Postoperative Chemoradiotherapy versus Radiotherapy Alone in Major Salivary Gland Cancers: A Stratified Study Based on the External Validation of the Distant Metastasis Risk Score Model
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants of the Study
2.2. Histology
2.3. Treatment and Definitions
2.4. Follow-Up
2.5. Statistical Analysis
3. Results
3.1. The Clinicopathological Characteristics
3.2. Variables Associated with DM in the Major SGM Following PORT
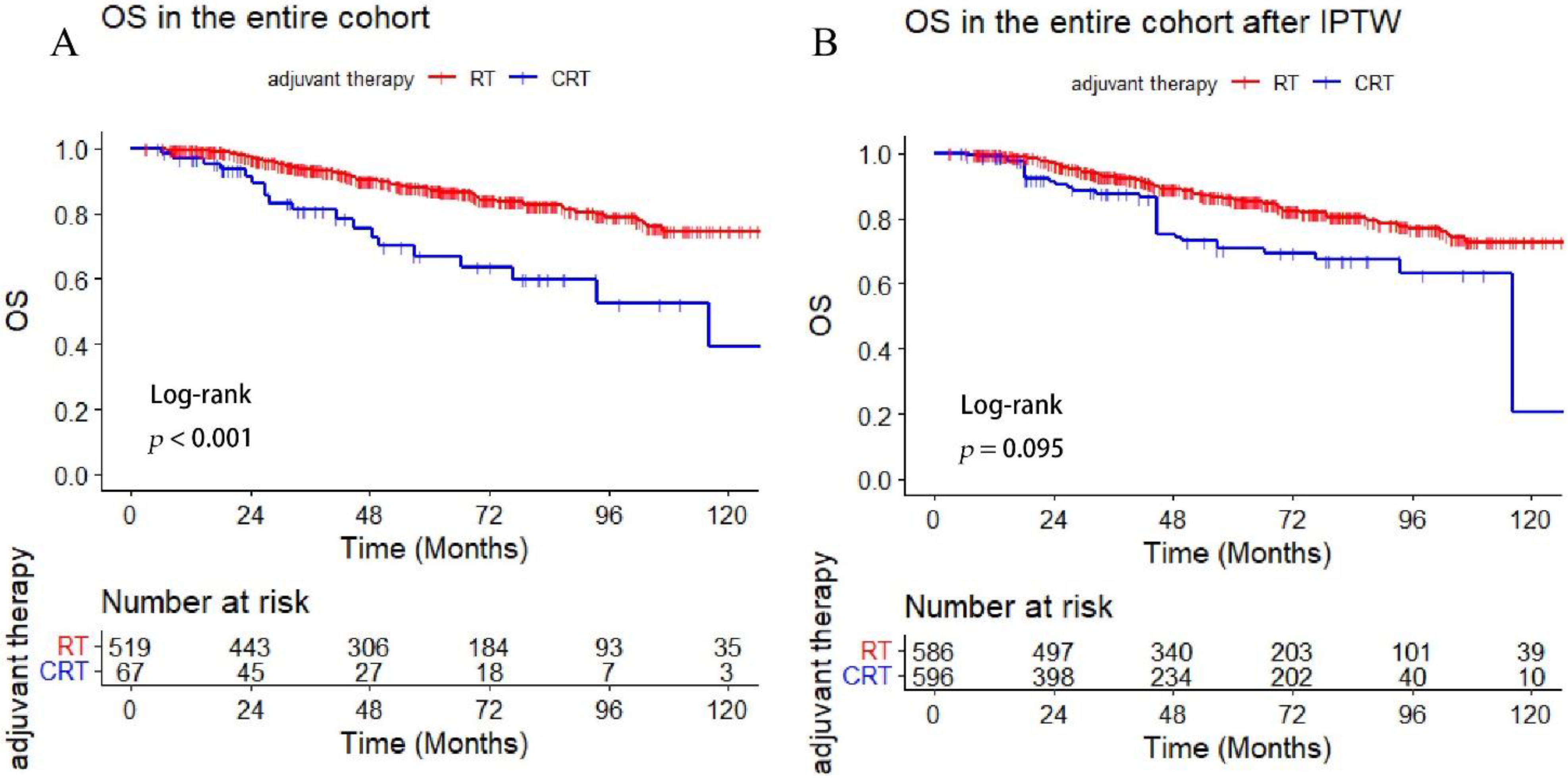
3.3. Comparison of Survival Outcome between the Adjuvant RT and CRT-Group
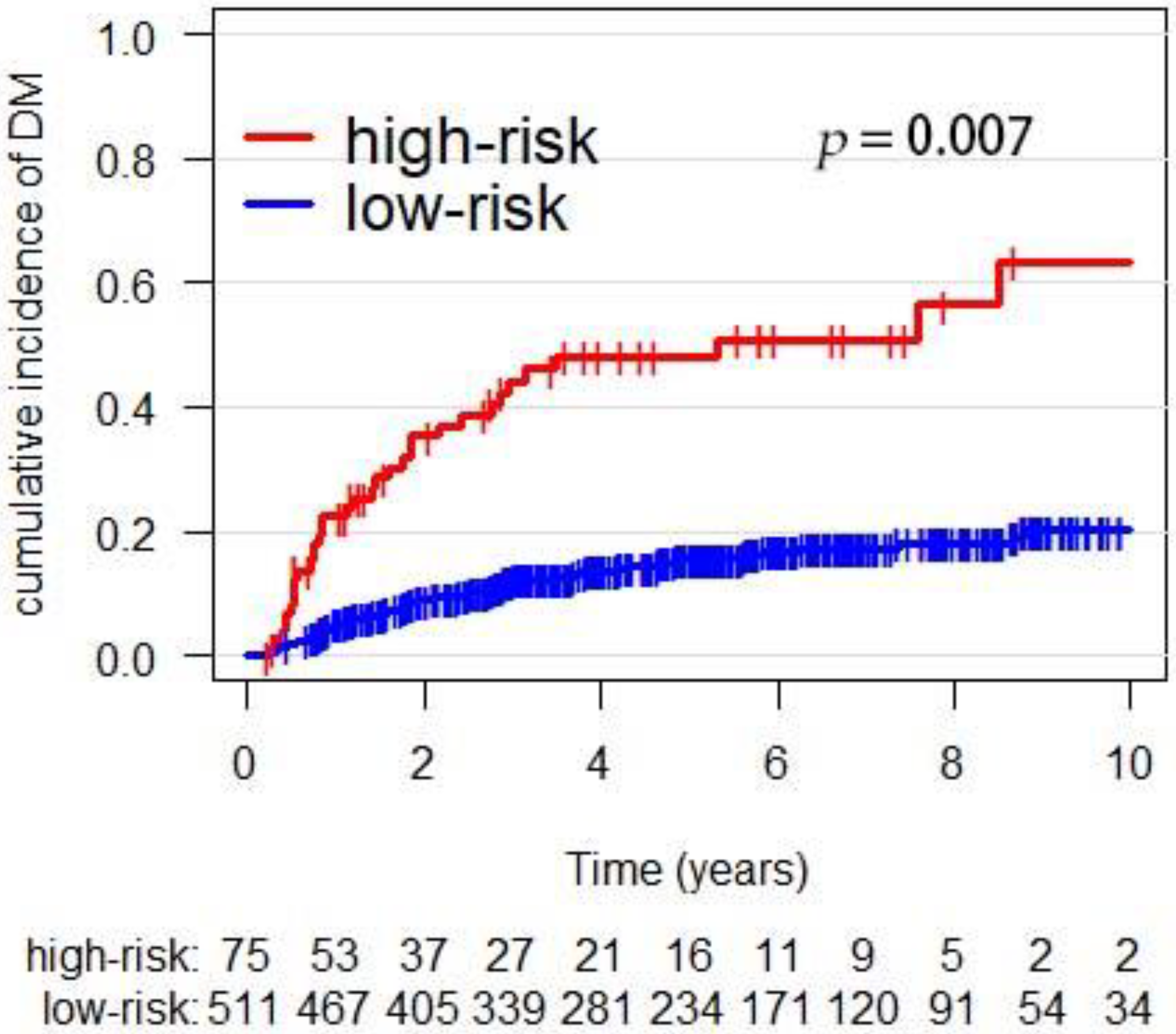
3.4. External Validation of the Prediction-Score Model
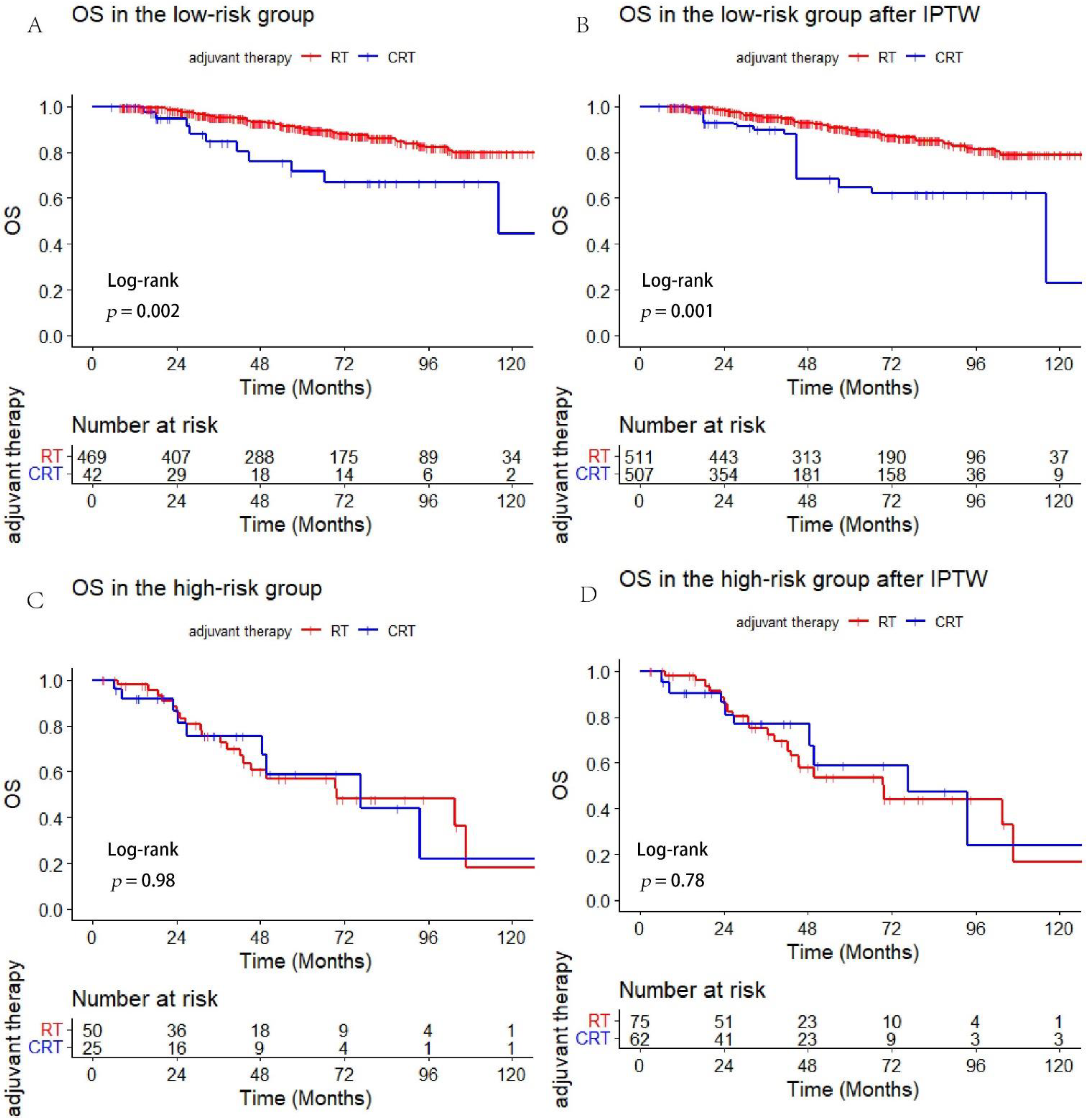
3.5. Association of Adjuvant CRT with Survival in High- and Low-Risk Subgroups
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Eppsteiner, R.W.; Fowlkes, J.W.; Anderson, C.M.; Robinson, R.A.; Pagedar, N.A. Aggressive salivary malignancies at early stage: Outcomes and implications for treatment. Ann. Otol. Rhinol. Laryngol. 2017, 126, 525–529. [Google Scholar] [CrossRef] [PubMed]
- Cheraghlou, S.; Kuo, P.; Mehra, S.; Agogo, G.O.; Bhatia, A.; Husain, Z.A.; Yarbrough, W.G.; Burtness, B.A.; Judson, B.L. Adjuvant therapy in major salivary gland cancers: Analysis of 8580 patients in the national cancer database. Head Neck. 2018, 40, 1343–1355. [Google Scholar] [CrossRef] [PubMed]
- Mahmood, U.; Koshy, M.; Goloubeva, O.; Suntharalingam, M. Adjuvant radiation therapy for high-grade and/or locally advanced major salivary gland tumors. Arch. Otolaryngol.—Head Neck. Surgery 2011, 137, 1025–1030. [Google Scholar] [CrossRef]
- Feinstein, T.M.; Lai, S.Y.; Lenzner, D.; Gooding, W.; Ferris, R.L.; Grandis, J.R.; Myers, E.N.; Johnson, J.T.; Heron, D.E.; Argiris, A.; et al. Prognostic factors in patients with high-risk locally advanced salivary gland cancers treated with surgery and postoperative radiotherapy. Head Neck. 2011, 33, 318–323. [Google Scholar] [CrossRef]
- Mendenhall, W.M.; Morris, C.G.; Amdur, R.J.; Werning, J.W.; Hinerman, R.W.; Villaret, D.B. Radiotherapy alone or combined with surgery for adenoid cystic carcinoma of the head and neck. Head Neck. 2004, 26, 154–162. [Google Scholar] [CrossRef]
- Al-Mamgani, A.; van Rooij, P.; Verduijn, G.M.; Meeuwis, C.A.; Levendag, P.C. Long-term outcomes and quality of life of 186 patients with primary parotid carcinoma treated with surgery and radiotherapy at the Daniel den Hoed Cancer Center. Int. J. Radiat. Oncol. Biol. Physics. 2012, 84, 189–195. [Google Scholar] [CrossRef] [PubMed]
- Mimica, X.; McGill, M.; Hay, A.; Karassawa Zanoni, D.; Shah, J.P.; Wong, R.J.; Ho, A.; Cohen, M.A.; Patel, S.G.; Ganly, I. Distant metastasis of salivary gland cancer: Incidence, management, and outcomes. Cancer. 2020, 126, 2153–2162. [Google Scholar] [CrossRef] [PubMed]
- Hosni, A.; Huang, S.H.; Goldstein, D.; Xu, W.; Chan, B.; Hansen, A.; Weinreb, I.; Bratman, S.V.; Cho, J.; Giuliani, M.; et al. Outcomes and prognostic factors for major salivary gland carcinoma following postoperative radiotherapy. Oral Oncol. 2016, 54, 75–80. [Google Scholar] [CrossRef] [PubMed]
- Cooper, J.S.; Pajak, T.F.; Forastiere, A.A.; Jacobs, J.; Campbell, B.H.; Saxman, S.B.; Kish, J.A.; Kim, H.E.; Cmelak, A.J.; Rotman, M.; et al. Postoperative concurrent radiotherapy and chemotherapy for high-risk squamous-cell carcinoma of the head and neck. N. Engl. J. Med. 2004, 350, 1937–1944. [Google Scholar] [CrossRef] [PubMed]
- Bernier, J.; Domenge, C.; Ozsahin, M.; Matuszewska, K.; Lefèbvre, J.-L.; Greiner, R.H.; Giralt, J.; Maingon, P.; Rolland, F.; Bolla, M.; et al. Postoperative irradiation with or without concomitant chemotherapy for locally advanced head and neck cancer. N. Engl. J. Med. 2004, 350, 1945–1952. [Google Scholar] [CrossRef]
- Seethala, R.R.; Stenman, G. Update from the 4th edition of the world health organization classification of head and neck tumours: Tumors of the salivary gland. Head Neck Pathol. 2017, 11, 55–67. [Google Scholar] [CrossRef] [PubMed]
- Amini, A.; Waxweiler, T.V.; Brower, J.V.; Jones, B.L.; McDermott, J.D.; Raben, D.; Ghosh, D.; Bowles, D.; Karam, S. Association of adjuvant chemoradiotherapy vs radiotherapy alone with survival in patients with resected major salivary gland carcinoma: Data from the national cancer data base. JAMA Otolaryngol. Head Neck Surgery. 2016, 142, 1100–1110. [Google Scholar] [CrossRef]
- Hsieh, C.-E.; Lin, C.-Y.; Lee, L.-Y.; Yang, L.-Y.; Wang, C.-C.; Wang, H.-M.; Chang, J.T.; Fan, K.H.; Liao, C.T.; Yen, T.C.; et al. Adding concurrent chemotherapy to postoperative radiotherapy improves locoregional control but Not overall survival in patients with salivary gland adenoid cystic carcinoma-a propensity score matched study. Radiat. Oncol. 2016, 11, 47. [Google Scholar] [CrossRef] [PubMed]
- Cheraghlou, S.; Schettino, A.; Zogg, C.K.; Otremba, M.D.; Bhatia, A.; Park, H.S.; Osborn, H.A.; Mehra, S.; Yarbrough, W.G.; Judson, B.L.; et al. Adjuvant Chemotherapy Is Associated With Improved Survival for Late-Stage Salivary Squamous Cell Carcinoma. Laryngoscope 2019, 129, 883–889. [Google Scholar] [CrossRef]
- Geiger, J.L.; Ismaila, N.; Beadle, B.; Caudell, J.J.; Chau, N.; Deschler, D.; Glastonbury, C.; Kaufman, M.; Lamarre, E.; Lau, H.Y.; et al. Management of Salivary Gland Malignancy: ASCO Guideline. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2021, 39, 1909–1941. [Google Scholar] [CrossRef]
- Gebhardt, B.J.; Ohr, J.P.; Ferris, R.L.; Duvvuri, U.; Kim, S.; Johnson, J.T.; Heron, D.E.; Clump, D.A., 2nd. Concurrent Chemoradiotherapy in the Adjuvant Treatment of High-risk Primary Salivary Gland Malignancies. Am. J. Clin. Oncol. 2018, 41, 888–893. [Google Scholar] [CrossRef]
- Lukovic, J.; Alfaraj, F.A.; Mierzwa, M.L.; Marta, G.N.; Xu, W.; Su, J.; Moraes, F.Y.; Huang, S.H.; Bratman, S.V.; O’Sullivan, B.; et al. Development and validation of a clinical prediction-score model for distant metastases in major salivary gland carcinoma. Ann. Oncol. Off. J. Eur. Soc. Med. Oncol. 2020, 31, 295–301. [Google Scholar] [CrossRef]
- Austin, P.C. An Introduction to Propensity Score Methods for Reducing the Effects of Confounding in Observational Studies. Multivar. Behav. Res. 2011, 46, 399–424. [Google Scholar] [CrossRef]
- Kim, J.Y.; Lee, S.; Cho, K.J.; Kim, S.Y.; Nam, S.Y.; Choi, S.H.; Roh, J.L.; Choi, E.K.; Kim, J.H.; Song, S.Y.; et al. Treatment results of post-operative radiotherapy in patients with salivary duct carcinoma of the major salivary glands. Br. J. Radiol. 2012, 85, e947–e952. [Google Scholar] [CrossRef]
- Nam, S.J.; Roh, J.-L.; Cho, K.-J.; Choi, S.-H.; Nam, S.Y.; Kim, S.Y. Risk Factors and Survival Associated with Distant Metastasis in Patients with Carcinoma of the Salivary Gland. Ann. Surg. Oncol. 2016, 23, 4376–4383. [Google Scholar] [CrossRef]
- Bakst, R.L.; Glastonbury, C.M.; Parvathaneni, U.; Katabi, N.; Hu, K.S.; Yom, S.S. Perineural Invasion and Perineural Tumor Spread in Head and Neck Cancer. Int. J. Radiat. Oncol. Biol. Phys. 2019, 103, 1109–1124. [Google Scholar] [CrossRef]
- Jegadeesh, N.; Liu, Y.; Prabhu, R.S.; Magliocca, K.R.; Marcus, D.M.; Higgins, K.A.; Vainshtein, J.M.; Trad Wadsworth, J.; Beitler, J.J. Outcomes and prognostic factors in modern era management of major salivary gland cancer. Oral Oncol. 2015, 51, 770–777. [Google Scholar] [CrossRef] [PubMed]
- Mariano, F.V.; da Silva, S.D.; Chulan, T.C.; de Almeida, O.P.; Kowalski, L.P. Clinicopathological factors are predictors of distant metastasis from major salivary gland carcinomas. Int. J. Oral Maxillofac. Surg. 2011, 40, 504–509. [Google Scholar] [CrossRef]
- Terhaard, C.H.J.; Lubsen, H.; Van der Tweel, I.; Hilgers, F.J.M.; Eijkenboom, W.M.H.; Marres, H.A.M.; Tjho-Heslinga, R.E.; de Jong, J.M.; Roodenburg, J.L. Salivary gland carcinoma: Independent prognostic factors for locoregional control, distant metastases, and overall survival: Results of the Dutch head and neck oncology cooperative group. Head Neck. 2004, 26, 681–693. [Google Scholar] [CrossRef] [PubMed]
- Nagliati, M.; Bolner, A.; Vanoni, V.; Tomio, L.; Lay, G.; Murtas, R.; Deidda, M.A.; Madeddu, A.; Delmastro, E.; Verna, R.; et al. Surgery and radiotherapy in the treatment of malignant parotid tumors: A retrospective multicenter study. Tumori 2009, 95, 442–448. [Google Scholar] [CrossRef] [PubMed]
- Mifsud, M.J.; Tanvetyanon, T.; McCaffrey, J.C.; Otto, K.J.; Padhya, T.A.; Kish, J.; Trotti, A.M.; Harrison, L.B.; Caudell, J.J. Adjuvant radiotherapy versus concurrent chemoradiotherapy for the management of high-risk salivary gland carcinomas. Head Neck. 2016, 38, 1628–1633. [Google Scholar] [CrossRef] [PubMed]
- Osborn, V.; Givi, B.; Lee, A.; Sheth, N.; Roden, D.; Schwartz, D.; Schreiber, D. Characterization, treatment and outcomes of salivary ductal carcinoma using the National Cancer Database. Oral Oncol. 2017, 71, 41–46. [Google Scholar] [CrossRef]
- Schoenfeld, J.D.; Sher, D.J.; Norris, C.M.; Haddad, R.I.; Posner, M.R.; Balboni, T.A.; Tishler, R.B. Salivary gland tumors treated with adjuvant intensity-modulated radiotherapy with or without concurrent chemotherapy. Int. J. Radiat. Oncol. Biol. Physics. 2012, 82, 308–314. [Google Scholar] [CrossRef]
- Tanvetyanon, T.; Qin, D.; Padhya, T.; McCaffrey, J.; Zhu, W.; Boulware, D.; DeConti, R.; Trotti, A. Outcomes of postoperative concurrent chemoradiotherapy for locally advanced major salivary gland carcinoma. Arch. Otolaryngol.—Head Neck Surg. 2009, 135, 687–692. [Google Scholar] [CrossRef]
- Tanvetyanon, T.; Fisher, K.; Caudell, J.; Otto, K.; Padhya, T.; Trotti, A. Adjuvant chemoradiotherapy versus with radiotherapy alone for locally advanced salivary gland carcinoma among older patients. Head Neck. 2016, 38, 863–870. [Google Scholar] [CrossRef]
| FUSCC Cohort (n = 586, %) | |
|---|---|
| Sex | |
| Male | 350 (60.3) |
| Female | 236 (39.4) |
| Age (median, range) | 49 (18–83) |
| Tumor Site | |
| Parotid | 421 (72.5) |
| Submandibular/sublingual | 165 (27.5) |
| p T stage | |
| T1–2 | 470 (80.5) |
| T3–4 | 116 (19.5) |
| p N stage | |
| N0 | 406 (68.5) |
| N+ | 180 (31.5) |
| Stage | |
| I | 119 (20.0) |
| II | 219 (37.0) |
| III | 98 (16.9) |
| IVa | 138 (24.1) |
| IVb | 12 (2.0) |
| PNI | |
| Negative | 438 (75.2) |
| Positive | 148 (24.8) |
| LVI | |
| Negative | 548 (93.2) |
| Positive | 38 (6.8) |
| Margin | |
| Negative | 557 (95.1) |
| Positive | 29 (4.9) |
| Pathology | |
| low-risk | 143 (23.6) |
| high-risk | 443 (76.4) |
| PTV (Gy) | 60 (45–70.4) |
| Adjuvant therapy | |
| RT alone | 519 (88.2) |
| CRT | 67 (11.8) |
| Histology | |
| Lymphepithelioma carcinoma (LEC) a | 147 (25.1) |
| Adenoid cystic carcinoma | 125 (21.3) |
| Mucoepidermoid carcinoma | 81 (13.8) |
| Salivary duct carcinoma | 79 (13.5) |
| Acinic cell carcinoma | 37 (6.3) |
| Carcinoma ex-pleomorphic adenoma a | 25 (4.3) |
| Adenocarcinoma | 18 (3.1) |
| Others a | 74 (12.6) |
| Univariable Analysis | Multivariate Analysis | |||
|---|---|---|---|---|
| HR (95% CI) | p-Value | HR (95% CI) | p-Value | |
| Age | 1.02 (1.01–1.04) | <0.001 | 1.01 (1.00–1.03) | 0.05 |
| Gender | 0.109 | |||
| Male | Ref. | |||
| Female | 0.72 (0.48–1.07) | |||
| T stage | 0.087 | |||
| T1–2 | Ref. | |||
| T3–4 | 1.44 (0.95–2.21) | |||
| N stage | <0.001 | <0.001 | ||
| N0 | Ref. | Ref. | ||
| N+ | 4.29 (2.92–6.30) | 3.36 (2.25–5.03) | ||
| Histology | <0.001 | 0.015 | ||
| Low-risk | Ref. | Ref. | ||
| High-risk | 3.88 (1.96–7.65) | 2.51 (1.19–5.30) | ||
| PNI | <0.001 | 0.007 | ||
| Negative | Ref. | Ref. | ||
| Positive | 2.42 (1.65–3.54) | 1.77 (1.168–2.67) | ||
| LVI | <0.001 | 0.004 | ||
| Negative | Ref. | Ref. | ||
| Positive | 3.92 (2.30–6.68) | 2.35 (1.31–4.23) | ||
| Surgical margin | 0.81 | |||
| Negative | Ref. | |||
| Positive | 0.88 (0.32–2.41) | |||
| CRT | 0.001 | 0.7 | ||
| No | Ref. | Ref. | ||
| Yes | 2.42 (1.46–4.03) | 1.11 (0.65–1.89) | ||
| RT (n = 519, %) | CRT (n = 67, %) | p-Value | |
|---|---|---|---|
| Sex | 0.898 | ||
| Male | 309 (59.5) | 41 (61.2) | |
| Female | 210 (40.5) | 26 (38.8) | |
| Age | 47.8 ± 14.7 | 49.0 ± 14.1 | 0.537 |
| T stage | 0.466 | ||
| T1–2 | 419 (80.9) | 51 (76.1) | |
| T3–4 | 100 (19.3) | 16 (23.9) | |
| N stage | <0.001 | ||
| N0 | 394 (75.1) | 12 (17.9) | |
| N+ | 125 (24.9) | 55 (82.1) | |
| PNI | 0.637 | ||
| Negative | 390 (75.1) | 48 (71.6) | |
| Positive | 129 (24.9) | 19 (28.4) | |
| LVI | 0.064 | ||
| Negative | 489 (94.2) | 59 (88.1) | |
| Positive | 30 (5.8) | 8 (11.9) | |
| Margin | 0.128 | ||
| Negative | 496 (95.6) | 61 (91.0) | |
| Positive | 23 (4.4) | 6 (9.0) | |
| Pathology * | <0.001 | ||
| low-risk | 139 (26.8) | 4 (5.9) | |
| high-risk | 380 (73.2) | 63 (94.1) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yan, W.; Huang, L.; Jiang, J.; Shen, C.; Ou, X.; Hu, C. Postoperative Chemoradiotherapy versus Radiotherapy Alone in Major Salivary Gland Cancers: A Stratified Study Based on the External Validation of the Distant Metastasis Risk Score Model. Cancers 2022, 14, 5583. https://doi.org/10.3390/cancers14225583
Yan W, Huang L, Jiang J, Shen C, Ou X, Hu C. Postoperative Chemoradiotherapy versus Radiotherapy Alone in Major Salivary Gland Cancers: A Stratified Study Based on the External Validation of the Distant Metastasis Risk Score Model. Cancers. 2022; 14(22):5583. https://doi.org/10.3390/cancers14225583
Chicago/Turabian StyleYan, Wenbin, Lili Huang, Jianyun Jiang, Chunying Shen, Xiaomin Ou, and Chaosu Hu. 2022. "Postoperative Chemoradiotherapy versus Radiotherapy Alone in Major Salivary Gland Cancers: A Stratified Study Based on the External Validation of the Distant Metastasis Risk Score Model" Cancers 14, no. 22: 5583. https://doi.org/10.3390/cancers14225583
APA StyleYan, W., Huang, L., Jiang, J., Shen, C., Ou, X., & Hu, C. (2022). Postoperative Chemoradiotherapy versus Radiotherapy Alone in Major Salivary Gland Cancers: A Stratified Study Based on the External Validation of the Distant Metastasis Risk Score Model. Cancers, 14(22), 5583. https://doi.org/10.3390/cancers14225583









