Electromotive Enhanced Drug Administration in Oncology: Principles, Evidence, Current and Emerging Applications
Abstract
Simple Summary
Abstract
1. Introduction
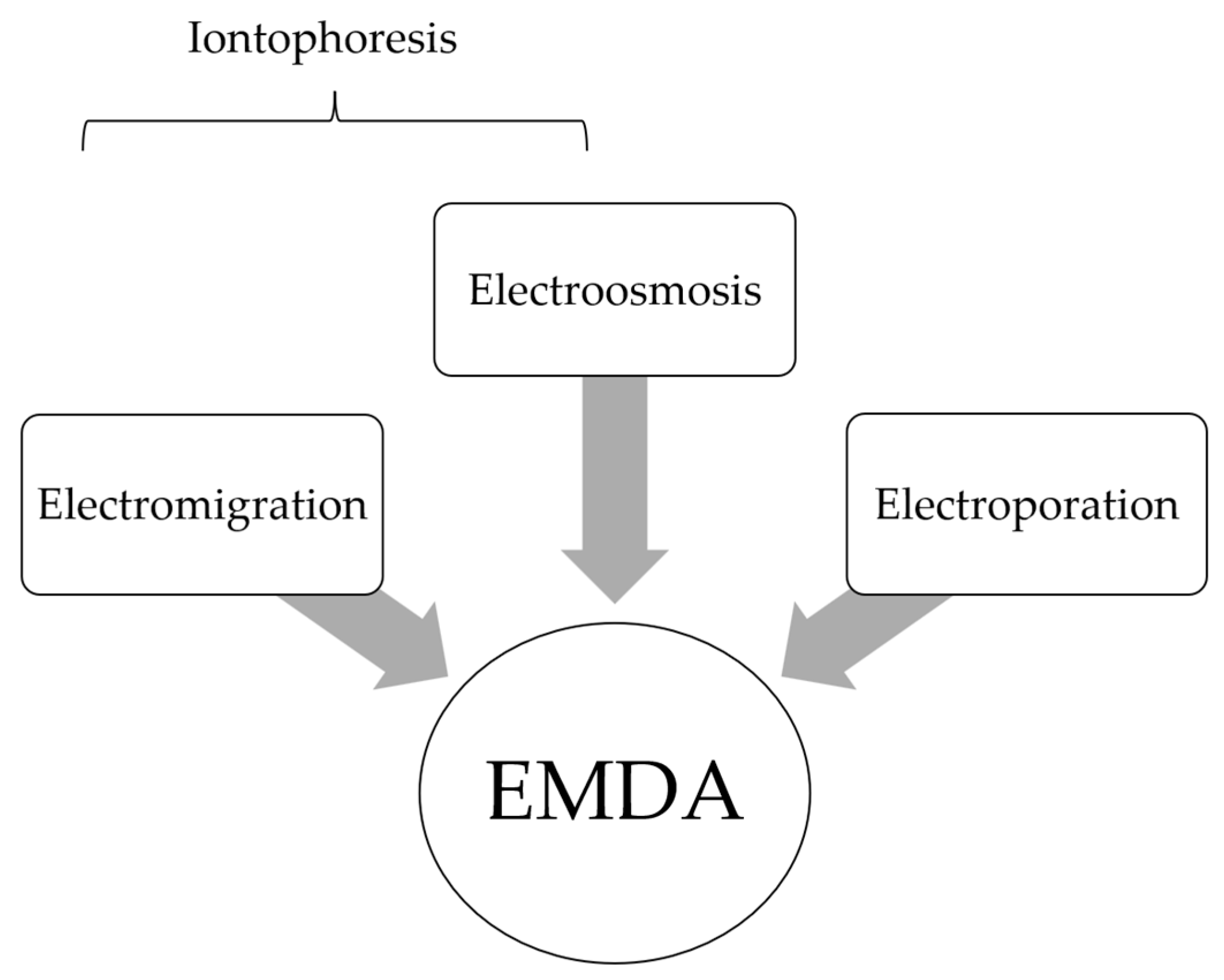
2. Fundamental Principles in EMDA
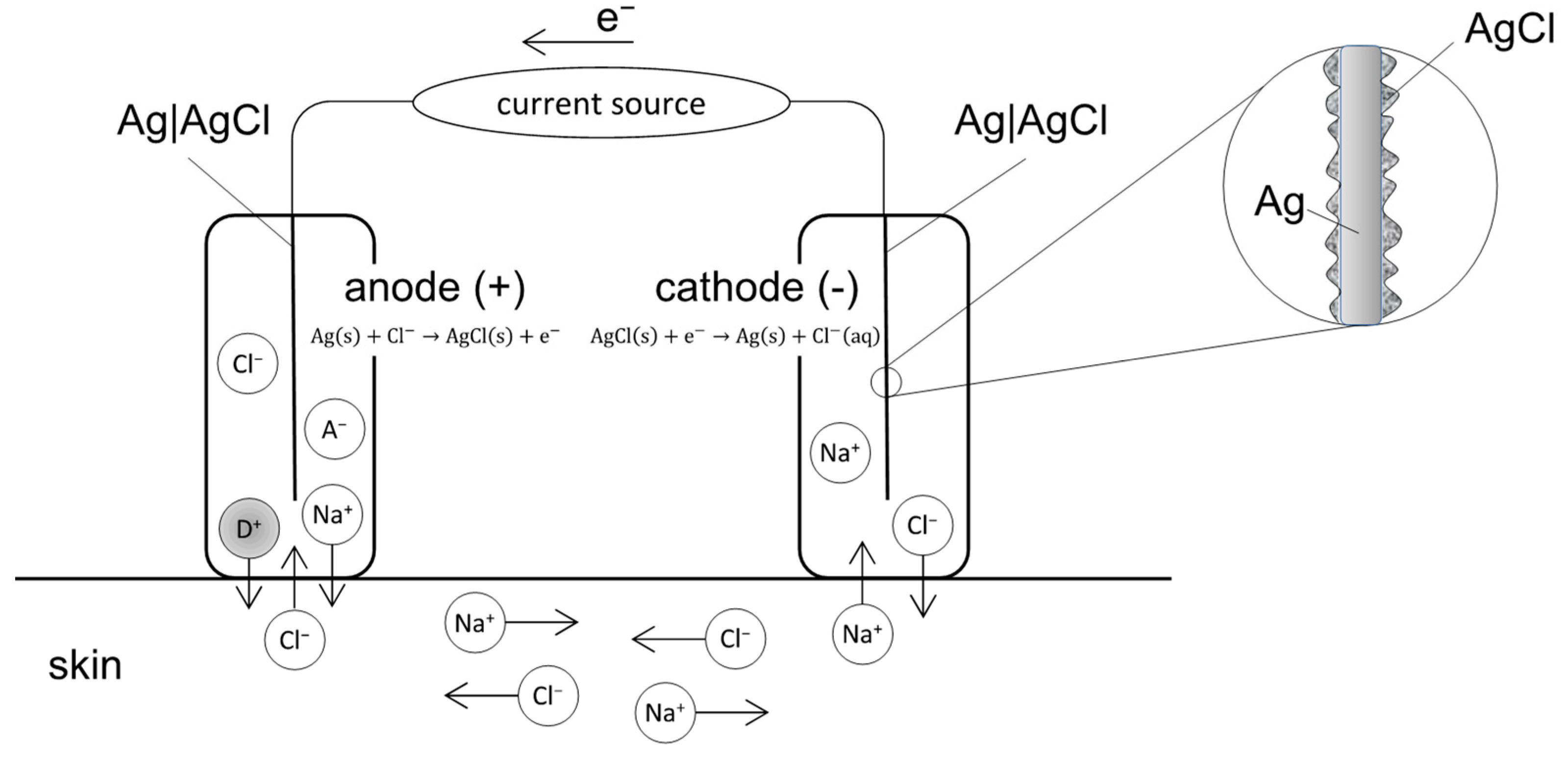
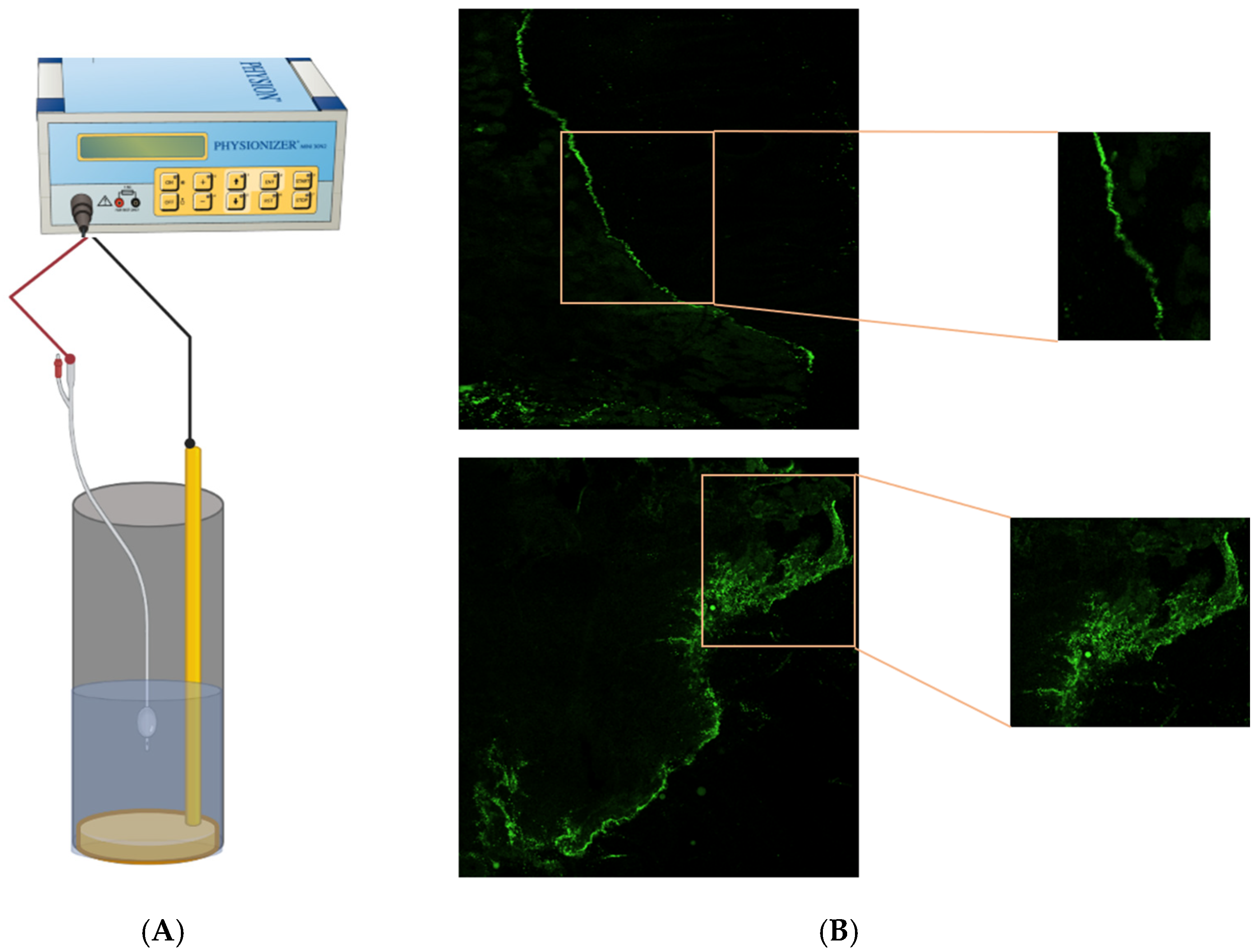
2.1. EMDA Devices
2.2. Relationship between Current Intensity, Ion Valency and EMDA
2.3. Relationship between Drug Physicochemical Properties and EMDA
2.4. Membrane or Barrier Properties and EMDA
3. Pre-Clinical Studies and Clinical Applications of EMDA in Oncology
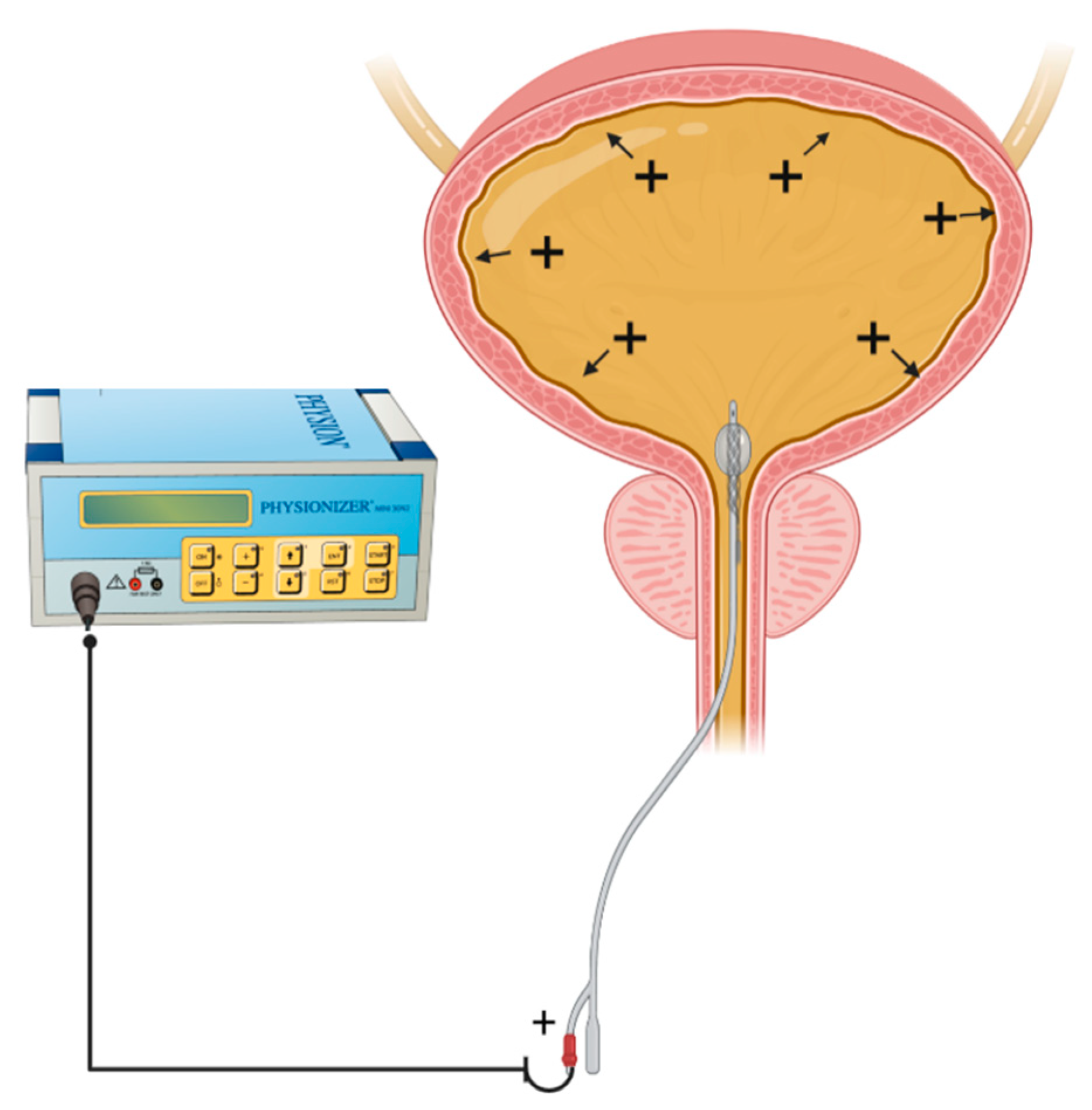
3.1. Urological Cancer
3.2. Skin Cancer
3.3. Ophthalmic Cancer
3.4. Other Cancer Types
4. Future Outlook
4.1. Cellular Pathways Mediating EMDA
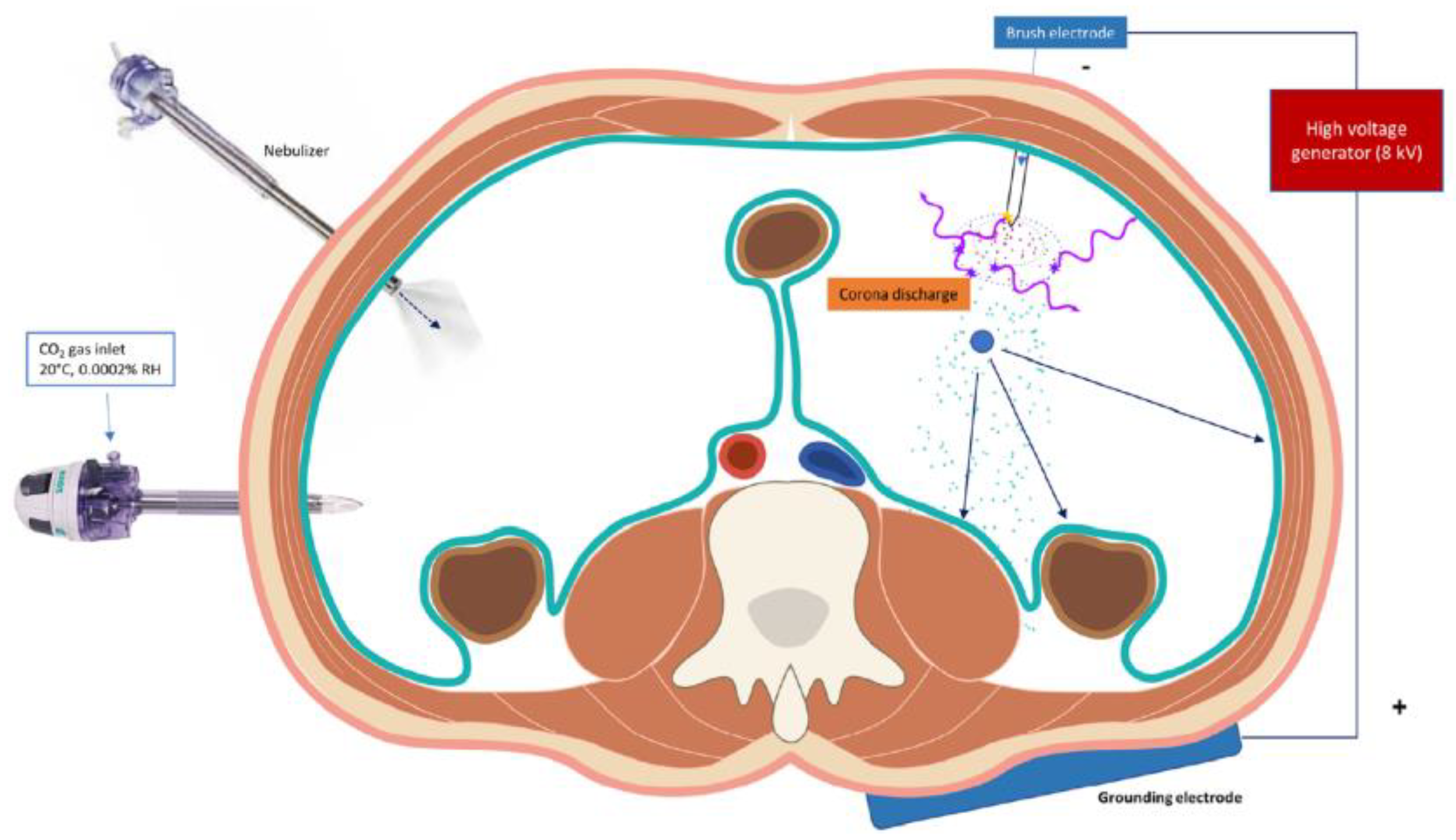
4.2. Extrapolating Current Evidence for Intra-Peritoneal Applications
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Petrak, K. Essential properties of drug-targeting delivery systems. Drug Discov. Today 2005, 10, 1667–1673. [Google Scholar] [CrossRef]
- Collins, J.M. Pharmacologic rationale for regional drug delivery. J. Clin. Oncol. 1984, 2, 498–504. [Google Scholar] [CrossRef] [PubMed]
- Joice, G.A.; Bivalacqua, T.J.; Kates, M. Optimizing pharmacokinetics of intravesical chemotherapy for bladder cancer. Nat. Rev. Urol. 2019, 16, 599–612. [Google Scholar] [CrossRef] [PubMed]
- Ceelen, W.; Demuytere, J.; de Hingh, I. Hyperthermic Intraperitoneal Chemotherapy: A Critical Review. Cancers 2021, 13, 3114. [Google Scholar] [CrossRef] [PubMed]
- Ceelen, W.P.; Flessner, M.F. Intraperitoneal therapy for peritoneal tumors: Biophysics and clinical evidence. Nat. Rev. Clin. Oncol. 2010, 7, 108–115. [Google Scholar] [CrossRef] [PubMed]
- Mishina, T.; Watanabe, H.; Kobayashi, T.; Maegawa, M.; Nakao, M.; Nakagawa, S. Absorption of anticancer drugs through bladder epithelium. Urology 1986, 27, 148–157. [Google Scholar] [CrossRef]
- Lasič, E.; Višnjar, T.; Kreft, M.E. Properties of the Urothelium that Establish the Blood-Urine Barrier and Their Implications for Drug Delivery. Rev. Physiol. Biochem. Pharmacol. 2015, 168, 1–29. [Google Scholar]
- Dedrick, R.L. Theoretical and experimental bases of intraperitoneal chemotherapy. Semin. Oncol. 1985, 12 (Suppl. S4), 1–6. [Google Scholar]
- Heldin, C.H.; Rubin, K.; Pietras, K.; Ostman, A. High interstitial fluid pressure—An obstacle in cancer therapy. Nat. Rev. Cancer 2004, 4, 806–813. [Google Scholar] [CrossRef]
- Hashemi, S.; Sahai, A.; Malde, S. Applications of electromotive drug administration in urology. Urol. Ann. 2020, 12, 301–308. [Google Scholar]
- Charoo, N.A.; Rahman, Z.; Repka, M.A.; Murthy, S.N. Electroporation: An avenue for transdermal drug delivery. Curr. Drug Deliv. 2010, 7, 125–136. [Google Scholar] [CrossRef] [PubMed]
- Turnell, W.J. Therapeutic action of constant current. Proc. R. Soc. Med. 1921, 14, 41–52. [Google Scholar]
- Leduc, S. Introduction of medicinal substances into the depth of tissues by electric current. Ann. D’Eleetrobiol. 1900, 3, 545–560. [Google Scholar]
- Leduc, S. Electric Ions and Their Use in Medicine; Rebman Ltd.: London, UK, 1908.
- Banga, A.K.; Chien, Y.W. Iontophoretic delivery of drugs: Fundamentals, developments and biomedical application. J. Control. Release 1988, 7, 1–24. [Google Scholar] [CrossRef]
- Phipps, J.B.; Padmanabhan, R.B.; Lattin, G.A. Iontophoretic delivery of model inorganic and drug ions. J. Pharm. Sci. 1989, 48, 365. [Google Scholar] [CrossRef] [PubMed]
- Hughes, L.; Maurice, D.M. A fresh look at iontophoresis. Arch. Ophthalmol. 1984, 102, 1825–1829. [Google Scholar] [CrossRef]
- Shelley, W.B.; Horwath, P.; Weidrnan, F.; Pillsbury, D.M. Experimental milaria in man. Production of sweat retention anhidrosis and vesicles by means of iontophoresis. J. Investig. Dermatol. 1948, 11, 275–291. [Google Scholar] [CrossRef]
- Harris, R. Treatment of post-therapeutic neuralgia. Lancet 1957, 269, 378–379. [Google Scholar] [CrossRef]
- Stephen, R.; Miotti, D.; Bettaglio, R.; Rossi, C.; Bonezzi, C. Electromotive administration of a new morphine formulation: Morphine citrate. Artif. Organs 1994, 18, 461–465. [Google Scholar] [CrossRef]
- Gangarosa, L.P.; Park, N.H.; Wiggins, C.A.; Hill, J.M. Increased penetration of nonelectrolytes into mouse skin during iontophoretic water transport (iontohydrokinesis). J. Pharmacol. Exp. Ther. 1980, 212, 377–381. [Google Scholar]
- Prausnitz, M.R.; Bose, V.G.; Langer, R.; Weaver, J.C. Electroporation of mammalian skin: A mechanism to enhance transdermal drug delivery. Proc. Natl. Acad. Sci. USA 1993, 90, 10504–10508. [Google Scholar] [CrossRef] [PubMed]
- Guy, R.H.; Kalia, Y.N.; Delgado-Charro, M.B.; Merino, V.; López, A.; Marro, D. Iontophoresis: Electrorepulsion and electroosmosis. J. Control. Release 2000, 64, 129–132. [Google Scholar] [CrossRef]
- Kalia, Y.N.; Naik, A.; Garrison, J.; Guy, R.H. Iontophoretic drug delivery. Adv. Drug Deliv. Rev. 2004, 56, 619–658. [Google Scholar] [CrossRef] [PubMed]
- Bard, A.J.; Faulkner, L.R. Electrochemical Methods: Fundamentals and Applications, 1st ed.; Wiley: New York, NY, USA, 1980. [Google Scholar]
- Fick, A.V. On liquid diffusion. Lond. Edinb. Dublin Philos. Mag. J. Sci. 1855, 63, 30–39. [Google Scholar] [CrossRef]
- Li, S.K.; Hao, J.; Liddell, M.R. Electrotransport across membranes in biological media: Electrokinetic theories and applications in drug delivery. In Transport in Biological Media; Becker, S., Kuznetsoy, A., Eds.; Elsevier: Philadelphia, PA, USA, 2013; pp. 417–454. [Google Scholar]
- Banga, A.K.; Bose, S.; Ghosh, T.K. Iontophoresis and electroporation: Comparisons and contrasts. Int. J. Pharm. 1999, 179, 1–19. [Google Scholar] [CrossRef]
- Available online: https://www.federalregister.gov/documents/2016/07/26/2016-17609/physical-medicine-devices-reclassification-of-iontophoresis-device-intended-for-any-other-purposes (accessed on 7 October 2022).
- Stillwell, G.K. Electrical stimulation and iontophoresis. In Handbook of Physical Medicine and Rehabilitation, 2nd ed.; Krussen, F.H., Ed.; W.B. Saunders Company: St. Louis, MO, USA, 1971; Chapter 14. [Google Scholar]
- Harding, J.W.; Felix, D. Quantification of angiotensin iontophoresis. J. Neurosci. Methods 1987, 19, 209–215. [Google Scholar] [CrossRef]
- Sloan, J.B.; Soltani, K. Iontophoresis in dermatology. A review. J. Am. Acad. Dermatol. 1986, 15 Pt 1, 671–684. [Google Scholar] [CrossRef]
- Sun, Y.; Siddiqui, O.; Liu, J.C.; Chien, Y.W. Transdermal modulated delivery of polypeptides: Effect of DC pulse wave-form on enhancement. In Proceedings of the 13th International Symposium on Controlled Release of Bioactive Materials, Controlled Release Society, Lincolnshire, IL, USA, 23–25 July 1986. [Google Scholar]
- Gangarosa, L.P.; Park, N.H.; Fong, B.C.; Scott, D.F.; Hill, J.M. Conductivity of drugs used for iontophoresis. J. Pharm. Sci. 1978, 67, 1439–1443. [Google Scholar] [CrossRef]
- Narasimha Murthy, S.; Wiskirchen, D.E.; Bowers, C.P. Iontophoretic drug delivery across human nail. J. Pharm. Sci. 2007, 96, 305–311. [Google Scholar] [CrossRef]
- Hadgraft, J. Skin, the final frontier. Int. J. Pharm. 2001, 224, 1–18. [Google Scholar] [CrossRef]
- Chopra, P.; Hao, J.; Li, S.K. Iontophoretic transport of charged macromolecules across human sclera. Int. J. Pharm. 2010, 388, 107–113. [Google Scholar] [CrossRef] [PubMed]
- Li, S.K.; Liddell, M.R.; Wen, H. Effective electrophoretic mobilities and charges of anti-VEGF proteins determined by capillary zone electrophoresis. J. Pharm. Biomed. Anal. 2011, 55, 603–607. [Google Scholar] [CrossRef] [PubMed]
- Di Stasi, S.M.; Giannantoni, A.; Massoud, R.; Dolci, S.; Navarra, P.; Vespasiani, G.; Stephen, R.L. Electromotive versus passive diffusion of mitomycin C into human bladder wall: Concentration-depth profiles studies. Cancer Res. 1999, 59, 4912–4918. [Google Scholar] [PubMed]
- Lugnani, F.; Mazza, G.; Cerulli, N.; Rossi, C.; Stephen, R. Iontophoresis of drugs in the bladder wall: Equipment and preliminary studies. Artif. Organs 1993, 17, 8–17. [Google Scholar] [CrossRef]
- Di Stasi, S.M.; Vespasiani, G.; Giannantoni, A.; Massoud, R.; Dolci, S.; Micali, F. Electromotive delivery of mitomycin C into human bladder wall. Cancer Res. 1997, 57, 875–880. [Google Scholar]
- Volpe, A.; Racioppi, M.; D’Agostino, D.; Cappa, E.; Filianoti, A.; Bassi, P.F. Mitomycin C for the treatment of bladder cancer. Minerva Urol. Nefrol. 2010, 62, 133–144. [Google Scholar]
- Gürpinar, T.; Truong, L.D.; Wong, H.Y.; Griffith, D.P. Electromotive drug administration to the urinary bladder: An animal model and preliminary results. J. Urol. 1996, 156, 1496–1501. [Google Scholar] [CrossRef]
- Di Stasi, S.M.; Giannantoni, A.; Stephen, R.L.; Capelli, G.; Navarra, P.; Massoud, R.; Vespasiani, G. Intravesical electromotive mitomycin C versus passive transport mitomycin C for high risk superficial bladder cancer: A prospective randomized study. J. Urol. 2003, 170, 777–782. [Google Scholar] [CrossRef]
- Di Stasi, S.M.; Giannantoni, A.; Giurioli, A.; Valenti, M.; Zampa, G.; Storti, L.; Attisani, F.; De Carolis, A.; Capelli, G.; Vespasiani, G.; et al. Sequential BCG and electromotive mitomycin versus BCG alone for high-risk superficial bladder cancer: A randomised controlled trial. Lancet Oncol. 2006, 7, 43–51. [Google Scholar] [CrossRef]
- Di Stasi, S.M.; Valenti, M.; Verri, C.; Liberati, E.; Giurioli, A.; Leprini, G.; Masedu, F.; Ricci, A.R.; Micali, F.; Vespasiani, G. Electromotive instillation of mitomycin immediately before transurethral resection for patients with primary urothelial non-muscle invasive bladder cancer: A randomised controlled trial. Lancet Oncol. 2011, 12, 871–879. [Google Scholar] [CrossRef]
- Brausi, M.; Campo, B.; Pizzocaro, G.; Rigatti, P.; Parma, A.; Mazza, G.; Vicini, A.; Stephen, R. Intravesical electromotive administration of drugs for treatment of superficial bladder cancer: A comparative Phase II study. Urology 1998, 51, 506–509. [Google Scholar] [CrossRef]
- Riedl, C.R.; Knoll, M.; Plas, E.; Pflüger, H. Intravesical electromotive drug administration technique: Preliminary results and side effects. J. Urol. 1998, 159, 1851–1856. [Google Scholar] [CrossRef]
- Gan, C.; Amery, S.; Chatterton, K.; Khan, M.S.; Thomas, K.; O’Brien, T. Sequential bacillus Calmette-Guérin/Electromotive Drug Administration of Mitomycin C as the Standard Intravesical Regimen in High Risk Nonmuscle Invasive Bladder Cancer: 2-Year Outcomes. J. Urol. 2016, 195, 1697–1703. [Google Scholar] [CrossRef] [PubMed]
- Carando, R.; Zazzara, M.; Cotrufo, S.; Ludovico, G.M. Intravesical Treatment with Electro-Mediated Administration of Mytomicin C as Prophylaxis for Intermediate and High-Risk Nonmuscle-Invasive Bladder Cancer: A Retrospective Multicenter Study. Urol. Int. 2019, 103, 285–290. [Google Scholar] [CrossRef]
- Carando, R.; Soldini, E.; Cotrufo, S.; Zazzara, M.; Ludovico, G.M. Electro-mediated drug administration of mitomycin C in preventing non-muscle-invasive bladder cancer recurrence and progression after transurethral resection of the bladder tumour in intermediate- and high-risk patients. Arab. J. Urol. 2020, 19, 71–77. [Google Scholar] [CrossRef]
- Zazzara, M.; Nazaraj, A.; Scarcia, M.; Cardo, G.; Carando, R.; Ludovico, G.M. Electromotive Drug Administration of Mitomycin C (EMDA/MMC) versus Intravesical Immunotherapy with Bacillus Calmette-Guérin (BCG) in Intermediate and High Risk Non Muscle Invasive Bladder Cancer. Urol. Int. 2021, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Colombo, R.; Brausi, M.; Da Pozzo, L.; Salonia, A.; Montorsi, F.; Scattoni, V.; Roscigno, M.; Rigatti, P. Thermo-chemotherapy and electromotive drug administration of mitomycin C in superficial bladder cancer eradication. a pilot study on marker lesion. Eur. Urol. 2001, 39, 95–100. [Google Scholar] [CrossRef]
- Decaestecker, K.; Lumen, N.; Van Camp, C.; Oosterlinck, W. Single ablative intravesical electromotive mitomycin C administration for small non-muscle-invasive bladder cancer: A prospective study. Acta Clin. Belg. 2018, 73, 324–327. [Google Scholar] [CrossRef]
- Racioppi, M.; Di Gianfrancesco, L.; Ragonese, M.; Palermo, G.; Sacco, E.; Bassi, P.F. ElectroMotive drug administration (EMDA) of Mitomycin C as first-line salvage therapy in high risk "BCG failure" non muscle invasive bladder cancer: 3 years follow-up outcomes. BMC Cancer 2018, 18, 1224. [Google Scholar] [CrossRef]
- Marks, R. The stratum corneum barrier: The final frontier. J. Nutr. 2004, 134, 2017S–2021S. [Google Scholar] [CrossRef]
- Yokouchi, M.; Kubo, A. Maintenance of tight junction barrier integrity in cell turnover and skin diseases. Exp. Dermatol. 2018, 27, 876–883. [Google Scholar] [CrossRef] [PubMed]
- Luxenberg, M.N.; Guthrie, T.H., Jr. Chemotherapy of basal cell and squamous cell carcinoma of the eyelids and periorbital tissues. Ophthalmology 1986, 93, 504–510. [Google Scholar] [CrossRef]
- Chang, B.K.; Guthrie, T.H., Jr.; Hayakawa, K.; Gangarosa, L.P., Sr. A pilot study of iontophoretic cisplatin chemotherapy of basal and squamous cell carcinomas of the skin. Arch. Dermatol. 1993, 129, 425–427. [Google Scholar] [CrossRef] [PubMed]
- Bacro, T.R.; Holladay, E.B.; Stith, M.J.; Maize, J.C.; Smith, C.M. Iontophoresis treatment of basal cell carcinoma with cisplatin: A case report. Cancer Detect. Prev. 2000, 24, 610–619. [Google Scholar]
- Welch, M.L.; Grabski, W.J.; McCollough, M.L.; Skelton, H.G.; Smith, K.J.; Menon, P.A.; Anderson, L.L. 5-fluorouracil iontophoretic therapy for Bowen’s disease. J. Am. Acad. Dermatol. 1997, 36 Pt 1, 956–958. [Google Scholar] [CrossRef]
- Tsuji, T. Bleomycin iontophoretic therapy for verrucous carcinoma. Arch. Dermatol. 1991, 127, 973–975. [Google Scholar] [CrossRef]
- Smith, K.J.; Konzelman, J.L.; Lombardo, F.A.; Skelton, H.G., 3rd; Holland, T.T.; Yeager, J.; Wagner, K.F.; Oster, C.N.; Chung, R. Iontophoresis of vinblastine into normal skin and for treatment of Kaposi’s sarcoma in human immunodeficiency virus-positive patients. The Military Medical Consortium for Applied Retroviral Research. Arch. Dermatol. 1992, 128, 1365–1370. [Google Scholar] [CrossRef]
- Taveira, S.F.; Nomizo, A.; Lopez, R.F. Effect of the iontophoresis of a chitosan gel on doxorubicin skin penetration and cytotoxicity. J. Control. Release 2009, 134, 35–40. [Google Scholar] [CrossRef]
- Huber, L.A.; Pereira, T.A.; Ramos, D.N.; Rezende, L.C.; Emery, F.S.; Sobral, L.M.; Leopoldino, A.M.; Lopez, R.F. Topical Skin Cancer Therapy Using Doxorubicin-Loaded Cationic Lipid Nanoparticles and lontophoresis. J. Biomed. Nanotechnol. 2015, 11, 1975–1988. [Google Scholar] [CrossRef]
- Kigasawa, K.; Kajimoto, K.; Nakamura, T.; Hama, S.; Kanamura, K.; Harashima, H.; Kogure, K. Noninvasive and efficient transdermal delivery of CpG-oligodeoxynucleotide for cancer immunotherapy. J. Control. Release 2011, 150, 256–265. [Google Scholar] [CrossRef]
- Toyoda, M.; Hama, S.; Ikeda, Y.; Nagasaki, Y.; Kogure, K. Anti-cancer vaccination by transdermal delivery of antigen peptide-loaded nanogels via iontophoresis. Int. J. Pharm. 2015, 483, 110–114. [Google Scholar] [CrossRef] [PubMed]
- Labala, S.; Mandapalli, P.K.; Kurumaddali, A.; Venuganti, V.V. Layer-by-layer polymer coated gold nanoparticles for topical delivery of imatinib mesylate to treat melanoma. Mol. Pharm. 2015, 12, 878–888. [Google Scholar] [CrossRef] [PubMed]
- Venuganti, V.V.; Saraswathy, M.; Dwivedi, C.; Kaushik, R.S.; Perumal, O.P. Topical gene silencing by iontophoretic delivery of an antisense oligonucleotide-dendrimer nanocomplex: The proof of concept in a skin cancer mouse model. Nanoscale 2015, 7, 3903–3914. [Google Scholar] [CrossRef] [PubMed]
- Lemos, C.N.; de Souza, J.G.; Simão, P.S.; Lopez, R.F. Iontophoresis Improved Growth Reduction of Invasive Squamous Cell Carcinoma in Topical Photodynamic Therapy. PLoS ONE 2016, 11, e0145922. [Google Scholar] [CrossRef] [PubMed]
- Labala, S.; Jose, A.; Venuganti, V.V. Transcutaneous iontophoretic delivery of STAT3 siRNA using layer-by-layer chitosan coated gold nanoparticles to treat melanoma. Colloids Surf. B Biointerfaces 2016, 146, 188–197. [Google Scholar] [CrossRef]
- Labala, S.; Jose, A.; Chawla, S.R.; Khan, M.S.; Bhatnagar, S.; Kulkarni, O.P.; Venuganti, V.V.K. Effective melanoma cancer suppression by iontophoretic co-delivery of STAT3 siRNA and imatinib using gold nanoparticles. Int. J. Pharm. 2017, 525, 407–417. [Google Scholar] [CrossRef]
- Petrilli, R.; Eloy, J.O.; Saggioro, F.P.; Chesca, D.L.; de Souza, M.C.; Dias, M.V.S.; da Silva, L.L.P.; Lee, R.J.; Lopez, R.F.V. Skin cancer treatment effectiveness is improved by iontophoresis of EGFR-targeted liposomes containing 5-FU compared with subcutaneous injection. J. Control. Release 2018, 283, 151–162. [Google Scholar] [CrossRef]
- Hasan, T.; Ortel, B.; Moor, A.C.; Pogue, B.W. Photodynamic therapy of cancer. In Cancer Medicine, 9th ed.; Kufe, D.W., Pollock, R.E., Weichselbaum, R.R., Bast, R.C., Gansler, T.S., Holland, J.F., Frei, E., Eds.; B.C. Decker: Hamilton, ON, USA, 2017; p. 537. [Google Scholar]
- Lopez, R.F.R.; Bentley, M.V.; Begoña Delgado-Charro, M.; Guy, R.H. Optimization of aminolevulinic acid delivery by iontophoresis. J. Control. Release 2003, 88, 65–70. [Google Scholar] [CrossRef]
- HBodde, E.; Roemele, P.E.H.; Star, W.M. Quantification of topically delivered 5-aminolevulinic acid by iontophoresis across ex vivo human stratum corneum. Photochem. Photobiol. 2002, 75, 418–423. [Google Scholar]
- Merclin, N.; Bramer, T.; Edsman, K. Iontophoretic delivery of 5-aminolevulinic acid and its methyl ester using a carbopol gel as vehicle. J. Control. Release 2004, 98, 57–65. [Google Scholar] [CrossRef]
- Fang, J.Y.; Lee, W.R.; Shen, S.C.; Fang, Y.P.; Hu, C.H. Enhancement of topical 5-aminolaevulinic acid delivery by erbium:YAG laser and microdermabrasion: A comparison with iontophoresis and electroporation. Br. J. Dermatol. 2004, 151, 132–140. [Google Scholar] [CrossRef] [PubMed]
- Perez, V.L.; Wirostko, B.; Korenfeld, M.; From, S.; Raizman, M. Ophthalmic Drug Delivery Using Iontophoresis: Recent Clinical Applications. J. Ocul. Pharmacol. Ther. 2020, 36, 75–87. [Google Scholar] [CrossRef] [PubMed]
- Hayden, B.; Jockovich, M.E.; Murray, T.G.; Kralinger, M.T.; Voigt, M.; Hernandez, E.; Feuer, W.; Parel, J.M. Iontophoretic delivery of carboplatin in a murine model of retinoblastoma. Investig. Ophthalmol. Vis. Sci. 2006, 47, 3717–3721. [Google Scholar] [CrossRef]
- Hayden, B.C.; Jockovich, M.E.; Murray, T.G.; Voigt, M.; Milne, P.; Kralinger, M.; Feuer, W.J.; Hernandez, E.; Parel, J.M. Pharmacokinetics of systemic versus focal Carboplatin chemotherapy in the rabbit eye: Possible implication in the treatment of retinoblastoma. Investig. Ophthalmol. Vis. Sci. 2004, 45, 3644–3649. [Google Scholar] [CrossRef]
- Eljarrat-Binstock, E.; Domb, A.J.; Orucov, F.; Dagan, A.; Frucht-Pery, J.; Pe’Er, J. In vitro and in vivo evaluation of carboplatin delivery to the eye using hydrogel-iontophoresis. Curr. Eye Res. 2008, 33, 269–275. [Google Scholar] [CrossRef]
- Gratieri, T.; Kalia, Y.N. Targeted local simultaneous iontophoresis of chemotherapeutics for topical therapy of head and neck cancers. Int. J. Pharm. 2014, 460, 24–27. [Google Scholar] [CrossRef]
- Matos, B.N.; Pereira, M.N.; Bravo, M.O.; Cunha-Filho, M.; Saldanha-Araújo, F.; Gratieri, T.; Gelfuso, G.M. Chitosan nanoparticles loading oxaliplatin as a mucoadhesive topical treatment of oral tumors: Iontophoresis further enhances drug delivery ex vivo. Int. J. Biol. Macromol. 2020, 154, 1265–1275. [Google Scholar] [CrossRef] [PubMed]
- Komuro, M.; Suzuki, K.; Kanebako, M.; Kawahara, T.; Otoi, T.; Kitazato, K.; Inagi, T.; Makino, K.; Toi, M.; Terada, H. Novel iontophoretic administration method for local therapy of breast cancer. J. Control. Release 2013, 168, 298–306. [Google Scholar] [CrossRef]
- Byrne, J.D.; Jajja, M.R.; O’Neill, A.T.; Bickford, L.R.; Keeler, A.W.; Hyder, N.; Wagner, K.; Deal, A.; Little, R.E.; Moffitt, R.A.; et al. Local iontophoretic administration of cytotoxic therapies to solid tumors. Sci. Trans. Med. 2015, 7, 273ra14. [Google Scholar] [CrossRef]
- Byrne, J.D.; Jajja, M.R.; Schorzman, A.N.; Keeler, A.W.; Luft, J.C.; Zamboni, W.C.; DeSimone, J.M.; Yeh, J.J. Iontophoretic device delivery for the localized treatment of pancreatic ductal adenocarcinoma. Proc. Natl. Acad. Sci. USA 2016, 113, 2200–2205. [Google Scholar] [CrossRef]
- Byrne, J.D.; Jajja, M.R.; O’Neill, A.T.; Schorzman, A.N.; Keeler, A.W.; Luft, J.C.; Zamboni, W.C.; Desimone, J.M.; Yeh, J.J. Impact of formulation on the iontophoretic delivery of the FOLFIRINOX regimen for the treatment of pancreatic cancer. Cancer Chemother. Pharmacol. 2018, 81, 991–998. [Google Scholar] [CrossRef] [PubMed]
- Hama, S.; Kimura, Y.; Mikami, A.; Shiota, K.; Toyoda, M.; Tamura, A.; Nagasaki, Y.; Kanamura, K.; Kajimoto, K.; Kogure, K. Electric stimulus opens intercellular spaces in skin. J. Biol. Chem. 2014, 289, 2450–2456. [Google Scholar] [CrossRef] [PubMed]
- Rominiyi, O.; Vanderlinden, A.; Clenton, S.J.; Bridgewater, C.; Al-Tamimi, Y.; Collis, S.J. Tumour treating fields therapy for glioblastoma: Current advances and future directions. Br. J. Cancer. 2021, 124, 697–709, Epub 2020, 125, 623. [Google Scholar] [CrossRef] [PubMed]
- Mun, E.J.; Babiker, H.M.; Weinberg, U.; Kirson, E.D.; Von Hoff, D.D. Tumor-Treating Fields: A Fourth Modality in Cancer Treatment. Clin. Cancer Res. 2018, 24, 266–275. [Google Scholar] [CrossRef]
- Tan, A.C.; Ashley, D.M.; López, G.Y.; Malinzak, M.; Friedman, H.S.; Khasraw, M. Management of glioblastoma: State of the art and future directions. CA Cancer J. Clin. 2020, 70, 299–312. [Google Scholar] [CrossRef]
- Aguilar, A.A.; Ho, M.C.; Chang, E.; Carlson, K.W.; Natarajan, A.; Marciano, T.; Bomzon, Z.; Patel, C.B. Permeabilizing Cell Membranes with Electric Fields. Cancers 2021, 13, 2283. [Google Scholar] [CrossRef] [PubMed]
- Rahimi-Gorji, M.; Van de Sande, L.; Debbaut, C.; Ghorbaniasl, G.; Braet, H.; Cosyns, S.; Remaut, K.; Willaert, W.; Ceelen, W. Intraperitoneal aerosolized drug delivery: Technology, recent developments, and future outlook. Adv. Drug Deliv. Rev. 2020, 160, 105–114. [Google Scholar] [CrossRef] [PubMed]
- Van de Sande, L.; Cosyns, S.; Willaert, W.; Ceelen, W. Albumin-based cancer therapeutics for intraperitoneal drug delivery: A review. Drug Deliv. 2020, 27, 40–53. [Google Scholar] [CrossRef]
- Van de Sande, L.; Rahimi-Gorji, M.; Giordano, S.; Davoli, E.; Matteo, C.; Detlefsen, S.; D’Herde, K.; Braet, H.; Shariati, M.; Remaut, K.; et al. Electrostatic Intraperitoneal Aerosol Delivery of Nanoparticles: Proof of Concept and Preclinical Validation. Adv. Healthc. Mater. 2020, 9, e2000655. [Google Scholar] [CrossRef]
| Study/Y | Design | Clinical Context | Control Group | Intervention Group | Oncological Outcomes (Compare Control vs. Intervention) | |||
|---|---|---|---|---|---|---|---|---|
| No. of Patients | Treatment Regime | No. of Patients | Treatment Regime | Histological pCR | Recurrence and Survival | |||
| Adjuvant Treatment in NMIBC | ||||||||
| Brausi 1998 [47] | Multi-center Cohort | Primary or recurrent stage Ta or T1, Grade 1 or 2 TCC | 12 | 40 mg PD MMC (2 h dwell time) weekly over 8 weeks | 15 | 40 mg EMDA-MMC 15 mA over 20 min wkly over 8 wks | 41.6% vs. 40% | RR: 60% vs. 33% DFI: 10.5 vs. 14.5 months |
| Riedl 1998 [48] | Single arm prospective | Ta or T1 TCC | NA | NA | 22 | 40 mg EMDA-MMC 15 mA 20 min weekly over 4 weeks | 56.6% (no control, at 14 months) | RR: 44% |
| Di Stasi 2003 [44] | Multi-center RCT | Ta or T1 TCC | 36 36 | A: 40 mg PD MMC (1 h dwell time) weekly over 6 weeks +/− additional course for non-responders B: BCG alone (2 h dwell time) | 36 | 40 mg EMDA-MMC 20 mA 30 min weekly over 6 wks +/− additional course for non-responders | 31% (Control A) vs. 58% (at 6 months) | RR: 75% and 52.8 (Controls A and B) vs. 52.8% DFI: 19.5 and 26 (Controls A and B) vs. 35 months * |
| Di Stasi 2006 [45] | Multi-center RCT | Primary or recurrent stage Ta or T1 TCC | 105 | BCG alone (2 h dwell time) weekly over 6 wks In CR, followed by BCG maintenance (to 10 months) | 107 | BCG (2 h dwell time) weekly over 2 weeks + 40 mg EMDA-MMC 20 mA 30 min weekly over 3 weeks In CR, followed by BCG + EMDA-MMC maintenance (to 10 months) | 57.1% vs. 69% | RR: 57.9% vs. 41.9% * DFI: 21 vs. 69 months * DSS: 83.8% vs. 94.4% * |
| Gan 2016 [49] | Single arm prospective | High risk NMIBC | NA | NA | 107 | BCG (2 h dwell time) + 40 mg EMDA-MMC 20 mA 30 min weekly over 3 wks | 71% (No control, at 1 year) | NR |
| Carando 2019# [50] | Retrospective single-arm | Intermediate and high risk NMIBC | NA | NA | 65 | 40 mg EMDA-MMC 20 to 23 mA 30 min weekly over 8 weeks | 82% (at 6 months) | NR |
| Carando 2020# [51] | Retrospective single-arm | Adjuvant setting Intermediate and high risk NMIBC | NA | NA | 101 | 40 mg EMDA-MMC 20 to 23 mA 30 min weekly over 8 weeks Followed by EMDA-MMC maintenance (up to 12 months) | 75% (at 6 months) | PFS (at 3 months): 94% PFS (at 6 months): 90% |
| Zazzara 2021# [52] | Retrospective cohort | Adjuvant setting Intermediate and high risk NMIBC | 104 | BCG alone (1.5 to 2 h dwell time) weekly over 6-weeks Followed by BCG maintenance (up to 12 months) | 140 | 40 mg EMDA-MMC 20 to 23 mA 30 min weekly over 8 weeks Followed by EMDA-MMC maintenance (up to 12 months) | NR | RR: 20 vs. 14% (at maintenance phase) |
| Neoadjuvant treatment in NMIBC | ||||||||
| Colombo 2001 [53] | Single center Cohort | Ta or T1, Grade 1 or 2, <2 cm tumors | 36 | 40 mg PD MMC (1 h dwell time) weekly over 4 wks | 15 | 40 mg EMDA-MMC 15 mA 20 min weekly over 4 wks | 27.7% vs. 40% | NR |
| Di Stasi 2011 [46] | Multi-center RCT | Neoadjuvant setting Ta or T1 TCC | 116 119 | A: TURBT alone B: TURBT + 40 mg PD MMC (1 h dwell time) | 117 | Pre-TURBT 40 mg EMDA-MMC 20 mA 30 min | NA | RR: 64% and 59% (Controls A and B) vs. 44% * DFI: 2 and 16 (Controls A and B) vs. 52 months * |
| Decae- Stecker 2018 [54] | Single-arm prospective | Neoadjuvant setting Primary or recurrent stage Ta or T1, tumor <2 cm | NA | NA | 36 | Pre-TURBT 60 mg EMDA-MMC 25 mA 25 min | 25% (no control,1 year) | NR |
| Other | ||||||||
| Racioppi 2018 [55] | Single-arm prospective | Salvage bladder-sparing BCG-refractory High grade NMIBC | NA | NA | 26 | 40 mg EMDA-MMC 20 mA 30 min weekly over 6 wks Followed by maintenance over 6 months | NA | High-grade disease-free rate 61.5% |
| Study/Year | Design | Clinical Context | No. of Patients | Drug | EMDA Protocol | Oncological Outcomes |
|---|---|---|---|---|---|---|
| Luxenberg 1986 [58] | Case series | BCC or SCC (non-surgical candidates treated with systemic chemotherapy) Tumor site: Peri-orbital EMDA applied to recurrent or residual lesions after systemic chemotherapy | 5 | Cisplatin | 0.5 to 1.5 mA DC over 20 min | PR in all patients |
| Chang 1993 [59] | Case series | BCC or SCC (non-surgical candidates) Tumor site: Face and others | 12 | Cisplatin | 0.5 to 1.5 mA DC over 20 to 30 min Daily for 3–5 days or once/week | CR 26.7% of lesions, PR 46.7%, minimal response 26.7% Factors associated with CR:
|
| Bacro 2000 [60] | Case report | BCC Tumor site: Lower extremity | 1 | Cisplatin | Daily for 5 days over 4 weeks | CR was achieved |
| Welch 1997 [61] | Case series | SCC-in-situ Tumor site: Head and Neck, Trunk, Upper and lower extremities | 26 | 5-FU | 4 mA DC over 10 min Twice/week over 4 weeks | CR in 96% (25 out of 26) patients |
| Tsuji 1991 [62] | Case study | Verrucous carcinoma (not surgical candidate) Tumor site: Lips | 1 | Bleomycin | 2 mA DC over 30 min Trice/week over 2 weeks | CR was achieved Recurrence-free at 6 months |
| Smith 1992 [63] | Case series | Kaposi’s sarcoma in HIV patients Tumor site: Upper and lower extremities, face, tongue | 4 | Vinblastine sulfate | 4 mA DC over 10 min once | CR in 29% of lesions, PR in 71% |
| Study/Year | Study Type | Experimental Tissue/Skin Cancer Type | Drug Used | Carrier (if Any) or Carrier Solution | EMDA Protocol | Outcomes |
|---|---|---|---|---|---|---|
| Taveira 2009 [64] | In vitro | Porcine skin Melanoma (Murine cells) | Doxorubicin (cationic) | Chitosan gel (cationic), water, HEC gel (non-ionic) | 0.5 mA/cm2 over 6 hrs | Doxorubicin with chitosan gel carrier result in improved epidermal penetration (in porcine skin) with EMDA when compared with passive diffusion (PD) 3-fold increase in doxorubicin cytotoxicity with EMDA in cancer cells over PD |
| Huber 2015 [65] | In vitro and In vivo | Porcine skin SCC (Murine model) | Doxorubicin (cationic) | Solid lipid nanoparticles (SLN), water | 0.5 mA/cm2 over 6 h | 2-fold improvement of doxorubicin penetration (in porcine skin) with SLN carrier Significantly improved SCC tumour inhibition using doxorubicin-SLN with EMDA over PD |
| Kigasawa 2011 [66] | In vivo | Melanoma (Murine model) | CpG-ODN | NaCl | 0.3 mA/cm2 over 1 h | Improved penetration and distribution of drug in epidermis and dermis with EMDA over PD Significant inhibition of tumour growth with local or distant application of drug with EMDA compared with non-treated controls |
| Toyoda 2015 [67] | In vitro and In vivo | Porcine Skin Melanoma (Murine model) | Cancer-antigen gp-100 peptide | PEG modified Nanogels | 0.4 mA/cm2 over 1 h | Significant increase in no. of Langerhans cells in epidermis with gp100 and EMDA over non-treated controls Significant tumour growth suppression with antigen peptide-loaded nanogels and EMDA over non-treated controls |
| Labala 2015 [68] | In vitro | Porcine skin Melanoma (Murine cells) | Imatinib mesylate | Gold nanoparticles | 0.47 mA/cm2 over 4 h | 6.2-fold improvement of drug-nanocomplex penetration with EMDA over PD Significant inhibition of tumour cell viability by nanocomplex over non-treated controls |
| Venuganti 2015 [69] | In vitro and In vivo | Porcine skin Skin tumor (Murine model) | Anti-sense Oligonucleotide (ASO) | PAMAM Dendrimer | 0.3 mA/cm2 over 4 h | Increased penetration of ASO-Dendrimer complex into epidermis with EMDA over PD Significant apoptosis of skin tumour with drug complex over non-treated controls |
| Lemos 2016 [70] | In vivo | SCC (Murine model) | ZnPcS4 * (anionic) | HEC Gel (Non-ionic) | 0.5 mA/cm2 over 30 min | 15-fold improvement of drug uptake by tumour with EMDA over PD 2.8-fold reduction in tumour volume in the group of mice treated with photosensitizer with EMDA followed by photodynamic therapy compared with non-treated controls |
| Labala 2016 [71] | In vitro | Porcine skin Melanoma (Murine cells) | anti-STAT3 SiRNA | Gold nanoparticles (Chitosan capped) | 0.47 mA/cm2 over 4 h | Complex was able to successfully penetrate skin with EMDA Nanoparticle complex inhibited cell growth by 50% |
| Labala 2017 [72] | In vitro and In vivo | Melanoma (Murine cells) Melanoma (Murine model) | Co-delivery of anti-STAT3 SiRNA and Imatinib mesylate | Gold nanoparticles | 0.5 mA/cm2 over 2 h | Combination of 2 drugs resulted in greater suppression of STAT3, reduction in tumour cell viability when compared with single drug delivery Transdermal EMDA delivery of nanocomplex resulted in significant reduction in tumour volume, weight, and STAT3 suppression (similar to direct intra-tumoral injection) over non-treated controls |
| Petrilli 2018 [73] | In vitro and In vivo | Porcine skin SCC (Murine model) | 5-FU | EGFR-targeted immunoliposome | 0.5 mA/cm2 over 6 h | 2-fold penetration of drug in skin with immunoliposomes with EMDA compared with control liposomes 2-fold reduction in tumour volume with transdermal 5-FU containing liposomes and EMDA compared with subcutaneous injection |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Min, J.W.S.; Saeed, N.; Coene, A.; Adriaens, M.; Ceelen, W. Electromotive Enhanced Drug Administration in Oncology: Principles, Evidence, Current and Emerging Applications. Cancers 2022, 14, 4980. https://doi.org/10.3390/cancers14204980
Min JWS, Saeed N, Coene A, Adriaens M, Ceelen W. Electromotive Enhanced Drug Administration in Oncology: Principles, Evidence, Current and Emerging Applications. Cancers. 2022; 14(20):4980. https://doi.org/10.3390/cancers14204980
Chicago/Turabian StyleMin, Jolene Wong Si, Nidda Saeed, Annelies Coene, Mieke Adriaens, and Wim Ceelen. 2022. "Electromotive Enhanced Drug Administration in Oncology: Principles, Evidence, Current and Emerging Applications" Cancers 14, no. 20: 4980. https://doi.org/10.3390/cancers14204980
APA StyleMin, J. W. S., Saeed, N., Coene, A., Adriaens, M., & Ceelen, W. (2022). Electromotive Enhanced Drug Administration in Oncology: Principles, Evidence, Current and Emerging Applications. Cancers, 14(20), 4980. https://doi.org/10.3390/cancers14204980







