; de Hullu, J.A.; Lok, C.A.R.; Buist, M.R.; Mourits, M.J.E.; Kets, C.M.; van der Kolk, L.E.; Oosterwijk, J.C.; et al. Genetic counseling of patients with ovarian carcinoma: Acceptance, timing, and psychological wellbeing. J. Community Genet. 2020, 11, 183–191. [Google Scholar] [CrossRef] [PubMed]
- Uner, F.O.; Korukcu, O. A prevalence and psychometric study on fear of cancer in women with abnormal cervical cytology undergoing colposcopy. Psycho-Oncology 2020, 29, 1850–1855. [Google Scholar] [CrossRef]
- Brandenbarg, D.; Maass, S.; Geerse, O.P.; Stegmann, M.E.; Handberg, C.; Schroevers, M.J.; Duijts, S.F.A. A systematic review on the prevalence of symptoms of depression, anxiety and distress in long-term cancer survivors: Implications for primary care. Eur. J. Cancer Care 2019, 28, e13086. [Google Scholar] [CrossRef]
- McQueen, A.; Vernon, S.W.; Meissner, H.I.; Rakowski, W. Risk perceptions and worry about cancer: Does gender make a difference? J. Health Commun. 2008, 13, 56–79. [Google Scholar] [CrossRef]
- Vermeer, N.C.A.; van der Valk, M.J.M.; Snijders, H.S.; Vasen, H.F.A.; Gerritsen van der Hoop, A.; Guicherit, O.R.; Liefers, G.J.; van de Velde, C.J.H.; Stiggelbout, A.M.; Peeters, K. Psychological distress and quality of life following positive fecal occult blood testing in colorectal cancer screening. Psycho-Oncology 2020, 29, 1084–1091. [Google Scholar] [CrossRef] [PubMed]
- McBride, K.A.; Ballinger, M.L.; Schlub, T.E.; Young, M.A.; Tattersall, M.H.N.; Kirk, J.; Eeles, R.; Killick, E.; Walker, L.G.; Shanley, S.; et al. Psychosocial morbidity in TP53 mutation carriers: Is whole-body cancer screening beneficial? Fam. Cancer 2017, 16, 423–432. [Google Scholar] [CrossRef]
- Keighley, M.R.; O’Morain, C.; Giacosa, A.; Ashorn, M.; Burroughs, A.; Crespi, M.; Delvaux, M.; Faivre, J.; Hagenmuller, F.; Lamy, V.; et al. Public awareness of risk factors and screening for colorectal cancer in Europe. Eur. J. Cancer Prev. 2004, 13, 257–262. [Google Scholar] [CrossRef]
- Willis, A.M.; Smith, S.K.; Meiser, B.; Ballinger, M.L.; Thomas, D.M.; Young, M.A. Sociodemographic, psychosocial and clinical factors associated with uptake of genetic counselling for hereditary cancer: A systematic review. Clin. Genet. 2017, 92, 121–133. [Google Scholar] [CrossRef]
- Andersen, M.R.; Smith, R.; Meischke, H.; Bowen, D.; Urban, N. Breast Cancer Worry and Mammography Use by Women with and without a Family History in a Population-based Sample. Cancer Epidemiol. Biomark. Prev. 2003, 12, 314–320. [Google Scholar]
- Vrinten, C.; McGregor, L.M.; Heinrich, M.; von Wagner, C.; Waller, J.; Wardle, J.; Black, G.B. What do people fear about cancer? A systematic review and meta-synthesis of cancer fears in the general population. Psycho-Oncology 2017, 26, 1070–1079. [Google Scholar] [CrossRef] [PubMed]
- Digby, J.; O’Carroll, R.E.; Chambers, J.A.; Steele, R.J.C. The impact of hypothetical PErsonalised Risk Information on informed choice and intention to undergo Colorectal Cancer screening colonoscopy in Scotland (PERICCS)-a randomised controlled trial. BMC Med. 2020, 18, 285. [Google Scholar] [CrossRef] [PubMed]
- Hedlin, D. Is there a ‘safe area’ where the nonresponse rate has only a modest effect on bias despite non-ignorable nonresponse? Int. Stat. Rev. 2020, 88, 642–657. [Google Scholar] [CrossRef]
- Sheeran, P. Intention—Behavior Relations: A Conceptual and Empirical Review. Eur. Rev. Soc. Psychol. 2002, 12, 1–36. [Google Scholar] [CrossRef]
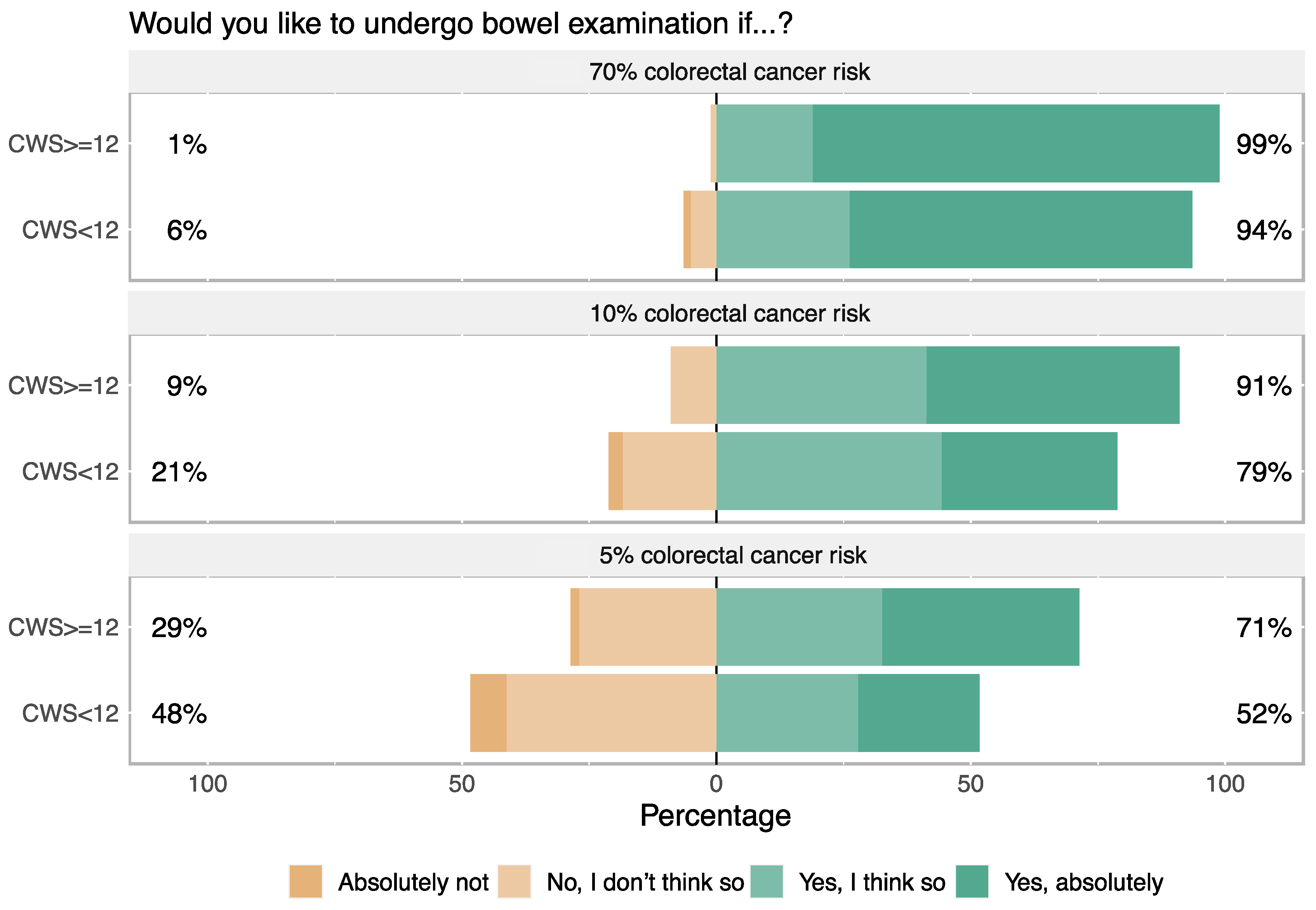
| Total | 6-Item CWS Score | 6-Item CWS Score Interval | ||||||
|---|---|---|---|---|---|---|---|---|
| Subgroup | N | Median (Min–Max) | Mean (Stand. Dev.) | p-Value t-Test/ANOVA | Low (CWS Score 6–11) N (%) † | High (CWS Score 12–24) N (%) † | p-Value Chi-Square Test | |
| Total | - | 853 | 9 (6–22) | 9.46 (2.72) | 674 (79) | 179 (21) | ||
| Gender | Women | 403 | 9 (6–22) | 9.91 (2.89) | 301 (75) | 102 (25) | ||
| Men | 450 | 9 (6–19) | 9.06 (2.49) | 373 (83) | 77 (17) | |||
| p < 0.001 *** | p = 0.004 ** | |||||||
| Age | 18–29 | 123 | 9 (6–19) | 9.28 (2.78) | 98 (80) | 25 (20) | ||
| 30–39 | 135 | 9 (6–22) | 9.65 (3.18) | 102 (76) | 33 (24) | |||
| 40–49 | 153 | 9 (6–17) | 9.45 (2.76) | 120 (78) | 33 (22) | |||
| 50–59 | 141 | 9 (6–17) | 9.67 (2.62) | 107 (76) | 34 (24) | |||
| 60–69 | 176 | 9 (6–17) | 9.41 (2.47) | 144 (82) | 32 (18) | |||
| 70–74 | 125 | 9 (6–18) | 9.28 (2.53) | 103 (82) | 22 (18) | |||
| p = 0.74 | p = 0.61 | |||||||
| Education ‡ | Lower | 344 | 9 (6–22) | 9.50 (2.69) | 267 (78) | 77 (22) | ||
| Middle | 271 | 9 (6–21) | 9.60 (2.81) | 213 (79) | 58 (21) | |||
| Higher | 234 | 9 (6–17) | 9.24 (2.67) | 190 (81) | 44 (19) | |||
| p = 0.31 | p = 0.58 | |||||||
| Country of birth § | Sweden | 785 | 9 (6–22) | 9.43 (2.69) | 622 (79) | 163 (21) | ||
| Other | 14 | 9 (6–19) | 9.68 (3.08) | 46 (77) | 14 (23) | |||
| p = 0.55 | p = 0.75 | |||||||
| Children ¶ | Yes | 540 | 9 (6–21) | 9.59 (2.65) | 419 (78) | 121 (22) | ||
| No | 308 | 8 (6–22) | 9.21 (2.80) | 252 (82) | 56 (18) | |||
| p = 0.06 | p = 0.17 | |||||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hawranek, C.; Maxon, J.; Andersson, A.; Van Guelpen, B.; Hajdarevic, S.; Numan Hellquist, B.; Rosén, A. Cancer Worry Distribution and Willingness to Undergo Colonoscopy at Three Levels of Hypothetical Cancer Risk—A Population-Based Survey in Sweden. Cancers 2022, 14, 918. https://doi.org/10.3390/cancers14040918
Hawranek C, Maxon J, Andersson A, Van Guelpen B, Hajdarevic S, Numan Hellquist B, Rosén A. Cancer Worry Distribution and Willingness to Undergo Colonoscopy at Three Levels of Hypothetical Cancer Risk—A Population-Based Survey in Sweden. Cancers. 2022; 14(4):918. https://doi.org/10.3390/cancers14040918
Chicago/Turabian StyleHawranek, Carolina, Johan Maxon, Andreas Andersson, Bethany Van Guelpen, Senada Hajdarevic, Barbro Numan Hellquist, and Anna Rosén. 2022. "Cancer Worry Distribution and Willingness to Undergo Colonoscopy at Three Levels of Hypothetical Cancer Risk—A Population-Based Survey in Sweden" Cancers 14, no. 4: 918. https://doi.org/10.3390/cancers14040918
APA StyleHawranek, C., Maxon, J., Andersson, A., Van Guelpen, B., Hajdarevic, S., Numan Hellquist, B., & Rosén, A. (2022). Cancer Worry Distribution and Willingness to Undergo Colonoscopy at Three Levels of Hypothetical Cancer Risk—A Population-Based Survey in Sweden. Cancers, 14(4), 918. https://doi.org/10.3390/cancers14040918






