Repurposing of Benzimidazole Anthelmintic Drugs as Cancer Therapeutics
Abstract
Simple Summary
Abstract
1. Introduction
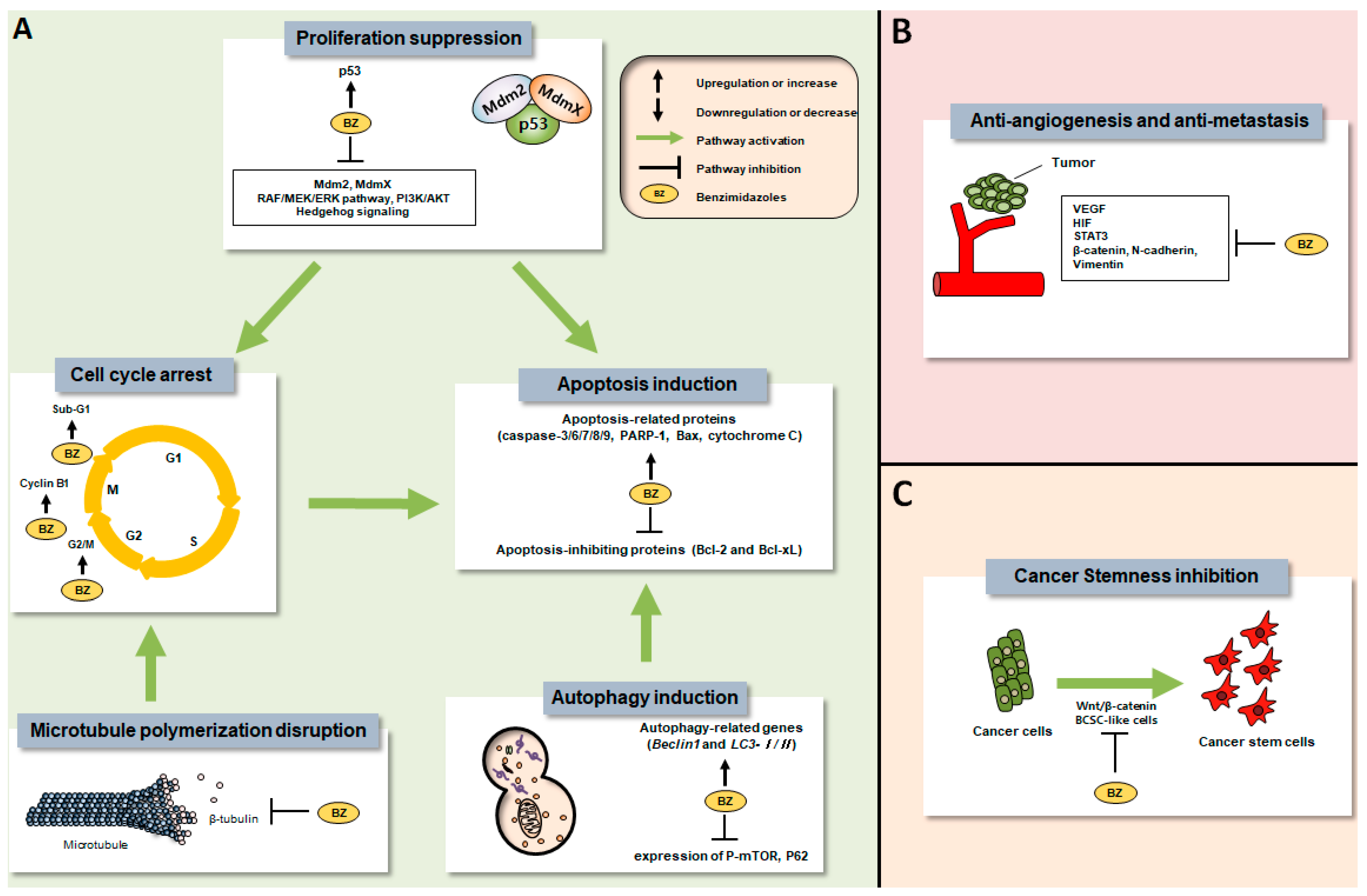
2. Preclinical Anti-Tumor Efficacies of Benzimidazole Anthelmintics
2.1. ABZ
2.1.1. In Vitro Anti-Tumor Effects
2.1.2. In Vivo Anti-Tumor Effects
2.2. FBZ
2.2.1. In Vitro Anti-Tumor Effects
2.2.2. In Vivo Anti-Tumor Effects
2.3. FLZ
2.3.1. In Vitro Anti-Tumor Effects
2.3.2. In Vivo Anti-Tumor Effects
2.4. MBZ
2.4.1. In Vitro Anti-Tumor Effects
2.4.2. In Vivo Anti-Tumor Effects
2.5. The Others
2.5.1. In Vitro Anti-Tumor Effects
2.5.2. In Vivo Anti-Tumor Effects
3. Clinical Properties of Benzimidazole Anthelmintics
Clinical Evidence
4. Pharmacokinetic Properties
5. Discussion
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Harris, P.; Finlay, I.G.; Cook, A.; Thomas, K.J.; Hood, K. Complementary and alternative medicine use by patients with cancer in Wales: A cross sectional survey. Complement. Ther. Med. 2003, 11, 249–253. [Google Scholar] [CrossRef]
- Kufel-Grabowska, J.; Bartoszkiewicz, M.; Litwiniuk, M. The use of complementary and alternative medicine among cancer patients. Polskie Arch. Med. Wewnetrznej 2021, 131, 83–85. [Google Scholar] [CrossRef] [PubMed]
- Eguchi, K.; Hyodo, I.; Saeki, H. Current status of cancer patients’ perception of alternative medicine in Japan. Support Care Cancer 2000, 8, 28–32. [Google Scholar] [CrossRef] [PubMed]
- Jung, K.-W.; Won, Y.-J.; Kong, H.-J.; Lee, E.S. Cancer statistics in Korea: Incidence, mortality, survival, and prevalence in 2016. Cancer Res. Treat. 2019, 51, 417. [Google Scholar] [CrossRef] [PubMed]
- American Cancer Society. Cancer Facts & Figures 2022; American Cancer Society: Atlanta, GA, USA, 2022. [Google Scholar]
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef] [PubMed]
- Finn, R.S.; Qin, S.; Ikeda, M.; Galle, P.R.; Ducreux, M.; Kim, T.-Y.; Kudo, M.; Breder, V.; Merle, P.; Kaseb, A.O. Atezolizumab plus bevacizumab in unresectable hepatocellular carcinoma. N. Engl. J. Med. 2020, 382, 1894–1905. [Google Scholar] [CrossRef]
- Debela, D.T.; Muzazu, S.G.; Heraro, K.D.; Ndalama, M.T.; Mesele, B.W.; Haile, D.C.; Kitui, S.K.; Manyazewal, T. New approaches and procedures for cancer treatment: Current perspectives. SAGE Open Med. 2021, 9, 20503121211034366. [Google Scholar] [CrossRef]
- Abou-Gharbia, M.; Childers, W.E. Discovery of innovative therapeutics: Today’s realities and tomorrow’s vision. 2. Pharma’s challenges and their commitment to innovation. J. Med. Chem. 2014, 57, 5525–5553. [Google Scholar] [CrossRef] [PubMed]
- Nath, J.; Paul, R.; Ghosh, S.K.; Paul, J.; Singha, B.; Debnath, N. Drug repurposing and relabeling for cancer therapy: Emerging benzimidazole antihelminthics with potent anticancer effects. Life Sci. 2020, 258, 118189. [Google Scholar] [CrossRef] [PubMed]
- Wouters, O.J.; McKee, M.; Luyten, J. Estimated research and development investment needed to bring a new medicine to market, 2009–2018. JAMA 2020, 323, 844–853. [Google Scholar] [CrossRef] [PubMed]
- Iragorri, N.; de Oliveira, C.; Fitzgerald, N.; Essue, B. The Out-of-Pocket Cost Burden of Cancer Care—A Systematic Literature Review. Curr. Oncol. 2021, 28, 1216–1248. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Zhou, L.; Xie, N.; Nice, E.C.; Zhang, T.; Cui, Y.; Huang, C. Overcoming cancer therapeutic bottleneck by drug repurposing. Signal Transduct. Target. Ther. 2020, 5, 113. [Google Scholar] [CrossRef] [PubMed]
- Son, D.-S.; Lee, E.-S.; Adunyah, S.E. The antitumor potentials of benzimidazole anthelmintics as repurposing drugs. Immune Netw. 2020, 20, e29. [Google Scholar] [CrossRef] [PubMed]
- Kim, E.-Y. Dog Dewormer Goes Out of Stock Amid Rumor of Efficacy for Cancer. Korea Biomedical Review, 27 December 2019. Available online: https://www.koreabiomed.com/news/articleView.html?idxno=7073 (accessed on 1 December 2021).
- Laudisi, F.; Marônek, M.; Di Grazia, A.; Monteleone, G.; Stolfi, C. Repositioning of anthelmintic drugs for the treatment of cancers of the digestive system. Int. J. Mol. Sci. 2020, 21, 4957. [Google Scholar] [CrossRef]
- U.S. Food & Drug Administration. Drugs@FDA: FDA-Approved Drugs. Available online: https://www.accessdata.fda.gov/scripts/cder/daf/index.cfm (accessed on 10 January 2022).
- Markowitz, D.; Ha, G.; Ruggieri, R.; Symons, M. Microtubule-targeting agents can sensitize cancer cells to ionizing radiation by an interphase-based mechanism. OncoTargets Ther. 2017, 10, 5633. [Google Scholar] [CrossRef]
- Liu, H.; Sun, H.; Zhang, B.; Liu, S.; Deng, S.; Weng, Z.; Zuo, B.; Yang, J.; He, Y. 18 F-FDG PET imaging for monitoring the early anti-tumor effect of albendazole on triple-negative breast cancer. Breast Cancer 2020, 27, 372–380. [Google Scholar] [CrossRef]
- Bai, R.-Y.; Staedtke, V.; Aprhys, C.M.; Gallia, G.L.; Riggins, G.J. Antiparasitic mebendazole shows survival benefit in 2 preclinical models of glioblastoma multiforme. Neuro Oncol. 2011, 13, 974–982. [Google Scholar] [CrossRef]
- Zhang, X.; Zhao, J.; Gao, X.; Pei, D.; Gao, C. Anthelmintic drug albendazole arrests human gastric cancer cells at the mitotic phase and induces apoptosis. Exp. Ther. Med. 2017, 13, 595–603. [Google Scholar] [CrossRef]
- Mrkvová, Z.; Uldrijan, S.; Pombinho, A.; Bartůněk, P.; Slaninová, I. Benzimidazoles downregulate Mdm2 and MdmX and activate p53 in MdmX overexpressing tumor cells. Molecules 2019, 24, 2152. [Google Scholar] [CrossRef]
- Čáňová, K.; Rozkydalová, L.; Vokurková, D.; Rudolf, E. Flubendazole induces mitotic catastrophe and apoptosis in melanoma cells. Toxicol. Vitr. 2018, 46, 313–322. [Google Scholar] [CrossRef]
- Sasaki, J.-I.; Ramesh, R.; Chada, S.; Gomyo, Y.; Roth, J.A.; Mukhopadhyay, T. The Anthelmintic Drug Mebendazole Induces Mitotic Arrest and Apoptosis by Depolymerizing Tubulin in Non-Small Cell Lung Cancer Cells 1 Supported in part by grants from the National Cancer Institute and the NIH Specialized Program of Research Excellence in Lung Cancer P-50-CA70907 and P01 CA78778-01A1 (both to JAR), by gifts to the Division of Surgery and Anesthesiology from Tenneco and Exxon for the Core Laboratory Facility, by The University of Texas MD Anderson Cancer Center Support Core Grant CA16672, by the WM Keck Foundation, and by a sponsored research agreement with Introgen Therapeutics, Inc. JAR is a scientific advisor for Introgen Therapeutics, Inc. 1. Mol. Cancer Ther. 2002, 1, 1201–1209. [Google Scholar] [PubMed]
- Yenjerla, M.; Cox, C.; Wilson, L.; Jordan, M.A. Carbendazim inhibits cancer cell proliferation by suppressing microtubule dynamics. J. Pharmacol. Exp. Ther. 2009, 328, 390–398. [Google Scholar] [CrossRef] [PubMed]
- Florio, R.; Veschi, S.; di Giacomo, V.; Pagotto, S.; Carradori, S.; Verginelli, F.; Cirilli, R.; Casulli, A.; Grassadonia, A.; Tinari, N. The benzimidazole-based anthelmintic parbendazole: A repurposed drug candidate that synergizes with gemcitabine in pancreatic cancer. Cancers 2019, 11, 2042. [Google Scholar] [CrossRef]
- Dogra, N.; Mukhopadhyay, T. Impairment of the ubiquitin-proteasome pathway by methyl N-(6-phenylsulfanyl-1H-benzimidazol-2-yl) carbamate leads to a potent cytotoxic effect in tumor cells: A novel antiproliferative agent with a potential therapeutic implication. J. Biol. Chem. 2012, 287, 30625–30640. [Google Scholar] [CrossRef] [PubMed]
- Oh, E.; Kim, Y.J.; An, H.; Sung, D.; Cho, T.M.; Farrand, L.; Jang, S.; Seo, J.H.; Kim, J.Y. Flubendazole elicits anti-metastatic effects in triple-negative breast cancer via STAT3 inhibition. Int. J. Cancer 2018, 143, 1978–1993. [Google Scholar] [CrossRef]
- Pinto, L.C.; Mesquita, F.P.; Soares, B.M.; da Silva, E.L.; Puty, B.; de Oliveira, E.H.C.; Burbano, R.R.; Montenegro, R.C. Mebendazole induces apoptosis via C-MYC inactivation in malignant ascites cell line (AGP01). Toxicol. In Vitro 2019, 60, 305–312. [Google Scholar] [CrossRef]
- Wei, K.-L.; Chen, F.-Y.; Lin, C.-Y.; Gao, G.-L.; Kao, W.-Y.; Yeh, C.-H.; Chen, C.-R.; Huang, H.-C.; Tsai, W.-R.; Jong, K.-J. Activation of aryl hydrocarbon receptor reduces carbendazim-induced cell death. Toxicol. Appl. Pharmacol. 2016, 306, 86–97. [Google Scholar] [CrossRef]
- Chen, Q.; Li, Y.; Zhou, X.; Li, R. Oxibendazole inhibits prostate cancer cell growth. Oncol. Lett. 2018, 15, 2218–2226. [Google Scholar] [CrossRef]
- Zhou, F.; Du, J.; Wang, J. Albendazole inhibits HIF-1α-dependent glycolysis and VEGF expression in non-small cell lung cancer cells. Mol. Cell. Biochem. 2017, 428, 171–178. [Google Scholar] [CrossRef]
- Sung, S.J.; Kim, H.-K.; Hong, Y.-K.; Joe, Y.A. Autophagy is a potential target for enhancing the anti-angiogenic effect of mebendazole in endothelial cells. Biomol. Ther. 2019, 27, 117. [Google Scholar] [CrossRef]
- Kralova, V.; Hanušová, V.; Caltová, K.; Špaček, P.; Hochmalová, M.; Skálová, L.; Rudolf, E. Flubendazole and mebendazole impair migration and epithelial to mesenchymal transition in oral cell lines. Chem. Biol. Interact. 2018, 293, 124–132. [Google Scholar] [CrossRef] [PubMed]
- Priotti, J.; Baglioni, M.V.; García, A.; Rico, M.J.; Leonardi, D.; Lamas, M.C.; Márquez, M.M. Repositioning of anti-parasitic drugs in cyclodextrin inclusion complexes for treatment of triple-negative breast cancer. AAPS PharmSciTech 2018, 19, 3734–3741. [Google Scholar] [CrossRef] [PubMed]
- Pourgholami, M.H.; Akhter, J.; Wang, L.; Lu, Y.; Morris, D.L. Antitumor activity of albendazole against the human colorectal cancer cell line HT-29: In vitro and in a xenograft model of peritoneal carcinomatosis. Cancer Chemother. Pharmacol. 2005, 55, 425–432. [Google Scholar] [CrossRef]
- Wang, L.-J.; Lee, Y.-C.; Huang, C.-H.; Shi, Y.-J.; Chen, Y.-J.; Pei, S.-N.; Chou, Y.-W.; Chang, L.-S. Non-mitotic effect of albendazole triggers apoptosis of human leukemia cells via SIRT3/ROS/p38 MAPK/TTP axis-mediated TNF-α upregulation. Biochem. Pharmacol. 2019, 162, 154–168. [Google Scholar] [CrossRef] [PubMed]
- Pourgholami, M.; Woon, L.; Almajd, R.; Akhter, J.; Bowery, P.; Morris, D. In vitro and in vivo suppression of growth of hepatocellular carcinoma cells by albendazole. Cancer Lett. 2001, 165, 43–49. [Google Scholar] [CrossRef]
- Shimomura, I.; Yokoi, A.; Kohama, I.; Kumazaki, M.; Tada, Y.; Tatsumi, K.; Ochiya, T.; Yamamoto, Y. Drug library screen reveals benzimidazole derivatives as selective cytotoxic agents for KRAS-mutant lung cancer. Cancer Lett. 2019, 451, 11–22. [Google Scholar] [CrossRef] [PubMed]
- Chu, S.W.; Badar, S.; Morris, D.L.; Pourgholami, M.H. Potent inhibition of tubulin polymerisation and proliferation of paclitaxel-resistant 1A9PTX22 human ovarian cancer cells by albendazole. Anticancer Res. 2009, 29, 3791–3796. [Google Scholar]
- Pourgholami, M.H.; Cai, Z.Y.; Lu, Y.; Wang, L.; Morris, D.L. Albendazole: A potent inhibitor of vascular endothelial growth factor and malignant ascites formation in OVCAR-3 tumor-bearing nude mice. Clin. Cancer Res. 2006, 12, 1928–1935. [Google Scholar] [CrossRef]
- Patel, K.; Doudican, N.A.; Schiff, P.B.; Orlow, S.J. Albendazole sensitizes cancer cells to ionizing radiation. Radiat. Oncol. 2011, 6, 160. [Google Scholar] [CrossRef]
- Patel, K.; Doudican, N.; Schiff, P.; Orlow, S. Albendazole Sensitizes Melanoma and Small Cell Lung Cancer Cells to Ionizing Radiation. Int. J. Radiat. Oncol. Biol. Phys. 2011, 81, S751–S752. [Google Scholar] [CrossRef]
- Tang, Y.; Liang, J.; Wu, A.; Chen, Y.; Zhao, P.; Lin, T.; Zhang, M.; Xu, Q.; Wang, J.; Huang, Y. Co-delivery of trichosanthin and albendazole by nano-self-assembly for overcoming tumor multidrug-resistance and metastasis. ACS Appl. Mater. Interfaces 2017, 9, 26648–26664. [Google Scholar] [CrossRef] [PubMed]
- Noorani, L.; Stenzel, M.; Liang, R.; Pourgholami, M.H.; Morris, D.L. Albumin nanoparticles increase the anticancer efficacy of albendazole in ovarian cancer xenograft model. J. Nanobiotechnol. 2015, 13, 25. [Google Scholar] [CrossRef] [PubMed]
- Choi, E.-K.; Kim, S.-W.; Nam, E.-J.; Paek, J.; Yim, G.-W.; Kang, M.-H.; Kim, Y.-T. Differential effect of intraperitoneal albendazole and paclitaxel on ascites formation and expression of vascular endothelial growth factor in ovarian cancer cell-bearing athymic nude mice. Reprod. Sci. 2011, 18, 763–771. [Google Scholar] [CrossRef] [PubMed]
- Hettiarachchi, G.; Samanta, S.K.; Falcinelli, S.; Zhang, B.; Moncelet, D.; Isaacs, L.; Briken, V. Acyclic cucurbit [n] uril-type molecular container enables systemic delivery of effective doses of albendazole for treatment of SK-OV-3 xenograft tumors. Mol. Pharm. 2016, 13, 809–818. [Google Scholar] [CrossRef] [PubMed]
- Ehteda, A.; Galettis, P.; Pillai, K.; Morris, D.L. Combination of albendazole and 2-methoxyestradiol significantly improves the survival of HCT-116 tumor-bearing nude mice. BMC Cancer 2013, 13, 86. [Google Scholar] [CrossRef] [PubMed]
- Ehteda, A.; Galettis, P.; Chu, S.W.L.; Pillai, K.; Morris, D.L. Complexation of albendazole with hydroxypropyl-β-cyclodextrin significantly improves its pharmacokinetic profile, cell cytotoxicity and antitumor efficacy in nude mice. Anticancer Res. 2012, 32, 3659–3666. [Google Scholar] [PubMed]
- Lai, S.R.; Castello, S.; Robinson, A.; Koehler, J. In vitro anti-tubulin effects of mebendazole and fenbendazole on canine glioma cells. Vet. Comp. Oncol. 2017, 15, 1445–1454. [Google Scholar] [CrossRef]
- Nygren, P.; Fryknäs, M.; Ågerup, B.; Larsson, R. Repositioning of the anthelmintic drug mebendazole for the treatment for colon cancer. J. Cancer Res. Clin. Oncol. 2013, 139, 2133–2140. [Google Scholar] [CrossRef]
- Dogra, N.; Kumar, A.; Mukhopadhyay, T. Fenbendazole acts as a moderate microtubule destabilizing agent and causes cancer cell death by modulating multiple cellular pathways. Sci. Rep. 2018, 8, 11926. [Google Scholar] [CrossRef]
- Duan, Q.; Liu, Y.; Rockwell, S. Fenbendazole as a potential anticancer drug. Anticancer Res. 2013, 33, 355–362. [Google Scholar]
- Michaelis, M.; Agha, B.; Rothweiler, F.; Löschmann, N.; Voges, Y.; Mittelbronn, M.; Starzetz, T.; Harter, P.N.; Abhari, B.A.; Fulda, S. Identification of flubendazole as potential anti-neuroblastoma compound in a large cell line screen. Sci. Rep. 2015, 5, 8202. [Google Scholar] [CrossRef]
- Hou, Z.-J.; Luo, X.; Zhang, W.; Peng, F.; Cui, B.; Wu, S.-J.; Zheng, F.-M.; Xu, J.; Xu, L.-Z.; Long, Z.-J. Flubendazole, FDA-approved anthelmintic, targets breast cancer stem-like cells. Oncotarget 2015, 6, 6326. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Guo, M.; Li, J.; Zheng, Y.; Zhang, S.; Xie, T.; Liu, B. Systems biology-based discovery of a potential Atg4B agonist (Flubendazole) that induces autophagy in breast cancer. Mol. Biosyst. 2015, 11, 2860–2866. [Google Scholar] [CrossRef] [PubMed]
- Zhen, Y.; Zhao, R.; Wang, M.; Jiang, X.; Gao, F.; Fu, L.; Zhang, L.; Zhou, X.-L. Flubendazole elicits anti-cancer effects via targeting EVA1A-modulated autophagy and apoptosis in Triple-negative Breast Cancer. Theranostics 2020, 10, 8080. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.-J.; Sung, D.; Oh, E.; Cho, Y.; Cho, T.-M.; Farrand, L.; Seo, J.H.; Kim, J.Y. Flubendazole overcomes trastuzumab resistance by targeting cancer stem-like properties and HER2 signaling in HER2-positive breast cancer. Cancer Lett. 2018, 412, 118–130. [Google Scholar] [CrossRef]
- Spagnuolo, P.A.; Hu, J.; Hurren, R.; Wang, X.; Gronda, M.; Sukhai, M.A.; Di Meo, A.; Boss, J.; Ashali, I.; Beheshti Zavareh, R. The antihelmintic flubendazole inhibits microtubule function through a mechanism distinct from Vinca alkaloids and displays preclinical activity in leukemia and myeloma. Blood 2010, 115, 4824–4833. [Google Scholar] [CrossRef]
- Lin, S.; Yang, L.; Yao, Y.; Xu, L.; Xiang, Y.; Zhao, H.; Wang, L.; Zuo, Z.; Huang, X.; Zhao, C. Flubendazole demonstrates valid antitumor effects by inhibiting STAT3 and activating autophagy. J. Exp. Clin. Cancer Res. 2019, 38, 293. [Google Scholar] [CrossRef]
- Li, Y.; Acharya, G.; Elahy, M.; Xin, H.; Khachigian, L.M. The anthelmintic flubendazole blocks human melanoma growth and metastasis and suppresses programmed cell death protein-1 and myeloid-derived suppressor cell accumulation. Cancer Lett. 2019, 459, 268–276. [Google Scholar] [CrossRef]
- Kipper, F.C.; Silva, A.O.; Marc, A.L.; Confortin, G.; Junqueira, A.V.; Neto, E.P.; Lenz, G. Vinblastine and antihelmintic mebendazole potentiate temozolomide in resistant gliomas. Investig. New Drugs 2018, 36, 323–331. [Google Scholar] [CrossRef]
- De Witt, M.; Gamble, A.; Hanson, D.; Markowitz, D.; Powell, C.; Al Dimassi, S.; Atlas, M.; Boockvar, J.; Ruggieri, R.; Symons, M. Repurposing mebendazole as a replacement for vincristine for the treatment of brain tumors. Mol. Med. 2017, 23, 50–56. [Google Scholar] [CrossRef]
- Skibinski, C.G.; Williamson, T.; Riggins, G.J. Mebendazole and radiation in combination increase survival through anticancer mechanisms in an intracranial rodent model of malignant meningioma. J. Neurooncol. 2018, 140, 529–538. [Google Scholar] [CrossRef] [PubMed]
- Bai, R.-Y.; Staedtke, V.; Wanjiku, T.; Rudek, M.A.; Joshi, A.; Gallia, G.L.; Riggins, G.J. Brain penetration and efficacy of different mebendazole polymorphs in a mouse brain tumor model. Clin. Cancer Res. 2015, 21, 3462–3470. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Dratver, M.B.; Yazal, T.; Dong, K.; Nguyen, A.; Yu, G.; Dao, A.; Dratver, M.B.; Duhachek-Muggy, S.; Bhat, K. Mebendazole potentiates radiation therapy in triple-negative breast cancer. Int. J. Radiat. Oncol. Biol. Phys. 2019, 103, 195–207. [Google Scholar] [CrossRef] [PubMed]
- He, L.; Shi, L.; Du, Z.; Huang, H.; Gong, R.; Ma, L.; Chen, L.; Gao, S.; Lyu, J.; Gu, H. Mebendazole exhibits potent anti-leukemia activity on acute myeloid leukemia. Exp. Cell Res. 2018, 369, 61–68. [Google Scholar] [CrossRef] [PubMed]
- Zhang, F.; Li, Y.; Zhang, H.; Huang, E.; Gao, L.; Luo, W.; Wei, Q.; Fan, J.; Song, D.; Liao, J. Anthelmintic mebendazole enhances cisplatin’s effect on suppressing cell proliferation and promotes differentiation of head and neck squamous cell carcinoma (HNSCC). Oncotarget 2017, 8, 12968. [Google Scholar] [CrossRef] [PubMed]
- Pinto, L.C.; Moreira-Nunes, C.d.F.A.; Soares, B.M.; Burbano, R.M.R.; de Lemos, J.A.R.; Montenegro, R.C. Mebendazole, an antiparasitic drug, inhibits drug transporters expression in preclinical model of gastric peritoneal carcinomatosis. Toxicol. Vitr. 2017, 43, 87–91. [Google Scholar] [CrossRef]
- Wang, X.; Lou, K.; Song, X.; Ma, H.; Zhou, X.; Xu, H.; Wang, W. Mebendazole is a potent inhibitor to chemoresistant T cell acute lymphoblastic leukemia cells. Toxicol. Appl. Pharmacol. 2020, 396, 115001. [Google Scholar] [CrossRef]
- Tan, Z.; Chen, L.; Zhang, S. Comprehensive modeling and discovery of mebendazole as a novel TRAF2-and NCK-interacting kinase inhibitor. Sci. Rep. 2016, 6, 33534. [Google Scholar] [CrossRef]
- Li, Y.; Thomas, D.; Deutzmann, A.; Majeti, R.; Felsher, D.W.; Dill, D.L. Mebendazole for differentiation therapy of acute myeloid leukemia identified by a lineage maturation index. Sci. Rep. 2019, 9, 16775. [Google Scholar] [CrossRef]
- Simbulan-Rosenthal, C.M.; Dakshanamurthy, S.; Gaur, A.; Chen, Y.-S.; Fang, H.-B.; Abdussamad, M.; Zhou, H.; Zapas, J.; Calvert, V.; Petricoin, E.F. The repurposed anthelmintic mebendazole in combination with trametinib suppresses refractory NRASQ61K melanoma. Oncotarget 2017, 8, 12576. [Google Scholar] [CrossRef]
- Larsen, A.R.; Bai, R.-Y.; Chung, J.H.; Borodovsky, A.; Rudin, C.M.; Riggins, G.J.; Bunz, F. Repurposing the antihelmintic mebendazole as a hedgehog inhibitor. Mol. Cancer Ther. 2015, 14, 3–13. [Google Scholar] [CrossRef] [PubMed]
- Bai, R.-Y.; Staedtke, V.; Rudin, C.M.; Bunz, F.; Riggins, G.J. Effective treatment of diverse medulloblastoma models with mebendazole and its impact on tumor angiogenesis. Neuro Oncol. 2015, 17, 545–554. [Google Scholar] [CrossRef]
- Younis, N.S.; Ghanim, A.M.; Saber, S. Mebendazole augments sensitivity to sorafenib by targeting MAPK and BCL-2 signalling in n-nitrosodiethylamine-induced murine hepatocellular carcinoma. Sci. Rep. 2019, 9, 19095. [Google Scholar] [CrossRef] [PubMed]
- Williamson, T.; Bai, R.-Y.; Staedtke, V.; Huso, D.; Riggins, G.J. Mebendazole and a non-steroidal anti-inflammatory combine to reduce tumor initiation in a colon cancer preclinical model. Oncotarget 2016, 7, 68571. [Google Scholar] [CrossRef] [PubMed]
- Doudican, N.A.; Byron, S.A.; Pollock, P.M.; Orlow, S.J. XIAP downregulation accompanies mebendazole growth inhibition in melanoma xenografts. Anticancer Drugs 2013, 24, 181–188. [Google Scholar] [CrossRef]
- Martarelli, D.; Pompei, P.; Baldi, C.; Mazzoni, G. Mebendazole inhibits growth of human adrenocortical carcinoma cell lines implanted in nude mice. Cancer Chemother. Pharmacol. 2008, 61, 809–817. [Google Scholar] [CrossRef] [PubMed]
- Rushworth, L.K.; Hewit, K.; Munnings-Tomes, S.; Somani, S.; James, D.; Shanks, E.; Dufès, C.; Straube, A.; Patel, R.; Leung, H.Y. Repurposing screen identifies mebendazole as a clinical candidate to synergise with docetaxel for prostate cancer treatment. Br. J. Cancer 2020, 122, 517–527. [Google Scholar] [CrossRef]
- Dayan, A. Albendazole, mebendazole and praziquantel. Review of non-clinical toxicity and pharmacokinetics. Acta Trop. 2003, 86, 141–159. [Google Scholar] [CrossRef]
- Atalay, P.B.; Kuku, G.; Tuna, B.G. Effects of carbendazim and astaxanthin co-treatment on the proliferation of MCF-7 breast cancer cells. In Vitro Cell. Dev. Biol. Anim. 2019, 55, 113–119. [Google Scholar] [CrossRef]
- Kawaratani, Y.; Matsuoka, T.; Hirata, Y.; Fukata, N.; Nagaoka, Y.; Uesato, S. Influence of the carbamate fungicide benomyl on the gene expression and activity of aromatase in the human breast carcinoma cell line MCF-7. Environ. Toxicol. Pharmacol. 2015, 39, 292–299. [Google Scholar] [CrossRef]
- Wales, C.T.; Taylor, F.R.; Higa, A.T.; McAllister, H.A.; Jacobs, A.T. ERK-dependent phosphorylation of HSF1 mediates chemotherapeutic resistance to benzimidazole carbamates in colorectal cancer cells. Anticancer Drugs 2015, 26, 657–666. [Google Scholar] [CrossRef] [PubMed]
- Xu, D.; Tian, W.; Jiang, C.; Huang, Z.; Zheng, S. The anthelmintic agent oxfendazole inhibits cell growth in non-small cell lung cancer by suppressing c-Src activation. Mol. Med. Rep. 2019, 19, 2921–2926. [Google Scholar] [CrossRef] [PubMed]
- Doudican, N.; Rodriguez, A.; Osman, I.; Orlow, S.J. Mebendazole induces apoptosis via Bcl-2 inactivation in chemoresistant melanoma cells. Mol. Cancer Res. 2008, 6, 1308–1315. [Google Scholar] [CrossRef] [PubMed]
- Belaz, K.R.A.; Denadai, M.; Almeida, A.P.; Lima, R.T.; Vasconcelos, M.H.; Pinto, M.M.; Cass, Q.B.; Oliveira, R.V. Enantiomeric resolution of albendazole sulfoxide by semipreparative HPLC and in vitro study of growth inhibitory effects on human cancer cell lines. J. Pharm. Biomed. Anal. 2012, 66, 100–108. [Google Scholar] [CrossRef] [PubMed]
- Králová, V.; Hanušová, V.; Stanková, P.; Knoppová, K.; Cánová, K.; Skálová, L. Antiproliferative effect of benzimidazole anthelmintics albendazole, ricobendazole, and flubendazole in intestinal cancer cell lines. Anticancer Drugs 2013, 24, 911–919. [Google Scholar] [CrossRef]
- Pourgholami, M.H.; Szwajcer, M.; Chin, M.; Liauw, W.; Seef, J.; Galettis, P.; Morris, D.L.; Links, M. Phase I clinical trial to determine maximum tolerated dose of oral albendazole in patients with advanced cancer. Cancer Chemother. Pharmacol. 2010, 65, 597–605. [Google Scholar] [CrossRef]
- Morris, D.L.; Jourdan, J.-L.; Pourgholami, M.H. Pilot study of albendazole in patients with advanced malignancy. Oncology 2001, 61, 42–46. [Google Scholar] [CrossRef]
- Dobrosotskaya, I.Y.; Hammer, G.D.; Schteingart, D.E.; Maturen, K.E.; Worden, F.P. Mebendazole monotherapy and long-term disease control in metastatic adrenocortical carcinoma. Endocr. Pract. 2011, 17, e59–e62. [Google Scholar] [CrossRef]
- Nygren, P.; Larsson, R. Drug repositioning from bench to bedside: Tumour remission by the antihelmintic drug mebendazole in refractory metastatic colon cancer. Acta Oncol. 2014, 53, 427–428. [Google Scholar] [CrossRef]
- An, G.; Murry, D.J.; Gajurel, K.; Bach, T.; Deye, G.; Stebounova, L.V.; Codd, E.E.; Horton, J.; Gonzalez, A.E.; Garcia, H.H. Pharmacokinetics, safety, and tolerability of oxfendazole in healthy volunteers: A randomized, placebo-controlled first-in-human single-dose escalation study. Antimicrob. Agents Chemother. 2019, 63, e02255-18. [Google Scholar] [CrossRef]
- Jung-Cook, H. Pharmacokinetic variability of anthelmintics: Implications for the treatment of neurocysticercosis. Expert Rev. Clin. Pharmacol. 2012, 5, 21–30. [Google Scholar] [CrossRef] [PubMed]
- Čáňová, K.; Rozkydalová, L.; Rudolf, E. Anthelmintic flubendazole and its potential use in anticancer therapy. Acta Med. 2017, 60, 5–11. [Google Scholar] [CrossRef] [PubMed]
- Hong, S.-T. Albendazole and praziquantel: Review and safety monitoring in Korea. Infect. Chemother. 2018, 50, 1035499. [Google Scholar] [CrossRef]
- Pantziarka, P.; Bouche, G.; Meheus, L.; Sukhatme, V.; Sukhatme, V.P. Repurposing Drugs in Oncology (ReDO)—Mebendazole as an anti-cancer agent. Ecancermedicalscience 2014, 8, 443. [Google Scholar] [CrossRef] [PubMed]
- Bach, T.; Galbiati, S.; Kennedy, J.K.; Deye, G.; Nomicos, E.Y.; Codd, E.E.; Garcia, H.H.; Horton, J.; Gilman, R.H.; Gonzalez, A.E. Pharmacokinetics, safety, and tolerability of oxfendazole in healthy adults in an open-label phase 1 multiple ascending dose and food effect study. Antimicrob. Agents Chemother. 2020, 64, e01018-20. [Google Scholar] [CrossRef] [PubMed]
- Capece, B.P.; Virkel, G.L.; Lanusse, C.E. Enantiomeric behaviour of albendazole and fenbendazole sulfoxides in domestic animals: Pharmacological implications. Vet. J. 2009, 181, 241–250. [Google Scholar] [CrossRef]
- Schulz, J.D.; Neodo, A.; Coulibaly, J.T.; Keiser, J. Pharmacokinetics of albendazole, albendazole sulfoxide, and albendazole sulfone determined from plasma, blood, dried-blood spots, and Mitra samples of hookworm-infected adolescents. Antimicrob. Agents Chemother. 2019, 63, e02489-18. [Google Scholar] [CrossRef]
- Ceballos, L.; Krolewiecki, A.; Juárez, M.; Moreno, L.; Schaer, F.; Alvarez, L.I.; Cimino, R.; Walson, J.; Lanusse, C.E. Assessment of serum pharmacokinetics and urinary excretion of albendazole and its metabolites in human volunteers. PLoS Negl. Trop. Dis. 2018, 12, e0005945. [Google Scholar] [CrossRef]
- Rudolf, K.; Rudolf, E. An analysis of mitotic catastrophe induced cell responses in melanoma cells exposed to flubendazole. Toxicol. Vitr. 2020, 68, 104930. [Google Scholar] [CrossRef]
- Rubin, J.; Mansoori, S.; Blom, K.; Berglund, M.; Lenhammar, L.; Andersson, C.; Loskog, A.; Fryknäs, M.; Nygren, P.; Larsson, R. Mebendazole stimulates CD14+ myeloid cells to enhance T-cell activation and tumour cell killing. Oncotarget 2018, 9, 30805. [Google Scholar] [CrossRef]
- Zhang, Q.-L.; Lian, D.-D.; Zhu, M.J.; Li, X.M.; Lee, J.K.; Yoon, T.-J.; Lee, J.-H.; Jiang, R.-H.; Kim, C.D. Antitumor effect of albendazole on cutaneous squamous cell carcinoma (SCC) cells. BioMed Res. Int. 2019, 2019, 3689517. [Google Scholar] [CrossRef] [PubMed]
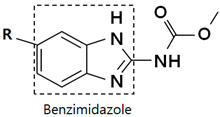 | |||
|---|---|---|---|
| R | Drug | R | Drug |
 | Albendazole |  | Nocodazole |
 | Fenbendazole |  | Oxfendazole |
 | Flubendazole |  | Oxibendazole |
 | Mebendazole |  | Ricobendazole |
 | Carbendazim |  | Parbendazole |
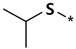 | Methiazole | ||
| Drug | Stage | Cancer Type | Number of Patients | Methods | Adverse Effects | Results | Identifier/Ref. |
|---|---|---|---|---|---|---|---|
| Albendazole | Phase 1 | Refractory solid tumors | 36 | Every day for 2 weeks, followed by 1 week of rest. Treatment was repeated in a 21-day cycle. 400–1200 mg b.i.d. p.o. | ABZ was well tolerated. Fatigue and mild gastrointestinal upset (Major). Myelosuppression. | 16% of patients showed a decrease in levels of tumor markers. Plasma VEGF level decreased in the first 8 h after ABZ administration. | [89] |
| Albendazole | Pilot Study | Colorectal cancer or hepatocellular carcinoma | 7 | 10 mg/kg/day, with 2 or 3 divided doses p.o. (28 d). The maximum tolerated dose was 1200 mg b.i.d. | ABZ was well tolerated. Severe neutropenia in three patients. | CEA decreased in two patients. CEA or α-feto protein stabilized in three patients. | [90] |
| Carbendazim | Phase 1 | Unspecified adult solid tumor | 25 | P.o weekly for 3 consecutive weeks, followed by 1 week of rest. Treatment repeated in a 28-day cycle. Determining dose. | No results posted. | No results posted. Actual study completion date: November 2000 | NCT00003709 |
| Mebendazole | Case report | Adrenal cancer | 1 | 100 mg b.i.d. p.o. for 19 months. | No significant adverse effects. | Metastases regressed. The patient’s disease remained stable for 19 months, but showed progression after 24 months. | [91] |
| Mebendazole | Case report | Refractory metastatic colon cancer | 1 | 100 mg b.i.d. p.o. for six weeks. | AST and ALT were increased up to > five times above the normal limit. | The metastases in the lungs and lymph nodes were near completely remissioned. A good portion of those in the liver were remissioned. | [92] |
| Mebendazole | Not applicable | Advanced hepatocellular carcinoma | 170 (recruiting) | 100 mg b.i.d. p.o. in combination with lenvatinib. | No results posted. | No results posted. Estimated study completion date: 19 June 2022 | NCT04443049 |
| Mebendazole | Phase 1 | High-grade glioma | 24 | T.i.d. p.o. in a 28-day cycle, in combination with temozolomide. Determining dose. | No results posted. | No results posted. Actual study completion date: 16 April 2021 | NCT01729260 |
| Mebendazole | Phase 1 | Recurrent pediatric brain cancers | 21 (recruiting) | T.i.d. p.o. Determining dose. | No results posted. | No results posted. Estimated study completion date: June 2022 | NCT02644291 |
| Mebendazole | Phase 1/2 | Pediatric gliomas | 36 (recruiting) | 50–200 mg/kg/day divided twice p.o., in combination with standard anti-tumor drugs | No results posted. | No results posted. Estimated study completion date: April 2023 | NCT01837862 |
| Mebendazole | Phase 2a | Advanced gastrointestinal cancer or cancer of unknown origin | 11 (Terminated due to lack of effect) | 50–4000 mg b.i.d. p.o. for 16 weeks. Determining dose. | No results posted. | No results posted. Actual study completion date: 16 January 2019 | NCT03628079 |
| Mebendazole | Phase 2 | Incurable and lethal cancers | 250 (recruiting) | Tolerable and safe doses for 10 to 12 months. Combination of two anti-protozoal drugs. | No results posted. | No results posted. Estimated study completion date: 31 December 2023 | NCT02366884 |
| Mebendazole | Phase 3 | Colorectal cancer | 40 (recruiting) | Folfox with avastin and MBZ. | No results posted. | No results posted. Estimated study completion date: December 2028 | NCT03925662 |
| Mebendazole | Phase 3 | Cancer | 207 (Not yet recruiting) | 100 mg q.d. in combination with atorvastatin, metformin, and doxycycline. | No results posted. | No results posted. Estimated study completion date: 22 September 2026 | NCT02201381 |
| Drug | Absorption | Distribution | Metabolism | Excretion | Ref. |
|---|---|---|---|---|---|
| Albendazole |
|
|
|
| [81,94,96,99,100,101] |
| Fenbendazole | - | - |
|
| [99] |
| Flubendazole |
| - |
|
| [95] |
| Mebendazole |
|
|
|
| [14,81,96,97] |
| Oxfendazole |
| - |
|
| [93,98] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Song, B.; Park, E.Y.; Kim, K.J.; Ki, S.H. Repurposing of Benzimidazole Anthelmintic Drugs as Cancer Therapeutics. Cancers 2022, 14, 4601. https://doi.org/10.3390/cancers14194601
Song B, Park EY, Kim KJ, Ki SH. Repurposing of Benzimidazole Anthelmintic Drugs as Cancer Therapeutics. Cancers. 2022; 14(19):4601. https://doi.org/10.3390/cancers14194601
Chicago/Turabian StyleSong, Bomi, Eun Young Park, Kwang Joon Kim, and Sung Hwan Ki. 2022. "Repurposing of Benzimidazole Anthelmintic Drugs as Cancer Therapeutics" Cancers 14, no. 19: 4601. https://doi.org/10.3390/cancers14194601
APA StyleSong, B., Park, E. Y., Kim, K. J., & Ki, S. H. (2022). Repurposing of Benzimidazole Anthelmintic Drugs as Cancer Therapeutics. Cancers, 14(19), 4601. https://doi.org/10.3390/cancers14194601










